Abstract
Background:
Octogenarians with aortic stenosis are an increasing population of patients admitted for surgical aortic valve replacement (SAVR) or transcatheter aortic valve implantation (TAVI). Although adequate sleep is important after illness and surgery, it has scarcely been studied in the immediate postoperative phase.
Aims:
To determine and compare the nature of self-reported sleep and insomnia, and recorded sleep–wake patterns in octogenarians during the in-hospital postoperative phase after SAVR or TAVI.
Methods:
A prospective cohort design was used that included octogenarian patients undergoing SAVR or TAVI at a regional university hospital. Self-reports were used to document sleep and insomnia, and actigraphy was used to record sleep–wake patterns. Data were collected at baseline preoperatively, and then daily for the first five postoperative days.
Results:
SAVR patients experienced the most insomnia on postoperative nights later in recovery, while TAVI patients experienced the most insomnia on postoperative nights early in recovery. The median total sleep time, as measured by actigraphy, was 6.4 h, and the median sleep efficiency was 79% for the five postoperative nights, but no differences were found between SAVR and TAVI patients on this parameter. All patients slept more during daytime than at night, with SAVR patients having significantly more total sleep hours for all five days than TAVI patients (p < 0.01).
Conclusion:
Octogenarians with aortic stenosis had disturbed self-reported sleep, increased insomnia, and disturbed sleep–wake patterns postoperatively, resulting in more daytime sleep and inactivity. In patients undergoing SAVR or TAVI, sleep evolves differently during the in-hospital postoperative phase.
Keywords: Aortic stenosis, SAVR, TAVI, in-hospital sleep, octogenarian
Introduction
As people live longer, aortic stenosis has become a severe cardiac health problem.1,2 The prevalence is 0.2% among adults (between 50 and 59 years) versus 9.8% in octogenarians.3 Fifty-six per cent of patients with aortic stenosis in Europe are more than 70 years old.4 Without surgical treatment, these patients have a poor prognosis and decreased quality of life.2 Traditionally, aortic valves are replaced with surgical aortic valve replacements (SAVR).5 As an increasing number of octogenarians are undergoing cardiac surgery,6 treatment modalities more suitable to older patients have been developed. For patients with severe comorbidities and advanced age, and thereby at a higher surgical risk, transcatheter aortic valve implantation (TAVI) provides an alternative treatment.7
Sleep is essential for health and well-being,8 and good sleep restores both psychological and physical energy,9 which is important for recovery after surgery.9,10 Both short and long duration of sleep are risk factors for inflammation and development of chronic health conditions, such as cardiovascular disease and diabetes. In addition to contributing to poor mood and decreased quality of life, disrupted sleep has also shown to increase mortality risk.8,11,12 Sleep pattern generally changes with age;13 older people often have problems initiating and maintaining sleep.14 Objective registrations have shown that less efficient sleep commonly leads to increased daytime napping.14,15 Sleep problems in older people may also be a consequence of comorbidities due to symptoms such as pain and dyspnoea,16 and patients with heart failure are reported to have both subjective and objective sleep disturbance17,18 and insomnia.19 Patients having cardiac surgery often complain about poor sleep and experience sleep disturbances during the early postoperative phase, as well as after several weeks.9 Cardiopulmonary bypass circulation, thoracic drainage and sternotomy may all induce disturbances in mood and sleep.9 Octogenarians, with a high prevalence of preoperative sleep problems, constitute a growing group in the cardiothoracic surgical ward.1,6 It is therefore important to consider that postoperative sleep disturbances (e.g. increased number of awakenings, decreased sleep at night, increased daytime napping) can affect mobilization and recovery. The majority of studies on sleep after cardiac surgery have been undertaken in patients undergoing coronary artery bypass grafting. Few studies have investigated sleep after SAVR.9 To the best of our knowledge, only one previous study has described sleep problems after TAVI.20 However, that study only gathered data from the first postoperative day, included less than 54 patients and involved no objective sleep measurements.
The aims of this study were therefore to determine and compare the nature of self-reported sleep and insomnia, and recorded sleep–wake patterns, in octogenarians during the in-hospital postoperative phase after SAVR or TAVI.
Methods
Study design and setting
An observational, prospective cohort study design was used. This single-centre study took place in a 1400-bed university hospital within the only cardiothoracic surgical centre in western Norway.
Patients
Patients were included between February 2011 and August 2013. Self-reported data were collected at baseline (the day before surgery) and daily for five postoperative days. We used an actigraph to continually record the patients’ activity data, starting immediately after surgery through the next five postoperative days. Inclusion criteria were age of 80+ years, having severe symptomatic aortic stenosis and had been accepted for SAVR or TAVI. Severe aortic stenosis was defined as having an indexed aortic valve area <0.6 cm2/m2, a mean valve gradient > 40 mmHg, and a peak velocity >4.0 m/s.2 Additionally, to be included the patients had to be able to speak and understand Norwegian and to give written informed consent.
Of the 162 patients who were admitted for aortic valve replacement during the study period, 144 agreed to participate. One patient withdrew consent before surgery; thus, 143 patients were included in the study. However, seven patients were either non-responsive due to sedation, or died within five days after surgery. As this is a sub-study of a larger study, power analysis was performed for the primary endpoint of the main study.21
Treatment procedure
As standard surgical premedication, patients undergoing SAVR received morphine scopolamine, and patients undergoing TAVI received oxazepam. SAVR was performed under general anaesthesia as an open cardiac procedure through a midline sternotomy and with cardiopulmonary bypass circulation. TAVI was performed under general anaesthesia and as a percutaneous procedure, using the femoral route or through the subclavian artery after cut-down. TAVI procedures took place in the cardiac catheterization laboratory. The mean length of stay at the hospital in patients undergoing TAVI was 8.8 days (SD 6.0) vs. 7.9 days (SD 4.7) after SAVR. One patient undergoing TAVI was discharged on the fourth postoperative day.21 Mean time in critical care was 20.7 h for patients after SAVR and 12.9 h for patients after TAVI.
