Abstract
Pluripotent stem cells only exist in a narrow window during early embryonic development, whereas multipotent stem cells are abundant throughout embryonic development and are retainedin various adult tissues and organs. While pluripotent stem cell lines have been established from several species, including mouse, rat, and human, it is still challenging to establish stable multipotent stem cell lines from embryonic or adult tissues. Based on current knowledge, we anticipate that by manipulating extrinsic and intrinsic signaling pathways, most if not all types of stem cells can be maintained in a long-term culture. In this article, we summarize current culture conditions established for the long-term maintenance of authentic pluripotent and multipotent stem cells and the signaling pathways involved. We also discuss the general principles of stem cell maintenance and propose several strategies on the establishment of novel stem cell lines through manipulation of signaling pathways. [BMB Reports 2015; 48(12): 668-676]
Keywords: Adult stem cells, Embryonic stem cell self-renewal, Neural stem cells, Stem cells, Wnt/β-catenin pathway
INTRODUCTION
All stem cells share two key properties: self-renewal and differentiation. The successful derivation of mouse embryonic stem cells (ESCs) in 1981 indicates that, under proper culture conditions, stem cells are capable of long-term self-renewal while retaining the ability to differentiate into multiple types of cells (1, 2). By using various culture conditions, stable pluripotent stem cell lines have been established from several species including mouse, rat, and human (3-9). However, the generation and long-term propagation of authentic multipotent stem cells has been hampered by the lack of well-established methods for maintenance such as those used for mouse ESC culture. We anticipate that successful generation and propagation of stable stem cell lines will require development of culture conditions that captures the specific stem cell state and facilitates their long-term self-renewal. The quantity of stem cells that can be isolated from embryonic or adult tissues is very limited; therefore, it is necessary to develop in vitro culture conditions to expand stem cells so that we can use these stem cells for a variety of biological studies, disease modeling, and stem cell-based therapy.
The major signaling pathways involved in embryonic development are likely also important in regulating stem cell self-renewal, as suggested by numerous studies on the molecular mechanisms of stem cell maintenance. Evidence from the current culture conditions developed for the maintenance of authentic stem cell lines suggests that manipulation of only a few signaling pathways may be sufficient to keep the stem cells at an undifferentiated state (10). In this article, we review the key signaling pathways related to the maintenance of different stem cell lines. We also discuss the general principles for stem cell maintenance and propose several strategies on how to develop culture conditions for the long-term maintenance of authentic stem cell lines.
CRITERIA FOR AUTHENTIC STEM CELL LINES
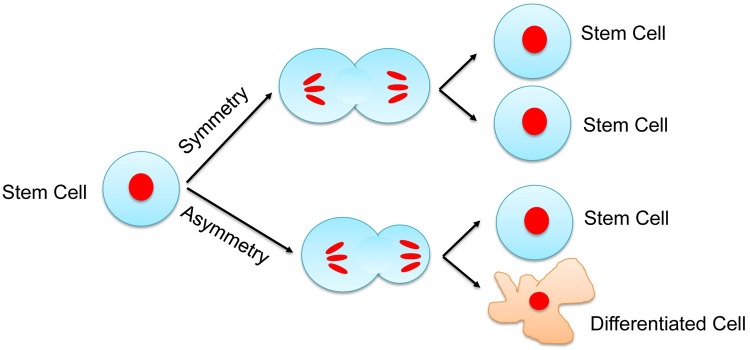
Stem cells undergo either symmetric or asymmetric division. When a stem cell undergoes a symmetric division, it produces two daughter cells that are identical to their mother. In asymmetric division, a stem cell divides to generate one daughter cell that is identical to the mother cell and another daughter cell with more restricted potential (Fig. 1). It is generally believed that most, if not all, of stem cells that reside in the body undergo asymmetric division to maintain tissue homeostasis. In this review, we focus on stem cell maintenance in vitro. In theory, long-term maintenance of stem cells in culture requires them to undergo symmetric division, because the number of stem cells will not increase if they divide asymmetrically. We propose that authentic stem cell lines must meet the first criterion: the majority of stem cells in the culture must undergo symmetric cell division.
Fig. 1. Symmetric and asymmetric stem cell division.
The second criterion for authentic stem cell lines is that they must be able to self-renew in long-term culture while retaining the same identity and developmental potential as the stem cells of origin in vivo. The identity of the stem cells can be determined by single cell RNA seq and genome-wide epigenetic analysis. Genetic and epigenetic patterns of cultured stem cells should be the same or similar to their in vivo counterparts. For example, in a mammalian female embryo, preimplantation inner cell mass (ICM) cells carry two active X chromosomes (11-13). This epigenetic signature of ground state pluripotency is shared with rodent ESCs and has become a criterion for the development of the naïve human ESC culture condition (5, 6, 14-19). Authentic stem cell lines should also retain the ability to differentiate into different progenies from their tissue of origin even after an extended period of expansion in vitro.
The third criterion is that these stem cells should remain karyotypically normal. The genomic stability during stem cell maintenance is critical, especially for the application of stem cell therapy. Stem cells should retain a normal karyotype over long-term passage to avoid culture-induced genomic alterations and transformation.
Although numerous stem cell lines from different tissue origins have been reported, few can be considered authentic stem cell lines that meet all three criteria. Below we list these established authentic stem cell lines (also see Table 1) and discuss their culture conditions and the major signaling pathways involved in their self-renewal.
