Abstract
Objectives:
To investigate whether electrolyte levels measured by using blood gas analyzers (ABG) and auto-analyzers (AA) are equivalent and can be used interchangeably.
Methods:
This observational prospective study was conducted in 100 patients admitted to the Intensive Care Unit, Adnan Menderes University School of Medicine, Aydin, Turkey, between March and August 2014. Samples for both AA and ABG analyzers were collected simultaneously from invasive arterial catheters of patients. The electrolyte levels were measured by using 2 methods.
Results:
The mean sodium level measured by ABG was 136.1±6.3 mmol/L and 137.8±5.4 mmol/L for AA (p=0.001). The Pearson’s correlation coefficient was 0.561 (p<0.001). The Bland-Altman 95% limits of agreement were -9.4 to 12.6 mmol/L. The mean potassium levels measured by ABG was 3.4±0.7 mmol/L and AA was 3.8±0.7 mmol/L (p=0.001). The Bland-Altman comparison limits were -0.58 to 1.24 and the associated Pearson’s correlation coefficient was 0.812 (p<0.001).
Conclusion:
The results of the 2 analyzing methods, in terms of sodium, were not equivalent and could not be used interchangeably. However, according to the statistical analyses results, by including, but not blindly trusting these findings, urgent and vital decisions could be made by the potassium levels obtained from the BGA, but a simultaneous follow-up sample had to be sent to the central laboratory for confirmation.
Electrolytes are very important for the continuation of the physiological functions of the human body. They play vital roles in: regulation of the cell membrane potential, steady process of neurohormonal pathways, energy transformation and the fluid, and acid-base balance in the body. Signs and symptoms of electrolyte disorders may be nonspecific in an intensive care unit (ICU) patient.1-3 The therapies directed for maintaining vital organ functions affect the electrolyte balance. Consequently, electrolyte disorders are more common in critically ill patients than non-critically ill patients.1 The incidence of electrolyte disorders is nearly 25% in ICU patients.2 In recent studies,4-6 it is shown that in ICU patients, serum sodium and potassium levels are significant predictors of mortality. Therefore, prompt and complete correction of electrolyte disorders in ICU patients is vitally important. Under these circumstances, the importance of obtaining the results of serum electrolyte levels at the earliest is obvious. In routine application, serum electrolytes are measured by the indirect ion-sensing (ISE) method using auto-analyzers (AA) located in the central laboratories of hospitals. In this analyzing method, the processing time is longer because of a delay in the transportation of the samples to the central laboratory on account of several reasons.7 Hence, point-of-care (POC) testing methods, such as, arterial blood gas (ABG) analyzers have been increasingly used in the daily assessments of ICU patients. Blood gas analyzers; use the direct ISE method with short processing time that provides time and rapidity to the physician in the patient’s treatment decisions.7,8 The United States Clinical Laboratory Improvement Amendments (US CLIA) accepts a 0.5 mmol/l difference in the measured potassium levels and a 4 mmol/l difference in the measured sodium levels, in the gold standard measure of the standard calibration solution.9 In some recent studies, the data revealed the difference in the electrolyte levels between the ABG and AA results.10,11 Furthermore, there are also studies that suggest that there is no significant difference between these measuring methods.12,13 Physicians want to trust the veracity of the ABG results of electrolytes such as sodium and potassium because, by this method, the delay in reaching the results is surpassed, and risks arising from this delay may be reduced. However, the results of the above-mentioned studies are confusing and still a diagnostic challenge for physicians. On account of the hesitation by the physicians, we decided to investigate whether the sodium and potassium levels measured by using ABG and AA were equivalent. We conducted a prospective study comparing the electrolyte level results measured in the arterial blood samples by 2 different methods. We tried to notice all limitations of previous similar studies and designed our study according to these points. Thereby, we aimed to improve the accuracy of our study results.
Methods
This was a prospective study of a consecutive cohort of adults admitted to the ICU, Adnan Menderes University School of Medicine, Aydin, Turkey, between March and August 2014. Related research studies were surveyed carefully from different databases. The present study was approved by the local Ethics Committee of Adnan Menderes University School of Medicine. All procedures were in accordance with the Declaration of Helsinki. Informed consent for sampling was taken from the patient or from the lawful custodian.
