Abstract
Thirty years have passed since the first report of portal vein embolization (PVE), and this procedure is widely adopted as a preoperative treatment procedure for patients with a small future liver remnant (FLR). PVE has been shown to be useful in patients with hepatocellular carcinoma (HCC) and chronic liver disease. However, special caution is needed when PVE is applied prior to subsequent major hepatic resection in cases with cirrhotic livers, and volumetric analysis of the liver segments in addition to evaluation of the liver functional reserve before PVE is mandatory in such cases. Advances in the embolic material and selection of the treatment approach, and combined use of PVE and transcatheter arterial embolization/chemoembolization have yielded improved outcomes after PVE and major hepatic resections. A novel procedure termed the associating liver partition and portal vein ligation for staged hepatectomy has been gaining attention because of the rapid hypertrophy of the FLR observed in patients undergoing this procedure, however, application of this technique in HCC patients requires special caution, as it has been shown to be associated with a high morbidity and mortality even in cases with essentially healthy livers.
Keywords: Hepatocellular carcinoma, Future liver remnant, Poral vein embolization, Liver functional reserve, The associating liver partition and portal vein ligation for staged hepatectomy
Core tip: Preoperative portal vein embolization (PVE) has been developed to secure the safety of a major hepatic resection by inducing the hypertrophy of the future liver remnant. PVE has been shown to be useful for patients with hepatocellular carcinoma and chronic liver disease. However, the indications should be carefully judged based on the volumetric analysis and evaluation of the liver functional reserve. Recently, a novel technique called the associating liver partition and portal vein ligation for staged hepatectomy (ALPPS) has been introduced to gain a rapid hypertrophy of the future liver remnant; however, at present, data supporting ALPPS in hepatocellular carcinoma with cirrhosis are still very weak.
INTRODUCTION
Currently, hepatic resection is the treatment of choice for large hepatocellular carcinomas (HCC), colorectal liver metastases (CLM) and hilar cholangiocarcinomas, and extensive liver resection is often required in patients with these malignancies. Preoperative portal vein embolization (PVE), which induces atrophy of the liver segments to be resected and hypertrophy of the future liver remnant (FLR), has been introduced in an attempt to expand the indications for major (the resection of 3 or more Couinaud segments[1]) hepatic resection and prevent postoperative liver insufficiency. Thirty years have passed since the first report of PVE by Makuuchi et al[2], and the usefulness of PVE is currently widely accepted. However, the beneficial effect of preoperative PVE may be impaired in patients with chronic liver disease, especially liver cirrhosis[3], and caution is required when PVE is applied in patients with large HCCs and underlying liver cirrhosis. In such patients, volumetric analysis of the liver segments in addition to evaluation of the liver functional reserve is mandatory[4]. On the other hand, some European groups have recently advocated the usefulness of a new procedure termed the associating liver partition and portal vein ligation for staged hepatectomy (ALPPS)[5], as this procedure has been shown to induce rapid hypertrophy of the FLR within a short interval[6]. However, application of ALPPS to HCC patients with underlying liver cirrhosis is debatable from the point of view of the safety. In this manuscript, we have reviewed the recent advances in preoperative PVE and other procedures aimed at increasing the FLR.
HISTORY OF PVE
In the first report, Makuuchi et al[7] applied preoperative PVE for patients with hilar cholangiocarcinoma. They stated that the purposes of PVE were: (1) to initiate compensatory hypertrophy of the FLR; and (2) to avoid a sudden increase of the portal venous pressure during and after the surgery[7]. The second goal is especially important in HCC patients with portal hypertension, where PVE may serve as a preoperative “tolerance test”; if the FLR cannot tolerate the higher portal pressure induced by PVE, sufficient hypertrophy of the FLR cannot be expected. Two approaches were used for PVE: Transileocolic portal embolization (TIPE) via laparotomy under general anesthesia, and percutaneous transhepatic portal embolization (PTPE) using a puncture technique with ultrasonic guidance under local anesthesia. The embolic material consisted of a mixture of absorbable gelatin powder, contrast material, and antibiotics.
Kinoshita et al[8] performed selective PVE (THPE), wherein they used a contralateral approach to occlude the portal vein branch bearing the HCC tumor. The aim of selective PVE was to enhance the effect of transcatheter arterial embolization (TAE) and the accompanying hypertrophy of the nonembolized segments. They used gelatin sponge, thrombin mixed with glucose, or an adhesive mixture of fibrin with contrast material as the embolic material.
