Abstract
It is essential that preventative vaccines for respiratory syncytial virus (RSV) elicit balanced T-cell responses. Immune responses dominated by type 2 T cells against RSV antigens are believed to cause exaggerated respiratory tract disease and may also contribute to unwanted inflammation in the airways that predisposes infants to wheeze through adolescence. Here we report on the construction and characterization of recombinant RSV (rRSV) strains with amino acids 151 to 221 or 178 to 219 of the attachment (G) glycoprotein deleted (rA2cpΔG150-222 or rA2cpΔG177-220, respectively). The central ectodomain was chosen for modification because a peptide spanning amino acids 149 to 200 of G protein has recently been shown to prime several strains of naïve inbred mice for polarized type 2 T-cell responses, and peripheral blood T cells from most human donors recognize epitopes within this region. Quantitative PCR demonstrated that synthesis of nascent rRSV genomes in human lung epithelial cell lines was similar to that for the parent virus (cp-RSV). Plaque assays further indicated that rRSV replication was not sensitive to 37°C, but pinpoint morphology was observed at 39°C. Both rRSV strains replicated in the respiratory tracts of BALB/c mice and elicited serum neutralization and anti-F-protein immunoglobulin G titers that were equivalent to those elicited by cp-RSV and contributed to a 3.9-log10-unit reduction in RSV A2 levels 4 days after challenge. Importantly, pulmonary eosinophilia was significantly diminished in BALB/c mice primed with native G protein and challenged with either rA2cpΔG150-222 or rA2cpΔG177-220. These findings are important for the development of attenuated RSV vaccines.
Respiratory syncytial virus (RSV) is a negative-strand, nonsegmented RNA virus and a member of the family Paramyxoviridae, genus Pneumovirus (7, 27). Respiratory tract disease caused by RSV imposes a significant burden on health care, and all age groups are infected. The most significant disease, however, occurs in young infants, aged adults, and patients with immunological abnormalities. It is estimated that lower respiratory tract (LRT) disease caused by RSV is responsible for 90% of bronchiolitis in infancy and 50% of all cases of pneumonia during the first 2 years of life. Thus, there is an immense need for vaccines against RSV. Both subunit and live-attenuated vaccine strategies have been followed to prevent LRT disease (35, 41). Unfortunately, neither tactic has thus far produced an acceptable vaccine. However, the recent advent of “reverse genetics” technology holds great promise for future vaccines (18, 39). With reverse genetics, recombinant RSV (rRSV) strains may be genetically engineered with defined mutations to ensure an attenuated phenotype or may include genes encoding cytokines to modify adaptive immune responses. One caveat, however, is that replication of rRSV in the airways may generate inflammatory responses that lead susceptible infants and toddlers to wheeze. RSV bronchiolitis is a well-documented major risk factor for wheeze up to the age of 13 years (38) and has even been suggested to set in motion immunological events that contribute to asthma (46).
The exact mechanisms whereby RSV infection brings about wheeze and asthma-like symptoms are not known. It is likely that both innate and adaptive immune responses are involved. Several reports have suggested that type 2 T-cell responses are dominant in human infants with LRT disease caused by RSV. Peripheral blood eosinophilia, RSV-specific immunoglobulin E (IgE) and IgG4, and increased secretion of interleukin-4 (IL-4) from peripheral blood mononuclear cells (PBMC) stimulated with an allergen or mitogen have been associated with acute bronchiolitis caused by RSV (4, 29, 42). Because type 2 T-cell responses and atopy are key factors in asthma (36), unbalanced T-cell responses to RSV antigens could contribute to harmful airway inflammation.
An antigen of primary interest in eliciting unbalanced T-cell responses is the attachment (G) protein. Findings from several laboratories have established that immunization with highly purified native or vaccinia virus-expressed recombinant G protein primed naïve BALB/c mice for pulmonary eosinophilia upon subsequent challenge with infectious RSV (17, 40). Eosinophilia was dependent on the presence of IL-5 and CD4+ T cells. In contrast, vaccination with vaccinia virus-expressed F protein (17, 40) or with natural F protein mixed with an appropriate adjuvant (22) did not prime for eosinophilia. Recently it was reported that PBMC from most human volunteers readily recognized epitopes located within the central ectodomain encompassed by amino acids 149 to 200 of G protein (20). A synthetic peptide composed of amino acids 149 to 200 and conjugated to a carrier primed several strains of inbred mice for pulmonary eosinophilia (24). The results presented here describe efforts to construct rRSV strains in which the region likely to induce unbalanced T-cell responses against G protein is deleted. By use of reverse genetics, rRSV strains were engineered with deletions of amino acids 151 to 221 (rA2cpΔG150-222) or 178 to 219 (rA2cpΔG177-220). We further characterized the mutants for attenuation, immunogenicity, and ability to recall type 2 T-cell responses in mice primed with native G protein.
MATERIALS AND METHODS
Cells and viruses.
A549 (ATCC CCL-185), HEp-2 (ATCC CCL-23), and Vero (ATCC CCL-81) cells (all from the American Type Culture Collection, Manassas, Va.) were maintained in culture as previously described (13). The viruses used in the present studies were RSV A2 (59), cp-RSV (8, 15, 33), cpts248/404 (9, 10, 58), rA2cpts248/404ΔSH (57), and rA2cpΔG213 (13).
Plasmid construction.
The rRSV strains rA2cpΔ150-222 and rA2cpΔ177-220 were constructed by using reverse genetics as previously described (13). Briefly, the RSV genome from strain cpts248/404 (9) was PCR amplified in three large fragments, Le-P, M-M2, and L, followed by ligation into plasmids. To remove the portion of the genome encoding amino acids 151 to 221 or 178 to 219 of the G protein, two DNA primers complementary to genomic sequences immediately flanking the nucleotides to be deleted were used to amplify the M-M2 plasmid (see Fig. 1). Each of these primers contained a BsaI restriction site. Extension from these primers on the M-M2 plasmid occurred in opposite directions such that the region of the G protein gene to be deleted was excluded from PCR amplification. Circularization of the PCR product occurred following digestion with BsaI. The inserts from the modified M-M2 plasmid were digested with the appropriate restriction enzymes and ligated into the previously generated cDNA clone (13). This approach resulted in a vector containing the full complement of RSV genes based on cp-RSV, with specific deletions in the G protein appropriate for this study (Fig. 1). This vector was used to transfect Vero cells and rescue infectious rRSV strains as previously described (13).
FIG. 1.
