Abstract
The move away from animal models for skin safety testing is inevitable. It is a question of when, not if. As skin safety studies move away from traditional animal-based approaches, a number of replacement technologies are becoming available. Human skin in organ culture is one such technology. Organ-cultured skin has several features that distinguish it from other technologies. First and foremost, organ-cultured skin is real skin. Almost by definition, therefore, it approximates the intact skin better than other alternative models. Organ culture is an easy-to-use and relatively inexpensive approach to preclinical safety assessment. Although organ culture is not likely to replace high-throughput enzyme assays or monolayer culture/skin equivalent cultures for initial compound assessment, organ culture should find use when the list of compounds to be evaluated is small and when simpler models have narrowed the dose range. Organ-cultured skin also provides a platform for mechanistic studies.
Keywords: contact irritation, contact sensitization, corrosivity, organ culture, organotypic culture, skin
Skin irritation is a term that covers a lot of territory. Skin irritation is often the issue in regard to agents that are intentionally applied to the skin for one purpose or another (soaps and skin-cleaning products, cosmetics and pharmaceuticals, and so on) and agents that the skin comes into contact with through daily living. Such agents can induce skin irritation through mechanisms that include corrosivity, contact irritation and contact sensitization. Agents with potential for causing skin damage through one or more of these mechanisms can, hopefully, be identified in preclinical screening models. Not only is it important to identify agents with the potential for skin injury (and the relevant concentrations that produce tissue damage), but it is also important to identify their mechanisms of action. Skin irritation induced by contact irritants can, for example, sometimes be controlled by appropriate dosing. Typically, this is not possible with contact- sensitizing agents.
Some agents may have significant therapeutic potential in spite of their irritancy. The tyrosine kinase antagonists currently being used in cancer treatment as well as in the treatment of other systemic diseases can produce a dose-limiting folliculitis. Many additional tyrosine kinase antagonists die in development due to their effects on the skin. It would be of significant value if, during discovery and early-stage development, the potential to elicit serious skin problems could be identified. It would also be of value to understand the mechanisms underlying the induction of skin irritation so that effective countermeasures can be utilized.
Systemic and localized inflammatory diseases are major contributors to skin irritation. Conditions such as atopic dermatitis, acne, rosacea and psoriasis, and so on drive large numbers of individuals to the dermatological clinic for relief.
In all of these conditions where skin irritation is a major (or sole) manifestation, investigators need appropriate tools for study. Animal models and in vitro culture models are needed during development to predict which structures will produce irritation. Models are also needed to help identify the pathophysiological mechanisms. Finally, relevant models are needed for the assessment of potential therapeutic interventions.
Research animals have long been used for these purposes. The Draze skin test, for example, has been the US FDA-accepted method to assess skin irritation since the 1940s [1]. Essentially, research animals (usually albino rats or rabbits) are exposed to the test agent over a range of concentrations, and parameters of inflammation, including redness and swelling, are recorded over time. Potential for corrosivity and contact irritation can be assessed in this manner. The local lymph node assay [2,3], usually in mice or guinea pigs, provides a way to determine a compound’s potential for contact sensitization. In this test, animals are exposed to the reagent in multiple doses. The final eliciting dose is followed by examination of the local draining lymph nodes for evidence of an immune response. There is a desire to move away from the animal models that have been used for years. The Draze test and other animal procedures are considered cruel by animal welfare advocates. They are also considered unscientific in that there is never a 100% correlation between events occurring in animals and the events that will be seen when humans are exposed to the same agent.
In vitro cell culture models have been considered as replacements for live animals for decades. Unfortunately, no single test or battery of tests has been validated, except for corrosivity [4,5]. Early efforts made use of cells in monolayer culture. Over the years, multiple different cell types have been proposed and multiple conditions have been employed. The problem, however, is that in monolayer cell culture, the range of cellular responses is too small to take into account all of the different ways in which the skin may be damaged. As an alternative to monolayer culture, 3D cultures, referred to as skin-equivalent or epidermal-equivalent cultures, have been developed [6–12]. Such cultures consist of isolated epidermal keratinocytes on a 3D matrix consisting of type I collagen with or without additional proteins and other components. In some cases (skin-equivalent cultures), dermal fibroblasts are embedded in the collagen. Keratinocytes on the surface proliferate to completely cover the surface. Concomitantly, the epithelial cells grow upwards to form a 3D layer with the proliferating cells at the base and the differentiating cells at the top. Such cultures represent intact skin more closely than do cells in monolayer culture. The technology for generating skin-equivalent cultures is well developed, and there are a number of commercial sources for these skin substitutes [101–104]. To date, however, it is not clear if such cultures will provide a model to replace most of the animals currently used in testing. 3D cultures may find more opportunity as part of mechanistic studies. At present, however, even this is not clear. What is becoming clear is that the technology will find use in wound-healing applications, particularly in the healing of chronic, difficult-to-close wounds [13].
Organ-cultured skin as an approach to irritation testing
The use of intact skin in organ culture provides another approach to skin irritation/skin damage research and testing. Unlike skin-equivalent cultures, which make use of isolated keratinocytes and fibroblasts in a reconstituted structure, organ culture makes use of small pieces of intact skin. Our laboratory has made extensive use of this approach, and the remainder of this review will (briefly) describe basic organ culture technology, describe some of the advantages (and disadvantages) that intact ex vivo skin has compared with other technologies, and indicate how it can be utilized to address issues that are relevant to preclinical development programs.
Technology
Human skin is available from a number of sources. Foreskin tissue obtained at circumcision is one common source of tissue. Adult tissue is often available after wound-closure procedures performed in dermatological surgery clinics or in cosmetic surgery clinics. Skin is also available after many general surgeries. In all such cases, tissue is obtained for nonresearch purposes and would otherwise be discarded. The major institutional review board (IRB) issues relate to collection of confidential patient information. Often, this information is not needed at all, and when the tissue is obtained de-identified, a waiver of IRB oversight may be obtained. If IRB oversight is required, the application would fall under the category of no direct involvement of human subjects. Volunteer tissue donors provide another source of skin. In typical protocols, up to 12 2-mm full-thickness biopsies from skin donors can be obtained. Sun-protected skin from the hip is commonly used, but depending on the research requirements, skin from the forearm (sun exposed) or skin from the scalp (containing hair follicles) may be utilized. For studies related to specific disease processes, lesional skin is often of value. Psoriatic plaque skin, for example, is used in ex vivo settings, although for testing antipsoriatic agents, the use of biopsied skin transplanted to severe combined immunodeficient mice seems to be the preferred approach [14–19].
