Abstract
Central leptin action is sufficient to restore euglycemia in insulinopenic type 1 diabetes (T1D); however, the underlying mechanism remains poorly understood. To examine the role of intracellular signal transducer and activator of transcription 3 (STAT3) pathways, we used LepRs/s mice with disrupted leptin-phosphorylated STAT3 signaling to test the effect of central leptin on euglycemia restoration. These mice developed streptozocin-induced T1D, which was surprisingly not associated with hyperglucagonemia, a typical manifestation in T1D. Further, leptin action on euglycemia restoration was abrogated in these mice, which was associated with refractory hypercorticosteronemia. To examine the role of fast-acting neurotransmitters glutamate and γ-aminobutyric acid (GABA), two major neurotransmitters in the brain, from leptin receptor (LepR) neurons, we used mice with disrupted release of glutamate, GABA, or both from LepR neurons. Surprisingly, all mice responded normally to leptin-mediated euglycemia restoration, which was associated with expected correction from hyperglucagonemia and hyperphagia. In contrast, mice with loss of glutamate and GABA appeared to develop an additive obesity effect over those with loss of single neurotransmitter release. Thus, our study reveals that STAT3 signaling, but not fast-acting neurotransmitter release, is required for leptin action on euglycemia restoration and that hyperglucagonemia is not required for T1D.
Introduction
Insulin administration remains the only option for treatment in patients with type 1 diabetes (T1D). However, the insulin therapy is associated with strong lipogenic effects and sometimes life-threatening hypoglycemia, which calls for alternative or combinational therapeutic strategies for T1D. Earlier studies (1–3) have demonstrated that leptin, in addition to its role in body weight regulation, enhances insulin sensitivity. Strikingly, leptin action on euglycemia restoration in T1D is independent of insulin action, and is associated with improved lipid homeostasis and less risk of hypoglycemia (4,5). Furthermore, euglycemia restoration by leptin is associated with reduction of glucagon concentration, suggesting a role for hyperglucagonemia in the pathogenesis of T1D. Indeed, glucagon receptor deficiency abrogates the hyperglycemia-producing effects by lesion of pancreatic β-cells (6). However, recent studies have also suggested an importance of hypercorticosteronemia in the development of T1D (7). Interestingly, leptin action in the brain alone is sufficient to restore euglycemia in T1D, which is again associated with glucagon reduction, whereas leptin action in the liver is not required (8,9). These results demonstrate a critical role for central leptin action in glucose homeostasis independent of insulin action. However, the underlying neural pathway responsible for leptin action in lowering glucose in T1D is not yet clear.
Leptin action is primarily mediated by isoform B leptin receptor (LepR) expressed in the brain (10). LepR-expressing neurons (LepR neurons) reside predominantly in the hypothalamus (11), but also in other brain regions such as brainstem nuclei and ventral tegmental area (12–16). LepR neurons can be broadly divided into GABAergic and glutamatergic subsets (17). LepRs in GABAergic neurons mediate a major part of the leptin action on body weight regulation, whereas those in glutamatergic neurons mediate a small part (17). We have recently demonstrated (18,19) that disruption of either γ-aminobutyric acid (GABA) or glutamate release from LepR neurons triggers obesity in mice. Importantly, LepR expression in GABAergic neurons is sufficient for leptin-mediated insulin-independent regulation of glucose homeostasis (20), suggesting a role for GABA release in mediating the leptin action. A recent study (21,22) suggests that leptin action in the ventromedial hypothalamic (VMH) neurons, which are mainly glutamatergic, is also sufficient to mediate glucose-lowering effects of central leptin action, suggesting a role for glutamate release in mediating the leptin action. However, whether GABA and/or glutamate release mediates the antidiabetic effects of leptin in T1D is unknown.
Leptin binding to LepR results in activation of multiple intracellular pathways including the signal transducer and activator of transcription (STAT) 3/STAT5, phosphoinositide 3-kinase (PI3K), mitogen-activated protein kinase, and mammalian target of rapamycin (mTOR) pathways (23). The STAT3 pathway mediates a major part of leptin action on body weight, but other pathways such as the STAT5 pathway also contribute significantly to normal body weight homeostasis (24,25). Mice with LepR → STAT3 signaling deficiency develop hyperglycemia and hyperinsulinemia (25); however, it is unknown whether it is due to a direct action or is secondary to development of severe obesity. The central action of other factors like fibroblast growth factor 19 and brain-derived neurotrophic factor also has a leptin-like effect in lowering glucose levels through an insulin-independent mechanism (26,27), suggesting a role for a common brain pathway in mediating the antidiabetic effect. A recent study (28) suggests that a noncanonical leptin signaling pathway (i.e., CREB-regulated transcription coactivator 1) mediates leptin action in lowering glucose levels in an insulin-dependent manner. However, it remains unclear which of the leptin signaling pathways mediates the leptin action.
This study was therefore designed to specifically examine the roles of glutamate and/or GABA released from LepR neurons and the leptin → phosphorylated STAT3 (pSTAT3) pathway in leptin-mediated euglycemia restoration in the T1D mouse model. Our results reveal that STAT3 signaling, but neither glutamate nor GABA release from LepR neurons, is required for the insulin-independent glucose-lowering effects of leptin.
Research Design and Methods
Animals
Vesicular GABA transporter (VGAT; encoded by Vgat, also known as Slc32a1) is required for presynaptic release of GABA, whereas vesicular glutamate transporter 2 (Vglut2; also known as Slc17a6) is responsible for presynaptic release of glutamate (29,30). Generation of LepR-Ires-Cre (LIC) mice and floxed Vgat (Vgatflox/flox) or Vglut2 (Vglut2flox/flox) mice was described previously (31–33). The same breeding strategy described in the previous studies was used to obtain LIC:Vgatflox/flox mice and LIC:Vglut2flox/flox mice (18,19). LIC:Vgaflox/flox:Vglut2flox/flox mice and their controls were obtained from breeding pairs LIC:Vgaflox/flox:Vglut2flox/flox and Vgatflox/flox:Vglut2flox/flox mice . Mice with specific disruption of the LepR → STAT3 signal in LepR neurons (LepRs/s) were obtained from The Jackson Laboratory (stock #008518) (25). All mice were housed with ad libitum access to water and food in a temperature-controlled room (21–22°C) with a 12:12 h light-dark cycle. Animal care and procedures were approved by the Animal Welfare Committee of The University of Texas Health Science Center at Houston.
Metabolic Measurements
From 4 to 20 weeks of age, body weight was monitored weekly in controls and LIC:Vgatflox/flox:Vglut2flox/flox mice fed standard mouse chow (Teklad F6 Rodent Diet 8664; Harlan Teklad, Madison, WI). At the end of this measurement, the body composition of all study subjects was measured with quantitative magnetic spectroscopy (Echo Medical Systems, Houston, TX).
Streptozocin-Induced T1D
Streptozocin (STZ) (Sigma-Aldrich, St. Louis, MO) dissolved in cold sterile acetate buffer, pH 4.5, was injected intraperitoneally into 6- to 8-week-old male mice at 150 mg/kg (two times at 1-week intervals). Nondiabetic control mice received intraperitoneal injection of acetate buffer alone. T1D was defined as a nonfasting blood glucose concentration of >400 mg/dL on 3 consecutive days.
