Abstract
Psychological stressors are known to affect colonic diseases but the mechanisms by which this occurs, and whether probiotics can prevent stressor effects, are not understood. Because inflammatory monocytes that traffic into the colon can exacerbate colitis, we tested whether CCL2, a chemokine involved in monocyte recruitment, was necessary for stressor-induced exacerbation of infectious colitis. Mice were exposed to a social disruption stressor that entails repeated social defeat. During stressor exposure, mice were orally challenged with Citrobacter rodentium to induce a colonic inflammatory response. Exposure to the stressor during challenge resulted in significantly higher colonic pathogen levels, translocation to the spleen, increases in colonic macrophages, and increases in inflammatory cytokines and chemokines. The stressor-enhanced severity of C. rodentium-induced colitis was not evident in CCL2−/− mice, indicating the effects of the stressor are CCL2-dependent. Additionally, we tested whether probiotic intervention could attenuate stressor-enhanced infectious colitis by reducing monocyte/macrophage accumulation. Treating mice with probiotic Lactobacillus reuteri reduced CCL2 mRNA levels in the colon, and attenuated stressor-enhanced infectious colitis. These data demonstrate that probiotic L. reuteri can prevent the exacerbating effects of stressor exposure on pathogen-induced colitis, and suggest that one mechanism by which this occurs is through a down-regulation of the chemokine CCL2.
Keywords: Inflammatory bowel diseases, psychological stress, pathogen induced colitis, macrophage, inflammatory monocyte, CCR2, CCL2, TNF-α, iNOS
Introduction
Chronic idiopathic inflammatory bowel diseases (IBD), including Crohn’s disease and ulcerative colitis, affect millions of Americans(1, 2). The exact etiology of IBD is unknown, but evidence from laboratory animals, as well as human patients, indicate that IBD involves disrupted homeostatic interactions between the microbiota and the mucosal immune system as a result of multiple genetic and environmental signals(3). One environmental factor that contributes to the severity of IBD is psychosocial stress. In animal models, a variety of different types of stressors, ranging from acute restraint to water avoidance stress, have been shown to exacerbate chemically-induced(4), spontaneous(5), and pathogen-induced colitis(6, 7). Moreover, multiple studies involving human participants indicate that perceived life stress is associated with increased disease severity as well as frequency of relapses(8–11). The mechanisms linking the physiological stress response to disease exacerbation, however, are not yet understood.
Macrophages in the large intestine are important contributors to diverse physiological processes, including maintenance of mucosal homeostasis, removal of pathogens, and development of tissue-damaging inflammatory responses(12, 13). These cells are continuously replenished by recruiting monocytes from the bone marrow through chemokine production(12). During periods of quiescence CX3CR1hiLy6Clo “patrolling” monocytes can migrate to the colon where they differentiate into CX3CR1hiLy6Clo resident macrophages that have high phagocytic capacity, but are not prolific producers of inflammatory cytokines(14). A second subset of monocytes, i.e., CX3CR1loLy6Chi inflammatory monocytes, also traffic into the intestines. In the absence of inflammation, these cells differentiate into resident macrophages(14). However, during periods of colitis, CCR2 expression on CX3CR1loLy6C+ inflammatory monocytes is necessary for recruitment from the bone marrow by the chemokine CCL2(15). During pathogen-induced colitis, these CCR2+CX3CR1loLy6Chi cells are necessary for clearance of the pathogen, but are also known to be prolific producers of inflammatory cytokines like tumor necrosis factor (TNF)-α(16). We have previously shown that exposure to a chronic stressor, namely prolonged restraint, resulted in significant increases in colonic TNF-α upon oral challenge with C. rodentium(7). Because of the ability of CCR2+CX3CR1loLy6Chi cells to produce TNF-α, this study determined whether stressor-enhanced severity of C. rodentium-induced colitis were CCL2-dependent.
Recently, we have employed the more ethologically-relevant stressor known as social disruption (SDR), which involves repeated social defeat by an aggressive intruder. Exposure to SDR results in an increase in the number of CD11b+Ly6Chi monocytes in the bone marrow, circulation, and spleen(17, 18). Transcriptomic analysis indicates that these cells have an increased capacity to produce inflammatory cytokines, including TNF-α(19). This is consistent with previous findings demonstrating that exposure to SDR primes splenic CD11b+ cells for enhanced reactivity to microbial stimulation(20). It is not yet known, however, whether exposure to SDR will enhance the ability of these cells to traffic to the colon in response to CCL2 during pathogen-challenge. Thus, mice were exposed to the SDR stressor during oral challenge with the colonic pathogen Citrobacter rodentium.
Citrobacter rodentium is a natural murine colonic pathogen, that induces a colonic inflammatory response resembling colonic inflammation in human IBD patients(21, 22) and includes infiltration of leukocytes, including inflammatory monocytes, production of inflammatory cytokines, such as TNF-α, and mediators, such as iNOS, and disruptions to the epithelial barrier. Interestingly, commensal probiotic microbes, including bacteria in the genus Lactobacillus, have the capacity to modify the progression of C. rodentium-induced colitis(7, 23). Our previous studies demonstrate that exposing animals to different types of stressors significantly disrupts the structure of the intestinal microbiota, with the most consistent finding being reductions in lactobacilli levels(6, 24–26). During exposure to a social disruption (SDR) stressor, the absolute abundance of colonic Lactobacillus reuteri was found to be significantly reduced(27). Because L. reuteri potently suppresses inflammatory responses(28), it is possible the loss of this protective bacteria may contribute to stressor-enhanced infectious colitis. Therefore we hypothesized that administering L. reuteri during stressor exposure would prevent the exacerbating effects of stress on C. rodentium-induced colitis.
Materials and Methods
Animals
Male C57BL/6 mice, 6–8 week of age, were purchased from Charles River Laboratories (Wilmington, MA). Genetically modified, CCL2-deficient mice (strain name: B6.129S4-Ccl2tm1Rol/J) were obtained from Jackson Laboratories (Bar Harbor, ME) and bred in our facility. CCR2-GFP mice were a gift from Eric Pamer(29). All mice were allowed to acclimate for 1 week prior to experimentation. Mice were housed in groups of 3 per cage and kept on a 12 hr light:dark schedule with lights on at 0600. Food and water were available ad libitum. Experiments consisted of 3 mice per group per condition, and data from 3 replicate experiments were used in the analyses for a final sample size of n = 9 per group per time point unless otherwise stated. All experimental procedures were approved by The Ohio State University’s Animal Care and Use Committee.
Social Disruption
Social disruption (SDR) occurred over a 2 hr period between 1630–1830. SDR was initiated by placing an aggressive male mouse into the home cage of the resident mice as previously reported(30). Agonistic behavior between the aggressor and residents were observed to ensure that the aggressor attacked and defeated all residents. After fighting was initiated, aggressors were left in the cages for 2 hrs. At the end of the 2 hr period, the aggressor was removed and resident mice were left undisturbed until the following day when SDR was repeated. The resident mice were exposed to a total of 6, two-hour cycles of SDR.
Infection
Citrobacter rodentium strain DBS120 (pCRP1::Tn5)(31) was grown in Lennox LB Broth overnight at 37°C. Mice were inoculated via oral gavage with 100 μl of bacteria (i.e., 1–3×106 CFU) in PBS, and deprived of food and water for 2 hrs after challenge. Fecal shedding of C. rodentium was determined every 3 days by plating feces from infected mice on MacConkey lactose agar (Becton Dickinson) supplemented with kanamycin (40 μg/ml). Pathogen levels were also determined is spleen homogenates plated in the same fashion as stool samples(7).
Probiotic
Lactobacillus reuteri was purchased from the American Type Culture Collection (ATCC 23272) and grown in Difco Lactobacilli MRS Broth (Becton Dickinson) for approximately 24 hr at 37°C with 5% CO2. The probiotic was washed with PBS, adjusted to 1×109 CFU/ml, and 100 μl was administered to mice via oral gavage (i.e., 1×108 CFU). Mice were given the probiotic after each exposure to the SDR stressor. Vehicle-treated mice were dosed orally with PBS as a control for the daily handling and repeated gavages.
Lamina Propria Lymphocyte Isolation
Colons were extracted from CCR2-GFP mice. Blood was collected for flow cytometry analysis. For colonic tissue, the Lamina Propria Dissociation Kit (Miltenyi-Biotec) protocol was followed for lamina propria lymphocyte (LPL) isolation. In brief, tissue was excised, rinsed, cut into 0.5cm pieces and placed in 20 mL of predigestion solution. Samples were incubated for 20 min at 37°C with rotation. After rinsing, tissue pieces were placed in the gentleMACS C tube with Lamina Propria Dissociation Enzyme Mix, and placed in the gentleMACS Dissociator (Miltenyi-Biotec). Dissociated cells were centrifuged, filtered and resuspended in RPMI/10% FBS at 5×106 cells/mL.
