Abstract
Background:
Aronia melanocarpa (Michx.) Elliot fruits are very rich in polyphenols – procyanidins, flavonoids, and phenolic acids.
Objective:
On rat hepatocytes, isolated by two-stepped collagenase perfusion, we investigated the effect of A. melanocarpa fruit juice (AMFJ) in two models of liver toxicity caused by (i) metabolic bioactivation of carbon tetrachloride (CCl4), and (ii) tert-butyl hydroperoxide (t-BuOOH)-induced oxidative stress.
Materials and Methods:
Isolated rat hepatocytes are a suitable model for hepatotoxicity studies. We determined the main parameters of the functional and metabolic status of rat hepatocytes: Cell viability (measured by trypan blue exclusion) and the levels of lactate dehydrogenase (LDH), reduced glutathione (GSH), and malondialdehyde (MDA). These parameters were used to investigate the protective effects of AMFJ in the two toxicity models. The effects of AMFJ were compared with those of silymarin. The cells were treated either with AMFJ or silymarin at increasing concentrations of 5 μg/ml, 10 μg/ml, 30 μg/ml, 50 μg/ml, and 100 μg/ml which were used for measuring of IC50.
Results:
In both toxicity models – CCl4 and t-BuOOH, AMFJ showed statistically significant cytoprotective and antioxidant activities. AMFJ prevented the loss of cell viability and GSH depletion, decreased LDH leakage and MDA production. The effects of AMFJ at the concentrations of 5, 10, 30, and 50 μg/ml were similar to those of the same concentrations of silymarin, while the effect of the highest AMFJ concentration of 100 μg/ml was higher than that of the same silymarin concentration. The effects were concentration-dependent and more prominent in the t-BuOOH model, compared to those in the CCl4 model.
Conclusion:
The cytoprotective and antioxidant effects of AMFJ established in this study might be due to its polyphenolic ingredients, which could influence the cytochrome P450-mediated metabolism of the experimental hepatotoxic substances (CCl4 and t-BuOOH) and could act as free radical scavengers. The stronger effects of the highest AMFJ concentration in comparison with that of silymarin were possibly due to the combined presence of different polyphenols in the juice.
SUMMARY
On rat hepatocytes, isolated by two-stepped collagenase perfusion, we investigated the effect of Aronia melanocarpa fruit juice (AMFJ) in two models of liver toxicity caused by i) metabolic bioactivation of carbon tetrachloride (CCl4), and ii) tert-butyl hydroperoxide (t-BuOOH)-induced oxidative stress. In both toxicity models – CCl4 and t-BuOOH, AMFJ showed statistically significant cytoprotective and antioxidant activities. AMFJ prevented the loss of cell viability and GSH depletion, decreased LDH leakage and MDA production. The effects of AMFJ at the concentrations of 5, 10, 30, and 50 μg/ml were similar to those of the same concentrations of silymarin, while the effect of the highest AMFJ concentration of 100 μg/ml was higher than that of the same silymarin concentration. The effects were concentration-dependent and were more prominent in the t-BuOOH model, compared to those in the CCl4 model.

Keywords: Aronia melanocarpa fruit juice, carbon tetrachloride, cytoprotection, isolated hepatocytes, tert-butyl hydroperoxide
INTRODUCTION
Convenient, well-controlled biological model system with high drug-metabolizing capacities, which can be used in experimental toxicology are isolated rat hepatocytes. This in vitro system is a part of recommended tests from the European Centre for the Validation of Alternative Methods (ECVAM). The main goal of ECVAM is to promote the acceptance of alternative methods, which are important for reducing, refining, and replacing the use of laboratory animals.[1]
The main enzymes, which play a role in the biotransformation of compounds from natural and synthetic origin, belong to the cytochrome P450 superfamily. These enzymes participate in the bioactivation and detoxification of numerous toxicants and pro-carcinogens. For example, carbon tetrachloride (CCl4) causes liver damage following its cleavage by cytochrome P450 to form the trichloromethyl free radical and the highly reactive trichloromethyl peroxy radical, which covalently bind to lipids and other cellular macromolecules.[2]
Oxidative damage is widely recognized as being involved in the development of many pathological conditions. The mechanism behind oxidative stress, considered to begin with lipid peroxidation in biomembranes, subsequently can cause structural and functional degeneration, and when it is severe, it may end in cell death. As a pro-oxidant, tert-butyl hydroperoxide (t-BuOOH) has been widely used, and many effects on cell metabolism have been described, e.g., changes in calcium homeostasis, increase of lipid peroxidation, and decrease of mitochondrial membrane potential.[3]
Experiments on isolated hepatocytes are thus a useful model system for evaluation of the toxic effect of various pro-oxidants, which act directly on mitochondrial enzymes.
