Abstract
Microvascular invasion is a crucial histopathologic prognostic factor for hepatocellular carcinoma. We reviewed the literature and aimed to draw attention to clinicopathologic and imaging findings that may predict the presence of microvascular invasion in hepatocellular carcinoma. Imaging findings suggesting microvascular invasion are disruption of capsule, irregular tumor margin, peritumoral enhancement, multifocal tumor, increased tumor size, and increased glucose metabolism on positron emission tomography-computed tomography. In the presence of typical findings, microvascular invasion may be predicted.
Hepatocellular carcinoma (HCC) is responsible for high incidence of cancer-related mortality worldwide (1, 2). Diagnosis of HCC is a well-known topic, and now it can be made solely by imaging findings in cirrhosis (3, 4). Advances in imaging procedures and intense follow-up of high-risk patients led to increased early detection of HCC and better treatment options (5, 6). However, even in good surgical candidates, long-term survival rates remain unsatisfactory because of high recurrence (7, 8). Presence of microvascular invasion (MVI) has been reported to be one of the most important risk factors related to postsurgery tumor recurrence (9–11). Presence of MVI alters the surgical procedure. MVI is a prognostic factor associated with lower survival and higher recurrence rates. Prediction of MVI may also affect locoregional treatments along with chemotherapy protocols. This review will focus on all aspects of MVI.
The effect of microvascular invasion on survival
Early tumor recurrence is linked to increased mortality rate. Lim et al. (9) reported that MVI is a more prominent tumor recurrence predictor than the Milan criteria for HCC after surgical resection. The patients who fulfill the Milan criteria should have a solitary tumor not exceeding 5 cm or three or fewer tumors with the largest not exceeding 3 cm, and no evidence of macrovascular invasion or extrahepatic metastasis (5, 9). Lim et al. (9) concluded that patients (without MVI) exceeding the Milan criteria could achieve comparable overall survival rates after surgical resection, relative to patients within the Milan criteria. However, overall survival decreased significantly with MVI, independently of the Milan criteria.
In a different study, authors grouped the patients into mild (one to five invaded vessels) and severe (more than five invaded vessels) MVI groups (5). They found that the three-year recurrence-free survival rates for patients with and without MVI were 27.7% and 62.5%, respectively. They also reported that recurrence-free survival rates at two years for patients without MVI, with mild MVI, and severe MVI were 75.9%, 47.2%, and 32.7%, respectively (5). Goh et al. (12) studied patients who had surgical resection of multifocal HCC and revealed that the presence of MVI and the number of nodules (which is also a result of MVI) were more important prognostic factors than tumor size (12). They also suggested that the current American Joint Committee for Cancer TNM staging system, which uses tumor size, but not MVI, as prognostic criteria for multifocal HCC, needs to be revised (12).
The indicators of microvascular invasion
Despite its significance in HCC assessment, MVI can rarely be diagnosed preoperatively. However, the presence of MVI may be predicted by key radiologic findings and specific laboratory tests. In this part of the paper, each characteristic finding that may be associated with MVI, will be discussed separately.
Capsule
A surrounding fibrous capsule may accompany HCC in 10%–70% of cases (13, 14). On imaging, the capsule is seen as a low-attenuating/hypointense ring on arterial phase and high-attenuating/hyperintense ring on delayed phase images (13, 15) (Fig. 1). The microvessels in the capsule are thought to be responsible for the late enhancement because of slow flow (13, 15).
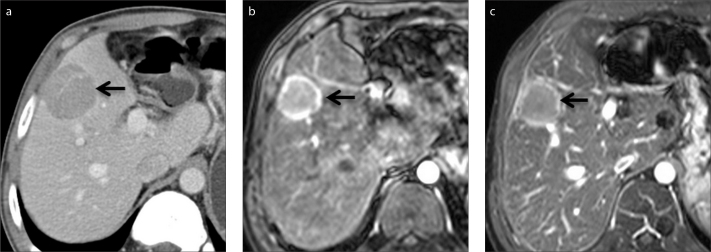
Figure 1.
a–c. A 38-year-old man with hepatocellular carcinoma (HCC). The capsule of HCC (arrow) is seen as a hyperattenuating ring on axial contrast-enhanced portal venous phase CT (a) and hyperintense ring on late arterial (b) and portal venous (c) phase gadolinium-enhanced MRI.
Lim et al. (13) revealed that the presence of a capsule on computed tomography (CT) was significantly correlated with histopathology (P < 0.001) and disruption of the capsule seen on CT was correlated with MVI (P < 0.001) (Fig. 2). They also reported that the presence of an intact capsule on CT was closely correlated with the absence of MVI (13). In a different study authors reported that there was no significant correlation between MVI and presence of radiologic capsule (16). On the other hand, Adachi et al. (17) reported that a fibrous capsule was a predictor of portal venous invasion since cancer cells frequently invade the vessels of the fibrous capsule. Witjes et al. (18) also reported that MVI was significantly associated with the presence of a capsule. However, the latter two studies did not clarify the phase that was used for capsule assessment. Capsule enhancement may occur on all phases according to its vascularity; thus, early enhancement of the capsule cannot be distinguished from peritumoral enhancement (16). Similarly, Ariizumi et al. (19) reported that there were no clinical or magnetic resonance imaging (MRI) findings to predict MVI, except for the radiologic capsule on dynamic studies and nonsmooth tumor margin on axial hepatobiliary phase (Fig. 3). For this reason, the presence of capsule should be assessed on delayed phase images; however, the correlation between the capsule and the presence of MVI remains unclear.