Construction of rRSV antigenomic cDNA with altered G protein. Plasmid pFL M-M2, containing genes encoding the M, SH, G, F, and M2 proteins of RSV, was genetically altered by PCR mutagenesis to exclude nucleotides corresponding to amino acids 151 to 221 or 178 to 219 of G protein from amplification. The resultant plasmids were then combined with other rRSV plasmids and transfected into Vero cells. The resulting rRSV strains were designated rA2cpΔG150-222 and rA2cpΔG177-220, respectively. CT, cytoplasmic tail; TM, transmembrane region; P, proline residues; C, cysteine residues; stalks with circles, potential O-linked carbohydrate acceptor sites; N, potential N-linked carbohydrate acceptor sites.
rRSV.
rRSV strains were purified from Vero cells over discontinuous sorbitol density gradients from cultures grown in complete Dulbecco's minimum essential medium (Gibco BRL, Grand Island, N.Y.) supplemented with 5% fetal bovine serum (HyClone, Logan, Utah), 2 mM l-glutamine (Gibco BRL), and 2% Pen-strep (Gibco BRL). In brief, supernatants were harvested when the cytopathic effect was at least 75% and were centrifuged at low speed (200 × g, 15 min, 4°C) to remove cellular debris. The clarified supernatant was added to a 50% PEG-NTE mixture (50% polyethylene glycol, 0.15 M NaCl, 0.05 M Tris, 1 nM EDTA) to yield a final concentration of 10% (vol/vol) PEG-NTE in the supernatant. After stirring for 2 h (4°C), the precipitate was pelleted (8,500 rpm, 30 min, 4°C) by using a Sorvall RC-5B Superspeed centrifuge with a GSA rotor. The resulting pellet was resuspended in 20% (wt/vol) sorbitol-NTE buffer and placed over the discontinuous sorbitol gradient. Purified virus was collected at the interface between 60 and 35% sorbitol-NTE and was stored at −70°C.
Genome sequence confirmation.
The consensus genome sequences of rescued viruses were confirmed by direct sequence analysis of reverse transcription-PCR (RT-PCR) products spanning the entire genome as previously described (13). Briefly, total RNA was extracted from amplified cell lysates with Trizol LS reagent (Invitrogen, Carlsbad, Calif.) and was used (1 μg) in the Prostar High Fidelity Single-Tube RT-PCR system (Stratagene, La Jolla, Calif.). The primer pairs were designed to amplify the viral genome in seven fragments of approximately 2 kb each. Control reactions that did not undergo reverse transcription and negative-control reactions with distilled H2O substituted for the RNA template were set up for each fragment. Amplification was performed in the GeneAmp 9700 (Applied Biosystems, Foster City, Calif.), and conditions were 48°C for 45 min; 95°C for 1 min; 40 cycles of 30 s at 94°C, 30 s at 58°C, and 6 min at 68°C; and a final extension step of 68°C for 7 min. The amplified fragments were purified by using the QIAquick PCR purification kit (QIAGEN, Valencia, Calif.), and cycle sequencing was performed on 50 to 100 ng of purified fragments by using the Big Dye Terminator v3.0 Ready Reaction Cycle Sequencing kit (Applied Biosystems). Unincorporated dyes were removed by using the DyeEx-96 kit (QIAGEN), and automated sequence analysis was carried out on the 3100 Genetic Analyzer (Applied Biosystems). Sequence data were aligned by using Sequencher (version 4.0.5; Gene Codes, Ann Arbor, Mich.).
Immunoblotting.
The protein concentrations of sorbitol density gradient-purified RSV strains rA2cpΔG150-222, rA2cpΔG177-220, cp-RSV, and wild-type A2 were determined by use of bicinchoninic acid (Pierce, Rockford, Ill.) according to the manufacturer's instructions. Then 5.0 μg of virus protein was admixed with Laemmli sample buffer (Bio-Rad Laboratories, Hercules, Calif.) containing 5% (vol/vol) β-mercaptoethanol (Sigma, St. Louis, Mo.), subjected to electrophoresis in sodium dodecyl sulfate (SDS)-12% polyacrylamide gels (Bio-Rad Laboratories), and transferred to nitrocellulose membranes (Bio-Rad Laboratories) for Western blot analysis. The immunoblots were incubated with monoclonal antibodies (MAbs) previously identified (13, 49) as reacting with regions of the G protein encompassed by amino acids 1 to 118 (131-2G), 174 to 193 (L9), or 215 to 298 (130-2G). The murine MAb K6-1, directed against the G protein, was also used in the studies. Reactivity was visualized by secondary incubation with horseradish peroxidase-conjugated goat anti-mouse IgG (Kirkegaard and Perry Laboratories, Gaithersburg, Md.), followed by incubation with 4-chloro-naphthol substrate (Sigma). Highly purified natural G protein (1 μg/lane) from the A2 strain of RSV served as a control.
qPCR.
RSV genome copy numbers in infected A549 monolayers were determined as previously described by quantitative PCR (qPCR) (13) using a DNA primer-probe set (Synthegen, LLC, Houston, Tex.) specific for the L gene of RSV. In brief, the sequences of the primers and probe were as follows: RSVAF forward primer, 5′-AGACAAGCTAAAATTACTAGCGAAATCA-3′; RSVAP FAM/TAMRA probe, 5′-TAGACTGGCAGTTACAGAGGTT-3′; and RSVARreverse primer, 5′-GTTGTGCACTTTTGGAGAATATTTTG-3′. The sequences were 100% conserved for all strains in the study. PCR cycling conditions were 50°C for 2 min, 95°C for 10 min, 40 cycles at 95°C for 15 s, and 60°C for 1 min. Equal loading was verified by using a TaqMan rRNA control reaction kit that amplified human 18S cDNA (Applied Biosystems). Variations between rRNA concentrations from different total cellular RNA isolations were in most instances less than 0.5%. PCR, fluorescence detection, and data analysis were performed on an ABI Prism 7700 sequence detector (Perkin-Elmer, Pittsburgh, Pa.).
Animal studies.
Female BALB/c and C57BL/6 mice (8 to 10 weeks old) were obtained, respectively, from Charles River Laboratories (Wilmington, Mass.) and Jackson Laboratories (Bar Harbor, Maine) and were housed in a facility accredited by the American Association for Accreditation of Laboratory Animal Care. Natural G glycoprotein was purified by immunoaffinity chromatography from Vero cells infected with the A2 strain of RSV (23). G protein was more than 90% pure as estimated by SDS-polyacrylamide gel electrophoresis (PAGE) and an antigen capture enzyme-linked immunosorbent assay (ELISA). Immunizations (1.0 μg per dose) with natural G protein prepared in phosphate-buffered saline (PBS) alone were intramuscular (0.1 ml). Additional control mice were injected with G protein admixed with CpG adjuvant (100 μg/dose; 5′-GCATGACGTTGAGCT-3′) as previously described (19). Intranasal (0.05 ml) immunizations by experimental infection or challenge with the A2 strain of RSV (at ∼106 PFU in either case) were performed under sedation (with ketamine and xylazine, 60 mg/kg of body weight; The Butler Co., Dublin, Ohio). Eosinophilia was assessed 7 days after challenge as previously described (23) following bronchoalveolar lavage and examination of at least 400 leukocytes in cytospin preparations of bronchoalveolar lavage fluids stained with Diff-Quik (Dade International, Miami, Fla.).
Plaque assays.