As oxidative stress is known to play a role in the pathogenesis of different diseases, the contribution of dietary polyphenols for their prevention has been widely investigated.[4] Dietary phenolic substances have been extensively studied because of their antioxidant activity. Berries are known as plant material very rich in polyphenols.
Aronia melanocarpa (Michx.) Elliot (black chokeberry) fruits are extremely rich in phenolic compounds such as procyanidins, flavonoids (mainly from the subclass of anthocyanins), and phenolic acids (chlorogenic and neochlorogenic). The anthocyanins in A. melanocarpa fruits are water-soluble plant pigments, responsible for the purple and even black color of the fruits.[5]
The aim of the following study is to investigate the possible cytoprotective effects of A. melanocarpa fruit juice (AMFJ) in two models on isolated rat hepatocytes caused by (i) metabolic bioactivation of CCl4, and (ii) t-BuOOH-induced oxidative stress. The effects of AMFJ were compared with those of the same concentrations of silymarin.
MATERIALS AND METHODS
Chemicals
The chemicals used in the experiments were: Pentobarbital sodium (Sanofi, France), (N-[2-Hydroxyethyl]piperazine-N’-[ethanesulfonic acid]) (HEPES) (Sigma-Aldrich, Germany), NaCl (Merck, Germany), KCl (Merck), D-glucose (Merck), NaHCO3 (Merck), KH2PO4 (Scharlau Chemie SA, Spain), CaCl2.2H2 O (Merck), MgSO4.7H2 O (Fluka AG, Germany), collagenase from Clostridium histolyticum type IV (Sigma-Aldrich), albumin, bovine serum fraction V, minimum 98% (Sigma-Aldrich), EGTA (Sigma-Aldrich), 2-thiobarbituric acid (4,6-dihydroxypyrimidine-2-thiol; TBA) (Sigma-Aldrich), trichloroacetic acid (TCA) (Valerus, Bulgaria), 2,2’- dinitro-5,5’- dithiodibenzoic acid (DTNB) (Merck), lactate dehydrogenase (LDH) kit (Randox, UK), t-BuOOH (Sigma-Aldrich), and CCl4 (Merck).
Aronia melanocarpa fruit juice
AMFJ was produced from A. melanocarpa Elliot fruits grown in the Balkan Mountains, Bulgaria, in the region of Troyan. They were handpicked in September, crushed, and squeezed. The juice was filtered, preserved with potassium sorbate (1.0 g/L) and stored at 0°C until the experiment. The contents of phenolic substances in 100 ml AMFJ were:[6] Total phenolics 546.1 mg as gallic acid equivalents, determined spectrophotometrically according to the Folin-Ciocalteu procedure;[7] total procyanidins 312.3 mg determined gravimetrically;[8] anthocyanins such as cyanidin-galactoside 14.3 mg, cyanidin-arabinoside 6.2 mg, cyanidin-xyloside 1.2 mg, and cyanidin-glycoside 0.44 mg, determined by high performance liquid chromatography (HPLC), and phenolic acids such as chlorogenic acid 58.5 mg and neochlorogenic acid 83 mg, determined by HPLC.
Animals
Male Wistar rats (body weight 200–250 g) were used. The rats were housed in plexiglass cages (3 per cage) in a 12/12 light/dark cycle, under standard laboratory conditions (ambient temperature 20 ± 2°C and humidity 72 ± 4%) with free access to water and standard pelleted rat food 53-3, produced according to ISO 9001:2008.
Animals were purchased from the National Breeding Center, Sofia, Bulgaria. At least 7 days of acclimatization was allowed before the commencement of the study. The health of animals was monitored regularly by a veterinary physician. The vivarium (certificate of registration of farm No 0072/01.08.2007) was inspected by the Bulgarian Drug Agency to check the husbandry conditions (No A-11-1081/03.11.2011). All performed procedures were approved by the Institutional Animal Care Committee and the principles stated in the European Convention for the Protection of Vertebrate Animals used for Experimental and other Scientific Purposes (ETS 123) (Council of Europe, 1991), and were strictly followed throughout the experiment.[9]
Experimental design
Isolation and incubation of hepatocytes
Rats were anesthetized with sodium pentobarbital (0.2 ml/100 g). An optimized in situ liver perfusion using less reagents and shorter time of cell isolation was performed. The method provided in a higher amount of live and metabolically active hepatocytes.[10]
After portal catheterization, the liver was perfused with HEPES buffer (pH = 7.85) +0.6 mM EDTA (pH = 7.85), followed by clean HEPES buffer (pH = 7.85), and finally HEPES buffer containing collagenase type IV (50 mg/200 ml) and 7 mM CaCl2 (pH = 7.85). The liver was excised, minced into small pieces, and hepatocytes were dispersed in Krebs-Ringer-bicarbonate (KRB) buffer (pH = 7.35) +1% bovine serum albumin.
Cells were counted under the microscope, and the viability was assessed by trypan blue exclusion (0.05%).[11] Initial viability averaged 89%.