Subsequently, the indication of preoperative PVE was expanded to other liver tumors, including CLM and HCC without cirrhosis. Among patients with CLM, PVE is indicated in patients with: (1) small multiple lesions of the right lobe; or (2) a small solitary tumor located adjacent to the hilum of the liver[9,10]. Reports dealing with PVE for HCC with underlying cirrhosis or chronic hepatitis were at first mainly small patient series from Asian countries, while documentations of large patient series have appeared after the year 2000[11-18]. The indications for PVE in cases of HCC is determined by the relationship between the liver functional reserve and the volumetric ratio of the FLR to the total liver volume. In general, major hepatic resection is contraindicated in Child-Pugh class B or C patients; these patients are therefore also not suitable candidates for PVE. In addition, Child-Pugh class A patients should undergo assessment by the indocyanine green retention rate at 15 min (ICG-R15). An ICG-R15 value of > 20% is generally considered as a contraindication for major hepatic resection and therefore also for PVE (Figure 1)[4].
Figure 1.
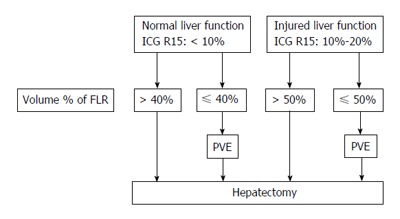
Indications of portal vein embolization for patients with hepatocellular carcinoma. PVE: Portal vein embolization; FLR: Future liver remnant; ICG R15: Indocyanine green retention ratio at 15 min.
MODIFICATION OF PVE
Approach
Several approaches have been advocated for PVE, which can be mainly categorized as TIPE or PTPE; the PTPE approach is further subdivided into an ipsilateral approach and a contralateral approach. TIPE is a safe approach; complete portography can be achieved using this approach, and insertion of the catheter into the segmental portal branches is relatively easy; however, it requires general anesthesia and laparotomy, and carries the risk of post-PVE bowel obstruction.
PTPE can be performed under local anesthesia, and is therefore considered to be a less invasive procedure; however, the possible risk of hemorrhage/subcapsular hematoma or peritonitis cannot be ignored, and if the contralateral approach is selected, injury to the vessels in the FLR may make the subsequent liver resection impossible. A meta-analysis showed that despite the absence of any significant difference in the rate of major complications between TIPE and PTPE, the rate for minor complications was significantly higher for PTPE[19].
Nagino et al[20] recommended the ipsilateral approach occluded the right anterior and posterior portal branches using different types of catheters. This technique is advantageous from the standpoint of safety, as the portal branch of the resected segments is punctured. Currently, PTPE using the ipsilateral approach, although the most technically demanding, is the most popularly used approach; however, the optimal approach must be selected according to the tumor location and past history of laparotomy.
Segment 4 embolization
When a more extended hepatic resection, such as right trisegmentectomy, is needed, embolization of the segment 4 portal branch in addition to the right portal vein branch may yield additional beneficial effects[21]. Embolization of the segment 4 branch is easy when the ipsilateral PTPE approach or TIPE is used. Two previous reports have confirmed the additional beneficial effect of embolization of the segment 4 portal branch on segment 2 + 3 hypertrophy, however, both reports dealt with non-injured livers, and no data are available for patients with underlying liver cirrhosis[22,23].
Embolic material
A number of embolic materials have been used for PVE, including gelatin sponge, gelatin powder, thrombin, fibrin glue, polyvinyl alcohol particles, absolute ethanol, cyanoacrylate, absolute ethanol, small spherical particles, and metallic coils[19]. The ideal agent would be the one that would lead to rapid, reproducible, and substantial functional hypertrophy of the FLR in the majority of patients without producing significant toxicity or adverse events. Currently, a combination of absolute ethanol and microcoils is widely used for HCC patients, as these agents have been shown to induce a greater degree of hypertrophy of the FLR as compared with other embolic materials[24]. However, there have been no randomized controlled trials to compare the efficiency of the embolic materials.
BASIC ASPECTS OF PVE
Liver regeneration after PVE
The mechanism of liver hypertrophy/regeneration after PVE has been widely studied using animal models or in clinical settings. Several experimental results imply that the mechanism of liver regeneration after PVE/portal vein ligation (PVL) is different from that after hepatectomy, as indicated by the different response to follistatin[25]. The difference is fundamentally attributed to maintained or enhanced arterial blood flow to the embolized liver segments after PVE, or the presence per se of the embolized segments, and the atrophying embolized liver segments are supposed to retain their specific functions. In addition, negative regulators of hepatocytes proliferation (such as transforming growth factor-β and interleukin-1β) are strongly expressed in the embolized segments. These factors in the embolized segments may modify the whole process of regeneration after PVE, although no definitive conclusions have been made yet[26].
