Abstract
Two novel cell-wall-less, acidophilic, mesophilic, organotrophic and facultatively anaerobic archaeal strains were isolated from acidic streamers formed on the surfaces of copper-ore-containing sulfidic deposits in south-west Spain and North Wales, UK. Cells of the strains varied from 0.1 to 2 μm in size and were pleomorphic, with a tendency to form filamentous structures. The optimal pH and temperature for growth for both strains were 1.0–1.2 and 37–40 °C, with the optimal substrates for growth being beef extract (3 g l− 1) for strain S5T and beef extract with tryptone (3 and 1 g l− 1, respectively) for strain PM4. The lipid composition was dominated by intact polar lipids consisting of a glycerol dibiphytanyl glycerol tetraether (GDGT) core attached to predominantly glycosidic polar headgroups. In addition, free GDGT and small relative amounts of intact and core diether lipids were present. Strains S5T and PM4 possessed mainly menaquinones with minor fractions of thermoplasmaquinones. The DNA G+C content was 37.3 mol% in strain S5T and 37.16 mol% for strain PM4. A similarity matrix of 16S rRNA gene sequences (identical for both strains) showed their affiliation to the order Thermoplasmatales, with 73.9–86.3 % identity with sequences from members of the order with validly published names. The average nucleotide identity between genomes of the strains determined in silico was 98.75 %, suggesting, together with the 16S rRNA gene-based phylogenetic analysis, that the strains belong to the same species. A novel family, Cuniculiplasmataceae fam. nov., genus Cuniculiplasma gen. nov. and species Cuniculiplasma divulgatum sp. nov. are proposed based on the phylogenetic, chemotaxonomic analyses and physiological properties of the two isolates, S5T and PM4 ( = JCM 30641 = VKM B-2940). The type strain of Cuniculiplasma divulgatum is S5T ( = JCM 30642T = VKM B-2941T).
The first genus and species of the Thermoplasmatales, Thermoplasma acidophilum, was described in 1970 (Darland et al., 1970), followed 25 years later by the genus Picrophilus (Schleper et al., 1995). Since then, the taxa Ferroplasma (Golyshina et al., 2000), Thermogymnomonas acidicola (Itoh et al., 2007) and Acidiplasma (Golyshina et al., 2009) were described in the first decade of the 21st century. All members of the Thermoplasmatales are characterized as micro-organisms that are difficult to isolate, having extremely acidic pH optima for growth (among the lowest known), with their cells being typically pleomorphic (with the only exception being members of Picrophilus), as a consequence of the lack of an intact cell wall.
In recent years, a number of extensive studies, mostly based on analysis of metagenomic data, have suggested a wide distribution of the Thermoplasmatales in low-pH environments. A number of lineages of uncultured euryarchaea affiliated with this group have been suggested, along with the prediction of their metabolic traits (Baker et al., 2006; Yelton et al., 2013). However, these studies have not led to success in isolation and taxonomic description of the novel archaeal taxa predicted in these environments.
Here, we report the isolation and characterization of strains representing a new family, a new genus and a novel species within the order Thermoplasmatales.
The sources of isolation of strains S5T and PM4 were acidic waters from the surfaces of copper mine sites taken from Cantareras, Spain, and Mynydd Parys, Anglesey, UK, in April and March 2011, respectively.
For enrichment cultures, modified medium 88 (DSMZ) was used, which contained (g l− 1): (NH4)2SO4, 1.3; KH2PO4, 0.28; MgSO4 . 0.7H2O, 0.25; CaCl2 . 2H2O, 0.07; FeCl3 . 6H2O, 0.02. Other salts in medium 88 were replaced with the trace element solution SL-10 from DSMZ medium 320 at a concentration of 1 : 1000. Beef extract (3 g l− 1) (with the addition of 1 g tryptone l− 1 for strain PM4), 0.06 % betaine and Kao and Michayluk's vitamin solution (Sigma-Aldrich) at 1 : 100 (v/v) were added to the medium. The medium was adjusted to pH 1.0–1.2 with concentrated H2SO4. The cultures were incubated for about 5 days at 37 °C with shaking at 120 r.p.m. Extinction to dilution was used for isolation of pure cultures, which were monitored by phase-contrast microscopic examination and by PCR amplification with bacteria- and archaea-specific primers, as described previously (Golyshina et al., 2009). The purified strains were cultured in 100 ml flasks in 20 ml medium with shaking at 120 r.p.m. Growth was monitored by measurements of the OD600 in a BioPhotometer Plus (Eppendorf).
Total lipid extracts were obtained from wet-frozen cell pellet samples using a modified Bligh and Dyer protocol (Sturt et al., 2004), after addition of an internal standard (phosphatidylcholine C21 : 0/C21 : 0) and 3–5 g pre-combusted sand. The samples were treated with a mixture of dichloromethane (DCM)/methanol/buffer (1 : 2 : 0.8, by vol.) and ultrasonicated, using an ultrasonic probe, for 10 min during each step. This procedure was performed in four steps, using phosphate buffer (K2HPO4, 50 mM at pH 7.4) for the first two steps and trichloroacetic acid buffer (50 g l− 1, pH 2) for the last two steps. After sonication, the mixture was centrifuged (10 min at 2000 r.p.m.) and the supernatant was collected in a separatory funnel. DCM and deionized Milli-Q water (1 : 1, v/v) were added to the combined supernatants, in a ratio of DCM/methanol/buffer of 2 : 1 : 0.8 (by vol.). Thereafter, the organic phase was separated and the remaining aqueous phase was washed three times with DCM. The collected organic phase (total lipid extract) was subsequently washed three times with Milli-Q water and afterwards reduced to dryness under a stream of nitrogen at a constant temperature of 23 °C. The obtained total lipid extract was stored at − 20 °C for further analysis.