The sample size of 17 patients achieved 80% power to detect a difference of 5.2 mmol/l for sodium with an alpha of 0.05.12 Patients with positive informed consent and aged between 18-70 were included in this study. Additionally, we included only patients whose paired blood samples could simultaneously be collected from an arterial catheter. Negative informed consent rendering, aged under 18 or >70, without an arterial catheter and the blood samples could not simultaneously be collected patients were excluded from our study. These samples were collected only if they were clinically indicated. All samples were collected by the specially trained nursing staff of the ICU. Two samples of arterial blood were collected at the bedside, at the same time. For the arterial blood gas analyzer, 1.6 ml blood was collected in commercially available plastic arterial blood gas syringes (BD A-LINE arterial blood gas collection syringe, 3.0 ml volume, 1.6 ml recommended draw volume, Becton, Dickinson Diagnostics®, Plymouth, UK) coated with 80 I.U Ca-heparin. These arterial blood gas samples were analyzed immediately, using Siemens Rapid Point 500 blood gas analyzer, which is located in the ICU. The blood gas analyzer was calibrated automatically. Every 4 hourly one point, every 8 hourly 2 points, and an external quality check was performed weekly. The second blood sample was collected 2 ml in a non-additive silicone-coated tube and sent to the central laboratory for measurement of serum electrolytes by Abott C 8000 Architect Auto-analyzer. Each sample was analyzed within a maximum of one hour after collection. The auto-analyzer was calibrated routinely every 24 hours by linear calibration, according to the manufacturer’s recommendations.
Statistical analysis
All data were tested for normality with the Kolmogorov-Smirnov test using the PASW Statistics for Windows, Version 18.0. (SPSS Inc., Chicago, IL,. USA). The paired sample t-test was used to detect the difference between the 2 dependent groups. Descriptive statistics were presented as mean ± standard deviation. Agreement between the 2 analyzers was assessed using the Bland-Altman approach.14 The linear relationship between the variables was assessed by the Pearson’s correlation analyzing method. A p-value of less than 0.05 was considered to indicate statistical significance.
Results
The mean sodium level measured by ABG was 136.1±6.3 mmol/L, and the result of AA for the mean sodium level was 137.8±5.4 mmol/L. There was a statistically significant difference on application of the paired sample t-test between the mean sodium levels of the 2 analyzing methods (p<0.001). The maximum difference in the sodium level was 16.1 mmol/L, and the minimum difference was -13 mmol/L. Consequently, the hypothesis suggesting that there was no difference between the 2 machines’ results was rejected. The Pearson’s correlation coefficient was r: 0.561 (p<0.001) (Figure 1A), suggesting a moderate correlation.
Figure 1.
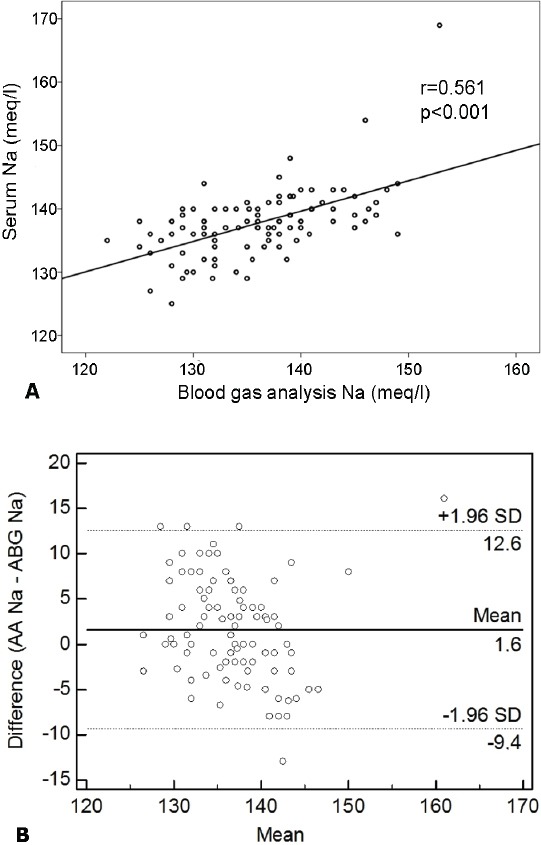
The correlation of sodium levels. A) A Bland-Altman comparison of AA and ABG for the Na measurement revealed that the limits of agreement were -9.4 to 12.6 mmol/L. Relationship between Na levels measured by 2 analyzing methods. Pearson’s correlation coefficient (r): (r: 0.561, p<0.001). B) Bland-Altman comparison of AA and ABG for Na showing the 95% limits of agreement. Plot of the difference of 2 methods against the mean of 2 methods for Na measurement. Solid line - mean difference, dashed lines - mean difference plus or minus 2 standard deviations, Na - Sodium, AA - auto-analyzer, ABG - arterial blood gas analyzer.