Skin is a robust tissue and can be maintained in organ culture under a variety of culture conditions and in a number of different media formulations [20–22]. In previous studies [23,24], the author has found that the optimal formulation is a basal medium without serum or growth factors, but in which the calcium concentration has been optimized for fibroblast function (i.e., approximately 1.5 mM [25]). A 2-mm punch biopsy can be maintained for several days or weeks in this culture medium when incubated at 37°C in an atmosphere of 95% air and 5% CO2. By contrast, if the culture conditions are optimized for keratinocyte proliferation (based on monolayer culture studies [26]), necrosis begins immediately, and both the epidermal keratinocytes and dermal fibroblasts will be dead within a few days. While many types of tissue require an air–liquid interface, organ-cultured skin remains viable and healthy when maintained submerged. Typically, the author incubate the tissue for 8–12 days with fresh culture medium (and any treatment) provided at 2- or 3-day intervals. The used culture medium obtained at each media change provides a rich source of secreted biomarkers. At the end of the incubation period, the tissue can be fixed in formalin for histology or in optimum cutting tissue (OCT) for immunohistology or RNA isolation. Organ-cultured skin is also amenable to electron microscopy and other advanced microscopic techniques.
Differences between organ-cultured skin & 3D skin-equivalent cultures
Two features distinguish organ-cultured skin from skin-equivalent cultures. First and foremost, skin-equivalent cultures consist of cells (keratinocytes with or without fibroblasts and perhaps other cells) that have been disaggregated and reconstituted into a 3D structure, while organ-cultured skin is intact tissue. There are numerous cell types in the skin in addition to keratinocytes and fibroblasts, and these cells are represented in organ culture. The microvasculature is still present as are the cells of the immune/inflammatory system. Whereas the keratinocytes and fibroblasts in skin-equivalent cultures synthesize a basement-like structure, a true basement membrane separates the epidermis and dermis in organ culture. Likewise, in organ culture, all of the connective tissue elements present in vivo are represented. By contrast, the connective tissue in skin-equivalent cultures primarily consist of solubilized and repolymerized type I collagen. Thus, organ-cultured skin is much more complex than is a skin-equivalent culture.
Another feature that distinguishes organ culture from skin-equivalent culture is variability. Typically, skin-equivalent cultures are generated from banks of cells that have been isolated and stored for that purpose. By contrast, every individual who serves as a skin donor brings the genetic and epigenetic variability that makes up the population. Is variability a strength or a weakness? While the inherent variability often raises the number of individual subjects needed to validate a given finding (a weakness), it concomitantly points to the subject-to-subject variability that will be seen in clinical studies (a strength).
Organ-cultured skin to assess chemically induced skin damage: corrosivity, contact irritation & contact sensitization
Skin organ culture technology can play an integral role in all aspects of skin biology/skin disease research. Regarding irritation studies per se, organ culture technology can be useful for helping to identify agents with potential for irritation, as well as agents that mitigate irritation. Perhaps more importantly, organ-cultured skin provides a tool for unraveling mechanistic events that contribute to irritation.
Chemically induced skin damage can be produced through multiple mechanisms. Generally speaking, agents that damage the skin upon direct exposure are often referred to as corrosives, contact irritants and contact sensitizers. While the end result may be similar, the mechanistic events that bring about the damage may be quite different. A number of different laboratories have demonstrated the applicability of organ culture technology to assessment of skin damage. Skin from humans [20–24,27–29], as well as from rodents, rabbits and pigs, has been adapted to organ culture [30–34], and skin from these different animals provide a way to assess agents for skin damage. Numerous end points have been used as markers of skin damage. Most commonly, this includes release of cytosolic contents such as lactate dehydrogenase and glutamic-oxaloacetate transaminase [30] but, in other cases, release of membrane fatty acids and their metabolites has been followed [33]. Often, assessment of biochemical markers has been correlated with histological changes. In other cases, special stains such as methyl green pyronine have been used to identify damage [21]. In most cases, the readouts effectively predict damage to the tissue. Cell injury, however, is not the only effect of irritant exposure. Hyperplastic changes, especially in the epidermis, may be seen under conditions in which cytotoxic injury does not occur.
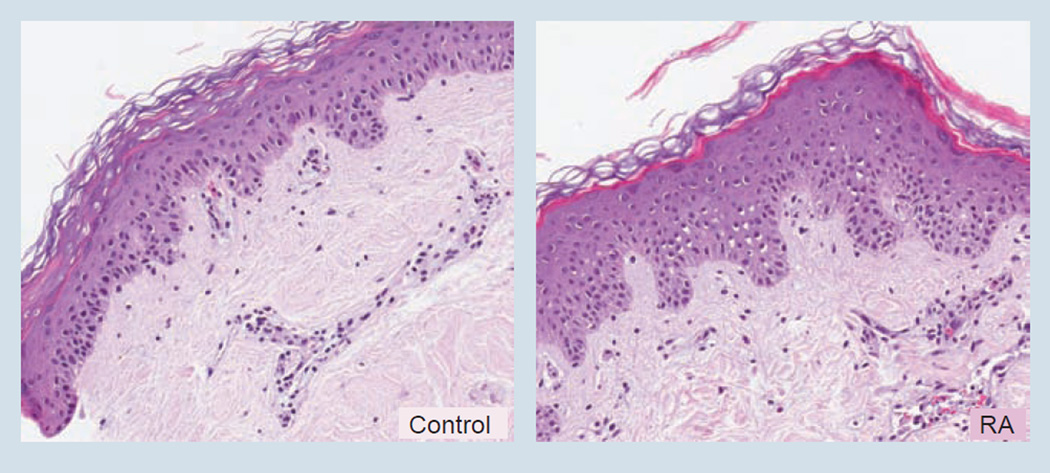
With this in mind, the author carried out a series of studies in which agents known to be corrosives, contact irritants and contact sensitizers were examined for their effects on organ cultures of human skin [35]. Contact irritants included all-trans retinoic acid (RA), sodium lauryl sulfate and benzylkonium chloride. Each of these three agents is a common ingredient used in various skincare or eyecare products. Contact sensitizers included three strong sensitizes (oxazolone, nickel sulfate and eugenol) and two characterized as moderate sensitizers (isoeugenol and ethylene glycol dimethacrylate). Finally, the author utilized an organic mercury compound, aminophenyl mercuric acetate (APMA), as a corrosive. Each agent was tested over a wide range of concentrations for effects on histological structure and biochemical function in skin cultures from several different donors. In parallel, each agent was tested for effects on human dermal fibroblasts and human epidermal keratinocytes in monolayer culture. To summarize the results of this study, the corrosive agent produced cytotoxic changes in skin that could be seen within hours of exposure. This included a loss of viable cells in both the epidermis and dermis, eventually leading to complete necrosis and separation of the two skin compartments at the dermal–epidermal junction. Concentrations as low as 1 µM or less were toxic. When epidermal keratinocytes and dermal fibroblasts were exposed to APMA in monolayer cell culture, cytotoxicity for both cell types occurred over the same concentration range. The skin irritants also produced cytotoxic changes observable in organ culture. However, several features distinguished the effects of all three contact irritants from those induced by the corrosive agent. First, whereas the corrosive agent was fully cytotoxic at a concentration of 1 µM or less, the contact irritants were active over the range of 1–10 µM. More importantly, while these agents were cytotoxic at high concentrations (5–10 µM), lower doses induced hyperplastic changes in the epidermis. Figure 1 shows the appearance of normal skin maintained for 8 days under control conditions or in the presence of a 1 µM solution of RA. Induction of epidermal hyperplasia is a complex event. The ability to induce epidermal hyperplasia is of interest since epidermal hyperplasia is commonly observed in intact skin when it is exposed to contact irritants, including RA and sodium lauryl sulfate [36,37]. The capacity to induce proliferative changes distinguishes these contact irritants from the corrosive.