Table 1. Current well-established stem cell lines and their culture conditions.
| Stem cell lines | Source of origin | Key components | Key signaling pathways | Species tested | Ref |
|---|---|---|---|---|---|
|
| |||||
| Pluripotent | |||||
| ESC | ICM | LIF + serum/BMP4 | JAK/STAT3, SMAD | Mouse | 3 |
| ICM | CHIR + PD03 | Wnt/β-catenin, MAPK | Mouse, rat | 4,5,6 | |
| EG cells | PGC | LIF + FGF2 + SCF | JAK/STAT3, FGF | Mouse | 27-30 |
| PGC | LIF + FGF2 + Forskolin | JAK/STAT3, FGF | Human | 25,26 | |
| PGC | LIF + CHIR + PD03 | JAK/STAT3, Wnt/β-catenin, MAPK | Mouse, rat | 31 | |
| EpiSC | Epiblast | Activin + FGF2 | TGF-β/SMAD, FGF | Mouse, human | 7,8 |
| Epiblast | CHIR + IWR-1 | Wnt/β-catenin | Mouse, human | 22 | |
| SSC | Testis | GDNF + FGF2 + LIF | PI3K/AKT, MAPK | Mouse, rat | 33 |
| Extraembryonic | |||||
| TS | Trophectoderm | FGF4 + heparin + Activin/Nodal | FGF/ERK | Mouse | 23 |
| XEN | Primitive endoderm | FGF | FGF/ERK | Mouse | 24 |
| Adult tissue | |||||
| NSC | Primitive neural ectoderm | LIF + CHIR | JAK/STAT, Wnt/β-catenin | Mouse, rat, human | 35-37 |
| Late embryonic or adult brain | FGF2 + EGF | MAPK, PI3K | Mouse, human | 38,39 | |
| Intestinal stem cells | Intestine | EGF + Noggin + R-spondin | Wnt | Mouse, human | 34 |
| Liver stem cells | Liver | EGF + Noggin + R-spondin + Wnt-3A + FGF10 + HGF + Nicotinamide | Wnt | Mouse, human | 44 |
ESCs
ESCs are derived from the ICM of the pre-implantation blastocyst. The ICM only exists during a short period of time in development. The derivation of ESCs proves that such a transient cell stage can probably be retained indefinitely in vitro given a proper culture condition. ESCs possess the capacity to become any type of cell in the body, and therefore represent a powerful tool for regenerative medicine, human disease modeling, and understanding biological development. Although ESCs have reportedly been derived from several species, including humans, only mouse and rat ESCs have been confirmed to be true ESCs through the gold-standard germline transmission test (1, 2, 5, 6). The study of rodent ESCs over the past three decades has provided a wealth of information indicating that these rodent ESCs meet the three criteria and therefore can be considered authentic stem cell lines. Genome-wide transcriptome analysis has further confirmed that rodent ESCs exhibit transcriptional similarities to the ICM cells (20).
Mouse ESC self-renewal is normally mediated by leukemia inhibitory factor (LIF)/signal transducer and activator of transcription 3 (STAT3) signaling (21). Alternatively, as we demonstrated, mouse ESC self-renewal can also be maintained if glycogen synthase kinase 3 (GSK3) and mitogen-activated protein kinase kinase (MEK) are simultaneously suppressed by addition of two small molecule inhibitors (2i), CHIR99021 and PD0325901 (4). It is also possible to derive and maintain rat ESCs using the 2i condition (5, 6).
Epiblast stem cells (EpiSCs)
EpiSCs are pluripotent stem cells derived from post-implantation epiblasts (7, 8). EpiSCs express core pluripotency markers Oct4, Sox2, and Nanog, and are able to differentiate into all three germ layers. However, EpiSCs are not competent to contribute to chimera formation and therefore are developmentally and functionally distinct from ESCs. Long-term self-renewal of mouse EpiSCs can be maintained in medium supplemented with fibroblast growth factor 2 (FGF2) and/or Activin A. Recently, we demonstrated that a combination of two small molecule inhibitors, CHIR99021 and IWR1, also maintains EpiSC self-renewal (22). IWR1, a tankyrase inhibitor, negatively regulates Wnt/β-catenin signaling through stabilization of Axin. CHIR99021 and IWR1 promote EpiSC self-renewal through stabilization of β-catenin and retention of β-catenin in the cytoplasm (22). Human ESCs are routinely cultured in medium supplemented with FGF2/Activin A. These human ESCs share defining features with mouse EpiSCs (7). Global gene expression profiling and epigenetic analysis have indicated that mouse EpiSCs and human ESCs are closely related to epiblast cells in the postimplantation embryos (7).
Extraembryonic stem cell lines
Two types of extraembryonic stem cell lines have been established so far. One is trophoblast stem (TS) cells derived from the trophectoderm lineage (23); another is extraembryonic endoderm stem (XEN) cells derived from primitive endoderm (24). TS cells are maintained in vitro in the presence of FGF4 and heparin while retaining the ability to differentiate into multiple cell types of the placenta. XEN cells can be established and continuously passaged using the same TS cell culture condition or medium containing serum. TS and XEN cells express markers typical of trophectoderm and extraembryonic endoderm derivatives, respectively. The identities of cultured TS and XEN cells are further confirmed by the chimera formation assay. Chimeras generated by injection of TS or XEN cells into preimplantation embryos show that the injected TS and XEN cells exclusively contribute to the trophoblastic component of the placenta and the extraembryonic endoderm cell types, respectively (23, 24).
Embryonic germ (EG) cells
Stable EG cell lines have been established from primordial germ cells (PGCs) isolated from species such as human, mouse, and rat (25-31). EG cells are very different from PGCs, their in vivo source of origin, in that EG cells are pluripotent whereas PGCs are unipotent and differentiate into only sperm or oocytes. The conversion of unipotent PGCs to pluripotent EG cells is considered a reprogramming event. Mouse and rat EG cells can be derived and maintained in 2i. Other factors that promote EG cell self-renewal include LIF, stem cell factor, and FGF2. When injected into blastocysts, mouse and rat EG cells maintained in 2i have been shown to contribute to the formation of chimeras as well as transmit through germline.