Analysis of the archaeal lipid composition was performed on the Dionex Ultimate 3000 UHPLC coupled to the Bruker maXis Ultra High Resolution tandem qToF equipped with electrospray interface (ESI).
We applied a recently developed reversed-phase HPLC-ESI-MS method, with analyte separation on an ACE3 C18 column (2.1 × 150 mm, 3 μm particle size; Advanced Chromatography Technologies) maintained at 45 °C at a flow rate of 0.2 ml min− 1 isocratically for 10 min with 100 % eluent A (methanol/formic acid/14.8 M NH4OH; 100 : 0.04 : 0.10, by vol.), followed by a linear gradient to 24 % eluent B (2-propanol/formic acid/14.8 M NH4OH; 100 : 0.04 : 0.10, by vol.) in 5 min, followed by a gradient to 65 % B in 55 min. The column was then flushed with 90 % B for 10 min and re-equilibrated with 100 % A for 10 min (Zhu et al., 2013). Lipid identification was based on retention time as well as accurate molecular mass and isotope pattern match with known sum formulas in full scan mode and MS2 fragment spectra. Lipid quantification was achieved by injecting an internal standard (C46-GTGT) along with the samples. Lipid abundances were corrected for response factors of commercially available as well as purified standards.
Mid-exponential phase-grown cells were fixed with glutardialdehyde and prepared for electron microscopic analysis as described in detail by Golyshina et al. (2000). Electron micrographs were recorded as zero-loss images with a 2k × 2k cooled CCD (SharpEye; Tröndle), bottom-mounted to an integrated energy-filtered transmission electron microscope Libra 120plus (Zeiss), operated at 120 kV acceleration voltage and an energy-slit width of 10 eV.
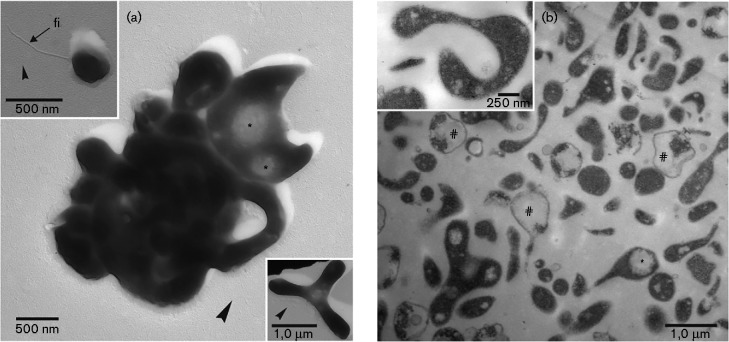
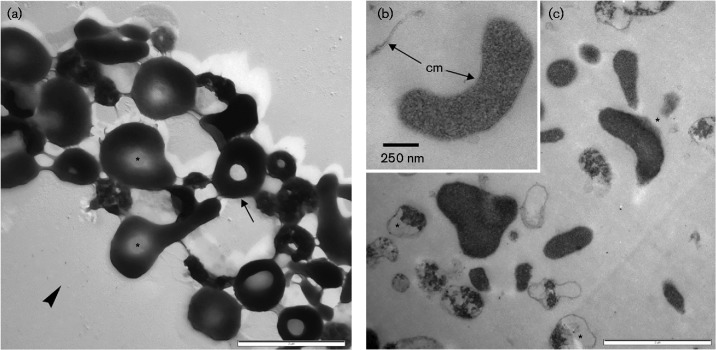
Both isolates PM4 and S5T lacked a cell wall, as revealed from ultrathin sections (Figs 1 and 2), and showed a pronounced pleomorphism, which resulted in the formation of ring- and Y-like cell forms, along with other morphotypes. In general, the cytoplasm was rather dense and was accompanied by electron-translucent voids (Figs 1 and 2; asterisks). A distinct surface coat was missing, and patch-like dark deposits were observed only occasionally on the surface (data not shown). The cytoplasmic membrane had a thickness of 5.30 ± 1.01 nm (n = 42; min. 3.61 nm; max. 7.01 nm) in the case of strain PM4 and 5.61 ± 0.90 nm (n = 41; min. 4.31 nm; max. 7.76 nm) for strain S5T. The thickness of the cytoplasmic membrane plus the outer electron-dense, discontinuous layer was 13.39 ± 1.94 nm (n = 15; min. 9.68 nm; max. 16.40 nm) for strain S5T and 14.38 ± 1.98 nm (n = 15; min. 11.23 nm; max. 19.12 nm) for strain PM4. Upon prolonged growth, cellular ghosts or membrane remnants became predominant. Here, especially with strain PM4, filamentous appendices could be observed, which may represent flaccid fimbria.
Fig. 1.
