Abstract
IκB kinase (IKK), a key regulator of immune and inflammatory responses, is known as an effector kinase mediating activation of the transcription factor NF-κB. Whether IKK also participates in other signaling events is not known. Here we show that IKK serves as an essential component of a signaling pathway that involves activation of the Tpl2 kinase and its downstream targets, MEK1 and ERK. Inhibition of IKKβ in macrophages eliminates Tpl2 activation and ERK phosphorylation induced by lipopolysaccharide and tumor necrosis factor alpha. Using IKK-deficient murine fibroblasts, we further demonstrate that IKKβ, but not IKKα, is required for Tpl2 activation. Moreover, this novel function of IKKβ appears to involve phosphorylation and degradation of the Tpl2 inhibitor NF-κB1/p105. These findings suggest that IKKβ exerts its immune-regulatory functions by targeting different downstream signaling pathways.
Inflammation is an important innate immune mechanism against microbial infections (18). A key step in the development of an inflammatory response is the release of proinflammatory cytokines and other soluble factors by tissue macrophages. The surface of macrophages expresses various Toll-like receptors (TLR), which recognize a variety of microbial components, including the bacterial endotoxin lipopolysaccharide (LPS) (1, 2). Through binding to TLR4, LPS triggers cascades of intracellular signaling events, including those that lead to activation of the IκB kinase (IKK) and three classes of mitogen-activated protein kinases (MAPK): the extracellular signal-regulated kinase (ERK), the c-jun N-terminal kinase (JNK), and p38 (36). These effector kinases each target the activation of specific transcription factors, which in turn cooperate in the activation of a large array of proinflammatory genes, such as those encoding the cytokines tumor necrosis factor α (TNF-α) and interleukin-1β, the prostaglandin-generating enzyme cycloxygenase 2 (COX-2), and intracellular nitric oxide synthase. Binding of the inflammatory cytokines to their receptors on macrophages and other cell types can further amplify the innate immune response.
IKK is known as the effector kinase mediating activation of NF-κB, a family of transcription factors that regulates macrophage activation as well as many other parts of the immune response (20, 34). The NF-κB proteins are normally sequestered in the cytoplasm as inactive complexes by physical interaction with specific inhibitors, including IκBα and related proteins (3). Upon activation by cellular stimuli, IKK phosphorylates IκBs, targeting these inhibitors for ubiquitination and proteasomal degradation, which allows released NF-κB factors to move to the nucleus and exert their transactivation function (19). In mammals, the NF-κB family consists of five members, RelA, RelB, c-Rel, NF-κB1, and NF-κB2, which function as homo- and heterodimers (33). A characteristic of NF-κB1 and NF-κB2 is that they are translated as large precursor proteins, p105 and p100, which upon proteasome-mediated processing generate the mature NF-κB subunits p50 and p52, respectively (11, 33). In addition to serving as precursor proteins, p105 and p100 also function as IκB molecules (26, 29). Recent studies suggest that the inducible processing of p100 controls a noncanonical NF-κB signaling pathway involved in B-cell maturation and lymphoid organogenesis (31, 41). In contrast to the processing of p100, the processing of p105 is constitutive and occurs cotranslationally (24). Interestingly, a large proportion of p105 remains as the unprocessed form, which undergoes degradation in response to certain cellular signals (4, 13, 15, 16, 28). However, the physiological role of p105 degradation has not been well defined.
IKK was originally isolated as a holoenzyme complex composed of two catalytic subunits, IKKα and IKKβ, and one regulatory subunit, IKKγ. Emerging evidence suggests that the different IKK components may also function in different kinase complexes (7, 40) and exert distinct functions (12). IKKα plays an essential role in mediating p100 processing and, thus, the noncanonical pathway of NF-κB activation (31). On the other hand, IKKβ and IKKγ are required for the canonical NF-κB signaling, specified by rapid degradation of IκBα and nuclear translocation of RelA-containing NF-κB dimers (12). Genetic studies reveal that inactivation of IKKβ, but not that of IKKα, severely cripples the activation of NF-κB by proinflammatory stimuli (22, 23, 37). IKKβ has consistently been shown to function as a key regulator of inflammatory responses (6). It remains unknown, however, whether IKKβ acts solely through NF-κB or possesses additional targets.
Recent studies on MAPKs have identified an ERK signaling pathway that specifically responds to immune stimuli. A key regulatory component of this immunoresponsive pathway is a MAPK kinase kinase (MAP3K), Tpl2 (also named Cot) (8). Unlike the MAP3K Raf-1 that regulates the classical ERK pathway, Tpl2 does not respond to mitogens (39) but acts as a target of various immune stimuli, including the bacterial component LPS, the proinflammatory cytokine TNF-α, and the B-cell stimulator CD40 ligand (8-10, 39). Genetic evidence suggests that the Tpl2/ERK signaling pathway plays a pivotal role in regulating the production of proinflammatory mediators by macrophages and antibody isotype switching in B cells (8-10, 39). However, how this novel signaling pathway is regulated has not been elucidated. Here we provide biochemical and genetic evidence that IKKβ is a key upstream regulator of the Tpl2 signaling pathway. Using both pharmacological inhibitors and IKK-deficient cells, we demonstrate that IKKβ as well as its regulatory subunit IKKγ are required for Tpl2 activation. These findings suggest that IKKβ exerts its immune-regulatory function by targeting different downstream signaling pathways, including those leading to activation of NF-κB and MAPK.
MATERIALS AND METHODS
Mice.
Mice deficient in the nfκb1 gene (32) and their closely related wild-type control (B6 129PF2/J) were purchased from Jackson Laboratories. All experiments were done in accordance with Institutional Guidelines of the Pennsylvania State University College of Medicine.
Plasmid constructs, antibodies, and other reagents.
The retroviral expression vector encoding wild-type p105 was generated by cloning human p105 cDNA into the pCLXSN vector (27). PCLXSN-p105SSS/AAA was generated by site-directed mutagenesis to replace three serines (921, 923, and 927) located at the IKK phosphorylation site of p105 with alanines. The plasmid vectors encoding Tpl2 and IKKβ were generated by cloning the corresponding cDNA into the pcDNA-hemagglutinin (HA) vector (14), and the bacterial expression vector encoding glutathione S-transferase (GST)-MEK1 was constructed by cloning human MEK1 into the pGEX4T-3 vector (Amersham/Pharmacia Biotech). GST-p105C and GST-p105C SSS/AAA were generated by cloning the C-terminal 535 amino acids of wild-type p105 or p105SSS/AAA into pGEX4T-1 vector (Amersham/Pharmacia Biotech). GST-IκBα(1-54) has been described previously (38). GST fusion proteins were produced in bacteria and were purified with glutathione-conjugated Sepharose beads (Amersham/Pharmacia Biotech) following the manufacturer's instruction.
The anti-p105 antibody was provided by Nancy Rice. The sources of the other antibodies were as described previously (39). The IKKβ inhibitor PS1145 was kindly provided by Millennium Pharmaceuticals, Inc. (5), and the IKK inhibitor 15dPGJ2 was purchased from Biomol Research Company, Inc. Both inhibitors were dissolved in dimethyl sulfoxide (DMSO). LPS derived from Escherichia coli O127:B8 and phorbol myristate acetate (PMA) were obtained from Sigma, and murine TNF-α was from BioSource. The recombinant IKKα and IKKβ, produced in insect cells, were kindly provided by Michael Karin.