Intracerebroventricular Leptin Infusion
Recombinant mouse leptin (purchased from A.F. Parlow, National Hormone and Peptide Program, Torrance, CA) dissolved in PBS (pH 8.0) was loaded into 14-day mini-osmotic pumps (DURECT Corporation, Cupertino, CA) that were connected with commercial brain infusion kits (DURECT Corporation). Pumps were surgically implanted in the mouse interscapular cavity 7 days after the establishment of T1D. All mice were anesthetized with ketamine (100 mg/kg) and xylazine (10 mg/kg), and then cannulas were stereotaxically positioned into the lateral cerebral ventricles using the following coordinates: −0.30 mm from the bregma, ±1.0 mm lateral, −2.5 mm of the skull. Leptin was continuously delivered at a speed of 50 ng/h (0.25 μL/h) to the ventricles. T1D mice receiving the same amount of vehicle by intracerebroventricular infusion were taken as controls.
From day 3 after intracerebroventricular surgery, blood glucose levels, food intake, and body weight of all animals were recorded once every 2 days. All measurements were performed between 10:00 and 12:00 a.m. Glucose levels were measured using tail vein blood with a OneTouch Glucometer.
Blood Collection and Tissue Processing
Blood samples were collected from all mice with deep anesthesia into a chilled tube containing 15 μL of EDTA. Plasma was isolated by centrifugation (3,000g at 4°C for 15 min), and then stored at −80°C. After blood collection, all mice were transcardially perfused with 0.9% normal saline followed by 10% formalin. Brain and pancreas were harvested and cryoprotected in 30% sucrose for further analysis. Plasma glucagon, insulin, and corticosterone were measured by the Vanderbilt University Hormone Assay & Analytical Services Core (Nashville, TN).
Immunohistochemistry
Brain sections were sliced with a microtome (Leica, Wetzlar, Germany) at a thickness of 30 μm, and pancreatic sections were sliced with a cryostat (Leica) at a thickness of 15 μm. pSTAT3 was immunostained with rabbit anti-pSTAT3 antibody (1:400; Cell Signaling Technology, Danvers, MA) in brain tissues from LIC:Vgatflox/flox, LIC: Vglut2flox/flox, and LIC:Vgatflox/flox:Vglut2flox/flox mice; phospho-S6 ribosomal protein (p-S6; Ser240/244) was immunostained with rabbit anti–p-S6 antibody (1:800; Cell Signaling Technology) in brain tissues from LepRs/s mice (34). All brain sections were incubated with the free-floating method, as previously described (35), and were visualized with Alexa Fluor 488–conjugated donkey anti-rabbit IgGs (Jackson ImmunoResearch Laboratories, West Grove, PA). Pancreatic sections were immunostained with insulin (mouse anti-insulin Ab-6 antibody; Thermo Scientific, Fremont, CA) and glucagon (rabbit anti-glucagon monoclonal antibody; Cell Signaling Technology) overnight at 4°C. Slides were then incubated with Alexa Fluor 488–conjugated donkey anti-rabbit and Alexa Fluor 594–conjugated donkey anti-mouse secondary antibodies (Jackson ImmunoResearch Laboratories) for 2 h at room temperature.
Microarray Analysis
Hypothalamic blocks were dissected from fresh brain tissues on a brain matrix and stored immediately in RNAlater (Sigma-Aldrich). Total hypothalamic RNA was isolated from each animal with TRI Reagent (Sigma-Aldrich) following the protocol provided by the manufacturer, and then microarray analysis was performed by the Microarray Core of The University of Texas Health Science Center at Houston (Houston, TX). Preprocessing was performed using GenomeStudio software (Illumina, Inc.). Differential expression was performed using the limma package from Bioconductor (36,37). Heat maps were generated using custom-designed software.
Statistical Analysis
All data were presented as mean ± SEM. Statistical analyses were performed using one-way ANOVA with Tukey post hoc test (GraphPad Prism; GraphPad Software, La Jolla, CA). A P value of <0.05 was considered to be significant.
Results
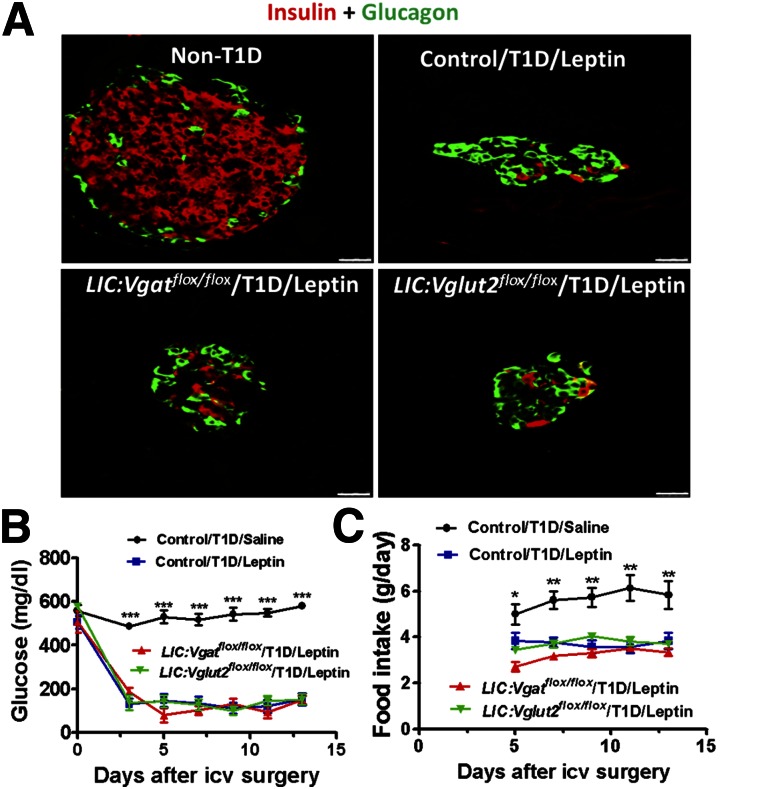
To examine whether glutamate or GABA release from LepR neurons mediates leptin action on euglycemia restoration in T1D, we used mice with disruption of glutamate release (LIC:Vglut2flox/flox mice) or GABA release from LepR neurons (LIC:Vgatflox/flox mice). To examine the deletion of Vglut2 or Vgat in LepR neurons, we performed dual color in situ hybridization for LepR and Vgat (LIC:Vgatflox/flox mice) or Vglut2 (LIC:Vglut2flox/flox mice). Both Vgat (Supplementary Fig. 1) and Vglut2 (Supplementary Fig. 2) were dramatically deleted in LepR neurons in the hypothalamus, confirming effective deletion mediated by LIC. As shown in Fig. 1A, after STZ administration, pancreatic islets in all genotypes showed a dramatic reduction in β-cell number. As expected, all mice showed hyperglycemia (>500 mg/dL) in response to STZ administration (Fig. 1B). Because Vglut2flox/flox and Vgatflox/flox mice exhibited comparable responses to saline or leptin, we pooled the data from these two groups as a control floxed group, in which glucose levels stayed above 500 mg/dL with intracerebroventricular administration of saline. For the same reason, we also pooled data from STZ-treated LIC:Vglut2flox/flox mice and LIC:Vgatflox/flox mice with saline infusion as a control Cre-floxed group, in which glucose levels also remained above 500 mg/dL, suggesting that these mice can have T1D induced by STZ in a fashion similar to that of wild-type mice (Fig. 1B). Surprisingly, both LIC:Vglut2flox/flox and LIC:Vgatflox/flox mice reduced glucose to a normal level with leptin infusion in a fashion similar to that of control mice (Fig. 1B).