Electron micrographs of cells of strain PM4 (4 days of growth). (a) Tight cluster of cells, showing general pleomorphism. Occasionally, cells are spherical (upper inset) or appear Y-shaped (lower inset). Appendices can be seen as fimbriae-like structures (see upper inset; fi). Arrowheads indicate direction of shadow casting. (b) Ultrathin sections show the rather obvious cell pleomorphism and, in detail (inset), it is seen that only a unit membrane surrounds the cytoplasm. The cytoplasm is rather dense but often contains electron-translucent areas of different sizes, which in general represent voids (*). Ghost-like remnants of cells (#) are present.
Fig. 2.
Ultrastructure of strain S5T. (a) Cluster of cells, which appear rather pleomorphic when prepared as whole-mount cells and PtC-shadow-contrasted (arrowhead, direction of shadow-cast; arrow, ring-shaped cell). When seen in ultrathin sections (b, c), cells are surrounded by a single unit membrane (cm) and in general show a densely packed cytoplasm without distinct inclusions.
The strains grew at 10–45 °C (PM4) and 10–48 °C (S5T); weak growth was detected at 5 °C (doubling time 168 and 144 h for strains PM4 and S5T, respectively) and even at 0 °C for strain PM4. Optimal growth was observed at 37 °C for strain PM4 and 40 °C for strain S5T. The pH range for growth in both cases was determined as pH 0.5–4, with optimal growth at about pH 1.0–1.2.
The strains were found to be obligate heterotrophs, requiring complex organic compounds such as yeast or beef extracts (0.05–0.5 %) for growth, with the best results for beef extract at 0.3–0.5 % (w/v). Strain PM4 also required the addition of tryptone at 0.1 % (w/v). The doubling time for both strains under optimal growth conditions was 17.8 h. No growth was found on galactose, fructose, trehalose, glucose, arabinose, sucrose, glycerol, malt extract, mannitol, starch, pullulan, curdlan, methyl β-dextrin, N-acetylglucosamine, galactomannan, sodium pyruvate, trisodium citrate, sodium acetate, methanol, l-cysteine, l-methionine, methylamine or dimethylsulfide (all at 0.1 %). Growth was also absent on pyrite and FeSO4 (medium 9K) in the presence of small amounts of beef or yeast extract (0.02 %). Both strains were observed to grow anaerobically, supplemented with 0.3 % (w/v) beef extract for strain S5T and beef extract (0.3 % w/v) and tryptone (0.1 % w/v) in the case of strain PM4, all in the presence of ferric iron citrate (1 mM), sulfur (0.1 M) and potassium nitrate (10 mM) as electron acceptors. Weak anaerobic growth was detected in the absence of ferric iron citrate, sulfur and nitrate.
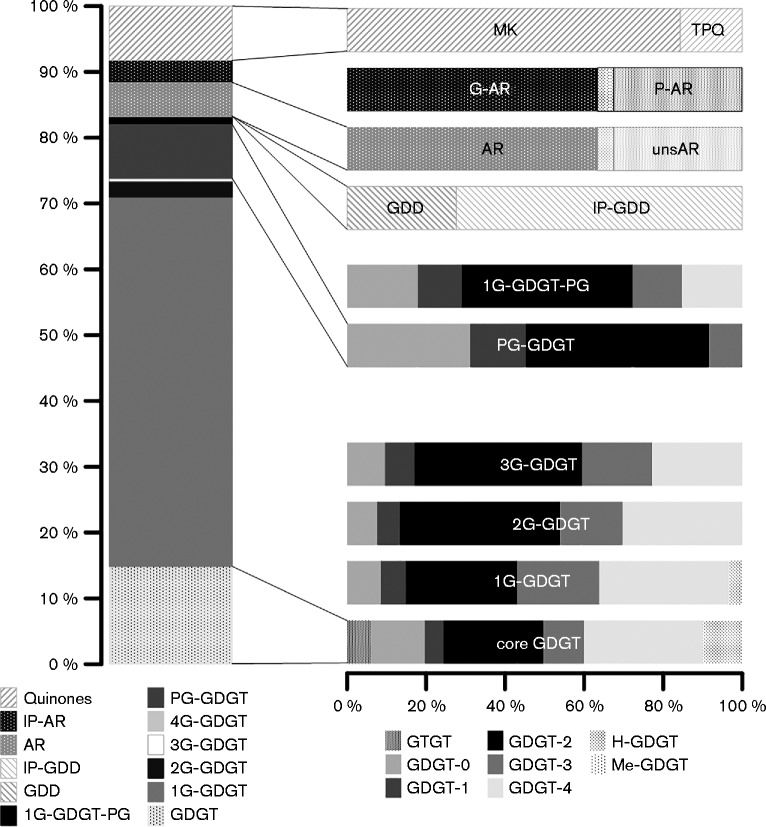
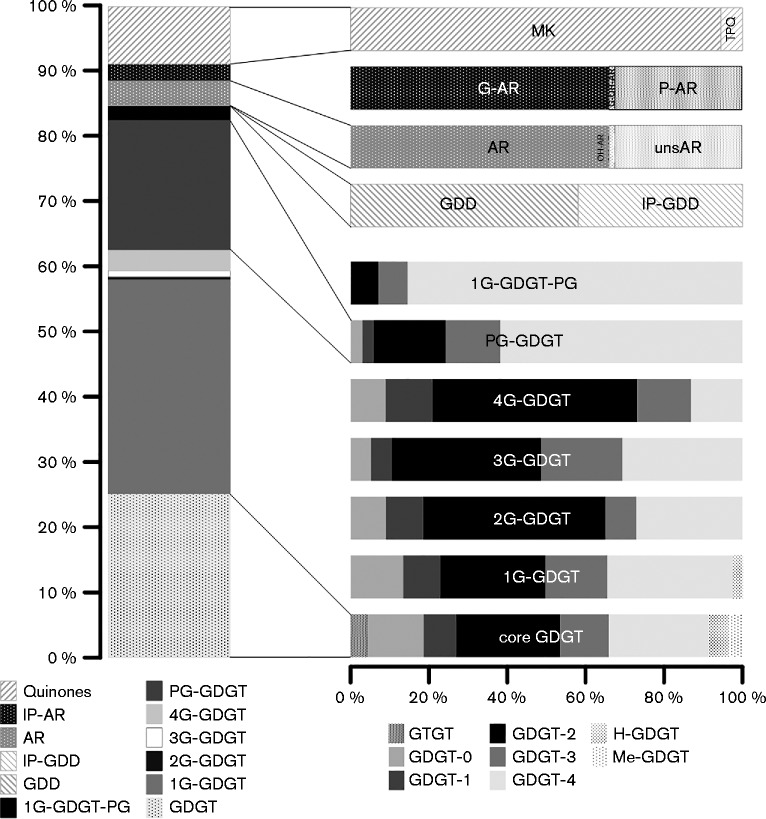
The lipid pools of strains S5T and PM4 were highly similar. The profiles of both strains were dominated by intact polar lipids (IPL) based on glycerol dibiphytanyl glycerol tetraether (GDGT) as core lipid, which is attached to variable polar headgroups. The IPL derivatives of GDGT were predominantly glycolipids, with monoglycosidic (1G), diglycosidic (2G), triglycosidic (3G) and, for strain PM4 only, tetraglycosidic (4G) headgroups. Mixed glyco-phospho (1G-PG) -based headgroups represented less than 5 % of the IPL GDGT pool, while phospho- (PG-) GDGT accounted for about 10 and 30 % in S5T and PM4, respectively. Free core GDGT were detected as regular GDGT and H-shaped (H-) GDGT with zero to four cyclopentane moieties (GDGT-0 to -4), with GDGT-2 and -4 being predominant. This general pattern of core GDGT distribution was also found among individual IPL GDGT types with the exception of PG-GDGT, which had GDGT-2 and GDGT-0 as major core lipids (Figs. 3 and 4 and Table S1, available in the online Supplementary Material).