Cells were diluted with KRB to make a suspension of about 3 × 106 hepatocytes/ml. Incubations were carried out in flasks containing 3 ml of the cell suspension (i.e. 9 × 106 hepatocytes) and were performed in a 5% CO2 + 95% O2 atmosphere.[11]
Cell incubation with Aronia melanocarpa fruit juice or silymarin
Cells were incubated in five increasing concentrations either of AMFJ or silymarin: 5 μg/ml, 10 μg/ml, 30 μg/ml, 50 μg/ml, and 100 μg/ml.[12,13,14]
On the basis of these five increasing concentrations, the IC50 was calculated.
Biochemical assays
Lactate dehydrogenase release
After incubation, the hepatocytes were centrifuged for 4 min at 500 rpm, and the supernatant was used for measuring LDH release spectrophotometrically by LDH kit.[15]
Reduced glutathione depletion
At the end of the incubation, isolated rat hepatocytes were centrifuged at 4°C, and the pellet was used for evaluating the level of intracellular reduced glutathione (GSH). It was assessed by measuring nonprotein sulfhydryls after precipitation of proteins with TCA, followed by measurement of thiols in the supernatant with DTNB. The absorbance was measured at 412 nm.[11]
Malondialdehyde assay
After incubation, 1 ml from hepatocyte suspension was taken and added to 0.67 ml of 20% (w/v) TCA. After centrifugation, 1 ml of the supernatant was added to 0.33 ml of 0.67% (w/v) TBA and heated at 100°C for 30 min. The absorbance was measured at 535 nm, and the amount of TBA-reactants was calculated using a molar extinction coefficient of malondialdehyde (MDA) 1.56 × 105/M/cm.[11]
Statistical analysis
For statistical analysis of the data, we used statistical program “MEDCALC.” Results are expressed as mean ± standard error of the mean for six experiments. The significance of the data was assessed using the nonparametric Mann–Whitney test. A level of P < 0.05 was considered significant. Three parallel samples were used. IC50 was calculated using GraphPad Prism Statistical Software (GraphPad Statistical, Inc.).
RESULTS
Effects of Aronia melanocarpa fruit juice and silymarin in carbon tetrachloride-induced toxicity model
The parameters (cell viability, LDH activity, MDA level, and GSH depletion) determined in the model of CCl4-induced toxicity (a result of metabolic bio-activation of CCl4) are presented in Table 1.
Table 1.
Effect AMFJ at concentrations of 5 μg/ml (AMFJ5), 10 μg/ml (AMFJ10), 30 μg/ml (AMFJ30), 50 μg/ml (AMFJ50), and 100 μg/ml (AMFJ100), and silymarin at concentrations of 5 μg/ml (S5), 10 μg/ml (S10), 30 μg/ml (S30), 50 μg/ml (S50), and 100 μg/ml (S100) in a model of CCl4-induced hepatotoxicity on cell viability, LDH activity, MDA level, and GSH depletion in isolated rat hepatocytes
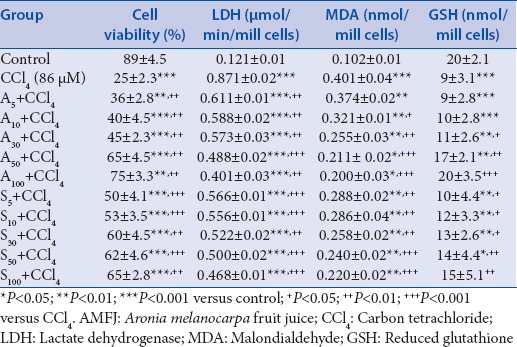
The incubation of hepatocytes in 86 μM of CCl4 resulted in a statistically significant reduction of cell viability by 72% (P < 0.001), increased LDH activity by 620% (P < 0.001), increased lipid peroxidation (MDA) by 293% (P < 0.001), and depletion of cell GSH by 55% (P < 0.001), compared to the control.
In combination with CCl4, AMFJ caused a statistically significant, concentration-dependent reduction of the damage caused by the hepatotoxic agent [Table 1].
AMFJ at the concentration of 5 μg/ml preserved cell viability by 44% (P < 0.01), at 10 μg/ml – by 60% (P < 0.01), at 30 μg/ml – by 80% (P < 0.01), at 50 μg/ml – by 160% (P < 0.01), and at 100 μg/ml – by 200% (P < 0.01) compared to CCl4 group. Silymarin preserved cell viability by 100% (P < 0.001) at the concentration of 5 μg/ml, by 112% (P < 0.001) at 10 μg/ml, by 140% (P < 0.01) at 30 μg/ml, by 148% (P < 0.001) at 50 μg/ml, and by 160% (P < 0.01) at 100 μg/ml compared to CCl4 group.