Immunohistochemistry
The entire colon was excised and transected longitudinally. One section was used for semi-quantitative real-time PCR, with the remaining section formalin fixed in 10% neutral buffered saline. These fixed tissues were processed routinely, paraffin embedded, sectioned at 5 μm, and stained with hematoxylin and eosin (H&E). Sections were scored by a board certified veterinary pathologist (Dr. Nicola Parry) who was blinded to experimental groups. Inflammation, hyperplasia, dysplasia, edema, and epithelial defects within intestinal tissue sections were graded on a scale of 0 to 4 at 0.5 intervals as described previously(32). These scores were summed to provide a colonic histological index that was used for statistical analyses. In addition to H&E staining, adjacent sections were stained for F4/80+ macrophages using published protocols(33). Total cell influx was analyzed by digital images of every 5th field of view for the entire length of the colon and quantified using histogram analysis in Adobe Photoshop CS2 software (F4/80+ pixilation)(33).
Cell Culture
The murine colonic epithelial cell line, CMT-93 (ATCC CCL-223), and murine macrophage cell line, RAW 264.7 (ATCC TIB-71), were used to determine the effects of L. reuteri on C. rodentium-induced gene transcription. CMT-93 epithelial cells and RAW 264.7 macrophages were cultured separately according to manufacturer’s guidelines.. Cells were allowed to adhere overnight in 6-well tissue culture treated plates at 1×106 cells/well prior to stimulation. To test the ability of L. reuteri, or its conditioned supernatant, to modulate C. rodentium-induced gene expression, two separate experiments were performed in each cell line. Initially, intact L. reuteri was added to either the CMT-93 or RAW 264.7 monolayer at concentrations ranging from 300:1 to 1:1 (L. reuteri:cell line) for 1 hour prior to C. rodentium challenge. Secondly, conditioned supernatants of L. reuteri were pH-adjusted to 7.0, filter-sterilized, and added to cells lines for 1 hour prior to challenge at a concentration of 5% (v/v). After the 1 hour pre-stimulation, cells were challenged with C. rodentium at a concentration of 100:1 of C. rodentium:cell line for 2 hours. At the end of C. rodentium challenge total RNA was harvested for mRNA analysis by semi-quantitative real-time PCR.
Semi-Quantitative Real-Time PCR
Total RNA was isolated from colonic tissue and in vitro cell lines using Trizol reagent as per manufacturer’s instructions (Invitrogen, Carlsbad, CA), and RNA was reverse transcribed to make complimentary cDNA using a commercially available kit (Promega, Madison, WI). Real-time PCR primers and probes were synthesized by Applied Biosystems and the 5′–3′ sequences are found in Table 1. Real-time PCR reactions were performed and analyzed as previously reported(6). In all cases, 18S was used as a housekeeping gene, and the relative amount of transcript was determined using the comparative cycle threshold (Ct) method as described by the manufacturer.
Table 1.
C. rodentium levels in the spleen
| Day Post-Challenge
|
|||||
|---|---|---|---|---|---|
| Pre | 1 | 6 | 12 | 24 | |
| Vehicle Treatment | |||||
| HCC Control | |||||
| Pathogen Prevalence | 0 | 0 | 0 | 12.5% (1/8) | 0 |
| Colonization | N.D. | N.D. | N.D. | 3.46 | N.D. |
| SDR Stressor | |||||
| Pathogen Prevalence | 0 | 0 | 50% (3/6)* | 62.5% (5/8)* | 0 |
| Colonization | N.D. | N.D. | 3.68±0.16† | 4.03±0.44‡ | N.D. |
| L. reuteri Treatment | |||||
| HCC Control | |||||
| Pathogen Prevalence | 0 | 0 | 0 | 12.5% (1/8) | 0 |
| Colonization | N.D. | N.D. | N.D. | 3.35 | N.D. |
| SDR Stressor | |||||
| Pathogen Prevalence | 0 | 0 | 33% (2/6) | 12.5% (1/8) | 0 |
| Colonization | N.D. | N.D. | 3.72±0.42§ | 3.65 | N.D. |
All data are log(10) CFU/g spleen mass.
N.D. = No Detectable Colonies;
is mean±S.E. from 3 mice;
is mean±S.E. from 5 mice;
is mean±range from 2 mice.
Flow Cytometry
Whole blood and isolated colonic LPL cells from CCR2-GFP mice were stained with PE-Conjugated Ly6C (Clone-AL/21) and APC-Conjugated CD11b+ (Clone-M1/70) (BD Pharmingen, San Jose, CA) at 4°C for 45 minutes. FACS Lysing Solution (BD, San Jose, CA) was added to the cells after incubation for 10 minutes, then a single rinse/spin cycle (400g for 5 min) was performed with FACS Buffer. Samples were analyzed with the BD FACSCalibur dual-laser flow cytometer (BD Immunocytometry Systems, San Jose, CA) with CellQuest Pro. Cells were gated based on forward and side-scatter and CD11b expression and classified based on Ly6C and GFP-CCR2 expression levels.
Statistical Analyses
Differences between SDR Stressor and HCC Control mice for all dependent variables were determined using a two-factor analysis of variance with group (i.e., SDR vs. HCC) and days post-challenge as the independent factors. To facilitate data interpretation, vehicle-treated mice were assessed independently of L. reuteri-treated mice. Protected t-tests using the modified Bon ferroni correction factor were used for post-hoc analyses. Chi-squared tests were performed on pathogen prevalence in the spleen. For all analyses, the level of statistical significance was set at p<.05. SPSS for Windows version 19 (SPSS, Chicago, IL) was used for all analyses.
Results
Stressor-Induced Increases in C. rodentium Levels Are Not Affected by L. reuteri
Exposure to social disruption (SDR) significantly increased the number of C. rodentium shed from the intestines over the 24 day experiment (p<0.01; Fig. 1A). This was noted primarily by increased C. rodentium CFU’s on Days 6 and 12 post-challenge (p<0.05). Importantly, treating mice with L. reuteri did not significantly impact fecal C. rodentium levels in SDR-exposed or non-stressed control mice (Fig. 1A).
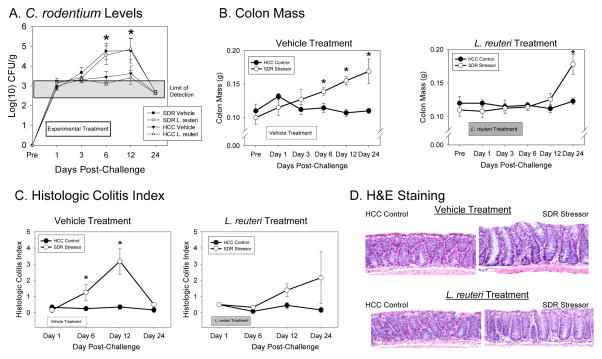
Figure 1.
Exposure to the SDR stressor significantly changed C. rodentium colonization, pathogen-induced increases in colon mass, and histopathology. Mice were exposed to the SDR stressor during oral challenge with 3–5 × 106 CFU of C. rodentium. A). Exposure to the SDR stressor led to increased levels of C. rodentium that could be cultured from the stool during the first 24 days post-challenge. Treatment with L. reuteri did not affect the stressor-induced increase in pathogen challenge. B). Colon mass was significantly increased in mice exposed to the SDR stressor during oral challenge with C. rodentium. This increase did not occur in mice treated with L. reuteri until Day 24 post-challenge. C). Colonic histopathology was significantly increased in mice exposed to the SDR stressor during oral challenge with C. rodentium. This increase was not evident in mice treated with L. reuteri. D). Representative images of H&E stained colonic sections (magnification = 20X). In all cases, the data are the mean ± S.E. * indicate p<.05 vs non-stressed HCC control mice at the same time point.
Lactobacillus reuteri Attenuates the Stressor-Induced Increase in Colonic Pathology
In addition to elevating C. rodentium levels, SDR-exposure during oral challenge with C. rodentium significantly increased colon mass on Days 6, 12, and 24 post-challenge (p<0.05; Fig. 1B). In mice treated with L. reuteri, however, SDR-exposure did not affect colon weight until Day 24 post-challenge (p<0.05; Fig. 1B). In addition to increasing colon weight, SDR-exposure during C. rodentium challenge increased C. rodentium-induced colitis in vehicle-treated mice (Fig. 1C, D). This was a main effect for stressor-exposure (p<0.05), indicating that in general, mice exposed to the stressor had a higher colitis score during the 24 days post-challenge. Protected t-tests were conducted on Days 6 and 12 post-challenge to test the a priori hypothesis that stressor exposure would enhance colitis during time points across the peak of infection. As predicted, C. rodentium-infected, vehicle-treated mice exposed to SDR had significantly higher colitis scores on Days 6 and 12 post-challenge (p<0.05; Fig. 1C, D). These effects were not as evident in L. reuteri-treated mice (Fig. 1C, D), although stressor exposure still resulted in a significant main effect for this group (p<0.05). In this case, however, protected t-tests indicated that there was no difference between colitis scores in non-stressed HCC Control and SDR-exposed mice on Day 6 post-challenge, and only a trend toward a significant difference on Day 12 post-challenge (p=0.08; Fig. 1C).