Fig. 3.
Lipid composition of strain S5T. The column on the left displays the relative abundances of the major lipid groups. The horizontal bars show the relative contributions of individual lipid structures to the major groups. 1G, Monoglycosidic; 1G-PG, monoglycosidic phosphatidylglycerol; 2G, diglycosidic; 3G, triglycosidic; 4G, tetraglycosidic; AR, archaeol; G-AR, glycosidic AR; GDD, glycerol dibiphytanyl diether; GDGT, glycerol dibiphytanyl glycerol tetraether; GDGT-0 to -4, GDGT with zero to four cyclopentane moieties; G-OH, glycosidic-hydroxylated; GTGT, glycerol trialkyl glycerol tetraether; H-GDGT, H-shaped GDGT; IP, intact polar; Me-GDGT, methylated GDGT; MK, menaquinone; OH, core hydroxylated; P-AR, phospho-AR; PG, phosphatidylglycerol; TPQ, thermoplasmaquinone; unsAR, unsaturated AR.
Fig. 4.
Lipid composition of strain PM4. See Fig. 3 for details.
Archaeol (AR) -based IPL possessed 1G and PG headgroups, while hydroxy-archaeol (OH-AR) was detected with 1G and 2G headgroups. Free diether-based core lipids such as AR, OH-AR and glycerol dibiphytanyl diethers (GDD) accounted for a minor fraction. Saturated AR was predominant among the core diether lipids, while unsaturated (uns) AR with one to six double bonds represented about one-third of the AR-based core lipid pool. In addition, OH-AR and GDD were minor constituents of this lipid pool (Figs 3 and 4, Table S1). Intact polar GDD was a minor component, and was present only as a 1G derivative (1G-GDD); the corresponding core GDD comprised less than 1 % of the core lipid pool (Figs. 3 and 4, Table S1).
Twenty-seven and 25 different quinones were identified in strains S5T and PM4, respectively. These were menaquinones (MK) and thermoplasmaquinones (TPQ) (Figs. 3 and 4, Table S1). The quinones were generally characterized by highly unsaturated side chains of four to ten isoprenoid units with one double bond per unit (4 : 4 to 10 : 10) (data not shown).
The 16S rRNA gene sequences derived from the whole-genome sequences of strains S5T and PM4 (GenBank accession numbers KT005320 and KT005321) appeared identical. These sequences, together with 16S rRNA gene sequences of all members of the Thermoplasmatales with validly published names, were aligned (a total of 11 sequences) using the muscle program (Edgar, 2004). Analysis of nucleotide substitution models using JModelTest version 2.1.7 (Darriba et al., 2012) suggested the general time reversible model (G+I, four categories; Tamura et al., 2013) as the best evolution model for the given dataset. The distances calculated using the above model suggested that the closest relatives for both S5T and PM4 were Thermogymnomonas acidicola (86.3 % sequence identity to the type strain), followed by Thermoplasma acidophilum (84.7 %), Thermoplasma volcanium (84.3 %), Picrophilus torridus and Picrophilus oshimae (both 84.9 %), with more distantly relatives being members of the Ferroplasmaceae; both species of Acidiplasma (77.0 %) and Ferroplasma acidiphilum (73.9 %). Evolutionary history was inferred in the mega6 program (Tamura et al., 2013) by using the maximum-likelihood method with 1000 repetitions. The phylogenetic tree with the highest log-likelihood ( − 4753.4401) is shown in Fig. 5. The tree is drawn to scale, with branch lengths measured in the number of substitutions per site with corrections associated with the model. All positions with less than 95 % site coverage were eliminated; that is, fewer than 5 % alignment gaps, missing data and ambiguous bases were allowed at any position. There were a total of 1323 positions in the final dataset.