Figure 1. Effects of retinoic acid on histology of human skin in organ culture.
The control skin section on the left was incubated in a standard serum-free, growth factor-free culture medium for 8 days with fresh medium added at 2-day intervals. The section on the right was incubated under the same conditions but in addition treated with 1 µM RA during the 8-day incubation period (hematoxylin and eosin stained). See [23,24] for details.
RA: Retinoic acid.
In comparison with the corrosive agent and skin irritants, the third group of chemical agents, the contact sensitizers, were unremarkable in their effects on organ culture histology. No consistent alterations were seen at concentrations below 100–200 µM (or higher), and even at these high concentrations, thinning of the still-viable epidermis rather than complete necrosis was the major finding. In parallel, when the author examined the contact sensitizers for effects on keratinocytes and fibroblast growth in monolayer culture, there was no inhibition at concentrations below 100–200 µM and no cytotoxicity. Figure 2 shows histological features in organ-cultured skin after 8 days in the presence of various contact-sensitizing agents.
Figure 2. Effects of different contact sensitizers on histology of human skin in organ culture.
The control skin section (upper left) was incubated in a standard serum-free, growth factor-free culture medium for 8 days with fresh medium added at 2-day intervals. The remaining sections were incubated under the same conditions but in addition treated with the indicated contact sensitizer (250 µM) during the 8-day incubation period (hematoxylin and eosin stained). See [35] for details.
EGDM: Ethylene glycol dimethacrylate.
As part of our initial study comparing the different classes of skin toxicants, soluble biomarkers of epidermal function were assessed in organ culture fluid. All three of the contact irritants induced an increase in amphiregulin (Figure 3). There was a relationship between the level of this EGF receptor agonist and the extent of the hyperplastic changes. Amphiregulin is well known to stimulate keratinocyte proliferation in monolayer culture [38,39], and it has been suggested that keratinocytes that are able to grow in monolayer culture represent the culture equivalent of the hyperplastic epidermis. Amphiregulin (along with heparin-binding [HB]-EGF) is also known to be upregulated in human skin in vivo, in response to RA [39–42]. By contrast, exposure of organ-cultured skin to the panel of contact-sensitizing agents did not induce amphiregulin expression. Neither did APMA.
Figure 3. Amphiregulin levels in human skin organ culture fluid.
Levels of the protein were assessed in day 2 culture fluids by enzyme-linked immunosorbant assay. The three contact irritants were used at concentrations of 1 µM (RA), 5 µM (SLS) and 10 µM (BZK), respectively. The five contact sensitizers were used at 250 µM. See [35] for details.
BZK: Benzylkonium chloride; EGDM: Ethylene glycol dimethacrylate; RA: Retinoic acid; SLS: Sodium lauryl sulfate.
Conditioned medium from contact irritant- exposed and contact sensitizer-exposed tissue was also used to assess a panel of proinflammatory and anti-inflammatory cytokines, as well as several other potentially useful biomarkers. Results from a study in which RA as a contact irritant and nickel sulfate as a contact sensitizer were compared in a series of subjects are shown for IL-6 in Figure 4. A very different pattern of response was seen with these two agents. With RA, organ-cultured skin from all eight subjects generated increased amounts of IL-6 relative to their respective (untreated) controls. Levels varied considerably among subjects, but the average increase was approximately twofold. With nickel sulfate as the stimulant, skin from five out of the eight same subjects showed no increase relative to control. Culture fluids from the other three subjects generated strong responses (threefold, 12-fold and 22-fold). These findings are consistent with the suggestion that essentially everyone responds to strong contact irritants because of their direct effects on resident cells of the skin. By contrast, the finding that only a subset of subjects responded to the known contact sensitizer is consistent with the fact that only a subset of individuals will typically demonstrate an allergic response to most sensitizing agents.
Figure 4. IL-6 levels in human skin organ culture fluid.
Skin biopsies from eight subjects were maintained in organ culture for 2 days under control conditions and treated with RA or nickel sulfate. IL-6 levels were assessed by enzyme-linked immunosorbant assay. RA was used at 1 µM and nickel sulfate was tested at 500 µM. Values are from individual subjects.
See [35] for details.
RA: Retinoic acid.
Organ-cultured skin for mechanistic studies
The mechanisms underlying chemically induced skin damage are not yet completely understood. Given that an intact organism is required for full manifestation of irritation, can the use of skin in organ culture provide insights into the pathophysiology? It might be possible to address this issue, perhaps through a case study with one well-known skin irritant, RA.