Lactobacillus reuteri Attenuates Stressor-Induced Increases in Colonic F4/80+ Macrophages During C. rodentium Challenge
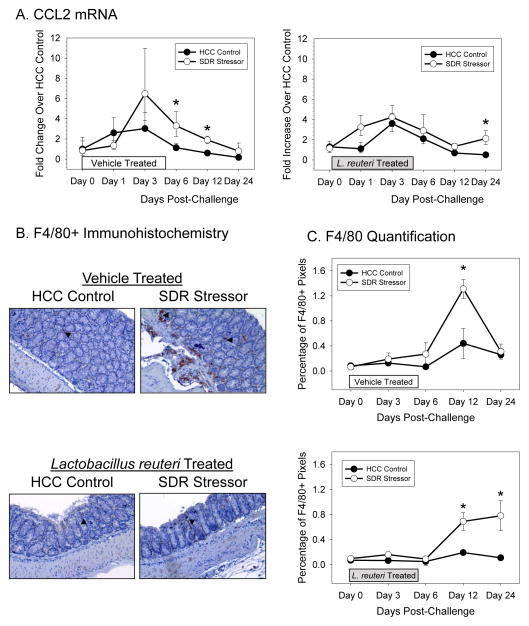
Stressor-exposure during challenge with C. rodentium significantly increased CCL2 mRNA levels in the colons of both vehicle treated mice (p<0.05; Fig. 2A) and L. reuteri-treated mice (p<0.05; Fig. 2A). Exposing vehicle-treated mice to SDR increased CCL2 mRNA on Days 6 and 12 post-challenge (p<0.05), whereas exposing L. reuteri-treated mice to SDR only resulted in increased mRNA on Day 24 post-challenge (p<0.05; Fig. 2A).
Figure 2.
Lactobacillus reuteri treatment attenuated the effects of SDR stressor exposure during oral challenge with C. rodentium on F4/80-positive cells in the colon. A). mRNA expression for the chemokine CCL2 was significantly increased in mice exposed to the SDR stressor during oral challenge with C. rodentium. This increase, however, did not occur in mice treated with L. reuteri until Day 24 post-challenge. B). Representative images of F4/80+ immunoreactivity. Arrows indicate F4/80+ cells. Magnification = 20X. C). Mice exposed to the SDR stressor during oral challenge with C. rodentium had more F4/80 staining in the colon. Although treatment with L. reuteri reduced F4/80 staining, mice exposed to the SDR stressor still had more F4/80+ cells in the colon than did non-stressed control mice. Data are the mean ± S.E. * indicate p<.05 vs. HCC control mice at the same time point.
Exposing vehicle-treated mice to SDR resulted in significantly increased F4/80 staining across the 24 days post-challenge with C. rodentium (p<0.05; Fig. 2B, C), with differences occurring on Day 12 post-challenge (p<0.05). Exposing L. reuteri-treated mice to SDR also increased F4/80+ staining across the 24 day period (p<0.05) with differences between stressed and control mice occurring on Days 12 and 24 (p<0.05; Fig. 2B, C). Although stressor exposure increased F4/80 staining in both vehicle-treated and L. reuteri-treated mice, overall F4/80 staining was lower in L. reuteri-treated mice than it was in vehicle-treated mice (p<0.05; Fig. 2B, C).
Lactobacillus reuteri Treatment Reduces the Elevated Levels of CD11b+Ly6ChiCCR2+Cells in the Blood and the Colonic Lamina Propria of Stressor-Exposed, C. rodentium-Challenged Mice
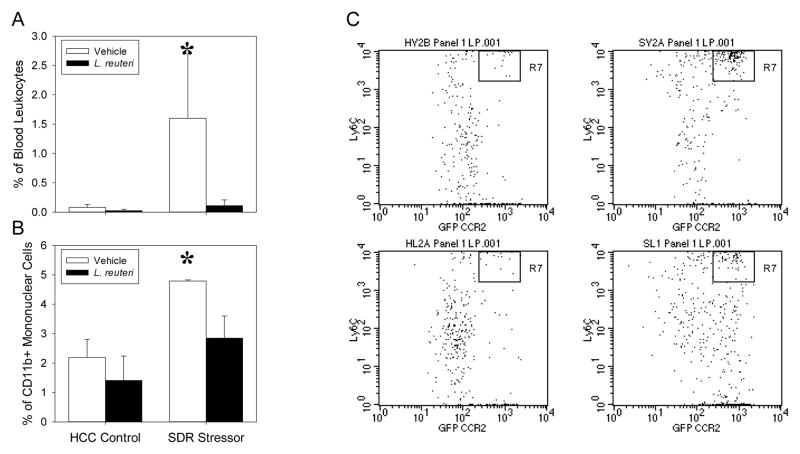
In order to determine whether SDR-exposure increases inflammatory monocyte trafficking to the colon in C. rodentium-challenged mice and if probiotic L. reuteri was capable of abrogating these changes, CD11b+Ly6ChiCCR2+ cells were identified in the colonic lamina propria, as well as the blood, via flow cytometry. Stressor-exposed, C. rodentium-challenged mice had significant increases in the proportion of CD11b+ cells that were Ly6ChiCCR2+ in the blood at 12 days post-infection (p<.05; Fig. 3A). This stressor-induced increase in inflammatory monocytes in the blood was not evident in mice treated with probiotic L. reuteri (Fig. 3A). Likewise, SDR enhanced the C. rodentium-induced increases in CD11b+Ly6ChiCCR2+ inflammatory monocytes within the colonic lamina propria on Day 12 post-infection (p<.05; Fig. 3B, 3C); this effect was not evident in mice gavaged with L. reuteri (Fig. 3C, 3D).
Figure 3.
Lactobacillus reuteri treatment modulates SDR Stressor-induced increases in inflammatory monocytes in the blood and lamina propria at 12 days post-infection with C. rodentium. A). Mice exposed to the SDR Stressor had an increased level of inflammatory monocytes in the blood as compared to HCC Control. This was reduced in L. reuteri-treated mice. Data are expressed as the mean (+/− SE) percentage of blood cells that were CD11b+CCR2+Ly6Chi. B). SDR Stressor exposed mice challenged with C. rodentium had significantly higher levels of lamina propria CD11b+ cells that were also CCR2+Ly6Chi on day 12 post-challenge. Treatment with L. reuteri prevented the stressor-induced increase in CD11b+CCR2+Ly6Chi cells in the lamina propria. C and D). Representative dot plots showing Ly6C and CCR2 staining on cells that were gated based on positive CD11b staining. Representative dot plots from vehicle treated mice are shown in C.) whereas representative dot plots from L. reuteri treated mice are shown in D.). * p<.05 vs. all other groups.
Exposure to SDR During Oral Challenge with C. rodentium Increases Colonic TNF-α and iNOS: L. reuteri Reduces the Stressor Effects
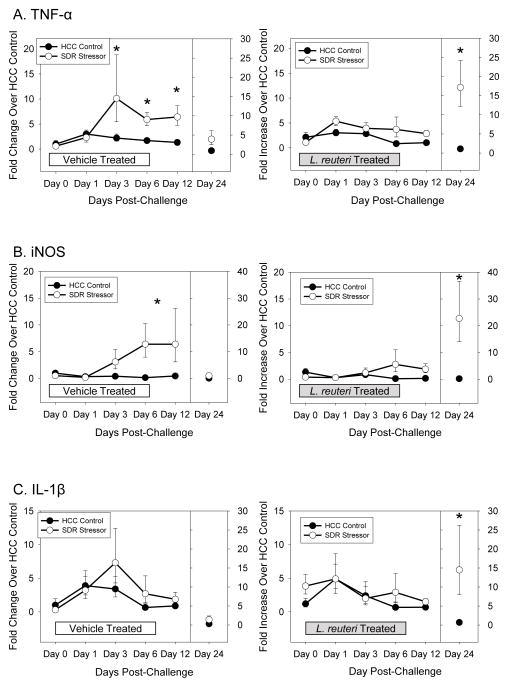
During the 3 weeks post-C. rodentium challenge, gene expression for both TNF-α (p<0.001; Fig. 4A) and iNOS (p<0.001; Fig. 4B) were significantly increased in vehicle-treated, SDR-exposed mice. Post-hoc testing indicated that C. rodentium-induced colonic TNF-α and iNOS mRNA levels were significantly higher in SDR-exposed mice on Days 3, 6, and 12 post-challenge (p<0.05 for each), but were back to baseline levels by Day 24 post-challenge. Although treating mice with L. reuteri reduced both TNF-α (p<0.05; Fig. 4A) and iNOS (p<0.01; Fig. 4B) expression across the 24 days, L. reuteri-treated mice exposed to the SDR stressor still had elevated pathogen-induced TNF-α (p<0.01) and iNOS (p<0.01) when compared to non-stressed controls. But, in this case, differences in TNF-α were due to a significant stressor-induced increase in gene expression on Day 24 post-challenge (p<0.05). In L. reuteri treated mice exposed to the SDR stressor, pathogen-induced iNOS expression was elevated on Days 6, 12, and 24 although the mean level of gene expression on Days 6 and 12 was only a 2 fold increase over non-stressed controls (compared to an approximately 5 fold increase in vehicle treated mice exposed to SDR) (Fig. 4A, B). IL-1β was not significantly increased in the stressor-exposed mice treated with vehicle on any day post C. rodentium challenge (Fig. 4C). However, colonic IL-1β mRNA was increased on Day 24 post-C. rodentium challenge in the mice treated with L. reuteri during stressor exposure (p<0.05; Fig. 4C).