ELISA.
Bone marrow-derived macrophages (BMDM) were seeded at a density of 106 cells/ml. After overnight starvation, cells were stimulated with LPS for 6 h in the presence or absence of P1145. Supernatant was then collected from the cells after the 6-h LPS stimulation and TNF-α was measured by using an enzyme-linked immunosorbent assay (ELISA) kit from Pharmigen (OptEIA TNF-α kit) following the manufacturer's protocol.
In vitro translation.
The pcDNA-based Tpl2 and IKKβ plasmid vectors were used for in vitro translations, because they carry the T7 promoter. The coupled TNT in vitro translation kit (Promega) was used.
Cells, in vitro stimulation, and retroviral infection.
BMDM were prepared and cultured as previously described (39). MEFs derived from wild-type and IKK-deficient mice were kindly provided by Michael Karin. The murine RAW264 macrophage cell line was from American Type Culture Collection. The cells were starved overnight in medium supplemented with 0.5% fetal bovine serum and then were stimulated for the indicated times. Following stimulation the cells were rapidly scraped into phosphate-buffered saline from culturing dishes, collected by centrifugation, and immediately preseeded for extract preparation. Retroviral infection of the nfκb1−/− BMDM was as described previously (39).
Immunoblotting, coIP, and protein kinase assays.
The procedures for immunoblotting, coimmunoprecipitation (coIP), and kinase assays were as previously described (39).
EMSA. Nuclear extracts were prepared from BMDM and were subjected to electrophoresis mobility shift assay (EMSA) with a 32P-radiolabeled κB oligonucleotide as previously reported (35).
RESULTS
IKKβ inhibition in macrophages impairs ERK activation by LPS and TNF-α.
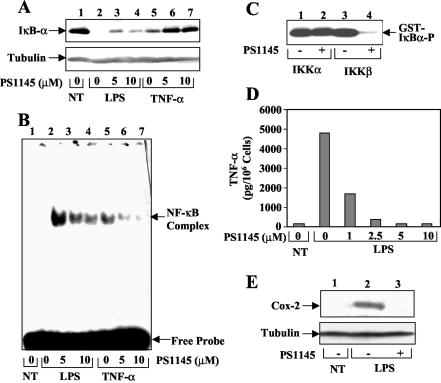
To investigate the role of IKK in regulating inflammatory responses, we examined the effect of a selective IKK inhibitor, PS1145 (5, 17), on NF-κB activation and production of inflammatory mediators in BMDM. As expected, both LPS and TNF-α induced degradation of the prototypical NF-κB inhibitor IκBα, with LPS being a stronger inducer (Fig. 1A, lanes 2 and 5). Preincubation of the cells with PS1145 partially inhibited the inducible degradation of IκBα by both LPS (lanes 3 and 4) and TNF-α (lanes 6 and 7). Furthermore, EMSA revealed a similar inhibitory effect of PS1145 on the activation of NF-κB (Fig. 1B). Using purified recombinant IKK components, we found that PS1145 selectively inhibited the kinase activity of IKKβ but had no effect on the function of IKKα (Fig. 1C). The inability of PS1145 to inhibit IKKα may explain the incomplete block of NF-κB activation in the drug-treated cells (Fig. 1B). We then examined the effect of PS1145 on two major proinflammatory mediators produced by LPS-stimulated macrophages, the cytokine TNF-α and the prostaglandin-generating enzyme COX-2. In the absence of PS1145, LPS potently stimulated the secretion of TNF-α (Fig. 1D) and expression of Cox-2 (Fig. 1E) by macrophages. More importantly, both of these LPS-mediated intracellular events were efficiently blocked by PS1145 (Fig. 1D and E).
FIG. 1.
Inhibitory effect of an IKKβ inhibitor on LPS-stimulated NF-κB activation and production of inflammatory mediators. (A) Immunoblot analysis of IκBα and the control tubulin in BMDM. The cells were preincubated with the indicated amounts of PS1145 for 90 min followed by stimulation with LPS (2.5 μg/ml) and TNF-α (10 ng/ml) for 20 min. Cytoplasmic extracts were subjected to immunoblot using anti-IκBα and anti-tubulin antibodies (39). (B) Nuclear extracts isolated from the BMDM described for panel A were subjected to EMSA to detect NF-κB DNA binding activity. (C) Inhibition of IKKβ, but not IKKα, by PS1145. Recombinant IKKα (10 ng) and IKKβ (5 ng) were incubated for 15 min (at 25°C) with either the solvent DMSO (−) or the IKK inhibitor PS1145 (10 μM), and their catalytic activity was analyzed by using GST-IκBα (1-54) as a substrate. (D) Inhibition of TNF-α production by PS1145. Macrophages were preincubated with the indicated amounts of PS1145 followed by stimulation with LPS for 6 h. The amounts of TNF-α secreted to the media were determined by ELISA and were expressed as pictograms per 106 cells. (E) Inhibition of COX-2 induction by PS1145. Macrophages were preincubated with (+) or without (−) PS1145 for 90 min, and the cells were either not treated (NT) or were stimulated for 2 h with LPS. COX-2 expression was analyzed by immunoblot.
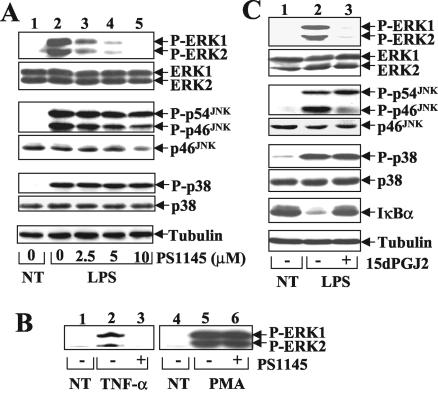
Given the potent anti-inflammatory function of PS1145, we examined whether it has additional targets in the LPS-signaling pathway. We performed in vivo phosphorylation assays to determine the effect of PS1145 on the activation of MAPKs. Remarkably, the LPS-stimulated activation of ERK1 and ERK2 was potently inhibited by the IKK inhibitor, although the activation of JNKs and p38 was not significantly influenced (Fig. 2A). Similar studies revealed that PS1145 also impaired TNF-α-stimulated ERK1 and ERK2 activation (Fig. 2B, lanes 1 to 3). Interestingly, however, this IKK inhibitor had no effect on ERK activation stimulated by the mitogen PMA (Fig. 2B, lanes 4 to 6). These results suggest that PS1145 inhibits the activation of ERK by specific inducers, namely the inflammatory elicitors LPS and TNF-α.
FIG. 2.
