Abstract
Lung cancer is a common malignancy in men and the second leading cause of cancer-related mortality in men in the western world. Phenolic cocoa ingredients have a strong antioxidative activity and the potential to have a protective effect against cancer. In the present study, we have evaluated the influence of cocoa beans subjected to different processing conditions on cell viability and apoptosis of human lung cancer cells (A549). We measured the viability of lung cells treated with cocoa beans, unroasted slates (US), roasted slates (RS), unroasted well fermented (UWF) cocoa, and roasted well fermented (RWF) cocoa for 24 h. Using an MTT assay, we observed a decrease in the viability of A549 cells after treatment with cocoa bean extracts. Flow cytometer analysis revealed that cocoa beans increased the percentage of cells in sub-G1 phase and promoted up to twofold increase of apoptotic cells when compared to the control group. Taken together, the present study suggests that cocoa beans may have a protective effect against lung cancer.
1. Introduction
Lung cancer is a major health concern since it is one of the leading causes of death worldwide [1, 2]. It is estimated that the annual incidence rate is nearly 1.23 million. In Brazil, it is the second most frequent type of cancer and it has the highest mortality rate, due to late diagnosis and the aggressiveness of the tumor type. In 80–90% of cases, it is caused by smoking and exposure to pollutants. The existing therapeutic strategies for cell lung cancer include surgery, radiotherapy, chemotherapy, and physical therapy. The survival rate of non-small-cell lung cancer patients is less than 1% [3, 4].
The proposals that cancer might be preventable, and that food and nutrition might influence the risk of cancer, were first made in the 19th and 20th centuries. Throughout recorded history, wise choices of food and drink, and of habitual behavior, have been recommended as a protective measure against cancer [5]. Researchers are still unsure about the role of diet in lung cancer. Bright yellow-orange beta-carotene is one of a number of carotenoids thought to have anticancer activity even greater than vitamin A. Other possible lung protectors are foods high in bioactive compounds such as vitamin C and other antioxidants present in fruits and vegetables. These nutrients may protect lung linings but cannot totally prevent damage [6].
Among the different bioactive compounds, phenolic compounds from fruits and vegetables have gained much attention over the years because of their antioxidant activity that indirectly reflects their potential effects on human health [7, 8]. Some studies reported that phenolic compounds found in cocoa beans may present different properties such as antioxidant, anticarcinogenic, and antiradical activities [9–11]. Polyphenols are the main antioxidant-active constituents of cocoa. Flavanols and procyanidins have previously been identified as the active antioxidant agents of cocoa [12]. The polyphenol content of cocoa products depends on many factors, especially the cultivated variety and the postharvest handling that includes fermentation, drying, and roasting of the beans and nibs. There is evidence that fermentation and roasting of the beans tend to reduce their flavanol content [13, 14]. The antioxidant activity of the phenolic compounds is primarily due to their redox properties that allow them to act as reducing agents, hydrogen donors, and scavengers of reactive oxygen species (ROS) and metal ions [8, 15–17]. The emergence of natural extracts with antioxidant properties may help reduce the current dependence on synthetic drugs.
Potential mechanisms for cancer prevention of bioactive compounds include prevention of DNA adduct formation enhanced carcinogen elimination, inhibition of inflammatory processes, and a direct cytotoxic effect on tumour cells [18–20]. In line with efforts to balance the conservation of biodiversity and encourage the controlled exploitation of plant resources for economic gain, especially in biopharming, waste of valuable resources should be minimized [17]. The aim of this study was to evaluate and compare the antioxidant activity of cocoa beans classified as slate, roasted, and unroasted with well fermented beans submitted to the same processing and their cytotoxic effects on human lung carcinoma cell line (A549).
2. Methods
2.1. Samples and Extractions
Samples of cocoa beans, unroasted slates (US), roasted slates (RS), unroasted well fermented (UWF) cocoa, and roasted well fermented (RWF) cocoa were harvested and preprocessed (fermented and dried) in the cocoa producing region of Ilhéus (Bahia, Brazil). These samples were classified according to their fermentation status and donated by a company of the cocoa sector from the same region. At least three different lots of each cocoa class were mixed to form the samples used in this study. The roasting as well as the fine grinding of all beans was carried out in the State University of Feira de Santana (BA). All samples were kept at −5°C and sent to the Functional Foods and Biotechnology Laboratory of the Federal University of Rio de Janeiro State (UNIRIO), where all analyses were conducted.
2.2. Extraction of Samples
The samples of cocoa were extracted with 3 different solution extractors: methanol (I), methanol 50% (II), and methanol 50% : acetone 70% (1 : 1) (III). 1.25 g of sample was weighed and suspended in 10 mL of extracting solution for 1 hour under stirring, protected from light. The crude extracts were filtered and completed in 25 mL with distilled water. For cellular analysis, 1.25 g of each sample was weighed and suspended in saline solution (PBS) at 2% dimethyl sulfoxide (DMSO).
2.3. Total Phenolic Assay
Total phenolic content of the extracts was determined according to the Folin-Ciocalteu method as described by Singleton and Rossi [21] with minor modifications. Aliquots of 0.5 mL of the extracts were added to 2.5 mL of Folin-Ciocalteu reagent and 2.0 mL of 4% sodium carbonate solution and the mixture was allowed to rest for 2 hours in the dark. Measurements were performed at 750 nm in triplicates, applying a Turner® 340 spectrophotometer. Gallic acid, in the concentration range of 0–100 mg/mL−1, was used to construct a calibration curve. The concentration of total phenolic compounds in the extract was expressed as gallic acid equivalents, which reflect the phenolic content as the amount of gallic acid in mg/100 g dry weight of the samples.
2.4. Antioxidant Activity Analyses
2.4.1. DPPH Assay
Aliquots of 0.5 mL of the extracts were mixed with 2.5 mL DPPH methanolic solution (0.06 mM) and allowed to react for 1 hour, in the dark. Measurements were performed at 515 nm applying a Turner 340 spectrophotometer. The analysis was performed in triplicates; the decline in the DPPH radical absorbance concentration caused by the extracts was compared to a trolox standard. The results were expressed as μmol trolox equivalents/g dry basis. [22].
2.4.2. Trolox Equivalent Antioxidant Capacity (ABTS/TEAC)
The TEAC•+ cation was prepared by mixing a TEAC stock solution (7 mM in water) with 2.45 mM potassium persulfate. This mixture was allowed to stand for 16 hours at room temperature until the reaction was completed and the absorbance was stable.
The antioxidant capacity assay was carried out by the improved ABTS/TEAC method as described by Rufino et al. [23]. TEAC solution (2.5 mL) was added to extracts or commercial antioxidant (trolox) and mixed thoroughly. Absorbance was recorded at 734 nm during 6 min. Aliquots of 5, 10, and 20 μL of the extracts were tested and their volume was completed to 0.5 mL with water. Results were expressed as μmol trolox/g dry basis.