Figure 4.
Lactobacillus reuteri treatment abrogated the SDR stressor-induced increase in inflammatory cytokine and chemokine mRNA levels in the colon after C. rodentium challenge. A). Exposing mice to the SDR stressor during oral challenge with C. rodentium significantly increased TNF-α mRNA levels. Treating mice with L. reuteri prevented the increase in TNF-α until Day 24 post-challenge. B). Exposure to the SDR stressor during C. rodentium challenge increased iNOS mRNA levels. L. reuteri reduced iNOS mRNA, but stressor exposure still resulted in significant increases in iNOS. C). Exposure to the stressor during C. rodentium challenge did not increase IL-1β mRNA levels in the colon. However, mice treated with L. reuteri during exposure to the SDR stressor had elevated levels of IL-1β on Day 24 post-challenge. Data are expressed as a fold increase over uninfected (i.e., Day 0) vehicle-treated HCC control mice and are the mean ± S.E. * indicate p<.05 vs. HCC control on each individual day.
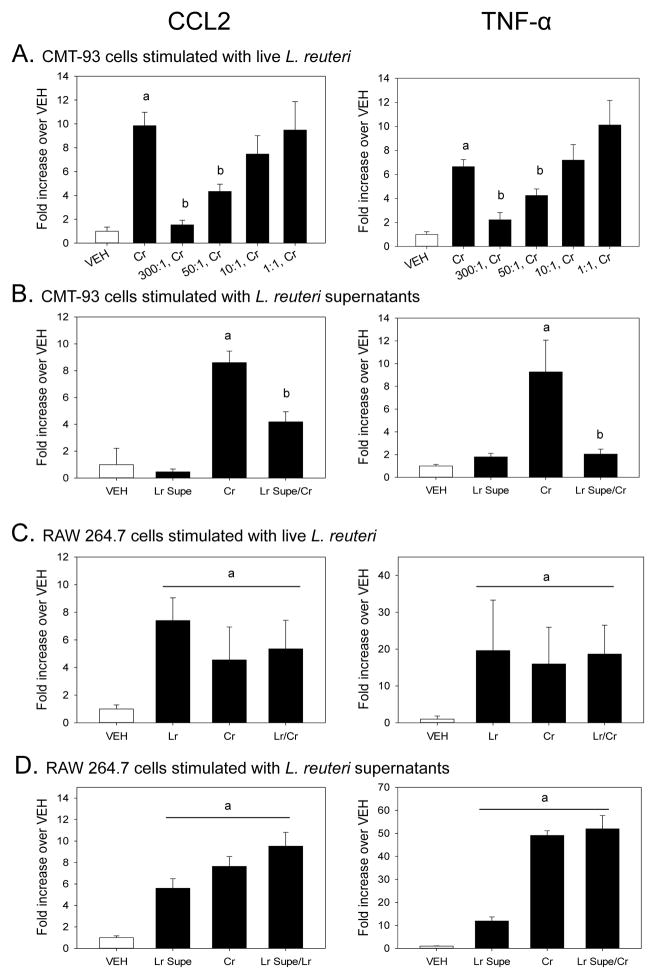
In Vitro Pretreatment With Live L. reuteri, or Conditioned Supernatants, Can Reduce C. rodentium-Enhanced Gene Expression in Colonic Epithelial Cells
Colonic epithelial cell (CMT-93) mRNA expression was determined after a 1 hour pretreatment of live L. reuteri followed by a 2 hour C. rodentium challenge. Epithelial cells treated with live L. reuteri alone did not produce a significant amount of CCL2 as compared to vehicle-treated control cells at any concentration tested (data not shown). There was, however, a significant increase in CCL2 mRNA expression after C. rodentium challenge (p<0.005) (Fig. 5A). This C. rodentium-induced expression of CCL2 was significantly reduced by pretreatment with live L. reuteri at concentrations of 300:1 (p<0.005), and 50:1 (p<0.0001). Live L. reuteri treatment caused a 3-fold increase in TNF-α mRNA expression in CMT-93 cells in only the 300:1 group (data not shown), while stimulation with C. rodentium caused a 14-fold increase in TNF-α mRNA expression compared to VEH-treated cells (p<0.005). When CMT-93 cells were pretreated with live L. reuteri prior to challenge with C. rodentium, pathogen-induced TNF-α gene expression was significantly reduced at concentrations of 300:1 (p<0.005) and 50:1 (p<0.05). Cell-free, pH-adjusted supernatants (5% v/v) from overnight cultures of L. reuteri were used to modulate C. rodentium-induced gene expression. Conditioned supernatants of L. reuteri did not change CMT-93 CCL2 or TNF-α gene expression over that of the vehicle-treated control cells (Fig. 5B), however pretreatment with conditioned supernatants were able to significantly reduce both C. rodentium-enhanced CCL2 (p<0.005) and TNF-α (p<0.005).
Figure 5.
Citrobacter rodentium-induced inflammatory mediator gene expression is modulated by pretreatment with live L. reuteri or its conditioned supernatants in intestinal epithelial cells, but not macrophages, in vitro. The colonic epithelial cell line, CMT-93, or RAW 264.7 macrophages were pretreated with either live, intact L. reuteri or pH-adjusted, filter sterilized L. reuteri supernatants for 1 hour prior to challenge with C. rodentium. At the end of the 2 hour pathogen challenge, total RNA was isolated in order to quantify mRNA expression by Real Time PCR. A). Epithelial CCL2 and TNF-α gene expression was significantly increased by 2 hours of C. rodentium challenge as compared to vehicle (PBS) control. Pretreatment with live L. reuteri significantly reduced C. rodentium-induced CCL2 and TNF-α mRNA expression at ratios of 300:1 and 50:1 (L. reuteri:CMT-93). a = treatment vs. VEH, p<0.005. b = Lr/Cr vs Cr, p<0.05. B). Conditioned supernatants from L. reuteri significantly reduce CCL2 and TNF-α mRNA expression in CMT-93 epithelial cells. a = treatment vs. VEH, p<0.05. b = Lr/Cr vs Cr, p<0.005. C). Live L. reuteri, C. rodentium, and L. reuteri pretreatment prior to pathogen challenge significantly enhances both CCL2 and TNF-α mRNA expression in RAW 264.7 macrophages. a = treatment vs. VEH, p<0.05. D). Conditioned supernatants also significantly enhance CCL2 and TNF-α mRNA expression in RAW 264.7 macrophages and fail to reduce C. rodentium-induced mRNA expression. a = treatment vs. VEH, p<0.001. Lr, L. reuteri treatment alone. Cr, C. rodentium treatment alone. Lr/Cr, L. reuteri pretreatment prior to C. rodentium challenge. Lr Supe, Cell-free, pH adjusted L. reuteri supernatant (5% v/v). n = 8/treatment. Data are the mean ± standard error.
Live L. reuteri or Conditioned Supernatants Do Not Attenuate C. rodentium-Enhanced Gene Expression in RAW 264.7 Macrophages
Stimulation with either live L. reuteri or C. rodentium caused a significant increase in CCL2 gene expression (p<0.05, Fig. 5C). Pretreatment of RAW 264.7 cells with live L. reuteri prior to challenge with C. rodentium failed to reduce C. rodentium-induced CCL2 expression. Similar to CCL2 expression, TNF-α mRNA expression was significantly enhanced by stimulation with live L. reuteri (p<0.001) and C. rodentium (p<0.001). There was no change in TNF-α gene expression when C. rodentium-challenged macrophages were pretreated with live L. reuteri. Treatment with L. reuteri conditioned supernatants alone was enough to significantly enhance CCL2 mRNA expression in RAW 264.7 macrophages to near similar levels as pathogen stimulation (p<0.0001, Fig. 5D). Pretreatment of macrophages with L. reuteri conditioned supernatants prior to challenge with C. rodentium did not reduce CCL2 gene expression as it did in CMT-93 cells. There was a significant increase in TNF-α mRNA gene expression in macrophages treated with L. reuteri supernatants only (p<0.0001), however this increase was significantly lower than that of pathogen-induced TNF-α expression. There was no change in C. rodentium-induced TNF-α mRNA expression when macrophages were pretreated with conditioned supernatants of L. reuteri.