A standard protocol for administering analgesics after SAVR was followed, using individual dosage of morphine or ketobemidone (intravenous administration in the intensive care unit (ICU) and subcutaneous in the ward) combined with paracetamol until the second postoperative day. From the second postoperative day oral codeine was given as required. Routinely, the SAVR patients remained in the thoracic surgery ICU for the first postoperative night. Provided there were no complications, they spent the second night at the step-down unit on the cardiothoracic surgery ward in a four-bed room and thereafter transferred to a standard 2–4 bed room on the ward. Patients having TAVI spent the first night in the cardiology observation unit in single rooms and, provided that no complications occurred, they were on the first postoperative day transferred to a standard 2–4 bed room on the cardiothoracic surgery ward.
Measures
Self-reported sleep was based on two items derived from the Uppsala Sleep Inventory, an instrument commonly used in sleep studies on older people.22 These were the number of hours of sleep per night, and how many hours the patient wished to sleep. The whole number of hours slept/needed, from four to 11, or the boundary values ‘3 or fewer hours’ and ‘more than 12 hours’.22 We also used the Minimal Insomnia Symptom Scale (MISS) to document sleep. The MISS is also a self-report tool that includes three items: difficulties initiating sleep, difficulties maintaining sleep, and difficulties with non-restorative sleep. Each item has five response categories, ranging from 0 to 4, with 0 indicating no difficulties and 4 indicating major difficulties. A sum score of 0–3 indicates no insomnia, 4–6 subclinical insomnia, 7–9 moderate insomnia, and 10–12 severe insomnia. The original version of the MISS is valid and reliable and commonly used among older people.23 Both instruments were translated from Swedish to Norwegian according to international guidelines.24 The Norwegian versions have not been tested for psychometric properties.
An Actiwatch 2 actigraph (Respironics, Philips Health Care, Best, Netherlands) was used to record sleep–wake patterns. The device has piezoelectric sensors which when placed on the patient’s non-dominant wrist measures the intensity and frequency of body movement. Recordings are then analysed by appropriate software to identify sleep and wakefulness.25 Actigraphy is a valid and reliable tool and is reported to have close to 90% agreement with polysomnography,26 but is easier and less costly to use.27 Actigraphy has previously been used successfully in cardiothoracic care settings.9 A standard algorithm was used, except for nocturnal sleep time, the latter of which was defined as 23:00 to 07:00 hours. Actigraphy data for daytime sleep on the day of surgery and on the fifth postoperative day were excluded from analysis, because the actigraph was put on and taken off at irregular times. The variables we analysed from the actigraphy recordings included total sleep time at night, sleep efficiency, wake time at night, and sleep time during day (between 07:00 and 23:00). The data were downloaded to a research computer and analysed using the software Respironics Actiware version 5. The software produced an actogram showing individual sleep–wake patterns.
We used the Charlson Comorbidity Index to quantitate comorbidities. It is a measure of comorbidity in which each of 17 different medical conditions is assigned a weight depending on the risk of mortality from that specific condition. Those weights are summarized to produce a total Charlson Comorbidity Index score, in which a higher score indicates a higher burden of comorbidity.28
The European System for Cardiac Operative Risk Evaluation (logistic EuroSCORE), was used to calculate predicted operative mortality for the patients undergoing cardiac surgery.29
Data collection
All data were collected by five research assistants trained to use the measures in the study in order to increase the reliability. They were nurses specialized in geriatric, cardiovascular or intensive care, and aware of the postoperative care routines and treatment performed. The research assistant visited the patient daily at noon, from the first to the fifth postoperative day, in order to assist the patient to fill out the questionnaires and to avoid any misunderstandings.
The patient medical records were used to record socio-demographic data and clinical variables at baseline, which are presented in Table 1.
Table 1.
Baseline socio-demographic and clinical characteristics of octogenarian patients scheduled for SAVR or TAVI (N = 143).