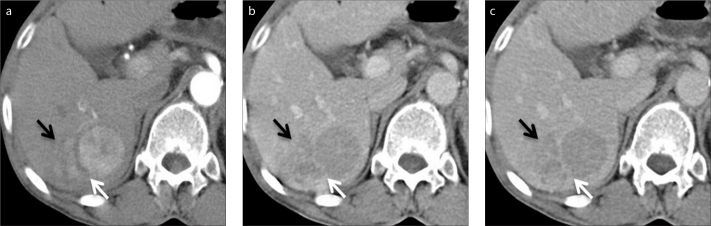
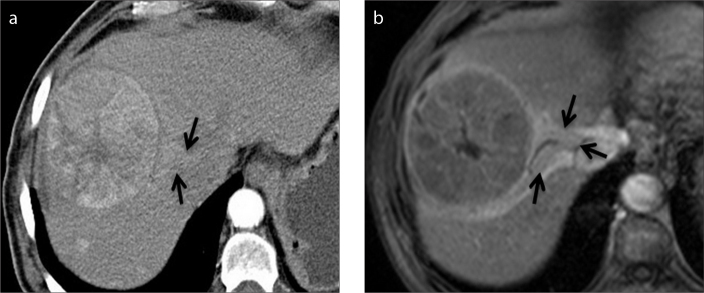
Figure 2.
a–c. A 68-year-old woman with HCC and elevated liver enzymes. Axial contrast-enhanced CT images in arterial (a), portal venous (b) and delayed (c) phase show the capsule of HCC is disrupted (white arrows) in the posterolateral side of the tumor. Tumor invasion adjacent to the disruption is also seen (black arrows).
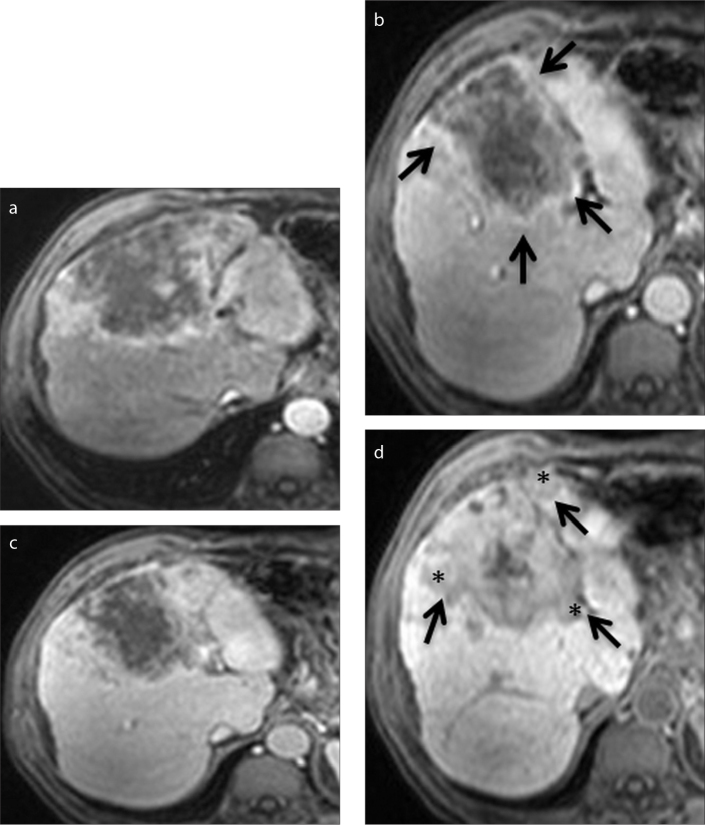
Figure 3.
a–d. A 52-year-old man with cirrhotic liver. Axial contrast-enhanced arterial (a), portal venous (b), delayed (c), and hepatobiliary (d) phase Gd-BOPTA-enhanced MRI scans show a heterogeneous mass that is histopathologically proven to be HCC. Capsule is best seen on the portal venous phase image (b, arrows), whereas tumor margin is best visualized on hepatobiliary phase images (d, arrows). Capsular disruption and irregular tumor margin are present (d, asterisks).
Tumor margin
The tumor margin may be either smooth or not. Depending on the appearance of the tumor margin, Chou et al. (20) concluded that nonsmooth tumor margins, detected on multiphasic CT, correlated with the histopathologic presence and location of MVI. In their study, three subtypes were defined as follows: simple nodular type, simple nodular type with extranodular growth, and confluent multinodular type (Fig. 4). They accepted last two subtypes as nonsmooth tumor margin and reported that nonsmooth tumor margins had 81.7% sensitivity and 88.1% specificity for predicting MVI. Nakashima et al. (21) also reported that extranodular growth and confluent multinodular type in small HCC (≤2 cm) had a higher frequency of portal vein invasion and intrahepatic metastases than single nodular type. Latest MRI protocols improved the visualization of tumor margin particularly on hepatobiliary phase images (19). Ariizumi et al. (19) reported that nonsmooth tumor margin in the hepatobiliary phase of Gd-EOB-DTPA-enhanced MRI (Fig. 3) can be used as a preoperative predictor of MVI in patients with HCC within the Milan criteria.
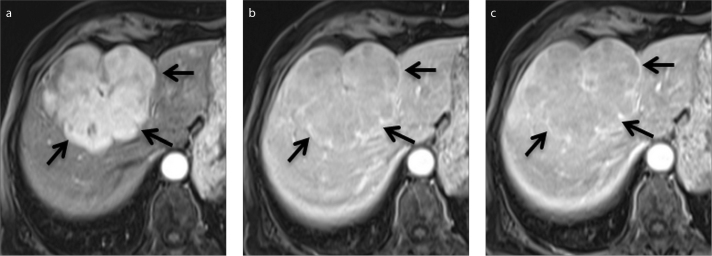
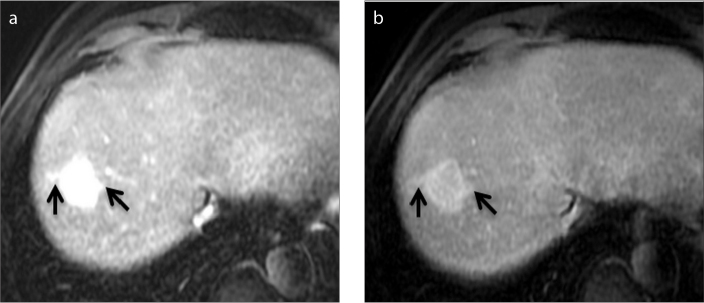
Figure 4.
a–c. An 82-year-old woman with a lobulated hypervascular liver mass. Axial contrast-enhanced arterial (a), portal venous (b) and delayed (c) phase Gd-BOPTA-enhanced MRI of HCC demonstrate a nonsmooth, multinodular tumor margin (arrows).