Figure 1.
Effect of central leptin infusion on glucose levels in T1D LIC:Vgatflox/flox and LIC:Vglut2flox/flox mice. A: Representative images of pancreata from non-T1D and T1D mice costained for insulin (red) and glucagon (green). Scale bars = 25 μm. Measurements for plasma glucose levels (B) and daily food intake (C). All data are presented as the mean ± SEM. *P < 0.05, **P < 0.01, and ***P < 0.001 vs. other groups (n = 5–8). icv, intracerebroventricular.
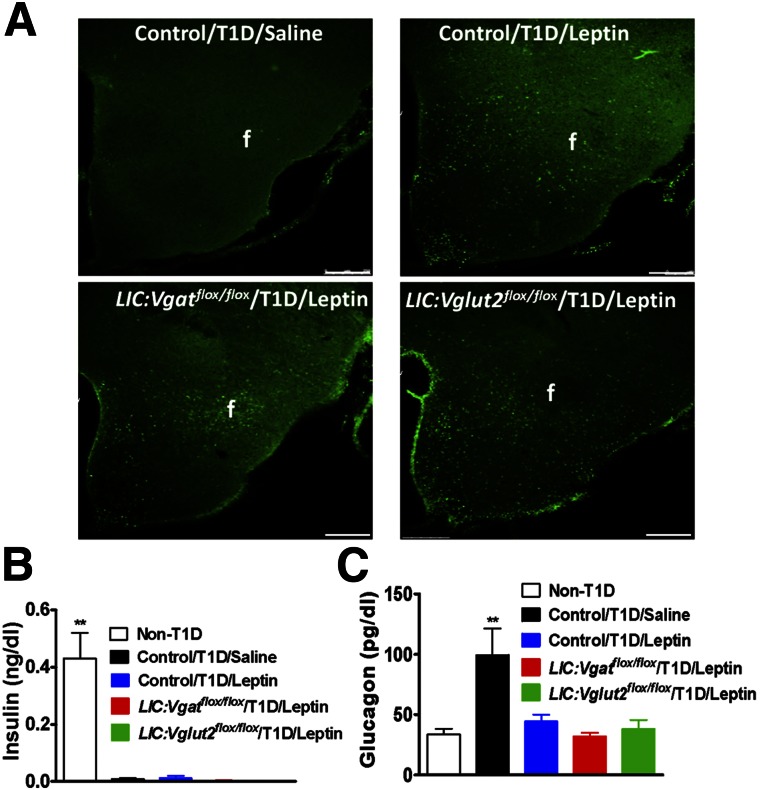
Interestingly, all mice with euglycemia restoration by leptin infusion exhibited lower food intake compared with those without euglycemia restoration (Fig. 1C), which is consistent with their corrected glucose levels. To confirm a successful leptin infusion, we performed immunohistochemistry on hypothalamic tissues for pSTAT3, a well-established indicator for leptin action. Although saline-treated animals showed no obvious pSTAT3 immunostaining (Fig. 2A), leptin produced robust expression of pSTAT3 in all genotypes tested (Fig. 2A). Additional statistical analysis on groups of mice showed that leptin induced a dramatic increase in pSTAT3 in the hypothalamus, compared with the saline-treated group (Supplementary Fig. 3), suggesting a successful leptin delivery to the brain. To ensure that euglycemia restoration in LIC:Vglut2flox/flox mice and LIC:Vgatflox/flox mice was not due to an augmented insulin action, we measured insulin concentrations in these mice. Compared with control mice without STZ treatment, all groups with STZ treatment showed insulin levels close to the detection limit (Fig. 2B), suggesting no augmented insulin action. Consistent with previous studies, higher glucagon levels, which were corrected with euglycemia restoration, were associated with T1D (Fig. 2C), suggesting an association between euglycemia restoration and reduced glucagon levels. These results, taken together, suggest that glutamate and GABA release from LepR neurons are not required for central leptin action on euglycemia restoration in T1D.
Figure 2.
Effects of central leptin infusion on glucagon levels in T1D LIC:Vgatflox/flox and LIC:Vglut2flox/flox mice. A: Expression of pSTAT3 in the hypothalamus of the indicated mouse groups. Scale bars = 250 μm. f, fornix. Plasma levels of insulin (B) and glucagon (C) were measured at day 13 of the experiment. All data are presented as the mean ± SEM. **P < 0.01 vs. the other groups (n = 4–7).
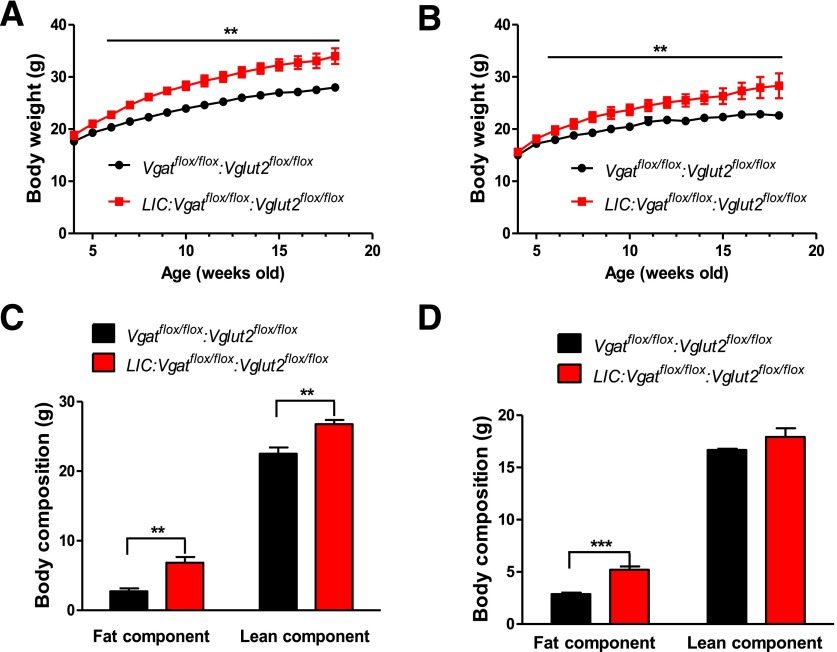
Because both glutamate and GABA released from LepR neurons are required for body weight regulation, it is possible that they are both engaged in leptin action on euglycemia restoration in T1D and that glutamate release can compensate for loss of GABA release and vice versa. To examine this possibility, we generated LIC:Vglut2flox/flox:Vgatflox/flox mice, in which the release of glutamate and GABA from LepR neurons will be simultaneously disrupted. In LIC:Vglut2flox/flox:Vgatflox/flox mice, there was increased body weight in both males (Fig. 3A) and females (Fig. 3B). The increase in body weight was largely due to increased fat tissues (Fig. 3C, males, and Fig. 3D, females), suggesting an obesity phenotype. Interestingly, there was also a significant increase in lean mass in male LIC:Vglut2flox/flox:Vgatflox/flox mice (Fig. 3C). Notably, male LIC:Vglut2flox/flox:Vgatflox/flox mice were 6 g heavier than controls at 18 weeks of age, whereas our previous results showed that both LIC:Vglut2flox/flox and LIC:Vgatflox/flox mice were ∼3 g heavier (18,19). Similarly, female LIC:Vglut2flox/flox:Vgatflox/flox mice were 5 g heavier than controls at 18 weeks of age, whereas our previous results showed that LIC:Vglut2flox/flox and LIC:Vgatflox/flox mice each were ∼2 g heavier (18,19). Thus, it appears that disruption of glutamate and GABA from LepR neurons produces an additive effect on body weight gain, suggesting that glutamate and GABA released from LepR neurons mediate body weight regulation involving parallel neural pathways.