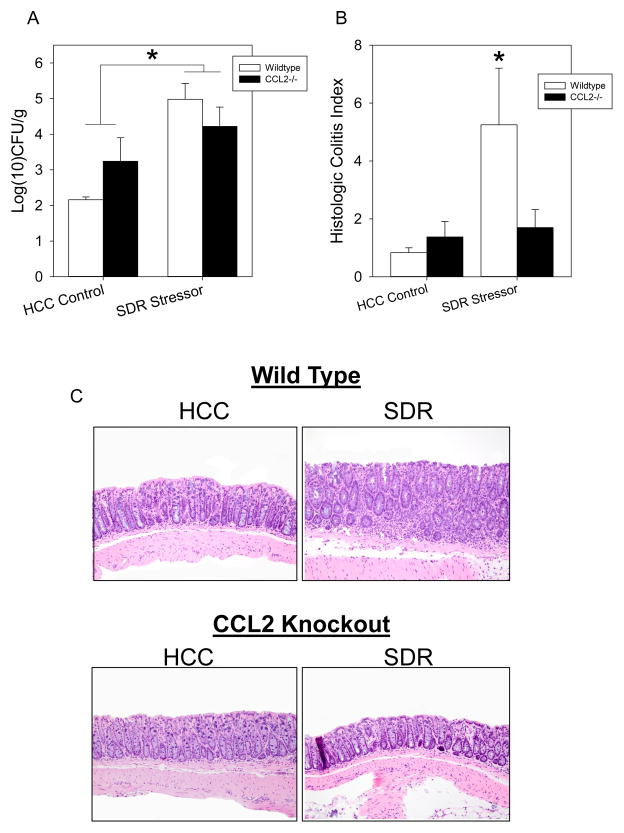
Stressor-Exposed, CCL2-Deficient Mice Have Reduced Colonic Histopathology Despite Increases in Pathogen Burden
Both C57BL/6 wild type (WT) and CCL2-deficient (CCL2−/−) mice were exposed to SDR and challenged with C. rodentium. There was a significant increase in C. rodentium burden in stressor exposed WT and CCL2−/− mice over their non-stressed counterparts on Day 12 post-challenge (p<0.05; Figure 6A). Colonic histopathology in C. rodentium-challenged mice was significantly increased in SDR-exposed, WT mice on Day 12 post-challenge (p<0.05; Fig. 6B); this effect was not evident in CCL2−/− mice (Fig. 6B).
Figure 6.
Stressor-enhanced, C. rodentium-induced infectious colitis is negated in CCL2−/− mice. Wild type (WT) and CCL2−/− mice were orally challenged with 3 × 106 CFU of C. rodentium during exposure to the SDR stressor. Social disruption continued for 5 days post infection. A). Exposure to the SDR stressor significantly increased C. rodentium colonization in WT and CCL2−/− mice. *p<0.05 HCC vs SDR. B). Stressor exposure significantly increased total pathology on Day 12 post-challenge. *p<0.001 SDR WT vs HCC WT. C). Representative images of H&E stained colonic sections (magnification = 20X). In all cases, the data are the mean ± S.E.
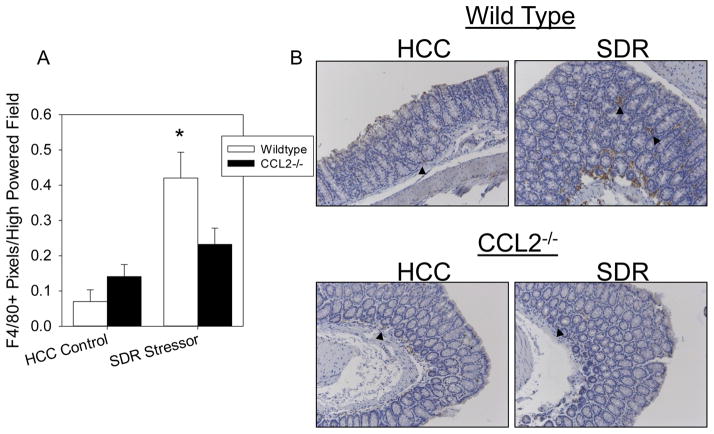
Stressor-Enhanced, C. rodentium-Induced Colonic Macrophage Accumulation is Reduced in CCL2−/− Mice
The colons of SDR-exposed, WT mice had a significant increase in macrophage accumulation on Day 12 post-C. rodentium challenge (p<0.05; Fig. 7A). This stressor-enhanced effect on colonic macrophage accumulation was not apparent in CCL2−/− mice exposed to the SDR stressor during C. rodentium challenge (Fig. 7B).
Figure 7.
Stressor exposure enhances pathogen-induced colonic macrophage accumulation in wild type mice. A). Colons of SDR-exposed WT mice had a significant increase in F4/80+ cells on day 12 post-challenge. This effect was reduced in CCL2−/−, stressor exposed mice. B). Representative images of F4/80+ immunoreactivity. Arrows indicate F4/80+ cells. Magnification = 20X. Data are the mean ± standard error. *p<0.05 SDR WT vs HCC WT.
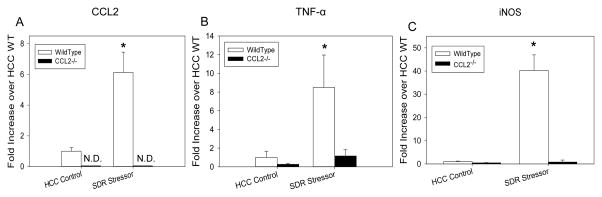
Stressor-Enhanced, C. rodentium-Induced Colonic Inflammatory Mediator Gene Expression is Negated in CCL2−/− Mice
On Day 12 post C. rodentium-challenge, there was a significant increase in CCL2 mRNA expression in SDR-exposed WT mice (p<0.001; Fig. 8A). Colonic CCL2 gene expression was not detected in CCL2−/− mice. Colonic TNF-α mRNA expression was also significantly increased on Day 12 post-C. rodentium challenge in SDR-exposed mice (p<0.001; Fig. 8B), however TNF-α mRNA levels were higher in WT mice compared to CCL2−/− mice (p<0.005; Fig. 8B). Similar to previous data, iNOS mRNA expression was also significantly increased in SDR-exposed WT mice on Day 12 post-C. rodentium challenge (p<0.01; Fig. 8C). This stressor-enhanced severity of C. rodentium-induced iNOS expression was diminished in CCL2−/− mice (Fig. 8C).
Figure 8.
Social disruption enhances C. rodentium-induced colonic gene expression in mice with intact CCL2. Stressor exposure significantly increased colonic mRNA expression of A) CCL2, B) TNF-α, and C) iNOS as compared to non-stressed controls on Day 12 post-challenge which was ablated in CCL2−/− mice. ND = Not Detectable. *p<0.01 SDR WT vs HCC WT. In TNF-α figure, a = HCC vs SDR, p<0.001. Data are the mean ± standard error.
Stressor-Induced Increases in C. rodentium Translocation are Reduced by L. reuteri
In vehicle-treated non-stressed control mice, C. rodentium was rarely found in the spleen. In fact, across the 24 day experiment, only 1 of the non-stressed control mice had detectable levels of C. rodentium in the spleen (Fig. 9A). In contrast, on Day 6 (p<0.05) and Day 12 (p<0.05) post-C. rodentium challenge, there were significant increases in the occurrence of C. rodentium in the spleens stressor-exposed mice (Table 1). Fifty percent of stressor-exposed mice had detectable levels of C. rodentium on Day 6 post-challenge, and 62.5% had detectable levels of C. rodentium on Day 12 post-challenge. As with previous measures, treating mice with L. reuteri reduced stressor-enhanced translocation of C. rodentium to the spleen with only 33% of stressor-exposed mice having detectable levels on Day 6 and 12.5% having detectable levels on Day 12. Although stressor exposure increased the likelihood that C. rodentium would translocate from the colon and be isolated from the spleen, all mice that were found to have C. rodentium in the spleen had similar levels of C. rodentium per gram of spleen mass (Table 1).