Peritumoral enhancement
It is known that peritumoral enhancement may accompany most malignant, particularly hypervascular, hepatic tumors and result in tumor size overestimation (22). This incident can be due to changes in the peritumoral liver parenchyma caused by parenchymal compression or portal venous obstruction by malignant liver tumors, or it can be due to a siphoning effect by hypervascular tumors (22).
Kim et al. (16) categorized the pattern of peritumoral enhancement as wedge-shaped and irregular circumferential enhancement (Figs. 5, 6). They reported that wedge-shaped enhancement could be due to nontumorous arterioportal shunts rather than tumorous ones and it was not a statistically significant risk factor for MVI; on the other hand, irregular circumferential peritumoral enhancement could be a preoperative predictor of MVI. The sensitivity, specificity, positive predictive value, and negative predictive value of irregular circumferential peritumoral enhancement in the prediction of MVI were 74.3%, 82.9%, 81.3%, and 76.3%, respectively (16). The crucial point in their study was obtaining four arterial-phase MRI scans at the same level. There was statistically significant correlation between the peritumoral enhancement and MVI on the third arterial phase (P = 0.004). They argued that the ability of single arterial phase image to evaluate peritumoral enhancement was inadequate compared with multiarterial phase images and suggested that peritumoral enhancement could be estimated more accurately with a multiarterial phase study (16). In a different study, Chou et al. (20) reported that peritumoral enhancement on multiphasic CT was not a significant risk factor for MVI. However, this contradiction may be due to lower contrast resolution of CT and single arterial phase imaging. Nishie et al. (23) evaluated the same topic from a different point of view and reported that the area of peritumoral enhancement might be larger in the MVI group, particularly in small HCCs (≤3 cm) with positive and negative predictive values of 71.4% and 75%, respectively (23).
Figure 5.
a, b. A 77-year-old man with HCC. Wedge-shaped peritumoral enhancement (arrows) is seen on axial contrast-enhanced arterial phase CT (a) and portal venous phase Gd-BOPTA-enhanced MRI (b).
Figure 6.
a, b. A 63-year-old man with chronic liver disease with HCC detection during follow-up. Irregular peritumoral enhancement (arrows) is seen on both sides of HCC on axial arterial (a) and portal venous (b) phase Gd-EOB-DTPA-enhanced MRI.
Multifocality
It is crucial to reveal if multiple lesions in the liver are multicentric tumors or metastases from a main tumor (24). Moreover, intrahepatic metastasis may be either due to intravascular micrometastases (i.e., MVI) or tumor satellite micronodules, which are isolated tumor microfoci in the surrounding parenchyma (25) (Fig. 7). However, such detailed differentiation is difficult to determine solely based on radiologic findings.
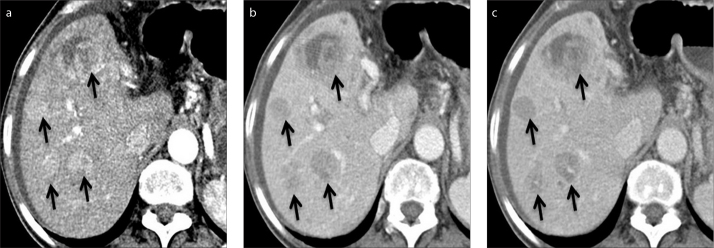
Figure 7.
a–c. A 73-year-old woman with chronic liver disease and ascites. Axial contrast-enhanced CT images in arterial (a), portal venous (b), and delayed (c) phase show multifocal HCC (arrows).
Chandarana et al. (26) reported that tumor multifocality, as detected on both MRI and pathologic examination, was the only parameter that may predict MVI. Their results showed that the presence of three or more tumors on MRI and four or more tumors at pathologic examination had high specificity (88.2% and 91.2%, respectively) for the prediction of MVI (26). However, multifocality was not associated with MVI in a recently published study (27).
The incidence of tumor multicentricity increases with progression and duration of chronic liver disease. Kubo et al. (24) evaluated clinicopathologic criteria for multicentric HCC and found that cumulative survival rate was significantly higher in multicentric HCC than in HCC with intrahepatic metastasis. Shi et al. (25) studied the micrometastases of solitary HCC and reported that the spread of the tumor satellite micronodules ranged from 0.10 to 0.80 cm, whereas intravascular micrometastases may range from 0.05 to 6.10 cm from the main tumor. Therefore, differentiation of tumor multiplicity from intrahepatic metastasis (i.e., MVI) is challenging by imaging. This drawback explains the aforementioned discrepancy in prediction of MVI by tumor multifocality. Thus, tumor multifocality, which is demonstrated solely by imaging, is a questionable parameter for prediction of MVI.
Tumor size
Due to increased peritumoral enhancement, size measurement may be overestimated by imaging (22). Therefore, hepatobiliary phase images should be used for accurate results (16). Tumor size showed statistically significant association with the presence of MVI in several studies (16, 20, 27, 28). Significant tumor size cutoffs for positive MVI were reported as >5 cm (P = 0.001) in Ahn et al. (27); 5.6±4.3 cm vs. 2.7±1.7 cm (P < 0.001) in Chou et al. (20); and ≥3cm (P = 0.0013) in Hirokawa et al. (28). Chandarana et al. (26) found no significant correlation between tumor size and microvascular invasion, but they emphasized that this contradiction might be due to patient selection bias with a mean tumor size of 2.1 cm (26).
Increased tumor metabolism – FDG
Fluorine-18 fluorodeoxyglucose positron emission tomography (18F-FDG PET) has a low sensitivity for detecting well-differentiated HCCs (29, 30). However, FDG accumulation is correlated with tumor aggressiveness, and high accumulation of FDG is associated with a worse prognosis (29, 30).