2.4.3. Ferric Reducing Ability (FRAP)
The extracts were measured for antioxidant activity by FRAP according to Rufino et al. [24]. Aliquots of 2.7 mL of TPTZ reagent (ferric 2,4,6-tripyridyl-s-triazine) were mixed with 0.5 mL of sample extract (aliquots 5, 10, and 20 μL). After 30 min at 37°C temperature, the absorbance was read at 595 nm. The antioxidant capacity (FRAP) was expressed as Fe3+ equivalents (μmol Fe3+/g dry basis).
2.4.4. Cell Culture and Treatment Protocol
Cell lines were obtained from the Rio de Janeiro Cell Bank which certified their identity and quality (INMETRO, Rio de Janeiro, RJ, Brazil). Human lung carcinoma cell line (A549) was plated in 25 cm2 tissue culture flasks (5.0 × 106 cells/flask) and maintained routinely in Dulbecco's Modified Eagle's Medium-high glucose (DMEM) supplemented with 10% Fetal Bovine Serum (FBS) and 1% Penicillin (PS), pH 7.4, under 5% CO2 atmosphere. Stock flasks were grown to 70% confluence and subcultured routinely. Medium renewal was done 3 times weekly. For each experiment, cells were seeded at 3.5 × 105 cells/cm2 and 2 × 104 cells/cm2 densities in 6-well plates and 96-well plates for cell cycle and cell proliferation analyses, respectively. After 24 h, medium was removed and cells were treated with increasing concentrations of cocoa nibs extract (100 to 10000 μg/mL) dissolved in DMEM. The controls, DMEM and DMEM + 2% DMSO, were included on each plate. The cells were then incubated for 48 hours.
2.4.5. Cell Viability
Cell viability was monitored by MTT assay (Amresco, Solon, OH). MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) is a pale yellow substrate that is reduced by living cells to yield a dark blue formazan product. This requires active mitochondria, and even recently dead cells do not reduce significant amounts of MTT. Exponentially growing cells were adjusted to 2.0 × 104/cm2 with DMEM, plated in 96-well plates (Corning, Tewksbury, MA) at 200 μL/well and incubated for 24 h according to the routine procedure. The cells were then incubated with cocoa nibs unroasted slates (US), roasted slates (RS), unroasted well fermented (UWF) cocoa, and roasted well fermented (RWF) cocoa (5–10 mg/mL) for 48 h (6 wells for each sample). Each well was also incubated with MTT (10 μL/well; 5 g/mL) for 4 h. After 85 μL/well the liquid was removed and 50 μL/well sodium dodecyl sulfate was added to dissolve the solid residue. Finally, the absorbance was measured using a microplate reader (POLARIS, CELER®) at 570 nm. The cell proliferation inhibition rate (CPIR) was calculated using the following formula: CPIR = (1 − average value of experimental group/average value of control group) × 100%.
2.4.6. Cell Cycle Analysis
Cells were rinsed briefly with calcium and magnesium-free phosphate-buffered saline and detached with trypsin at room temperature. After centrifugation, the cells were washed twice with phosphate-buffered saline; cells were resuspended in 500 μL of ice-cold Vindelov solution [25] containing 0.1% Triton X-100, 0.1% citrate buffer, 0.1 mg/mL RNase, and 50 mg/mL propidium iodide (Sigma Chemical Co., St. Louis, MO). After 15 min of incubation, cell suspension was analysed for DNA content by flow cytometry using a FACSCalibur flow cytometer (Becton Dickinson, Mountain View, CA). The relative proportions of cells with DNA content indicative of apoptosis (<2n), G0/G1 diploid (2n), S (phase >2n but <4n), and G2/M phase (4n) were obtained and analyzed using the CellQuest WinMDI 2.9. The percentage of cell population at a particular phase was estimated with FlowJo software. Cell dissociation procedure does not affect fluorescence under the experimental conditions that were used in this study or in any other studies of which we are aware. Nuclei of viable cells were gated according to FL-2W × FL2-A relation.
2.4.7. Apoptosis Assay
To measure the rate of apoptosis, the cells were subjected to staining with Annexin V conjugated to FITC (BD Pharmingen, San Diego, CA). The nonadherent cells were collected, and adherent cells were quickly washed with buffered saline solution (BSS) calcium/magnesium-free and were detached with trypsin/EDTA 0.125% (Sigma chemical Co., St. Louis, USA) at room temperature. Subsequently, apoptotic and necrotic cells were stained with Annexin VFITC/propidium iodide (PI) (BD Pharmingen, New Jersey, USA) according to the manufacturer's instructions, quantified by flow cytometer (FACSCalibur, BD Bioscience, New Jersey, USA), and analyzed using two specific programs, Cell Quest and FlowJo software.
2.5. Statistical Analysis
Results are presented as mean with the corresponding standard deviation of 3 independent experiments done in triplicates (n = 9). Data were analysed with the statistical software GraphPad Prism (version 5.04, GraphPad Software, San Diego, CA) and Statistica (version 7.0, StatSoft Inc., Tulsa, OK). One-way analysis of variance (ANOVA) test with the posttest of Tukey at a confidence level of 95% was used to test cell viability, cell cycle, and apoptosis.
3. Results and Discussion
3.1. Phenolic Compounds and Antioxidant Activity of Cocoa Beans Samples
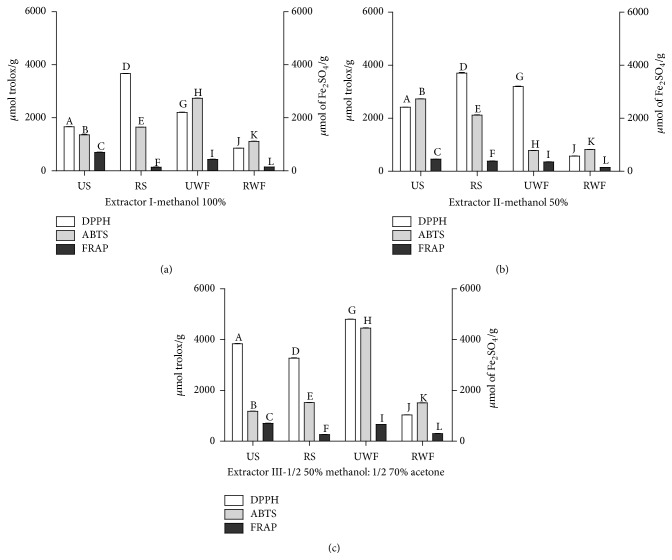
The method that yielded higher phenolic extracts was the one using acetone, in all samples tested, as can be observed in Figure 1. Dreosti [26] reported that 60% of the total phenolics in raw cocoa beans are flavanol monomers (epicatechin and catechin) and procyanidin oligomers (dimer to decamer) [27]. Benayad et al. [28], Cheng et al. [29], and Boulekbache-Makhlouf et al. [30] have shown that the use of acetone, when compared to the use of other polar organic compounds, potentiated the extraction of flavonoids and flavonols from different plant materials. In addition, extraction of procyanidins from cocoa with acetone solutions has been successfully accomplished at least since 1999.