IKK inhibition specifically impairs LPS-stimulated ERK activation. (A) LPS-stimulated activation of ERK but not other MAPKs is sensitive to PS1145. Macrophages were preincubated with increasing doses of PS1145 followed by LPS stimulation (15 min). The phosphorylated (P-) and total MAPKs (ERK1 and ERK2, JNKs, and p38) and the loading control tubulin were analyzed by immunoblot as previously described (39). (B) Effect of PS1145 on ERK activation induced by TNF-α and PMA. The cells were preincubated for 90 min with (+) or without (−) PS1145 followed by stimulation with TNF-α (10 ng/ml) or PMA (25 ng/ml) for 15 min. ERK1 and ERK2 activation was analyzed by immunoblotting with a phosphospecific anti-ERK antibody. (C) Inhibition of ERK but not other MAPKs by another IKK inhibitor, 15dPGJ2. The cells were preincubated for 70 min with (+) or without (−) 15dPGJ2 (4 μM) and then were left either not treated (NT) or stimulated with LPS for 15 min. Immunoblotting was performed as described for panel A.
To investigate whether the ERK signaling could also be inhibited by other IKK inhibitors, we tested the effect of another well-characterized IKK inhibitor, 15dPGJ2 (30), on LPS-stimulated ERK activation. At 4 μM, 15dPGJ2 blocked the degradation of IκBα (Fig. 2C) and activation of NF-κB (data not shown) in LPS-treated cells. More importantly, inhibition of IKK by 15dPGJ2 resulted in the elimination of LPS-stimulated ERK1 and ERK2 activation (Fig. 2C). Similar to that of PS1145, the ERK-inhibitory function of 15dPGJ2 was specific, because it had little effect on LPS-stimulated activation of JNK and p38 (Fig. 2C) and PMA-stimulated ERK1 and ERK2 activation (data not shown). Together with the previous finding that the ERK pathway is essential for LPS-stimulated production of TNF-α and COX-2 (8, 9), these results suggest that the potent anti-inflammatory effect of IKK inhibitors involves inhibition of both the NF-κB and ERK signaling pathways. These findings also imply that IKK may function as an essential component of the LPS-responsive ERK signaling pathway.
IKK inhibitors block activation of MAP3K Tpl2.
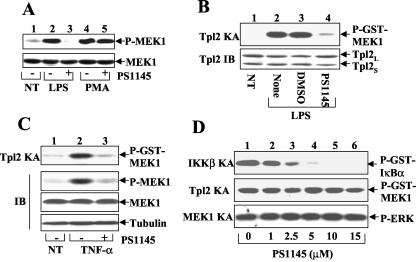
The finding that IKK is dispensable for PMA-stimulated ERK activation suggests that IKK does not directly regulate ERK but may target an upstream component in the ERK signaling pathway. In this regard, ERK activation is known to be mediated by a MAP2K, MEK1, which in turn is regulated by the MAP3K Tpl2 in the response to inflammatory signals (8, 10, 39). We first investigated the role of IKK in mediating the activation of MEK1. Stimulation of macrophages by LPS resulted in potent activation of MEK1, as demonstrated by its in vivo phosphorylation (Fig. 3A). More importantly, this signaling event was completely blocked by the IKKβ inhibitor PS1145 (Fig. 3A, lane 3). Similar to its effect on ERK activation, PS1145 did not inhibit the activation of MEK1 stimulated by PMA (Fig. 3A, lane 5). This result suggests that a more upstream kinase, instead of MEK1, may serve as the direct target of IKK.
FIG. 3.
IKK is required for Tpl2 activation. (A) Induction of MEK1 phosphorylation by LPS, but not by PMA, is sensitive to the IKK inhibitor. Macrophages were preincubated with (+) or without (−) PS1145 followed by stimulation for 15 min with LPS or PMA (25 ng/ml). Activated (upper panel) and total (lower panel) MEK1 proteins were detected by immunoblot using anti-phospho-MEK1 (P-MEK1) and anti-MEK1, respectively. (B) Macrophages were either not treated (NT) or were stimulated for 15 min with LPS following 90 min of preincubation with PS1145, carrier control DMSO, or medium (None). Tpl2 was isolated by immunoprecipitation and was subjected to kinase assays (KA) with GST-MEK1 as substrate (upper panel). The Tpl2 protein was monitored by immunoblot using a C-terminal-specific anti-Tpl2 antibody (anti-Cot M20; Santa Cruz). (C) Macrophages were preincubated with (+) or without (−) PS1145 followed by stimulation with TNF-α (10 ng/ml) for 15 min. In vitro kinase assays (top panel) and phosphospecific immunoblotting (second panel) were performed to measure the catalytic activity of Tpl2 and in vivo phosphorylation of MEK1, respectively. The expression levels of total MEK1 and the loading control tubulin were monitored by immunoblot (lower two panels). (D) Recombinant IKKβ purified from insect cells (top panel), recombinant Tpl2 purified from an in vitro translation system (middle panel), and recombinant MEK1 purified as a GST fusion protein from bacteria (bottom panel) were preincubated for 15 min (at 25°C) with increasing concentrations of PS1145 and then were subjected to kinase assays using GST-IκBa(1-54) (top panel), GST-MEK1 (middle panel), and ERK2 (bottom panel) as substrates, respectively.
Because Tpl2 is responsible for LPS-mediated MEK1 activation (8, 39), we performed kinase assays to examine the effect of PS1145 on LPS-mediated activation of Tpl2. As previously reported (39), LPS potently stimulated the catalytic activity of Tpl2 (Fig. 3B, lane 2). Interestingly, the Tpl2 activation was severely crippled when the cells were preincubated with PS1145 (lane 4). Similar results were obtained with the other IKK inhibitor, 15dPGJ2 (data not shown). A recent study suggests that the Tpl2 signaling pathway in macrophages is also targeted by the inflammatory cytokine TNF-α (10). We thus investigated whether the TNF-α-stimulated Tpl2 activation was also sensitive to IKK inhibitors. Like LPS, TNF-α stimulated the kinase activity of Tpl2, as demonstrated by the in vitro (Fig. 3C, top panel) and in vivo (second panel) phosphorylation of MEK1 (lane 2). Although TNF-α was a weaker Tpl2 inducer than LPS (data not shown), the TNF-α-stimulated Tpl2 activation was efficiently blocked by the IKK inhibitors (Fig. 3C, lane 3).
The above-described studies performed with IKK inhibitors suggested that the activation of Tpl2 is under the control of IKK. However, we also considered the possibility that the IKK inhibitors might directly affect the catalytic activity of Tpl2. To exclude this possibility, we performed detailed titration studies to examine the inhibitory effect of PS1145 on the catalytic activity of IKKβ and Tpl2. As expected, PS1145 efficiently inhibited the kinase activity of recombinant IKKβ in vitro (Fig. 3D, top panel). In contrast, the catalytic activity of recombinant Tpl2 was not significantly affected by PS1145 at doses up to 15 μM (middle panel). Similarly, PS1145 did not inhibit MEK1-mediated phosphorylation of ERK (lower panel). Parallel studies using 15dPGJ2 similarly revealed that this IKK inhibitor had no inhibitory effect on Tpl2 or MEK1 (data not shown). Therefore, it is likely that the IKK inhibitors have no direct effect on the catalytic activity of Tpl2 or its downstream kinase MEK1 but rather block the activation of these kinases through inhibition of IKK. These results further suggested that IKK is required for activation of the Tpl2 signaling pathway.
Activation of Tpl2 and phosphorylation of ERK are preceded by IKK activation.