Figure 3.
Obesity in LIC:Vgatflox/flox:Vglut2 flox/flox mice. A and B: Weekly body weight changes in mice fed with normal chow in males (A) and females (B). **P < 0.01 vs. Vgatflox/flox:Vglut2flox/flox groups. C and D: Body mass changes were measured at 18 weeks old in males (C) and females (D). **P < 0.01, ***P < 0.001 vs. Vgatflox/flox:Vglut2flox/flox groups. All data are reported as the mean ± SEM. Males n = 8–12; females n = 5–8.
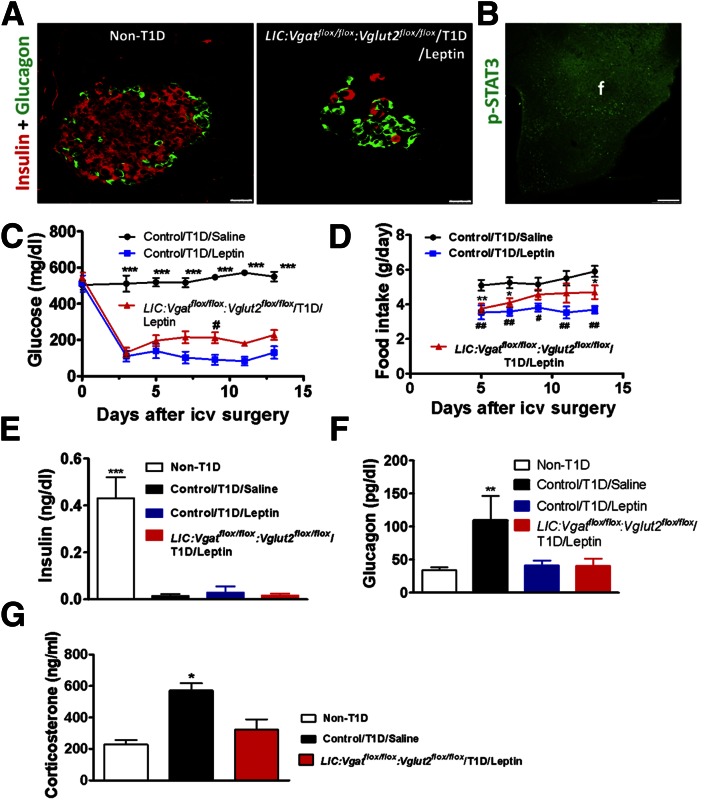
Compared with controls, STZ treatment led to a dramatic reduction in the number of β-cells in LIC:Vglut2flox/flox:Vgatflox/flox mice (Fig. 4A), suggesting the susceptibility of STZ-induced β-cell death in these mice. After 2 weeks of intracerebroventricular infusion of leptin, abundant expression of pSTAT3 was observed in the hypothalamus, compared with controls (Fig. 4B). Additional statistical analysis of groups of mice showed that leptin induced a dramatic increase in pSTAT3 in the hypothalamus compared with the saline-treated group (Supplementary Fig. 3), suggesting a successful delivery of leptin to the brain. In STZ-treated LIC:Vglut2flox/flox:Vgatflox/flox mice with intracerebroventricular saline infusion, glucose levels were maintained at >500 mg/dL throughout the experiment, suggesting the stable establishment of hyperglycemia. In leptin-treated mice, glucose levels, initially at ∼500 mg/dL at day 1, dropped to ∼150 mg/dL at day 3, and remained at this level for the rest of the period of the experiment (Fig. 4C), suggesting effective leptin action on reducing glucose levels. As expected, compared with saline-treated diabetic mice, leptin-treated mice showed reduced food intake (Fig. 4D). In addition, insulin levels in all STZ-treated animals were close to the detection limit (Fig. 4E), which is consistent with the loss of β-cells in the pancreatic islets, suggesting that euglycemia restoration in these mice was not due to an augmented insulin action. Consistent with the previous results, euglycemia restoration in these mice was associated with reduced glucagon levels (Fig. 4F). Because T1D is also associated with hypercorticosteronemia, we also measured corticosterone levels in these mice (7). Corticosterone levels were significantly higher in T1D, but were reduced to normal levels after leptin treatment (Fig. 4G), suggesting a tight association between glucose and corticosterone levels. Taken together, these data showed that glutamate and GABA release are not required for leptin action on euglycemia restoration in T1D.
Figure 4.
Effects of central leptin infusion in LIC:Vgatflox/flox:Vglut2flox/flox mice with T1D. A: Representative images of pancreata from non-T1D and T1D mice costained for insulin (red) and glucagon (green). Scale bars = 25 μm. B: Expression of pSTAT3 in the hypothalamus taken from LIC:Vgatflox/flox:Vglut2 flox/flox mice at day 13 of the experiment. Scale bar = 250 μm. f, fornix. C: Plasma glucose levels (n = 4–6). ***P < 0.001 vs. the other groups, #P < 0.05 vs. control/T1D/leptin group. D: Daily food intake (n = 5–6). *P < 0.05, **P < 0.01, #P < 0.05, ##P < 0.01 vs. control/T1D/saline group. E: Plasma insulin levels were measured at day 13 (n = 5). ***P < 0.001 vs. the other groups. Plasma glucagon (F) and corticosterone (G) levels were measured at day 13 (n = 5–6). *P < 0.05, **P < 0.01 vs. the other groups. All data are represented as the mean ± SEM. icv, intracerebroventricular.