Figure 1.
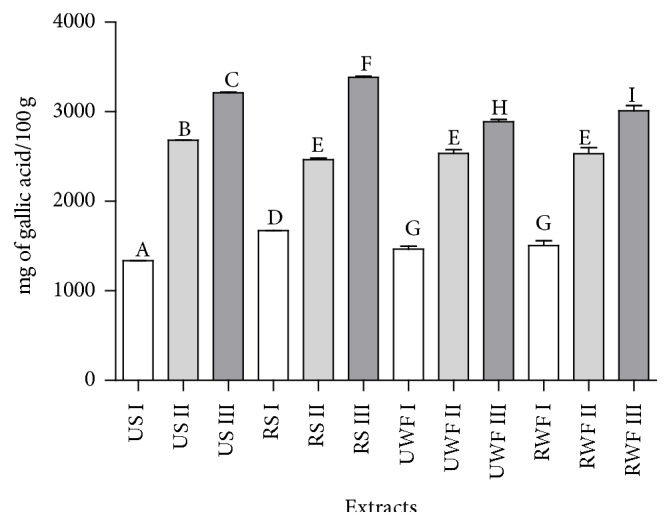
Total phenolic compounds of cocoa nibs unroasted slates (US), roasted slates (RS), unroasted well fermented (UWF) cocoa, and roasted well fermented (RWF) cocoa. Extracting solutions: I, methanol; II, methanol 50%; III, 1/2 50% methanol : 1/2 70% acetone. Means with different letters differ significantly (p < 0.05, Tukey's test).
The present work found amounts of phenolic substances (Table 1) compatible with the results displayed by Kadow et al. [31] for both raw and “fermentation-like” treated samples. Unprocessed cocoa beans usually present a high phenolic content of about 12–18% (dry weight) [32]. The processing of raw cocoa includes a number of stages and each stage in the processing alters cocoa's chemistry and composition [11]. When extractor III was considered, there were significant differences among all samples tested and slates showed higher phenolic content when compared to well fermented samples. Generally, it is expected that well fermented beans show lower phenolic contents, because low mass phenols are related to astringency and anthocyanins are related to the purple color of unfermented beans, both considered undesirable characteristics in chocolate [33, 34].
Table 1.
Effect of extracts of cocoa, cocoa nibs roasted slates (RS), and unroasted well fermented (UWF) cocoa (5–10 mg/mL) on cell cycle progression in human lung cancer cell line after 48 hours.
| Cell cycle phase | CT | 5 mg/mL | 10 mg/mL | |
|---|---|---|---|---|
| RS | sub-G1 | 6.65 ± 1.00 | 55.65 ± 0.49 | 77.10 ± 2.26 |
| G0/G1 | 75.75 ± 4.45 | 30.65 ± 0.92∗ | 20.63 ± 0.90∗ | |
| S | 5.40 ± 0.77 | 4.89 ± 0.16 | 0.74 ± 0.40∗ | |
| G2/M | 7.68 ± 1.77 | 6.73 ± 0.38 | 0.70 ± 0.64∗ | |
|
| ||||
| UWF | sub-G1 | 1.37 ± 0.29 | 13.55 ± 2.62∗ | 50.28 ± 7.09∗∗ |
| G0/G1 | 75.80 ± 6.02 | 25.65 ± 7.57∗ | 21.78 ± 5.35∗ | |
| S | 5.61 ± 1.17 | 2.64 ± 1.58∗ | 2.20 ± 1.01∗ | |
| G2/M | 11.58 ± 3.54 | 33.95 ± 3.04∗∗ | 16.08 ± 4.01∗ | |
Results are expressed as the percentage of total cells. The data represent mean ± SD values of triplicate experiments. Tukey's test; ∗ p < 0.05; ∗∗ p < 0.01.
Figures 2(a)–2(c) clearly show that the overall higher values were obtained for the DPPH method, followed by TEAC and FRAP, respectively. The slate samples showed higher antioxidant activity compared to well fermented beans or nibs. All these findings were in agreement with previous results as it was expected to obtain higher antioxidant activity in extracts with higher concentrations of phenolic compounds [14, 35–37].
Figure 2.
Antioxidant activity of cocoa nibs unroasted slates (US), roasted slates (RS), unroasted well fermented (UWF) cocoa, and roasted well fermented (RWF) cocoa for DPPH, TEAC, and FRAP assays by different extracting solutions (I–III). Means with different letters differ significantly (p < 0.05, Tukey's test).
The roasted slate (RS) samples showed a decrease in antioxidant activity after fermentation. Roasting is considered one of the processing steps of the cocoa nibs that leads to the loss of phenolic compounds and should cause a decrease in the antioxidant activity, as may be seen when comparing UWF and RWF samples (Figure 2). It is possible that, during roasting, while phenolic compounds are degraded, other antioxidant potential compounds are formed through the Maillard reaction, especially reductones and melanoidins [38, 39]. The reason why this formation may have been significant in slates but not in well fermented beans is probably related to the different compositions of these two samples before the roasting process.
FRAP assay showed a decrease in antioxidant activity after roasting. According to Apak et al. [40], FRAP methodology is not capable of measuring the antioxidant activity of compounds in complex matrix, since it takes more time to perform the analysis of antioxidant function, and not all antioxidants have the specific ability to reduce iron [41].
Recovery of antioxidant compounds from plant materials is typically accomplished through different extraction techniques, taking into account their chemistry and uneven distribution in the plant matrix. These compounds tend to present different polarities as well as other variable characteristics. Thus, the solubility in a particular solvent is a unique feature of the phytochemicals to be taken into account. Methanol and solutions of 50% methanol in water are commonly applied solvents that efficiently extract phenolic compounds. Methanol and acetone are also suitable solvents for anthocyanin extraction from various raw materials [42–44], and acetone-water mixtures have been suggested to grant better extraction results of procyanidins and phenols when compared with other extractors [45].