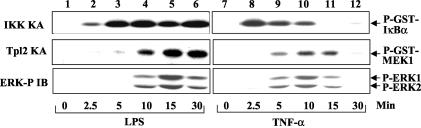
If IKK is indeed required for Tpl2 activation, its activation should precede that of Tpl2. We performed kinetic studies to examine this possibility. Upon LPS stimulation, IKK was activated as early as 2.5 min, activity reached the maximal level around 5 min, and the activity persisted until at least 30 min poststimulation (Fig. 4, top panel, lanes 2 to 6). The activation of Tpl2 was delayed compared to that of IKK. The Tpl2 activity was first detected at 5 min after LPS stimulation and reached the maximal level around 15 min (middle panel, lanes 2 to 6). Consistent with the role of Tpl2 in MEK1 activation (8), in vivo phosphorylation of the MEK1 substrate, ERK1 and ERK2, was also more delayed than the activation of IKK (bottom panel, lanes 2 to 6).
FIG. 4.
Activation of IKK precedes the activation of Tpl2 and ERK phosphorylation. BMDM were stimulated with either LPS or TNF-α for the indicated time periods. IKK and Tpl2 were isolated by immunoprecipitation using anti-IKKγ and anti-Tpl2 (anti-Cot M20), respectively, and were subjected to kinase assays (KA) as described in the legend to Fig. 3D. ERK phosphorylation was analyzed by immunoblotting (IB) using the phosphospecific anti-ERK antibody.
Parallel studies using TNF-α revealed that the overall signaling kinetics induced by this proinflammatory cytokine are more rapid and transient than that stimulated by LPS. Maximal IKK activation was detected at 2.5 min after TNF-α stimulation, and the activity became largely diminished around 15 min (Fig. 4, top panel, lanes 8 to 12). Importantly, the activation of Tpl2 and phosphorylation of ERK again exhibited delayed kinetics (middle and lower panels, lanes 8 to 12).
Genetic evidence for the essential role of IKKβ and IKKγ in Tpl2 activation.
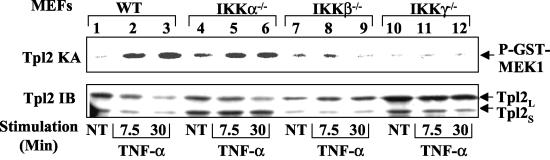
To obtain genetic evidence for the regulatory role of IKK in Tpl2 activation, we examined this signaling event by using mouse embryonic fibroblasts (MEF) harboring deficiencies in different IKK subunits. Although the wild-type MEFs did not appreciably respond to LPS (data not shown), they supported the Tpl2 activation by TNF-α (Fig. 5, upper panel, lanes 2 and 3). The MEFs deficient in IKKα (IKKα−/−) or IKKβ (IKKβ−/−) exhibited a residual basal activity of Tpl2 (lanes 4 and 7). Interestingly, the IKKα−/− MEFs were fully responsive to TNF-α-induced Tpl2 activation (lanes 4 to 6), suggesting that this IKK component is dispensable for this signaling event. In contrast, the TNF-α-stimulated Tpl2 activation was completely defective in the IKKβ−/− MEFs (lanes 7 to 9). Consistent with its essential role in modulating IKKβ activation, the regulatory subunit IKKγ was also required for TNF-α-mediated Tpl2 activation (lanes 10 to 12). We also tried to analyze the phosphorylation of ERK in the mutant MEFs. Unfortunately, these MEFs exhibited high constitutive ERK activity even after serum starvation (data not shown). Nevertheless, such ERK activity is independent of Tpl2, because Tpl2 remains inducible in these cells (Fig. 5) and because the constitutive ERK activity is insensitive to the IKK inhibitor (data not shown). Nevertheless, these genetic studies clearly demonstrate that IKKβ and IKKγ are required for Tpl2 activation.
FIG. 5.
IKKβ and IKKγ, but not IKKα, are essential for Tpl2 activation. MEFs derived from wild-type (WT) mice or the indicated IKK knockout mice were stimulated for the indicated times with TNF-α (50 ng/ml) followed by protein kinase assays (KA) to detect Tpl2 kinase activity (upper panel). The level of Tpl2 was monitored by immunoblot (IB) (lower panel).
IKK is required for LPS-stimulated liberation of Tpl2 from its inhibitor, p105.
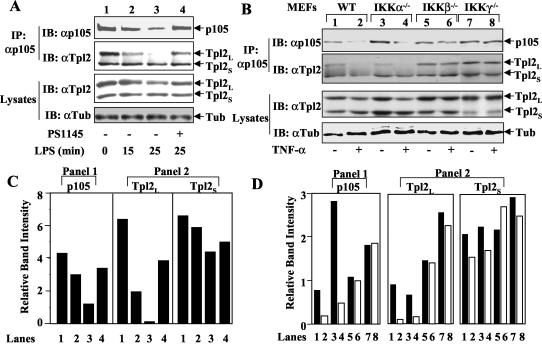
Recent studies suggest that the nfκb1 gene product p105 forms a stable complex with Tpl2 (4) and functions as a physiological inhibitor of Tpl2 (39). The activation of Tpl2 appears to involve the release of its long isoform (Tpl2L) from p105, although the underlying mechanisms have not been resolved (39). We examined whether the IKK inhibitors affected this critical step of Tpl2 activation. Consistent with the results of previous studies (4, 39), both isoforms of Tpl2 (Tpl2L and Tpl2S) were coprecipitated with p105 in a coIP assay (Fig. 6A, second panel, lane 1), suggesting their stable association with p105. Further, in response to LPS stimulation, Tpl2L was rapidly released from the p105 complex (lanes 2 and 3). Remarkably, the dissociation of Tpl2 from p105 was completely blocked by both of the IKK inhibitors, PS1145 (Fig. 6A, second panel, lane 4) and 15dPGJ2 (data not shown).
FIG. 6.
IKKβ and IKKγ are required for signal-induced release of Tpl2 from p105. (A) Macrophages were preincubated with (+) or without (−) PS1145 followed by LPS stimulation for the indicated times. P105 and its associated Tpl2 were isolated by immunoprecipitation (IP) using a C-terminal-specific anti-p105 antibody (29). The precipitated p105 (top panel) and Tpl2 isoforms (panel 2) were detected by immunoblot (IB) using anti-p105 and horseradish peroxidase-conjugated anti-Tpl2, respectively. The total amount of Tpl2 (panel 3) and the control tubulin (Tub) (bottom panel) in the cell lysates was monitored by direct immunoblot. (B) MEFs derived from wild-type (WT) mice or the indicated IKK knockout mice were either not treated (−) or were stimulated for 15 min with TNF-α (50 ng/ml) followed by coIP assays to detect the level of p105 (top panel) and p105-associated Tpl2 (panel 2). The total amounts of Tpl2 (panel 3) and tubulin (panel 2) in the cell lysates were monitored by direct immunoblotting (bottom panel). (C) Densitometry quantitation of protein bands presented in the first two gels of panel A. The lanes indicated below the graphs are the same as those indicated in panel A. (D) Densitometry quantitation of protein bands presented in the first two gels of panel B. The lanes indicated below the graphs correspond to the lanes of panel B.