RA is a classic example of a skin pharmaceutical that also has the capacity to produce skin irritation. Following early studies demonstrating improved appearance in the skin of rodents with topical RA application, controlled clinical studies demonstrated the same phenomenon in both photodamaged skin [43,44] and chronologically aged skin [45,46]. Experimental studies showed that RA treatment decreased production of the major connective tissue-degrading enzymes in the skin, including matrix metalloproteinase (MMP)-1 (collagenase-1), MMP-3 (stromelysin-1) and MMP-9 (gelatinase B) [47–49]. Later, it was shown that RA also inhibited MMP function, in part by raising production of the major MMP inhibitor in the skin, tissue inhibitor of metalloproteinases-1 [50]. Topical RA use results in a demonstrable increase in type I collagen in treated skin [51], but it is not clear if increased biosynthesis of the precursor molecule (type I procollagen) is primarily responsible for this, or whether increased collagen deposition is a result of decreased breakdown.
While effects of RA on collagen turnover have been well studied in relation to skin repair, topical retinoid use also stimulates keratinocyte proliferation and increased epidermal thickening [36,38–42]. The epidermal effects probably contribute to improved appearance, but how important this is to skin repair is uncertain. Keratinocyte proliferation is associated with motility [52], and one could envision both contributing to rapid wound closure. Mechanistically, proliferation and motility in the epidermis are dependent not only on autocrine and paracrine growth factors but also on the structure of the underlying dermis. Both proliferation and motility are impaired when the associated connective tissue is fragmented [53,54].
While RA has proven to be effective in the repair of photo- skin and age-damaged skin, most people who use topical RA experience some degree of irritation. The retinoid irritation response is characterized by redness/edema, dryness, cracking, burning, itching and flaking at the treated site. Irritation is a major cause of retinoid noncompliance among users prescribed with tretinoin/Renova®. Packaging labels on tretinoin indicate that up to 40% of users will experience irritation. In the studies on topical retinol (vitamin A) use in aged individuals, there was a 35% dropout rate over a 16-week period [46].
The cellular and molecular basis of retinoid irritation is not fully understood. There is an assumption that epidermal hyperplasia (and the concomitant alterations in epidermal cohesion/differentiation/barrier function that occur in conjunction with hyperplastic growth) [55] are directly related to inflammatory changes that underlie irritation. Hyperplastic growth and barrier disruption are associated with upregulation of proinflammatory cytokines such as IL-1, IL-6, IL-8 and macrophage chemotactic peptide-1 [56,57]. In turn, proinflammatory cytokine production leads to the upregulation of leukocyte adhesion molecules such as intercellular adhesion molecule-1, vascular cell adhesion molecule, E-selectin and P-selectin on microvascular endothelial cells. This drives the recruitment of inflammatory cells from the circulation [58]. While recruitment of circulating leukocytes cannot occur in organ culture, the author found that the same changes in cytokine production and adhesion molecule expression could be seen when skin biopsies were exposed to RA ex vivo [59]. Of interest, in the same study, the author found that when a nonirritating experimental retinoid was used in place of RA, cytokine production and adhesion molecule expression were not upregulated [59].
One might ask how valuable organ culture technology is to understanding retinoid-induced skin irritation, since the same information could be achieved by treating subjects topically with RA and then assessing their response. The answer is twofold. First, while RA is approved for human use, the goal in most cases is to assess agents for skin irritation potential while they are still in preclinical development. The nonirritating retinoid used in our study (Molecular Design International [MDI]-301 [59]) is not yet approved for testing in subjects. Perhaps more importantly from the standpoint of mechanistic insight, it is possible to induce changes in organ-cultured skin that are of interest and then use interventional approaches to modify the response. In this way, it is possible to tease out mechanisms of action. Past studies, for example, have shown that the IL-1 receptor antagonism effectively blocks events that directly lead to tissue damage in the skin [60].
Skin organ culture has also been useful in dissecting the mechanism of RA-induced epidermal hyperplasia per se. Specifically, it has been shown that retinoid treatment leads to induction of two EGF receptor ligands, HB-EGF and amphiregulin, and to activation of the EGF receptor signaling pathway [42]. As noted above, similar events had been previously shown in intact skin. Unique to organ culture, however, was the capacity to demonstrate that while at least two EGR receptor ligands were upregulated, a neutralizing anti-HB-EGF antibody strongly inhibited the hyperplastic changes, while anti-amphiregulin was less effective. Not surprisingly, a powerful EGF receptor tyrosine kinase antagonist proved to be highly effective in blocking hyperplasia from developing in response to RA treatment [42]. Equally important, it was found that suppressing EGF receptor signaling in organ cultures of normal skin had little effect on structure and function. By contrast, suppressing another growth factor-signaling pathway – that is, the IGF-1 pathway – proved to be growth suppressing and cytotoxic [61]. Finally, studies with a nonirritating retinoid (MDI-301) allowed us to dissociate, at least in part, irritation from epidermal hyperplasia. MDI-301 was comparable to RA in inducing epidermal thickening without inducing proinflammatory events [59]. Of interest in this regard, other investigators have shown that RA-induced EGF receptor activation is critical for hyaluronan production in keratinocytes [41]. Hyaluronan deposition in the epidermis itself may underlie epidermal thickening. In addition, modulation of the matrix influences proliferation. Thus, it might be anticipated that modulation of hyaluronan synthesis is a key feature in EGF-induced epidermal proliferation.
Overall, from these studies it appears reasonable to suggest that what is observed in organ culture in a variety of situations and under a variety of conditions appears to replicate what is seen in intact skin. The advantage of the ex vivo situation is that it provides a mechanism to utilize interventions that could not be used in the human subjects, either because the therapeutic treatment was not approved for human use or because the intervention would be too difficult in the intact host.
Expert commentary
The move away from animal models for skin safety studies is inevitable. It is a question of when, not if. The move away from animals, especially in the cosmetic field, reflects societal demands that have little to do with science. Beyond this, however, there is the understanding that results in animals do not always reflect what will be seen when humans are topically exposed to a new chemical agent. For example, if dogs were used in preclinical testing, poison ivy would be approved for human use. As skin safety studies move away from traditional animal-based models that have been used for decades, there are a number of technologies that can be considered as replacements. Human skin in organ culture is one such technology. Organ-cultured skin has several features that distinguish it from other technologies. First and foremost, organ-cultured skin is real skin. Almost by definition, therefore, it approximates responses in the intact skin better than other alternative models. Organ culture is an easy-to-use and relatively inexpensive approach to preclinical assessments. Although organ culture may not replace high-throughput enzyme assays or monolayer culture/skin equivalent cultures for initial compound assessments, organ culture should find use when the list of compounds to be evaluated has been reduced and when simpler models have narrowed the dose range.
