Abstract
Background:
Peroxisome proliferator-activated receptors (PPAR)-γ is widely used as an attractive target for the treatment of type 2 diabetes mellitus. Thiazolidinediones, the agonists of PPARγ, has been popularly utilized as insulin sensitizers in the therapy of type 2 diabetes whereas numerous severe side-effects may also occur concomitantly.
Objective:
The PPARγ activation activity of different polar extracts, including petroleum ether, ethyl acetate, n-butanol, residual of ethanol, the precipitate part of water and the supernatant of water extracts, from 37 traditional Chinese medicines were systematically evaluated.
Materials and Methods:
HeLa cells were transiently co-transfected with the re-constructed plasmids of GAL4-PPARγ-ligand binding domain and pGL4.35. The activation of PPARγ by different polarity extracts were evaluated based on the PPARγ transactivation assay and rosiglitazone was used as positive control.
Results:
Seven medicines (root bark of Lycium barbarum, Anoectochilus sroxburghii, the rhizome of Phragmites australis, Pterocephalus hookeri, Polygonatum sibiricum, fruit of Gleditsia sinensis, and Epimedium brevicornu) were able to significantly activate PPARγ.
Conclusion:
As seven medicines were able to activate PPARγ, the anti-diabetic activity of them is likely to be mediated by this nuclear receptor.
SUMMARY
Lots of the tested medicinal products had activation effects on activating PPARγ
Ethyl acetate extracts of root bark of L.barbarum, rhizome of P.saustralis and fruit of G.siasinensis showed good PPARγ activation effect similar or higher than that of positive control, 0.5 μg/mL rosiglitazone
Petroleum ether extracts of A.roxburghii, P. hookeri, P. sibiricum, E.brevicornu also can significantly activate PPARγ, the effects of them were higher than t0.5 μg/mL rosiglitazone
Schisandra chinensis (Turcz.) Baill., the fruit Cornus officinalis Siebold and Zucc., Alisma plantago-aquatica L. and the root of Trichosanthes Kirilowii Maxim., traditional anti-diabetic mediciness in China, had no effects on the activation of PPARγ.
Abbreviations used: PPARγ: Peroxisome Proliferator-activated Receptors-γ, TCMs: Traditional Chinese medicines, TZDs: Thiazolidinediones, LBD: Ligand binding domain, DMSO: Dimethyl sulfoxide, FBS: Fetal bovine serum, DMEM: Dulbecco's modified Eagle's medium.
Keywords: Diabetes mellitus, different polarity extracts, peroxisome proliferator-activated receptor-γ, traditional anti-diabetic medicines
INTRODUCTION
Diabetes mellitus is a metabolic disease characterized by insulin resistance coupled with a lackage of enough insulin to control blood glucose.[1] It is well-known that chronic hyperglycemia results in many diabetic complications such as diabetic nephropathy, obesity, and atherosclerosis.[2]
Peroxisome proliferator-activated receptors (PPARs) have been implicated to participate in many critical physiological and pathological processes, especially in the treatment of diabetes mellitus, obesity, and atherosclerosis.[3,4] They play important roles in the expression of various genes which are crucial to lipid and glucose metabolism.[5] There are three PPAR subtypes, PPARα, PPARβ/δ, and PPARγ, which have respective expression patterns and regulate different biological processes based on the requirement of a specific tissue.[6] PPARγ is expressed in adipose cells as well as islet beta cells.[7] Activation of PPARγ can improve insulin sensitization and reduce the risk of the insulin resistance in adipose tissue, liver tissue, and et al.[8,9] The activation of it affects the glucose homeostasis and lipid metabolism as well as inflammation.[10] Increasing evidence indicated that the activation of PPARγ can promote the adipocyte differentiation, decrease the accumulation of glucose in adipose tissue.[11,12] Moreover, PPARγ plays an important role in the regulation of pancreatic functions and activation of PPARγ can decrease β-cell apoptosis.[13] It is also known that PPARγ affects glucose-stimulated insulin secretion.[14,15] Therefore, the screening of drugs which can activate PPARγ has great significance in the treatment of diabetes mellitus and related metabolic syndromes.
