Introduction
Noma, also known as cancrum oris or the face of poverty, is a rapidly progressive polymicrobial opportunistic infection resulting in orofacial gangrene and eventually death if untreated. The etiopathogenesis of the disease has been attributed to extreme malnutrition, dehydration, and inadequate oral hygiene.1, 2 Noma is primarily observed among children younger than 6 years in countries with a low human development index, with the highest incidence occurring in the noma belt, which extends from sub-Saharan West Africa to central Sudan.3 Cases in adults and citizens of high income countries are exceedingly rare and have been limited to those with immunodysfunction.4, 5, 6 We report a case of noma in an immunocompetent man who presented to an urban hospital in the United States.
Case Report
A 73-year-old black man from urban Atlanta, Georgia, presented to the emergency room with a 1-day history of fever, altered mental status, and dyspnea. His past medical history was significant for hypertension, a remote history of a cerebrovascular accident, and a 15-year history of osteoarthritis and degenerative disc disease. Medications included metoprolol and lisinopril and periodic analgesics for his musculoskeletal pain. His medical records indicated that he was coming only once yearly to see an internist for these medical conditions, and his last evaluation was 6 months before presentation. He had no history of illicit drug use. He had a 30-pack-year history of tobacco smoking and consumed 6 to 8 beers per day for many years. He used a wheelchair for mobility and was not employed, and his primary income source was social security benefits. He had lived independently until approximately 6 months before admission, when, according to close family members, his new girlfriend moved in with him and reportedly began exploiting him financially.
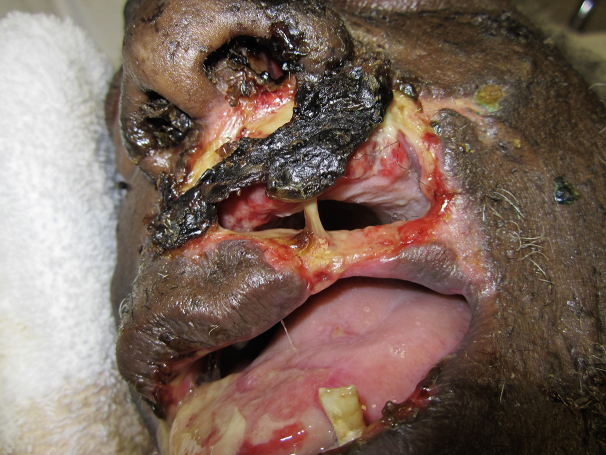
The patient was admitted with a diagnosis of severe acute respiratory distress syndrome (partial pressure of arterial oxygen/fraction of inspired oxygen = 98), acute kidney injury, and septic shock presumed secondary to aspiration pneumonia. On examination, he appeared diaphoretic, frail, and malnourished. Initial medical management consisted of mechanical ventilation, vasopressors, continuous renal replacement therapy, and antibiotic treatment with vancomycin and piperacillin/tazobactam. Laboratory values obtained during his admission compared with those obtained 6 months prior are displayed in Table I. Blood culture was positive for Streptococcus viridans and Staphylococcus epidermis and respiratory culture grew Enterobacter aerogenes and Citrobacter koseri. On day 4 of hospitalization, perioral erosions with hemorrhagic crust were noted. The differential diagnosis included mucocutaneous herpetic infection, Wegener's granulomatosis, necrotizing ulcerative gingivitis, mucormycosis, ecthyma gangrenosum, and extranodal natural killer/T-cell lymphoma nasal type (formerly known as lethal midline granuloma). By day 8, eschars had developed on the left upper and lower lips and nasal ala. White blood cell count had increased from 2.3 × 103/μL at admission to 19 × 103/μL (reference range, 4.0-8.5 × 103/μL) with a 95% neutrophilic predominance. Serologic findings for hepatitis B and C, syphilis, and HIV were negative, as were toxicology and antineutrophil cytoplasmic antibody screens. Piperacillin/tazobactam use was discontinued, and acyclovir was started for presumed mucocutaneous herpes simplex virus (HSV) infection. Oral surgery was consulted, and the bottom right central incisor and upper left cuspid were extracted to reduce aspiration risk. By hospital day 11, the patient's perioral eschars had evolved into disfiguring necrosis of the orofacial soft tissue. This necrosis extended superficially from the right philtral crest past the left nasolabial fold and invaded deep through the orbicularis oris to the alveolar mucosa, resulting in exposure of the oral cavity (Fig 1). Copious oral secretions, malodorous breath, and dental exfoliation were seen. Skin biopsy found a mixed inflammatory infiltrate with no evidence of organisms; however, HSV culture of the lip ulceration was positive.
Table I.