Infectious virus titers in culture supernatants or lungs after experimental infection or challenge were determined by a plaque assay using HEp-2 cell monolayers as previously described (22, 23). The sensitivities of mutant viruses to temperature were determined at 32, 37, 39, and 40°C (13). Pulmonary tissues were collected 4 and 7 days after the primary experimental infection or in efficacy studies 4 days after challenge.
Serum antibody determinations.
An end point ELISA and the plaque reduction neutralization test were used to ascertain geometric mean serum anti-F protein IgG and neutralization titers, respectively (22, 23). Neutralization titers were determined against the A2 strain of RSV in the presence or absence of 5% (vol/vol) guinea pig serum (BioWhittaker, Walkersville, Md.) as a source of complement. The neutralization titer was calculated as the reciprocal of the serum dilution that showed 60% reduction in the number of foci per well (relative to the number for the virus control).
Statistical analyses.
Significant differences (P < 0.05) were determined after log transformation by a Tukey-Kramer honestly significant difference multiple comparison or Student's t test using JMP statistical discovery software (SAS Institute Inc., Cary, N.C.). Data are expressed as geometric means ± 1 standard deviation. All data were confirmed in separate studies.
RESULTS
Construction of rRSV strains with altered G protein.
The recall responses of T cells from mice (24) and humans (20) against G protein appear to be directed primarily against epitopes within the ectodomain encompassed by amino acids 149 to 200. To diminish T-cell responses to G protein and improve safety, antigenomic cDNAs with deletions of nucleotides in this region were constructed (Fig. 1). From one cDNA, 123 nucleotides (positions 5221 to 5344) encoding 41 amino acids (residues 178 to 219) were deleted. From the second cDNA, 210 nucleotides (positions 5140 to 5350) encoding 70 amino acids (residues 151 to 221) were deleted. The rRSV strains were rescued as previously reported (13) and designated rA2cpΔG177-220 and rA2cpΔG150-222, respectively. The appropriate mutations for each rRSV strain were confirmed following consensus genomic sequencing of RT-PCR products spanning the entire genome. No additional alterations in the genome or the gene for the G protein were detected (data not shown).
Further confirmation that the engineered deletions were correct was provided by immunoblotting of sorbitol density gradient-purified rA2cpΔG177-220 and rA2cpΔG150-222 with MAbs reported (13, 49) to react with specific regions of G protein. Both rA2cpΔG177-220 and rA2cpΔG150-222 were visualized when probed with a MAb (130-2G) that reacted with an epitope within the C-terminal region spanned by amino acids 214 to 298 (Fig. 2D). As expected, the staining pattern also indicated that the G protein of rA2cpΔG150-222 migrated farther during SDS-PAGE than that of rA2cpΔG177-220 or cp-RSV. When probed with a MAb (L9) that binds the region encompassed by amino acids 174 to 193, G proteins from rA2cpΔG177-220 and rA2cpΔG150-222 were not visualized (Fig. 2C). Probing of HEp-2 cell monolayers with MAb 130-2G 3 days after infection with rA2cpΔG150-222 or rA2cpΔG177-220 resulted in positive staining (Table 1). In contrast, plaques were not stained following probing with MAb L9 (Table 1). Of interest were results obtained after probing with MAb 131-2G (Table 1 and Fig. 2A). When infected with rA2cpΔG150-222, HEp-2 cell monolayers did not react with 131-2G, and G protein was not visualized on immunoblots. Thus, deletion of amino acids 151 to 221 appeared to affect a conformational epitope in the region spanned by amino acids 1 to 118. It was also noteworthy that construction of the rRSV strains enabled identification of the epitope recognized by MAb K6-1. The results in Table 1 and the immunoblot shown in Fig. 2B demonstrated that the epitope recognized by MAb K6-1 is located in the region of the G protein spanned by amino acids 150 to 174. As expected, following infection with cp-RSV, G protein was visualized by all MAbs (Table 1 and Fig. 2). Following infection with rA2cpΔG213, plaques were not positively stained with MAb 130-2G (Table 1).
FIG. 2.
Western blot analysis of rRSV strains with altered G protein. rA2cpΔG150-222, rA2cpΔG177-220, and cp-RSV were purified from Vero cells over discontinuous sorbitol density gradients and compared to native G protein by immunoblotting using MAb K6-1 (B) or a MAb that reacted with the region of the G protein spanned by amino acids 1 to 118 (A), 174 to 193 (C), or 215 to 298 (D). Positions of molecular weight markers (in thousands) are given on the right.
TABLE 1.
MAb mapping of rA2cpΔG150-222 and rA2cpΔG177-220
| Virusa | Reactivity with a MAbb that binds the region spanned by amino acids:
|
|||
|---|---|---|---|---|
| 1-118 (131-2G) | 1-298 (K6-1) | 174-193 (L9) | 214-298 (130-2G) | |
| rA2cpΔG150-222 | Neg. | Neg. | Neg. | Pos. |
| rA2cpΔG177-220 | Pos. | Pos. | Neg. | Pos. |
| rA2cpΔG213 | Pos. | Pos. | Pos. | Neg. |
| cp-RSV | Pos. | Pos. | Pos. | Pos. |
HEp-2 cell monolayers were infected with the indicated virus. Three days postinfection, the monolayers were probed with an anti-G protein MAb.
The following MAbs were used to confirm genetic alterations of G protein: 131-2G (spanning amino acids 1 to 118), K6-1 (spanning amino acids 1 to 298), L9 (spanning amino acids 174 to 193), and 130-2G (spanning amino acids 214 to 298). Neg. and Pos., negative and positive staining, respectively, of HEp-2 cell monolayers infected with rRSV strains.
In vitro growth characteristics of rA2cpΔG177-220 and rA2cpΔG150-222.
The replication of the mutant viruses was characterized by qPCR and standard plaque assays using human lung epithelial cells (A549) cultured at 37°C. Table 2 shows results from a qPCR assay in which temporal increases in the RSV genome copy number following infection with rA2cpΔG177-220 or rA2cpΔG150-222 were contrasted with those for RSV strains cp-RSV (parent virus), rA2cpΔG213 (13), A2, and cpts248/404 (temperature sensitive) 14). The results indicated that the rate of genome synthesis following infection with rA2cpΔG177-220 or rA2cpΔG150-222 was slightly lower than that of the parent virus (cp-RSV) or RSV A2. After 72 h, the genome copy numbers of the rRSV strains were approximately 47,000 and 70,000, respectively, numbers two- and fourfold lower than the nearly 200,000 copies detected in A549 monolayers infected with cp-RSV. In comparison, only 17,300 (11-fold decrease) and 1,500 (123-fold decrease) copies of the attenuated rA2cpΔG213 (13) and temperature sensitive cpts248/404 genomes, respectively, were detected by qPCR.
TABLE 2.