A Bland-Altman comparison of AA and ABG for the sodium measurement results revealed that the limits of agreement were -9.4 to 12.6 mmol/L. As shown in Figure 1B, the 95% limits of agreement for sodium were very wide and this was not clinically acceptable.
The sodium analysis results were stratified according to the standard laboratory values; ≤134 mmol/L was considered as hyponatremia, 135-145 mmol/L was considered as normonatremia, and values ≥145 mmol/L were considered as hypernatremia. The difference in sodium levels through the 2 analyzing methods was statistically significant in the normonatremia group (p=0.007; null hypothesis rejected) (Table 1).
Table 1.
Stratified analysis of difference between electrolyte levels measured by AA and ABG.
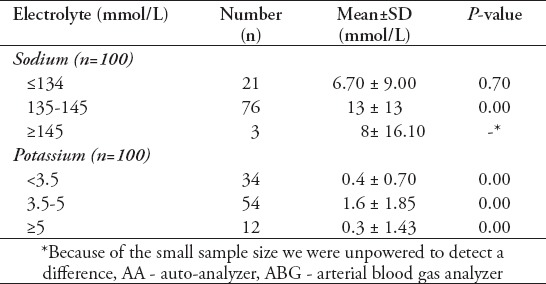
Additionally, the mean difference between AA and ABG was 13 mmol/L, which was not within the acceptable limit for sodium defined by CLIA. There was no significant difference between the 2 methods for the hyponatremia group (p=0.702); however, the mean difference was 6.70 mmol/L, which was also not within the acceptable limits of CLIA. In the hypernatremia group, because of the small sample size of the group (n=3), we were unable to detect a difference, but the mean difference between the 2 methods was above the defined acceptable limits of CLIA.
The mean potassium levels measured by ABG was 3.4±0.7 mmol/L and AA was 3.8±0.7 mmol/L. There was a statistically significant difference between the 2 analyzing methods in terms of the potassium levels (p<0.001). The maximum difference was 1.8 mmol/L, and the minimum difference was -1.6 mmol/L. However, the Pearson’s correlation coefficient was 0.812 (p<0.001) (Figure 2A), suggesting a strong correlation between the 2 different analyzing methods. Additionally, the Bland-Altman comparison of the ABG and AA data revealed that the limits of agreement were - 0.58 to 1.24 mmol/L (Figure 2B). These limits were clinically acceptable.
Figure 2.