Enhancement of the effect of PVE
Various factors have been shown to influence the effect of PVE: Age, gender, body mass index, nutrition status, previous chemotherapy, diabetes mellitus, etc[26]. It has been shown that liver regeneration is impaired in chronically diseased livers[3]. Sugawara et al[14] have examined the clinical factors associated with liver hypertrophy after PVE in HCC patients, and have found that the hypertrophic effect was significantly enhanced when PVE was combined with TAE. Recently, Beppu et al[27] have shown a favorable effect of branched-chain amino acid supplementation on functional liver regeneration after PVE.
CLINICAL IMPLICATIONS OF PVE
Clinical outcomes after PVE and major hepatic resection
Clinically, the percent increase in the volume of the FLR in cirrhotic livers within the first 2-3 wk after PVE is reported to in the range of 5% to 10%[10-12], and the hypertrophy ratio of the FLR has also been reported to be approximately 1.3 to 1.5[10,11,13]. Others have reported a rate of hypertrophy in cirrhotic livers of 9 cm2/d at 2 wk[14]. These figures are significantly smaller than those reported in non-cirrhotic livers[14-17]. Nevertheless, most previous reports have documented the safety of the PVE procedure and of subsequent major hepatic resection even in cases with a cirrhotic liver[28-32].
Previous reports have documented satisfactory long-term results after PVE and subsequent major hepatic resection for HCC (Table 1)[11-18]. The reported 5-year survival rates range from 44% to 72%, and the reported 5-year disease-free survival rates range from 21% to 56%. These figures are comparable to those after major hepatic resections for HCC without PVE. It may be deduced that PVE does not have any adverse effect on the risk of oncogenesis (i.e., intrahepatic HCC recurrence or development of new primary lesions) in the FLR after hepatic resection.
Table 1.
Clinical outcomes of portal vein embolization for hepatocellular carcinoma
| Ref. | Year | Technique | No. of patients | Morbidity (%) | Mortality (%) | 5-yr disease-free survival (%) | 5-yr overall survival (%) |
| Azoulay et al[11] | 2000 | PVE | 10 | 55 | 0 | 21 | 44 |
| Tanaka et al[12] | 2000 | PVE | 33 | - | 3 | 33 | 50 |
| Wakabayashi et al[13] | 2001 | PVE | 26 | - | 11.5 | 40 | 46 |
| Sugawara et al[14] | 2002 | PVE | 66 | - | 0 | 37.9 | 58.9 |
| Aoki et al[15] | 2004 | TACE + PVE | 24 | 24 | 0 | 47 | 56 |
| Ogata et al[16] | 2006 | TACE + PVE | 18 | 39 | - | 37 | - |
| PVE | 18 | 56 | - | 19 | - | ||
| Seo et al[17] | 2007 | PVE | 32 | 19 | 0 | 37 | 72 |
| Palavecino et al[18] | 2009 | PVE | 21 | 24 | 0 | 56 | 72 |
PVE: Portal vein embolization; TACE: Transcatheter arterial chemoembolization.
PVE also has significance as a preoperative “tolerance test”. Indeed, if the liver cannot tolerate PVE, sufficient hypertrophy of the FLR cannot be expected, and a subsequent major hepatic resection is precluded. In addition to the volumetric increase of the FLR, the kinetic growth rate (speed of increase in the volume of the FLR) has also been shown to be a predictor of the morbidity and mortality after subsequent major hepatic resections[33].
Tumor growth after PVE
On the other hand, tumors in the nonembolized liver segments have been reported to grow more rapidly than tumors in the embolized segments. Alternatively, tumors in the nonembolized segments show an enhanced rate of progression as compared to their natural history. This possible underlying mechanisms for this observation are that: (1) the increased arterial blood supply to the nonembolized liver segments after PVE can promote tumor growth; and (2) the cytokines associated with the atrophy-hypertrophy complex can also promote the progression of tumors. Several previous reports have addressed this issue. Despite some conflicting results, accumulating evidence suggests an adverse effect of PVE on tumor growth[34-38], although most previous studies investigating the risk of tumor growth after PVE have dealt with patients having colorectal liver metastases.
Tumor growth after PVE, especially tumor growth in the nonembolized FLR and/or extrahepatic tumor progression, may preclude curative resection. Indeed, a recent meta-analysis reported that about 15% of patients could not undergo curative resection after PVE, and about a half of these patients showed severe tumor progression or extrahepatic tumor spread[19].
Sequential transcatheter arterial chemoembolization and PVE and two-staged hepatectomy
As mentioned above, the risk of tumor growth after PVE may counteract the beneficial effect of PVE. Therefore, measures to prevent tumor growth during the waiting period before hepatectomy should be considered.