Replication of rRSV strains with genetically altered G protein in A549 lung epithelial cells
| Groupa | Value for the indicated virus at the following time postinfection:
|
||||||||
|---|---|---|---|---|---|---|---|---|---|
| 24 h
|
48 h
|
72 h
|
|||||||
| Genome copy no. (103) | Fold Δb | 103 PFU/ml of culture medium | Genome copy no. (103) | Fold Δ | 103 PFU/ml of culture medium | Genome copy no. (103) | Fold Δ | 103 PFU/ml of culture medium | |
| rA2cpΔG150-222 | 14.2 | 1.2 | 23.0 | 46.1 | 2.0 | 40.5 | 70.0 | 2.1 | 115.0 |
| rA2cpΔG177-220 | 12.6 | 1.3 | 4.3 | 35.6 | 2.6 | 34.0 | 47.2 | 4.0 | 95.0 |
| rA2cpΔG213 | 3.2 | 5.1 | 0.005 | 12.4 | 7.3 | 0.2 | 17.3 | 11.0 | 2.3 |
| A2 | 23.0 | 0.7 | 25.0 | 101.9 | 0.9 | 360.0 | 183.5 | 1.0 | 2,700.0 |
| cp-RSV | 16.5 | 22.5 | 90.63 | 315.0 | 189.4 | 3,300.0 | |||
| cpts248/404 | 0.3 | 66.0 | NDc | 0.9 | 103.0 | 0.1 | 1.5 | 123.0 | 0.9 |
| Control | ND | ND | ND | ND | ND | ND | |||
A549 cells were infected with the indicated virus at a multiplicity of infection of 0.09.
Fold difference in genome copy number relative to that of cp-RSV.
ND, not detected.
To characterize the replication of rA2cpΔG177-220 and rA2cpΔG150-222 further, standard plaque assays were used to detect infectious virus in the culture supernatants (Table 2). Infectious virus titers recovered from A549 cells infected with rA2cpΔG177-220 or rA2cpΔG150-222 were similar to those of cp-RSV at 24 h postinfection. However, at 48 and 72 h postinfection, the titers were approximately 10-fold less than those of the cp-RSV and A2 strains of RSV. As previously described, the replication of strains rA2cpΔG213 (13) and cpts248/404 (14) was significantly restricted in vitro. Infectious virus titers were 1,000-fold lower those of cp-RSV or A2 (Table 2). Thus, the rate of replication of rA2cpΔG177-220 and rA2cpΔG150-222 in A549 cells at 37°C was less than that of parent virus, but not restricted to the level of rA2cpΔG213 or cpts248/404.
We next ascertained the sensitivities of rA2cpΔG177-220 and rA2cpΔG150-222 to temperature. HEp-2 cell monolayers were infected, and after 3 to 5 days of culture at 32, 37, 39, or 40°C, plaques were visualized by immunostaining for F protein (Fig. 3). When the plaque morphology from cultures infected with rA2cpΔG177-220 or rA2cpΔG150-222 was compared to that of cp-RSV cultures, little difference was noted. Indeed, pinpoint plaques were readily observed at 39°C. In monolayers infected with cpts248/404, plaques were observed at 32 but not 37°C, in agreement with the previously reported (14) shutoff temperature. Thus, deletion of amino acids 151 to 221 did not confer biologically relevant sensitivity to temperature.
FIG. 3.
Plaque morphologies of rRSV strains with altered G protein. A549 monolayers were infected with the indicated strain and cultured for 3 to 4 days at the temperature given. Plaques were visualized by immunostaining with a MAb reactive with F protein.
Immunogenicity and efficacy of rRSV vaccines.
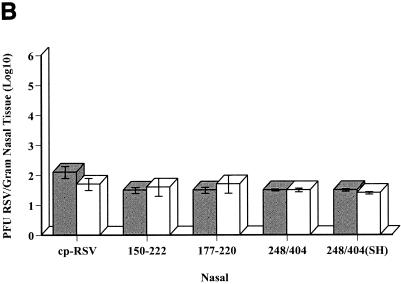
The capacity of the genetically altered viruses to replicate in the respiratory tract was examined after experimental infection of BALB/c mice. The results indicated that deletion of amino acids 151 to 221 did not lead to restricted replication in vivo. Virus titers in the lungs (Fig. 4A) and nasal tissues (Fig. 4B) 4 days after infection with rA2cpΔG177-220 or rA2cpΔG150-222 were not significantly less than titers of the parent virus. As previously reported (5, 14), replication of the cpts248/404 and rA2cpts248/404ΔSH viruses was severely limited in vivo. Because the G protein is a major protective antigen, several studies were also performed to ascertain the capacity of rA2cpΔG177-220 or rA2cpΔG150-222 to induce efficacious immune responses in BALB/c mice. Four weeks after primary infection with rA2cpΔG177-220 or rA2cpΔG150-222, noteworthy serum complement-assisted neutralization titers were observed (Table 3). The titers were comparable to those elicited following infection with cp-RSV and significantly greater than titers generated after infection with the temperature-sensitive cpts248/404 or rA2cpts248/404ΔSH strain. Genetic alteration of the G protein did not affect the induction of anti-F protein IgG titers. These also were significantly greater than titers generated after infection with temperature-sensitive strains and comparable to those of the parent virus (Table 3). Most importantly, the immune responses induced following infection were efficacious. Four days after challenge with the A2 strain of RSV, a 3.9-log10-unit reduction in the titer of infectious virus in the lungs was observed.
FIG. 4.
Replication of rRSV strains in the respiratory tracts of BALB/c mice. Naïve BALB/c mice were infected (∼1 × 106 PFU) with the indicated viruses. Lung (A) and nasal (B) tissues were collected 4 and 7 days postinfection for determination of the infectious virus titer (±1 standard deviation) by a plaque assay. The lower limit of detection for both data sets was approximately 1.5 log10 units. There were five mice per group.
TABLE 3.
Immune responses of BALB/c mice vaccinated with rA2cpΔ150-222 or rA2cpΔG177-220
| Virus | Log10 PFU of RSV/g of tissueb | Antibody titer (log10)a
|
||
|---|---|---|---|---|
| Anti-F protein IgG | Neutralizing
|
|||
| + C | − C | |||
| cp-RSV | <1.7 ± 0.03 | 4.6 ± 0.4 | 1.5 ± 0.1 | <1.0 |
| rA2cpΔG150-222 | <1.7 ± 0.04 | 5.3 ± 0.4d | 2.2 ± 0.4d | <1.3 ± 0.2 |
| rA2cpΔG177-220 | <1.7 ± 0.03 | 5.2 ± 0.4d | 2.1 ± 0.6d | <1.0 |
| cpts248/404 | 2.0 ± 0.7 | 3.8 ± 0.2 | 1.3 ± 0.3 | <1.0 |
| rA2cpts248/404ΔSH | <1.7 ± 0.1 | 4.0 ± 0.2 | 1.3 ± 0.1 | <1.0 |
| PBS | 5.6 ± 0.04c | <1.7 | <1.0 | <1.0 |
Titers are geometric means ± 1 standard deviation derived from serum samples collected 4 weeks after primary experimental infection. Neutralization titers were ascertained in the presence (+) or absence (−) of 5% serum as a source of complement (C). There were five mice per group.
Measured 4 days after challenge with the A2 strain of RSV. Numbers are geometric means ± 1 standard deviation. There were five mice per group.