| Total | SAVR | TAVI | p-valuea | ||||
|---|---|---|---|---|---|---|---|
| Participants, N (%) | 143 | (100) | 78 | (55) | 65 | (45) | |
| Age, mean (SD) | 83 | (2.7) | 82 | (2.0) | 85 | (2.8) | < 0.001 |
| Sex, counts (%): | 0.21 | ||||||
| Men | 62 | (43) | 38 | (49) | 24 | (37) | |
| Women | 81 | (57) | 40 | (51) | 41 | (63) | |
| Education, counts (%): | 0.76 | ||||||
| Primary school | 99 | (69) | 52 | (67) | 47 | (72) | |
| Secondary school | 30 | (21) | 18 | (23) | 12 | (18) | |
| College/university | 14 | (10) | 8 | (10) | 6 | (9) | |
| Marital status, counts (%): | 0.27 | ||||||
| Married/cohabitation | 77 | (59) | 46 | (59) | 31 | (48) | |
| Unmarried | 4 | (1) | 1 | (1) | 3 | (5) | |
| Divorced | 1 | (1) | 1 | (1) | 0 | (0) | |
| Widow/widower | 61 | (38) | 30 | (38) | 31 | (48) | |
| NYHA, counts (%): | < 0.001 | ||||||
| I | 2 | (2) | 2 | (3) | 0 | (0) | |
| II | 46 | (36) | 35 | (49) | 11 | (20) | |
| III | 74 | (58) | 35 | (49) | 39 | (70) | |
| IV | 6 | (5) | 0 | (0) | 6 | (11) | |
| LVEF, counts (%): | 0.07 | ||||||
| < 30 | 3 | (2) | 2 | (3) | 1 | (2) | |
| 30–50 | 34 | (24) | 13 | (17) | 21 | (32) | |
| > 50 | 106 | (74) | 63 | (81) | 43 | (66) | |
| Aorta insufficiency, mean (SD) | 2.3 | (2.2) | 2.1 | (2.3) | 2.4 | (2.2) | 0.49 |
| Aortic valve area, mean (SD) | 0.4 | (0.2) | 0.4 | (0.2) | 0.4 | (0.1) | 0.23 |
| Aortic valve gradient, mean gradient mmHg, mean (SD) | 48 | (17) | 50 | (17) | 46 | (16) | 0.08 |
| Aortic valve gradient, max gradient mmHg, mean (SD) | 79 | (25) | 84 | (25) | 74 | (24) | 0.03 |
| Logistic euroSCORE, mean (SD) | 14 | (9) | 9 | (4) | 20 | (11) | < 0.001 |
| Charlson Comorbidity Index, mean (SD) | 2.1 | (1.2) | 1.8 | (1.0) | 2.5 | (1.3) | < 0.001 |
Pearson’s chi-square test, Fisher’s exact test, or Welch’s two-sample t-test, as appropriate.
LVEF: left ventricular ejection fraction; NYHA: New York Heart Association functional classification; SAVR: surgical aortic valve replacement; TAVI: transcatheter aortic valve implantation
Ethical considerations
The basis for conducting this study was the ethical principles in the Declaration of Helsinki.30 All patients provided informed written consent after they received oral and written information about the study. The study was approved by the Regional Committee for Ethics in Medical Research (2010/2936-6).
Statistical analysis
Descriptive statistics with means, standard deviations, medians, quartiles, frequencies and percentages were used to characterize the data. For comparing continuous and ordinal variables between treatment groups, Welch’s t-test and the Mann–Whitney test were used, respectively. For comparing sleep differences between days and nights (within patients), the Friedman rank sum test was used. For analysing the three sleep questionnaire items, a longitudinal linear model was fitted with an ARMA correlation structure, using generalized least squares. The ARMA parameters used were chosen based on the Akaike information criterion (AIC) of the possible models. Data management and initial statistical analyses were done in IBM SPSS Statistics version 21 (IBM Corp., Armonk, NY), while R version 3.1.131 was used for the reported statistical analyses. All results with p-values < 0.05 were considered statistically significant.
Results
Socio-demographic and clinical variables are presented in Table 1. The two treatment groups were similar with regard to sex, education and marital status. The TAVI patients were older, had a higher New York Heart Association (NYHA) functional class, higher scores regarding the Charlson comorbidity index and the logistic EuroSCORE, and lower maximal gradient across the aortic valve.
Self-reported sleep and insomnia
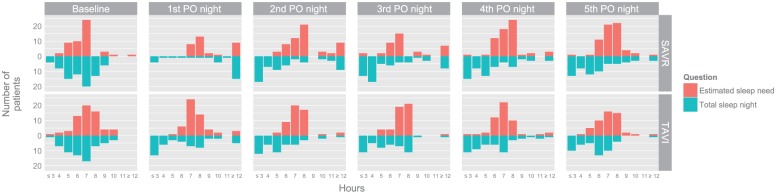
At baseline the patients estimated their sleep need as 7 h. The self-reported nocturnal sleep was 6.8 h (median 6 h) at baseline for all patients during the postoperative phase. Patients reported most nocturnal sleep time and most estimated sleep need the first postoperative night, and both decreased during the postoperative phase, with 5 h the second to the fourth postoperative night (Table 2 and Figure 1). There were significant differences between the SAVR and TAVI patients for the first postoperative night, where SAVR patients reported much longer needed and actual sleep times than TAVI patients.
Table 2.
Self-reported sleep and insomnia in octogenarian patients after SAVR or TAVI, at baseline, and on the first five postoperative nights (N = 143).