Kornberg et al. (31) reported that preoperative 18F-FDG uptake on PET was a reliable predictor of MVI with high positive and negative predictive values (87.5% and 88.5%, respectively). They also concluded that patients beyond the Milan criteria and negative preoperative PET scans could achieve a post-transplant three-year recurrence-free survival, which is comparable to patients within the Milan criteria (31). However, lack of quantitative uptake measurement was a limitation of their study (31). On the other hand, Ahn et al. (27) reported that the ratio of tumor maximum standardized uptake values (SUV) to normal liver mean SUV (TSUVmax/LSUVmean) of 1.2 or more had a statistically significant association with MVI (P < 0.001) (Fig. 8).
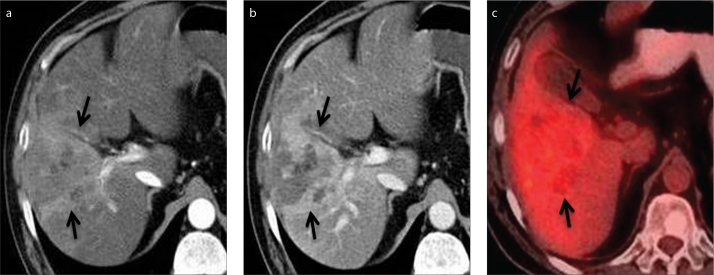
Figure 8.
a–c. A 55-year-old man with HCC and elevated liver enzymes. Axial contrast-enhanced CT images in arterial (a) and portal venous (b) phase show HCC in the right lobe of the liver (arrows). PET-CT image (c) shows 18F-FDG uptake (arrows) in the right lobe of the liver with a SUVmax value of 4.2.
From a different point of view, Ochi et al. (30) showed that SUVmax significantly correlated with the distance of the microsatellite lesion (due to MVI) from the main tumor. They concluded that the cutoff value of SUVmax for microsatellite distance >1 cm was 8.8, with a negative predictive value of 97.3% (30).
In the future, PET/MRI scanners will probably be the modality of choice for predicting MVI, due to high contrast resolution and the ability to measure the degree of metabolic activity, which is associated with the presence of MVI.
Tumor markers
As tumor markers, alpha-fetoprotein (AFP), particularly AFP-L3 form, des-gamma-carboxy prothrombin (DCP), gamma-glutamyltransferase (GGT) and protein induced by vitamin K absence or antagonist II (PIVKA II) are widely used for the detection of HCC (28, 32). However, recent studies focused on predicting MVI rather than detection of HCC, by serum levels of tumor markers.
Hirokawa et al. (28) showed that PIVKA-II (≥150 mAU/mL) and positivity of AFP-L3 are independent risk factors for MVI in solitary HCC. AFP-L3 positivity and failure of AFP-L3 conversion to negativity or further elevation of AFP-L3 are associated with aggressive tumor behavior and worse histologic grade results in early tumor recurrence (i.e., in MVI) (28, 33). In a different study, DCP >100 mAU/mL was proposed as a reliable predictor for MVI even in small HCCs (≤2 cm), with a positive predictive value of 79.3% (34). They also reported that DCP was more specific than AFP for MVI (34).
Fan et al. (35) proposed AFP level (>100 μg/L) as a predictor of MVI (P = 0.004) (35). However, as a limitation of their study, they did not analyze AFP glycoforms separately. It is known that AFP-L3 form is not proportional to AFP; thus, it can be used as an independent and significant factor for diagnosis of HCC, and it is associated with tumor aggressiveness (28, 32).
Shirabe et al. (36) designed a scoring system using tumor size, serum DCP levels, and values of SUVmax to predict MVI. They reported that when the tumor size is ≥3.6 cm, SUVmax ≥4.2, and the serum DCP level ≥101 mAU/mL, MVI can be predicted precisely with a sensitivity of 100% and specificity of 90.9% (36).
Zhao et al. (37) studied preoperative predictors of MVI in multinodular HCC. They determined serum AFP level >400 mg/L, serum GGT level >130 U/L, total tumor diameter >8 cm, and tumor number >3 as preoperative predictors of MVI in patients with multinodular HCC.
Needle biopsy
HCCs are classified as well-differentiated, moderately differentiated, poorly differentiated, or undifferentiated according to the Edmondson–Steiner grading system (38). Studies showed that tumor grade is an independent predictor of MVI (16, 20). However, Pawlik et al. (39) demonstrated that the needle core biopsy of HCC to determine histologic grade was inaccurate due to heterogeneous tumor differentiation resulting in sampling errors. Also, there is a risk for needle-tract implantation as a complication of biopsy (40). In such cases, resection of the needle tract should be performed during curative surgery. Thus, needle core biopsy is not an effective method to show MVI before surgical treatment of HCC.
The effect of microvascular invasion on treatment
Surgery
Surgical treatments in HCC include liver transplantation, as well as anatomical and limited liver resections. Liver transplantation is proposed as the gold standard treatment option for early HCC, particularly in cirrhotic patients (41). However, in practice, transplantation promotes worse results than resection due to lack of donors and long waiting time (41). Thus, partial resection remains as a suitable option (42).
Partial liver resection can be performed as limited resection with an acceptable tumor-free margin (1 cm or greater) or as anatomical resection removing the portal unit along with the tumor and at least one Couinaud’s segment (42).
Resection of the cirrhotic liver may result in postoperative hepatic failure, which is linked to the degree of parenchymal loss. Anatomic resection is more challenging because of its technical difficulties and wider extent of parenchymal loss (42). However, in cirrhotic yet good functioning livers with a small HCC (≤4 cm), anatomic resection results in better disease-free survival rates than limited resection (5–8 year disease-free survival, 54%–45% vs. 35%–6%) without increasing the postoperative risk (42). Additionally, radiofrequency ablation (RFA) achieved comparable three- and five-year overall survival rates with surgical resection in patients with very early stage HCC (≤2 cm), although surgery resulted in better disease-free survival than RFA (43). However, in a different study, anatomic resection achieved better recurrence-free survival rate in small HCC (≤3 cm) accompanied by three or fewer nodules (44). They also determined that success rate was more prominent in nonboundary type HCC (single nodular type with extranodular growth, confluent multinodular type, and invasive type) than in the boundary type (single nodular type) (44). They also revealed that the distance of micrometastases from the main mass was significantly longer in the nonboundary type (9.5 mm) than in the boundary type (3.1 mm) (44). Anatomic resection is more effective in nonboundary type HCC, due to increased risk of MVI and intrahepatic metastasis. In a recently published study, anatomic resection had significantly better overall and disease-free survival rates than limited resection in solitary small HCC (≤5 cm) with MVI (35).