Organ-cultured skin provides a platform for generating sophisticated data on compounds before they reach the stage of clinical testing. Our own studies have shown that agents with corrosive activity, agents that are contact irritants and that are contact sensitizers produce markedly different responses in intact skin in organ culture. While the number of agents that have been tested (in the laboratory, at least) is small, the data generated to date are compelling enough to suggest that larger studies should be undertaken.
One might argue that having a more sophisticated test for corrosivity is not needed. However, being able to identify potential for contact irritation and, even more importantly, to identify potential for contact sensitization would be highly valued. Will the use of organ-cultured skin be able to accomplish this? Only time and additional studies will tell. Whether or not organ-cultured skin ultimately provides a reliable approach for assessing skin toxicity and for distinguishing among classes of skin toxicants, there is no doubt that organ culture approaches are useful for dissecting the underlying cellular and molecular mechanisms that lead to toxicity in the skin.
Five-year view
If the use of organ-cultured human skin is going to provide an alternative to currently used animal models for skin safety studies, several things will need to happen during the next 5 years. First and foremost, studies with a large number of chemical agents will have to be carried out. Agents of various chemical structures that are known to be corrosives, contact irritants and contact sensitizers will all need to be evaluated. On the basis of the data generated with a small sample of such agents, there is reason to hope that larger, more definitive studies will be successful. It must be kept in mind, of course, that no single in vitro approach is likely to be completely successful. Not all corrosives will damage the skin in the same manner, likewise with contact irritants. Furthermore, it cannot be expected that all contact sensitizers will be inert, if for no other reason than that some contact sensitizers will also have contact irritant capacity. At high concentrations, most chemical agents will, in fact, have a direct cytotoxic effect. A successful in vitro culture system will need to be capable of elucidating toxicity occurring through a variety of mechanisms at concentrations reflecting those at which the skin is likely to come in contact with them. This means having enough suitable biochemical and/or histological end points that, together, they encompass all of the relevant mechanisms responsible for tissue damage.
The technology or combination of technologies that, ultimately, replace currently employed animal models must be as reliable as the models they replace. Hopefully, the new technologies will be more predictive. Beyond reliability, whatever technology or combination of technologies come to replace existing methods, it will have to be reproducible, easy to use, inexpensive and widely available. If organ-cultured skin is to be part of this mix, there are multiple logistical issues that will need to be addressed in the coming years.
Having a reliable source of tissue is one such issue. While skin that is available from surgical and other medical procedures is readily accessible in many instances, the variability in tissue from such sources is a challenge. An alternative is to use tissue obtained from skin donors who have been recruited for that purpose. While this adds a layer of regulatory compliance, the consistency from tissue piece to tissue piece makes the subsequent work easier. As noted above, there is significant biological variability in the use of whole-skin organ culture; that is, variability based on subject-to-subject differences. One does not need additional variability due to differences in tissue handling or due to associated diseases or traumas that necessitated the surgical procedure in the first place. Other questions that need to be addressed relate to how the tissue is handled during acquisition, transport to the testing laboratory and processing. Appropriate responses to all of the logistical and regulatory issues will determine whether the benefits of organ culture technology are widely available for safety studies, or whether the use of organ-cultured skin will only be available in a few selected research laboratories.
Key issues.
It will be important to confirm with a large number of chemical agents that the results obtained with human skin in organ culture predict effects that can be expected upon human exposure. In particular, it will be important to determine how effectively this technology is capable of distinguishing corrosive agents, contact irritants and contact sensitizers from one another.
It will be necessary to identify and overcome the roadblocks that prevent human skin from being readily available for this purpose. Both logistical and regulatory issues need to be addressed.
We need to determine how best to utilize the technology – that is, what end points are of value, how best to measure these end points, and so on, in order to establish corrosive activity, contact irritant activity and contact sensitization activity.
We will need to validate the use of organ culture as a primary approach to elucidating mechanisms of action. While this may not be a high priority in the cosmetic field, understanding mechanisms of action and mechanisms of toxicity are critical to pharmaceutical development.
Acknowledgments
This work was supported in part by grants from the United States Public Health Service. The author wishes to acknowledge that he has a financial position in a company (JV Biolabs) that utilizes technologies discussed in the review for commercial purposes.
Footnotes
Financial & competing interests disclosure
The author has no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
References
Papers of special note have been highlighted as:
• of interest
- 1. Draize JH, Woodard G, Calvery HO. Methods for the study of irritation and toxicity of substances applied topically to the skin and mucous membranes. J. Pharmacol. Exp. Ther. 1944;82:377–390. • Established animal-based technology for assessing skin damage. Replacing tests described here and similar tests are the goal of the alternative technologies described in this article.
- 2.van Och FM, Vandebriel RJ, Prinsen MK, De Jong WH, Slob W, van Loveren H. Comparison of dose-responses of contact allergens using the guinea pig maximization test and the local lymph node assay. Toxicology. 2001;167(3):207–215. doi: 10.1016/s0300-483x(01)00425-5. [DOI] [PubMed] [Google Scholar]
- 3.Basketter DA, Scholes EW. Comparison of the local lymph node assay with the guinea-pig maximization test for the detection of a range of contact allergens. Food Chem. Toxicol. 1992;30(1):65–69. doi: 10.1016/0278-6915(92)90138-b. [DOI] [PubMed] [Google Scholar]
- 4.Botham PA, Earl LK, Fentem JH, Rouguet R, van de Sandt JJM. Alternative methods for skin irritation testing. Altern. Lab. Anim. 1998;26:195–211. [PubMed] [Google Scholar]
- 5. Botham PA. The validation of in vitro methods for skin irritation. Toxicol. Lett. 2004;149(13):387–390. doi: 10.1016/j.toxlet.2003.12.048. • Validated in vitro assay for corrosivity.