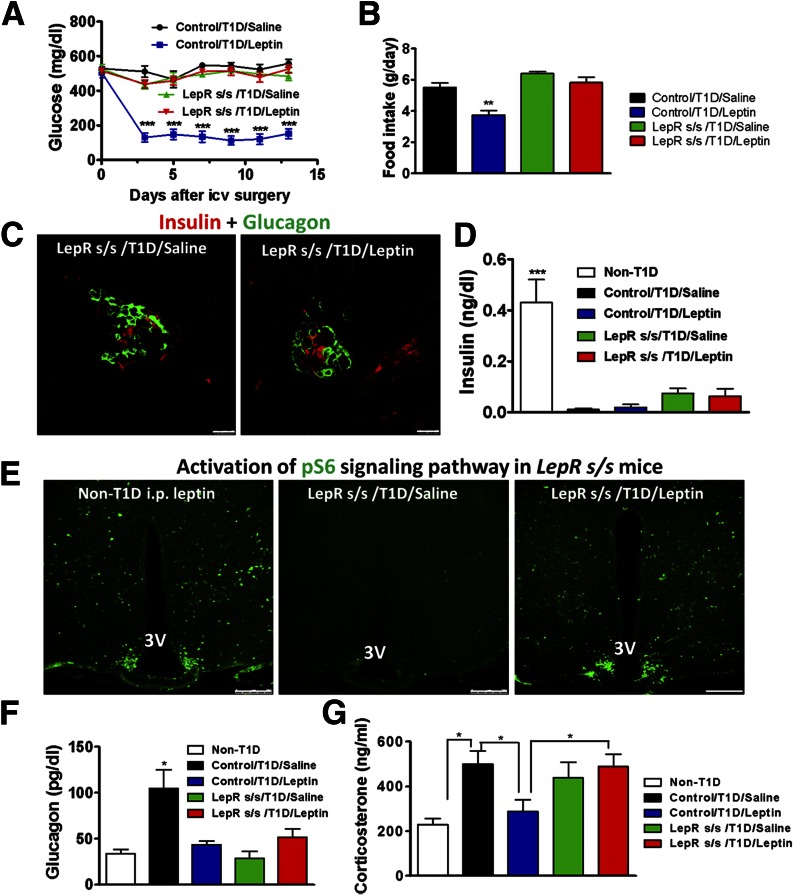
Several intracellular pathways mediate leptin action on body weight, among which the STAT3 pathway plays a major role (25,38). When treated with STZ, LepRs/s mice exhibited T1D with glucose levels of >500 mg/dL and remained hyperglycemic throughout the 14-day experimental period with intracerebroventricular saline infusion (Fig. 5A). Interestingly, with intracerebroventricular leptin infusion, glucose levels in these mice also remained at >500 mg/dL throughout the experimental period (Fig. 5A), suggesting that central leptin infusion failed to reduce glucose levels. Consistent with the glucose profile, although leptin-treated controls reduced food intake, leptin-treated LepRs/s mice remained hyperphagic at a level similar to that of saline-treated diabetic mice (Fig. 5B). STZ treatment led to a dramatic reduction in β-cell number in both saline- and leptin-treated LepRs/s mice (Fig. 5C), and insulin levels in both groups were near detection limit (Fig. 5D). Consistent with deficient LepR → pSTAT3 signaling in LepRs/s mice, leptin fusion failed to induce pSTAT3 in the hypothalamus of these mice (Supplementary Fig. 3). Central leptin activates ribosome protein S6 kinase, which is part of LepR-mTOR signaling, a distinct pathway from STAT3 signaling (34). Although the saline-treated hypothalamus exhibited no obvious p-S6 immunostaining, the one with intraperitoneal leptin induced robust p-S6 immunostaining, demonstrating the effective induction of p-S6 by leptin action (Fig. 5E). Central leptin infusion produced p-S6 expression comparable with that of intraperitoneal leptin injection (Fig. 5E). Additional statistical analysis of groups of mice showed that leptin induced a dramatic increase in p-S6 in the hypothalamus, compared with the saline-treated group (Supplementary Fig. 4), suggesting a successful delivery of leptin infusion. Taken together, these results suggest that disruption of the leptin → pSTAT3 pathway abrogates euglycemia restoration by central leptin in T1D.
Figure 5.
Disruption of the LepR → STAT3 signaling pathway abrogated the antidiabetic effect of central leptin. A: Plasma glucose levels (n = 4–7). ***P < 0.001 vs. the other groups. B: Daily food intake (n = 4–7). **P < 0.01 vs. the other groups. C: Representative images of pancreata from saline- and leptin-treated T1D LepRs/s mice costained for insulin (red) and glucagon (green). Scale bars = 25 μm. D: Plasma insulin levels were measured at day 13 (n = 4–6). ***P < 0.001 vs. all other groups. E: Expression of p-S6 by intraperitoneal (i.p.) leptin treatment in the hypothalamus of the indicated mouse groups. Scale bars = 250 μm. 3V, third ventricle. Levels of plasma glucagon (F) and corticosterone (G) were measured at day 13. *P < 0.05 vs. the other groups. All data are represented as the mean ± SEM. icv, intracerebroventricular.
Of interest, despite apparent uncontrolled hyperglycemia in LepRs/s mice treated with intracerebroventricular saline, glucagon levels in these mice were not increased and were maintained at a level comparable to that of control nondiabetic mice (Fig. 5F). In contrast to control mice in which treatment with intracerebroventricular leptin reduced glucagon levels, the same treatment failed to further reduce glucagon levels in LepRs/s mice (Fig. 5F). However, compared with non-T1D mice, both saline-treated T1D groups developed hypercorticosteronemia (Fig. 5G). Notably, although intracerebroventricular leptin treatment reduced corticosterone in control diabetic mice, it failed to do so in LepRs/s mice (Fig. 5G). These results suggest that hyperglucagonemia is not required for T1D development.
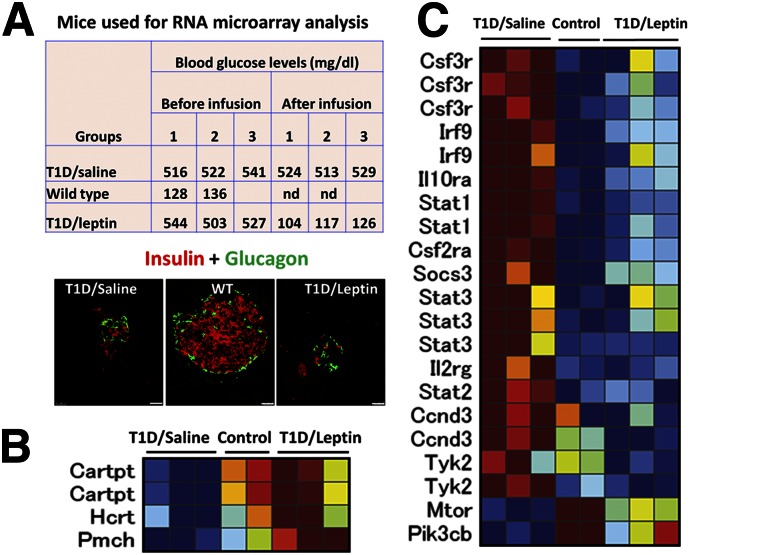
To further explore the mechanism by which central leptin reverses diabetes, we performed an RNA microarray analysis of hypothalamic tissues from mice with non-T1D, STZ-induced T1D, or STZ-induced T1D corrected by central leptin treatment. We were interested in those genes that respond to STZ-induced T1D and are reversed by leptin treatment. We performed a three-way comparison of the non-T1D, STZ-induced T1D, and leptin groups using an empirical Bayes approach (39) with a strict cutoff (false discovery rate <0.05, and at least twofold change in expression). We found that 179 genes showed increased, and 5 genes showed reduced, expression in STZ-induced T1D, which was reversed by leptin treatment (Gene Expression Omnibus accession number GSE 72227). Notably, levels of peptides, including CART, MCH, and hypocretin (Fig. 6B), were reduced by STZ and the reduction was reversed by leptin. Similarly, mTOR and PI3K levels were reduced by STZ and the reduction was reversed by leptin (Fig. 6C). Surprisingly, the expression of cytokine pathways, including interleukin-10 signaling, tyrosine kinase 2 (Tyk2), a known non–LepR-activated tyrosine kinase, and STAT1–STAT3, was increased by STZ treatment and also reversed by leptin (Fig. 6C). The observed changes in the expression of STAT1, STAT3, Carptpt, Ccnd3 (cyclin D3), and Tyk2 were independently confirmed by quantitative PCR (Supplementary Fig. 5). These results suggest a response in non–leptin-mediated cytokine pathways to STZ-induced T1D and its reversal by leptin treatment.