In a retrospective analysis of 1566 patients with liver transplantation for HCC from 36 centers, MVI was the most significant parameter associated with poor survival and tumor recurrence rate (45).
Locoregional treatment
Local treatments play a major role in the management of HCC. RFA is the preferred local thermal ablation therapy method for HCC compared with ethanol and acetic acid injection (46). Also, with the recent embolic microspheres that release the chemotherapeutic agent in a controlled fashion, transcatheter arterial chemoembolization (TACE) was shown to significantly increase safety and effectiveness, particularly for multinodular and recurrent HCC (47).
Even small tumors (≤3 cm) may be accompanied with MVI and associated with increased risk of local recurrence (28, 34, 48). Nakashima et al. (21) and Ueno et al. (44) reported that the distance between the micrometastasis and the main tumor was 10 mm or shorter in most of the patients. Thus, it is crucial to ablate the main tumor with an adequate surrounding liver tissue of ≥10 mm to prevent the recurrence and local control of MVI (28, 48). Local recurrence rate was shown to be higher after RFA compared with anatomic resection (49). However, in a recently published review, authors concluded that RFA should be preferred over resection in small HCC (≤2 cm) (50). Furthermore, in a recently published meta-analysis, authors concluded that in early HCCs (<5 cm; within Milan criteria) there was no significant difference between RFA and hepatic resection in overall survival, recurrence-free survival, and in-hospital mortality. Nevertheless, RFA group had higher recurrence, yet lower complication rates and shorter hospitalization period (51).
Jin et al. (52) compared the treatment outcomes of TACE and surgery/RFA according to the presence of MVI in patients with recurrent HCC with a median tumor size of 1.5 cm (1–10 cm). They revealed that TACE provided significantly better overall survival among early (≤12 months) recurrent MVI-positive patients than surgery/RFA after curative resection for HCC (52). However, they suggested that a large-scale study is needed to confirm the results of their study.
Chemotherapy
Patients with early HCC are treated by surgical resection, locoregional treatment, or both. In advanced HCC, treatment options are limited due to tumor characteristics and underlying cirrhosis. Thus, aforementioned invasive methods remain inefficient in most cases.
Sorafenib is an oral multikinase inhibitor of the vascular endothelial growth factor receptor and platelet-derived growth factor receptor (53–55). It inhibits tumor-cell proliferation and tumor angiogenesis, and thus, increases the rate of apoptosis in a wide range of tumor models (55). In phase 3 randomized, double-blind, placebo-controlled trials, patients with advanced HCC (vascular invasion and extrahepatic spread) treated with sorafenib had longer median survival and time to progression than those given placebo (53–55).
Conclusion
In this review, we highlighted the clinicopathologic and imaging parameters that may predict the presence of MVI in HCC. Radiologists and oncologists should be aware of the importance of MVI and any significant findings that may predict MVI.
Main points.
Microvascular invasion (MVI) is an important histopathologic prognostic factor in the assessment of hepatocellular carcinoma.
MVI may be predicted with typical imaging findings such as capsular disruption, irregular tumor margin, peritumoral enhancement, multifocal tumor, increased tumor size, and increased uptake on PET/CT.
However, imaging findings cannot precisely determine the presence of MVI in every case. Moreover, needle core biopsy may also fail to demonstrate MVI due to sampling errors.
Footnotes
Conflict of interest disclosure
The authors declared no conflicts of interest.
References
- 1.Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin. 2005;55:74–108. doi: 10.3322/canjclin.55.2.74. http://dx.doi.org/10.3322/canjclin.55.2.74. [DOI] [PubMed] [Google Scholar]
- 2.Yang JD, Roberts LR. Epidemiology and management of hepatocellular carcinoma. Infect Dis Clin North Am. 2010;24:899–919. doi: 10.1016/j.idc.2010.07.004. http://dx.doi.org/10.1016/j.idc.2010.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Baron RL, Oliver JH, 3rd, Dodd GD, 3rd, Nalesnik M, Holbert BL, Carr B. Hepatocellular carcinoma: evaluation with biphasic, contrast-enhanced, helical CT. Radiology. 1996;199:505–511. doi: 10.1148/radiology.199.2.8668803. http://dx.doi.org/10.1148/radiology.199.2.8668803. [DOI] [PubMed] [Google Scholar]
- 4.Kim T, Murakami T, Oi H, et al. Detection of hypervascular hepatocellular carcinoma by dynamic MRI and dynamic spiral CT. J Comput Assist Tomogr. 1995;19:948–954. doi: 10.1097/00004728-199511000-00020. http://dx.doi.org/10.1097/00004728-199511000-00020. [DOI] [PubMed] [Google Scholar]
- 5.Sumie S, Nakashima O, Okuda K, et al. The significance of classifying microvascular invasion in patients with hepatocellular carcinoma. Ann Surg Oncol. 2014;21:1002–1009. doi: 10.1245/s10434-013-3376-9. http://dx.doi.org/10.1245/s10434-013-3376-9. [DOI] [PubMed] [Google Scholar]