| Total (N = 143) | SAVR (n = 78) | TAVI (n = 65) | p-valuea | ||||
|---|---|---|---|---|---|---|---|
| Self-reported sleep, median hours | |||||||
| At baseline | |||||||
| Estimated sleep need | 7.0 | 7.5 | 7.0 | 0.87 | |||
| Total sleep night | 6.8 | 6.8 | 6.8 | 0.75 | |||
| 1st PO night | |||||||
| Estimated sleep need | 8.0 | 8.0 | 7.0 | < 0.001 | |||
| Total sleep night | 7.0 | > 11 | 6.3 | < 0.001 | |||
| 2nd PO night | |||||||
| Estimated sleep need | 7.8 | 8.0 | 7.0 | 0.05 | |||
| Total sleep night | 5.0 | 5.0 | 5.0 | 0.46 | |||
| 3rd PO night | |||||||
| Estimated sleep need | 8.0 | 8.0 | 7.0 | 0.13 | |||
| Total sleep night | 5.0 | 5.0 | 6.0 | 0.68 | |||
| 4th PO night | |||||||
| Estimated sleep need | 7.0 | 7.3 | 7.0 | 0.07 | |||
| Total sleep night | 5.0 | 5.0 | 5.8 | 0.75 | |||
| 5th PO night | |||||||
| Estimated sleep need | 7.0 | 7.0 | 7.0 | 0.21 | |||
| Total sleep night | 6.0 | 5.0 | 6.0 | 0.91 | |||
| Insomnia, counts (%) | |||||||
| At baseline | 0.98 | ||||||
| No insomnia | 94 | (66) | 52 | (67) | 42 | (66) | |
| Subclinical insomnia | 35 | (25) | 18 | (23) | 17 | (27) | |
| Moderate insomnia | 12 | (8) | 7 | (9) | 5 | (8) | |
| Severe insomnia | 1 | (1) | 1 | (1) | 0 | (0) | |
| (Missing) | 1 | 0 | 1 | ||||
| 1st PO night | < 0.001 | ||||||
| No insomnia | 63 | (62) | 35 | (76) | 28 | (50) | |
| Subclinical insomnia | 20 | (20) | 7 | (15) | 13 | (23) | |
| Moderate insomnia | 15 | (15) | 4 | (9) | 11 | (20) | |
| Severe insomnia | 4 | (4) | 0 | (0) | 4 | (7) | |
| (Missing) | 9 | 32 | 41 | ||||
| 2nd PO night | 0.33 | ||||||
| No insomnia | 41 | (36) | 25 | (40) | 16 | (31) | |
| Subclinical insomnia | 35 | (31) | 18 | (29) | 17 | (33) | |
| Moderate insomnia | 20 | (18) | 7 | (11) | 13 | (25) | |
| Severe insomnia | 18 | (16) | 12 | (19) | 6 | (12) | |
| (Missing) | 13 | 16 | 29 | ||||
| 3rd PO night | 0.24 | ||||||
| No insomnia | 49 | (41) | 24 | (35) | 25 | (49) | |
| Subclinical insomnia | 32 | (27) | 19 | (28) | 13 | (25) | |
| Moderate insomnia | 25 | (21) | 19 | (28) | 6 | (12) | |
| Severe insomnia | 13 | (11) | 6 | (9) | 7 | (14) | |
| (Missing) | 14 | 10 | 24 | ||||
| 4th PO night | 0.08 | ||||||
| No insomnia | 52 | (43) | 24 | (38) | 28 | (50) | |
| Subclinical insomnia | 30 | (25) | 14 | (22) | 16 | (29) | |
| Moderate insomnia | 23 | (19) | 17 | (27) | 6 | (11) | |
| Severe insomnia | 15 | (12) | 9 | (14) | 6 | (11) | |
| (Missing) | 9 | 14 | 23 | ||||
| 5th PO night | 0.09 | ||||||
| No insomnia | 58 | (49) | 28 | (43) | 30 | (56) | |
| Subclinical insomnia | 24 | (20) | 12 | (18) | 12 | (22) | |
| Moderate insomnia | 19 | (16) | 15 | (23) | 4 | (7) | |
| Severe insomnia | 18 | (15) | 10 | (15) | 8 | (15) | |
| (Missing) | 11 | 13 | 24 | ||||
Values are presented as medians or counts (percentages). Total: the total sample of SAVR and TAVI patients. The number of patients varied between the different nights: 1st PO night: TAVI 50–54, SAVR 30–33; 2nd PO night: TAVI 47–51, SAVR 58–59; 3rd PO night: TAVI 49–50, SAVR 61–66; 4th PO night: TAVI 52, SAVR 62; 5th PO night: TAVI 49–51, SAVR 63.
All p-values are based on Mann–Whitney tests on the original (non-categorized) values.
PO: postoperative; SAVR: surgical aortic valve replacement; TAVI: transcatheter aortic valve implantation.
Figure 1.
Self-reported sleep and sleep need in octogenarian patients after SAVR or TAVI at baseline and on the first five postoperative nights (N = 143).
Some patients reported fractional number of hours. These are rounded down (e.g. 4.5 becomes 4) in this graph.
PO: postoperative; SAVR: surgical aortic valve replacement; TAVI: transcatheter aortic valve implantation.
At baseline, 9% of the patients had self-reported insomnia (moderate-to-severe insomnia scores) which worsened to 31–34% postoperatively (Table 2); there were no differences between the SAVR and TAVI patients on this variable. Self-reported insomnia was lowest on the first postoperative night (in total 19%, with 9% for SAVR and 27% for TAVI), but increased on the following nights. On the first postoperative night, the SAVR patients had less self-reported insomnia than the TAVI patients, although this also increased and varied during the postoperative phase. On the second postoperative night, the TAVI patients had the highest self-reported insomnia (37%), which decreased during the subsequent nights (Table 2).
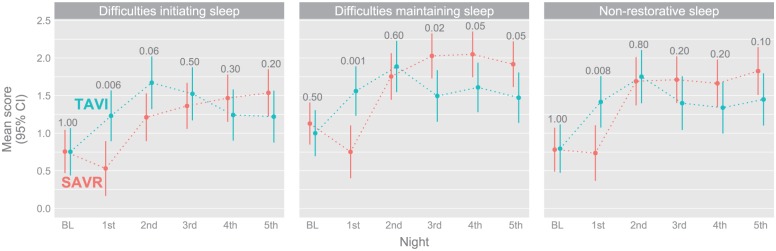
The patients reported more difficulties in initiating sleep, difficulties maintaining sleep and more problems with non-restorative sleep during the postoperative phase compared with baseline. The only exception was the SAVR patients on the first postoperative night (Figure 2). The SAVR patients also had somewhat fewer difficulties maintaining sleep than the TAVI patients for the last three postoperative nights. For both groups, there were few sleep differences from the second postoperative night and onward.