We further confirmed the requirement of IKK in the induction of Tpl2/p105 complex dissociation by using IKK-deficient MEFs. As seen with LPS-stimulated macrophages, TNF-α stimulation of wild-type MEFs resulted in efficient release of Tpl2L from p105 (Fig. 6B, second panel, lane 2). This step of Tpl2 signaling does not require IKKα, because it remained normal in IKKα−/− MEFs (lane 4). In contrast, the TNF-α-induced Tpl2 and p105 dissociation was completely blocked in the IKKβ−/− (lane 6) and IKKγ−/− MEFs (lane 8). These genetic studies further confirmed the essential role for IKKβ as well as the regulatory subunit (IKKγ) of canonical IKK in regulating the activation of Tpl2.
Proteasome inhibitors block LPS-stimulated Tpl2/ERK signaling.
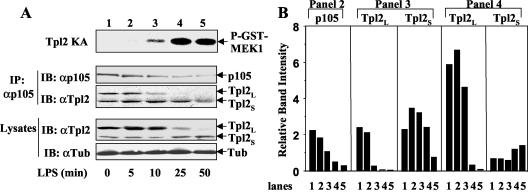
We noticed that the LPS-stimulated release of Tpl2L from p105 was associated with a partial loss of p105 (Fig. 6A, top panel, lanes 2 and 3). Interestingly, the p105 degradation was blocked by the IKKβ inhibitor PS1145 (lane 4). These findings suggest that the LPS-induced Tpl2 liberation and activation may result from IKK-mediated degradation of p105. We further examined this possibility by performing a more detailed time course experiment to correlate p105 degradation with Tpl2 activation, Tpl2L liberation, and degradation. A partial loss of p105 was noticeable at 10 min of LPS stimulation and became more prominent at 25 min (Fig. 7A, second panel). This dynamic change was correlated with the release of Tpl2L from p105 (third panel) and the activation of Tpl2 kinase activity (top panel). The kinetics of Tpl2L degradation (bottom panel) was evidently delayed compared to that of its liberation (third panel). These results support the hypothesis that activation of Tpl2 involves degradation of p105 and liberation of Tpl2L. We noticed that p105 was not completely degraded even after extended LPS stimulation (50 min), indicating that the degradation signal might preferentially target a proportion of p105. This idea is supported by the finding that the p105/Tpl2S complex was not significantly dissociated following LPS stimulation (Fig. 7A, third panel).
FIG. 7.
A time course analysis to correlate Tpl2 activation with p105 degradation and Tpl2L release. (A) BMDM were stimulated with LPS for the indicated time periods and then were subjected to Tpl2 kinase assay (KA) (top panel) as described in the legend to Fig. 3B. In parallel, the p105/Tpl2 complexes were isolated by immunoprecipitation (IP) using anti-p105C antibody followed by immunoblot (IB) detection of precipitated p105 (panel 2) and Tpl2 (panel 3) using specific antibodies. The expression levels of the Tpl2 isoforms (panel 4) and tubulin (bottom panel) in the cell lysates were analyzed by direct immunoblot. (B) Densitometry analysis of the protein bands from panels 2, 3, and 4 of panel A. The lanes indicated below the figure correspond to the lanes in panel A.
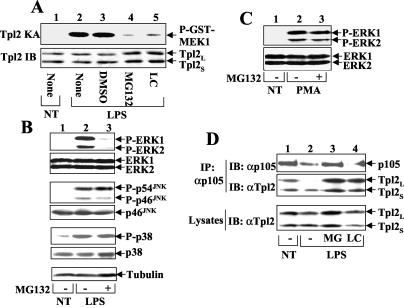
The inducible degradation of p105 is known to be mediated through the proteasome pathway (15). To further address the role of p105 degradation in mediating Tpl2 activation, we examined the effect of proteasome inhibitors on LPS-stimulated activation of Tpl2 and downstream signaling events. Remarkably, incubation of macrophages with two proteasome inhibitors, MG132 and lactocystine, efficiently blocked the LPS-stimulated activation of Tpl2 (Fig. 8A). Consistent with this activity, LPS-stimulated activation of the Tpl2-specific downstream kinases ERK1 and ERK2 was also inhibited by MG132 (Fig. 8B) and lactocystine (data not shown). This effect was highly specific, because the proteasome inhibitors had no inhibitory effect on the activation of JNKs and p38 (Fig. 8B). Further, the PMA-stimulated ERK1 and ERK2 activation, which is independent of Tpl2 (8, 39), was not inhibited by MG132 (Fig. 8C). Parallel coIP assays revealed that the proteasome inhibitors prevented the dissociation of Tpl2 from its inhibitor, p105 (Fig. 8D, middle panel). This action was associated with the blockade of LPS-stimulated degradation of p105 (Fig. 8D, upper panel). Together, these data indicate that the inducible degradation of p105 contributes to the release of Tpl2L and activation of Tpl2 catalytic activity, although the possibility for involvement of proteolysis in certain other steps of Tpl2 activation cannot be excluded. Nevertheless, it is clear that LPS-stimulated Tpl2/ERK signaling requires both IKK and the proteasome.
FIG. 8.
LPS-stimulated Tpl2/ERK signaling is sensitive to proteasome inhibitors. (A) Macrophages were either not treated (NT) or were stimulated for 15 min with LPS following preincubation (60 min) with medium (None), carrier control (DMSO), or the proteasome inhibitors MG132 and lactacysteine (LC). Tpl2 kinase activity (upper panel) and protein level (lower panel) were determined by kinase assays (KA) and immunoblotting (IB). (B) Cells were either not treated (NT) or were stimulated for 15 min with LPS following preincubation (30 min) with (+) or without (−) MG132. Activated (upper panels) and total (lower panels) MAPKs were detected by immunoblot using corresponding phosphospecific (P-) and pan-antibodies. As a loading control, an immunoblot with antitubulin was included. (C) Macrophages were either not treated (NT) or were stimulated for 15 min with PMA following preincubation with MG132 (30 min). Activated (upper panel) and total (lower panel) ERK proteins were detected by immunoblot using anti-phospho-ERK (P-ERK) and anti-ERK, respectively. (D) Macrophages were preincubated without (−) or with MG132 (MG) or lactacysteine (LC) and then left either not treated (NT) or stimulated with LPS for 25 min. The release of Tpl2L from p105 was determined by coIP (top and middle panels) as described in the legend to Fig. 6. The expression level of Tpl2 isoforms in the cell lysates was determined by direct immunoblot (bottom panel).
Site-specific phosphorylation of p105 is an essential step in LPS-stimulated MEK and ERK signaling.