Thiazolidinediones (TZDs), the agonists of PPARγ, had been demonstrated to have a variety of clinical effects such as enhancing insulin sensitivity and improving glucose tolerance.[16,17] However, recently evidence demonstrates that TZDs had several side-effects such as cardiovascular risks, liver damage, weight gain, and peripheral edema.[18,19] On the other hand, traditional Chinese medicines (TCMs) with fewer side-effects have proven historically to be a potential source for drug discovery. Therefore, numbers of researches have been done to explore the activators of PPARγ from TCMs.[10,20]
In previous study, we developed a cell-based PPARs screening model. The model is a stable and effective assay tool to characterize the interactions between PPAR subtype and PPARs activating drugs. Only when a drug bound and activated GAL4-PPARγ-ligand binding domain (LBD) could the luciferase be expressed. The Z’-factor, which was a useful tool and a statistical characteristic of any given assay, was used to evaluate the reliability and the stability of our model. When the value of Z’-factor was between 0.5 and 1, suggesting that the model was suitable for drug screening. The value of Z’-factor of our model was 0.64.[21]
Herein, this study aimed to preliminary screen and to evaluate the activation of PPARγ by different polarity extracts from 37 TCMs on the basis of PPARγ transactivation assay, laying the basis of further identifying the effects of active extracts on PPARγ-mediated gene expression, and biological responses and isolating the active compounds from active extracts.
MATERIALS AND METHODS
Cells and reagents
HeLa cells were purchased from the Cell Center of the Chinese Academy of Medicinal Sciences (Shanghai, China). Fetal bovine serum (FBS) and Dulbecco’s modified Eagle’s medium (DMEM) were products of Gibco BRL (Gaithersburg, MD, USA). FUGene® HD Transfection Reagent and Bright-Glo™ Luciferase Assay were purchased from Promega (Madison, WI, USA). Dimethyl sulfoxide (DMSO), penicillin, streptomycin, and rosiglitazone were provided from Sigma-Aldrich Chemical Co, Ltd (St. Louis, MO, USA).
Plasmid construction
pGL4.35 (luc2P/9XGAL4UAS/Hygro) Vector (Product No. E1370) and GAL4-GR Vector (Product No. E1581) were from Promega Corporation (Madison, WI, USA). The synthetic PPARγ-LBD gene sequences were inserted into GAL4-GR vector, and the sequencing of PPARγ-LBD genes was analyzed by restriction enzyme digestion (XhoI and XbaI) and gel electrophoresis. Then PPARγ-LBD replaced glucocorticoid receptor-LBD (GR-LBD) gene fusion in original GAL4-GR Vector and then formed the GAL4-PPARγ-LBD fusion protein. The pGL4.35 (luc2P/9XGAL4UAS/Hygro) vector containing GAL4 special response element of firefly luciferase was used as reporter gene.