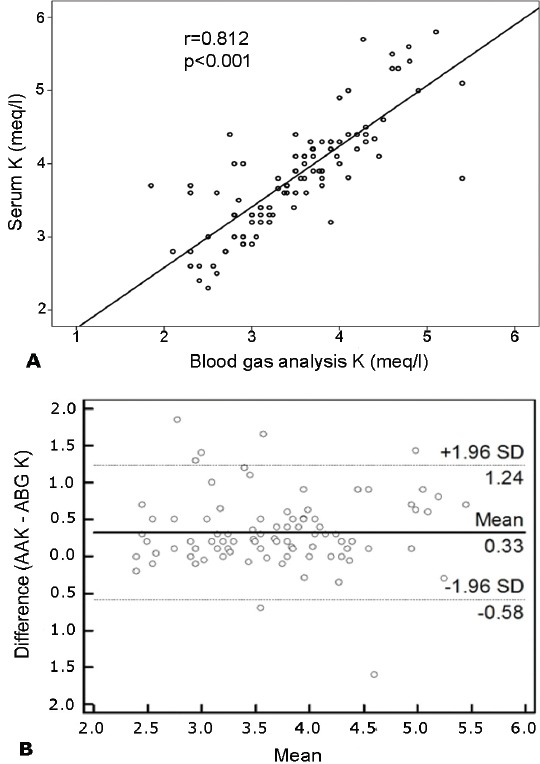
The correlation of potassium (K) levels. A) Relationship between K levels measured by 2 analyzing methods. Pearson’s correlation coefficient(r) (r: 0.812, p<0.001). B) Bland-Altman comparison of AA and ABG for K showing the 95% limits of agreement. Plot of the difference of 2 methods against the mean of 2 methods for K measurement. Solid line - mean difference, dashed lines - mean difference ±2 SD, AA - auto-analyzer, ABG - arterial blood gas analyzer
The potassium values were stratified based upon the standard laboratory values. The mean difference in potassium levels through ABG and AA were statistically significant in the hypokalemia (p=0.002), normokalemia (p<0.001), and hyperkalemia (p=0.001) groups. However, the mean differences in patients with hypokalemia was 0.5 mmol/L and hyperkalemia was 0.3 mmol/L, all of which were within the acceptable limits of the CLIA guidelines9 (Table 1).
Discussion
Intensive care unit patients require more frequent monitoring of their electrolyte levels. This increases the hospital laboratory costs of the patients. However, if the staff takes the sample and processes it at the bedside of the patient with POC testing instruments, both labor costs, and reagent costs are reduced when compared with the central laboratory (AA) analyzing costs.15 Furthermore, POC testing results are rapidly available, which enables early detection of adverse events and an early start of the appropriate treatment.16 Furthermore, it is known that patients in ICUs are critically ill and tend to have low blood protein levels. The ABG results are not affected by serum protein levels, which make the ABG electrolyte results more accurate for those critically ill patients.17 All the advantages mentioned above refer physicians to use ABG testing in ICUs and emergency departments more frequently.
In the present study, we wanted to identify if the sodium and potassium levels measured by using different methods and instruments were equivalent and could be used interchangeably.
In the study conducted by Morimatsu et al,10 it was revealed that results with AA and ABG differed significantly for the plasma sodium and chloride levels. Moreover, these differences significantly affected the calculation of the anion gap values, which could consequently lead physicians to different assessments of the acid-base and electrolyte levels. Furthermore, Chacko et al11 also concluded that the differences in the measured sodium levels between the 2 methods were significant. Consistent with the literature, in our present study, there was a significant difference between the 2 methods for sodium levels. Although the mean sodium level differences were very small between AA and ABG, the Bland Altman’s 95% limits of agreement for sodium were very wide, which was not clinically acceptable. Seventy-six percent of the total patients were normonatremic, and there was a significant difference between the 2 analyzing methods in this normonatremia group. At this point, considering our study data, making clinical decisions for sodium according to the ABG results seemed to be unreliable. There was a statistically significant difference between the mean potassium levels measured by ABG and AA. Several reasons might explain this difference between the 2 analyzing methods. Each instrument used a different sample types, such as, serum or whole blood. Hemolysis caused a release of potassium and other intracellular components from the erythrocytes into the extracellular fluid, leading to elevate levels of serum potassium. Hemolysis might result from prolonged storage at low temperatures or a prolonged time between sampling and analysis, use of alcohol for disinfection, and inappropriate sampling needles.18
The factors mentioned above seem most likely to affect the potassium instead of the sodium levels. Moreover, removing adequate amount of the discarded volume is very important for avoiding dilution with flush fluid when sampling from a catheter. Furthermore, the different heparin volumes in ABG sampling syringes dilute the whole blood and lower the levels of measured electrolytes in ABG testing. For this reason, pre-heparinized dry and balanced syringes are recommended for ABG sampling. Even with the use of pre-heparinized syringes, lower than 5% bias was reported.19
In our study, we tried to minimize these external contributing factors by considereing other study limitations. For the standardization of sampling and to edge out all affecting factors, as mentioned above; we used pre-heparinized standardized syringes for sampling. Furthermore, only previously informed, trained, and selected staff of the ICU performed the sampling, to prevent sampling errors. The same simultaneously obtained arterial catheter samples were used for both analyses instead of using different sampling areas. Moreover, we analyzed both the ABG and AA samples within a maximum of one hour after collection, to avoid hemolysis.