Stressor-Enhanced Pathogen Translocation to the Spleen is Diminished in CCL2−/− Mice
Stressor-exposed WT mice had a significant increase in spleen mass during C. rodentium challenge (p<0.05; Table 2), a phenomenon which was negated in SDR-exposed CCL2−/− mice. The significant increase in spleen mass in SDR-exposed WT mice coincided with a significant increase in the likelihood of detecting C. rodentium in the spleen. Stressor exposure failed to increase C. rodentium translocation in CCL2−/− mice.
Table 2.
C. rodentium levels in the spleens of CCL2−/− mice
| Spleen Mass(g)
|
Prevalence(%)
|
Colonization
|
|
|---|---|---|---|
| Wild Type | |||
| HCC Control | 0.079±0.006 | 0 (0/3) | N.D. |
| SDR Stressor | 0.138±0.018* | 67 (4/6)* | 6133±5800 |
| CCL2-deficient | |||
| HCC Control | 0.061±0.005 | 25 (1/4) | 150±50 |
| SDR Stressor | 0.072±0.007 | 17 (1/6) | 100±5 |
Colonization data are CFU/spleen.
N.D. = No Detectable Colonies.
p<0.05 vs HCC Control.
Discussion
Murine infection with C. rodentium results in colonic inflammation that resembles intestinal inflammation found in patients with IBD(21, 34). In IBD patients, psychological stress may increase symptom flares or cause relapse from remission(35). Despite this realization, the mechanisms by which this occurs, and whether probiotic microbes may ameliorate stressor-induced exacerbation, are not yet known. In our study, infecting mice with a lower dose of C. rodentium resulted in low pathogen colonization, and little evidence of colonic inflammation. However, exposing mice to a social stressor significantly increased the colonic inflammatory response to C. rodentium, demonstrating the relevance of the model system to the observation that psychosocial stress in humans can increase the severity of IBD(9–11). The overall histologic colitis index was significantly increased on Days 6 and 12 post-challenge in stressor-exposed mice.
Mice, as well as humans, exposed to social stress have an increase in bone marrow-derived Ly6ChiCD11b+ monocytes in circulation(19). Both gene expression profiling and ex vivo assays demonstrate that these cells have an increased capacity to produce inflammatory cytokines (e.g., TNF-α) and reactive oxygen and nitrogen intermediates (e.g., iNOS and peroxynitrite) upon stimulation with microbial antigen(19, 30). During infection, innate immune cells, such as CD11b+Ly6Chi inflammatory monocytes are recruited from the bone marrow in part by chemokines, such as CCL2(15). This chemokine was significantly elevated in the stressor-exposed mice on Days 3, 6, and 12 post-C. rodentium challenge. Consistent with these data, F4/80+ macrophages and CD11b+Ly6ChiCCR2+ cells were increased in the colon during the first two weeks post-challenge. This finding is important, because previous studies have shown that newly recruited Ly6ChiCCR2+ inflammatory monocytes can differentiate into pro-inflammatory macrophages, which are the primary producers of TNF-α(16, 36), and other mediators, such as iNOS, during C. rodentium challenge(37). It is well recognized that production of TNF-α or reactive nitrogen intermediates (produced as a result of expressing the iNOS enzyme) helps to control microbial pathogen levels in the colon(38, 39). However, excessive production results in tissue damage in both experimental colitis and in humans with inflammatory bowel diseases(38, 39).
The exacerbating effects of stressor exposure on the severity of pathogen-induced colitis were not apparent in CCL2−/− mice. These mice had low levels of F4/80+ macrophages in the colon at the peak of C. rodentium challenge. Additionally, the ability of the stressor to exacerbate TNF-α and iNOS mRNA was prevented in CCL2−/− mice further demonstrating the importance of CCL2 in stressor-induced exacerbation of pathogen-induced colitis. The production of CCL2 can be influenced by probiotic microbes, and our data demonstrated that L. reuteri (strain ATCC 23272) reduces the ability of murine colonic epithelial cells to produce CCL2. While other strains of L. reuteri are able to reduce cytokine and chemokine production from monocytic cells(40) the strain of L. reuteri used in the current study was unable to prevent cytokine and chemokine production by RAW 264.7 macrophage cell line or by primary splenic CD11b+ monocytes/macrophages (data not shown).
The ability of L. reuteri to reduce colonic cytokine and chemokine production was also evident in vivo. Administering L. reuteri on each day of stressor-exposure significantly reduced the effects of the stressor on the severity of C. rodentium-induced colitis. Overall colitis scores were significantly reduced, as were the accumulation of macrophages and the elevated TNF-α and iNOS mRNA. We hypothesize that L. reuteri-secreted factors are able to stabilize colonic epithelial cells from overexpressing proinflammatory cytokines and chemokines following stressor-exposure during C. rodentium challenge. In previous studies, it was demonstrated that a cocktail of two rat (R2LC and JCM 5869) and two human (ATCC PTA 4659 and ATCC 55730) L. reuteri isolates were able to reduce chemically-induced colitis via the reduction of P-selectin-associated leukocyte-endothelial cell interactions(41). Thus, it is possible the reduction of these adhesion molecules, coupled with the reduction of colonic CCL2, contributes to the reduction of stressor-enhanced colonic macrophage accumulation, thereby reducing stressor-exacerbated colitis.
Stressor exposure during C. rodentium challenge increased the ability of C. rodentium to translocate from the lumen of the intestines to abdominal organs. Citrobacter rodentium does not possess the pathogenicity factors necessary to invade across epithelial cells(42, 43). Thus, paracellular permeability of the epithelial barrier must be enhanced for the pathogen to migrate from the colon to the interior of the body. Stressor-exposed mice during C. rodentium challenge were more likely to have detectable levels of C. rodentium in the spleen, indicating that epithelial permeability had been increased in stressor-exposed mice. Mice treated with L. reuteri during exposure to the SDR stressor did not have an increase in C. rodentium in the spleen after oral challenge. While it has been documented that L. reuteri can enhance epithelial barrier integrity even when not reducing cytokine production(7, 44), it is likely that the increased barrier permeability in stressor-exposed mice was related to colonic inflammation. There was no evidence that stressor exposure enhanced barrier permeability in CCL2−/− mice, suggesting that leukocyte recruitment was involved in the disruption of the epithelial barrier.
The beneficial effects of L. reuteri occurred even though L. reuteri did not significantly change C. rodentium levels in the colon. This is important, because, L. reuteri can produce a compound in the presence of glycerol, called reuterin, that can kill enteric pathogens in vitro(45, 46). The results of this study indicate that the anti-inflammatory effects of L. reuteri are not due to decreases in colonic C. rodentium levels. As reuterin production by L. reuteri was not induced within our studies we do not currently believe this is the mechanism by which stressor-enhanced infectious colitis is reduced. Rather, the results indicate that L. reuteri has a more direct effect on the colonic inflammatory response. Our results are consistent with previous studies indicating that L. reuteri attenuates colitis in germfree mice challenged with the closely related pathogen, enterohemorrhagic E. coli, even though pathogen levels were not reduced until 3 weeks post-challenge(28).
The effects of L. reuteri were not permanent. In this study, probiotic L. reuteri was only given through Day 5 post-challenge (i.e., through the last day of stressor exposure) to test the hypothesis that lactobacilli could prevent stressor effects. However, by Day 24 post-challenge, which is 19 days after terminating L. reuteri treatment, stressor-exposed, C. rodentium-challenged mice that were treated with L. reuteri had more pathology in the colon than did mice in any other group, including vehicle-treated, stressor-exposed mice. It is not clear why pathology was higher in these mice, but others have demonstrated that probiotic lactobacilli, including L. reuteri, do not colonize the host for long periods of time(47, 48). This may be partly dependent upon host factors, because human isolates of L. reuteri (such as the currently used ATCC 23272) do not adhere to murine colonic tissue(49). It is possible that rodent lactobacilli isolates that colonize the murine gastrointestinal (GI) tract(50), would persist for longer periods of time and prevent the observed increase in colitis even after probiotic termination. This hypothesis will be tested in future studies
Stressor exposure is recognized to exacerbate gastrointestinal illnesses, but the mechanisms by which this occurs are not well understood. The use of well-defined animal stressors and pathogen challenge to induce infectious colitis offers a means to begin understanding how stress can impact GI illness. When considered together, this study indicates that stressor exposure leads to increased production of CCL2 in the colon, which is needed to recruit inflammatory monocytes from the bone marrow(15). In the colon, these inflammatory monocytes are prolific producers of inflammatory cytokines and mediators, like TNF-α and iNOS, which are recognized to have both protective and tissue-damaging effects in the colon. This increased production of CCL2 can be reduced by probiotic L. reuteri, which also prevents stressor-induced exacerbation of the severity of C. rodentium colitis. While it is not clear how L. reuteri prevents the effects of stressor exposure on the severity of colonic inflammation, it is interesting to note that stressor exposure reduces commensal L. reuteri levels in the colonic mucosa(27). If providing probiotic L. reuteri prevents this stressor-induced reduction in commensal lactobacilli, it would suggest that commensal microbes play a role in stressor-induced exacerbation of colonic inflammation, and would provide for a mechanism by which probiotics exert their beneficial effects.