P < 0.05 for comparison with all viruses.
P < 0.05 for comparison with cpts248/404 and rA2cpts248/404ΔSH.
We next estimated the potential of rA2cpΔG177-220 or rA2cpΔG150-222 to induce type 2 T-cell responses and pulmonary eosinophilia. To accomplish this, polarized type 2 T-cell responses were first induced in naïve BALB/c mice following immunization with native G protein prepared in PBS. Control mice were vaccinated with G protein admixed with a CpG adjuvant previously shown to increase type 1 T-cell responses (19). Thereafter the capacity of the rRSV strain to recall type 2 T-cell responses after challenge was determined. Figure 5A shows levels of pulmonary eosinophilia in mice primed with G protein and challenged 2 weeks afterwards with rA2cpΔG150-222 or cp-RSV. The data demonstrated that the level of eosinophilia was significantly reduced (from 54% ± 8% to 23% ± 3%) in the lungs relative to that with cp-RSV. Similar results were observed for mice vaccinated on weeks 0 and 2 with G protein and challenged with rRSV strains with altered G protein (Fig. 5B). As expected, pulmonary eosinophilia was not observed after challenge of mice immunized with G/CpG. Thus, genetic elimination of amino acids 151 to 221 or 178 to 219 removed epitopes important in the generation of T-cell responses that lead to pulmonary eosinophilia in BALB/c mice. Pulmonary eosinophilia was not affected in C57BL/6 mice primed with G protein and challenged with rA2cpΔG150-222.
FIG. 5.
Eosinophilia in the lungs of native-G-protein-sensitized mice after challenge with rRSV. BALB/c (H-2d) and C57BL/6 (H-2b) mice were injected intramuscularly on week 0 (A) or weeks 0 and 2 (B) with natural G protein (1 μg/dose) prepared in PBS alone. The mice were challenged with the indicated virus 2 weeks after the last injection, and levels of pulmonary eosinophilia (±1 standard deviation) were assessed 7 days thereafter. Eosinophilia levels were significantly (P < 0.05) reduced in BALB/c mice challenged with rRSV. Asterisk indicates control mice vaccinated with G protein admixed with CpG (100 μg/dose) and challenged with the cp-RSV (A) or A2 (B) strain of RSV. The lower limit of detection for both data sets was approximately1.5 log10 units. There were five mice per group.
DISCUSSION
Asthma is a chronic inflammatory disease of the airways characterized by inflammation, hyperreactive airways, bronchial obstruction, and recurrent episodes of wheezing. In severe cases asthma is also distinguished by significant airway remodeling (11). Asthma imposes a substantial burden on societies worldwide. In the United States alone, the incidence of asthma increased dramatically in the past 20 years, especially among children below the age of 5 years, where the rate increased approximately 160% (16). For many individuals, asthma becomes evident within the first few years of life. Although the precipitating events are not fully understood, both genetic (11, 28) and environmental factors are likely involved. Regarding environmental factors, there is increased interest in LRT disease caused by RSV (37). LRT disease in infancy caused by RSV places pediatric populations at increased risk for persistent wheezing and asthma-like symptoms until age 13 (38). Indeed, the increased incidence of hospitalizations related to RSV bronchiolitis during the past 2 decades is similar to the upsurge in asthma (45). Thus, a successful prophylactic vaccine for RSV would not only lessen disease and prevent hospitalizations caused by acute bronchiolitis but also significantly diminish wheezing illnesses (and possibly asthma) from infancy through adolescence.
Two challenges confront the successful development of RSV vaccines for naïve infants. The vaccine must be sufficiently immunogenic in the presence of maternal antibody to be efficacious, yet not predispose the recipient for immunopathology in the airways. Both subunit and attenuated RSV vaccine strategies are currently being pursued. For highly purified protein-based or vectored subunit vaccines, the putative attachment (G) and fusion (F) proteins are of primary interest. Both proteins are located in the envelope and are major protective antigens. Hence, immunization with combination subunit vaccines containing both antigens would likely generate more-efficacious immune responses (1, 21). It is critical, however, that subunit vaccines do not elicit adaptive immune responses dominated by type 2 T cells. The exacerbated disease observed in RSV-naïve human infants to whom formalin-inactivated vaccines were administered (31, 34) was associated with unbalanced type 2 T-cell responses (17, 32). Asthma is associated with type 2 T cells and atopy (36). The dominance of type 2 T cells in naïve rodents can be overcome through formulation of highly purified proteins with adjuvants that target toll-like receptors (19). However, for vectors such as vaccinia virus, type 2 T-cell responses against G protein remain dominant (40). Only alphavirus RNA replicons encoding F and G proteins elicited balanced T-cell responses following intranasal administration to naïve rodents (6). When the replicons were injected subcutaneously, however, pulmonary pathology was still observed. Thus, for subunit vaccines, special measures are required to ensure the generation of balanced T-cell responses.
Great care must also be exercised in designing safe and efficacious attenuated RSV vaccines. Finding the appropriate level of attenuation for naïve infants without sacrificing immunogenicity, and ensuring no reversion to a less attenuated phenotype, is not without complication. In addition, infants are prone to generate type 2 T-cell responses in the first few months of life, presumably because of the Th2 cytokine-enriched environment in utero (25). Type 2 T-cell responses have been observed in infants with LRT disease caused by RSV (3, 42-44). Elevated serum IgE levels and peripheral blood eosinophilia at the time of RSV infection have been further associated with asthma (56). Thus, immune responses following infection with an attenuated virus of limited replication could, in the susceptible recipient, induce unbalanced T-cell responses.
Presented here are our efforts to increase the safety profile of attenuated vaccines through construction of rRSV strains with deletions of 41 (residues 178 to 219) or 70 (residues 151 to 221) amino acids in the central ectodomain of G protein. These rRSV strains were designated rA2cpΔG177-220 and rA2cpΔG150-222, respectively. Our strategy was based on results from studies that indicated that PBMC from most adult human donors were readily activated upon stimulation with peptide antigens from the region of G protein spanned by amino acids 149 to 200 (20). It was further demonstrated that epitopes within the region primed several inbred strains of mice for pulmonary eosinophilia (24). At first, it was not clear what impact genetic alteration of this magnitude would have on attenuation, immunogenicity, and pulmonary eosinophilia. Studies demonstrated that an rRSV strain in which 26 amino acids containing the highly conserved cystine noose were deleted could be rescued without adverse effects on replication (52). Indeed, an rRSV strain with the G protein completely deleted was successfully rescued (51, 53). However, the complete absence of G protein severely restricted replication in vivo and thus limited the potential of the rRSV strain as a vaccine. Alternatively, efficacious immunity was observed (47) following immunization of BALB/c mice with vaccinia virus-expressed G protein in which the region shown to be responsible for eosinophilia was altered by a frameshift (50, 55). Following challenge with the Long strain of RSV, eosinophilia was not observed.