Figure 2.
Difficulties initiating sleep, difficulties maintaining sleep, and non-restorative sleep in octogenarian patients after SAVR or TAVI at baseline and on the first five postoperative nights.
Estimated mean scores, with 95% CIs, based on a longitudinal model. The number of patients varied between the different nights and the three questions. At baseline: TAVI 64–65, SAVR 78; 1st PO night: TAVI 56, SAVR 46–48; 2nd PO night: TAVI 52, SAVR 63; 3rd PO night: TAVI 51, SAVR 68; 4th PO night: TAVI 56, SAVR 65–66; 5th PO night: TAVI 54, SAVR 65–66.
CI: confidence interval; PO: postoperative; SAVR: surgical aortic valve replacement; TAVI: transcatheter aortic valve implantation.
Sleep–wake pattern
Median total sleep time for all patients at night measured by actigraphy was 381 min, median sleep efficiency was 79%, and median wake time at night was 43 min for the entire five postoperative nights. For all sleep and wake variables, there were significant differences between the postoperative nights (all p-values < 0.003). The shortest length of total sleep time and the lowest sleep efficiency was observed on the fifth postoperative night (Table 3).
Table 3.
Objective sleep–wake patterns including total sleep time, wake time at night, and sleep time during day in octogenarian patients after SAVR or TAVI (n = 143).
|
Total (N=143) |
SAVR (n=78) |
TAVI (n=65) |
p-valuea | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Median | Quartiles | Median | Quartiles | Median | Quartiles | |||||
| Total sleep time – night, min | ||||||||||
| 1st PO night | 401 | 325 | 438 | 416 | 320 | 442 | 385 | 332 | 432 | 0.42 |
| 2nd PO night | 392 | 293 | 434 | 400 | 322 | 445 | 372 | 286 | 410 | 0.05 |
| 3rd PO night | 376 | 292 | 419 | 359 | 264 | 410 | 380 | 307 | 423 | 0.45 |
| 4th PO night | 383 | 298 | 421 | 382 | 298 | 420 | 384 | 293 | 421 | 0.95 |
| 5th PO night | 370 | 327 | 422 | 356 | 326 | 421 | 396 | 328 | 424 | 0.48 |
| Sleep efficiency, % | ||||||||||
| 1st PO night | 84 | 68 | 91 | 86 | 67 | 92 | 80 | 69 | 90 | 0.45 |
| 2nd PO night | 82 | 61 | 91 | 83 | 67 | 93 | 78 | 60 | 85 | 0.04 |
| 3rd PO night | 78 | 60 | 87 | 75 | 55 | 86 | 80 | 64 | 88 | 0.44 |
| 4th PO night | 80 | 62 | 88 | 80 | 62 | 88 | 80 | 61 | 88 | 0.95 |
| 5th PO night | 77 | 68 | 88 | 74 | 68 | 87 | 82 | 68 | 88 | 0.38 |
| Wake time – night, min | ||||||||||
| 1st PO night | 30 | 17 | 55 | 27 | 17 | 48 | 40 | 21 | 72 | 0.03 |
| 2nd PO night | 39 | 19 | 68 | 27 | 19 | 60 | 44 | 22 | 70 | 0.16 |
| 3rd PO night | 46 | 30 | 78 | 52 | 32 | 83 | 44 | 29 | 75 | 0.29 |
| 4th PO night | 41 | 24 | 72 | 42 | 20 | 66 | 40 | 27 | 72 | 0.63 |
| 5th PO night | 46 | 26 | 71 | 46 | 26 | 75 | 40 | 27 | 66 | 0.96 |
| Sleep time – day, min | ||||||||||
| 1st PO day | 665 | 559 | 772 | 744 | 615 | 824 | 592 | 507 | 676 | < 0.01 |
| 2nd PO day | 561 | 452 | 684 | 574 | 513 | 754 | 514 | 394 | 650 | 0.001 |
| 3rd PO day | 536 | 420 | 674 | 568 | 436 | 734 | 502 | 379 | 653 | 0.02 |
| 4th PO day | 503 | 397 | 643 | 552 | 434 | 653 | 439 | 382 | 601 | 0.02 |
Total: the total sample of SAVR and TAVI patients.
All p-values are based on Mann–Whitney tests. The number of patients varied between the different nights: 1st PO night: TAVI 54, SAVR 63–64; 2nd PO night: TAVI 54, SAVR 63–64; 3rd PO night: TAVI 54, SAVR 62–64; 4th PO night: TAVI 53, SAVR 60–61; 5th PO night: TAVI 45–51, SAVR 58–60.
PO: postoperative; SAVR: surgical aortic valve replacement; TAVI: transcatheter aortic valve implantation.
Median sleep time during the day was 570 min for the first four postoperative days. The patients slept the most on the first postoperative day and the least on the fourth postoperative day, with a significant difference between the days (p < 0.001). There were significant differences between the patient groups, with the SAVR patients sleeping more than the TAVI patients for all the days (Table 3).