Acknowledgments
Supported by NIH grant AT006552-01A1 (M.T.B), P30-ES002109 (J.G.F), P30-CA016058, and T32-DE014320.
References
- 1.Farrell D, Savage E. Symptom burden: a forgotten area of measurement in inflammatory bowel disease. International journal of nursing practice. 2012;18(5):497–500. doi: 10.1111/j.1440-172X.2012.02063.x. Epub 2012/09/27. [DOI] [PubMed] [Google Scholar]
- 2.Sicilia B, Vicente R, Gomollon F. Epidemiology of inflammatory bowel disease: controversies in classical epidemiology. Acta gastroenterologica Latinoamericana. 2009;39(2):135–45. Epub 2009/08/12. Enfermedad de Crohn y colitis ulcerosa: discusion de la epidemiologia clasica. [PubMed] [Google Scholar]
- 3.Sartor RB. Microbial influences in inflammatory bowel diseases. Gastroenterology. 2008;134(2):577–94. doi: 10.1053/j.gastro.2007.11.059. Epub 2008/02/05. [DOI] [PubMed] [Google Scholar]
- 4.Reber SO, Obermeier F, Straub RH, Falk W, Neumann ID. Chronic intermittent psychosocial stress (social defeat/overcrowding) in mice increases the severity of an acute DSS-induced colitis and impairs regeneration. Endocrinology. 2006;147(10):4968–76. doi: 10.1210/en.2006-0347. Epub 2006/06/24. [DOI] [PubMed] [Google Scholar]
- 5.Savignac HM, Hyland NP, Dinan TG, Cryan JF. The effects of repeated social interaction stress on behavioural and physiological parameters in a stress-sensitive mouse strain. Behavioural brain research. 2011;216(2):576–84. doi: 10.1016/j.bbr.2010.08.049. Epub 2010/09/10. [DOI] [PubMed] [Google Scholar]
- 6.Bailey MT, Dowd SE, Parry NM, Galley JD, Schauer DB, Lyte M. Stressor exposure disrupts commensal microbial populations in the intestines and leads to increased colonization by Citrobacter rodentium. Infection and immunity. 2010;78(4):1509–19. doi: 10.1128/IAI.00862-09. Epub 2010/02/11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mackos AR, Eubank TD, Parry NM, Bailey MT. Probiotic Lactobacillus reuteri Attenuates the Stressor-Enhanced Severity of Citrobacter rodentium Infection. Infection and immunity. 2013;81(9):3253–63. doi: 10.1128/IAI.00278-13. Epub 2013/06/27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mackner LM, Crandall WV. Psychological factors affecting pediatric inflammatory bowel disease. Current opinion in pediatrics. 2007;19(5):548–52. doi: 10.1097/MOP.0b013e3282ef4426. Epub 2007/09/22. [DOI] [PubMed] [Google Scholar]
- 9.Mackner LM, Clough-Paabo E, Pajer K, Lourie A, Crandall WV. Psychoneuroimmunologic factors in inflammatory bowel disease. Inflammatory bowel diseases. 2011;17(3):849–57. doi: 10.1002/ibd.21430. Epub 2010/08/20. [DOI] [PubMed] [Google Scholar]
- 10.Sajadinejad MS, Asgari K, Molavi H, Kalantari M, Adibi P. Psychological issues in inflammatory bowel disease: an overview. Gastroenterology research and practice. 2012;2012:106502. doi: 10.1155/2012/106502. Epub 2012/07/11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mardini HE, Kip KE, Wilson JW. Crohn’s disease: a two-year prospective study of the association between psychological distress and disease activity. Digestive diseases and sciences. 2004;49(3):492–7. doi: 10.1023/b:ddas.0000020509.23162.cc. Epub 2004/05/14. [DOI] [PubMed] [Google Scholar]
- 12.Mowat AM, Bain CC. Mucosal macrophages in intestinal homeostasis and inflammation. Journal of innate immunity. 2011;3(6):550–64. doi: 10.1159/000329099. Epub 2011/10/26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sagaert X, Tousseyn T, De Hertogh G, Geboes K. Macrophage-related diseases of the gut: a pathologist’s perspective. Virchows Archiv: an international journal of pathology. 2012;460(6):555–67. doi: 10.1007/s00428-012-1244-9. Epub 2012/05/12. [DOI] [PubMed] [Google Scholar]
- 14.Bain CC, Scott CL, Uronen-Hansson H, Gudjonsson S, Jansson O, Grip O, et al. Resident and pro-inflammatory macrophages in the colon represent alternative context-dependent fates of the same Ly6Chi monocyte precursors. Mucosal immunology. 2013;6(3):498–510. doi: 10.1038/mi.2012.89. Epub 2012/09/20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Serbina NV, Shi C, Pamer EG. Monocyte-mediated immune defense against murine Listeria monocytogenes infection. Advances in immunology. 2012;113:119–34. doi: 10.1016/B978-0-12-394590-7.00003-8. Epub 2012/01/17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Platt AM, Bain CC, Bordon Y, Sester DP, Mowat AM. An independent subset of TLR expressing CCR2-dependent macrophages promotes colonic inflammation. Journal of immunology (Baltimore, Md: 1950) 2010;184(12):6843–54. doi: 10.4049/jimmunol.0903987. Epub 2010/05/21. [DOI] [PubMed] [Google Scholar]
- 17.Wohleb ES, Powell ND, Godbout JP, Sheridan JF. Stress-induced recruitment of bone marrow-derived monocytes to the brain promotes anxiety-like behavior. The Journal of neuroscience: the official journal of the Society for Neuroscience. 2013;33(34):13820–33. doi: 10.1523/JNEUROSCI.1671-13.2013. Epub 2013/08/24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Stark JL, Avitsur R, Padgett DA, Campbell KA, Beck FM, Sheridan JF. Social stress induces glucocorticoid resistance in macrophages. American journal of physiology Regulatory, integrative and comparative physiology. 2001;280(6):R1799–805. doi: 10.1152/ajpregu.2001.280.6.R1799. Epub 2001/05/17. [DOI] [PubMed] [Google Scholar]
- 19.Powell ND, Sloan EK, Bailey MT, Arevalo JM, Miller GE, Chen E, et al. Social stress up-regulates inflammatory gene expression in the leukocyte transcriptome via beta-adrenergic induction of myelopoiesis. Proceedings of the National Academy of Sciences of the United States of America. 2013 doi: 10.1073/pnas.1310655110. Epub 2013/09/26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bailey MT, Engler H, Powell ND, Padgett DA, Sheridan JF. Repeated social defeat increases the bactericidal activity of splenic macrophages through a Toll-like receptor-dependent pathway. American journal of physiology Regulatory, integrative and comparative physiology. 2007;293(3):R1180–90. doi: 10.1152/ajpregu.00307.2007. Epub 2007/06/29. [DOI] [PubMed] [Google Scholar]
- 21.Mundy R, MacDonald TT, Dougan G, Frankel G, Wiles S. Citrobacter rodentium of mice and man. Cellular microbiology. 2005;7(12):1697–706. doi: 10.1111/j.1462-5822.2005.00625.x. Epub 2005/11/29. [DOI] [PubMed] [Google Scholar]
- 22.Eckmann L. Animal models of inflammatory bowel disease: lessons from enteric infections. Annals of the New York Academy of Sciences. 2006;1072:28–38. doi: 10.1196/annals.1326.008. Epub 2006/10/24. [DOI] [PubMed] [Google Scholar]
- 23.Rodrigues DM, Sousa AJ, Johnson-Henry KC, Sherman PM, Gareau MG. Probiotics are effective for the prevention and treatment of Citrobacter rodentium-induced colitis in mice. The Journal of infectious diseases. 2012;206(1):99–109. doi: 10.1093/infdis/jis177. Epub 2012/03/21. [DOI] [PubMed] [Google Scholar]
- 24.Bailey MT, Coe CL. Maternal separation disrupts the integrity of the intestinal microflora in infant rhesus monkeys. Developmental psychobiology. 1999;35(2):146–55. Epub 1999/08/26. [PubMed] [Google Scholar]
- 25.Bailey MT, Lubach GR, Coe CL. Prenatal stress alters bacterial colonization of the gut in infant monkeys. Journal of pediatric gastroenterology and nutrition. 2004;38(4):414–21. doi: 10.1097/00005176-200404000-00009. Epub 2004/04/16. [DOI] [PubMed] [Google Scholar]
- 26.Bailey MT. Psychological stress, immunity, and the effects on the indigenous microflora. In: Lyte M, Freestone PP, editors. Microbial Endocrinology. New York: Springer; 2010. pp. 191–212. [Google Scholar]