- 6.Bernhofer LP, Seiberg M, Martin KM. The influence of the response of skin equivalent systems to topically applied consumer products by epithelial–mesenchymal interactions. Toxicol. In Vitro. 1999;13(2):219–229. doi: 10.1016/s0887-2333(98)00087-3. [DOI] [PubMed] [Google Scholar]
- 7.Bernhofer LP, Barkovic S, Appa Y, Martin KM. IL-1alpha and IL-1ra secretion from epidermal equivalents and the prediction of the irritation potential of mild soap and surfactant-based consumer products. Toxicol. In Vitro. 1999;13(2):231–239. doi: 10.1016/s0887-2333(98)00088-5. [DOI] [PubMed] [Google Scholar]
- 8.Boelsma E, Gibbs S, Faller C, Ponec M. Characterization and comparison of reconstructed skin models: morphological and immunohistochemical evaluation. Acta Derm. Venereol. 2000;80(2):82–88. [PubMed] [Google Scholar]
- 9.Coquette A, Berna N, Vandenbosch A, Rosdy M, De Wever B, Poumay Y. Analysis of interleukin-1alpha (IL-1alpha) and interleukin-8 (IL-8) expression and release in in vitro reconstructed human epidermis for the prediction of in vivo skin irritation and/or sensitization. Toxicol. In Vitro. 2003;17(3):311–321. doi: 10.1016/s0887-2333(03)00019-5. [DOI] [PubMed] [Google Scholar]
- 10.Leh CL, Jacobs JJ, Elliott GR, Das PK. A two-centre evaluation of the human organotypic skin explant culture model for screening contact allergens. Altern. Lab. Anim. 2003;31(6):553–561. doi: 10.1177/026119290303100604. [DOI] [PubMed] [Google Scholar]
- 11.Ponec M, Kempenaar J. Use of human skin recombinants as an in vitro model for testing the irritation potential of cutaneous irritants. Skin Pharmacol. 1995;8(12):49–59. doi: 10.1159/000211330. [DOI] [PubMed] [Google Scholar]
- 12.Ponec M, Boelsma E, Gibbs S, Mommaas M. Characterization of reconstructed skin models. Skin Pharmacol. Appl. Skin Physiol. 2002;15(Suppl. 1):4–17. doi: 10.1159/000066682. [DOI] [PubMed] [Google Scholar]
- 13. Damodaran G, Syed M, Leigh I, Myers S, Navsaria H. Clinical application of skin substitutes. Expert Rev. Dermatol. 2008;3:345–356. • Review on surgical application of skin substitutes.
- 14.Bhagavathula N, Nerusu KC, Fisher GJ, et al. Amphiregulin and epidermal hyperplasia: amphiregulin is required to maintain the psoriatic phenotype of human skin grafts on severe combined immunodeficient mice. Am. J. Pathol. 2005;166(4):1009–1016. doi: 10.1016/S0002-9440(10)62322-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bhagavathula N, Nerusu K, Reddy M, et al. BP-1007: a novel synthetic thiazoladinedione that inhibits epidermal hyperplasia in psoriatic skin SCID mouse transplants following topical application. J. Pharmacol. Exp. Ther. 2005;315:996–1004. doi: 10.1124/jpet.105.091066. [DOI] [PubMed] [Google Scholar]
- 16.Bhagavathula N, Nerusu KC, Hanosh A, et al. 7-chloro-5-(4-hydroxyphenyl)-1-methyl-3-(naphthalen-2-ylmethyl)-4,5- dihydro-1H-benzo[b][1,4]diazepin-2(3H)-one (Bz-423), a benzodiazepine, suppresses keratinocyte proliferation and has antipsoriatic activity in the human skin-severe, combined immunodeficient mouse transplant model. J. Pharmacol. Exp. Ther. 2008;324(3):938–947. doi: 10.1124/jpet.107.130955. [DOI] [PubMed] [Google Scholar]
- 17.Blumberg H, Dinh H, Dean C, Jr, et al. IL-1RL2 and its ligands contribute to the cytokine network in psoriasis. J. Immunol. 2010;185(7):4354–4362. doi: 10.4049/jimmunol.1000313. [DOI] [PubMed] [Google Scholar]
- 18.Ellis CN, Varani J, Fisher GJ, et al. Troglitazone improves psoriasis and normalizes models of proliferative skin disease: ligands for peroxisome proliferator-activated receptor-gamma inhibit keratinocyte proliferation. Arch. Dermatol. 2000;136(5):609–616. doi: 10.1001/archderm.136.5.609. [DOI] [PubMed] [Google Scholar]
- 19.Zeigler ME, Chi Y, Varani J, et al. Comparative effects of cyclosporin A and clobetasol propionate (Temovate) on normal and psoriatic human skin transplanted to scid mice. Lab. Invest. 2001;81:1253–1261. doi: 10.1038/labinvest.3780339. [DOI] [PubMed] [Google Scholar]
- 20.Agren UM, Tammi M, Tammi R. Hydrocortisone regulation of hyaluronan metabolism in human skin organ culture. J. Cell. Physiol. 1995;164(2):240–248. doi: 10.1002/jcp.1041640204. [DOI] [PubMed] [Google Scholar]
- 21.Jacobs JJ, Leh C, Cammans KD, Das PK, Elliott GR. An in vitro model for detecting skin irritants: methyl green-pyronine staining of human skin explant cultures. Toxicol. In Vitro. 2002;16(5):581–588. doi: 10.1016/s0887-2333(02)00039-5. [DOI] [PubMed] [Google Scholar]
- 22.Jacobs JJ, Leh CL, Cammans KD, Das PK, Elliott GR. Assessment of contact allergens by dissociation of irritant and sensitizing properties. Toxicol. In Vitro. 2004;18(5):681–690. doi: 10.1016/j.tiv.2004.03.007. [DOI] [PubMed] [Google Scholar]
- 23.Varani J, Fligiel SE, Schuger L, et al. Effects of all-trans retinoic acid and Ca on human skin in organ culture. Am. J. Pathol. 1993;142(1):189–198. [PMC free article] [PubMed] [Google Scholar]
- 24.Varani J, Perone P, Griffiths CE, Inman DR, Fligiel SE, Voorhees JJ. All-trans retinoic acid (RA) stimulates events in organ-cultured human skin that underlie repair. Adult skin from sun-protected and sun-exposed sites responds in an identical manner to RA while neonatal foreskin responds differently. J. Clin. Invest. 1994;94(5):1747–1756. doi: 10.1172/JCI117522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Varani J, Mitra RS, Gibbs D, et al. All-trans retinoic acid stimulates growth and extracellular matrix production in growth-inhibited cultured human skin fibroblasts. J. Invest. Dermatol. 1990;94(5):717–723. doi: 10.1111/1523-1747.ep12876294. [DOI] [PubMed] [Google Scholar]