Collection of medicines material
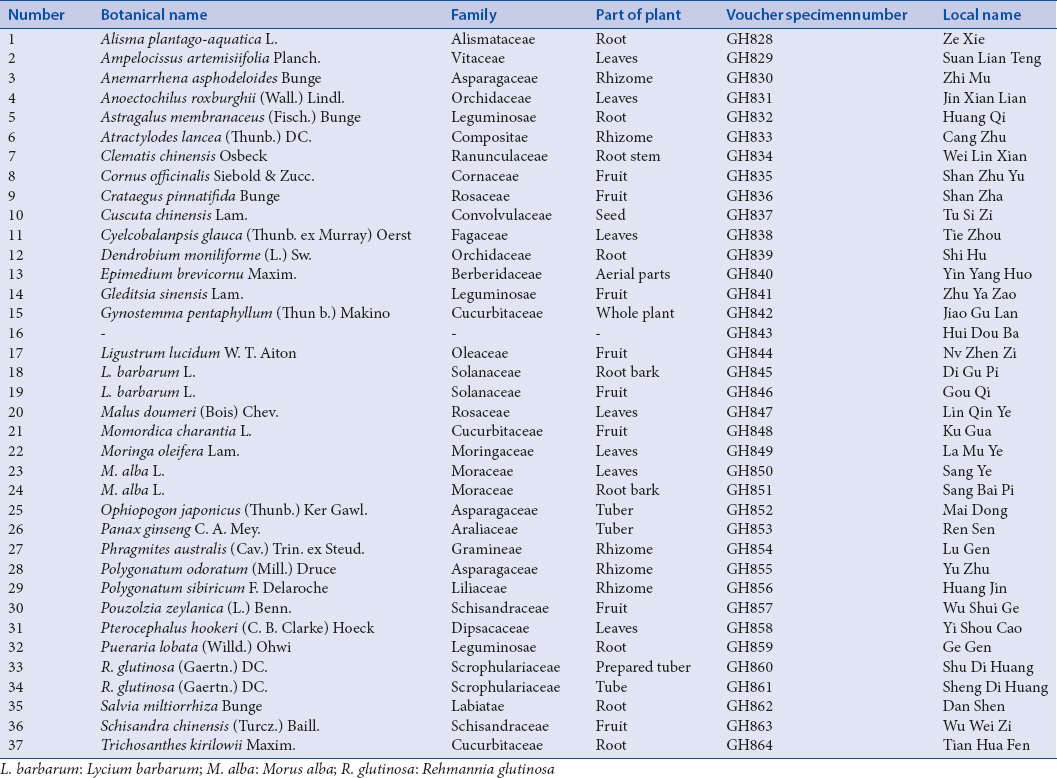
The TCMs were obtained from the local drug stores, TCMs markets or production place [Table 1]. Further identification of the specimens was authenticated by Professor GuoyueZhong (Jiangxi University of TCM). All voucher specimens have been deposited at Chongqing Academy of Chinese Materia Medica, Chongqing, China.
Table 1.
The plants species tested for potential peroxisome proliferator-activated receptor gamma activation properties in the screening model
Preparation of different polarity extracts
The dried materials of 37 TCMs were ground into fine powder in a pulverizer, respectively. Reflux extraction was conducted in turn by petroleum ether, 70% ethanol, and water. Then, 70% ethanol extract was liquid-liquid extracted by ethyl acetate, while the concentrated segments of water were treated with absolute ethanol until the absolute ethanol reached to 60%. Afterward, the precipitate and the supernatant were collected, respectively. Finally, petroleum ether, ethyl acetate, the residual of ethanol, the precipitate, and the supernatant of water extracts were obtained. Each part was concentrated to dryness in a vacuum to afford samples for biological tests.
The positive drugs, petroleum ether, and ethyl acetate extracts were dissolved in DMSO. The residual of ethanol, the precipitate part of water, and the supernatant of water extracts were dissolved in water. All of them were stored at −20°C.
Cell culture
HeLa cells were grown in DMEM, containing 10% FBS and antibiotics (100 units/mL penicillin and 100 μg/mL streptomycin). All cells were cultivated at 37°C in 5%CO2 atmosphere.
IN VITRO PEROXISOME PROLIFERATOR-ACTIVATED RECEPTORS γ TRANSACTIVATION BIOASSAY
HeLa cells were seeded into 6 well-plates at a density of 2 × 105 cells/well in DMEM containing 10% FBS, 100 units/mL penicillin, and 100 μg/mL streptomycin for 24 h. Then, 0.4 μg GAL4-PPARγ-LBD vector and 1.6 μg of pGL4.35 vector were transiently co-transfections into cells under the condition of FuGENE® HD Transfection reagent. After an overnight culture, the cells were replaced into 96 well-plates with new culture medium. 24 h later, negative control, 0.5 μg/mL rosiglitazone, or test samples were added, respectively. After 24 h, the luciferase activity was detected with Bright-Glo™.
Statistical analysis
Each of these samples was measured in triplicate at various concentrations. Each concentration was tested for sextuplicate. All data are expressed as the mean ± standard deviation. The results were analyzed by one-way, and significant differences were determined by the origin 8.0. Statistical significance is displayed as P < 0.05.
