Abstract
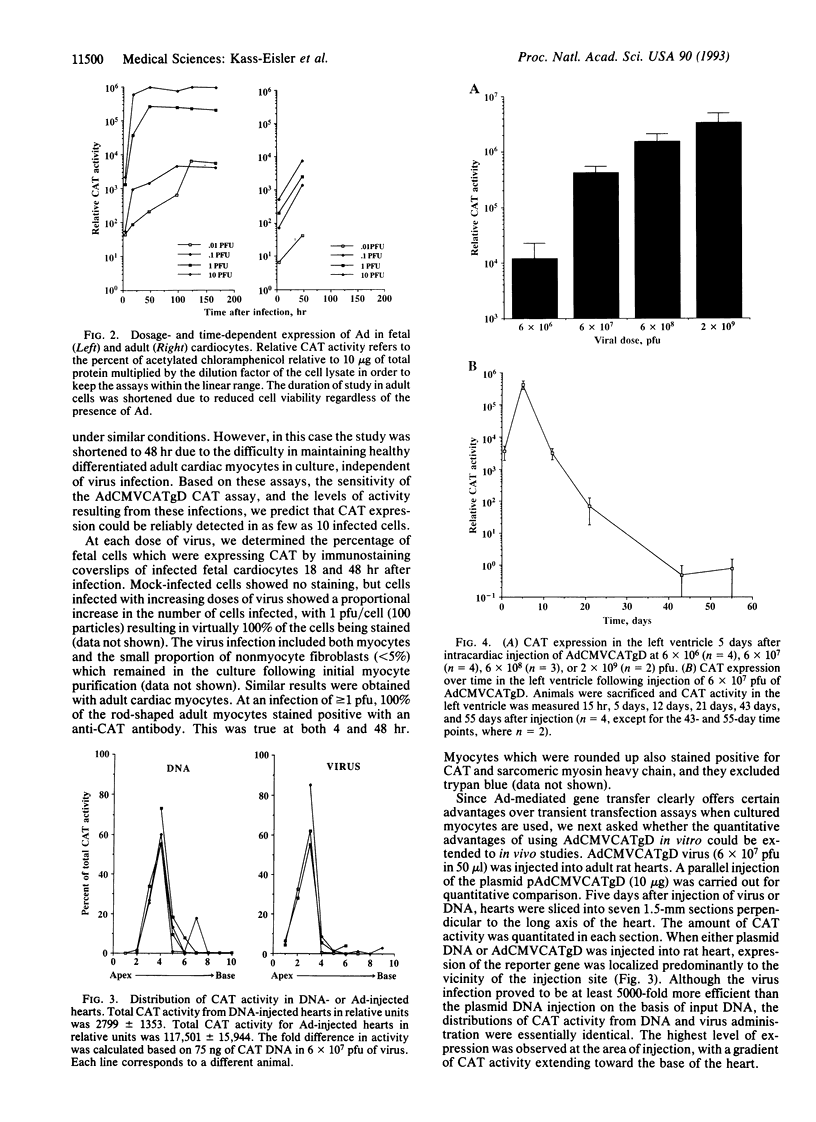
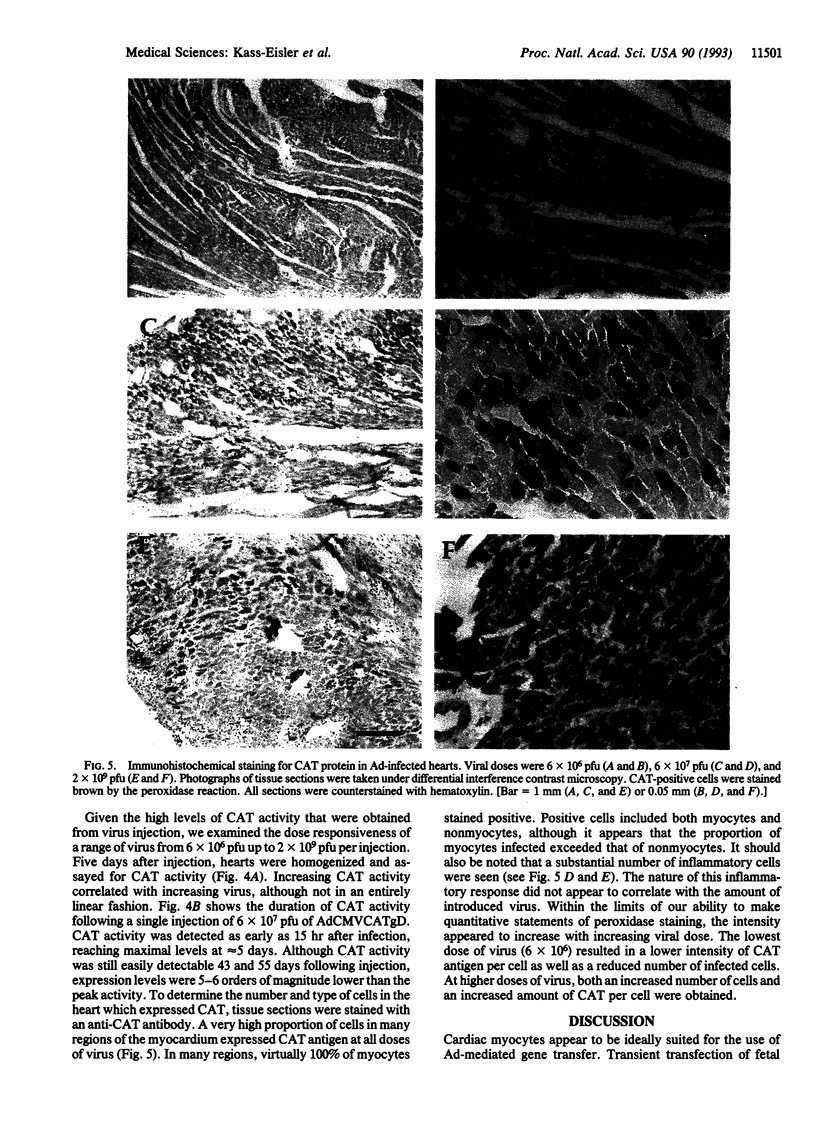
To optimize the use of modified adenoviruses as vectors for gene delivery to the myocardium, we have characterized infection of cultured fetal and adult rat cardiac myocytes in vitro and of adult cardiac myocytes in vivo by using a replication-defective adenovirus carrying the chloramphenicol acetyltransferase (CAT) reporter gene driven by the cytomegalovirus promoter (AdCMVCATgD). In vitro, virtually all fetal or adult cardiocytes express the CAT gene when infected with 1 plaque-forming unit of virus per cell. CAT enzymatic activity can be detected in these cells as early as 4 hr after infection, reaching near-maximal levels at 48 hr. In fetal cells, CAT expression was maintained without a loss in activity for at least 1 week. Using in vitro studies as a guide, we introduced the AdCMVCATgD virus directly into adult rat myocardium and compared the expression results obtained from virus injection with those obtained by direct injection of pAdCMVCATgD plasmid DNA. The amount of CAT activity resulting from adenovirus infection of the myocardium was orders of magnitude higher than that seen from DNA injection and was proportional to the amount of input virus. Immunostaining for CAT protein in cardiac tissue sections following adenovirus injection demonstrated large numbers of positive cells, reaching nearly 100% of the myocytes in many regions of the heart. Expression of genes introduced by adenovirus peaked at 5 days but was still detectable 55 days following infection. Adenoviruses are therefore a very useful tool for high-efficiency gene transfer into the cardiovascular system.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Acsadi G., Jiao S. S., Jani A., Duke D., Williams P., Chong W., Wolff J. A. Direct gene transfer and expression into rat heart in vivo. New Biol. 1991 Jan;3(1):71–81. [PubMed] [Google Scholar]