It is now recognized that diet and nutrients play an import role in cancer development and progress, with many dietary components found to be associated with cancer risk. However, almost all the clinical intervention trials with isolated nutrients, such as vitamin A, vitamin E, vitamin C, and phenolic compounds supplements, failed to demonstrate their protective effects against cancer. Due to the complexity of cocoa matrix, it is very difficult to characterize all components and even say which major component is responsible for the cytotoxic effect, due to the synergistic and antagonistic effects.
3.2. Cells Results
3.2.1. Effect of Cocoa Extracts on Cell Viability
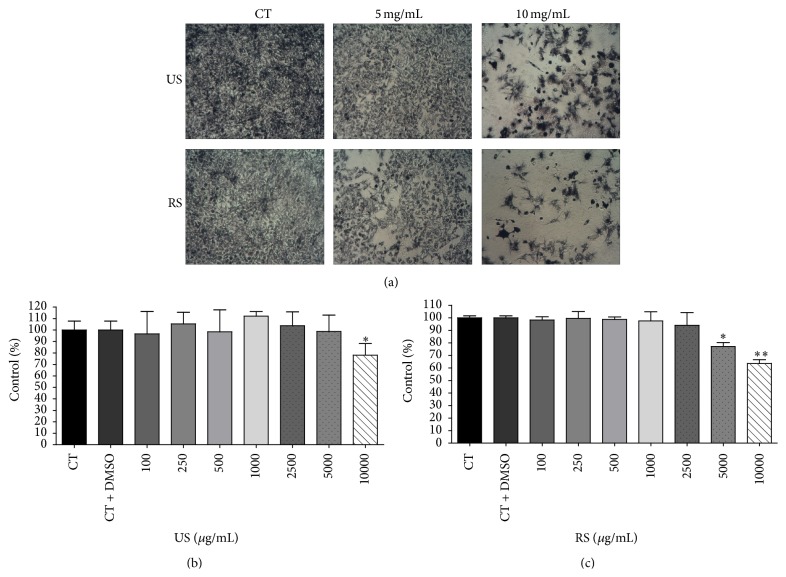
The slate nibs were the supplement which caused the largest decrease in viability compared to control (34.45%, Figure 3), while cells exposed to US had the highest percentage of viability, 78.07%, at a concentration of 10 mg/mL (Figure 3(b)). However, surprisingly, crisp sample obtained higher potential reduction in cell viability (Figure 3(c)), with cell viability of 77.15% (5000 μg/mL) and 63.55% (10.000 μg/mL).
Figure 3.
The effect of unroasted slates (US) and roasted slates (RS) nibs extract in cell control (a). Cocoa nibs US (b) and cocoa nibs RS (c), after forty-eight hours on viability A549 cells after exposure using MTT assays. The results are expressed as mean ± standard error and significant differences between cells treated with US and RS nibs extract (100–10000 μg/mL) were compared using Tukey's test (∗ p < 0.05; ∗∗ p < 0.01).
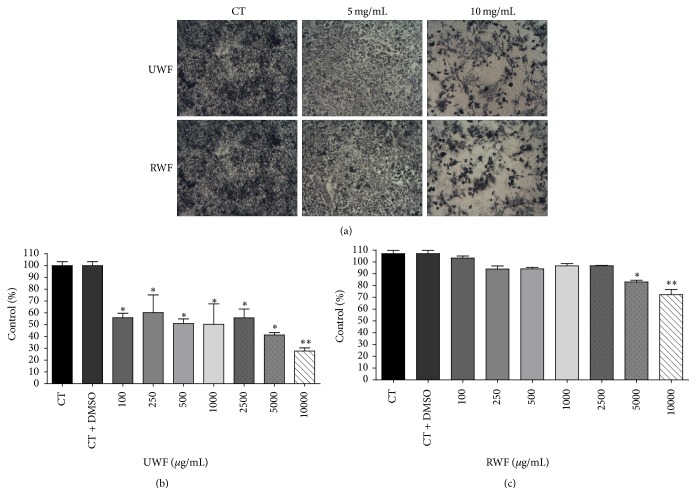
Well fermented cocoa bean extract decreased the number of viable A549 cells within 48 hours. In UWF sample, cell viability decreased from the concentration of 100 μg/mL by 45% compared with the control group (p < 0.05) (Figures 4(a) and 4(b)). The concentrations that caused the largest decrease in cell viability were 5000 and 10000 μg/mL, reduced by 58.77% and 72.35%, respectively (Figure 3(b)). For RWF sample, the reduction was smaller (Figure 3(c)), with effect only at concentrations of 5000 (83.07% viable cells) and 10000 μg/mL (72.20% viable cells).
Figure 4.
The effect of unroasted well fermented (UWF) nibs and roasted well fermented (RWF) nibs extracts in cell control (a). UWF cocoa nibs (b) and RWF cocoa nibs (c), after forty-eight hours on viability A549 cells after exposure using MTT assays. The experiment is expressed as mean ± standard error and significant differences between cells treated with UWF and RWF nibs extract (100–10000 μg/mL) were compared using Tukey's test (∗ p < 0.05; ∗∗ p < 0.01).
Cell culture studies constitute a useful tool to elucidate the molecular mechanisms of action of cocoa extracts and their polyphenolic compounds in different cancer cell lines. It has been shown that cocoa components induced a time-dependent regulation of survival/proliferation pathways in HepG2 liver cells [46]. Moreover, a cocoa procyanidin fraction inhibited TPA-induced neoplastic transformation of JB6P+ mouse epidermal cells, COX-2 expression, and phosphorylation of MEK and p90 ribosomal s6 kinase and attenuated activator protein-1 (AP-1) and NF-κB stimulations [47].
The physiological impact of polyphenols depends on their absorption. However, it is important to bear in mind that the most common polyphenols in diet are not necessarily the most bioavailable, since their structure plays an important role. Most native polyphenols in foods are in glycoside form (flavonols, flavones, flavanones, isoflavones, and anthocyanins), together with the less frequent oligomers (proanthocyanidins), which cannot be absorbed in the intestinal mucosa [48]. Through this, we can observe that the sample that most reduced the viability of human lung carcinoma cells was again the sample which had the parent compounds of the modified crude cocoa, such as well fermented sample, and also the sample which has not been roasted, since many bioactive compounds are lost during this processing.
3.2.2. Effect of Cocoa Extracts on Cell Cycle Progression
Uncontrolled cell proliferation is a characteristic of cancer [49], and extracts of cocoa beans have been shown to inhibit the proliferation of A549 cells. Previous trials of antioxidant activity and MTT have determined which samples have the greatest potential for use in the analysis of cell cycle and apoptosis. To probe inhibition of cell growth mediated by RS and UWF extracts, we examined the cell cycle by flow cytometry. The effects of the RS and UWF extracts on cell cycle progression in A549 cells are shown in Table 1. After 48 h of growth, the population control in the sub-G1 G0/G1 phase reached values between 6.65 and 1.37% and between 75.75% and 75.80%, respectively. Meanwhile, cells treated in the sub-G1 population increased those treated in G0/G1 phase decreased in a dose-dependent manner in both treatments. The loss of the ability to regulate the cell cycle is characteristic of cancer cells and results in uncontrollable proliferation. The cell progression through the first gap phase of the cell cycle (G1) is a step which is frequently disordered in cancer [50].