Spermatogonial stem cells (SSCs)
SSCs are rare, representing only 0.03% of all germ cells in rodent testes. Like other adult stem cells, SSCs maintain tissue homeostasis by undergoing both self-renewal and differentiation. In vivo, the niche factor glial cell line-derived neurotrophic factor (GDNF) plays a pivotal role in regulating SSC self-renewal (32). GDNF is also important for the long-term expansion of SSCs in vitro, along with the other SSC self-renewal factors epidermal growth factor (EGF), FGF2, and LIF (33). Through manipulation of these self-renewal signaling pathways, a uniform SSC population with similar developmental potential can be expanded in a culture dish. Under these culture conditions, differentiated cells are not favored for proliferation and usually will be selected out through passages. The extrinsic modulation of signaling pathways combined with the cell-cell interaction between undifferentiated SSCs is sufficient to maintain the stem cell identity in vitro.
Intestinal stem cells
In contrast to stem cells from embryonic tissues, most adult stem cells reside in a quiescent state but readily exit quiescence and undergo self-renewal and differentiation in response to stress, injury, and other environmental stimuli. In vivo, adult stem cells often divide asymmetrically to generate two sister cells with one retaining the same identity as the mother cell and the other becoming a more differentiated progeny. Hans Clever’s group developed an organoid culture system for the long-term maintenance of intestinal stem cells in vitro (34). In this method, Lgr5+ stem cells are mixed with laminin-rich Matrigel along with growth factors including R-spondin 1, EGF, and Noggin. In response to the culture condition, Lgr5+ intestinal stem cells generate both Lgr5+ cells and differentiated intestinal cells. The undifferentiated and differentiated cells support each other’s growth and form crypt-villus structures. Since the Lgr5+ cell population expands rapidly, it is likely that the majority of the Lgr5+ cells within the organoids undergo symmetric cell division to produce more stem cells. The organoid culture system could potentially be applied to the maintenance of adult stem cells from other tissues.
Neural stem cells (NSCs)
Developmental studies have defined at least two stages of NSCs: primitive and definitive NSCs. Primitive NSCs resemble the earliest stage of neural cells arising during development and can be isolated from the neuroectoderm of the E7.5 mouse embryo (35). These primitive NSCs are responsive to LIF, but not FGF2/EGF for self-renewal (35-37). Definitive NSCs, which are more committed to a neural identity than primitive NSCs, first appear in the anterior neural plate of the E8.5 mouse embryo and persist throughout life. These definitive NSCs can be transiently maintained in the presence of FGF2 and EGF (38, 39).
Other putative stem cell lines
It has been reported that dual inhibition of GSK3 and mammalian target of rapamycin (mTOR) promotes hematopoietic stem cell (HSC) self-renewal (40). Several other culture conditions have been developed for the expansion of HSCs. However, in all cases, HSCs were only maintained for a short period time after which they underwent differentiation. Although muscle stem cells can be expanded in vitro, their capacity to regenerate damaged muscles is greatly diminished compared to freshly isolated muscle stem cells (41, 42). Recently, through mimicry of the endogenous microenvironment, four cytokines secreted by T cells (IL-1α, IL-13, TNF-α, and IFN-γ) have been determined to promote long-term expansion of muscle stem cells (43). In contrast to the conventional method, muscle stem cells maintained in medium supplemented with these four cytokines retain the capacity to effectively regenerate muscle after transplantation. The expansion of muscle stem cells without apparent loss of their differentiation potential has been an important step towards the derivation of authentic muscle stem cells. However, more systematic characterization is needed to validate the identity of these cultured muscle stem cell lines.
UNANSWERED QUESTIONS IN STEM CELL BIOLOGY
In the three decades since the first derivation of mouse ESCs, significant progress has been made in understanding the molecular mechanisms underlying stem cell self-renewal and differentiation. Nonetheless, several key questions in stem cell biology remain unanswered. Is it possible to establish true ESCs from species other than mice and rats? So far, germline competent ESC lines have been established only from mice and rats. We and many other groups have tried but failed to derive true ESC lines from different species including pigs, rabbits, cows, chicken, and zebrafishes. Is the mechanism of ESC self-renewal shared or distinct among different species? Is it possible to maintain tissue-specific stem cells in culture indefinitely in a similar manner to mouse and rat ESCs? The majority of tissue-specific stem cell types cannot be maintained under current available culture conditions. Some tissue-specific stem cells, such as NSCs, intestinal stem cells, and skin stem cells, can be expanded in culture for an extended period of time. Whether these expanded stem cells retain the same identity as the original stem cells remains unknown.
HYPOTHESES AND MODELS ON STEM CELL MAINTENACE
To help develop strategies for addressing the above questions, we propose some new hypotheses and working models on stem cell maintenance. Our first hypothesis is that the fundamental mechanism underlying the maintenance of the same type of stem cells is likely conserved among different species. Stem cells from the same tissue origin but different species are likely to have differences in terms of their requirements for self-renewal and their characteristics. However, these differences might be subtle and are attributable to the variations in the response of the stem cells to specific microenvironments and stimuli. We came up this hypothesis based on the following evidence: 1. Both mouse and rat ESCs can be derived and maintained in the same 2i condition. More recently, naïve state human ESCs resembling mouse ESCs were generated using 2i-containing media. 2. Primed state pluripotent stem cells from mouse, rat, and human can all be maintained in the same FGF2/Activin A or CHIR99021/IWR1 condition (7, 8, 22). 3. NSCs from different species can be derived and maintained in similar culture conditions. This is also true for other types of tissue-specific stem cells.