Our group has employed combined transcatheter arterial chemoembolization (TACE) with PVE as a preoperative treatment in HCC patients. The antitumor effect of TACE in cases of HCC has been reported previously[39]. TACE is also useful for embolizing the arterio-portal shunts in the tumor. Thus, the combination of TACE plus PVE before planned major hepatic resection may strengthen the effect of PVE while simultaneously preventing tumor progression. Our study showed satisfactory short- and long-term outcomes after sequential preoperative TACE and PVE in 17 patients with HCC[15]. During the waiting period after PVE, tumor progression, as evaluated by measurements of the tumor volume, serum alpha-fetoprotein level, and plasma des-γ-carboxy prothrombin level, was significantly suppressed.
Another European group compared 18 patients who underwent sequential preoperative TACE and PVE with 18 patients who underwent PVE alone prior to hepatic resection[16]. All the patients underwent a right hepatectomy 4-8 wk after the PVE. They found that the degree of hypertrophy of the FLR was greater in the TACE + PVE group, and that the recurrence-free survival period was also significantly longer in the TACE + PVE group than that in the PVE alone group.
A potential concern of sequential TACE and PVE is infarction or necrosis of the non-cancerous liver parenchyma. Our previous results showed, however, that necrosis of the non-cancerous liver parenchyma in the resected specimens was minimal. Possibly, recanalization of the hepatic artery abrogates the possible adverse effect of dual embolization.
Two-stage hepatectomy with preoperative PVE can also be applied in patients with metastatic liver tumors[40]. The tumors in the FLR are removed by limited resections as the first step, PVE is performed as the second step, and finally, the major hepatic resection is carried out as the third and final step. This strategy is fascinating, but is rarely performed in HCC patients as the surgical indications for bilobar multiple HCC are extremely limited.
ALTERNATIVES TO PVE
PVE vs PVL vs ALPPS
In general, PVL at the right branch is believed to induce canalization of the intrahepatic communications of the peripheral portal branches within a few days, therefore, PVE is considered to be more efficient as compared to PVL. However, a meta-analysis has shown only a borderline difference in the increase of the FLR volume after PVE and PVL. The morbidity and mortality of the two procedures are similar[41].
Recently, European groups have reported a novel approach to rapid liver regeneration in patients scheduled for extended right hepatectomy. This procedure, termed ALPPS, consists of right portal ligation and in situ splitting of the liver parenchyma on the right side of the umbilical portion of the portal vein. Schnitzbauer et al[5], who published the first report of this procedure, reported a marked and rapid hypertrophy of about 75% of the left lateral lobe within a median of 9 d. This growth rate has been reported to be 11 times higher as compared to that after PVE/PVL, and comparable to that in donors after living donor liver transplantation[42]. The mechanisms of the apparent profound hepatic growth of the FLR after ALPPS are unknown, although probably this noteworthy phenomenon may be attributable to an abruption of the arterial blood flow between the two parts of the liver. The same group and others also documented that ALPPS significantly improved the chance of curative resection for initially unresectable liver tumors as compared to conventional PVE/PVL[6,43].
The concern about this procedure, however, is the high morbidity and mortality rates associated with it[44,45]. The reported 90-d mortality after ALPPS is 15%, while that after PVE/PVL is 6%, and the odds ratio for perioperative death was 2.7-fold higher in the patients who underwent ALPPS[6]. In addition, a high recurrence rate within a short follow-up period has also been reported[46]. Based on these observations, Shindoh et al[47] concluded that PVE (right portal branch plus segment 4) and interval surgery remain the standard for patients with small FLRs.
Is ALPPS applicable to HCC patients with cirrhosis? The ALPPS series included some patients with HCC (about 10% of the patients), and some recent papers have documented that ALPPS can be safely performed in HCC patients with cirrhosis; however, no detailed data are available because of the small number of patients[5,6,48]. Currently, the indications of ALPPS for HCC patients are extremely limited and each patient should be carefully examined as to his/her suitability to undergo ALPPS.
Radioembolization
Our group has applied a combination of preoperative TACE and PVE to prevent tumor progression during the waiting period before surgery. An alternative to this strategy is radioembolization, which treats the tumor in the embolized lobe along with induction of contralateral hypertrophy. An increase in the size of the non-embolized lobe by 42% after radioembolization has been reported in cirrhotic livers[49]. A comparison of PVE and radioembolization in non-cirrhotic livers has shown that PVE induces a greater degree of hypertrophy of the FLR than that radioembolization[50]. Nevertheless, this novel procedure is promising, as it enables both embolization and treatment of the tumor(s) in a single step.
CONCLUSION
Much basic and clinical evidence associated with PVE has been accumulated, however, especially for cases of HCC with underlying liver cirrhosis or chronic hepatitis, the available clinical data are limited. Development of safe and reliable novel approaches that can be used in combination with PVE to induce rapid hypertrophy of FLR, which can be applied even to chronically diseased livers, is needed.
Footnotes
Conflict-of-interest statement: The authors have no conflict of interest to report.