Treatments with RS resulted in the highest percentage of cells in sub-G1 (77.01%, 10 mg/mL) and in G0/G1 (30.65%, 5 mg/mL) phases. As the G0/G1 phase population increased the G2/M phase population of cells decreased, whereas the population of phase S cells showed no difference within 48 h. Treatment with UWF cocoa at a concentration of 10 mg/mL showed the highest value for sub-G1 (50.27%) reduction within phases G0/G1 and S and an increase in the G2/M phase. Treatment with 5 mg/mL of UWF cocoa showed the smallest number of cells in sub-G1 and the G0/G1 phases and therefore a greater cell population in the G2/M phase (33.95%).
Cocoa-derived pentameric procyanidin (pentamer) caused a G0/G1 cell cycle arrest in human breast cancer MDA MB-231, MDA MB-436, MDA MB-468, SKBR-3, and MCF-7 cells and in benzo(a)pyrene-immortalized 184A1N4 and 184B5 cells, whereas normal human mammary epithelial cells in primary culture and spontaneously immortalized MCF-10A cells were resistant [51]. Similarly, procyanidin-enriched extracts from cocoa caused growth inhibition with blockade of the cell cycle at G2/M phase in human colonic Caco-2 cells [52], and EC induced S phase arrest in the cell cycle progression in LoVo colon cancer cells [53].
Our results indicate that cocoa nibs extracts were able to modify cell cycle. The deregulation in cell cycle control is a fundamental aspect in the development of cancer. Faults in the cell cycle regulation process can cause a greater proliferation of cancer cells. However, the reversal of this process leads to a delay in growth and induces cell death [54].
3.2.3. Apoptosis
We examined the effect of RS and UWF cocoa on apoptotic death in A549 cells. Table 2 shows the percentages of viable, early apoptotic, late apoptotic, and nonapoptotic cells treated with 5 mg/mL and 10 mg/mL RS and UWF cocoa. Figure 4 shows the influence of the extracts of cocoa nibs on the rate of apoptosis. Cells treated with 5 mg/mL and 10 mg/mL RS and UWF cocoa for 48 hours resulted in a significant increase in the percentage of apoptotic cells compared with untreated cells (control). The concentrations used in this cellular model promoted a change in cell cycle and induction of apoptosis by lower doses than isolated compounds or cocoa products used in studies with human models. [55–57]. This has been demonstrated in a study by Ottaviani et al. [57], where adult males were given 1.5 mg/kg of a concentrated cocoa solution, and two hours after ingestion it was noted that there were many metabolites of flavonols in cocoa in the blood of the volunteers.
Table 2.
Effect of extracts of cocoa nibs roasted slates and unroasted well fermented cocoa (5–10 mg/mL) on programmed cell death in human lung cancer cell line after 48 hours.
| Stages of cell death | CT | RS (mg/mL) | UWF (mg/mL) | ||
|---|---|---|---|---|---|
| 5 | 10 | 5 | 10 | ||
| Viable cells (Annexin V−/PI−) |
95.80 ± 0.57 | 89.10 ± 2.26 | 86.73 ± 1.96 | 91.37 ± 1.33 | 72.03 ± 4.67∗ |
| Early apoptosis (Annexin V+/PI−) |
2.16 ± 1.03 | 1.80 ± 0.83 | 8.22 ± 2.48∗∗ | 3.87 ± 1.22 | 4.85 ± 2.05 |
| Late apoptosis (Annexin V+/PI+) |
0.90 ± 0.25 | 2.56 ± 1.30 | 4.92 ± 0.95∗ | 2.23 ± 0.67 | 8.13 ± 0.06∗∗ |
| Nonapoptotic cells (Annexin V−/PI+) |
1.12 ± 0.69 | 6.54 ± 0.13∗ | 0.14 ± 0.69 | 2.52 ± 0.68 | 14.93 ± 0.42∗∗ |
Results are expressed as percentage of total cells. The experiment is expressed as mean ± standard deviation; significant differences between untreated cells (CT) and cells treated with lycopene (5–10 μM) were compared by one-way ANOVA with the posttest of Tukey (∗ p < 0.05; ∗∗ p < 0.01).
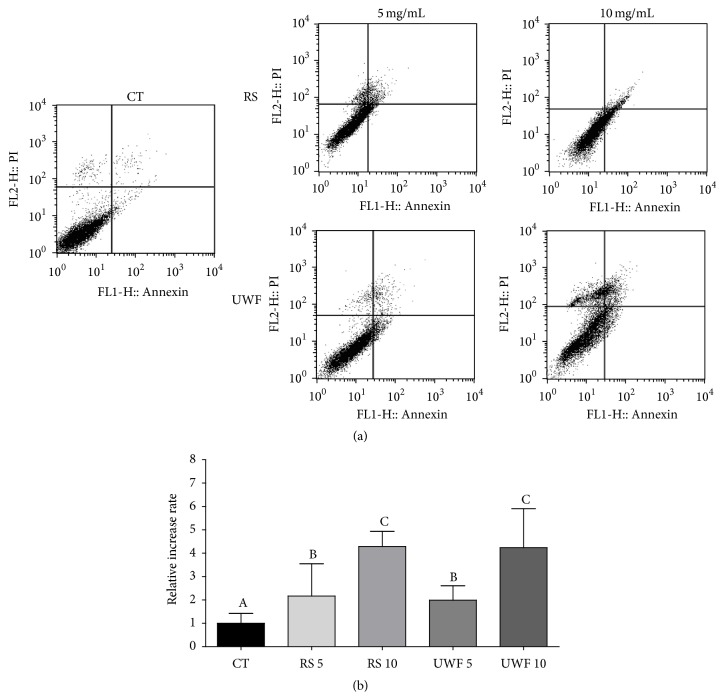
Increase of early apoptosis was observed in A549 cells treated with 10 mg/mL RS, whereas increase in late apoptosis was observed in cells treated with 10 mg/mL UWF cocoa for 48 h. A549 cells incubated with 5 mg/mL and 10 mg/mL RS UQG for 48 hours showed a decrease in the population of viable cells and an increase of up to 4.3 times in the percentage of apoptotic cells compared with control, in a dose-dependent manner (Table 2 and Figure 5).
Figure 5.