- Babich A., Feldman L. T., Nevins J. R., Darnell J. E., Jr, Weinberger C. Effect of adenovirus on metabolism of specific host mRNAs: transport control and specific translational discrimination. Mol Cell Biol. 1983 Jul;3(7):1212–1221. doi: 10.1128/mcb.3.7.1212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Babiss L. E., Friedman J. M., Darnell J. E., Jr Cellular promoters incorporated into the adenovirus genome: effects of viral regulatory elements on transcription rates and cell specificity of albumin and beta-globin promoters. Mol Cell Biol. 1986 Nov;6(11):3798–3806. doi: 10.1128/mcb.6.11.3798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beltz G. A., Flint S. J. Inhibition of HeLa cell protein synthesis during adenovirus infection. Restriction of cellular messenger RNA sequences to the nucleus. J Mol Biol. 1979 Jun 25;131(2):353–373. doi: 10.1016/0022-2836(79)90081-0. [DOI] [PubMed] [Google Scholar]
- Berkner K. L., Sharp P. A. Expression of dihydrofolate reductase, and of the adjacent EIb region, in an Ad5-dihydrofolate reductase recombinant virus. Nucleic Acids Res. 1984 Feb 24;12(4):1925–1941. doi: 10.1093/nar/12.4.1925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedman J. M., Babiss L. E., Clayton D. F., Darnell J. E., Jr Cellular promoters incorporated into the adenovirus genome: cell specificity of albumin and immunoglobulin expression. Mol Cell Biol. 1986 Nov;6(11):3791–3797. doi: 10.1128/mcb.6.11.3791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gustafson T. A., Markham B. E., Bahl J. J., Morkin E. Thyroid hormone regulates expression of a transfected alpha-myosin heavy-chain fusion gene in fetal heart cells. Proc Natl Acad Sci U S A. 1987 May;84(10):3122–3126. doi: 10.1073/pnas.84.10.3122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrison T., Graham F., Williams J. Host-range mutants of adenovirus type 5 defective for growth in HeLa cells. Virology. 1977 Mar;77(1):319–329. doi: 10.1016/0042-6822(77)90428-7. [DOI] [PubMed] [Google Scholar]
- Herz J., Gerard R. D. Adenovirus-mediated transfer of low density lipoprotein receptor gene acutely accelerates cholesterol clearance in normal mice. Proc Natl Acad Sci U S A. 1993 Apr 1;90(7):2812–2816. doi: 10.1073/pnas.90.7.2812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones N., Shenk T. Isolation of adenovirus type 5 host range deletion mutants defective for transformation of rat embryo cells. Cell. 1979 Jul;17(3):683–689. doi: 10.1016/0092-8674(79)90275-7. [DOI] [PubMed] [Google Scholar]