IC50 for cell viability was 7.5 μg/ml for AMFJ, and lower than 5 μg/ml for silymarin.
The activity of LDH was decreased by AMFJ at the concentration of 5 μg/ml by 30% (P < 0.01), at 10 μg/ml – by 32% (P < 0.01), at 30 μg/ml – by 34% (P < 0.01), at 50 μg/ml – by 44% (P < 0.001), and at 100 μg/ml – by 54% (P < 0.01), compared to CCl4 group. Silymarin decreased LDH activity at 5 μg/ml by 35% (P < 0.001), at 10 μg/ml – by 36% (P < 0.001), at 30 μg/ml – by 40% (P < 0.01), at 50 μg/ml – by 43% (P < 0.001), and at 100 μg/ml – by 46% (P < 0.001), compared to CCl4 group.
Thus, IC50 for LDH leakage was 100 μg/ml for both AMFJ and silymarin.
The production of MDA was not significantly affected by AMFJ at the concentration of 5 μg/ml. It was significantly decreased by AMFJ at concentrations of 10 μg/ml by 20% (P < 0.05), at 30 μg/ml – by 36% (P < 0.01), at 50 μg/ml – by 47% (P < 0.001), and at 100 μg/ml – by 50% (P < 0.001). Silymarin decreased MDA production at the concentration of 5 μg/ml by 28% (P < 0.01), at 10 μg/ml – by 29% (P < 0.01), at 30 μg/ml – by 36% (P < 0.01), at 50 μg/ml – by 40% (P < 0.001), and at 100 μg/ml – by 45% (P < 0.001), compared to CCl4 group.
IC50 for MDA production was100 μg/ml for AMFJ and up to 100 μg/ml for silymarin.
AMFJ at concentrations of 5 μg/ml and 10 μg/ml did not prevent CCl4-induced GSH depletion while the concentration of 30 μg/ml prevented it by 22% (P < 0.05), the concentration of 50 μg/ml – by 89% (P < 0.05), and the concentration of 100 μg/ml – by 122% (P < 0.05). Silymarin at the concentration of 5 μg/ml did not prevent CCl4-induced GSH depletion, while the concentrations of 10 μg/ml prevented it by 33% (P < 0.05), 30 μg/ml – by 44% (P < 0.05), 50 μg/ml – by 56% (P < 0.01), and 100 μg/ml – by 67% (P < 0.001), compared to CCl4 group.
IC50 for GSH depletion was 40 μg/ml for AMFJ and 50 μg/ml for silymarin.
Effects of Aronia melanocarpa fruit juice and silymarin in tert-butyl hydroperoxide-induced toxicity model
The parameters (cell viability, LDH activity, MDA level, and GSH depletion) determined in the model of t-BuOOH-induced toxicity (a model of oxidative stress) are presented in Table 2.
Table 2.
Effect AMFJ at concentrations of 5 μg/ml (AMFJ5), 10 μg/ml (AMFJ10), 30 μg/ml (AMFJ30), 50 μg/ml (AMFJ50), and 100 μg/ml (AMFJ100), and silymarin at concentrations of 5 μg/ml (S5), 10 μg/ml (S10), 30 μg/ml (S30), 50 μg/ml (S50), and 100 μg/ml (S100) in a model of t-BuOOH-induced oxidative stress on cell viability, LDH activity, MDA level, and GSH depletion in isolated rat hepatocytes
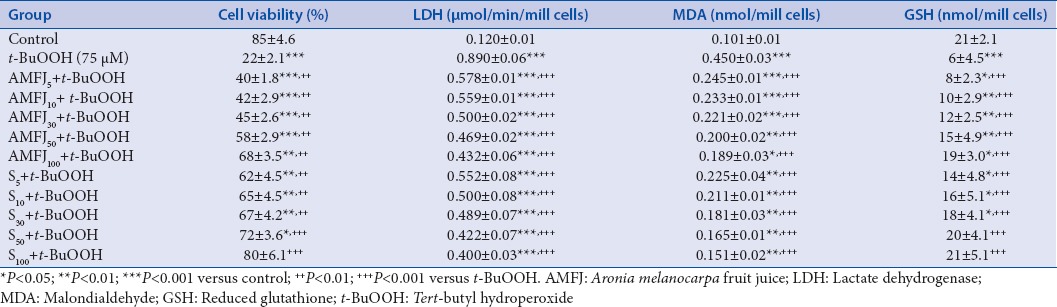
Hepatocytes incubation with 75 μM t-BuOOH resulted in a statistically significant reduction of cell viability by 74% (P < 0.001), increased LDH activity by 642% (P < 0.001), increased lipid peroxidation (MDA) by 346% (P < 0.001), and depletion of cell GSH by 71% (P < 0.001), compared to the control.
In combination with t-BuOOH, AMFJ caused a statistically significant, concentration-dependent reduction of the damage induced by the hepatotoxic agent [Table 2].