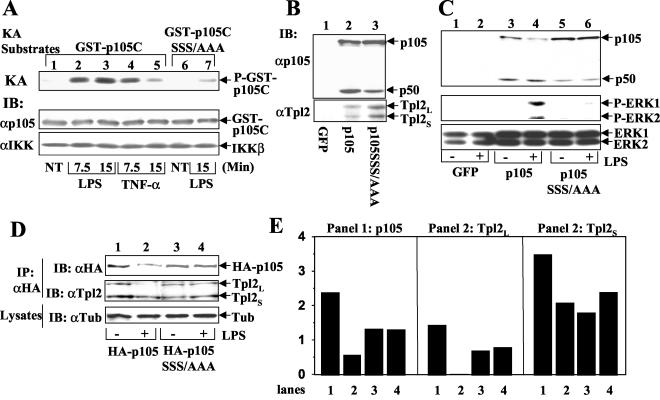
Previous studies have shown that IKK phosphorylates p105 at three C-terminal serines (16, 28), although the physiological role of this signaling event has remained unclear. Consistent with previous findings, the IKK complex isolated from LPS-stimulated macrophages efficiently phosphorylated p105 (Fig. 9A, lanes 2 and 3). Similar results were obtained with TNF-α-treated cells, although the activation of IKK was more transient (lanes 4 and 5). Further, the phosphorylation of p105 requires its C-terminal serines, because a mutant form lacking these serines (p105SSS/AAA) was only weakly phosphorylated (lane 7). To address the physiological role of p105 phosphorylation in Tpl2 activation, we employed a genetic model system involving reconstitution of the nfκb1−/− macrophages with p105 or p105SSS/AAA. In this regard, it was previously shown that the nfκb1−/− cells have a deficiency in Tpl2 expression and LPS-stimulated ERK activation, which can be rescued by expression of p105 (39). As expected, the steady expression of Tpl2 in nfκb1−/− cells was restored upon p105 expression (Fig. 9B, lower panel, lane 2). The same result was obtained by expression of p105SSS/AAA (lane 3), thus suggesting the dispensability of p105 phosphorylation in Tpl2 stabilization. However, unlike the wild-type p105, the p105SSS/AAA mutant was unable to rescue the signaling defect of nfκb1−/− macrophages in LPS-stimulated ERK activation (Fig. 9C, middle panel). This functional deficiency of p105SSS/AAA was apparently due to its inability to respond to LPS signal for degradation. Indeed, the wild-type p105 (Fig. 9C, upper panel, lane 4) but not p105SSS/AAA (lane 6) was degraded by the LPS signal. These genetic studies further suggest an essential role for IKK-mediated p105 phosphorylation in LPS-stimulated ERK signaling.
FIG. 9.
The IKK phosphorylation site of p105 is essential for mediating Tpl2 activation in macrophages. (A) Site-specific phosphorylation of p105 by LPS-stimulated IKK in macrophages. Macrophages were stimulated with the indicated inducers. IKK complex was isolated by immunoprecipitation (IP) using anti-IKKγ followed by kinase assays (KA) (top panel) using GST-p105C or GST-p105C harboring serine-to-alanine mutations at the IKK phosphorylation site of p105 (p105C SSS/AAA). Following autoradiography, the kinase filter was subjected to immunoblotting (IB) using anti-p105C (middle panel) and anti-IKKα/β (H-470; Santa Cruz) (bottom panel) to detect the levels of substrates and IKKβ. (B) nfκb1−/− macrophages were infected with retroviruses encoding green fluorescence protein (GFP), wild-type p105, or a p105 mutant harboring serine-to-alanine mutations at the IKK phosphorylation site (p105SSS/AAA). The exogenous p105 proteins (upper panel) and rescued Tpl2 isoforms (lower panel) were detected by immunoblot using N-terminal-specific anti-p105 and C-terminal-specific anti-Tpl2 antibodies, respectively. (C) The retrovirus-infected nfκb1−/− macrophages were either not treated (−) or were stimulated for 15 min with LPS followed by immunoblot to analyze the degradation of p105 (top panel), activation of ERK1 and ERK2 (middle panel), and total ERK1 and ERK2 (bottom panel). (D) Tpl2L cannot be released from a phosphorylation-defective p105 mutant. Raw264 murine macrophage cells were infected with retroviruses encoding HA-tagged wild-type p105 or a phosphorylation-defective p105 mutant (p105SSS/AAA). Bulk infected cells were either not treated (−) or were stimulated (+) with LPS for 15 min followed by isolation of the HA-tagged p105/Tpl2 complexes by using anti-HA antibody. The exogenous p105 and coprecipitated Tpl2 proteins were detected by immunoblotting using anti-HA and horseradish peroxidase-conjugated anti-Tpl2 (anti-Cot M20), respectively. The use of horseradish peroxidase-conjugated anti-Tpl2 was essential to avoid interference from the immunoglobulin heavy chain. A tubulin immunoblot was included as a loading control (bottom panel). (E) Densitometry quantitation of protein bands from the first two gels of panel D. The lanes indicated below the graphs correspond to the lanes in panel D.
We next determined whether p105 phosphorylation is essential for the liberation of Tpl2L. For these studies, wild-type p105 and p105SSS/AAA were stably expressed in the Raw264 macrophage cell line, and then their association with Tpl2 under untreated and LPS-stimulated conditions was analyzed. Both wild-type p105 and p105SSS/AAA formed a stable complex with Tpl2 in untreated cells (Fig. 9D, middle panel, lanes 1 and 3). However, upon LPS stimulation, Tpl2L was quickly dissociated from wild-type p105 (lane 2) but remained associated with p105SSS/AAA (lane 4). Thus, the phosphorylation site of p105 is essential for mediating the signal-induced liberation of Tpl2L.
DISCUSSION
Macrophage activation by microbial components is a key step in the development of an innate immune response. This step involves the integration of cascades of intracellular signaling pathways, including those that lead to activation of NF-κB and different classes of MAPKs. Compelling evidence suggests that IKK plays a pivotal role in macrophage activation as well as in many other parts of immune responses (20, 34). Up to now, IKK has been thought to function as a specific effector kinase mediating activation of NF-κB. The results presented in the present study, however, suggest that this key immune regulator also acts through activating other signaling pathways.
Using both IKK inhibitors and IKK-deficient MEFs, we have shown that IKK is required for activation of Tpl2, a MAP3K that specifically mediates MEK and ERK activation by immune stimuli (8, 10, 39). This novel function of IKK appears to be mediated by its canonical complex. Indeed, IKKβ, the key component of the canonical IKK (12), is essential for Tpl2 activation. Further, the regulatory subunit of the canonical IKK complex, IKKγ, is also indispensable for this signaling function. In contrast, the activation of Tpl2 does not require the noncanonical IKK component IKKα. We have shown that inhibition of IKKβ by the inhibitor PS1145 does not completely eliminate LPS-stimulated NF-κB activation. This is likely due to the functional compensation by IKKα, as suggested by IKKβ knockout studies (21). Surprisingly, however, the IKKβ inhibitor is effective in blocking the Tpl2/ERK signaling in macrophages. This result suggests that the role of IKKβ in Tpl2 activation cannot be compensated for by IKKα. This idea is further supported by the finding that IKKβ−/− MEFs are completely defective in Tpl2 activation.
Although our data clearly demonstrate the requirement for IKKβ in Tpl2 activation, the role of IKKβ in mediating ERK activation is less clear. Because ERK activation can be mediated through either Tpl2-dependent or -independent mechanisms, the effect of IKKβ on this downstream pathway may vary among cell types and cellular inducers. In this regard, previous Tpl2 knockout studies suggest that ERK activation by LPS and TNF-α in macrophages is dependent on Tpl2 (8, 9). We have consistently shown that inhibition of Tpl2 by the IKKβ inhibitor abolishes ERK activation by LPS and TNF-α. On the other hand, a recent study revealed that conditional knockout of IKKβ in hepatocytes has no effect on ERK activation in response to LPS administration (25). Because LPS likely induces hepatocyte effects through TNF-α, this finding indicates that IKKβ may not be required for TNF-α-induced ERK activation in hepatocytes. Because a major role of Tpl2 is the regulation of ERK signaling in macrophages (8), mutant mice with IKKβ deficiency in macrophages will be important for understanding the in vivo role of the IKKβ/Tpl2 signaling pathway in macrophage activation and inflammatory responses.