- 6.Takayama T, Makuuchi M, Hirohashi S, et al. Early hepatocellular carcinoma as an entity with a high rate of surgical cure. Hepatology. 1998;28:1241–1246. doi: 10.1002/hep.510280511. http://dx.doi.org/10.1002/hep.510280511. [DOI] [PubMed] [Google Scholar]
- 7.Nagasue N, Uchida M, Makino Y, et al. Incidence and factors associated with intrahepatic recurrence following resection of hepatocellular carcinoma. Gastroenterology. 1993;105:488–494. doi: 10.1016/0016-5085(93)90724-q. [DOI] [PubMed] [Google Scholar]
- 8.Poon RT, Fan ST, Lo CM, Liu CL, Wong J. Intrahepatic recurrence after curative resection of hepatocellular carcinoma: long-term results of treatment and prognostic factors. Ann Surg. 1999;229:216–222. doi: 10.1097/00000658-199902000-00009. http://dx.doi.org/10.1097/00000658-199902000-00009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lim KC, Chow PK, Allen JC, et al. Microvascular invasion is a better predictor of tumor recurrence and overall survival following surgical resection for hepatocellular carcinoma compared to the Milan criteria. Ann Surg. 2011;254:108–113. doi: 10.1097/SLA.0b013e31821ad884. http://dx.doi.org/10.1097/SLA.0b013e31821ad884. [DOI] [PubMed] [Google Scholar]
- 10.Shah SA, Greig PD, Gallinger S, et al. Factors associated with early recurrence after resection for hepatocellular carcinoma and outcomes. J Am Coll Surg. 2006;202:275–283. doi: 10.1016/j.jamcollsurg.2005.10.005. http://dx.doi.org/10.1016/j.jamcollsurg.2005.10.005. [DOI] [PubMed] [Google Scholar]
- 11.Sumie S, Kuromatsu R, Okuda K, et al. Microvascular invasion in patients with hepatocellular carcinoma and its predictable clinicopathological factors. Ann Surg Oncol. 2008;15:1375–1382. doi: 10.1245/s10434-008-9846-9. http://dx.doi.org/10.1245/s10434-008-9846-9. [DOI] [PubMed] [Google Scholar]
- 12.Goh BK, Chow PK, Teo JY, et al. Number of nodules, Child-Pugh status, margin positivity, and microvascular invasion, but not tumor size, are prognostic factors of survival after liver resection for multifocal hepatocellular carcinoma. J Gastrointest Surg. 2014;18:1477–1485. doi: 10.1007/s11605-014-2542-0. http://dx.doi.org/10.1007/s11605-014-2542-0. [DOI] [PubMed] [Google Scholar]
- 13.Lim JH, Choi D, Park CK, Lee WJ, Lim HK. Encapsulated hepatocellular carcinoma: CT-pathologic correlations. Eur Radiol. 2006;16:2326–2333. doi: 10.1007/s00330-006-0203-8. http://dx.doi.org/10.1007/s00330-006-0203-8. [DOI] [PubMed] [Google Scholar]
- 14.Ros PR, Murphy BJ, Buck JL, Olmedilla G, Goodman Z. Encapsulated hepatocellular carcinoma: radiologic findings and pathologic correlation. Gastrointest Radiol. 1990;15:233–237. doi: 10.1007/BF01888783. http://dx.doi.org/10.1007/BF01888783. [DOI] [PubMed] [Google Scholar]
- 15.Grazioli L, Olivetti L, Fugazzola C, et al. The pseudocapsule in hepatocellular carcinoma: correlation between dynamic MR imaging and pathology. Eur Radiol. 1999;9:62–67. doi: 10.1007/s003300050629. http://dx.doi.org/10.1007/s003300050629. [DOI] [PubMed] [Google Scholar]
- 16.Kim H, Park MS, Choi JY, et al. Can microvessel invasion of hepatocellular carcinoma be predicted by pre-operative MRI? Eur Radiol. 2009;19:1744–1751. doi: 10.1007/s00330-009-1331-8. http://dx.doi.org/10.1007/s00330-009-1331-8. [DOI] [PubMed] [Google Scholar]
- 17.Adachi E, Maeda T, Kajiyama K, et al. Factors correlated with portal venous invasion by hepatocellular carcinoma: univariate and multivariate analyses of 232 resected cases without preoperative treatments. Cancer. 1996;77:2022–2031. doi: 10.1002/(SICI)1097-0142(19960515)77:10<2022::AID-CNCR9>3.0.CO;2-S. http://dx.doi.org/10.1002/(SICI)1097-0142(19960515)77:10<2022::AID-CNCR9>3.0.CO;2-S. [DOI] [PubMed] [Google Scholar]
- 18.Witjes CD, Willemssen FE, Verheij J, et al. Histological differentiation grade and microvascular invasion of hepatocellular carcinoma predicted by dynamic contrast-enhanced MRI. J Magn Reson Imaging. 2012;36:641–647. doi: 10.1002/jmri.23681. http://dx.doi.org/10.1002/jmri.23681. [DOI] [PubMed] [Google Scholar]
- 19.Ariizumi S, Kitagawa K, Kotera Y, et al. A non-smooth tumor margin in the hepatobiliary phase of gadoxetic acid disodium (Gd-EOB-DTPA)-enhanced magnetic resonance imaging predicts microscopic portal vein invasion, intrahepatic metastasis, and early recurrence after hepatectomy in patients with hepatocellular carcinoma. J Hepatobiliary Pancreat Sci. 2011;18:575–585. doi: 10.1007/s00534-010-0369-y. http://dx.doi.org/10.1007/s00534-010-0369-y. [DOI] [PubMed] [Google Scholar]
- 20.Chou CT, Chen RC, Lin WC, Ko CJ, Chen CB, Chen YL. Prediction of microvascular invasion of hepatocellular carcinoma: preoperative CT and histopathologic correlation. AJR Am J Roentgenol. 2014;203:W253–259. doi: 10.2214/AJR.13.10595. http://dx.doi.org/10.2214/AJR.13.10595. [DOI] [PubMed] [Google Scholar]
- 21.Nakashima Y, Nakashima O, Tanaka M, Okuda K, Nakashima M, Kojiro M. Portal vein invasion and intrahepatic micrometastasis in small hepatocellular carcinoma by gross type. Hepatol Res. 2003;26:142–147. doi: 10.1016/s1386-6346(03)00007-x. http://dx.doi.org/10.1016/S1386-6346(03)00007-X. [DOI] [PubMed] [Google Scholar]