Our second hypothesis is that by manipulating a few signaling pathways, most, if not all, pluripotent and tissue-specific stem cells can be maintained in culture. Signaling pathways have evolved into complex networks of interactions during millions of years’ evolution. The development of a single zygote into a whole organism involves proliferation, differentiation, and patterning of a wide variety of cells and tissues. Surprisingly, the whole development process is controlled mainly by seven major signaling pathways including Wnt, receptor tyrosine kinase, Notch, Hedgehog, transforming growth factor-β (TGF-β), JAK/STAT, and nuclear receptor pathways (45). In retrospect, we found that most of the established authentic stem cell lines can be maintained in culture by manipulating just two of the seven pathways. We anticipate that this is also largely true for the maintenance of other stem cell lines that have yet to be established.
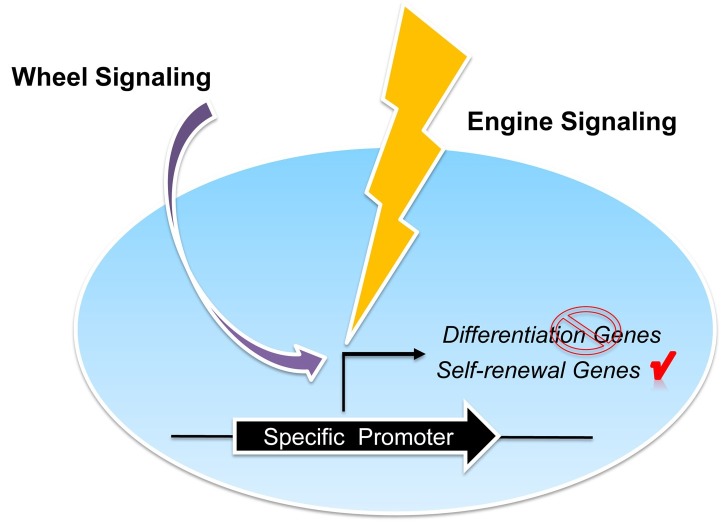
There are two key elements for the maintenance of stem cells: proliferation and prevention of differentiation. We propose an ‘Engine-and-Wheel’ model to explain stem cell maintenance in vitro. The engine provides the motion of the car while the steering wheel controls the direction of that motion. We predict that the maintenance of most if not all types of stem cells in culture might require modulation of just two signaling pathways, with one acting as the ‘Engine’ signaling pathway and the other as the ‘Wheel’ signaling pathway (Fig. 2). Using mouse ESCs as an example, we consider Wnt/β-catenin the ‘Engine’ signal and mitogen-activated protein kinase (MAPK) pathway the ‘Wheel’ signal. Activation of Wnt/β-catenin by CHIR99021-mediated inhibition of GSK3 drives both self-renewal and differentiation in mouse ESCs. The differentiation-inducing effect of CHIR99021 is suppressed by PD0325901 through inhibition of MAPK signaling, resulting in mouse ESC self-renewal. It is likely that for some types of stem cells, there is more than one ‘Engine’ pathway or ‘Wheel’ pathway. For example, LIF/STAT3 is another ‘Engine’ signaling pathway for mouse ESCs. LIF/STAT3 maintains mouse ESC self-renewal in collaboration with TGF-β superfamily member BMP4, which acts as the ‘Wheel’ pathway. ‘Engine’ signaling and ‘Wheel’ signaling work together to achieve a certain degree of balance that is required to keep stem cells undifferentiated while they are proliferating. Studies on stem cell maintenance so far have implicated Wnt/β-catenin to be the key ‘Engine’ signaling pathway for a panel of stem cells including naïve ESCs, NSCs, HSCs, and intestinal stem cells.
Fig. 2. Illustration of the ‘Engine-and-Wheel’ model in stem cell maintenance. In stem cells, the ‘Engine’ signaling drives the expression of genes important for self-renewal as well as genes that induce differentiation. The ‘Wheel’ signaling works together with the ‘Engine’ signaling to maintain stem cell self-renewal by suppressing the expression of the differentiation genes.