Figure 6.
Changes in the expression of selected genes in the microarray study. A: Plasma glucose levels and representative images of pancreata from T1D/saline mice, control mice, and T1D/leptin mice costained for insulin (red) and glucagon (green). nd, not determined. Scale bars = 25 μm. B: Heat map of RNA microarray showing that changes in the expression of representative neuropeptide genes by STZ-induced T1D were corrected by intracerebroventricular leptin infusion. C: Heat map of RNA microarray showing that changes in the expression of the representative genes in cytokine pathways by STZ-induced T1D were corrected by intracerebroventricular leptin infusion. The value of each gene expression was centered and normalized for clustering and display in the heat map.
Discussion
Previous studies have demonstrated that central leptin administration restores euglycemia in rodent models of T1D (4,8,20,21). The mechanism whereby leptin action in the brain improves glucose metabolism is largely unknown, and many published studies focused on the level of LepR (8,20,21). Thus, the current study focused on the role of intracellular signaling pathways mediating LepR function and that of neurotransmitter release mediating LepR neuron function. We found that the leptin → pSTAT3 signaling, but neither glutamate nor GABA release, is required for central leptin action on euglycemia restoration in T1D. In addition, we showed that LepRs/s mice developed T1D without concomitant hyperglucagonemia and that the failure of central leptin action in lowering glucose levels was correlated with a failure in reducing corticosterone levels, suggesting the importance of hypercorticosteronemia, but not hypergluconemia, in the development of T1D in STZ-treated LepRs/s mice.
LepR expression in GABAergic neurons is sufficient for leptin action on euglycemia restoration in T1D (20), suggesting a potential importance for GABA in mediating LepR neurons in insulin-independent glucose homeostasis. Surprisingly, our results showed that GABA release from LepR neurons was not required for the leptin action on euglycemia restoration. Notably, the negative result is not due to a compensatory action from glutamate release because the loss of release of either glutamate alone or glutamate and GABA leads to a normal response to leptin in euglycemia restoration. Thus, neither GABA nor glutamate from LepR neurons is required for leptin action on euglycemia restoration, suggesting that other neurotransmitters or neuropeptides mediate the leptin action.
Notably, the melanocortin signaling has been demonstrated to be required for leptin action in the insulin-independent glucose-lowering effect (40). Consistently, AgRP neurons and AgRP release from AgRP neurons may play an important role in reducing blood glucose levels in mice with deficient leptin action and hyperglycemia (41). Of note, a previous study (21) suggests that leptin in the VMH is sufficient but not required for leptin action on insulin-independent euglycemia restoration. Given the glutamatergic nature of VMH neurons (22), this result raises the possibility that glutamate release from VMH neurons is not required, but may be sufficient for the leptin action.
Loss of glutamate or GABA release from LepR neurons causes obesity (18,19), and our current results showed that the concurrent loss of both appeared to cause an additive effect over individual effects on obesity development. However, disrupted release of each individual or both from LepR neurons had no impact on the leptin effect in euglycemia restoration. These contrasting results indicate that the pathways mediating leptin action on body weight and glucose homeostasis can be segregated. Indeed, previous results show that the restoration of LepR in the arcuate nucleus of LepR-deficient mice dramatically improves glucose homeostasis while having mild effects on body weight (42). In addition, segregated pathways mediate the action of melanocortin-4 receptors in body weight and glucose homeostasis (43). Further studies are required to reveal the mechanism underlying the differential requirement and sufficiency of LepRs and neurotransmitters mediating leptin action on euglycemia restoration in T1D.
Within neurons, leptin action is mediated by several signaling pathways (23). Among them, the pSTAT3 pathway is the predominant one for body weight regulation, although other signaling pathways such as pSTAT5 are also important (38). Notably, mice with deficient LepR → pSTAT3 pathways show obesity that is comparable to that of LepR-deficient db/db mice, but they exhibit much lower blood glucose levels (25). Surprisingly, the current study showed that pSTAT3 was required for leptin action on euglycemia restoration in T1D. Given insulin deficiency in T1D, these results suggest that the improved glucose homeostasis in LepRs/s mice might be due to an augmented insulin action. Consistent with this, the insulin level in LepRs/s mice is higher than that in db/db mice (25), suggesting a role for non–LepR → pSTAT3 signaling in augmenting insulin levels. Compared with control mice, central leptin action produced a comparable signaling of p-S6 in the hypothalamus of T1D LepRs/s mice, whereas it failed to lower blood glucose levels, suggesting that the LepR-mTOR pathway is not sufficient for leptin action on insulin-independent glucose homeostasis. It will be of interest to examine whether the LepR → pSTAT3 pathway is sufficient to restore euglycemia in T1D. Because the central action of fibroblast growth factor 19 and brain-derived neurotrophic factor is also able to restore euglycemia (27), it is likely that the same pathway mediates their action. It is thus of interest to test whether their action depends on leptin → pSTAT3 signaling.
Most T1D models are accompanied by hyperglucagonemia, and glucose-lowering effects by leptin are also associated with reduced glucagon levels, suggesting a potential role for hyperglucagonemia in the development of T1D (4,5,20). Indeed, STZ-induced pancreatic β-cell ablation is not sufficient to produce hyperglycemia in mice with deficiency of glucagon receptor (6). In this regard, it is striking that our result showed that STZ-treated LepRs/s mice developed T1D without hyperglucagonemia, suggesting that hyperglucagonemia is not necessary for the development of T1D. Consistent with our result, mice with ablation of pancreatic α-cells are still susceptible to STZ-induced T1D development (44). Taken together, these results suggest that a basal level, but not an enhanced level, of glucagon receptor activation is required for T1D development. In addition, our results showed that central leptin action caused no changes in glucagon levels in LepRs/s mice, whereas it normalized glucagon levels from hyperglucagonemia in control mice, suggesting a potential role for LepR → pSTAT3 signaling in glucagon homeostasis.