The effect of unroasted well fermented cocoa and roasted slates on the process of programmed death in A549 cells after treatment for 48 h. (a) Flow cytometry analysis of UWF cocoa and RS according to the exposure time and concentration of the compounds. (b) Quantitative effects of UWF cocoa and RS at 5 mg/mL and 10 mg/mL on A549 cells after exposure for 48 h. The results are expressed as mean ± SD, with significant differences between untreated cells (CT) and cells treated with UWF cocoa and RS (5–10 mg/mL) compared by 1-way ANOVA followed by Tukey's multiple comparison post hoc test. ∗ p < 0.05. ∗∗ p < 0.01.
Apoptosis is characterized by a series of distinct changes in cell morphology, loss of cell attachment, cytoplasmic contraction, DNA fragmentation, and other biochemical changes, including the activation of caspases through extrinsic and/or intrinsic mitochondrial pathways [58]. Increased resistance to apoptosis is a hallmark of many tumor cells. The functional inhibition of specific antiapoptotic factors may provide a rational basis for the development of novel therapeutic strategies. Therefore, apoptotic deficiency is considered to be a major cause of therapeutic resistance of tumors, since many chemotherapeutic and radiotherapeutic agents act through the induction of apoptosis [59]. However, the apoptotic effect of the active ingredient of cocoa bean samples on A549 cells has not yet been studied in detail.
4. Conclusion
In conclusion, cocoa extract presented itself as a potent antioxidant agent, with antioxidant capability influenced by the processing method and extraction. Slate cocoa beans extract inhibited cell proliferation, arrested cell cycle in different phases, and increased apoptosis in human lung carcinoma cells, in a time-dependent and dose-dependent manner. Our study has far reaching health relevance as cocoa could be projected as functional foods which, in addition to providing nutrition, would provide preventive therapeutic value against the development of cancer.
Acknowledgments
The authors would like to thank Dr. Osnat Hakimi for her careful reading of the final text and valuable suggestions.
Competing Interests
The authors declare that they have no competing interests.
References
- 1.WHO. World Health Statistics 2013. World Health Organization; 2013. [Google Scholar]
- 2.Al Zeyadi M., Dimova I., Ranchich V., et al. Whole genome microarray analysis in non-small cell lung cancer. Biotechnology and Biotechnological Equipment. 2015;29(1):111–118. doi: 10.1080/13102818.2014.989179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.INCA. Estimativa 2014: Incidência de Câncer no Brasil. Ministério da Saúde; 2014. [Google Scholar]
- 4.Ni M., Shi X., Qu Z., Jiang H., Chen Z., Hu J. Epithelial mesenchymal transition of non–small–cell lung cancer cells A549 induced by SPHK1. Asian Pacific Journal of Tropical Medicine. 2015;8(2):142–146. doi: 10.1016/S1995-7645(14)60305-9. [DOI] [PubMed] [Google Scholar]
- 5.World Cancer Research Fund and American Institute for Cancer Research. Food, Nutrition, Physical Activity, and the Prevention of Cancer: A Global Perspective. 2007. [Google Scholar]
- 6.Stadler K. M. Extension Specialist, Human Nutrition and Foods. Virginia Tech; 2009. The diet and cancer connection. [Google Scholar]
- 7.Hazirah A. R., Zainal B., Abdah M. A. Total phenolic content, antioxidant and cytotoxic activity of cocoa (Theobroma cacao L.) polyphenols extracts on cancer cell lines. Malaysian Journal of Nutrition. 2013;19(2):223–232. [Google Scholar]
- 8.Turati F., Rossi M., Pelucchi C., Levi F., La Vecchia C. Fruit and vegetables and cancer risk: a review of southern European studies. British Journal of Nutrition. 2015;113(2, supplement):S102–S110. doi: 10.1017/s0007114515000148. [DOI] [PubMed] [Google Scholar]
- 9.Sanbongi C., Osakabe N., Natsume M., Takizawa T., Gomi S., Osawa T. Antioxidative Polyphenols Isolated from Theobroma cacao . Journal of Agricultural and Food Chemistry. 1998;46(2):454–457. doi: 10.1021/jf970575o. [DOI] [PubMed] [Google Scholar]
- 10.Ren W., Qiao Z., Wang H., Zhu L., Zhang L. Flavonoids: promising anticancer agents. Medicinal Research Reviews. 2003;23(4):519–534. doi: 10.1002/med.10033. [DOI] [PubMed] [Google Scholar]
- 11.Wollgast J., Anklam E. Review on polyphenols in Theobroma cacao: changes in composition during the manufacture of chocolate and methodology for identification and quantification. Food Research International. 2000;33(6):423–447. doi: 10.1016/s0963-9969(00)00068-5. [DOI] [Google Scholar]
- 12.Arlorio M., Locatelli M., Travaglia F., et al. Roasting impact on the contents of clovamide (N-caffeoyl-L-DOPA) and the antioxidant activity of cocoa beans (Theobroma cacao L.) Food Chemistry. 2008;106(3):967–975. doi: 10.1016/j.foodchem.2007.07.009. [DOI] [Google Scholar]
- 13.Miller K. B., Hurst W. J., Payne M. J., et al. Impact of alkalization on the antioxidant and flavanol content of commercial cocoa powders. Journal of Agricultural and Food Chemistry. 2008;56(18):8527–8533. doi: 10.1021/jf801670p. [DOI] [PubMed] [Google Scholar]
- 14.Payne M. J., Hurst W. J., Miller K. B., Rank C., Stuart D. A. Impact of fermentation, drying, roasting, and dutch processing on epicatechin and catechin content of cacao beans and cocoa ingredients. Journal of Agricultural and Food Chemistry. 2010;58(19):10518–10527. doi: 10.1021/jf102391q. [DOI] [PubMed] [Google Scholar]
- 15.Rice-Evans C. A., Miller N. J., Paganga G. Antioxidant properties of phenolic compounds. Trends in Plant Science. 1997;2(4):152–159. doi: 10.1016/S1360-1385(97)01018-2. [DOI] [Google Scholar]
- 16.Crozier A., Del Rio D., Clifford M. N. Bioavailability of dietary flavonoids and phenolic compounds. Molecular Aspects of Medicine. 2010;31(6):446–467. doi: 10.1016/j.mam.2010.09.007. [DOI] [PubMed] [Google Scholar]