- 26.Varani J, Nickoloff BJ, Dixit VM, Mitra RS, Voorhees JJ. All-trans retinoic acid stimulates growth of adult human keratinocytes cultured in growth factor-deficient medium, inhibits production of thrombospondin and fibronectin, and reduces adhesion. J. Invest. Dermatol. 1989;93(4):449–454. doi: 10.1111/1523-1747.ep12284020. [DOI] [PubMed] [Google Scholar]
- 27.Tammi R, Tammi M. Influence of retinoic acid on the ultrastructure and hyaluronic acid synthesis of adult human epidermis in whole skin organ culture. J. Cell. Physiol. 1986;126(3):389–398. doi: 10.1002/jcp.1041260309. [DOI] [PubMed] [Google Scholar]
- 28.Iwata M, Iwata S, Everett MA, Fuller BB. Hormonal stimulation of tyrosinase activity in human foreskin organ cultures. In Vitro Cell. Dev. Biol. 1990;26(6):554–560. doi: 10.1007/BF02624203. [DOI] [PubMed] [Google Scholar]
- 29.Van de Sandt JJM, Rutten AAJJL, Koster HBWM. Cutaneous toxicity testing in organ culture: neutral red uptake and reduction of tetrazolium dye. Toxicol. In Vitro. 1993;7:81–86. doi: 10.1016/0887-2333(93)90115-l. [DOI] [PubMed] [Google Scholar]
- 30.Bartnik FG, Pittermann WF, Mendorf N, Tillman U, Küstler K. Skin organ culture for the study of skin irritancy. Toxicol. In Vitro. 1990;4(45):293–301. doi: 10.1016/0887-2333(90)90067-4. [DOI] [PubMed] [Google Scholar]
- 31.Varani J, Perone P, Merfert MG, Moon SE, Larkin D, Stevens MJ. All-trans retinoic acid improves structure and function of diabetic rat skin in organ culture. Diabetes. 2002;51(12):3510–3516. doi: 10.2337/diabetes.51.12.3510. [DOI] [PubMed] [Google Scholar]
- 32.Lateef H, Stevens M, Varani J. Topical pretreatment of diabetic rats with all-trans retinoic acid improves healing of subsequently-induced superficial wounds. Diabetes. 2005;54:855–861. doi: 10.2337/diabetes.54.3.855. [DOI] [PubMed] [Google Scholar]
- 33.van de Sandt JJ, Maas WJ, Doornink PC, Rutten AA. Release of arachidonic and linoleic acid metabolites in skin organ cultures as characteristics of in vitro skin irritancy. Fundam. Appl. Toxicol. 1995;25:20–28. doi: 10.1006/faat.1995.1036. [DOI] [PubMed] [Google Scholar]
- 34.Dame M, Paruchuri T, DaSilva M, Bhagavathula N, Ridder W, Varani J. The Gottingen minipig for assessment of retinoid efficacy in the skin: comparison of topically treated animals with results from organ-cultured skin. In Vitro Develop. Cell Biol. 2009;45(9):551–557. doi: 10.1007/s11626-009-9221-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Varani J, Perone P, Spahlinger DM, et al. Human skin in organ culture and human skin cells (keratinocytes and fibroblasts) in monolayer culture for assessment of chemically induced skin damage. Toxicol. Pathol. 2007;35(5):693–701. doi: 10.1080/01926230701481907. [DOI] [PubMed] [Google Scholar]
- 36.Fisher GJ, Esmann J, Griffiths CE, et al. Cellular, immunologic and biochemical characterization of topical retinoic acid-treated human skin. J. Invest. Dermatol. 1991;96(5):699–707. doi: 10.1111/1523-1747.ep12470632. [DOI] [PubMed] [Google Scholar]
- 37.Marrakchi S, Maibach HI. Sodium lauryl sulfate-induced irritation in the human face: regional and age-related differences. Skin Pharmacol. Physiol. 2006;19(3):177–180. doi: 10.1159/000093112. [DOI] [PubMed] [Google Scholar]
- 38.Cook PW, Mattox PA, Keeble WW, Pittelkow MR, Plowman GD. Human keratinocyte autocrine factor is similar or identical to amphiregulin. Mol. Cell Biol. 1991;11:2547–2557. doi: 10.1128/mcb.11.5.2547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Cook PW, Pittelkow MP, Shipley GD. Growth factor-independent proliferation of normal human neonatal keratinocytes: production of autocrine and paracrine mitogenic factors. J. Cell. Physiol. 1991;146:277–289. doi: 10.1002/jcp.1041460213. [DOI] [PubMed] [Google Scholar]
- 40.Ritti L, Varani J, Kang S, Fisher GJ, Voorhees JJ. Retinoid-induced epidermal hyperplasia is mediated by epidermal growth factor receptor activation via specific induction of its ligands heparin binding-EGF and amphiregulin in human skin in vivo. J. Invest. Dermatol. 2006;126:732–739. doi: 10.1038/sj.jid.5700202. [DOI] [PubMed] [Google Scholar]
- 41.Pasonen-Seppänen SM, Maytin EV, Törrönen KJ, et al. All-trans retinoic acid-induced hyaluronan production and hyperplasia are partly mediated by EGFR signaling in epidermal keratinocytes. J. Invest. Dermatol. 2008;128(4):797–807. doi: 10.1038/sj.jid.5701098. [DOI] [PubMed] [Google Scholar]
- 42.Varani J, Zeigler ME, Dame MK, et al. HB-EGF activation of keratinocyte ErbB receptors mediates epidermal hyperplasia following retinoid treatment: a prominent side effect of retinoid therapy. J. Invest. Dermatol. 2001;117:1335–1341. doi: 10.1046/j.0022-202x.2001.01564.x. [DOI] [PubMed] [Google Scholar]
- 43.Kligman AM, Grove GL, Hirose R, Leyden JJ. Topical tretinoin for photoaged skin. J. Am. Acad. Dermatol. 1986;15(4 Pt 2):836–859. doi: 10.1016/s0190-9622(86)70242-9. [DOI] [PubMed] [Google Scholar]
- 44. Weiss JS, Ellis CN, Headington JT, Tincoff T, Hamilton TA, Voorhees JJ. Topical tretinoin improves photoaged skin. A double-blind vehicle-controlled study. JAMA. 1988;259(4):527–532. • Definitive study of retinoic acid and skin repair.