RESULTS
The dose relationship between positive drug and the activation of peroxisome proliferator-activated receptors γ
To select an appropriate concentration of positive drug, the activation of PPARγ by rosiglitazone was tested (concentration from 0.5 μg/mL to 4.0 μg/mL) by PPARγ transactivation bioassay. The fold activation of rosiglitazone in dose-dependence manner is shows in Figure 1. The maximum fold on the activation of PPARγ was 1 μg/mL, and the range of fold activation was from 3.8-to 5.8-folds activation compared to vehicle control (DMSO). On the other hand, there was little change of the fold activation when the concentration of rosiglitazone exceeded 0.5 μg/mL (the range of fold activation was from 3.2 to 5.1 folds activation). Hence, we chose 0.5 μg/mL rosiglitazone as a positive control in later experiments.
Figure 1.
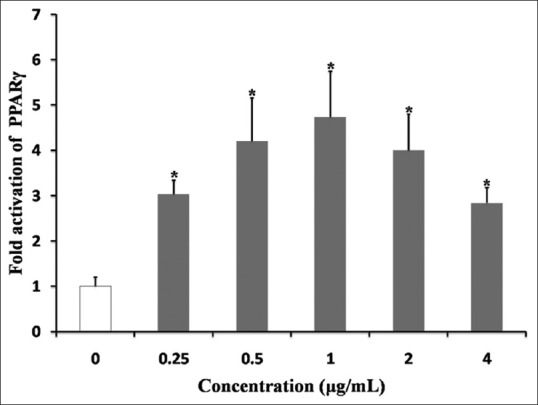
Fold activation of PPARγ by different dosages of rosiglitazone. The data are expressed as the means and standard deviations of three independent experiments with triplicate well. *P < 0.05 vehicle control dimethyl sulfoxide (set to 1.0)
Effects of different polarity extracts on the activation of peroxisome proliferator-activated receptors γ