Open-Access: This article is an open-access article which was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
Peer-review started: May 8, 2015
First decision: September 8, 2015
Article in press: March 9, 2016
P- Reviewer: de Santibañes E, Edeline J S- Editor: Gong ZM L- Editor: A E- Editor: Liu SQ
References
- 1.Couinaud C. Le Foie, Etude Anatomiques et Chirurgicales. Paris, France: Masson; 1957. [Google Scholar]
- 2.Makuuchi M, Takayasu K, Takuma T, Yamazaki S, Hasegawa H, Nishimura S, Shimamura Y. Preoperative transcatheter embolization of the portal venous branch for patients receiving extended lobectomy due to the bile duct carcinoma. J Jpn Soc Clin Surg. 1984;45:14–20. [Google Scholar]
- 3.Chen MF, Hwang TL, Hung CF. Human liver regeneration after major hepatectomy. A study of liver volume by computed tomography. Ann Surg. 1991;213:227–229. doi: 10.1097/00000658-199103000-00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kubota K, Makuuchi M, Kusaka K, Kobayashi T, Miki K, Hasegawa K, Harihara Y, Takayama T. Measurement of liver volume and hepatic functional reserve as a guide to decision-making in resectional surgery for hepatic tumors. Hepatology. 1997;26:1176–1181. doi: 10.1053/jhep.1997.v26.pm0009362359. [DOI] [PubMed] [Google Scholar]
- 5.Schnitzbauer AA, Lang SA, Goessmann H, Nadalin S, Baumgart J, Farkas SA, Fichtner-Feigl S, Lorf T, Goralcyk A, Hörbelt R, et al. Right portal vein ligation combined with in situ splitting induces rapid left lateral liver lobe hypertrophy enabling 2-staged extended right hepatic resection in small-for-size settings. Ann Surg. 2012;255:405–414. doi: 10.1097/SLA.0b013e31824856f5. [DOI] [PubMed] [Google Scholar]
- 6.Schadde E, Ardiles V, Slankamenac K, Tschuor C, Sergeant G, Amacker N, Baumgart J, Croome K, Hernandez-Alejandro R, Lang H, et al. ALPPS offers a better chance of complete resection in patients with primarily unresectable liver tumors compared with conventional-staged hepatectomies: results of a multicenter analysis. World J Surg. 2014;38:1510–1519. doi: 10.1007/s00268-014-2513-3. [DOI] [PubMed] [Google Scholar]
- 7.Makuuchi M, Thai BL, Takayasu K, Takayama T, Kosuge T, Gunvén P, Yamazaki S, Hasegawa H, Ozaki H. Preoperative portal embolization to increase safety of major hepatectomy for hilar bile duct carcinoma: a preliminary report. Surgery. 1990;107:521–527. [PubMed] [Google Scholar]
- 8.Kinoshita H, Sakai K, Hirohashi K, Igawa S, Yamasaki O, Kubo S. Preoperative portal vein embolization for hepatocellular carcinoma. World J Surg. 1986;10:803–808. doi: 10.1007/BF01655244. [DOI] [PubMed] [Google Scholar]
- 9.Kawasaki S, Makuuchi M, Kakazu T, Miyagawa S, Takayama T, Kosuge T, Sugihara K, Moriya Y. Resection for multiple metastatic liver tumors after portal embolization. Surgery. 1994;115:674–677. [PubMed] [Google Scholar]
- 10.Imamura H, Shimada R, Kubota M, Matsuyama Y, Nakayama A, Miyagawa S, Makuuchi M, Kawasaki S. Preoperative portal vein embolization: an audit of 84 patients. Hepatology. 1999;29:1099–1105. doi: 10.1002/hep.510290415. [DOI] [PubMed] [Google Scholar]
- 11.Azoulay D, Castaing D, Krissat J, Smail A, Hargreaves GM, Lemoine A, Emile JF, Bismuth H. Percutaneous portal vein embolization increases the feasibility and safety of major liver resection for hepatocellular carcinoma in injured liver. Ann Surg. 2000;232:665–672. doi: 10.1097/00000658-200011000-00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tanaka H, Hirohashi K, Kubo S, Shuto T, Higaki I, Kinoshita H. Preoperative portal vein embolization improves prognosis after right hepatectomy for hepatocellular carcinoma in patients with impaired hepatic function. Br J Surg. 2000;87:879–882. doi: 10.1046/j.1365-2168.2000.01438.x. [DOI] [PubMed] [Google Scholar]