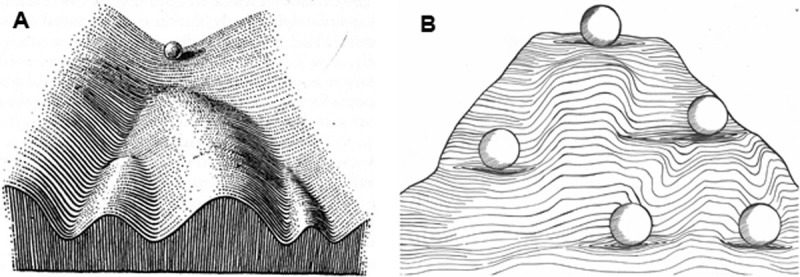
In Waddington’s epigenetic landscape model (Fig. 3A), the ball (representing a totipotent cell) can roll into different paths of the valleys (representing different cell fates). As the ball rolls down the valleys, the available paths and final destinations become more restricted, illustrating the increased differentiation of the cell (46). In Fig. 3B, we present a modified version of the model to highlight the potency and plasticity of stem cells maintained in culture. Stem cells exist in different forms throughout life. Their potency becomes more and more restricted as development proceeds. The plateaus where the balls stand represent the different developmental stages at which the potency of the stem cells can be captured and retained in vitro. Within the plateau, the ball can move around easily, implicating a certain degree of plasticity possessed by the stem cells. In the mouse, the developmental stages between the zygote and the 8-cell stage embryo can be considered as the ‘totipotency plateau’, because any individual blastomeres possess the plasticity to become a zygote and develop into a whole embryo by culturing under defined conditions. The maintenance of mouse ESCs in culture is another plateau representing developmental stages between the E2.5 morula embryo and the E4.5 late blastocyst stage embryo. Global transcriptome and epigenome studies revealed that mouse ESCs cultured in 2i are similar to cells in the E2.5-3.5 embryo while mouse ESCs maintained in LIF resemble epiblast cells in the E4.5 late blastocyst. Although these two types of ESCs have distinct functional potency and transcriptional identities, they are interconvertible by a mere switch of the culture conditions. We designate this state as the ‘ESC plateau’. The next potency level is the ‘EpiSC plateau’ in which the primed state of mouse EpiSCs are maintained in either FGF2/Activin A or the CHIR99021/IWR1 condition. EpiSCs maintained in these two conditions represent epiblast cells of different developmental stages (21, 47), yet they too are readily converted to another developmental state by adjustment of the culture condition. Stem cells in the ‘ESC plateau’ can be converted to stem cells in the ‘EpiSC plateau’ simply by switching the culture condition. The conversion EpiSCs to more potent ESCs, however, requires forced expression of one or more reprogramming factors (48). We predict that such plateaus also exist in different tissue-specific stem cells. It is likely that each type of tissue-specific stem cells has two or more ‘potency plateaus’. We anticipate that these plateaus will be gradually defined as more and better culture conditions are developed for the maintenance of authentic tissue-specific stem cells.
Fig. 3. Comparison of epigenetic landscape in development and stem cell maintenance. (A) Waddington’s epigenetic landscape (adapted from reference 46). (B) The modified version of epigenetic landscape for stem cell maintenance. Valleys represent the paths a stem cell can follow towards distinct cell fate. Ridges in between the valleys block the cells from crossing into other independent lineages. Stem cell lines established in vitro represent a range of stem cells in vivo. This range of area is represented by the plateau each of the ball stands on.
STRATEGIES FOR DEVELOPING NEW CULTURE CONDITIONS FOR STEM CELL MAINTENACE
Until now, the approaches used to search for better culture conditions for the maintenance of authentic stem cells still remain mostly empirical. While the empirical approach has resulted in a few successes, including the development of ESC and EpiSC culture conditions, most of such attempts have failed and we still do not understand how to maintain the long-term self-renewal of the majority of stem cells from different tissue origins of different species. Below, we present several strategies that might accelerate the development of new stem cell culture conditions.
Establishing reliable reporter systems
In most tissues, stem cells are rare, representing only a minority of the population, and are difficult to be identified by morphology alone. Establishing a reporter system (e.g. stem cell specific promoter-driven GFP) that faithfully recapitulates the undifferentiated status of the original stem cells will greatly facilitate the identification of self-renewal-promoting factors. The recent advance in next-generation sequencing and CRISPR/cas9 gene editing technologies has provided powerful tools for the establishment of a good reporter system both in cultured cells and in animals. A reporter system could be based on a set of gene expression in combination, as well as the expression of other regulatory elements such as enhancers or miRNAs. For the convenience of observation and quantification, the reporter system can be established by knocking a fluorescent cassette into the locus of a gene that is specifically expressed in the stem cells of interest. As an alternative approach, a dual-luciferase reporter system can be established when a more sensitive measurement is desired. The reporter construct must be experimentally verified to confirm it indeed recapitulates the endogenous gene expression pattern. Even a subtle difference within the reporter systems could result in the selection of a different cell population. In mouse, occupancy at Oct4 enhancer regulatory regions show a distinct binding preference between ESCs and EpiSCs (49). Specifically, Oct4 transcription activity in naïve ESCs is associated with distal Oct4 enhancer (DE), whereas EpiSCs depend upon proximal enhancer (PE) of Oct4. A reporter system driven by the DE of Oct4 has been used to screen factors that promote the maintenance of naïve human ESCs (15, 16).
Identifying the ‘Engine’ and the ‘Wheel’ signaling in stem cells
Germline-competent true ESCs have so far only been established from mice and rats. According to our first hypothesis, ESCs from other species most likely use a similar combination of ‘Engine’ and ‘Wheel’ signaling to control their self-renewal as mouse and rat ESCs do. We and others have tried to use either LIF/BMP4 or the 2i condition to culture ICM cells isolated from non-rodent species, but have failed to establish any ESC lines. This could be simply due to the variations in the response of ICM cells to LIF/BMP4 or 2i, because as we know the activation/suppression levels of the signals induced by these factors play a critical role in determining the outcome (50, 51). Recent studies have revealed that LIF/STAT3 and the 2i-mediated signaling pathways are also important for the maintenance of naïve human ESCs (14-19). It is likely that true ESCs can be established from other species through manipulation of the two pairs of ‘Engine-and-Wheel’ signaling pathways in ESCs: LIF/STAT3 and BMP/SMAD, or Wnt/β-catenin and MAPK.