AMFJ preserved cell viability at the concentration of 5 μg/ml by 82% (P < 0.01), at 10 μg/ml – by 91% (P < 0.01), at 30 μg/ml – by 105% (P < 0.01), at 50 μg/ml – by 164% (P < 0.01), and at 100 μg/ml – by 209% (P < 0.01), compared to the t-BuOOH group. Silymarin preserved cell viability by 182% (P < 0.01) at the concentration of 5 μg/ml, by 195% (P < 0.01) at 10 μg/ml, by 205% (P < 0.01) at 30 μg/ml, by 227% (P < 0.001) at 50 μg/ml, and by 264% (P < 0.001) at 100 μg/ml, compared to the t-BuOOH group.
IC50 for cell viability both for AMFJ and silymarin was lower than 5 μg/ml.
The activity of LDH was decreased by AMFJ by 35% (P < 0.001) at 5 μg/ml, by 37% (P < 0.001) at 10 μg/ml, by 44% (P < 0.001) at 30 μg/ml, by 47% (P < 0.001) at 50 μg/ml, and by 51% (P < 0.001) at 100 μg/ml, as well as by silymarin by 38% (P < 0.001) at 5 μg/ml, by 44% (P < 0.001) at 10 μg/ml, by 45% (P < 0.001) at 30 μg/ml, by 53% (P < 0.001) at 50 μg/ml, and by 55% (P < 0.001) at 100 μg/ml, compared to t-BuOOH.
IC50 for LDH leakage was 100 μg/ml for AMFJ and 50 μg/ml for silymarin.
MDA level was decreased by AMFJ as follows: By 46% (P < 0.001) at the concentration of 5 μg/ml, by 48% (P < 0.001) at 10 μg/ml, by 51% (P < 0.001) at 30 μg/ml, by 56% (P < 0.001) at 50 μg/ml, and by 58% (P < 0.001) at 100 μg/ml, compared to t-BuOOH group. MDA level was also decreased by silymarin at the concentration of 5 μg/ml by 50% (P < 0.001), at 10 μg/ml by 53% (P < 0.001), at 30 μg/ml by 60% (P < 0.001), at 50 μg/ml by 63% (P < 0.001), and at 100 μg/ml by 67% (P < 0.001), compared to t-BuOOH group.
IC50 for MDA production was 30 μg/ml for AMFJ and 5 μg/ml for silymarin.
AMFJ prevented t-BuOOH-induced GSH depletion at the concentration of 5 μg/ml by 33% (P < 0.001), at 10 μg/ml – by 67% (P < 0.001), at 30 μg/ml – by 100% (P < 0.001), at 50 μg/ml – by 150% (P < 0.001), and at 100 μg/ml – by 217% (P < 0.001), compared to t-BuOOH group. Silymarin prevented GSH depletion by 133% (P < 0.001) at the concentration of 5 μg/ml, by 167% (P < 0.001) at 10 μg/ml, by 200% (P < 0.001) at 30 μg/ml, by 233% (P < 0.001) at 50 μg/ml, and by 250% (P < 0.001) at 100 μg/ml, compared with t-BuOOH group.
IC50 for the prevention of GSH depletion was 7.5 μg/ml for AMFJ and lower than 5 μg/ml for silymarin.
DISCUSSION
The main mechanisms of xenobiotics-induced hepatotoxicity involve the process of cytochrome P450 (CYP)-mediated bio-activation which leads to the formation of reactive metabolites and oxidative stress. Some of the most widely used toxic agents in hepatotoxicity models acting by different mechanisms are CCl4 and t-BuOOH.
In experimental toxicology, the in vitro systems are widely used for the investigation of the xenobiotics biotransformation, and for revealing the possible mechanisms of toxic stress and its prevention. Isolated liver cells are a convenient model system for evaluation of the cytoprotective effects of some promising biologically active compounds, both newly synthesized and derived from plants.
Many natural compounds investigated on isolated rat hepatocytes had their own metabolism and toxicity. For example, benzophenones, isolated from Hypericum annulatum, exerted toxic effects at the concentration of 100 μM, which might be due to the formation of toxic metabolites – benzhydrol and p-hydroxybenzophenone.[10] In our preliminary data, we found that administered alone, A. melanocarpa juice showed cytotoxic effects on isolated rat hepatocytes. We suggest that some of the juice components (probably polyphenols) had their own metabolism and possibly formed toxic metabolites, which damaged the cells.