- 27.Galley JD, Nelson MC, Yu Z, Dowd SE, Walter J, Kumar PS, et al. Exposure to a social stressor disrupts the community structure of the colonic mucosa-associated microbiota. BMC microbiology. 2014;14:189. doi: 10.1186/1471-2180-14-189. Epub 2014/07/17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Eaton KA, Honkala A, Auchtung TA, Britton RA. Probiotic Lactobacillus reuteri ameliorates disease due to enterohemorrhagic Escherichia coli in germfree mice. Infection and immunity. 2011;79(1):185–91. doi: 10.1128/IAI.00880-10. Epub 2010/10/27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Serbina NV, Hohl TM, Cherny M, Pamer EG. Selective expansion of the monocytic lineage directed by bacterial infection. Journal of immunology (Baltimore, Md: 1950) 2009;183(3):1900–10. doi: 10.4049/jimmunol.0900612. Epub 2009/07/15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Allen RG, Lafuse WP, Powell ND, Webster Marketon JI, Stiner-Jones LM, Sheridan JF, et al. Stressor-induced increase in microbicidal activity of splenic macrophages is dependent upon peroxynitrite production. Infection and immunity. 2012;80(10):3429–37. doi: 10.1128/IAI.00714-12. Epub 2012/07/25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Schauer DB, Falkow S. The eae gene of Citrobacter freundii biotype 4280 is necessary for colonization in transmissible murine colonic hyperplasia. Infection and immunity. 1993;61(11):4654–61. doi: 10.1128/iai.61.11.4654-4661.1993. Epub 1993/11/01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Borenshtein D, Schlieper KA, Rickman BH, Chapman JM, Schweinfest CW, Fox JG, et al. Decreased expression of colonic Slc26a3 and carbonic anhydrase iv as a cause of fatal infectious diarrhea in mice. Infection and immunity. 2009;77(9):3639–50. doi: 10.1128/IAI.00225-09. Epub 2009/06/24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Eubank TD, Roberts RD, Khan M, Curry JM, Nuovo GJ, Kuppusamy P, et al. Granulocyte macrophage colony-stimulating factor inhibits breast cancer growth and metastasis by invoking an anti-angiogenic program in tumor-educated macrophages. Cancer research. 2009;69(5):2133–40. doi: 10.1158/0008-5472.CAN-08-1405. Epub 2009/02/19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Borenshtein D, McBee ME, Schauer DB. Utility of the Citrobacter rodentium infection model in laboratory mice. Current opinion in gastroenterology. 2008;24(1):32–7. doi: 10.1097/MOG.0b013e3282f2b0fb. Epub 2007/11/29. [DOI] [PubMed] [Google Scholar]
- 35.Bernstein CN, Singh S, Graff LA, Walker JR, Miller N, Cheang M. A prospective population-based study of triggers of symptomatic flares in IBD. The American journal of gastroenterology. 2010;105(9):1994–2002. doi: 10.1038/ajg.2010.140. Epub 2010/04/08. [DOI] [PubMed] [Google Scholar]
- 36.Zuo L, Huang Z, Dong L, Xu L, Zhu Y, Zeng K, et al. Targeting delivery of anti-TNFalpha oligonucleotide into activated colonic macrophages protects against experimental colitis. Gut. 2010;59(4):470–9. doi: 10.1136/gut.2009.184556. Epub 2009/12/03. [DOI] [PubMed] [Google Scholar]
- 37.Vallance BA, Deng W, De Grado M, Chan C, Jacobson K, Finlay BB. Modulation of inducible nitric oxide synthase expression by the attaching and effacing bacterial pathogen citrobacter rodentium in infected mice. Infection and immunity. 2002;70(11):6424–35. doi: 10.1128/IAI.70.11.6424-6435.2002. Epub 2002/10/16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Goncalves NS, Ghaem-Maghami M, Monteleone G, Frankel G, Dougan G, Lewis DJ, et al. Critical role for tumor necrosis factor alpha in controlling the number of lumenal pathogenic bacteria and immunopathology in infectious colitis. Infection and immunity. 2001;69(11):6651–9. doi: 10.1128/IAI.69.11.6651-6659.2001. Epub 2001/10/13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lebeis SL, Sherman MA, Kalman D. Protective and destructive innate immune responses to enteropathogenic Escherichia coli and related A/E pathogens. Future microbiology. 2008;3(3):315–28. doi: 10.2217/17460913.3.3.315. Epub 2008/05/29. [DOI] [PubMed] [Google Scholar]
- 40.Lin YP, Thibodeaux CH, Pena JA, Ferry GD, Versalovic J. Probiotic Lactobacillus reuteri suppress proinflammatory cytokines via c-Jun. Inflammatory bowel diseases. 2008;14(8):1068–83. doi: 10.1002/ibd.20448. Epub 2008/04/22. [DOI] [PubMed] [Google Scholar]
- 41.Schreiber O, Petersson J, Phillipson M, Perry M, Roos S, Holm L. Lactobacillus reuteri prevents colitis by reducing P-selectin-associated leukocyte- and platelet-endothelial cell interactions. American journal of physiology Gastrointestinal and liver physiology. 2009;296(3):G534–42. doi: 10.1152/ajpgi.90470.2008. Epub 2009/01/17. [DOI] [PubMed] [Google Scholar]
- 42.Luperchio SA, Schauer DB. Molecular pathogenesis of Citrobacter rodentium and transmissible murine colonic hyperplasia. Microbes and infection/Institut Pasteur. 2001;3(4):333–40. doi: 10.1016/s1286-4579(01)01387-9. Epub 2001/05/04. [DOI] [PubMed] [Google Scholar]
- 43.Wiles S, Clare S, Harker J, Huett A, Young D, Dougan G, et al. Organ specificity, colonization and clearance dynamics in vivo following oral challenges with the murine pathogen Citrobacter rodentium. Cellular microbiology. 2004;6(10):963–72. doi: 10.1111/j.1462-5822.2004.00414.x. Epub 2004/09/02. [DOI] [PubMed] [Google Scholar]
- 44.Dicksved J, Schreiber O, Willing B, Petersson J, Rang S, Phillipson M, et al. Lactobacillus reuteri maintains a functional mucosal barrier during DSS treatment despite mucus layer dysfunction. PloS one. 2012;7(9):e46399. doi: 10.1371/journal.pone.0046399. Epub 2012/10/03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Schaefer L, Auchtung TA, Hermans KE, Whitehead D, Borhan B, Britton RA. The antimicrobial compound reuterin (3-hydroxypropionaldehyde) induces oxidative stress via interaction with thiol groups. Microbiology (Reading, England) 2010;156(Pt 6):1589–99. doi: 10.1099/mic.0.035642-0. Epub 2010/02/13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Spinler JK, Taweechotipatr M, Rognerud CL, Ou CN, Tumwasorn S, Versalovic J. Human-derived probiotic Lactobacillus reuteri demonstrate antimicrobial activities targeting diverse enteric bacterial pathogens. Anaerobe. 2008;14(3):166–71. doi: 10.1016/j.anaerobe.2008.02.001. Epub 2008/04/09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Preidis GA, Saulnier DM, Blutt SE, Mistretta TA, Riehle KP, Major AM, et al. Probiotics stimulate enterocyte migration and microbial diversity in the neonatal mouse intestine. FASEB journal: official publication of the Federation of American Societies for Experimental Biology. 2012;26(5):1960–9. doi: 10.1096/fj.10-177980. Epub 2012/01/24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Saxami G, Ypsilantis P, Sidira M, Simopoulos C, Kourkoutas Y, Galanis A. Distinct adhesion of probiotic strain Lactobacillus casei ATCC 393 to rat intestinal mucosa. Anaerobe. 2012;18(4):417–20. doi: 10.1016/j.anaerobe.2012.04.002. Epub 2012/05/05. [DOI] [PubMed] [Google Scholar]
- 49.Frese SA, Benson AK, Tannock GW, Loach DM, Kim J, Zhang M, et al. The evolution of host specialization in the vertebrate gut symbiont Lactobacillus reuteri. PLoS genetics. 2011;7(2):e1001314. doi: 10.1371/journal.pgen.1001314. Epub 2011/03/08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hoffmann M, Rath E, Holzlwimmer G, Quintanilla-Martinez L, Loach D, Tannock G, et al. Lactobacillus reuteri 100-23 transiently activates intestinal epithelial cells of mice that have a complex microbiota during early stages of colonization. The Journal of nutrition. 2008;138(9):1684–91. doi: 10.1093/jn/138.9.1684. Epub 2008/08/22. [DOI] [PubMed] [Google Scholar]