Fig. 5.
16S rRNA gene-based maximum-likelihood tree showing the phylogenetic positions of strains S5T and PM4 and type strains of all members of the order Thermoplasmatales with validly published names; the analysis involved 11 nucleotide sequences. There were a total of 1323 positions in the final dataset. Methanomassiliicoccus luminyensis B10T (GenBank accession no. HQ896499.1) was used as an outgroup (not shown). Bar, 5 substitutions per 100 nucleotide positions.
The genomes of strains S5T and PM4 (O. V. Golyshina, unpublished) exhibited average nucleotide identity of 98.75 % (Goris et al., 2007), suggesting that both belong to the same species.
The genomic DNA G+C contents were 37.3 mol% for strain S5T and 37.16 mol% for strain PM4, based on in silico calculations using the complete genome sequences of the strains (O. V. Golyshina, unpublished).
Strains S5T and PM4 are acidophilic, mesophilic, facultatively anaerobic, heterotrophic, cell-wall-deficient archaea. Generally, all these features are common in the archaea of the order Thermoplasmatales (Table 1), but certain distinctive traits for strains S5T and PM4 must be emphasized. Being isolated from sulfide ore mines, these strains did not oxidize any tested iron(II) donors, a common feature in archaea of the family Ferroplasmaceae and potentially anticipated in micro-organisms living in these environments. Acid mine drainage systems are, with some exceptions (e.g. Richmond Mine, Iron Mountain, USA), situated in low-to-moderately temperate conditions, and this explains the mostly mesophilic nature of bacteria and archaea that occupy these biotopes. However, mesophily is rather rare among members of the order Thermoplasmatales, and is indicative solely for some archaea of the family Ferroplasmaceae (Golyshina, 2011). Furthermore, in relation to the temperature, one can note that the Mynydd Parys site is one of the acid mine drainage systems with the lowest annual temperatures (Méndez-Garcia et al., 2015) and is thus definitely distinct in this sense from the Cantareras site (south-west Spain). Nevertheless, both strains exhibited mesophilic temperature ranges for growth in vitro, with a very slight difference in their temperature optima. Both strains could be characterized as psychrotolerant, due to growth at temperatures of about 10 °C, which is remarkably low in comparison to characteristics of other thermophilic archaea of the order Thermoplasmatales. Furthermore, strains S5T and PM4 showed some potential to grow at temperatures as low as 5 °C (and even at 0 °C, in the case of strain PM4). The pH optima for growth of both strains were similar to the lowest values recorded for other representatives of Thermoplasmatales, and the pH range for growth reflected the environmental conditions at the sites of isolation.
Table 1. Main physiological characteristics of archaea of the order Thermoplasmatales and strains S5T and PM4.
Reference taxa: 1, Thermoplasma (data from Darland et al., 1970; Segerer et al., 1988); 2, Picrophilus (Schleper et al., 1995); 3, Ferroplasma acidiphilum (Golyshina et al., 2000); 4, Acidiplasma (Golyshina et al., 2009); 5, Thermogymnomonas acidicola (Itoh et al., 2007).
| Characteristic | 1 | 2 | 3 | 4 | 5 | S5T | PM4 |
|---|---|---|---|---|---|---|---|
| Cell wall/S-layer | − | + | − | − | − | − | − |
| Growth temperature (°C) | |||||||
| Range | 33–67 | 47–65 | 15–45 | 15/22–65 | 38–68 | 10–48 | 10–45 |
| Optimum | 69 | 60 | 35/37 | 45/53.5 | 60 | 40 | 37 |
| Growth pH | |||||||
| Range | 0.5–4 | 0–3.5 | 1.3–2.2 | 0/0.4–1.8/4 | 1.8–4 | 0.5–4 | 0.5–4 |
| Optimum | 1–2 | 0.7 | 1.7 | 1–1.6 | 3 | 1–1.2 | 1–1.2 |
| Fe2+ oxidation | − | − | + | + | − | − | − |
| Anaerobic growth | + | − | − /+ | + | − | + | + |
| DNA G+C content (mol%) | 38–46 | 36 | 37 | 34–36 | 56 | 37 | 37 |
As for the majority of other organisms from the order Thermoplasmatales, cells of strains S5T and PM4 were surrounded by only cytoplasmic membranes. Based on the obligatory use of polypeptides as growth substrates, one can suggest a ‘scavenger’-type lifestyle for these archaea in natural biotopes.
Analyses of metagenomics data from samples obtained in a number of geographically separated regions have revealed 16S rRNA gene sequences identical to those of strains S5T and PM4 (Yelton et al., 2013; Méndez-Garcia et al., 2014), suggesting that these archaea are ubiquitous in acidic environments.