Discussion
To the best of our knowledge, this is the first study to systematically determine self-reported sleep and insomnia, with recorded sleep–wake patterns during the complete in-hospital postoperative phase in patients after TAVI. Octogenarians undergoing SAVR or TAVI had disturbed self-reported sleep and increased insomnia during the postoperative phase compared with baseline. SAVR patients experienced most disturbed sleep during the late postoperative phase (i.e. second postoperative night and later). Actigraphy of sleep–wake patterns revealed that sleep was poor during all postoperative nights, and that the patients slept more during daytime compared with at night. SAVR patients had more disturbed sleep–wake patterns during the later postoperative phase, while the TAVI patients displayed more disturbed sleep–wake patterns on the first postoperative day.
The present results that quantified disrupted sleep with actigraphy and sleep self-reports correspond with those of previous reports on self-reported sleep after cardiac surgery. These reports showed that sleep was severely disturbed and insomnia increased.9 However, the patients of previous studies experienced the most severe insomnia during the first postoperative night.9,20 This is in contrast to the present study, which found that octogenarian patients had few self-reported sleep disturbances and obtained the lowest insomnia scores on the first postoperative night (Table 2). This finding should be considered in light of a study by Bihari et al.,32 where older ICU patients slept better than younger patients.
In the present study, the amount of self-reported sleep was highest for the first postoperative night, particularly for the SAVR patients. We believe that octogenarians likely felt more comfortable and less anxious with more available nursing staff, as in the ICU, but were more anxious with fewer staff present, as was the case on the surgical ward.32 The impact of the duration of treatment is also important as the longer the treatment time in the operation room, the more the patient will be affected by sleep disorders.33,34 Furthermore, the analgesics used during the postoperative phase is an important factor as octogenarians are usually more sedated than younger patients.32,35
Disturbed sleep after surgery is, in general, related to surgical trauma, in which the duration of each step of the surgery is important.34 Particularly, cardiopulmonary bypass circulation is assumed to be an influential factor on sleep quality.9 However, our results do not support this as TAVI patients reported the most disturbed sleep and insomnia during the early postoperative phase, while SAVR patients experienced a higher degree of sleep disturbances and most insomnia in the late postoperative phase (Figure 2). Analgesic use is a factor that affects sleep architecture.36 This might explain why the SAVR patients, who received opioids on the first two postoperative nights, reported more sleep during the early postoperative phase than the TAVI patients.35 Pain is reported to have an impact on postoperative sleep,9,16 and as the analgesics were reduced for the SAVR patients during the postoperative phase, their sleep quality was negatively affected.
Wake time at night, as measured by actigraphy, was negatively affected during the first night and varied during the postoperative phase (Table 3). These results are comparable to those of previous post-cardiac surgery studies.9 Poor sleep is defined as a sleep efficiency of <85%.26 The octogenarians in this study had a median sleep efficiency of 79% for the postoperative phase. In previous post-cardiac surgery studies, sleep efficiency varied between 58% and 68%.9 In contrast to previous data on decreased sleep in the ICU,9,11,37 data obtained in the present study revealed that the octogenarians had better scores for all sleep–wake variables assessed on the first postoperative night than for the rest of the postoperative phase. This indicates that they experienced better sleep in the ICU than in the cardiothoracic surgical ward. However, this should be viewed in the context of the effects of surgical anaesthesia.38 On the first postoperative night, the patients were still sedated after anaesthesia, which might have affected their sleep.33,34 Since the patients were sedated and moving less the first night, the actigraph may have registered this falsely as sleep. One disadvantage with actigraphy is that it may overestimate total sleep time and underestimate wake time among patients with fragmented sleep and when patients lie relatively motionless in bed.39 Especially old patients and patients with hypoactive delirium may lie still at night.32,37 The incidence of postoperative delirium was high in this group of patients.21
An important finding in this study was the amount of total sleep time, and that the octogenarians slept the most during daytime. Daytime sleep has been associated with little or no restorative sleep,39 and less functional recovery.37 However, reduced sleep at night can result in low energy during daytime;40 therefore, it has been assumed that it is important to let patients sleep during the day.9 Sleep disturbances at night may negatively affect the possibility for getting deep, restorative sleep.9,11 Disrupting factors for this type of sleep might be the hospital environment in general,37 with noise and light being especially disruptive,41 as well as interventions from health-care personnel.32 It is also well known that many older people take a daytime nap.27 Nevertheless, this does not explain the length of sleep time during the day in the present study, in which the patients slept more during the day than at night. This result differs from results of previous post-cardiac surgery studies, in which patients slept 1.7–4.4 h, up to 50% of the total sleep time, during the day, but not more than during the night.9,11 In comparison, stroke patients slept for 1.4 h,39 and in post-acute rehabilitation, older patients slept 2.1 h a day.40 Patients with severe aortic stenosis may suffer from different symptoms (e.g. dyspnoea and nocturia due to heart failure, chest pain and vertigo) prior to surgery. This can cause secondary insomnia and a disturbed sleep–wake pattern (e.g. increased daytime napping).16,19 TAVI patients had the most disturbed sleep–wake pattern throughout the postoperative phase, while SAVR patients slept most both during day and night. Medical devices, including thoracic drainage and central and peripheral vascular lines may prevent SAVR patients from moving during the first postoperative nights. This may contribute even more as a sleep-disrupting factor after the level of sedation and analgesics has been lowered. This points to the possibility that for octogenarians, TAVI is less discomforting than SAVR during the early postoperative phase. However, it is possible that the patients’ sleep would improve by going home earlier.42 Although, to our knowledge, no studies have yet been published on this issue.