- 13.Wakabayashi H, Ishimura K, Okano K, Izuishi K, Karasawa Y, Goda F, Maeba T, Maeta H. Is preoperative portal vein embolization effective in improving prognosis after major hepatic resection in patients with advanced-stage hepatocellular carcinoma? Cancer. 2001;92:2384–2390. doi: 10.1002/1097-0142(20011101)92:9<2384::aid-cncr1586>3.0.co;2-h. [DOI] [PubMed] [Google Scholar]
- 14.Sugawara Y, Yamamoto J, Higashi H, Yamasaki S, Shimada K, Kosuge T, Takayama T, Makuuchi M. Preoperative portal embolization in patients with hepatocellular carcinoma. World J Surg. 2002;26:105–110. doi: 10.1007/s00268-001-0189-y. [DOI] [PubMed] [Google Scholar]
- 15.Aoki T, Imamura H, Hasegawa K, Matsukura A, Sano K, Sugawara Y, Kokudo N, Makuuchi M. Sequential preoperative arterial and portal venous embolizations in patients with hepatocellular carcinoma. Arch Surg. 2004;139:766–774. doi: 10.1001/archsurg.139.7.766. [DOI] [PubMed] [Google Scholar]
- 16.Ogata S, Belghiti J, Farges O, Varma D, Sibert A, Vilgrain V. Sequential arterial and portal vein embolizations before right hepatectomy in patients with cirrhosis and hepatocellular carcinoma. Br J Surg. 2006;93:1091–1098. doi: 10.1002/bjs.5341. [DOI] [PubMed] [Google Scholar]
- 17.Seo DD, Lee HC, Jang MK, Min HJ, Kim KM, Lim YS, Chung YH, Lee YS, Suh DJ, Ko GY, et al. Preoperative portal vein embolization and surgical resection in patients with hepatocellular carcinoma and small future liver remnant volume: comparison with transarterial chemoembolization. Ann Surg Oncol. 2007;14:3501–3509. doi: 10.1245/s10434-007-9553-y. [DOI] [PubMed] [Google Scholar]
- 18.Palavecino M, Chun YS, Madoff DC, Zorzi D, Kishi Y, Kaseb AO, Curley SA, Abdalla EK, Vauthey JN. Major hepatic resection for hepatocellular carcinoma with or without portal vein embolization: Perioperative outcome and survival. Surgery. 2009;145:399–405. doi: 10.1016/j.surg.2008.10.009. [DOI] [PubMed] [Google Scholar]
- 19.Abulkhir A, Limongelli P, Healey AJ, Damrah O, Tait P, Jackson J, Habib N, Jiao LR. Preoperative portal vein embolization for major liver resection: a meta-analysis. Ann Surg. 2008;247:49–57. doi: 10.1097/SLA.0b013e31815f6e5b. [DOI] [PubMed] [Google Scholar]
- 20.Nagino M, Nimura Y, Kamiya J, Kondo S, Kanai M. Selective percutaneous transhepatic embolization of the portal vein in preparation for extensive liver resection: the ipsilateral approach. Radiology. 1996;200:559–563. doi: 10.1148/radiology.200.2.8685357. [DOI] [PubMed] [Google Scholar]
- 21.Nagino M, Nimura Y, Kamiya J, Kondo S, Uesaka K, Kin Y, Kutsuna Y, Hayakawa N, Yamamoto H. Right or left trisegment portal vein embolization before hepatic trisegmentectomy for hilar bile duct carcinoma. Surgery. 1995;117:677–681. doi: 10.1016/s0039-6060(95)80012-3. [DOI] [PubMed] [Google Scholar]
- 22.Nagino M, Kamiya J, Kanai M, Uesaka K, Sano T, Yamamoto H, Hayakawa N, Nimura Y. Right trisegment portal vein embolization for biliary tract carcinoma: technique and clinical utility. Surgery. 2000;127:155–160. doi: 10.1067/msy.2000.101273. [DOI] [PubMed] [Google Scholar]
- 23.Kishi Y, Madoff DC, Abdalla EK, Palavecino M, Ribero D, Chun YS, Vauthey JN. Is embolization of segment 4 portal veins before extended right hepatectomy justified? Surgery. 2008;144:744–751. doi: 10.1016/j.surg.2008.05.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Madoff DC, Hicks ME, Abdalla EK, Morris JS, Vauthey JN. Portal vein embolization with polyvinyl alcohol particles and coils in preparation for major liver resection for hepatobiliary malignancy: safety and effectiveness--study in 26 patients. Radiology. 2003;227:251–260. doi: 10.1148/radiol.2271012010. [DOI] [PubMed] [Google Scholar]
- 25.Kogure K, Omata W, Kanzaki M, Zhang YQ, Yasuda H, Mine T, Kojima I. A single intraportal administration of follistatin accelerates liver regeneration in partially hepatectomized rats. Gastroenterology. 1995;108:1136–1142. doi: 10.1016/0016-5085(95)90212-0. [DOI] [PubMed] [Google Scholar]