How is it possible to identify the ‘Engine’ and the ‘Wheel’ signaling for a given stem cell type? According to our ‘Engine-and-Wheel’ model, the key step is to find the engine signaling pathway. It is likely that the engine signaling pathway for most stem cell types belongs to one of the seven major signaling pathways. Genetic and epigenetic studies in the past several decades should have provided rich information on the contribution of different genes and signaling pathways to the maintenance of different stem cell population both in vitro and in vivo. One good example is the initial study of the germline stem cell niche that has led to the identification of GDNF as a niche factor produced by Sertoli cells to regulate SSC selfrenewal. GDNF later proved to be the engine signaling pathway that promotes long-term SSC self-renewal through activation of the PI3K-AKT pathway (32, 33). In intestinal stem cells, Wnt signaling was again found to be the key engine pathway for their self-renewal (52). In many of the established protocols for stem cell maintenance, controlling the engine pathways often times is sufficient to support short-term self-renewal of the stem cells, like mouse ESCs which could be partially and temporarily retained in an undifferentiated state with the addition of LIF or CHIR alone.
The stimuli used to induce the engine signaling pathway typically triggers a cascade of reactions that leads to the activation of both self-renewal and differentiation-inducing signaling pathways. Therefore, modulation of the wheel signal is often required in order to achieve maximum self-renewal effect driven by the engine signaling. One example is the effect of MEK inhibitor or BMP4 in the LIF-mediated ESC self-renewal. Besides the JAK/STAT3 pathway, LIF can also activate the MAPK pathway which is known to induce mouse ESCs differentiation (53). Therefore, the suppression of MAPK signaling is beneficial and can further enhance LIF-mediated ESC self-renewal (54). Alternatively, BMP4 controls the stem cell balance by suppression of neural differentiation in the presence of LIF (3). This diversification of signaling pathways downstream of receptor activation is a common phenomenon. The short-term expansion of HSC based on dual inhibition of GSK3 and mTOR is another example. GSK3 inhibition releases the suppression of self-renewal pathway, but also activates mTOR which promotes lineage-commitment and stem cell depletion (40). FGFs are widely used to promote proliferation of many types of stem cells. However, at least four main signaling cascades will respond to the FGF/FGF receptor interaction, including JAK/STAT3, PLCγ, PI3K, and MAPK pathways, and this diverse reaction may potentially explain why the usage of FGF is often accompanied by other extra factors (55).
CONCLUDING REMARKERS
Stem cells hold great promise for tissue repair and regeneration. There are several obstacles must be overcome before the full potential of stem cells can be realized. One of the key obstacles is the lack of well-established methods for the longterm expansion of stem cells, especially tissue-specific stem cells and non-rodent ESCs. Accumulated evidence on stem cell research has indicated that the fine-tuning of signaling pathways is the key for stem cell maintenance. We anticipate that most types of stem cells can be maintained in vitro for an extended period of time by manipulating just two of the seven major signaling pathways.
The establishment of reliable report systems will greatly facilitate the identification of signaling pathways important for stem cell maintenance
Acknowledgments
We thank Edward Trope and My Thuy Vu for critical reading of this manuscript and Naibo Zhang for the drawing of Fig. 3B. This research was supported by National Institutes of Health (R01 OD010926), California Institute for Regenerative Medicine (CIRM) New Faculty Award II (RN2-00938), CIRM Scientific Excellence through Exploration and Development (SEED) Grant (RS1-00327), and Chen Yong Foundation of the Zhongmei Group.
References
- 1.Evans MJ, Kaufman MH. Establishment in culture of pluripotential cells from mouse embryos. Nature. (1981);292:154–156. doi: 10.1038/292154a0. [DOI] [PubMed] [Google Scholar]
- 2.Martin GR. Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc Natl Acad Sci U S A. (1981);78:7634–7638. doi: 10.1073/pnas.78.12.7634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ying Q, Nichols J, Chambers I, Smith A. BMP induction of Id proteins suppresses differentiation and sustains embryonic stem cell self-renewal in collaboration with STAT3. Cell. (2003);115:281–292. doi: 10.1016/S0092-8674(03)00847-X. [DOI] [PubMed] [Google Scholar]
- 4.Ying QL, Wray J, Nichols J, et al. The ground state of embryonic stem cell self-renewal. Nature. (2008);453:519–523. doi: 10.1038/nature06968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Buehr M, Meek S, Blair K, et al. Capture of authentic embryonic stem cells from rat blastocysts. Cell. (2008);135:1287–1298. doi: 10.1016/j.cell.2008.12.007. [DOI] [PubMed] [Google Scholar]
- 6.Li P, Tong C, Mehrian-Shai R, et al. Germline competent embryonic stem cells derived from rat blastocysts. Cell. (2008);135:1299–1310. doi: 10.1016/j.cell.2008.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tesar PJ, Chenoweth JG, Brook FA, et al. New cell lines from mouse epiblast share defining features with human embryonic stem cells. Nature. (2007);448:196–199. doi: 10.1038/nature05972. [DOI] [PubMed] [Google Scholar]