In the present experiments, on hepatotoxicity models, we used silymarin as a reference compound. There are data that silybin (the main compound in silymarin) inactivates human cytochrome 3A4 (CYP3A4) and cytochrome 2C9, as well as the main liver glucuronosyltransferases.[16]
In this experiment, we used CCl4, a substance subjected to CYP-mediated bio-activation. It is known that CCl4 is activated by cytochrome 2E1 (CYP2E1), as well as CYP2B1, and possibly CYP3A, to form the trichloromethyl radical (●CCl3) which initiates the chain reaction of lipid peroxidation.[2] Preincubation of the hepatocytes with AMFJ resulted in protection against CCl4-induced toxicity. The effects of AMFJ on the examined parameters were similar to those of silymarin at concentrations of 5 μg/ml, 10 μg/ml, 30 μg/ml, and 50 μg/ml. At the highest concentration of 100 μg/ml, AMFJ had stronger effects on all parameters, compared to silymarin at the same concentration.
There are data that in human liver microsomes, some flavonoids exert inhibitory effects on CYP3A activity.[17] The study of Krajka-Kuzniak et al. showed that the forced feeding with A. melanocarpa juice alone decreased the activities of cytochrome CYP1A1 and CYP 1A2, and the pretreatment with the juice further reduced the activity of CYP2E1 which was decreased by N-nitrosodiethylamine.[18] In another study, Krajka-Kuzniak et al. demonstrated that plant phenols such as protocatechuic and tannic acids were weak inhibitors of CYP2E1 in rats.[19] In previous studies, we found that anthocyanins, isolated from Bulgarian red wine, showed strong hepatoprotective and antioxidant effects in CCl4-induced hepatotoxicity in vivo comparable to the effect of Vitamin E.[20] They also decreased the lipid dystrophy caused by CCl4.[20]
The cytoprotective effect of AMFJ in CCl4-induced toxicity model might be due to possible inhibiting effects on CYP3A and CYP2E1 resulting in a decreased production of toxic CCl4 metabolites. These possible CYP-inhibiting effects might be either due to the polyphenolic components of the juice, or metabolites resulting from their CYP-mediated metabolism.
The other model on isolated rat hepatocytes was t-BuOOH-induced oxidative stress. Two mechanisms for t-BuOOH action were proposed: Depletion of GSH cellular stores and oxidation of functionally important SH groups on mitochondrial enzymes, and/or changes of mitochondrial membrane integrity induced by peroxidation of membrane lipids.[3]
The metabolism of t-BuOOH to free radicals passes through several steps. In microsomal suspension (in the absence of NADPH), t-BuOOH undergoes one-electron oxidation to a peroxyl radical (reaction 1), whereas in the presence of NADPH – one-electron reduction to an alkoxyl radical (reaction 2). The toxic agent undergoes β-scission to the methyl radical (reaction 3) in isolated mitochondria and intact cells. All these radicals cause lipid peroxidation.[21,22]
(CH3)3 COOH → (CH3)3 COO● + e− + H+ (reaction 1)
(CH3)3 COOH + e− → (CH3)3 CO● + OH− (reaction 2)
(CH3)3 CO● → (CH3)2 CO + ●CH3 (reaction 3)
The results from the present study showed that in the model of t-BuOOH-induced oxidative stress, AMFJ had statistically significant cytoprotective and antioxidant activities, similar to those of silymarin at concentrations of 5 μg/ml, 10 μg/ml, 30 μg/ml, and 50 μg/ml. At the highest concentration of 100 μg/ml, AMFJ had stronger effects on all parameters in comparison with the effects of same concentration of silymarin.
Both AMFJ and silymarin had stronger cytoprotective and antioxidant activities in the t-BuOOH model in comparison with the CCl4 model.
The cytoprotective effects of AMFJ on t-BuOOH-induced oxidative stress were possibly related to its activity as a scavenger of reactive oxygen species (ROS), and these effects were comparable to the effects of silymarin a well-known ROS scavenger.[16] These data correlate with literature data on the pronounced radical scavenging effects of AMFJ, an important mechanism of its antioxidant activity,[23] demonstrated by many authors and reviewed by Chrubasik et al.,[24] Kokotkiewicz et al.,[25] and Denev et al.[26]
CONCLUSION
In this study, in two different models of toxicity on isolated rat hepatocytes, AMFJ showed statistically significant, concentration-dependent, cytoprotective and antioxidant effects, more prominent in the t-BuOOH compared to the CCl4 model. We suggest that some of the compounds in AMFJ (probably polyphenols) or their metabolites might have inhibitory effects on some CYP isoforms, playing a role in the metabolism of CCl4. Thus, in the CCl4-induced hepatotoxicity model, the cytoprotective effects of AMFJ might be predominantly due to inhibition of the metabolic bio-activation of CCl4 (possibly by CYP3A4 and CYP2E1). As polyphenols act as powerful radical scavengers, the cytoprotective effects of AMFJ in the t-BuOOH-induced hepatotoxicity model were probably predominantly due to effects on oxidative stress. The effects of AMFJ were comparable to those of silymarin, a well-known hepatoprotector and antioxidant. We suggest that the combined presence of different polyphenolic substances contributes to the stronger cytoprotective and antioxidant effects of the highest AMFJ concentration in comparison with the same concentration of silymarin, in both models of toxicity.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
ABOUT AUTHORS

Magdalena Kondeva-Burdina
Magdalena Kondeva-Burdina, PhD
Current position:
Associate Professor, Laboratory “Drug metabolism and drug toxicity”, Department of Pharmacology, Pharmacotherapy and Toxicology, Medical University-Sofia, Bulgaria.