Comparison of laboratory values
| Value (normal range) | Six months before presentation | At presentation |
|---|---|---|
| Weight/BMI | 150 lbs/22.1 kg/m2 | 120 lbs/17.7 kg/m2 |
| Albumin (3.5-5.5 g/dL) | 3.8 g/dL | 2.3 g/dL |
| Triglycerides (65-227 mg/dL) | 77 mg/dL | 26 mg/dL |
| Hemoglobin (13.5-17.5 g/dL) | 16 g/dL | 12.2 g/dL |
| Iron (50-170 μg/dL) | — | <10 g/dL |
| % Iron saturation (20%-50%) | — | <10% |
| Prealbumin (17-34 mg/dL) | — | 9 mg/dL |
| 25-hydroxy vitamin D (30-100 pg/mL) | 12.0 pg/mL | 5 pg/mL |
| Folate (>7.7 ng/mL) | 12.5 ng/mL | 5.3 ng/mL |
| Vitamin B12 (180-914 pg/mL) | — | 716.0 pg/mL |
BMI, Body mass index.
Fig 1.
Acute noma. Extensive gangrenous necrosis extending through the perioral soft tissue and muscle. Photograph taken on hospital day 11.
Based on our patient's clinical findings along with laboratory data supporting severe malnutrition (Table I), the diagnosis of noma was rendered. To treat a broader spectrum of aerobic bacteria as well as anaerobic bacteria, metronidazole and meropenem were added to his treatment regimen. With aggressive management, progression of the acute necrotic process was halted. His fever resolved and white blood cell count normalized. The patient began following commands and mouthing words. He was eventually discharged to a skilled nursing facility with plans for surgical reconstruction, but he died shortly after discharge.
Discussion
Noma is a devastating and disfiguring disease of the world's poorest citizens. It is largely preventable by adequate nutrition and living conditions. Early recognition is crucial. Fever, fetid breath, local edema, and copious oral secretions classically accompany rapid unilateral spread and formation of a well-delineated eschar, which, when removed, reveals full-thickness tissue loss. Noma may be considered an evolution of acute necrotizing gingivitis (trench mouth) with extension past the periodontal tissue. In a study in Niger, the same bacterial microbiota were found in both diseases.7 Additionally, herpesvirus infection may contribute to the onset or progression of noma, as 81% of cases in Niger were reportedly positive for HSV antibodies.1 Similarly, HSV has been implicated in the pathogenesis of acute necrotizing ulcerative gingivitis; polymerase chain reaction detected HSV in 23% of cases among Nigerian children.8 The pathogenesis of noma is unclear; recognized contributing factors include anaerobic bacterial infection in the setting of severe malnutrition and impaired host immunity.1, 2, 7 Unlike necrotizing fasciitis, noma is not restricted to soft tissue; extension through muscle is characteristic. Mortality rate is reduced to 8% when adequately treated with antibiotics.1 We present this case to illustrate that a disease of poverty can present and progress to debilitation in one of the world's wealthiest nations.
Footnotes
Funding sources: None.
Conflicts of interest: None declared.
References
- 1.Baratti-Mayer D., Gayet-Ageron A., Hugonnet S. Risk factors for noma disease: a 6-year, prospective, matched case-control study in Niger. The Lancet Global Health. 2013;1:e87–e96. doi: 10.1016/S2214-109X(13)70015-9. [DOI] [PubMed] [Google Scholar]
- 2.Enwonwu C.O. Noma-The Ulcer of Extreme Poverty. N Engl J Med. 2006;354:221–224. doi: 10.1056/NEJMp058193. [DOI] [PubMed] [Google Scholar]
- 3.Marck K.W. A history of noma, the “Face of Poverty”. Plast Reconstr Surg. 2003;15:1702–1707. doi: 10.1097/01.PRS.0000055445.84307.3C. [DOI] [PubMed] [Google Scholar]
- 4.Chiandussi S., Luzzati R., Tirelli G., Di Lenarda R., Biasotto M. Cancrum oris in developed countries. Aging Clin Exp Res. 2009;21:475–477. doi: 10.1007/BF03327447. [DOI] [PubMed] [Google Scholar]
- 5.Silva K.N., Twaddell W.S., Powers D.B. A 40-year-old man with a perforated cheek. Am J Med Sci. 2011;341:399–403. doi: 10.1097/MAJ.0b013e3181e2ed3e. [DOI] [PubMed] [Google Scholar]
- 6.Lembo S., DeLeonibus C., Francia M.G., Lembo C., Ayala F. Cancrum oris in a boy with Down syndrome. J Am Acad Dermatol. 2011;64:1200–1202. doi: 10.1016/j.jaad.2009.08.048. [DOI] [PubMed] [Google Scholar]
- 7.Bolivar I., Whiteson K., Stadelmann B. Bacterial diversity in oral samples of children in Niger with acute noma, acute necrotizing gingivitis, and healthy controls. PLoS Negl Trop Dis. 2012;6(3):e1556. doi: 10.1371/journal.pntd.0001556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Contreras A., Falkler W.A., Jr., Enwonwu C.O. Human Herpesviridae in acute necrotizing ulcerative gingivitis in children in Nigeria. Oral Microbiol Immunol. 1997;12:259–265. doi: 10.1111/j.1399-302x.1997.tb00389.x. [DOI] [PubMed] [Google Scholar]