Limitations
A limitation of the present study is that we did not record whether the patients slept in a shared room, and thereby that sleep potentially was disturbed by other patients. To reduce burden to the patients, we tried to include as few questionnaire items as possible; therefore we used two items drawn from the Uppsala Sleep Inventory to measure self-reported sleep, an instrument commonly used in the elderly. Actigraphy has been found to be reliable and valid in comparable studies.9 However, as noted, it can overestimate total sleep time and underestimate wake time. Polysomnography including electroencephalogram would have documented sleep–wake patterns more precisely, but it is a more resource-demanding measurement that can be difficult to use in an intensive care setting, especially for elderly patients. Another limitation is that baseline data of pre-hospital sleep patterns was only obtained by subjective data, as compared with postoperative data, which included both subjective and objective measurements.
Conclusion
The octogenarians undergoing SAVR or TAVI reported sleep disturbances with more sleep time during day than at night, but SAVR patients slept more overall than TAVI patients. However, the treatment modalities displayed a different pattern throughout the postoperative phase, as SAVR patients slept more in the early phase while TAVI patients slept more in the later phase.
Acknowledgments
We thank the patients who participated in the study and our colleagues at the thoracic surgical ward. We thank the Kavli Research Centre for Geriatrics and Dementia, the Norwegian Nurses Association, and Bergen University College for supporting the data collection.
Footnotes
Conflict of interest: The authors declare that there is no conflict of interest.
Funding: The CARDELIR study received fuding from the Kavli Research Center for Aging and Dementia, University of Bergen, Bergen University College and the Norwegian Nurses Association.
Implications for practice
- Both patients after transcatheter aortic valve implantation and patients after surgical aortic valve replacement have documented sleep disturbance following treatment, and treatment of insomnia is a challenge in the early postoperative phase.
- Organizing interventions to improve sleep (e.g. focusing on sleep hygiene; with light, noise, temperature, analgesics, availability of nurses and sleeping routines) in order to reduce the time to recovery and length of hospital stay is of importance.
- Nurses should encourage daytime mobilization, and provide information on the negative effect of immobility and too much daytime napping.
- The study carries important messages for clinical care related to sleep and postoperative mobility, since octogenarians are vulnerable and will need more care than younger patients.
References
- 1. Forman DE, Rich MW, Alexander KP, et al. Cardiac care for older adults. Time for a new paradigm. J Am Coll Cardiol 2011; 57: 1801–1810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Vahanian A, Alfieri O, Andreotti F, et al. Guidelines on the management of valvular heart disease (version 2012): The Joint Task Force on the Management of Valvular Heart Disease of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS). Eur J Cardiothorac Surg 2012; 42: S1–S44. [DOI] [PubMed] [Google Scholar]
- 3. Otto CM, Prendergast B. Aortic-valve stenosis – from patients at risk to severe valve obstruction. N Engl J Med 2014; 371: 744–756. [DOI] [PubMed] [Google Scholar]
- 4. Finn M, Green P. Transcatheter aortic valve implantation in the elderly: Who to refer? Prog Cardiovasc Dis 2014; 57: 215–225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Smith CR, Leon MB, Mack MJ, et al. Transcatheter versus surgical aortic-valve replacement in high-risk patients. N Engl J Med 2011; 364: 2187–2198. [DOI] [PubMed] [Google Scholar]
- 6. Lam BK, Hendry PJ. Patients over 80 years: Quality of life after aortic valve replacement. Age Ageing 2004; 33: 307–309. [DOI] [PubMed] [Google Scholar]
- 7. Minutello RM, Wong SC, Swaminathan RV, et al. Costs and in-hospital outcomes of transcatheter aortic valve implantation versus surgical aortic valve replacement in commercial cases using a propensity score matched model. Am J Cardiol 2015; 115: 1443–1447. [DOI] [PubMed] [Google Scholar]
- 8. Magee CA, Caputi P, Iverson DC. Relationships between self-rated health, quality of life and sleep duration in middle aged and elderly Australians. Sleep Med 2011; 12: 346–350. [DOI] [PubMed] [Google Scholar]
- 9. Liao WC, Huang CY, Huang TY, et al. A systematic review of sleep patterns and factors that disturb sleep after heart surgery. J Nurs Res 2011; 19: 275–288. [DOI] [PubMed] [Google Scholar]
- 10. Tranmer JE, Minard J, Fox LA, et al. The sleep experience of medical and surgical patients. Clin Nurs Res 2003; 12: 159–173. [DOI] [PubMed] [Google Scholar]
- 11. Hsu SM, Ko WJ, Liao WC, et al. Associations of exposure to noise with physiological and psychological outcomes among post-cardiac surgery patients in ICUs. Clinics (Sao Paulo) 2010; 65: 985–989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Cappuccio FP, D’Elia L, Strazzullo P, et al. Sleep duration and all-cause mortality: A systematic review and meta-analysis of prospective studies. Sleep 2010; 33: 585–592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Martin JL, Fung CH. Quality indicators for the care of sleep disorders in vulnerable elders. J Am Geriatr Soc 2007; 55(Suppl. 2): S424–S430. [DOI] [PubMed] [Google Scholar]