- 45.Kligman AM, Dogadkina D, Lavker RM. Effects of topical tretinoin on non-sunexposed protected skin of the elderly. J. Am. Acad. Dermatol. 1993;29(1):25–33. doi: 10.1016/0190-9622(93)70147-l. [DOI] [PubMed] [Google Scholar]
- 46.Kafi R, Kwak HSR, Schumacher WE, et al. Improvement of naturally aged skin with vitamin A (retinol) Arch. Dermatol. 2007;143:606–612. doi: 10.1001/archderm.143.5.606. [DOI] [PubMed] [Google Scholar]
- 47.Fisher GJ, Datta SC, Talwar HS, et al. Molecular basis of sun-induced premature skin ageing and retinoid antagonism. Nature. 1996;379(6563):335–339. doi: 10.1038/379335a0. [DOI] [PubMed] [Google Scholar]
- 48.Fisher GJ, Wang ZQ, Datta SC, Varani J, Kang S, Voorhees JJ. Pathophysiology of premature skin aging induced by ultraviolet light. N. Engl. J. Med. 1997;337(20):1419–1428. doi: 10.1056/NEJM199711133372003. [DOI] [PubMed] [Google Scholar]
- 49.Varani J, Warner RL, Phan SH, Datta SC, Fisher GJ, Voorhees JJ. Vitamin A antagonizes decreased cell growth, and elevated collagen-degrading matrix metalloproteinases and stimulates collagen accumulation in naturally-aged human skin. J. Invest. Dermatol. 2000;114:480–486. doi: 10.1046/j.1523-1747.2000.00902.x. [DOI] [PubMed] [Google Scholar]
- 50.Lateef H, Sevens M, Varani J. All-trans retinoic acid suppresses matrix metalloproteinase production/activation and increases collagen synthesis in diabetic skin in organ culture. Am. J. Pathol. 2004;165:167–174. doi: 10.1016/S0002-9440(10)63285-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Griffiths CE, Russman AN, Majmudar G, Singer RS, Hamilton TA, Voorhees JJ. Restoration of collagen formation in photodamaged human skin by tretinoin (retinoic acid) N. Engl. J. Med. 1993;329(8):530–535. doi: 10.1056/NEJM199308193290803. [DOI] [PubMed] [Google Scholar]
- 52.Nickoloff BJ, Mitra RS, Riser BL, Dixit VM, Varani J. Modulation of keratinocyte motility. Correlation with production of extracellular matrix molecules in response to growth promoting and anti-proliferative factors. Am. J. Pathol. 1988;132:543–551. [PMC free article] [PubMed] [Google Scholar]
- 53.Pilcher BK, Dumin JA, Sudbeck BD, Krane SM, Welgus HG, Parks WC. The activity of collagenase-1 is required for keratinocyte migration on a type I collagen matrix. J. Cell Biol. 1997;137(6):1445–1457. doi: 10.1083/jcb.137.6.1445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Varani J, Perone P, Deming MO, et al. Impaired keratinocyte function on matrix metalloproteinase-1 (MMP-1) damaged collagen. Arch. Dermatol. Res. 2009;301(7):497–506. doi: 10.1007/s00403-009-0948-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Jetten AM, George MA, Pettit GR, Herald CL, Rearick JL. Action of phorbol esters, bryostatins and retinoic acid on cholesterol sulfate synthesis: relation to the multistep process of differentiation in human epidermal keratinocytes. J. Invest. Dermatol. 1989;93:108–115. doi: 10.1111/1523-1747.ep12277374. [DOI] [PubMed] [Google Scholar]
- 56.Maas-Szabowski N, Shimotoyodome A, Fusenig NE. Keratinocyte growth regulation in fibroblast cocultures via a double paracrine mechanism. J. Cell. Sci. 1999;112(Pt 12):1843–1853. doi: 10.1242/jcs.112.12.1843. [DOI] [PubMed] [Google Scholar]
- 57.Maas-Szabowski N, Stark HJ, Fusenig NE. Keratinocyte growth regulation in defined organotypic cultures through IL-1-induced keratinocyte growth factor expression in resting fibroblasts. J. Invest. Dermatol. 2000;114(6):1075–1084. doi: 10.1046/j.1523-1747.2000.00987.x. [DOI] [PubMed] [Google Scholar]
- 58.Mulligan MS, Varani J, Dame MK. Role of endothelial cell-leukocyte adhesion molecule-1 (E-selectin in neutrophil-mediated lung injury in rats. J. Clin. Invest. 1991;88:1396–1406. doi: 10.1172/JCI115446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Varani J, Fay K, Perone P. MDI 301, a non-irritating retinoid, induces changes in human skin that underlie repair. Arch. Dermatol. Res. 2007;298(9):439–448. doi: 10.1007/s00403-006-0720-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Moon SE, Dame MK, Remick DR, Elder JT, Varani J. Induction of matrix metalloproteinase-1 (MMP-1) during epidermal invasion of the stroma in human skin organ culture: keratinocyte stimulation of fibroblast MMP-1 production. Br. J. Cancer. 2001;85(10):1600–1605. doi: 10.1054/bjoc.2001.2122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Tavakkol A, Varani J, Elder JT, Zouboulis CC. Maintenance of human skin in organ culture: role for insulin-like growth factor-1 receptor and epidermal growth factor receptor. Arch. Dermatol. Res. 1999;291(12):643–651. doi: 10.1007/s004030050469. [DOI] [PubMed] [Google Scholar]
Websites
- 101.Mat Tech. Corp. www.mattek.com.
- 102.Cell n Tec Advanced Cell Systems. www.cellntec.com.
- 103.Organogenesis, Inc. www.organogenesis.com.
- 104.Smith & Nephew. www.smith-nephew.com.