The results presented here extend these previous observations and demonstrate that an rRSV strain with 70 amino acids removed from the central ectodomain may be rescued and replicate without significant restriction. The rRSV strains were not sensitive to physiological temperature and replicated as well as cp-RSV in vivo. Positive reactivity with MAbs suggested that, in addition, deletion of the central ectodomain did not significantly alter the structure of the C-terminal one-third of the G protein. As vaccines, the rRSV strains were immunogenic and elicited efficacious immune responses. Most importantly, upon challenge of native-G-protein-primed BALB/c mice with rA2cpΔG177-220 or rA2cpΔG150-222, pulmonary eosinophilia was significantly diminished. Thus, the potential of the rRSV strains to elicit dominant type 2 responses was significantly lessened without apparent loss of immunogenicity.
The data presented here do not address the mechanisms whereby type 2 T cells dominate immune responses in human infants against RSV antigens. As noted, some human infants may be susceptible to this scenario because of a Th2 cytokine-rich environment in utero (25). However, it was recently suggested (2) that infection with RSV decreased the capacity of myeloid dendritic cells to induce type 1 T-cell responses. Thus, during acute LRT disease, reduced secretion of gamma interferon from RSV-infected myeloid dendritic cells could contribute to increased type 2 T-cell responses. Immune responses against G protein could also lead to wheezing by contributing to neurogenic inflammation, which plays an important role in the pathogenesis of asthma. RSV infection was shown to upregulate the expression of nerve growth factor and its receptors (26), and the presence of G protein was associated with the secretion of substance P, a tachykinin neuropeptide (54). It was also recently demonstrated that IL-13 alone was adequate for G protein-induced eosinophilia (30). Thus, through secretion of cytokines such as IL-13, type 2 T cells in the lungs against G protein could contribute directly to airway hyperresponsiveness, mucus hypersecretion, and inflammation (12, 60).
Replacement of wild-type genes with genes encoding genetically altered G proteins from both A and B strains of RSV will facilitate the development of subunit vaccines, as well as strategies based on attenuated strains of RSV. Importantly, the risk of inducing unbalanced T-cell responses in naïve populations will be inherently lower. However, it should be noted that pulmonary eosinophilia was observed after challenge of native-G-protein-primed C57BL/6 mice with rA2cpΔG150-222. In addition, following challenge with rA2cpΔG150-222, eosinophilia was not reduced to background levels in BALB/c mice primed with natural G protein. Thus, innate inflammatory events subsequent to virus replication in the airways might contribute to pulmonary eosinophilia. Alternatively, secondary epitopes outside the central ectodomain may yield type 2 T-cell responses against G protein that result in eosinophilia. Previous studies (24, 48, 55) have indicated that dominant T-cell epitopes in H-2b and H-2d mice are located in the central ectodomain of G protein. Whether unbalanced T-cell responses against secondary epitopes would arise in naïve recipients in the presence of attenuated virus replication, or following subunit vaccination, remains to be determined.
Acknowledgments
We acknowledge the technical assistance of K. Condello and thank J. H. Eldridge and S. A. Udem for critical reviews of the manuscript.
REFERENCES
- 1.Anderson, L. J., P. Bingham, and J. C. Hierholzer. 1988. Neutralization of respiratory syncytial virus by individual and mixtures of F and G protein monoclonal antibodies. J. Virol. 62:4232-4238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bartz, H., O. Turkel, S. Hoffjan, T. Rothoeft, A. Gonschorek, and U. Schauer. 2003. Respiratory syncytial virus decreases the capacity of myeloid dendritic cells to induce interferon-gamma in naive T cells. Immunology 109:49-57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bendelja, K., A. Gagro, A. Bae, R. Lokar-Kolbas, V. Krulovi-Hrei, V. Drazenovi, G. Mlinaric-Galinovi, and S. Rabati. 2000. Predominant type-2 response in infants with respiratory syncytial virus (RSV) infection demonstrated by cytokine flow cytometry. Clin. Exp. Immunol. 121:332-338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bui, R. H., G. A. Molinaro, J. D. Kettering, D. C. Heiner, D. T. Imagawa, and J. W. St Geme, Jr. 1987. Virus-specific IgE and IgG4 antibodies in serum of children infected with respiratory syncytial virus. J. Pediatr. 110:87-90. [DOI] [PubMed] [Google Scholar]
- 5.Bukreyev, A., S. Whitehead, B. Murphy, and P. Collins. 1997. Recombinant respiratory syncytial virus from which the entire SH gene has been deleted grows efficiently in cell culture and exhibits site-specific attenuation in the respiratory tract of the mouse. J. Virol. 71:8973-8982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chen, M., K.-F. Hu, B. Rozell, C. Orvell, B. Morein, and P. Liljestrom. 2002. Vaccination with recombinant alphavirus or immune-stimulating complex antigen against respiratory syncytial virus. J. Immunol. 169:3208-3216. [DOI] [PubMed] [Google Scholar]
- 7.Collins, P. L., Y. T. Huang, and G. W. Wertz. 1984. Identification of a tenth mRNA of respiratory syncytial virus and assignment of polypeptides to the 10 viral genes. J. Virol. 49:572-578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Connors, M., J. E. Crowe, Jr., C.-Y. Firestone, B. R. Murphy, and P. L. Collins. 1995. A cold-passaged, attenuated strain of human respiratory syncytial virus contains mutations in the F and L genes. Virology 208:478-484. [DOI] [PubMed] [Google Scholar]
- 9.Crowe, J. E., Jr., P. T. Bui, A. R. Davis, R. M. Chanock, and B. R. Murphy. 1994. A further attenuated derivative of a cold-passaged temperature-sensitive mutant of human respiratory syncytial virus retains immunogenicity and protective efficacy against wild-type challenge in seronegative chimpanzees. Vaccine 12:783-790. [DOI] [PubMed] [Google Scholar]
- 10.Crowe, J. E., Jr., C. Y. Firestone, S. S. Whitehead, P. L. Collins, and B. R. Murphy. 1996. Acquisition of the ts phenotype by a chemically mutagenized cold-passaged human respiratory syncytial virus vaccine candidate results from the acquisition of a single mutation in the polymerase (L) gene. Virus Genes 13:269-273. [DOI] [PubMed] [Google Scholar]