STZ-treated LepRs/s mice developed T1D associated with hypercorticosteronemia, and the failure of leptin action was also associated with a failure in normalization of corticosterone levels. This result is consistent with recent findings on the role of hypercorticosteronemia and augmented hypothalamus-pituitary-adrenal (HPA) axis in T1D and in mediating the leptin action on euglycemia restoration in T1D (7). In this regard, it is surprising that the action of leptin on euglycemia restoration in STZ-induced T1D does not require HPA inhibition and cannot be blocked by hypercorticosteronemia (45). Despite potential concerns in using STZ-treated mice to model T1D on lacking immunogenicity and high levels of stress, both hyperglucagonemia and hypercorticosteronemia appear to be a common feature of T1D, including genetic rodent models (7), and the STZ-induced T1D has been successfully used previously to examine the mechanism underlying leptin action on euglycemia restoration (5,8,21). Thus, the contrasting results may not be due to a difference in the T1D models used. Alternatively, it is possible that, although both hyperglucagonemia and hypercorticosteronemia contribute to uncontrolled glucose levels in T1D, there may be other unknown factors that are responsible for T1D development and mediate leptin action on insulin-independent euglycemia restoration (46).
To further explore mechanisms underlying leptin action on glucose-lowering effects in T1D, we performed an unbiased microarray assay. To focus on those genes that show changes specific to leptin action on T1D, we analyzed those genes that respond to STZ-induced T1D and can be reversed by leptin. Consistent with the known role of leptin action in the hypothalamus (23,47,48), the expression of CART, MCH, PI3K, and mTOR was reduced by STZ and reversed by leptin. The increased mTOR expression is also correlated with the fact that central leptin action increased p-S6 levels. Given the recent finding of a role for hypocretin neurons in activating the HPA axis (49), it is surprising that hypocretin levels were reduced by STZ and reversed by leptin. This result suggests that the enhanced HPA axis in STZ-induced T1D may not be mediated by hypocretin neurons. However, it is intriguing that the gene expression for a subset of cytokine pathways including STAT3 was increased by STZ and also reversed by central leptin action. Increased STAT3 transcription in STZ-mediated mice with T1D may not be due to diminished leptin action because leptin deficiency is instead associated with lower STAT3 expression in the hypothalamus (50). The reason for the upregulation is unknown but may involve an interaction between leptin-mediated signaling and non–leptin-mediated signaling. For example, augmented action of interleukin-10 and Tyk2 may promote the expression of STATs. Further studies on other genes that responded to STZ-induced T1D and were reversed by leptin may reveal important novel mediators for leptin action on euglycemia restoration in T1D.
Supplementary Material
Article Information
Funding. This work was supported by National Institutes of Health (NIH) grants TL1-TR-000371 (to J.T.C.), DK-056731 (to M.G.M.), and R01-DK-093587 (to Yo.X.) and NIH grant R01-DK-092605, JDRF grant 1-2011-153, a Grant-in-Aid Award from the American Heart Association, and American Diabetes Association Basic Research Award 1-15-BS-184 (all to Q.T.). Q.T. is the holder of the Cullen Chair in Molecular Medicine and is a Welch Research Scholar (L-AU0002) at the McGovern Medical School of The University of Texas Health Science Center at Houston.
Duality of Interest. No potential conflicts of interest relevant to this article were reported.
Author Contributions. Yu.X. and Q.T. conceived and performed the experiments and wrote the article. J.T.C. contributed to the microarray data analysis. M.G.M. and Yo.X. provided essential reagents and guidance. Q.T. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Prior Presentation. Parts of this study were presented in abstract form at the 75th Scientific Sessions of the American Diabetes Association, Boston, MA, 5–9 June 2015.
Footnotes
This article contains Supplementary Data online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1160/-/DC1.
References
- 1.Chinookoswong N, Wang JL, Shi ZQ. Leptin restores euglycemia and normalizes glucose turnover in insulin-deficient diabetes in the rat. Diabetes 1999;48:1487–1492 [DOI] [PubMed] [Google Scholar]
- 2.Morton GJ, Gelling RW, Niswender KD, Morrison CD, Rhodes CJ, Schwartz MW. Leptin regulates insulin sensitivity via phosphatidylinositol-3-OH kinase signaling in mediobasal hypothalamic neurons. Cell Metab 2005;2:411–420 [DOI] [PubMed] [Google Scholar]
- 3.Kamohara S, Burcelin R, Halaas JL, Friedman JM, Charron MJ. Acute stimulation of glucose metabolism in mice by leptin treatment. Nature 1997;389:374–377 [DOI] [PubMed] [Google Scholar]
- 4.Wang MY, Chen L, Clark GO, et al. Leptin therapy in insulin-deficient type I diabetes. Proc Natl Acad Sci USA 2010;107:4813–4819 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Yu X, Park BH, Wang MY, Wang ZV, Unger RH. Making insulin-deficient type 1 diabetic rodents thrive without insulin. Proc Natl Acad Sci USA 2008;105:14070–14075 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lee Y, Wang MY, Du XQ, Charron MJ, Unger RH. Glucagon receptor knockout prevents insulin-deficient type 1 diabetes in mice. Diabetes 2011;60:391–397 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Perry RJ, Zhang XM, Zhang D, et al. Leptin reverses diabetes by suppression of the hypothalamic-pituitary-adrenal axis. Nat Med 2014;20:759–763 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fujikawa T, Chuang JC, Sakata I, Ramadori G, Coppari R. Leptin therapy improves insulin-deficient type 1 diabetes by CNS-dependent mechanisms in mice. Proc Natl Acad Sci USA 2010;107:17391–17396 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Denroche HC, Levi J, Wideman RD, et al. Leptin therapy reverses hyperglycemia in mice with streptozotocin-induced diabetes, independent of hepatic leptin signaling. Diabetes 2011;60:1414–1423 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cohen P, Zhao C, Cai X, et al. Selective deletion of leptin receptor in neurons leads to obesity. J Clin Invest 2001;108:1113–1121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schwartz MW, Seeley RJ, Campfield LA, Burn P, Baskin DG. Identification of targets of leptin action in rat hypothalamus. J Clin Invest 1996;98:1101–1106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Scott MM, Lachey JL, Sternson SM, et al. Leptin targets in the mouse brain. J Comp Neurol 2009;514:518–532 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Myers MG Jr, Münzberg H, Leinninger GM, Leshan RL. The geometry of leptin action in the brain: more complicated than a simple ARC. Cell Metab 2009;9:117–123 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Grill HJ. Distributed neural control of energy balance: contributions from hindbrain and hypothalamus. Obesity (Silver Spring) 2006;14(Suppl. 5):216S–221S [DOI] [PubMed] [Google Scholar]
- 15.Fulton S, Pissios P, Manchon RP, et al. Leptin regulation of the mesoaccumbens dopamine pathway. Neuron 2006;51:811–822 [DOI] [PubMed] [Google Scholar]