- 26.Yokoyama Y, Nagino M, Nimura Y. Mechanisms of hepatic regeneration following portal vein embolization and partial hepatectomy: a review. World J Surg. 2007;31:367–374. doi: 10.1007/s00268-006-0526-2. [DOI] [PubMed] [Google Scholar]
- 27.Beppu T, Nitta H, Hayashi H, Imai K, Okabe H, Nakagawa S, Hashimoto D, Chikamoto A, Ishiko T, Yoshida M, et al. Effect of branched-chain amino acid supplementation on functional liver regeneration in patients undergoing portal vein embolization and sequential hepatectomy: a randomized controlled trial. J Gastroenterol. 2015;50:1197–1205. doi: 10.1007/s00535-015-1067-y. [DOI] [PubMed] [Google Scholar]
- 28.de Baere T, Roche A, Elias D, Lasser P, Lagrange C, Bousson V. Preoperative portal vein embolization for extension of hepatectomy indications. Hepatology. 1996;24:1386–1391. doi: 10.1053/jhep.1996.v24.pm0008938166. [DOI] [PubMed] [Google Scholar]
- 29.Lee KC, Kinoshita H, Hirohashi K, Kubo S, Iwasa R. Extension of surgical indications for hepatocellular carcinoma by portal vein embolization. World J Surg. 1993;17:109–115. doi: 10.1007/BF01655721. [DOI] [PubMed] [Google Scholar]
- 30.Nagino M, Nimura Y, Kamiya J, Kondo S, Uesaka K, Kin Y, Hayakawa N, Yamamoto H. Changes in hepatic lobe volume in biliary tract cancer patients after right portal vein embolization. Hepatology. 1995;21:434–439. [PubMed] [Google Scholar]
- 31.Yamanaka N, Okamoto E, Kawamura E, Kato T, Oriyama T, Fujimoto J, Furukawa K, Tanaka T, Tomoda F, Tanaka W. Dynamics of normal and injured human liver regeneration after hepatectomy as assessed on the basis of computed tomography and liver function. Hepatology. 1993;18:79–85. [PubMed] [Google Scholar]
- 32.Shimamura T, Nakajima Y, Une Y, Namieno T, Ogasawara K, Yamashita K, Haneda T, Nakanishi K, Kimura J, Matsushita M, et al. Efficacy and safety of preoperative percutaneous transhepatic portal embolization with absolute ethanol: a clinical study. Surgery. 1997;121:135–141. doi: 10.1016/s0039-6060(97)90282-8. [DOI] [PubMed] [Google Scholar]
- 33.Shindoh J, Truty MJ, Aloia TA, Curley SA, Zimmitti G, Huang SY, Mahvash A, Gupta S, Wallace MJ, Vauthey JN. Kinetic growth rate after portal vein embolization predicts posthepatectomy outcomes: toward zero liver-related mortality in patients with colorectal liver metastases and small future liver remnant. J Am Coll Surg. 2013;216:201–209. doi: 10.1016/j.jamcollsurg.2012.10.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Elias D, De Baere T, Roche A, Mducreux J, Lasser P. During liver regeneration following right portal embolization the growth rate of liver metastases is more rapid than that of the liver parenchyma. Br J Surg. 1999;86:784–788. doi: 10.1046/j.1365-2168.1999.01154.x. [DOI] [PubMed] [Google Scholar]
- 35.Kokudo N, Tada K, Seki M, Ohta H, Azekura K, Ueno M, Ohta K, Yamaguchi T, Matsubara T, Takahashi T, et al. Proliferative activity of intrahepatic colorectal metastases after preoperative hemihepatic portal vein embolization. Hepatology. 2001;34:267–272. doi: 10.1053/jhep.2001.26513. [DOI] [PubMed] [Google Scholar]
- 36.Barbaro B, Di Stasi C, Nuzzo G, Vellone M, Giuliante F, Marano P. Preoperative right portal vein embolization in patients with metastatic liver disease. Metastatic liver volumes after RPVE. Acta Radiol. 2003;44:98–102. [PubMed] [Google Scholar]
- 37.Hayashi S, Baba Y, Ueno K, Nakajo M, Kubo F, Ueno S, Aikou T, Komokata T, Nakamura N, Sakata R. Acceleration of primary liver tumor growth rate in embolized hepatic lobe after portal vein embolization. Acta Radiol. 2007;48:721–727. doi: 10.1080/02841850701424514. [DOI] [PubMed] [Google Scholar]
- 38.Hoekstra LT, van Lienden KP, Doets A, Busch OR, Gouma DJ, van Gulik TM. Tumor progression after preoperative portal vein embolization. Ann Surg. 2012;256:812–817; discussion 817-818. doi: 10.1097/SLA.0b013e3182733f09. [DOI] [PubMed] [Google Scholar]