- 11.Davies, D. E., J. Wicks, R. M. Powell, S. M. Puddicombe, and S. T. Holgate. 2003. Airway remodeling in asthma: new insights. J. Allergy Clin. Immunol. 111:215-225. [DOI] [PubMed] [Google Scholar]
- 12.Elias, J. A., C. G. Lee, T. Zheng, Y. Shim, and Z. Zhu. 2003. Interleukin-13 and leukotrienes: an intersection of pathogenetic schema. Am. J. Respir. Cell Mol. Biol. 28:401-404. [DOI] [PubMed] [Google Scholar]
- 13.Elliott, M. B., K. S. Pryharski, R. Lerch, C. L. Parks, Q. Yu, C. K. Gupta, V. B. Randolph, N. A. LaPierre, K. M. Heers Dack, T. S. Laughlin, and G. E. Hancock. 2004. Recombinant respiratory syncytial viruses lacking the C-terminal third of the attachment (G) protein are immunogenic and attenuated in vivo and in vitro. J. Virol. 78:5773-5783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Firestone, C. Y., S. S. Whitehead, P. L. Collins, B. R. Murphy, and J. E. Crowe, Jr. 1996. Nucleotide sequence analysis of the respiratory syncytial virus subgroup A cold-passaged (cp) temperature sensitive (ts) cpts-248/404 live attenuated virus vaccine candidate. Virology 225:419-422. [DOI] [PubMed] [Google Scholar]
- 15.Friedewald, W. T., B. R. Forsyth, C. B. Smith, M. A. Gharpure, and R. M. Chanock. 1968. Low-temperature-grown RS virus in adult volunteers. JAMA 203:690-694. [PubMed] [Google Scholar]
- 16.Fuhlbrigge, A. L., R. J. Adams, T. W. Guilbert, E. Grant, P. Lozano, S. L. Janson, F. Martinez, K. B. Weiss, and S. T. Weiss. 2002. The burden of asthma in the United States: level and distribution are dependent on interpretation of the National Asthma Education and Prevention Program Guidelines. Am. J. Respir. Crit. Care Med. 166:1044-1049. [DOI] [PubMed] [Google Scholar]
- 17.Graham, B. S., T. R. Johnson, and R. S. Peebles. 2000. Immune-mediated disease pathogenesis in respiratory syncytial virus infection. Immunopharmacology 48:237-247. [DOI] [PubMed] [Google Scholar]
- 18.Grosfeld, H., M. Hill, and P. Collins. 1995. RNA replication by respiratory syncytial virus (RSV) is directed by the N, P, and L proteins; transcription also occurs under these conditions but requires RSV superinfection for efficient synthesis of full-length mRNA. J. Virol. 69:5677-5686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hancock, G. E., K. M. Heers, K. S. Pryharski, J. D. Smith, and L. Tiberio. 2003. Adjuvants recognized by toll-like receptors inhibit the induction of polarized type 2 T cell responses by natural attachment (G) protein of respiratory syncytial virus. Vaccine 21:4348-4358. [DOI] [PubMed] [Google Scholar]
- 20.Hancock, G. E., C. A. Scheuer, R. Sierzega, K. S. Pryharski, J. T. McBride, L. F. Watelet, P. W. Tebbey, and J. D. Smith. 2001. Adaptive immune responses of patients with asthma to the attachment (G) glycoprotein of respiratory synctial virus. J. Infect. Dis. 184:1589-1593. [DOI] [PubMed] [Google Scholar]
- 21.Hancock, G. E., J. D. Smith, and K. M. Heers. 2000. Serum neutralizing antibody titers of seropositive chimpanzees immunized with vaccines coformulated with natural fusion and attachment proteins of respiratory syncytial virus. J. Infect. Dis. 181:1768-1771. [DOI] [PubMed] [Google Scholar]
- 22.Hancock, G. E., D. J. Speelman, P. J. Frenchick, M. M. Mineo-Kuhn, R. B. Baggs, and D. J. Hahn. 1995. Formulation of the purified fusion protein of respiratory syncytial virus with the saponin QS-21 induces protective immune responses in Balb/c mice that are similar to those generated by experimental infection. Vaccine 13:391-400. [DOI] [PubMed] [Google Scholar]
- 23.Hancock, G. E., D. J. Speelman, K. Heers, E. Bortell, J. Smith, and C. Cosco. 1996. Generation of atypical pulmonary inflammatory responses in BALB/c mice after immunization with the native attachment (G) glycoprotein of respiratory syncytial virus. J. Virol. 70:7783-7791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hancock, G. E., P. W. Tebbey, C. A. Scheuer, K. S. Pryharski, K. M. Heers, and N. A. LaPierre. 2003. Immune responses to the nonglycosylated ectodomain of respiratory syncytial virus attachment glycoprotein mediate pulmonary eosinophilia in inbred strains of mice with different MHC haplotypes. J. Med. Virol. 70:301-308. [DOI] [PubMed] [Google Scholar]
- 25.Holt, P. G. 2000. Key factors in the development of asthma: atopy. Am. J. Respir. Crit. Care Med. 161:172S-175S. [DOI] [PubMed] [Google Scholar]
- 26.Hu, C., K. Wedde-Beer, A. Auais, M. M. Rodriguez, and G. Piedimonte. 2002. Nerve growth factor and nerve growth factor receptors in respiratory syncytial virus-infected lungs. Am. J. Physiol. Lung Cell Mol. Physiol. 283:L494-L502. [DOI] [PubMed] [Google Scholar]
- 27.Huang, Y. T., and G. W. Wertz. 1983. Respiratory syncytial virus mRNA coding assignments. J. Virol. 46:667-672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hull, J., A. Thomson, and D. Kwiatkowski. 2000. Association of respiratory syncytial virus bronchiolitis with the interleukin 8 gene region in UK families. Thorax 55:1023-1027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Jackson, M., and R. Scott. 1996. Different patterns of cytokine induction in cultures of respiratory syncytial (RS) virus-specific human TH cell lines following stimulation with RS virus and RS virus proteins. J. Med. Virol. 49:161-169. [DOI] [PubMed] [Google Scholar]
- 30.Johnson, T. R., R. A. Parker, J. E. Johnson, and B. S. Graham. 2003. IL-13 is sufficient for respiratory syncytial virus G glycoprotein-induced eosinophilia after respiratory syncytial virus challenge. J. Immunol. 170:2037-2045. [DOI] [PubMed] [Google Scholar]
- 31.Kapikian, A. Z., R. H. Mitchell, R. M. Chanock, R. A. Shvedoff, and C. E. Stewart. 1969. An epidemiologic study of altered clinical reactivity to respiratory syncytial (RS) virus infection in children previously vaccinated with an inactivated RS virus vaccine. Am. J. Epidemiol. 89:405-421. [DOI] [PubMed] [Google Scholar]
- 32.Kim, H., S. Leikin, J. Arrobio, C. Brandt, R. Chanock, and R. Parrott. 1976. Cell-mediated immunity to respiratory syncytial virus induced by inactivated vaccine or by infection. Pediatr. Res. 10:75-78. [DOI] [PubMed] [Google Scholar]
- 33.Kim, H. W., J. O. Arrobio, G. Pyles, C. D. Brandt, E. Camargo, R. M. Chanock, and R. H. Parrott. 1971. Clinical and immunological response of infants and children to administration of low-temperature adapted respiratory syncytial virus. Pediatrics 48:745-755. [PubMed] [Google Scholar]