- 14. Roepke SK, Ancoli-Israel S. Sleep disorders in the elderly. Indian J Med Res 2010; 131: 302–310. [PubMed] [Google Scholar]
- 15. Lee CY, Low LP, Twinn S. Understanding the sleep needs of older hospitalized patients: A review of the literature. Contemp Nurse 2005; 20: 212–220. [DOI] [PubMed] [Google Scholar]
- 16. Chen Q, Hayman LL, Shmerling RH, et al. Characteristics of chronic pain associated with sleep difficulty in older adults: The Maintenance of Balance, Independent Living, Intellect, and Zest in the Elderly (MOBILIZE) Boston study. J Am Geriatr Soc 2011; 59: 1385–1392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Redeker NS. Sleep disturbance in people with heart failure: Implications for self-care. J Cardiovasc Nurs 2008; 23: 231–238. [DOI] [PubMed] [Google Scholar]
- 18. Chen HM, Clark AP, Tsai LM, et al. Self-reported sleep disturbance of patients with heart failure in Taiwan. Nurs Res 2009; 58: 63–71. [DOI] [PubMed] [Google Scholar]
- 19. Brostrom A, Stromberg A, Dahlstrom U, et al. Sleep difficulties, daytime sleepiness, and health-related quality of life in patients with chronic heart failure. J Cardiovasc Nurs 2004; 19: 234–242. [DOI] [PubMed] [Google Scholar]
- 20. Egerod I, Nielsen S, Lisby KH, et al. Immediate post-operative responses to transcatheter aortic valve implantation: An observational study. Eur J Cardiovasc Nurs 2015; 14: 232–239. [DOI] [PubMed] [Google Scholar]
- 21. Eide LS, Ranhoff AH, Fridlund B, et al. Comparison of frequency, risk factors, and time course of postoperative delirium in octogenarians after transcatheter aortic valve implantation versus surgical aortic valve replacement. Am J Cardiol 2015; 115: 802–809. [DOI] [PubMed] [Google Scholar]
- 22. Brostrom A, Hubbert L, Jakobsson P, et al. Effects of long-term nocturnal oxygen treatment in patients with severe heart failure. J Cardiovasc Nurs 2005; 20: 385–396. [DOI] [PubMed] [Google Scholar]
- 23. Hellstrom A, Hagell P, Fagerstrom C, et al. Measurement properties of the Minimal Insomnia Symptom Scale (MISS) in an elderly population in Sweden. BMC Geriatr 2010; 10: 84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Brislin RW. Back-translation for cross-cultural research. J Cross Cult Psychol 1970; 1: 185–216. [Google Scholar]
- 25. Martin JL, Hakim AD. Wrist actigraphy. Chest 2011; 139: 1514–1527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Sadeh A, Acebo C. The role of actigraphy in sleep medicine. Sleep Med Rev 2002; 6: 113–124. [DOI] [PubMed] [Google Scholar]
- 27. Goldman SE, Hall M, Boudreau R, et al. Association between nighttime sleep and napping in older adults. Sleep 2008; 31: 733–740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation. J Chronic Dis 1987; 40: 373–383. [DOI] [PubMed] [Google Scholar]
- 29. Pillai BS, Baloria KA, Selot N. Validation of the European System for Cardiac Operative Risk Evaluation-II model in an urban Indian population and comparison with three other risk scoring systems. Ann Card Anaesth 2015; 18: 335–342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. World Medical Association. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA 2000; 284: 3043–3045. [PubMed] [Google Scholar]
- 31. R Core Team. R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing, 2014. [Google Scholar]
- 32. Bihari S, Doug McEvoy R, Matheson E, et al. Factors affecting sleep quality of patients in intensive care unit. J Clin Sleep Med 2012; 8: 301–307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Loadsman JA, Hillman DR. Anaesthesia and sleep apnoea. Br J Anaesth 2001; 86: 254–266. [DOI] [PubMed] [Google Scholar]
- 34. Rosenberg-Adamsen S, Kehlet H, Dodds C, et al. Postoperative sleep disturbances: Mechanisms and clinical implications. Br J Anaesth 1996; 76: 552–559. [DOI] [PubMed] [Google Scholar]
- 35. Pergolizzi J, Boger RH, Budd K, et al. Opioids and the management of chronic severe pain in the elderly: Consensus statement of an International Expert Panel with focus on the six clinically most often used World Health Organization Step III opioids (buprenorphine, fentanyl, hydromorphone, methadone, morphine, oxycodone). Pain Pract 2008; 8: 287–313. [DOI] [PubMed] [Google Scholar]
- 36. Cronin AJ, Keifer JC, Davies MF, et al. Postoperative sleep disturbance: Influences of opioids and pain in humans. Sleep 2001; 24: 39–44. [DOI] [PubMed] [Google Scholar]
- 37. Patel M, Chipman J, Carlin BW, et al. Sleep in the intensive care unit setting. Crit Care Nurs Q 2008; 31: 309–318; quiz 19–20. [DOI] [PubMed] [Google Scholar]
- 38. Karkela J, Vakkuri O, Kaukinen S, et al. The influence of anaesthesia and surgery on the circadian rhythm of melatonin. Acta Anaesthesiol Scand 2002; 46: 30–36. [DOI] [PubMed] [Google Scholar]
- 39. Bakken LN, Lee KA, Kim HS, et al. Sleep–wake patterns during the acute phase after first-ever stroke. Stroke Res Treat 2011; 2011: 936298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Alessi CA, Martin JL, Webber AP, et al. More daytime sleeping predicts less functional recovery among older people undergoing inpatient post-acute rehabilitation. Sleep 2008; 31: 1291–1300. [PMC free article] [PubMed] [Google Scholar]
- 41. Lane T, East LA. Sleep disruption experienced by surgical patients in an acute hospital. Br J Nurs 2008; 17: 766–771. [DOI] [PubMed] [Google Scholar]
- 42. Lauck SB, Wood DA, Achtem L, et al. Risk stratification and clinical pathways to optimize length of stay after transcatheter aortic valve replacement. Can J Cardiol 2014; 30: 1583–1587. [DOI] [PubMed] [Google Scholar]