- Kirshenbaum L. A., MacLellan W. R., Mazur W., French B. A., Schneider M. D. Highly efficient gene transfer into adult ventricular myocytes by recombinant adenovirus. J Clin Invest. 1993 Jul;92(1):381–387. doi: 10.1172/JCI116577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kitsis R. N., Buttrick P. M., McNally E. M., Kaplan M. L., Leinwand L. A. Hormonal modulation of a gene injected into rat heart in vivo. Proc Natl Acad Sci U S A. 1991 May 15;88(10):4138–4142. doi: 10.1073/pnas.88.10.4138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lemarchand P., Jones M., Yamada I., Crystal R. G. In vivo gene transfer and expression in normal uninjured blood vessels using replication-deficient recombinant adenovirus vectors. Circ Res. 1993 May;72(5):1132–1138. doi: 10.1161/01.res.72.5.1132. [DOI] [PubMed] [Google Scholar]
- Lin H., Parmacek M. S., Morle G., Bolling S., Leiden J. M. Expression of recombinant genes in myocardium in vivo after direct injection of DNA. Circulation. 1990 Dec;82(6):2217–2221. doi: 10.1161/01.cir.82.6.2217. [DOI] [PubMed] [Google Scholar]
- Mittal S. K., McDermott M. R., Johnson D. C., Prevec L., Graham F. L. Monitoring foreign gene expression by a human adenovirus-based vector using the firefly luciferase gene as a reporter. Virus Res. 1993 Apr;28(1):67–90. doi: 10.1016/0168-1702(93)90090-a. [DOI] [PubMed] [Google Scholar]
- Quantin B., Perricaudet L. D., Tajbakhsh S., Mandel J. L. Adenovirus as an expression vector in muscle cells in vivo. Proc Natl Acad Sci U S A. 1992 Apr 1;89(7):2581–2584. doi: 10.1073/pnas.89.7.2581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rindt H., Gulick J., Knotts S., Neumann J., Robbins J. In vivo analysis of the murine beta-myosin heavy chain gene promoter. J Biol Chem. 1993 Mar 5;268(7):5332–5338. [PubMed] [Google Scholar]
- Rosenfeld M. A., Siegfried W., Yoshimura K., Yoneyama K., Fukayama M., Stier L. E., Päkkö P. K., Gilardi P., Stratford-Perricaudet L. D., Perricaudet M. Adenovirus-mediated transfer of a recombinant alpha 1-antitrypsin gene to the lung epithelium in vivo. Science. 1991 Apr 19;252(5004):431–434. doi: 10.1126/science.2017680. [DOI] [PubMed] [Google Scholar]