- 17.Osman H., Nasarudin R., Lee S. L. Extracts of cocoa (Theobroma cacao L.) leaves and their antioxidation potential. Food Chemistry. 2004;86(1):41–46. doi: 10.1016/j.foodchem.2003.08.026. [DOI] [Google Scholar]
- 18.Talalay P. Chemoprotection against cancer by induction of Phase 2 enzymes. BioFactors. 2000;12(1–4):5–11. doi: 10.1002/biof.5520120102. [DOI] [PubMed] [Google Scholar]
- 19.Surh Y.-J. Cancer chemoprevention with dietary phytochemicals. Nature Reviews Cancer. 2003;3(10):768–780. doi: 10.1038/nrc1189. [DOI] [PubMed] [Google Scholar]
- 20.Martin K. R. Targeting apoptosis with dietary bioactive agents. Experimental Biology and Medicine. 2006;231(2):117–129. doi: 10.1177/153537020623100201. [DOI] [PubMed] [Google Scholar]
- 21.Singleton V. L., Rossi J. A. J. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. American Journal of Enology and Viticulture. 1965;16(3):144–158. [Google Scholar]
- 22.Brand-Williams W., Cuvelier M. E., Berset C. Use of a free radical method to evaluate antioxidant activity. LWT—Food Science and Technology. 1995;28(1):25–30. doi: 10.1016/s0023-6438(95)80008-5. [DOI] [Google Scholar]
- 23.Rufino M. D. S. M., Alves R. E., De Brito E. S., et al. Metodologia Científica: Determinação da Atividade Antioxidante Total em Frutas pela Captura do Radical Livre. Comunicado Técnico. 2007;(127)
- 24.Rufino M. D. S. M., Alves R. E., De Brito E. S., De Morais S. M., Sampaio C. D. G., Saura-Calixto F. D. Comunicado Técnico. 125. Embrapa Agroindústria Tropical; 2006. Metodologia Científica: Determinação da Atividade Antioxidante Total em Frutas pelo Método de Redução do Ferro (FRAP) [Google Scholar]
- 25.Vindelov L. L. Flow microfluorometric analysis of nuclear DNA in cells from solid tumors and cell suspensions. A new method for rapid isolation and staining of nuclei. Virchows Archiv Abteilung B: Cell Pathology. 1977;24(3):227–242. [PubMed] [Google Scholar]
- 26.Dreosti I. E. Antioxidant polyphenols in tea, cocoa, and wine. Nutrition. 2000;16(7-8):692–694. doi: 10.1016/S0899-9007(00)00304-X. [DOI] [PubMed] [Google Scholar]
- 27.Othman A., Ismail A., Abdul Ghani N., Adenan I. Antioxidant capacity and phenolic content of cocoa beans. Food Chemistry. 2007;100(4):1523–1530. doi: 10.1016/j.foodchem.2005.12.021. [DOI] [Google Scholar]
- 28.Benayad Z., Martinez-Villaluenga C., Frias J., Gomez-Cordoves C., Es-Safi N. E. Phenolic composition, antioxidant and anti-inflammatory activities of extracts from Moroccan Opuntia ficus-indica flowers obtained by different extraction methods. Industrial Crops and Products. 2014;62:412–420. doi: 10.1016/j.indcrop.2014.08.046. [DOI] [Google Scholar]
- 29.Cheng V. J., Bekhit A. E.-D. A., McConnell M., Mros S., Zhao J. Effect of extraction solvent, waste fraction and grape variety on the antimicrobial and antioxidant activities of extracts from wine residue from cool climate. Food Chemistry. 2012;134(1):474–482. doi: 10.1016/j.foodchem.2012.02.103. [DOI] [Google Scholar]
- 30.Boulekbache-Makhlouf L., Medouni L., Medouni-Adrar S., Arkoub L., Madani K. Effect of solvents extraction on phenolic content and antioxidant activity of the byproduct of eggplant. Industrial Crops and Products. 2013;49:668–674. doi: 10.1016/j.indcrop.2013.06.009. [DOI] [Google Scholar]
- 31.Kadow D., Niemenak N., Rohn S., Lieberei R. Fermentation-like incubation of cocoa seeds (Theobroma cacao L.)—reconstruction and guidance of the fermentation process. LWT—Food Science and Technology. 2015;62(1):357–361. doi: 10.1016/j.lwt.2015.01.015. [DOI] [Google Scholar]
- 32.Elwers S., Zambrano A., Rohsius C., Lieberei R. Differences between the content of phenolic compounds in Criollo, Forastero and Trinitario cocoa seed (Theobroma cacao L.) European Food Research and Technology. 2009;229(6):937–948. doi: 10.1007/s00217-009-1132-y. [DOI] [Google Scholar]
- 33.Araujo Q. R., Fernandes C. A. F., Ribeiro D. O., et al. Cocoa quality index—a proposal. Food Control. 2014;46:49–54. doi: 10.1016/j.foodcont.2014.05.003. [DOI] [Google Scholar]
- 34.Krähmer A., Engel A., Kadow D., et al. Fast and neat— determination of biochemical quality parameters in cocoa using near infrared spectroscopy. Food Chemistry. 2015;181:152–159. doi: 10.1016/j.foodchem.2015.02.084. [DOI] [PubMed] [Google Scholar]
- 35.Afoakwa E. O., Ofosu-Ansah E., Takrama J. F., Budu A. S., Mensah-Brown H. Changes in chemical quality of cocoa butter during roasting of pulp pre-conditioned and fermented cocoa (Theobroma cacao) beans. International Food Research Journal. 2014;21(6):2221–2227. [Google Scholar]
- 36.Afoakwa E. O., Paterson A., Fowler M., Ryan A. Flavor formation and character in cocoa and chocolate: a critical review. Critical Reviews in Food Science and Nutrition. 2008;48(9):840–857. doi: 10.1080/10408390701719272. [DOI] [PubMed] [Google Scholar]
- 37.Suazo Y., Davidov-Pardo G., Arozarena I. Effect of fermentation and roasting on the phenolic concentration and antioxidant activity of cocoa from nicaragua. Journal of Food Quality. 2014;37(1):50–56. doi: 10.1111/jfq.12070. [DOI] [Google Scholar]
- 38.Silván J. M., van de Lagemaat J., Olano A., del Castillo M. D. Analysis and biological properties of amino acid derivates formed by Maillard reaction in foods. Journal of Pharmaceutical and Biomedical Analysis. 2006;41(5):1543–1551. doi: 10.1016/j.jpba.2006.04.004. [DOI] [PubMed] [Google Scholar]
- 39.Wang H.-Y., Qian H., Yao W.-R. Melanoidins produced by the Maillard reaction: structure and biological activity. Food Chemistry. 2011;128(3):573–584. doi: 10.1016/j.foodchem.2011.03.075. [DOI] [Google Scholar]