- 8.Brons IGM, Smithers LE, Trotter MWB, et al. Derivation of pluripotent epiblast stem cells from mammalian embryos. Nature. (2007);448:191–195. doi: 10.1038/nature05950. [DOI] [PubMed] [Google Scholar]
- 9.Thomson JA, Itskovitz-eldor J, Shapiro SS, et al. Embryonic Stem Cell Lines Derived from Human Blastocysts. Science. (1998);282:1145–1147. doi: 10.1126/science.282.5391.1145. [DOI] [PubMed] [Google Scholar]
- 10.He S, Nakada D, Morrison SJ. Mechanisms of stem cell self-renewal. Annu Rev Cell Dev Biol. (2009);25:377–406. doi: 10.1146/annurev.cellbio.042308.113248. [DOI] [PubMed] [Google Scholar]
- 11.Rastan S, Robertson EJ. X-chromosome deletions in embryo-derived (EK) cell lines associated with lack of X-chromosome inactivation. J Embryol Exp Morphol. (1985);90:379–388. [PubMed] [Google Scholar]
- 12.Mak W, Nesterova TB, de Napoles M, et al. Reactivation of the paternal X chromosome in early mouse embryos. Science. (2004);303:666–669. doi: 10.1126/science.1092674. [DOI] [PubMed] [Google Scholar]
- 13.Okamoto I, Otte AP, Allis CD, Reinberg D, Heard E. Epigenetic dynamics of imprinted X inactivation during early mouse development. Science. (2004);303:644–649. doi: 10.1126/science.1092727. [DOI] [PubMed] [Google Scholar]
- 14.Takashima Y, Guo G, Loos R, et al. Resetting Transcription Factor Control Circuitry toward Ground-State Pluripotency in Human. Cell. (2014);158:1254–1269. doi: 10.1016/j.cell.2014.08.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Theunissen T, Powell B, Wang H, et al. Systematic Identification of Culture Conditions for Induction and Maintenance of Naive Human Pluripotency. Cell Stem Cell. (2014);15:471–487. doi: 10.1016/j.stem.2014.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gafni O, Weinberger L, Mansour AA, et al. Derivation of novel human ground state naive pluripotent stem cells. Nature. (2013);504:282–286. doi: 10.1038/nature12745. [DOI] [PubMed] [Google Scholar]
- 17.Ware CB, Nelson AM, Mecham B, et al. Derivation of naive human embryonic stem cells. Proc Natl Acad Sci U S A. (2014);111:4484–4489. doi: 10.1073/pnas.1319738111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chan YS, Göke J, Ng JH, et al. Induction of a Human Pluripotent State with Distinct Regulatory Circuitry that Resembles Preimplantation Epiblast. Cell Stem Cell. (2013);13:663–675. doi: 10.1016/j.stem.2013.11.015. [DOI] [PubMed] [Google Scholar]
- 19.Hanna J, Cheng AW, Saha K, et al. Human embryonic stem cells with biological and epigenetic characteristics similar to those of mouse ESCs. Proc Natl Acad Sci U S A. (2010);107:9222–9227. doi: 10.1073/pnas.1004584107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Huang K, Maruyama T, Fan G. The Naive State of Human Pluripotent Stem Cells: A Synthesis of Stem Cell and Preimplantation Embryo Transcriptome Analyses. Cell Stem Cell. (2014);15:410–415. doi: 10.1016/j.stem.2014.09.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Niwa H, Burdon T, Chambers I, Smith A. Self-renewal of pluripotent embryonic stem cells is mediated via activation of STAT3. Genes Dev. (1998);12:2048–2060. doi: 10.1101/gad.12.13.2048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kim H, Wu J, Ye S, et al. Modulation of β-catenin function maintains mouse epiblast stem cell and human embryonic stem cell self-renewal. Nat Commun. (2013);4:2403. doi: 10.1038/ncomms3403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tanaka S, Kunath T, Hadjantonakis AK, Nagy A, Rossant J. Promotion of trophoblast stem cell proliferation by FGF4. Science. (1998);282:2072–2075. doi: 10.1126/science.282.5396.2072. [DOI] [PubMed] [Google Scholar]
- 24.Kunath T, Arnaud D, Uy GD, et al. Imprinted X-inactivation in extra-embryonic endoderm cell lines from mouse blastocysts. Development. (2005);132:1649–1661. doi: 10.1242/dev.01715. [DOI] [PubMed] [Google Scholar]
- 25.Shamblott MJ, Axelman J, Wang S, et al. Derivation of pluripotent stem cells from cultured human primordial germ cells. Proc Natl Acad Sci U S A. (1998);95:13726–13731. doi: 10.1073/pnas.95.23.13726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Turnpenny L, Brickwood S, Spalluto, Cosma M, et al. Derivation of Human Embryonic Germ Cells: An Alternative Source of Pluripotent Stem Cells. Stem Cells. (2003);21:598–609. doi: 10.1634/stemcells.21-5-598. [DOI] [PubMed] [Google Scholar]
- 27.Matsui Y, Zsebo K, Hogan BL. Derivation of pluripotential embryonic stem cells from murine primordial germ cells in culture. Cell. (1992);70:841–847. doi: 10.1016/0092-8674(92)90317-6. [DOI] [PubMed] [Google Scholar]
- 28.Resnick JL, Bixler LS, Cheng L, Donovan PJ. Long-term proliferation of mouse primordial germ cells in culture. Nature. (1992);359:550–551. doi: 10.1038/359550a0. [DOI] [PubMed] [Google Scholar]
- 29.Tada T, Tada M, Hilton K, et al. Epigenotype switching of imprintable loci in embryonic germ cells. Dev Genes Evol. (1998);207:551–561. doi: 10.1007/s004270050146. [DOI] [PubMed] [Google Scholar]
- 30.Durcova-Hills G, Ainscough J, McLaren a. Pluripotential stem cells derived from migrating primordial germ cells. Differentiation. (2001);68:220–226. doi: 10.1046/j.1432-0436.2001.680409.x. [DOI] [PubMed] [Google Scholar]
- 31.Leitch HG, Blair K, Mansfield W, et al. Embryonic germ cells from mice and rats exhibit properties consistent with a generic pluripotent ground state. Development. (2010);137:2279–2287. doi: 10.1242/dev.050427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Meng X, Lindahl M, Hyvönen ME, et al. Regulation of cell fate decision of undifferentiated spermatogonia by GDNF. Science. (2000);287:1489–1493. doi: 10.1126/science.287.5457.1489. [DOI] [PubMed] [Google Scholar]