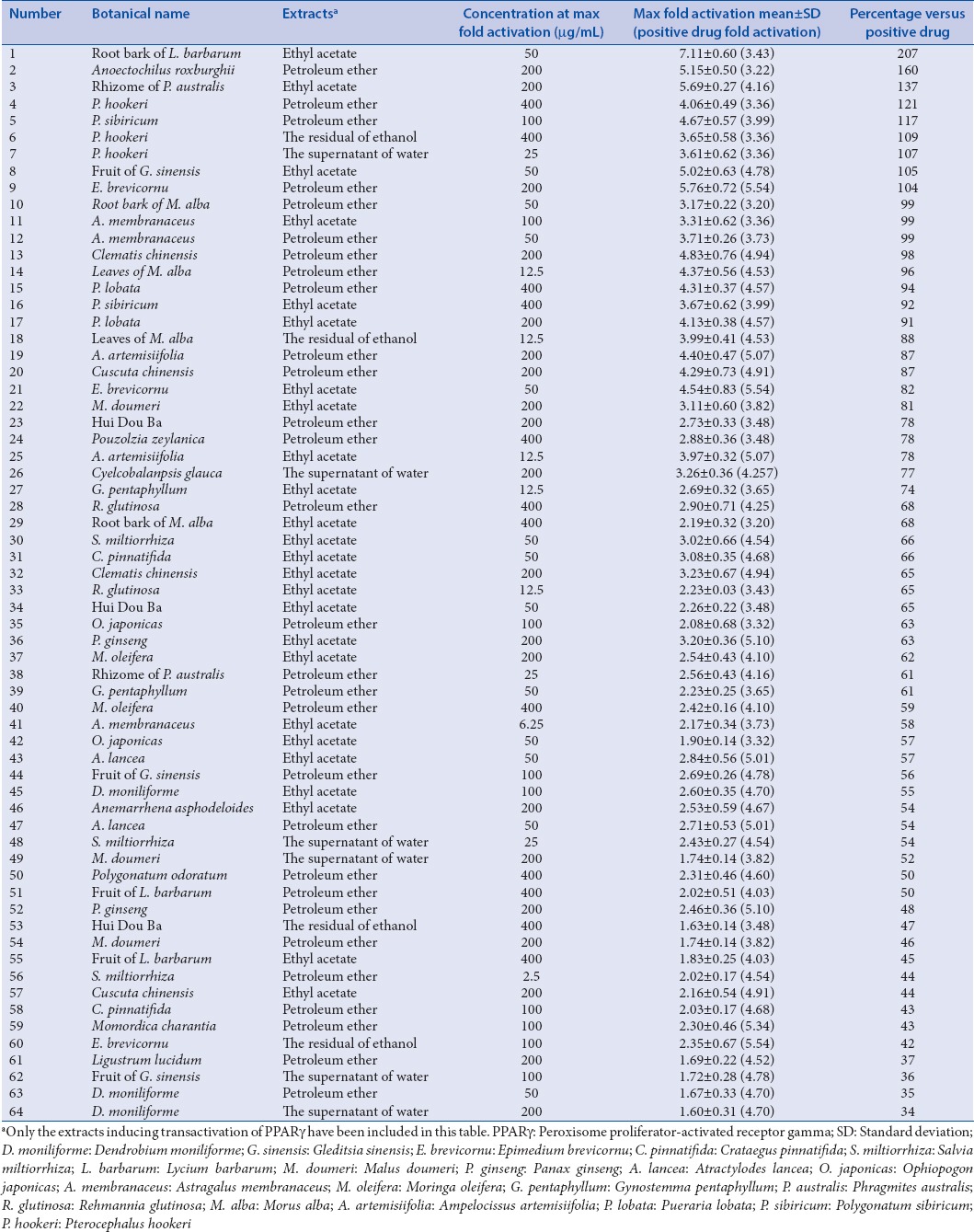
The activation of PPARγ resulted from 185 different kinds of prepared extracts from selected TCMs were tested for six concentrations, respectively. The results [Table 2] indicated that nine extracts resulted in stronger activation on PPARγ than rosiglitazone based on the comparisons of the maximum folds on the activation of PPARγ.
Table 2.
Summary of the results obtained by applying the screening model to 185 different polarity extracts from 37 traditional Chinese medicines
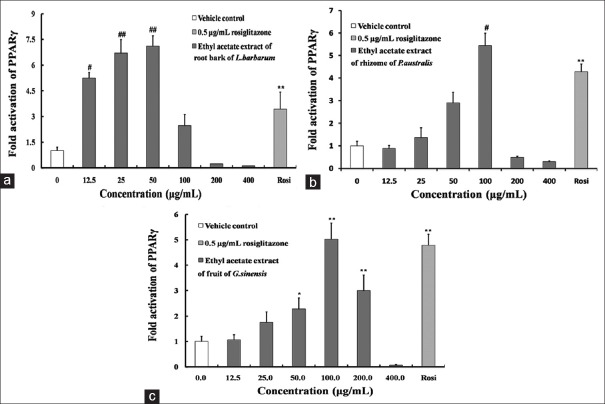
Ethyl acetate extract of root bark of Lycium barbarum had the strongest effect on activating PPARγ in all extracts. The maximum fold was 7.11-fold (50 μg/mL), which equal to 207% of positive drug [Figure 2a]. Besides, ethyl acetate extracts of rhizome of Phragmites australis and fruit of Gleditsia sinensis also could significantly activate PPARγ. The maximum fold on the activation of PPARγ by rhizome of P. saustralis was 5.69 fold, which was 137% of 0.5 μg/mL rosiglitazone [Figure 2b]. The maximum fold activation of the fruit of G. sinensis was 5.02-fold, about 105% that of positive control [Figure 2c].
Figure 2.