- 39.Llovet JM, Bruix J. Systematic review of randomized trials for unresectable hepatocellular carcinoma: Chemoembolization improves survival. Hepatology. 2003;37:429–442. doi: 10.1053/jhep.2003.50047. [DOI] [PubMed] [Google Scholar]
- 40.Jaeck D, Oussoultzoglou E, Rosso E, Greget M, Weber JC, Bachellier P. A two-stage hepatectomy procedure combined with portal vein embolization to achieve curative resection for initially unresectable multiple and bilobar colorectal liver metastases. Ann Surg. 2004;240:1037–1049; discussion 1049-1051. doi: 10.1097/01.sla.0000145965.86383.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pandanaboyana S, Bell R, Hidalgo E, Toogood G, Prasad KR, Bartlett A, Lodge JP. A systematic review and meta-analysis of portal vein ligation versus portal vein embolization for elective liver resection. Surgery. 2015;157:690–698. doi: 10.1016/j.surg.2014.12.009. [DOI] [PubMed] [Google Scholar]
- 42.Croome KP, Hernandez-Alejandro R, Parker M, Heimbach J, Rosen C, Nagorney DM. Is the liver kinetic growth rate in ALPPS unprecedented when compared with PVE and living donor liver transplant? A multicentre analysis. HPB (Oxford) 2015;17:477–484. doi: 10.1111/hpb.12386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Alvarez FA, Ardiles V, de Santibañes M, Pekolj J, de Santibañes E. Associating liver partition and portal vein ligation for staged hepatectomy offers high oncological feasibility with adequate patient safety: a prospective study at a single center. Ann Surg. 2015;261:723–732. doi: 10.1097/SLA.0000000000001046. [DOI] [PubMed] [Google Scholar]
- 44.Schadde E, Ardiles V, Robles-Campos R, Malago M, Machado M, Hernandez-Alejandro R, Soubrane O, Schnitzbauer AA, Raptis D, Tschuor C, et al. Early survival and safety of ALPPS: first report of the International ALPPS Registry. Ann Surg. 2014;260:829–836; discussion 836-838. doi: 10.1097/SLA.0000000000000947. [DOI] [PubMed] [Google Scholar]
- 45.Schadde E, Schnitzbauer AA, Tschuor C, Raptis DA, Bechstein WO, Clavien PA. Systematic review and meta-analysis of feasibility, safety, and efficacy of a novel procedure: associating liver partition and portal vein ligation for staged hepatectomy. Ann Surg Oncol. 2015;22:3109–3120. doi: 10.1245/s10434-014-4213-5. [DOI] [PubMed] [Google Scholar]
- 46.Sala S, Ardiles V, Ulla M, Alvarez F, Pekolj J, de Santibañes E. Our initial experience with ALPPS technique: encouraging results. Updates Surg. 2012;64:167–172. doi: 10.1007/s13304-012-0175-y. [DOI] [PubMed] [Google Scholar]
- 47.Shindoh J, Vauthey JN, Zimmitti G, Curley SA, Huang SY, Mahvash A, Gupta S, Wallace MJ, Aloia TA. Analysis of the efficacy of portal vein embolization for patients with extensive liver malignancy and very low future liver remnant volume, including a comparison with the associating liver partition with portal vein ligation for staged hepatectomy approach. J Am Coll Surg. 2013;217:126–133; discussion 133-134. doi: 10.1016/j.jamcollsurg.2013.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Vennarecci G, Laurenzi A, Levi Sandri GB, Busi Rizzi E, Cristofaro M, Montalbano M, Piselli P, Andreoli A, D’Offizi G, Ettorre GM. The ALPPS procedure for hepatocellular carcinoma. Eur J Surg Oncol. 2014;40:982–988. doi: 10.1016/j.ejso.2014.04.002. [DOI] [PubMed] [Google Scholar]
- 49.Edeline J, Lenoir L, Boudjema K, Rolland Y, Boulic A, Le Du F, Pracht M, Raoul JL, Clément B, Garin E, et al. Volumetric changes after (90)y radioembolization for hepatocellular carcinoma in cirrhosis: an option to portal vein embolization in a preoperative setting? Ann Surg Oncol. 2013;20:2518–2525. doi: 10.1245/s10434-013-2906-9. [DOI] [PubMed] [Google Scholar]
- 50.Garlipp B, de Baere T, Damm R, Irmscher R, van Buskirk M, Stübs P, Deschamps F, Meyer F, Seidensticker R, Mohnike K, et al. Left-liver hypertrophy after therapeutic right-liver radioembolization is substantial but less than after portal vein embolization. Hepatology. 2014;59:1864–1873. doi: 10.1002/hep.26947. [DOI] [PubMed] [Google Scholar]