- 33.Kanatsu-Shinohara M, Ogonuki N, Inoue K, et al. Long-term proliferation in culture and germline transmission of mouse male germline stem cells. Biol Reprod. (2003);69:612–616. doi: 10.1095/biolreprod.103.017012. [DOI] [PubMed] [Google Scholar]
- 34.Sato T, Vries RG, Snippert HJ, et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature. (2009);459:262–265. doi: 10.1038/nature07935. [DOI] [PubMed] [Google Scholar]
- 35.Hitoshi S, Seaberg RM, Koscik C, et al. Primitive neural stem cells from the mammalian epiblast differentiate to definitive neural stem cells under the control of Notch signaling. Genes Dev. (2004);18:1806–1811. doi: 10.1101/gad.1208404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Li W, Sun W, Zhang Y, et al. Rapid induction and long-term self-renewal of primitive neural precursors from human embryonic stem cells by small molecule inhibitors. Proc Natl Acad Sci U S A. (2011);108:8299–8304. doi: 10.1073/pnas.1014041108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Xi G, Hu P, Qu C, Qiu S, Tong C, Ying QL. Induced neural stem cells generated from rat fibroblasts. Genomics Proteomics Bioinformatics. (2013);11:312–319. doi: 10.1016/j.gpb.2013.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Reynolds BA, Weiss S. Clonal and population analyses demonstrate that an EGF-responsive mammalian embryonic CNS precursor is a stem cell. Dev Biol. (1996);175:1–13. doi: 10.1006/dbio.1996.0090. [DOI] [PubMed] [Google Scholar]
- 39.Tropepe V, Sibilia M, Ciruna BG, Rossant J, Wagner EF, van der Kooy D. Distinct neural stem cells proliferate in response to EGF and FGF in the developing mouse telencephalon. Dev Biol. (1999);208:166–188. doi: 10.1006/dbio.1998.9192. [DOI] [PubMed] [Google Scholar]
- 40.Huang J, Nguyen-McCarty M, Hexner EO, Danet-Desnoyers G, Klein PS. Maintenance of hematopoietic stem cells through regulation of Wnt and mTOR pathways. Nat Med. (2012);18:1778–1785. doi: 10.1038/nm.2984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Montarras D, Morgan J, Collins C, et al. Direct isolation of satellite cells for skeletal muscle regeneration. Science. (2005);309:2064–2067. doi: 10.1126/science.1114758. [DOI] [PubMed] [Google Scholar]
- 42.Sacco A, Doyonnas R, Kraft P, Vitorovic S, Blau HM. Self-renewal and expansion of single transplanted muscle stem cells. Nature. (2008);456:502–506. doi: 10.1038/nature07384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Fu X, Xiao J, Wei Y, et al. Combination of inflammation-related cytokines promotes long-term muscle stem cell expansion. Cell Res. (2015);25:655–673. doi: 10.1038/cr.2015.58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Huch M, Gehart H, van Boxtel R, et al. Long-term culture of genome-stable bipotent stem cells from adult human liver. Cell. (2015);160:299–312. doi: 10.1016/j.cell.2014.11.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Pires-daSilva A, Sommer RJ. The evolution of signalling pathways in animal development. Nat Rev Genet. (2003);4:39–49. doi: 10.1038/nrg977. [DOI] [PubMed] [Google Scholar]
- 46.Waddington C. The strategy of the genes a discussion of some aspects of theoretical biology. Allen & Unwin; London: (1957). [Google Scholar]
- 47.Wu J, Okamura D, Li M, et al. An alternative pluripotent state confers interspecies chimaeric competency. Nature. (2015);521:316–321. doi: 10.1038/nature14413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Guo G, Yang J, Nichols J, et al. Klf4 reverts developmentally programmed restriction of ground state pluripotency. Development. (2009);136:1063–069. doi: 10.1242/dev.030957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Yeom YI, Fuhrmann G, Ovitt CE, et al. Germline regulatory element of Oct-4 specific for the totipotent cycle of embryonal cells. Development. (1996);122:881–894. doi: 10.1242/dev.122.3.881. [DOI] [PubMed] [Google Scholar]
- 50.Tai CI, Schulze EN, Ying QL. Stat3 signaling regulates embryonic stem cell fate in a dose-dependent manner. Biol Open. (2014);3:958–965. doi: 10.1242/bio.20149514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Huang G, Yan H, Ye S, Tong C, Ying QL. STAT3 phosphorylation at tyrosine 705 and serine 727 differentially regulates mouse ESC fates. Stem Cells. (2014);32:1149–1160. doi: 10.1002/stem.1609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Clevers H, Loh KM, Nusse R. Stem cell signaling. An integral program for tissue renewal and regeneration: Wnt signaling and stem cell control. Science. (2014);346:1248012. doi: 10.1126/science.1248012. [DOI] [PubMed] [Google Scholar]
- 53.Burdon T, Stracey C, Chambers I, Nichols J, Smith A. Suppression of SHP-2 and ERK signalling promotes self-renewal of mouse embryonic stem cells. Dev Biol. (1999);210:30–43. doi: 10.1006/dbio.1999.9265. [DOI] [PubMed] [Google Scholar]
- 54.Kunath T, Saba-El-Leil MK, Almousailleakh M, Wray J, Meloche S, Smith A. FGF stimulation of the Erk1/2 signalling cascade triggers transition of pluripotent embryonic stem cells from self-renewal to lineage commitment. Development. (2007);134:2895–2902. doi: 10.1242/dev.02880. [DOI] [PubMed] [Google Scholar]
- 55.Lanner F, Rossant J. The role of FGF/Erk signaling in pluripotent cells. Development. (2007);137:3351–3360. doi: 10.1242/dev.050146. [DOI] [PubMed] [Google Scholar]