- 16.Lam DD, Leinninger GM, Louis GW, et al. Leptin does not directly affect CNS serotonin neurons to influence appetite. Cell Metab 2011;13:584–591 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Vong L, Ye C, Yang Z, Choi B, Chua S Jr, Lowell BB. Leptin action on GABAergic neurons prevents obesity and reduces inhibitory tone to POMC neurons. Neuron 2011;71:142–154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Xu Y, O’Brien WG 3rd, Lee CC, Myers MG Jr, Tong Q. Role of GABA release from leptin receptor-expressing neurons in body weight regulation. Endocrinology 2012;153:2223–2233 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Xu Y, Kim ER, Zhao R, Myers MG Jr, Munzberg H, Tong Q. Glutamate release mediates leptin action on energy expenditure. Mol Metab 2013;2:109–115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fujikawa T, Berglund ED, Patel VR, et al. Leptin engages a hypothalamic neurocircuitry to permit survival in the absence of insulin. Cell Metab 2013;18:431–444 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Meek TH, Matsen ME, Dorfman MD, et al. Leptin action in the ventromedial hypothalamic nucleus is sufficient, but not necessary, to normalize diabetic hyperglycemia. Endocrinology 2013;154:3067–3076 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tong Q, Ye C, McCrimmon RJ, et al. Synaptic glutamate release by ventromedial hypothalamic neurons is part of the neurocircuitry that prevents hypoglycemia. Cell Metab 2007;5:383–393 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Morris DL, Rui L. Recent advances in understanding leptin signaling and leptin resistance. Am J Physiol Endocrinol Metab 2009;297:E1247–E1259 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Robertson S, Ishida-Takahashi R, Tawara I, et al. Insufficiency of Janus kinase 2-autonomous leptin receptor signals for most physiologic leptin actions. Diabetes 2010;59:782–790 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bates SH, Stearns WH, Dundon TA, et al. STAT3 signalling is required for leptin regulation of energy balance but not reproduction. Nature 2003;421:856–859 [DOI] [PubMed] [Google Scholar]
- 26.Meek TH, Wisse BE, Thaler JP, et al. BDNF action in the brain attenuates diabetic hyperglycemia via insulin-independent inhibition of hepatic glucose production. Diabetes 2013;62:1512–1518 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Morton GJ, Matsen ME, Bracy DP, et al. FGF19 action in the brain induces insulin-independent glucose lowering. J Clin Invest 2013;123:4799–4808 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kim GH, Szabo A, King EM, Ayala J, Ayala JE, Altarejos JY. Leptin recruits Creb-regulated transcriptional coactivator 1 to improve hyperglycemia in insulin-deficient diabetes. Mol Metab 2015;4:227–236 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wojcik SM, Katsurabayashi S, Guillemin I, et al. A shared vesicular carrier allows synaptic corelease of GABA and glycine. Neuron 2006;50:575–587 [DOI] [PubMed] [Google Scholar]
- 30.Fremeau RT Jr, Voglmaier S, Seal RP, Edwards RH. VGLUTs define subsets of excitatory neurons and suggest novel roles for glutamate. Trends Neurosci 2004;27:98–103 [DOI] [PubMed] [Google Scholar]
- 31.Leshan RL, Opland DM, Louis GW, et al. Ventral tegmental area leptin receptor neurons specifically project to and regulate cocaine- and amphetamine-regulated transcript neurons of the extended central amygdala. J Neurosci 2010;30:5713–5723 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tong Q, Ye CP, Jones JE, Elmquist JK, Lowell BB. Synaptic release of GABA by AgRP neurons is required for normal regulation of energy balance. Nat Neurosci 2008;11:998–1000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Madisen L, Zwingman TA, Sunkin SM, et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat Neurosci 2010;13:133–140 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gong Y, Ishida-Takahashi R, Villanueva EC, Fingar DC, Münzberg H, Myers MG Jr. The long form of the leptin receptor regulates STAT5 and ribosomal protein S6 via alternate mechanisms. J Biol Chem 2007;282:31019–31027 [DOI] [PubMed] [Google Scholar]
- 35.Xu Y, Wu Z, Sun H, et al. Glutamate mediates the function of melanocortin receptor 4 on Sim1 neurons in body weight regulation. Cell Metab 2013;18:860–870 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ritchie ME, Phipson B, Wu D, et al. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res 2015;43:e47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Gentleman RC, Carey VJ, Bates DM, et al. Bioconductor: open software development for computational biology and bioinformatics. Genome Biol 2004;5:R80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Patterson CM, Villanueva EC, Greenwald-Yarnell M, et al. Leptin action via LepR-b Tyr1077 contributes to the control of energy balance and female reproduction. Mol Metab 2012;1:61–69 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Efron B. Empirical Bayes estimates for large-scale prediction problems. J Am Stat Assoc 2009;104:1015–1028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Meek TH, Matsen ME, Damian V, Cubelo A, Chua SC Jr, Morton GJ. Role of melanocortin signaling in neuroendocrine and metabolic actions of leptin in male rats with uncontrolled diabetes. Endocrinology 2014;155:4157–4167 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Gonçalves GH, Li W, Garcia AV, Figueiredo MS, Bjørbæk C. Hypothalamic agouti-related peptide neurons and the central melanocortin system are crucial mediators of leptin’s antidiabetic actions. Cell Reports 2014;7:1093–1103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Coppari R, Ichinose M, Lee CE, et al. The hypothalamic arcuate nucleus: a key site for mediating leptin’s effects on glucose homeostasis and locomotor activity. Cell Metab 2005;1:63–72 [DOI] [PubMed] [Google Scholar]
- 43.Rossi J, Balthasar N, Olson D, et al. Melanocortin-4 receptors expressed by cholinergic neurons regulate energy balance and glucose homeostasis. Cell Metab 2011;13:195–204 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Thorel F, Damond N, Chera S, et al. Normal glucagon signaling and β-cell function after near-total α-cell ablation in adult mice. Diabetes 2011;60:2872–2882 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Morton GJ, Meek TH, Matsen ME, Schwartz MW. Evidence against hypothalamic-pituitary-adrenal axis suppression in the antidiabetic action of leptin. J Clin Invest 2015;2015:4587–4591 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Edgerton DS, Cherrington AD. Glucagon as a critical factor in the pathology of diabetes. Diabetes 2011;60:377–380 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Huang Q, Viale A, Picard F, Nahon J, Richard D. Effects of leptin on melanin-concentrating hormone expression in the brain of lean and obese Lep(ob)/Lep(ob) mice. Neuroendocrinology 1999;69:145–153 [DOI] [PubMed] [Google Scholar]
- 48.Kristensen P, Judge ME, Thim L, et al. Hypothalamic CART is a new anorectic peptide regulated by leptin. Nature 1998;393:72–76 [DOI] [PubMed] [Google Scholar]
- 49.Bonnavion P, Jackson AC, Carter ME, de Lecea L. Antagonistic interplay between hypocretin and leptin in the lateral hypothalamus regulates stress responses. Nat Commun 2015;6:6266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Håkansson-Ovesjö ML, Collin M, Meister B. Down-regulated STAT3 messenger ribonucleic acid and STAT3 protein in the hypothalamic arcuate nucleus of the obese leptin-deficient (ob/ob) mouse. Endocrinology 2000;141:3946–3955 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.