Fold activation of PPARγ by ethyl acetate extracts of Root bark of Lycium barbarum, (a) rhizome of Phragmites australis, (b) fruit of Gleditsia sinensis. (c) The data are expressed as the means and standard deviations of three independent experiments with triplicate well. *P < 0.05 versus vehicle control dimethyl sulfoxide, **P < 0.01 versus vehicle control dimethyl sulfoxide (set to 1.0), #P < 0.05 versus 0.5 μg/mL rosiglitazone, ##P < 0.01 versus 0.5 μg/mL rosiglitazone
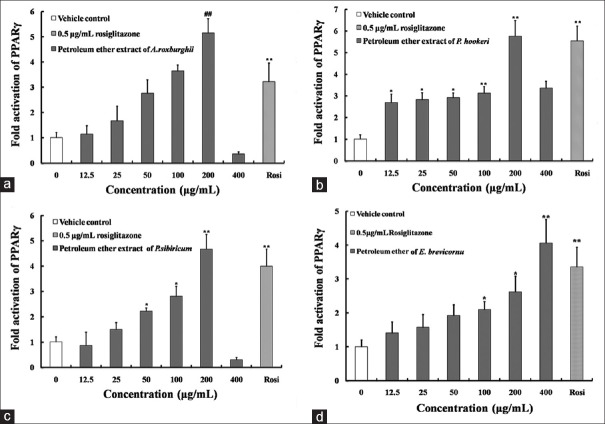
Petroleum ether extracts of Anoectochilusroxburghii, Pterocephalushookeri, Polygonatumsibiricum, Epimediumbrevicornu also can significantly activate PPARγ. The maximum folds on the activation of PPARγ by them were higher than that of positive drug. The maximum fold to activate PPARγ by A. sroxburghii was 5.15 fold, about 160% compared to 0.5 μg/mL rosiglitazone [Figure 3a]. The maximum fold to activate PPARγ by P. hookeri was 4.06 fold, 121% of positive control [Figure 3b]. The maximum folds on the activation of PPARγ by P. sibiricum and E. brevicornu were 4.67 fold and 5.76 fold, which equaled to 117% and 104% of 0.5 μg/mL rosiglitazone [Figure 3c and d].
Figure 3.
Fold activation of peroxisome proliferator-activated receptors-γ by n-hexane extracts of Anoectochilu sroxburghii, (a) Pterocephalus hookeri, (b) Polygonatum sibiricum, (c) Epimedium brevicornu (d). The data are expressed as the means and standard deviations of three independent experiments with triplicate well. *P < 0.05 versus vehicle control dimethyl sulfoxide, **P < 0.01 versus vehicle control dimethyl sulfoxide (set to 1.0), #P < 0.05 versus 0.5 μg/mL rosiglitazone, ##P < 0.01 versus 0.5 μg/mL rosiglitazone
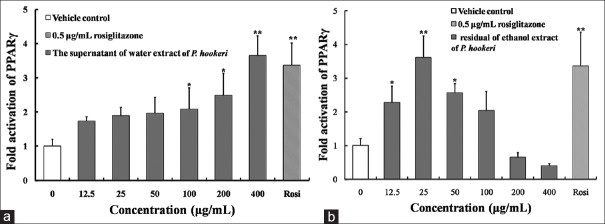
In addition, the residual of ethanol and the supernatant of water extracts of P. hookeri also had remarkable activation effects on PPARγ [Figure 4a and b]. The maximum folds on the activation of PPARγ by them were similar to positive drug. The maximum folds to activate PPARγ by the residual of ethanol and the supernatant of water extracts were 3.65 fold and 3.61 fold, which were 107% and 104% that of positive control, respectively.
Figure 4.
Fold activation of peroxisome proliferator-activated receptors-γ of the supernatant of water extract (a) and residual of ethanol extract (b) of Pterocephalus hookeri. The data are expressed as the means and standard deviations of three independent experiments with triplicate well. *P < 0.05 versus vehicle control dimethyl sulfoxide, **P < 0.01 versus vehicle control dimethyl sulfoxide (set to 1.0)
However, traditionally anti-diabetic medicine including Schisandra chinensis, the fruit of Calendula officinalis, Alisma plantago-aquatica and the root of Trichosanthes kirilowii, had no effects on the activation of PPARγ.