- 22.Kanematsu M, Hoshi H, Yamada T, et al. Overestimating the size of hepatic malignancy on helical CT during arterial portography: equilibrium phase CT and pathology. J Comput Assist Tomogr. 1997;21:713–719. doi: 10.1097/00004728-199709000-00006. http://dx.doi.org/10.1097/00004728-199709000-00006. [DOI] [PubMed] [Google Scholar]
- 23.Nishie A, Yoshimitsu K, Asayama Y, et al. Radiologic detectability of minute portal venous invasion in hepatocellular carcinoma. AJR Am J Roentgenol. 2008;190:81–87. doi: 10.2214/AJR.07.2810. http://dx.doi.org/10.2214/AJR.07.2810. [DOI] [PubMed] [Google Scholar]
- 24.Kubo S, Nishiguchi S, Hirohashi K, et al. Clinicopathological criteria for multicentricity of hepatocellular carcinoma and risk factors for such carcinogenesis. Jpn J Cancer Res. 1998;89:419–426. doi: 10.1111/j.1349-7006.1998.tb00580.x. http://dx.doi.org/10.1111/j.1349-7006.1998.tb00580.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Shi M, Zhang CQ, Zhang YQ, Liang XM, Li JQ. Micrometastases of solitary hepatocellular carcinoma and appropriate resection margin. World J Surg. 2004;28:376–381. doi: 10.1007/s00268-003-7308-x. http://dx.doi.org/10.1007/s00268-003-7308-x. [DOI] [PubMed] [Google Scholar]
- 26.Chandarana H, Robinson E, Hajdu CH, Drozhinin L, Babb JS, Taouli B. Microvascular invasion in hepatocellular carcinoma: is it predictable with pretransplant MRI? AJR Am J Roentgenol. 2011;196:1083–1089. doi: 10.2214/AJR.10.4720. http://dx.doi.org/10.2214/AJR.10.4720. [DOI] [PubMed] [Google Scholar]
- 27.Ahn SY, Lee JM, Joo I, et al. Prediction of microvascular invasion of hepatocellular carcinoma using gadoxetic acid-enhanced MR and F-FDG PET/CT. Abdom Imaging. 2015;40:843–851. doi: 10.1007/s00261-014-0256-0. http://dx.doi.org/10.1007/s00261-014-0256-0. [DOI] [PubMed] [Google Scholar]
- 28.Hirokawa F, Hayashi M, Miyamoto Y, et al. Outcomes and predictors of microvascular invasion of solitary hepatocellular carcinoma. Hepatol Res. 2014;44:846–853. doi: 10.1111/hepr.12196. http://dx.doi.org/10.1111/hepr.12196. [DOI] [PubMed] [Google Scholar]
- 29.Cheung TT, Chan SC, Ho CL, et al. Can positron emission tomography with the dual tracers [11 C]acetate and [18 F]fludeoxyglucose predict microvascular invasion in hepatocellular carcinoma? Liver Transpl. 2011;17:1218–1225. doi: 10.1002/lt.22362. http://dx.doi.org/10.1002/lt.22362. [DOI] [PubMed] [Google Scholar]
- 30.Ochi H, Hirooka M, Hiraoka A, et al. F-FDG-PET/CT predicts the distribution of microsatellite lesions in hepatocellular carcinoma. Mol Clin Oncol. 2014;2:798–804. doi: 10.3892/mco.2014.328. http://dx.doi.org/10.3892/mco.2014.328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kornberg A, Freesmeyer M, Barthel E, et al. 18F-FDG-uptake of hepatocellular carcinoma on PET predicts microvascular tumor invasion in liver transplant patients. Am J Transplant. 2009;9:592–600. doi: 10.1111/j.1600-6143.2008.02516.x. http://dx.doi.org/10.1111/j.1600-6143.2008.02516.x. [DOI] [PubMed] [Google Scholar]
- 32.Zhao YJ, Ju Q, Li GC. Tumor markers for hepatocellular carcinoma. Mol Clin Oncol. 2013;1:593–598. doi: 10.3892/mco.2013.119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zhang XF, Lai EC, Kang XY, et al. Lens culinaris agglutinin-reactive fraction of alpha-fetoprotein as a marker of prognosis and a monitor of recurrence of hepatocellular carcinoma after curative liver resection. Ann Surg Oncol. 2011;18:2218–2223. doi: 10.1245/s10434-011-1613-7. http://dx.doi.org/10.1245/s10434-011-1613-7. [DOI] [PubMed] [Google Scholar]
- 34.Yamashita Y, Tsuijita E, Takeishi K, et al. Predictors for microinvasion of small hepatocellular carcinoma ≤ 2 cm. Ann Surg Oncol. 2012;19:2027–2034. doi: 10.1245/s10434-011-2195-0. http://dx.doi.org/10.1245/s10434-011-2195-0. [DOI] [PubMed] [Google Scholar]
- 35.Fan LF, Zhao WC, Yang N, Yang GS. Alpha-fetoprotein: the predictor of microvascular invasion in solitary small hepatocellular carcinoma and criterion for anatomic or non-anatomic hepatic resection. Hepatogastroenterology. 2013;60:825–836. doi: 10.5754/hge121039. [DOI] [PubMed] [Google Scholar]
- 36.Shirabe K, Toshima T, Kimura K, et al. New scoring system for prediction of microvascular invasion in patients with hepatocellular carcinoma. Liver Int. 2014;34:937–941. doi: 10.1111/liv.12459. http://dx.doi.org/10.1111/liv.12459. [DOI] [PubMed] [Google Scholar]
- 37.Zhao WC, Fan LF, Yang N, Zhang HB, Chen BD, Yang GS. Preoperative predictors of microvascular invasion in multinodular hepatocellular carcinoma. Eur J Surg Oncol. 2013;39:858–864. doi: 10.1016/j.ejso.2013.04.003. http://dx.doi.org/10.1016/j.ejso.2013.04.003. [DOI] [PubMed] [Google Scholar]
- 38.Edmondson HA, Steiner PE. Primary carcinoma of the liver: a study of 100 cases among 48,900 necropsies. Cancer. 1954;7:462–503. doi: 10.1002/1097-0142(195405)7:3<462::aid-cncr2820070308>3.0.co;2-e. http://dx.doi.org/10.1002/1097-0142(195405)7:3<462::AID-CNCR2820070308>3.0.CO;2-E. [DOI] [PubMed] [Google Scholar]