Scientific interests:
Hepatoprotection and possible toxicity of biologically active substances from plant and synthetic origin, Processes of bioactivation and detoxication, Evaluation of the relationship structure-toxicityckers, Neuroprotection.
Member of:
Bulgarian Toxicological Society, Bulgarian Pharmaceutical Society, EUROTOX, IUTOX.

Stefka Valcheva-Kuzmanova
Stefka Valcheva-Kuzmanova, MD, PhD, D.Sc.
Current position:
Associate Professor, Head of Department of Preclinical and Clinical Pharmacology, Medical University – Varna, Bulgaria.
Scientific interests:
Pharmacology and clinical pharmacology of biologically active substances of plant origin, Pharmacology of histamine, Pharmacology of calcium channel blockers, Pharmacoeconomics of drug use.
Member of:
Bulgarian Pharmacological Society, Bulgarian Society of Clinical Pharmacology and Therapeutics, Bulgarian Medical Association.
Acknowledgments
We thank the Laboratory of Biologically Active Substances – Plovdiv, Institute of Organic Chemistry with Centre of Phytochemistry – BAS, for determining the contents of polyphenolic substances in AMFJ.
REFERENCES
- 1.Blaauboer BJ, Boobis AR, Castell JV, Coecke S, Groothuis GM, Guillouzo MA, et al. The practical applicability of hepatocyte cultures in routine testing. Altern Lab Anim. 1994;22:231–41. [Google Scholar]
- 2.Weber LW, Boll M, Stampfl A. Hepatotoxicity and mechanism of action of haloalkanes: Carbon tetrachloride as a toxicological model. Crit Rev Toxicol. 2003;33:105–36. doi: 10.1080/713611034. [DOI] [PubMed] [Google Scholar]
- 3.Drahota Z, Kriváková P, Cervinková Z, Kmonícková E, Lotková H, Kucera O, et al. Tert-butyl hydroperoxide selectively inhibits mitochondrial respiratory-chain enzymes in isolated rat hepatocytes. Physiol Res. 2005;54:67–72. doi: 10.33549/physiolres.930578. [DOI] [PubMed] [Google Scholar]
- 4.Xu S, Touyz RM. Reactive oxygen species and vascular remodelling in hypertension: Still alive. Can J Cardiol. 2006;22:947–51. doi: 10.1016/s0828-282x(06)70314-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Oszmianski J, Wojdylo A. Aronia melanocarpa phenolics and their anti-oxidant activity. Eur Food Res Technol. 2005;221:809–13. [Google Scholar]
- 6.Valcheva Kuzmanova S, Denev P, Krachanova M, Surleva A, Belcheva A. Composition and anti-oxidant activity of Aronia melanocarpa fruit juice. Varna Med Forum. 2014;3:19–24. [Google Scholar]
- 7.Singleton VL, Rossi JA. Colorimetry of total phenolics with phosphomolybdic phosphotungstic acid reagents. Am J Enol Vitic. 1965;16:144–58. [Google Scholar]
- 8.Howell AB, Reed JD, Krueger CG, Winterbottom R, Cunningham DG, Leahy M. A-type cranberry proanthocyanidins and uropathogenic bacterial anti-adhesion activity. Phytochemistry. 2005;66:2281–91. doi: 10.1016/j.phytochem.2005.05.022. [DOI] [PubMed] [Google Scholar]
- 9.Council of Europe. European Convention for the Protection of Vertebrate Animals Used for Experimental and other Scientific Purposes. CETS No. 123. 1991. [Displayed on 2007 May 30]. Available from: http://www.conventions.coe.int/treaty/Commun/QueVoulezVous.asp?NT=123ECL=ENG .