Advanced HPLC-MS analysis of the lipidome allowed us to characterize more than 70 different major glycerol-based membrane lipids. The distribution of GDGT, here detected as glyco-, phospho- and glyco-phospho-derivatives, resembles that of other members of the Thermoplasmatales (Schleper et al., 1995; Shimada et al., 2002; Itoh et al., 2007; Golyshina et al., 2009; Golyshina, 2011). An increasing number of cyclopentane rings of GDGTs has been interpreted to reflect adaptation to decreasing pH and increasing temperatures for the thermophilic acidophile Thermoplasma acidophilum HO-62 (Shimada et al., 2008) and other thermophilic acidophiles (e.g. Boyd et al., 2011). Strains S5T and PM4, however, are acidophilic mesophiles and show a distinct ring distribution dominated by GDGT-2 and GDGT-4, which suggest that the low pH causes high degrees of cycloalkylation in these strains. The mean number of glycosidic moieties attached to GDGT lipids was found to increase with decreasing environmental pH in Thermoplasma acidophilum, and can be interpreted to function as a shield against protons for microbial cells in low-pH environments (Shimada et al., 2008; Wang et al., 2012). This phenomenon could explain the prevalence of up to four glycosidic headgroups in the lipid pools of strains S5T and PM4 as an adaptation to extremely acidic conditions. The presence of OH-AR, which is usually abundant in halophilic and methanogenic euryarchaea of the order Halobacteriales (Kates, 1993) as well as the orders Methanococcales, Methanosarcinales and Methanopyrales (Koga & Morii, 2005), is consistent with earlier reports of this compound in other members of the Thermoplasmatales (Swain et al., 1997; Golyshina et al., 2000; and references therein). Unsaturated AR derivatives have so far not been described for the Thermoplasmatales, however, for euryarchaea of the orders Halobacteriales, Thermococcales, Methanopyrales and Methanosarcinales (e.g. Dawson et al., 2012; Gonthier et al., 2001; Hafenbradl et al., 1993; Nichols et al., 2004), with a broad range of growth temperatures. This indicates that the abundance of these compounds in strains S5T and PM4 is not necessarily linked to temperature; hence, understanding their role in the archaeal membrane will require further research.
The respiratory quinones identified were MK and TPQ with a diverse set of unsaturated chains from 4 : 4 to 10 : 10. This results in a more diverse quinone portfolio for strains S5T and PM4 compared with the quinone inventory described for other members of the Thermoplasmatales (e.g. Shimada et al., 2001; Itoh et al., 2007; Golyshina et al., 2009; Elling et al., 2015).
Both the identical 16S rRNA gene sequences of strains S5T and PM4 and the 98.75 % average nucleotide identity between their genomes suggest their affiliation to the same species.
The results of phylogenetic analysis suggest an outlying position of the novel organisms (Fig. 5) within the Thermoplasmatales, which, along with their physiological, morphological and chemotaxonomic properties, allows the proposal of the novel family Cuniculiplasmataceae fam. nov., containing the genus Cuniculiplasma gen. nov. with the type species Cuniculiplasma divulgatum gen. nov., sp. nov.
Description of Cuniculiplasma gen. nov.
Cuniculiplasma (Cu.ni′cu.li.plas′ma. L. n. cuniculum a cave, mine, subterranean path; Gr. neut. n. plasma something shaped or moulded; N.L. neut. n. Cuniculiplasma a form living in a mine or underground).
Cells are pleomorphic without cell walls, with a tendency to form ring- and Y-like morphologies. Facultatively anaerobic. Organotrophic. Mesophilic, by which the genus is distinct from other members of the Thermoplasmatales with exclusively organotrophic metabolism from the genera Picrophilus, Thermogymnomonas and Thermoplasma. Acidophilic. 16S rRNA gene sequences place the genus within the order Thermoplasmatales, phylum Euryarchaeota. Known habitats are acidic streamers. The type species is Cuniculiplasma divulgatum.
Description of Cuniculiplasma divulgatum sp. nov.
Cuniculiplasma divulgatum (di.vul.ga′tum. L. neut. adj. divulgatum common, widespread).
In addition to the features described for the genus, the temperature range for growth is 10–48 °C with the optimum at 37–40 °C. Grows at pH 0.5–4.0, with an optimum at pH 1.0–1.2. Grows organoheterotrophically with beef and yeast extracts (and tryptone for strain PM4). Polar lipids consist of a GDGT core with glyco-headgroups, dominated by monoglycosidic derivatives, smaller amounts of phospho, diglycosidic and triglycosidic derivatives and a smaller fraction of diether lipids such as archaeols. Strain PM4 shows less dominance of monoglycosidic derivatives in favour of phospho, triglycosidic and tetraglycosidic GBGT lipids. The main respiratory quinones are represented by menaquinones and minor thermoplasmaquinones.
The type strain is strain S5T ( = VKM B-2941T = JCM 30642T), isolated from an acidic streamer in Cantareras Mine (Spain). Strain PM4 was isolated from an acidic streamer of Mynydd Parys, Anglesey, UK. The DNA G+C content of strain S5T is 37.3 mol% and that of strain PM4 is 37.16 mol%.
Description of Cuniculiplasmataceae fam. nov.
Cuniculiplasmataceae (Cu.ni′cu.li.plas.ma.ta′ce.ae. N.L. neut.n. Cuniculiplasma type genus of the family; L. suff. -aceae ending to denote a family; N.L. fem. pl. n. Cuniculiplasmataceae the family of the genus Cuniculiplasma).