- 39.Pawlik TM, Gleisner AL, Anders RA, Assumpcao L, Maley W, Choti MA. Preoperative assessment of hepatocellular carcinoma tumor grade using needle biopsy: implications for transplant eligibility. Ann Surg. 2007;245:435–442. doi: 10.1097/01.sla.0000250420.73854.ad. http://dx.doi.org/10.1097/01.sla.0000250420.73854.ad. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kim SH, Lim HK, Lee WJ, Cho JM, Jang HJ. Needle-tract implantation in hepatocellular carcinoma: frequency and CT findings after biopsy with a 19.5-gauge automated biopsy gun. Abdom Imaging. 2000;25:246–250. doi: 10.1007/s002610000025. http://dx.doi.org/10.1007/s002610000025. [DOI] [PubMed] [Google Scholar]
- 41.Llovet JM, Fuster J, Bruix J. Intention-to-treat analysis of surgical treatment for early hepatocellular carcinoma: resection versus transplantation. Hepatology. 1999;30:1434–1440. doi: 10.1002/hep.510300629. http://dx.doi.org/10.1002/hep.510300629. [DOI] [PubMed] [Google Scholar]
- 42.Regimbeau JM, Kianmanesh R, Farges O, Dondero F, Sauvanet A, Belghiti J. Extent of liver resection influences the outcome in patients with cirrhosis and small hepatocellular carcinoma. Surgery. 2002;131:311–317. doi: 10.1067/msy.2002.121892. http://dx.doi.org/10.1067/msy.2002.121892. [DOI] [PubMed] [Google Scholar]
- 43.Wang JH, Wang CC, Hung CH, Chen CL, Lu SN. Survival comparison between surgical resection and radiofrequency ablation for patients in BCLC very early/early stage hepatocellular carcinoma. J Hepatol. 2012;56:412–418. doi: 10.1016/j.jhep.2011.05.020. http://dx.doi.org/10.1016/j.jhep.2011.05.020. [DOI] [PubMed] [Google Scholar]
- 44.Ueno S, Kubo F, Sakoda M, et al. Efficacy of anatomic resection vs. nonanatomic resection for small nodular hepatocellular carcinoma based on gross classification. J Hepatobiliary Pancreat Surg. 2008;15:493–500. doi: 10.1007/s00534-007-1312-8. http://dx.doi.org/10.1007/s00534-007-1312-8. [DOI] [PubMed] [Google Scholar]
- 45.Mazzaferro V, Llovet JM, Miceli R, et al. Predicting survival after liver transplantation in patients with hepatocellular carcinoma beyond the Milan criteria: a retrospective, exploratory analysis. Lancet Oncol. 2009;10:35–43. doi: 10.1016/S1470-2045(08)70284-5. http://dx.doi.org/10.1016/S1470-2045(08)70284-5. [DOI] [PubMed] [Google Scholar]
- 46.Germani G, Pleguezuelo M, Gurusamy K, Meyer T, Isgro G, Burroughs AK. Clinical outcomes of radiofrequency ablation, percutaneous alcohol and acetic acid injection for hepatocellular carcinoma: a meta-analysis. J Hepatol. 2010;52:380–388. doi: 10.1016/j.jhep.2009.12.004. http://dx.doi.org/10.1016/j.jhep.2009.12.004. [DOI] [PubMed] [Google Scholar]
- 47.Lencioni R. Loco-regional treatment of hepatocellular carcinoma. Hepatology. 2010;52:762–773. doi: 10.1002/hep.23725. http://dx.doi.org/10.1002/hep.23725. [DOI] [PubMed] [Google Scholar]
- 48.Minami Y, Nishida N, Kudo M. Therapeutic response assessment of RFA for HCC: contrast-enhanced US, CT and MRI. World J Gastroenterol. 2014;20:4160–4166. doi: 10.3748/wjg.v20.i15.4160. http://dx.doi.org/10.3748/wjg.v20.i15.4160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Dong W, Zhang T, Wang ZG, Liu H. Clinical outcome of small hepatocellular carcinoma after different treatments: a meta-analysis. World J Gastroenterol. 2014;20:10174–10182. doi: 10.3748/wjg.v20.i29.10174. http://dx.doi.org/10.3748/wjg.v20.i29.10174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Cucchetti A, Piscaglia F, Cescon M, Ercolani G, Pinna AD. Systematic review of surgical resection vs. radiofrequency ablation for hepatocellular carcinoma. World J Gastroenterol. 2013;19:4106–4118. doi: 10.3748/wjg.v19.i26.4106. http://dx.doi.org/10.3748/wjg.v19.i26.4106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wang Y, Luo Q, Li Y, Deng S, Wei S, Li X. Radiofrequency ablation versus hepatic resection for small hepatocellular carcinomas: a meta-analysis of randomized and nonrandomized controlled trials. PLoS One. 2014;9:e84484. doi: 10.1371/journal.pone.0084484. http://dx.doi.org/10.1371/journal.pone.0084484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Jin YJ, Lee JW, Lee OH, et al. Transarterial chemoembolization versus surgery/radiofrequency ablation for recurrent hepatocellular carcinoma with or without microvascular invasion. J Gastroenterol Hepatol. 2014;29:1056–1064. doi: 10.1111/jgh.12507. http://dx.doi.org/10.1111/jgh.12507. [DOI] [PubMed] [Google Scholar]
- 53.Cheng AL, Kang YK, Chen Z, et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009;10:25–34. doi: 10.1016/S1470-2045(08)70285-7. http://dx.doi.org/10.1016/S1470-2045(08)70285-7. [DOI] [PubMed] [Google Scholar]
- 54.Cheng AL, Guan Z, Chen Z, et al. Efficacy and safety of sorafenib in patients with advanced hepatocellular carcinoma according to baseline status: subset analyses of the phase III Sorafenib Asia-Pacific trial. Eur J Cancer. 2012;48:1452–1465. doi: 10.1016/j.ejca.2011.12.006. http://dx.doi.org/10.1016/j.ejca.2011.12.006. [DOI] [PubMed] [Google Scholar]
- 55.Llovet JM, Ricci S, Mazzaferro V, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378–390. doi: 10.1056/NEJMoa0708857. http://dx.doi.org/10.1056/NEJMoa0708857. [DOI] [PubMed] [Google Scholar]