- 10.Mitcheva M, Kondeva M, Vitcheva V, Nedialkov P, Kitanov G. Effect of benzophenones from Hypericum annulatum on carbon tetrachloride-induced toxicity in freshly isolated rat hepatocytes. Redox Rep. 2006;11:3–8. doi: 10.1179/135100006X100968. [DOI] [PubMed] [Google Scholar]
- 11.Fau D, Berson A, Eugene D, Fromenty B, Fisch C, Pessayre D. Mechanism for the hepatotoxicity of the antiandrogen, nilutamide. Evidence suggesting that redox cycling of this nitroaromatic drug leads to oxidative stress in isolated hepatocytes. J Pharmacol Exp Ther. 1992;263:69–77. [PubMed] [Google Scholar]
- 12.Sharifi R, Pasalar P, Kamalinejad M, Dehpour AR, Tavangar SM, Paknejad M, et al. The effect of silymarin (Silybum marianum) on human skin fibroblasts in an in vitro wound healing model. Pharm Biol. 2013;51:298–303. doi: 10.3109/13880209.2012.721789. [DOI] [PubMed] [Google Scholar]
- 13.Essid E, Dernawi Y, Petzinger E. Apoptosis induction by OTA and TNF-α in cultured primary rat hepatocytes and prevention by silibinin. Toxins (Basel) 2012;4:1139–56. doi: 10.3390/toxins4111139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Persiani S, Sala F, Cole R, Webber G, Caselli G, Vaghi P, et al. Silibinin hemisuccinate binding to proteins in plasma and blood cell/plasma partitioning in mouse, rat, dog and man in vitro . Drug Metabol Drug Interact. 2013;28:115–22. doi: 10.1515/dmdi-2013-0013. [DOI] [PubMed] [Google Scholar]
- 15.Fau D, Eugene D, Berson A, Letteron P, Fromenty B, Fisch C, et al. Toxicity of the antiandrogen flutamide in isolated rat hepatocytes. J Pharmacol Exp Ther. 1994;269:954–62. [PubMed] [Google Scholar]
- 16.Sridar C, Goosen TC, Kent UM, Williams JA, Hollenberg PF. Silybin inactivates cytochromes P450 3A4 and 2C9 and inhibits major hepatic glucuronosyltransferases. Drug Metab Dispos. 2004;32:587–94. doi: 10.1124/dmd.32.6.587. [DOI] [PubMed] [Google Scholar]
- 17.Tsujimoto M, Horie M, Honda H, Takara K, Nishiguchi K. The structure-activity correlation on the inhibitory effects of flavonoids on cytochrome P450 3A activity. Biol Pharm Bull. 2009;32:671–6. doi: 10.1248/bpb.32.671. [DOI] [PubMed] [Google Scholar]
- 18.Krajka-Kuzniak V, Szaefer H, Ignatowicz E, Adamska T, Oszmianski J, Baer-Dubowska W. Effect of Chokeberry (Aronia melanocarpa) juice on the metabolic activation and detoxication of carcinogenic N-nitrosodiethylamine in rat liver. J Agric Food Chem. 2009;57:5071–7. doi: 10.1021/jf803973y. [DOI] [PubMed] [Google Scholar]
- 19.Krajka-Kuzniak V, Szaefer H, Baer-Dubowska W. Modulation of 3-methylcholanthrene-induced rat hepatic and renal cytochrome P450 and phase II enzymes by plant phenols: Protocatechuic and tannic acids. Toxicol Lett. 2004;152:117–26. doi: 10.1016/j.toxlet.2004.04.016. [DOI] [PubMed] [Google Scholar]
- 20.Mitcheva M, Astroug H, Drenska D, Popov A, Kassarova M. Biochemical and morphological studies on the effects of anthocyans and vitamin E on carbon tetrachloride induced liver injury. Cell Mol Biol (Noisy-le-grand) 1993;39:443–8. [PubMed] [Google Scholar]
- 21.Ollinger K, Brunk UT. Cellular injury induced by oxidative stress is mediated through lysosomal damage. Free Radic Biol Med. 1995;19:565–74. doi: 10.1016/0891-5849(95)00062-3. [DOI] [PubMed] [Google Scholar]
- 22.O’Donnell V, Burkitt MJ. Mitochondrial metabolism of a hydroperoxide to free radicals in human endothelial cells: An electron spin resonance spin-trapping investigation. Biochem J. 1994;304(Pt 3):707–13. doi: 10.1042/bj3040707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Valcheva-Kuzmanova S, Blagovic B, Valic S. Electron spin resonance measurement of radical scavenging activity of Aronia melanocarpa fruit juice. Pharmacogn Mag. 2012;8:171–4. doi: 10.4103/0973-1296.96583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chrubasik C, Li G, Chrubasik S. The clinical effectiveness of chokeberry: A systematic review. Phytother Res. 2010;24:1107–14. doi: 10.1002/ptr.3226. [DOI] [PubMed] [Google Scholar]
- 25.Kokotkiewicz A, Jaremicz Z, Luczkiewicz M. Aronia plants: A review of traditional use, biological activities, and perspectives for modern medicine. J Med Food. 2010;13:255–69. doi: 10.1089/jmf.2009.0062. [DOI] [PubMed] [Google Scholar]
- 26.Denev PN, Kratchanov CG, Ciz M, Lojek A, Kratchanova MG. Bioavailability and antioxidant activity of black chokeberry (Aronia melanocarpa) polyphenols: In vitro and in vivo evidences and possible mechanisms of action. A review. Compr Rev Food Sci Food Saf. 2012;11:471–89. [Google Scholar]