The description is identical to that of the genus Cuniculiplasma. The type genus is Cuniculiplasma.
Acknowledgements
We gratefully acknowledge the help of Professor Bernhard Schink (Uni. Konstanz) with nomenclature. This work was supported by the Royal Society UK–Russia Exchange grant IE 130218 , EU Horizon 2020 grant no. 634486 ‘INMARE’ and EU FP7 grant no. 287589 ‘MicroB3’. Lipid analyses were supported by Deutsche Forschungsgemeinschaft grant HI 616## provided through the Gottfried Wilhelm Leibniz Program. We thank Professor Barrie Johnson (Bangor University, UK) for providing the sample of acidic streamer from Cantareras Mine, Spain. The skilful work of Mrs Inge Kristen (HZI, Central Unit of Microscopy, Braunschweig, Germany) is gratefully acknowledged.
Supplementary Data
Supplementary Data
Abbreviations:
- GDD
glycerol dibiphytanyl diether
- GDGT
glycerol dibiphytanyl glycerol tetraether
- IPL
intact polar lipid.
References
- Baker B. J., Tyson G. W., Webb R. I., Flanagan J., Hugenholtz P., Allen E. E., Banfield J. F. (2006). Lineages of acidophilic archaea revealed by community genomic analysis Science 314 1933–1935 10.1126/science.1132690 . [DOI] [PubMed] [Google Scholar]
- Boyd E. S., Pearson A., Pi Y., Li W. J., Zhang Y. G., He L., Zhang C. L., Geesey G. G. (2011). Temperature and pH controls on glycerol dibiphytanyl glycerol tetraether lipid composition in the hyperthermophilic crenarchaeon Acidilobus sulfurireducens Extremophiles 15 59–65 10.1007/s00792-010-0339-y . [DOI] [PubMed] [Google Scholar]
- Darland G., Brock T. D., Samsonoff W., Conti S. F. (1970). A thermophilic, acidophilic mycoplasma isolated from a coal refuse pile Science 170 1416–1418 10.1126/science.170.3965.1416 . [DOI] [PubMed] [Google Scholar]
- Darriba D., Taboada G. L., Doallo R., Posada D. (2012). jModelTest 2: more models, new heuristics and parallel computing Nat Methods 9 772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dawson K. S., Freeman K. H., Macalady J. L. (2012). Molecular characterization of core lipids from halophilic archaea grown under different salinity conditions Org Geochem 48 1–8 10.1016/j.orggeochem.2012.04.003. [DOI] [Google Scholar]
- Edgar R. C. (2004). muscle: multiple sequence alignment with high accuracy and high throughput Nucleic Acids Res 32 1792–1797 10.1093/nar/gkh340 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elling F. J., Becker K. W., Könneke M., Schröder J. M., Kellermann M. Y., Thomm M., Hinrichs K.-U. (2015). Respiratory quinones in Archaea: phylogenetic distribution and application as biomarkers in the marine environment Environ Microbiol (in press) . [DOI] [PubMed] [Google Scholar]
- Golyshina O. V. (2011). Environmental, biogeographic, and biochemical patterns of archaea of the family Ferroplasmaceae Appl Environ Microbiol 77 5071–5078 10.1128/AEM.00726-11 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Golyshina O. V., Pivovarova T. A., Karavaiko G. I., Kondratéva T. F., Moore E. R., Abraham W. R., Lünsdorf H., Timmis K. N., Yakimov M. M., Golyshin P. N. (2000). Ferroplasma acidiphilum gen. nov., sp. nov., an acidophilic, autotrophic, ferrous-iron-oxidizing, cell-wall-lacking, mesophilic member of the Ferroplasmaceae fam. nov., comprising a distinct lineage of the Archaea Int J Syst Evol Microbiol 50 997–1006 10.1099/00207713-50-3-997 . [DOI] [PubMed] [Google Scholar]
- Golyshina O. V., Yakimov M. M., Lünsdorf H., Ferrer M., Nimtz M., Timmis K. N., Wray V., Tindall B. J., Golyshin P. N. (2009). Acidiplasma aeolicum gen. nov., sp. nov., a euryarchaeon of the family Ferroplasmaceae isolated from a hydrothermal pool, and transfer of Ferroplasma cupricumulans to Acidiplasma cupricumulans comb. nov Int J Syst Evol Microbiol 59 2815–2823 10.1099/ijs.0.009639-0 . [DOI] [PubMed] [Google Scholar]
- Gonthier I., Rager M. N., Metzger P., Guezennec J., Largeau C. (2001). A di-O-dihydrogeranylgeranyl glycerol from Thermococcus S 557, a novel ether lipid, and likely intermediate in the biosynthesis of diethers in Archaea Tetrahedron Lett 42 2795–2797 10.1016/S0040-4039(01)00305-7. [DOI] [Google Scholar]
- Goris J., Konstantinidis K. T., Klappenbach J. A., Coenye T., Vandamme P., Tiedje J. M. (2007). DNA-DNA hybridization values and their relationship to whole-genome sequence similarities Int J Syst Evol Microbiol 57 81–91 10.1099/ijs.0.64483-0 . [DOI] [PubMed] [Google Scholar]
- Hafenbradl D., Keller M., Thiericke R., Stetter K. O. (1993). A novel unsaturated archaeal ether core lipid from the hyperthermophile Methanopyrus kandleri Syst Appl Microbiol 16 165–169 10.1016/S0723-2020(11)80463-7. [DOI] [Google Scholar]