- 40.Apak R., Güçlü K., Özyürek M., Karademir S. E. Novel total antioxidant capacity index for dietary polyphenols and vitamins C and E, using their cupric ion reducing capability in the presence of neocuproine: CUPRAC method. Journal of Agricultural and Food Chemistry. 2004;52(26):7970–7981. doi: 10.1021/jf048741x. [DOI] [PubMed] [Google Scholar]
- 41.Oldoni T. L. C. Prospecção e identificação de compostos bioativos de subprodutos agroindustriais [Ph.D. thesis] Universidade de São Paulo; 2010. [Google Scholar]
- 42.de Almeida Melo E., Maciel M. I. S., de Lima V. L. A. G., do Nascimento R. J. Capacidade antioxidante de frutas. Revista Brasileira de Ciências Farmacêuticas. 2008;44(2) doi: 10.1590/S1516-93322008000200005. [DOI] [Google Scholar]
- 43.Skrede G., Wrolstad R. E., Durst R. W., Skrede G. Changes in Anthocyanins and Polyphenolics During Juice Processing of Highbush Blueberries (Vaccinium corymbosum L. J. Food Sci. 2000;65(2):357–364. [Google Scholar]
- 44.Vizzotto M., Pereira M. C. Amora-preta (Rubus sp.): otimização do processo de extração para determinação de compostos fenólicos antioxidantes. Revista Brasileira de Fruticultura. 2011;33(4):1209–1214. doi: 10.1590/s0100-29452011000400020. [DOI] [Google Scholar]
- 45.Yilmaz Y., Toledo R. T. Oxygen radical absorbance capacities of grape/wine industry byproducts and effect of solvent type on extraction of grape seed polyphenols. Journal of Food Composition and Analysis. 2006;19(1):41–48. doi: 10.1016/j.jfca.2004.10.009. [DOI] [Google Scholar]
- 46.Granado-Serrano A. B., Martín M. A., Haegeman G., Goya L., Bravo L., Ramos S. Epicatechin induces NF-κB, activator protein-1 (AP-1) and nuclear transcription factor erythroid 2p45-related factor-2 (Nrf2) via phosphatidylinositol-3-kinase/protein kinase B (PI3K/AKT) and extracellular regulated kinase (ERK) signalling in HepG2 cells. British Journal of Nutrition. 2010;103(2):168–179. doi: 10.1017/s0007114509991747. [DOI] [PubMed] [Google Scholar]
- 47.Kang N. J., Lee K. W., Lee D. E., et al. Cocoa procyanidins suppress transformation by inhibiting mitogen-activated protein kinase kinase. The Journal of Biological Chemistry. 2008;283(30):20664–20673. doi: 10.1074/jbc.M800263200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Valdés L., Cuervo A., Salazar N., Ruas-Madiedo P., Gueimonde M., González S. The relationship between phenolic compounds from diet and microbiota: impact on human health. Food Funct. 2015;6(8):2424–2439. doi: 10.1039/C5FO00322A. [DOI] [PubMed] [Google Scholar]
- 49.Soule B. P., Simone N. L., DeGraff W. G., Choudhuri R., Cook J. A., Mitchell J. B. Loratadine dysregulates cell cycle progression and enhances the effect of radiation in human tumor cell lines. Radiation Oncology. 2010;5(1, article 8) doi: 10.1186/1748-717x-5-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.da Costa Pereira Soares N., Teodoro A. J., Oliveira F. L., et al. Lycopene induce apoptosis in human prostate cells and alters the expression of Bax and Bcl-2 genes. LWT—Food Science and Technology. 2014;59(2):1290–1297. doi: 10.1016/j.lwt.2014.04.028. [DOI] [Google Scholar]
- 51.Ramljak D., Romanczyk L. J., Metheny-Barlow L. J., et al. Pentameric procyanidin from Theobroma cacao selectively inhibits growth of human breast cancer cells. Molecular Cancer Therapeutics. 2005;4(4):537–546. doi: 10.1158/1535-7163.mct-04-0286. [DOI] [PubMed] [Google Scholar]
- 52.Carnésecchi S., Schneider Y., Lazarus S. A., Coehlo D., Gossé F., Raul F. Flavanols and procyanidins of cocoa and chocolate inhibit growth and polyamine biosynthesis of human colonic cancer cells. Cancer Letters. 2002;175(2):147–155. doi: 10.1016/S0304-3835(01)00731-5. [DOI] [PubMed] [Google Scholar]
- 53.Tan X., Hu D., Li S., Han Y., Zhang Y., Zhou D. Differences of four catechins in cell cycle arrest and induction of apoptosis in LoVo cells. Cancer Letters. 2000;158(1):1–6. doi: 10.1016/S0304-3835(00)00445-6. [DOI] [PubMed] [Google Scholar]
- 54.Murad L. D., da Soares C. P., Brand C., Monteiro M. C., Teodoro A. J. Effects of caffeic and 5-caffeoylquinic acids on cell viability and cellular uptake in human colon adenocarcinoma cells. Nutrition and Cancer. 2015;67(3):532–542. doi: 10.1080/01635581.2015.1004736. [DOI] [PubMed] [Google Scholar]
- 55.Ottaviani J. I., Momma T. Y., Heiss C., Kwik-Uribe C., Schroeter H., Keen C. L. The stereochemical configuration of flavanols influences the level and metabolism of flavanols in humans and their biological activity in vivo. Free Radical Biology and Medicine. 2011;50(2):237–244. doi: 10.1016/j.freeradbiomed.2010.11.005. [DOI] [PubMed] [Google Scholar]
- 56.Neukam K., Stahl W., Tronnier H., Sies H., Heinrich U. Consumption of flavanol-rich cocoa acutely increases microcirculation in human skin. European Journal of Nutrition. 2007;46(1):53–56. doi: 10.1007/s00394-006-0627-6. [DOI] [PubMed] [Google Scholar]
- 57.Ottaviani J. I., Momma T. Y., Kuhnle G. K., Keen C. L., Schroeter H. Structurally related (−)-epicatechin metabolites in humans: assessment using de novo chemically synthesized authentic standards. Free Radical Biology and Medicine. 2012;52(8):1403–1412. doi: 10.1016/j.freeradbiomed.2011.12.010. [DOI] [PubMed] [Google Scholar]
- 58.Tuli H. S., Sandhu S. S., Sharma A. K., Gandhi P. Anti-angiogenic activity of the extracted fermentation broth of an entomopathogenic fungus, Cordyceps militaris 3936. International Journal of Pharmacy and Pharmaceutical Sciences. 2014;6(7):581–583. [Google Scholar]
- 59.Teodoro A. J., Oliveira F. L., Martins N. B., Maia G. D. A., Martucci R. B., Borojevic R. Effect of lycopene on cell viability and cell cycle progression in human cancer cell lines. Cancer Cell International. 2012;12(1, article 36) doi: 10.1186/1475-2867-12-36. [DOI] [PMC free article] [PubMed] [Google Scholar]