Along with the statistically significant difference between the mean potassium levels, the mean difference between AA and ABG was 0.32 mmol/L for potassium. The magnitude of difference in our study was consistent with the literature data (0.1-0.7 mmol/L), as reported earlier.8 This value was within the acceptable limits of CLIA. Furthermore, there was a strong correlation between the results of the 2 instruments, concomitant with the acceptable 95% limits of agreement in the Bland Altman analysis. Even though statistically significant, the mean differences in the potassium levels of hypokalemic was 0.4 mmol/L and hyperkalemic patients was 0.3 mmol/L, which were all within the acceptable limits of CLIA.
However, the answer to the question of what that statistical significance meant to the clinical difference is not clear yet. It is not possible to determine whether the AA or ABG electrolyte values are closer to the real values. The observed differences between AA and ABG electrolyte values arise from the combination of various factors, as discussed above. Using a correction factor may compensate these variations. The usage of a correction factor may minimize the differences between the analyzing instruments. We cannot recommend this method for the sodium results of our study. Although, if a correction factor of 0.32 mmol/L is used for the potassium values, the results of AA and ABG instruments may be used interchangeably. However, this correction factor should be determined individually for each hospital. Therefore, it is important for each center to perform its own studies with regard to concordance.
Under these circumstances, we conclude that, similar to the study of Jain et al,12 including, but not blindly trusting these findings; critical, urgent, and vital decisions can be made by using the potassium levels obtained from the ABG, but a simultaneously follow-up sample must be sent to the central laboratory for confirmation.
Study limitations
Some of the limitations of our study were the use of only one type of AA and one type of ABG instrument. Different types of measurement instruments might provide more information on variance and accuracy. However, these were the only instruments available in our hospital. Furthermore, these 2 methods and instruments mentioned above were used by numerous hospitals in developing countries, making our study more relevant and generalizable with those localities using the same instruments. Another limitation was to leave the serum protein levels out of count. As well known, the serum protein levels, which might be low in critically ill patients, could affect the measured electrolyte results.
In conclusion, our study emphasizes the importance of determining the concordance between the electrolyte values obtained by AA and ABG, individually, for each hospital. The usage of a correction factor may minimize the differences between the analyzing instruments. We cannot advice the use of sodium results interchangeably because the results differ between AA and ABG. Conversely, we think that; including, but not blindly trusting these findings can make critical, urgent, and vital decisions made by the potassium levels obtained from the ABG machines.
Footnotes
Ethical Consent.
All manuscripts reporting the results of experimental investigations involving human subjects should include a statement confirming that informed consent was obtained from each subject or subject’s guardian, after receiving approval of the experimental protocol by a local human ethics committee, or institutional review board. When reporting experiments on animals, authors should indicate whether the institutional and national guide for the care and use of laboratory animals was followed.