- Rosenfeld M. A., Yoshimura K., Trapnell B. C., Yoneyama K., Rosenthal E. R., Dalemans W., Fukayama M., Bargon J., Stier L. E., Stratford-Perricaudet L. In vivo transfer of the human cystic fibrosis transmembrane conductance regulator gene to the airway epithelium. Cell. 1992 Jan 10;68(1):143–155. doi: 10.1016/0092-8674(92)90213-v. [DOI] [PubMed] [Google Scholar]
- Schneider R. J., Shenk T. Impact of virus infection on host cell protein synthesis. Annu Rev Biochem. 1987;56:317–332. doi: 10.1146/annurev.bi.56.070187.001533. [DOI] [PubMed] [Google Scholar]
- Stratford-Perricaudet L. D., Makeh I., Perricaudet M., Briand P. Widespread long-term gene transfer to mouse skeletal muscles and heart. J Clin Invest. 1992 Aug;90(2):626–630. doi: 10.1172/JCI115902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Subramaniam A., Jones W. K., Gulick J., Wert S., Neumann J., Robbins J. Tissue-specific regulation of the alpha-myosin heavy chain gene promoter in transgenic mice. J Biol Chem. 1991 Dec 25;266(36):24613–24620. [PubMed] [Google Scholar]
- Tantravahi J., Alvira M., Falck-Pedersen E. Characterization of the mouse beta maj globin transcription termination region: a spacing sequence is required between the poly(A) signal sequence and multiple downstream termination elements. Mol Cell Biol. 1993 Jan;13(1):578–587. doi: 10.1128/mcb.13.1.578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thummel C., Tjian R., Grodzicker T. Expression of SV40 T antigen under control of adenovirus promoters. Cell. 1981 Mar;23(3):825–836. doi: 10.1016/0092-8674(81)90447-5. [DOI] [PubMed] [Google Scholar]
- White R. L., Wittenberg B. A. NADH fluorescence of isolated ventricular myocytes: effects of pacing, myoglobin, and oxygen supply. Biophys J. 1993 Jul;65(1):196–204. doi: 10.1016/S0006-3495(93)81058-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Carvalho A. C., Tanowitz H. B., Wittner M., Dermietzel R., Roy C., Hertzberg E. L., Spray D. C. Gap junction distribution is altered between cardiac myocytes infected with Trypanosoma cruzi. Circ Res. 1992 Apr;70(4):733–742. doi: 10.1161/01.res.70.4.733. [DOI] [PubMed] [Google Scholar]