It was previously shown that Tpl2 activation involves the release of its long isoform, Tpl2L, from the NF-κB1 precursor protein p105 (39). Our present results suggest that the Tpl2L liberation requires p105 phosphorylation by IKKβ, because Tpl2L cannot be liberated from a p105 mutant lacking its IKK phosphorylation site. Further, both the release of Tpl2L from p105 and the activation of Tpl2 catalytic activity are blocked in cells lacking IKKβ or treated with IKKβ inhibitors. Several lines of evidence indicate that the phosphorylation-coupled degradation of p105 contributes to Tpl2L liberation. First, partial degradation of p105 is associated with Tpl2L release. Second, inhibition of p105 degradation by proteasome inhibitors blocks the release of Tpl2L and the activation of Tpl2. Third, the defect in Tpl2L/p105 dissociation in IKKβ−/− and IKKγ−/− MEFs is also associated with the blockade in p105 degradation. On the other hand, our data do not exclude the possibility of the involvement of proteolytic events in other signaling steps of Tpl2 activation.
It is presently unclear how cellular signals specifically target the dissociation of p105/Tpl2L complex but not that of the p105/Tpl2S complex. One possibility is that the p105/Tpl2 dissociation requires modifications (e.g., phosphorylation) of both p105 and Tpl2. In this regard, Tpl2L contains a unique N-terminal region that is missing in Tpl2S. The modification of Tpl2L may also serve to trigger its catalytic activity. Given that overexpressed Tpl2 induces p105 degradation (4), it is possible that the activated Tpl2L participates in signal-induced degradation of p105. Future studies will determine the precise sequence requirement for signal-induced liberation of Tpl2L from p105. Additionally, it is also important to examine whether any upstream kinases other than IKKβ are required for Tpl2 activation. Given the high specificity of the Tpl2 activation signals, it is conceivable that activation of this signaling pathway requires both IKKβ and additional regulators. Notwithstanding these issues, our results have uncovered an essential function of IKKβ in Tpl2 activation and suggest the idea that the immunoregulatory function of IKKβ is beyond the activation of NF-κB. It remains possible that IKK may target additional downstream pathways in other parts of the immune response.
Acknowledgments
We thank Millennium Pharmaceuticals, Inc., for the IKK inhibitor PS1145, Michael Karin for the MEFs and recombinant IKK, and N. R. Rice for the anti-p105 antibody.
This work was supported by research grant 1 R01 AI057555, awarded to S.-C.S. and M.Z., from the National Institutes of Health. M.W. and W.R. were supported by a predoctoral and postdoctoral training grant (5T32CA60395-10) from the National Institutes of Health.
REFERENCES
- 1.Aderem, A., and R. J. Ulevitch. 2000. Toll-like receptors in the induction of the innate immune response. Nature 406:782-787. [DOI] [PubMed] [Google Scholar]
- 2.Akira, S., K. Takeda, and T. Kaisho. 2001. Toll-like receptors: critical proteins linking innate and acquired immunity. Nat. Immunol. 2:675-680. [DOI] [PubMed] [Google Scholar]
- 3.Baldwin, A. S., Jr. 1996. The NF-κB and IκB proteins: new discoveries and insights. Annu. Rev. Immunol. 14:649-683. [DOI] [PubMed] [Google Scholar]
- 4.Belich, M. P., A. Salmeron, L. H. Johnston, and S. C. Ley. 1999. TPL-2 kinase regulates the proteolysis of the NF-κB-inhibitory protein NF-κB1 p105. Nature 397:363-368. [DOI] [PubMed] [Google Scholar]
- 5.Castro, A. C., L. C. Dang, F. Soucy, L. Grenier, H. Mazdigasni, M. Hottelet, L. Parent, C. Pien, V. Palombella, and J. Adams. 2003. Novel IKK inhibitors: β-carbonlines. Bioorgan. Med. Chem. Lett. 13:2419-2422. [DOI] [PubMed] [Google Scholar]
- 6.Chen, L. W., L. Egan, Z. W. Li, F. R. Greten, M. F. Kagnoff, and M. Karin. 2003. The two faces of IKK and NF-κB inhibition: prevention of systemic inflammation but increased local injury following intestinal ischemia-reperfusion. Nat. Med. 9:575-581. [DOI] [PubMed] [Google Scholar]
- 7.Dejardin, E., N. M. Droin, M. Delhase, E. Haas, Y. Cao, C. Makris, Z. W. Li, M. Karin, C. F. Ware, and D. R. Green. 2002. The lymphotoxin-beta receptor induces different patterns of gene expression via two NF-κB pathways. Immunity 17:525-535. [DOI] [PubMed] [Google Scholar]
- 8.Dumitru, C. D., J. D. Ceci, C. Tsatsanis, D. Kontoyiannis, K. Stamatakis, J. H. Lin, C. Patriotis, N. A. Jenkins, N. G. Copeland, G. Kollias, and P. N. Tsichlis. 2000. TNF-α induction by LPS is regulated posttranscriptionally via a Tpl2/ERK-dependent pathway. Cell 103:1071-1083. [DOI] [PubMed] [Google Scholar]
- 9.Eliopoulos, A. G., C. D. Dumitru, C.-C. Wang, J. Cho, and P. N. Tsichlis. 2002. Induction of COX-2 by LPS in macrophages is regulated by Tpl2-dependent CREB activation signals. EMBO J. 21:4831-4840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Eliopoulos, A. G., C.-C. Wang, C. D. Dumitru, and P. N. Tsichlis. 2003. Tpl2 transduces CD40 and TNF signals that activate ERK and regulates IgE induction by CD40. EMBO J. 15:3855-3864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fan, C. M., and T. Maniatis. 1991. Generation of p50 subunit of NF-κB by processing of p105 through an ATP-dependent pathway. Nature 354:395-398. [DOI] [PubMed] [Google Scholar]
- 12.Ghosh, S., and M. Karin. 2002. Missing pieces in the NF-κB puzzle. Cell 109:S81-S96. [DOI] [PubMed] [Google Scholar]
- 13.Harhaj, E. W., S. B. Maggirwar, and S.-C. Sun. 1996. Inhibition of p105 processing by NF-κB proteins in transiently transfected cells. Oncogene 12:2385-2392. [PubMed] [Google Scholar]
- 14.Harhaj, E. W., and S.-C. Sun. 1999. IKKg serves as a docking subunit of the IkB kinase and mediates interaction of IKK with the human T-cell leukemia virus Tax protein. J. Biol. Chem. 274:22911-22914. [DOI] [PubMed] [Google Scholar]
- 15.Heissmeyer, V., D. Krappmann, E. N. Hatada, and C. Scheidereit. 2001. Shared pathways of IκB kinase-induced SCFβTrCP-mediated ubiquitination and degradation for the NF-κB precursor p105 and IκBα. Mol. Cell. Biol. 21:1024-1035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Heissmeyer, V., D. Krappmann, F. G. Wulczyn, and C. Scheidereit. 1999. NF-κB p105 is a target of IκB kinases and controls signal induction of Bcl-3-p50 complexes. EMBO J. 18:4766-4778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hideshima, T., D. Chanhan, P. Richardson, N. Mitsiades, T. Hayashi, N. Munshi, L. Dang, A. Castro, V. Palombella, J. Adams, and K. C. Anderson. 2002. NF-kB as a therapeutic target in multiple myeloma. J. Biol. Chem. 277:16639-16647. [DOI] [PubMed] [Google Scholar]
- 18.Janeway, C. A., P. Travers, M. Walport, and M. Shlomchik. 2001. Immunobiology, 5th ed. Garland, New York, N.Y.