References
- 1.Hoekstra M, Vogelzang M, Drost JT, Janse M, Loef BG, van der Horst IC, et al. Implementation and evaluation of a nurse-centered computerized potassium regulation protocol in the intensive care unit--a before and after analysis. BMC Med Inform Decis Mak. 2010;10:5. doi: 10.1186/1472-6947-10-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Vincent JL, Abraham E, Moore FA, Kochanek P, Mitchell P. Textbook of Critical Care. 6th ed. Philadelphia (PA): Saunders; 2011. [Google Scholar]
- 3.Buckley MS, LeBlanc JM, Cawley MU. Electrolyte disturbances associated with commonly prescribed medications in the intensive care unit. Crit Care Med. 2010;38(6 Suppl):S253–S264. doi: 10.1097/CCM.0b013e3181dda0be. [DOI] [PubMed] [Google Scholar]
- 4.Whelan B, Bennett K, O'Riordan D, Silke B. Serum sodium as a risk factor for in-hospital mortality in acute unselected medical patients. QJM. 2009;102:175–182. doi: 10.1093/qjmed/hcn165. [DOI] [PubMed] [Google Scholar]
- 5.Chawla A, Sterns RH, Nigwekar SU, Cappuccio JD. Mortality and serum sodium: do patients die from or with hyponatremia? Clin J Am Soc Nephrol. 2011;6:960–965. doi: 10.2215/CJN.10101110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mousavi SA, Shahabi S, Mostafapour E, Purfakharan M, Fereshtehnejad SM, Amini J, et al. Comparison of the serum electrolyte levels among patients died and survived in the intensive care unit. Tanaffos. 2012;11:36–42. [PMC free article] [PubMed] [Google Scholar]
- 7.Dimeski G, Morgan TJ, Presneill JJ, Venkatesh B. Disagreement between ion selective electrode direct and indirect sodium measurements: estimation of the problem in a tertiary referral hospital. J Crit Care. 2012;27:326.e9–3260e16. doi: 10.1016/j.jcrc.2011.11.003. [DOI] [PubMed] [Google Scholar]
- 8.Scott MG, LeGrys VA, Klutts JS. Tietz Textbook of Clinical Chemistry and Molecular Diagnostics. 4th ed. Philadelphia (PA): Saunders; 2006. Electrolytes and blood gases; pp. 983–1018. [Google Scholar]
- 9.Ehrmeyer SS, Laessig RH, Leinweber JE, Oryall JJ. 1990 Medicare/CLIA final rules for proficiency testing: minimum intralaboratory performance characteristics (CV and bias) needed to pass. Clin Chem. 1990;36:1736–1740. [PubMed] [Google Scholar]
- 10.Morimatsu H, Rocktäschel J, Bellomo R, Uchino S, Goldsmith D, Gutteridge G. Comparison of point-of-care versus central laboratory measurement of electrolyte concentrations on calculations of the anion gap and the strong ion difference. Anesthesiology. 2003;98:1077–1084. doi: 10.1097/00000542-200305000-00009. [DOI] [PubMed] [Google Scholar]
- 11.Chacko B, Peter JV, Patole S, Fleming JJ, Selvakumar R. Electrolytes assessed by point-of-care testing- Are the values comperable with results obtained from the central laboratory? Indian J Crit Care Med. 2011;15:24–29. doi: 10.4103/0972-5229.78219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Jain A, Subhan I, Joshi M. Comparison of the point-of-care blood gas analyzer versus the laboratory auto-analyzer for the measurement of electrolytes. Int J Emerg Med. 2009;2:117–120. doi: 10.1007/s12245-009-0091-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhang JB, Lin J, Zhao XD. Analysis of bias in measurements of potassium, sodium and hemoglobin by an emergency department-based blood gas analyzer relative to hospital laboratory autoanalyzer results. PloS One. 2015;10:e0122383. doi: 10.1371/journal.pone.0122383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet. 1986;1:307–310. [PubMed] [Google Scholar]
- 15.Statland BE, Brzys K. Evaluating STAT testing alternatives by calculating annual laboratory costs. Chest. 1990;97:198–203. doi: 10.1378/chest.97.5_supplement.198s. [DOI] [PubMed] [Google Scholar]
- 16.Burtis C, Bruns D, editors. Tietz Fundamentals of Clinical Chemistry and Molecular Diagnostics. 7th ed. Philadelphia (PA): Saunders; 2014. [Google Scholar]
- 17.King RI, Mackay RJ, Florkowsky CM, Lynn AM. Electrolytes in sick neonates-which sodium is the right answer? Arch Dis Child Fetal Neonatal Ed. 2013;98:F74–F76. doi: 10.1136/archdischild-2011-300929. [DOI] [PubMed] [Google Scholar]
- 18.Sakyi A, Laing E, Ephraim R, Asibey O, Sadique O. Evaluation of analytical errors in a clinical chemistry laboratory: a 3 year experience. Ann Med Health Sci Res. 2015;5:8–12. doi: 10.4103/2141-9248.149763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Van BM, Scharnhost V. Electrolyte-balanced heparin in blood gas syringes can introduce a significant bias in measurement of positively charged electrolytes. Clin Chem Lab Med. 2011;49:249–252. doi: 10.1515/CCLM.2011.047. [DOI] [PubMed] [Google Scholar]