- Itoh T., Yoshikawa N., Takashina T. (2007). Thermogymnomonas acidicola gen. nov., sp. nov., a novel thermoacidophilic, cell wall-less archaeon in the order Thermoplasmatales, isolated from a solfataric soil in Hakone, Japan Int J Syst Evol Microbiol 57 2557–2561 10.1099/ijs.0.65203-0 . [DOI] [PubMed] [Google Scholar]
- Kates M. (1993). Biology of halophilic bacteria, part II. Membrane lipids of extreme halophiles: biosynthesis, function and evolutionary significance Experientia 49 1027–1036 10.1007/BF01929909 . [DOI] [PubMed] [Google Scholar]
- Koga Y., Morii H. (2005). Recent advances in structural research on ether lipids from archaea including comparative and physiological aspects Biosci Biotechnol Biochem 69 2019–2034 10.1271/bbb.69.2019 . [DOI] [PubMed] [Google Scholar]
- Méndez-García C., Mesa V., Sprenger R. R., Richter M., Diez M. S., Solano J., Bargiela R., Golyshina O. V., Manteca Á., other authors (2014). Microbial stratification in low pH oxic and suboxic macroscopic growths along an acid mine drainage ISME J 8 1259–1274 10.1038/ismej.2013.242 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Méndez-García C., Peláez A. I., Mesa V., Sánchez J., Golyshina O. V., Ferrer M. (2015). Microbial diversity and metabolic networks in acid mine drainage habitats Front Microbiol 6 475 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nichols D. S., Miller M. R., Davies N. W., Goodchild A., Raftery M., Cavicchioli R. (2004). Cold adaptation in the Antarctic archaeon Methanococcoides burtonii involves membrane lipid unsaturation J Bacteriol 186 8508–8515 10.1128/JB.186.24.8508-8515.2004 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schleper C., Puehler G., Holz I., Gambacorta A., Janekovic D., Santarius U., Klenk H. P., Zillig W. (1995). Picrophilus gen. nov., fam. nov.: a novel aerobic, heterotrophic, thermoacidophilic genus and family comprising archaea capable of growth around pH 0 J Bacteriol 177 7050–7059 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Segerer A., Langworthy T. A., Stetter K. O. (1988). Thermoplasma acidophilum and Thermoplasma volcanium sp. nov. from solfatara fields Syst Appl Microbiol 10 161–171 10.1016/S0723-2020(88)80031-6. [DOI] [Google Scholar]
- Shimada H., Shida Y., Nemoto N., Oshima T., Yamagishi A. (2001). Quinone profiles of Thermoplasma acidophilum HO-62 J Bacteriol 183 1462–1465 10.1128/JB.183.4.1462-1465.2001 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimada H., Nemoto N., Shida Y., Oshima T., Yamagishi A. (2002). Complete polar lipid composition of Thermoplasma acidophilum HO-62 determined by high-performance liquid chromatography with evaporative light-scattering detection J Bacteriol 184 556–563 10.1128/JB.184.2.556-563.2002 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimada H., Nemoto N., Shida Y., Oshima T., Yamagishi A. (2008). Effects of pH and temperature on the composition of polar lipids in Thermoplasma acidophilum HO-62 J Bacteriol 190 5404–5411 10.1128/JB.00415-08 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sturt H. F., Summons R. E., Smith K., Elvert M., Hinrichs K.-U. (2004). Intact polar membrane lipids in prokaryotes and sediments deciphered by high-performance liquid chromatography/electrospray ionization multistage mass spectrometry — new biomarkers for biogeochemistry and microbial ecology Rapid Commun Mass Spectrom 18 617–628 10.1002/rcm.1378 . [DOI] [PubMed] [Google Scholar]
- Swain M., Brisson J.-R., Sprott G. D., Cooper F. P., Patel G. B. (1997). Identification of β-l-gulose as the sugar moiety of the main polar lipid Thermoplasma acidophilum Biochim Biophys Acta 1345 56–64 10.1016/S0005-2760(96)00163-4 . [DOI] [PubMed] [Google Scholar]
- Tamura K., Stecher G., Peterson D., Filipski A., Kumar S. (2013). mega6: molecular evolutionary genetics analysis version 6.0 Mol Biol Evol 30 2725–2729 10.1093/molbev/mst197 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X., Lv B., Cai G., Fu L., Wu Y., Wang X., Ren B., Ma H. (2012). A proton shelter inspired by the sugar coating of acidophilic archaea Sci Rep 2 892 10.1038/srep00892 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yelton A. P., Comolli L., Justice N. B., Castelle C., Denef V. J., Thomas B. C., Banfield J. F. (2013). Comparative genomics in acid mine drainage biofilm communities reveals metabolic and structural differentiation of co-occurring archaea BMC Genomics 14 485 10.1186/1471-2164-14-485 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu C., Lipp J. S., Wörmer L., Becker K. W., Schröder J., Hinrichs K.-U. (2013). Comprehensive glycerol ether lipid fingerprints through a novel reversed phase liquid chromatography-mass spectrometry protocol Org Geochem 65 53–62 10.1016/j.orggeochem.2013.09.012. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Data