- 34.Kim, H. W., J. G. Canchola, C. D. Brandt, G. Pyles, R. M. Chanock, K. Jensen, and R. H. Parrott. 1969. Respiratory syncytial virus disease in infants despite prior administration of antigenic inactivated vaccine. Am. J. Epidemiol. 89:422-434. [DOI] [PubMed] [Google Scholar]
- 35.Kneyber, M. C., and J. L. Kimpen. 2002. Current concepts on active immunization against respiratory syncytial virus for infants and young children. Pediatr. Infect. Dis. J. 21:685-696. [DOI] [PubMed] [Google Scholar]
- 36.Larche, M., D. S. Robinson, and A. B. Kay. 2003. The role of T lymphocytes in the pathogenesis of asthma. J. Allergy Clin. Immunol. 111:450-463. [DOI] [PubMed] [Google Scholar]
- 37.Lemanske, R. F. 2002. The Childhood Origins of Asthma (COAST) study. Pediatr. Allergy Immunol. 13:38-43. [DOI] [PubMed] [Google Scholar]
- 38.Martinez, F. D. 2003. Respiratory syncytial virus bronchiolitis and the pathogenesis of childhood asthma. Pediatr. Infect. Dis. J. 22:S76-S82. [DOI] [PubMed] [Google Scholar]
- 39.Murphy, B. R., and P. L. Collins. 2002. Live-attenuated virus vaccines for respiratory syncytial and parainfluenza viruses: applications of reverse genetics. J. Clin. Investig. 110:21-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Openshaw, P. J., S. L. Clarke, and F. M. Record. 1992. Pulmonary eosinophilic response to respiratory syncytial virus infection in mice sensitized to the major surface glycoprotein G. Int. Immunol. 4:493-500. [DOI] [PubMed] [Google Scholar]
- 41.Piedra, P. A. 2003. Clinical experience with respiratory syncytial virus vaccines. Pediatr. Infect. Dis. J. 22:S94-S99. [DOI] [PubMed] [Google Scholar]
- 42.Rabatic, S., A. Gagro, R. Lokar-Kolbas, V. Krsulovic-Hresic, Z. Vrtar, T. Popow-Kraupp, V. Drazenovic, and G. Mlinaric-Galinovic. 1997. Increase in CD23+ B cells in infants with bronchiolitis is accompanied by appearance of IgE and IgG4 antibodies specific for respiratory syncytial virus. J. Infect. Dis. 175:32-37. [DOI] [PubMed] [Google Scholar]
- 43.Renzi, P. M., J. P. Turgeon, J. P. Yang, S. P. Drblik, J. E. Marcotte, L. Pedneault, and S. Spier. 1997. Cellular immunity is activated and a TH-2 response is associated with early wheezing in infants after bronchiolitis. J. Pediatr. 130:584-593. [DOI] [PubMed] [Google Scholar]
- 44.Roman, M., W. J. Calhoun, K. L. Hinton, L. F. Avendano, V. Simon, A. M. Escobar, A. Gaggero, and P. V. Diaz. 1997. Respiratory syncytial virus infection in infants is associated with predominant Th-2-like response. Am. J. Respir. Crit. Care Med. 156:190-195. [DOI] [PubMed] [Google Scholar]
- 45.Shay, D. K., R. C. Holman, R. D. Newman, L. L. Liu, J. W. Stout, and L. J. Anderson. 1999. Bronchiolitis-associated hospitalizations among US children, 1980-1996. JAMA 282:1440-1446. [DOI] [PubMed] [Google Scholar]
- 46.Sigurs, N. 2002. A cohort of children hospitalised with acute RSV bronchiolitis: impact on later respiratory disease. Paediatr. Respir. Rev. 3:177-183. [DOI] [PubMed] [Google Scholar]
- 47.Sparer, T. E., S. Matthews, T. Hussell, A. J. Rae, B. Garcia-Barreno, J. A. Melero, and P. J. Openshaw. 1998. Eliminating a region of respiratory syncytial virus attachment protein allows induction of protective immunity without vaccine-enhanced lung eosinophilia. J. Exp. Med. 187:1921-1926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Srikiatkhachorn, A., W. Chang, and T. J. Braciale. 1999. Induction of Th-1 and Th-2 responses by respiratory syncytial virus attachment glycoprotein is epitope and major histocompatibility complex independent. J. Virol. 73:6590-6597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sullender, W. 1995. Antigenic analysis of chimeric and truncated G proteins of respiratory syncytial virus. Virology 209:70-79. [DOI] [PubMed] [Google Scholar]
- 50.Tebbey, P. W., M. Hagen, and G. E. Hancock. 1998. Atypical pulmonary eosinophilia is mediated by a specific amino acid sequence of the attachment (G) protein of respiratory syncytial virus. J. Exp. Med. 188:1967-1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Techaarpornkul, S., N. Barretto, and M. E. Peeples. 2001. Functional analysis of recombinant respiratory syncytial virus deletion mutants lacking the small hydrophobic and/or attachment glycoprotein gene. J. Virol. 75:6825-6834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Teng, M. N., and P. L. Collins. 2002. The central conserved cystine noose of the attachment G protein of human respiratory syncytial virus is not required for efficient viral infection in vitro or in vivo. J. Virol. 76:6164-6171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Teng, M. N., S. S. Whitehead, and P. L. Collins. 2001. Contribution of the respiratory syncytial virus G glycoprotein and its secreted and membrane-bound forms to virus replication in vitro and in vivo. Virology 289:283-296. [DOI] [PubMed] [Google Scholar]
- 54.Tripp, R. A., A. Dakhama, L. P. Jones, A. Barskey, E. W. Gelfand, and L. J. Anderson. 2003. The G glycoprotein of respiratory syncytial virus depresses respiratory rates through the CX3C motif and substance P. J. Virol. 77:6580-6584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Varga, S. M., E. L. Wissinger, and T. J. Braciale. 2000. The attachment (G) glycoprotein of respiratory syncytial virus contains a single immunodominant epitope that elicits both Th1 and Th2 CD4+ T cell responses. J. Immunol. 165:6487-6495. [DOI] [PubMed] [Google Scholar]
- 56.Welliver, R. C. 2003. Respiratory syncytial virus and other respiratory viruses. Pediatr. Infect. Dis. J. 22:S6-S12. [DOI] [PubMed] [Google Scholar]
- 57.Whitehead, S. S., A. Bukreyev, M. N. Teng, C.-Y. Firestone, M. St. Claire, W. R. Elkins, P. L. Collins, and B. R. Murphy. 1999. Recombinant respiratory syncytial virus bearing a deletion of either the NS2 or SH gene is attenuated in chimpanzees. J. Virol. 73:3438-3442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Whitehead, S. S., C.-Y. Firestone, P. L. Collins, and B. R. Murphy. 1998. A single nucleotide substitution in the transcription start signal of the M2 gene of respiratory syncytial virus vaccine candidate cpts248/404 is the major determinant of the temperature-sensitive and attenuation phenotypes. Virology 247:232-239. [DOI] [PubMed] [Google Scholar]
- 59.Wright, P. F., M. A. Gharpure, D. S. Hodes, and R. M. Chanock. 1973. Genetic studies of respiratory syncytial virus temperature-sensitive mutants. Arch. Gesamte Virusforsch. 41:238-247. [DOI] [PubMed] [Google Scholar]
- 60.Wynn, T. A. 2003. IL-13 effector functions. Annu. Rev. Immunol. 21:425-456. [DOI] [PubMed] [Google Scholar]