- 19.Karin, M., and Y. Ben-Neriah. 2000. Phosphorylation meets ubiquitination: the control of NF-[κ]B activity. Annu. Rev. Immunol. 18:621-663. [DOI] [PubMed] [Google Scholar]
- 20.Karin, M., and M. Delhase. 2000. The IκB kinase (IKK) and NF-κB: key elements of proinflammatory signalling. Semin. Immunol. 12:85-98. [DOI] [PubMed] [Google Scholar]
- 21.Li, Q., G. Estepa, S. Memet, A. Israel, and I. M. Verma. 2000. Complete lack of NF-κB activity in IKK1 and IKK2 double-deficient mice: additional defect in neurulation. Genes Dev. 14:1729-1733. [PMC free article] [PubMed] [Google Scholar]
- 22.Li, Q., D. Van Antwerp, F. Mercurio, K. F. Lee, and I. M. Verma. 1999. Severe liver degeneration in mice lacking the IκB kinase 2 gene. Science 284:321-325. [DOI] [PubMed] [Google Scholar]
- 23.Li, Z. W., W. Chu, Y. Hu, M. Delhase, T. Deerinck, M. Ellisman, R. Johnson, and M. Karin. 1999. The IKKbeta Subunit of IκB kinase (IKK) is essential for nuclear factor κB activation and prevention of apoptosis. J. Exp. Med. 189:1839-1845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lin, L., G. N. DeMartino, and W. C. Greene. 1998. Cotranslational biogenesis of NF-κB p50 by the 26S proteasome. Cell 92:819-828. [DOI] [PubMed] [Google Scholar]
- 25.Maeda, S., L. Chang, Z. W. Li, J. L. Luo, H. Leffert, and M. Karin. 2003. IKKβ is required for prevention of apoptosis mediated by cell-bound but not by circulating TNF-α. Immunity 19:725-737. [DOI] [PubMed] [Google Scholar]
- 26.Mercurio, F., J. A. DiDonato, C. Rosette, and M. Karin. 1993. p105 and p98 precursor proteins play an active role in NF-κB-mediated signal transduction. Genes Dev. 7:705-718. [DOI] [PubMed] [Google Scholar]
- 27.Naviaux, R. N., E. Costanzi, M. Haas, and I. M. Verma. 1996. The pCL vector system: rapid production of helper-free high-titer, recombinant retroviruses. J. Virol. 70:5701-5705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Orian, A., H. Gonen, B. Bercovich, I. Fajerman, E. Eytan, A. Israel, F. Mercurio, K. Iwai, A. L. Schwartz, and A. Ciechanover. 2000. SCF(beta)(-TrCP) ubiquitin ligase-mediated processing of NF-κB p105 requires phosphorylation of its C-terminus by IκB kinase. EMBO J. 19:2580-2591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rice, N. R., M. L. MacKichan, and A. Israel. 1992. The precursor of NF-κB p50 has IκB-like functions. Cell 71:243-253. [DOI] [PubMed] [Google Scholar]
- 30.Rossi, A., P. Kapahi, G. Natoli, T. Takahashi, Y. Chen, M. Karin, and M. G. Santoro. 2000. Anti-inflammatory cyclopentene prostaglandins are direct inhibitors of IκB kinase. Nature 403:103-108. [DOI] [PubMed] [Google Scholar]
- 31.Senftleben, U., Y. Cao, G. Xiao, G. Kraehn, F. Greten, Y. Chen, Y. Hu, A. Fong, S.-C. Sun, and M. Karin. 2001. Activation of IKKα of a second, evolutionary conserved, NF-κB signaling pathway. Science 293:1495-1499. [DOI] [PubMed] [Google Scholar]
- 32.Sha, W. C., H. C. Liou, E. I. Tuomanen, and D. Baltimore. 1995. Targeted disruption of the p50 subunit of NF-κ B leads to multifocal defects in immune responses. Cell 80:321-330. [DOI] [PubMed] [Google Scholar]
- 33.Siebenlist, U., G. Franzoso, and K. Brown. 1994. Structure, regulation and function of NF-κB. Annu. Rev. Cell Biol. 10:405-455. [DOI] [PubMed] [Google Scholar]
- 34.Silverman, N., and T. Maniatis. 2001. NF-κB signaling pathways in mammalian and insect innate immunity. Genes Dev. 15:2321-2342. [DOI] [PubMed] [Google Scholar]
- 35.Sun, S.-C., P. A. Ganchi, D. W. Ballard, and W. C. Greene. 1993. NF-κB controls expression of inhibitor IκBα: evidence for an inducible autoregulatory pathway. Science 259:1912-1915. [DOI] [PubMed] [Google Scholar]
- 36.Sweet, M. J., and D. A. Hume. 1996. Endotoxin signal transduction in macrophages. J. Leukoc. Biol. 60:8-26. [DOI] [PubMed] [Google Scholar]
- 37.Tanaka, M., M. E. Fuentes, K. Yamaguchi, M. H. Durnin, S. A. Dalrymple, K. L. Hardy, and D. V. Goeddel. 1999. Embryonic lethality, liver degeneration, and impaired NF-κ B activation in IKK-beta-deficient mice. Immunity 10:421-429. [DOI] [PubMed] [Google Scholar]
- 38.Uhlik, M., L. Good, G. Xiao, E. W. Harhaj, E. Zandi, M. Karin, and S.-C. Sun. 1998. NF-κB-inducing kinase and IκB kinase participate in human T-cell leukemia virus I Tax-mediated NF-κB activation. J. Biol. Chem. 273:21132-21136. [DOI] [PubMed] [Google Scholar]
- 39.Waterfield, M. R., M. Zhang, L. P. Norman, and S. C. Sun. 2003. NF-κB1/p105 regulates lipopolysaccharide-stimulated MAP kinase signaling by governing the stability and function of the Tpl2 kinase. Mol. Cell 11:685-694. [DOI] [PubMed] [Google Scholar]
- 40.Xiao, G., M. E. Cvijic, A. Fong, E. W. Harhaj, M. T. Uhlik, M. Waterfield, and S. C. Sun. 2001. Retroviral oncoprotein Tax induces processing of NF-κB2/p100 in T cells: evidence for the involvement of IKKα. EMBO J. 20:6805-6815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Xiao, G., E. W. Harhaj, and S. C. Sun. 2001. NF-κB-inducing kinase regulates the processing of NF-κB2 p100. Mol. Cell 7:401-409. [DOI] [PubMed] [Google Scholar]