DISCUSSION
Nuclear receptors are large family of receptors which allow an access to the control of gene regulation. The family contains steroid receptors, metabolic receptors, retinoid receptors, Vitamin D receptor and et al.[22,23] They play important roles in normal development, reproduction, and metabolism.[24,25,26] Most nuclear receptors, share a similar basic structure that contains two important domains named LBD and DNA-binding domain. The two domains are crucial for the regulation of the transcription of nuclear receptors. Especially, the LBD fulfills the functions of ligand binding, dimerization, and recruitment of co-regulators.[27,28,29] Ligand binding and activation of nuclear receptor induces a conformational change of it and participates in the regulation of transcription via many approaches such as activation of phosphorylation of the receptor, replacement of corepressors by coactivators and et al.[30,31]
Considering that nuclear receptor family binds to a wide range of lipophilic ligands derived from daily life, targeting nuclear receptors have been a major source for the development of new drugs.[32] It is noteworthy that researchers have found some active compounds from traditional medicines which are the ligands for some of these nuclear receptors.[23,33] For example, honoliol, a compound isolated from magnolia bark, is a partial PPARγ agonist which binds directly to PPARγ LBD.[20] On accounting that honokiol can prevent hyperglycemia and weight gain in mice, it becomes a clinically interesting compound which has opportunity to replace TZDs.
In the past few decades, the prevalence of diabetes mellitus has increased, especially type 2 diabetes. Type 2 diabetes mellitus are closely related to unhealthy diet, sedentary lifestyle, as well as the rise of obesity in the population, which in turn impel the search for new preventive and treatment strategies.[34,35] In recent years, metabolic receptors from nuclear receptor family get more and more attention due to their regulation of metabolic homeostasis.[36,37] It is noteworthy that PPARγ, which belongs to PPAR family, is widely used as an important target for the treatment of diabetes mellitus. Ligand binding of PPARγ can induce the expression of a lot of genes which in turns changes the lipids and glucose metabolism.[38,39] However, common agonists of PPARγ such as TZDs, have many serious side-effects such as cardiovascular risks, liver damage, weight gain, and peripheral edema.[40] Hence, significant research efforts have recently been undertaken to explore the potential drugs of activating PPARγ with less adverse effects.[40,41,42,43] TCMs seem to be an ideal replacement of TZDs for the treatment of hyperglycemia, insulin resistance, and the diabetic complications with less adverse effects.[44,45,46]
Based on the cell-based PPARs screening model.[47] In this study, thirty-seven traditionally used anti-diabetic TCMs were selected and to evaluate their activation activities of PPARγ. The activations of PPARγ-LBD by different polarity extracts from 37 TCMs were evaluated. The results indicated that the ethyl acetate extracts of root bark of L. barbarum, rhizome of P. saustralis and fruit of G. sinensis could significantly activate PPARγ. The activities of them were higher than that of 0.5 μg/mL rosiglitazone. Besides, petroleum ether extracts of A. sroxburghii, P. hookeri, P. sibiricum, E. brevicornu also can significantly activate PPARγ. The activities of them were also higher than that of 0.5 μg/mL rosiglitazone. Furthermore, the residual of ethanol and the supernatant of water extracts of P. hookeri had remarkable activation effects on PPARγ. Thus, three extracts (petroleum ether, the residual of ethanol and the supernatant of water) of P.hookeri were found to have a significant effect on the activation of PPARγ compared to 0.5 μg/mL rosiglitazone. On the other hand, previous studies showed that P. hookeri had good anti-inflammatory effects, and it was used to treat inflammation and analgesic in Tibet of China.[48] Other evidence also demonstrated that the ligands of PPARγ can treat inflammation in the development of diabetes mellitus.[49,50] Therefore, it is speculated that the significant activation of PPARγ by P. hookeri might be related to its remarkable anti-inflammatory activity.
On the contrary, we found that all extracts of S. chinensis, C. officinalis, A. plantago-aquatica, and the root of T. kirilowii could not activate PPARγ. Previous studies showed that extracts of S. chinensis were effective when used as aldosereductase inhibitors for the treatment of diabetes mellitus.[51] The petroleum ether extract of it had PTP1B and alpha-glucosidase inhibitory activities. Moreover, schisandrols A and B, schisandrins A and B from S. chinensis were the ligands of pregnane X receptor, a xenobiotic receptor from nuclear receptor family.[23] Thus, it was speculated that the anti-diabetic effect of S. chinensis was through activating mulitiple targets except for PPARγ. Similar to S. chinensis, the anti-diabetic effects of C. officinalis, A. plantago-aquatica and the root of T. kirilowii were not through activating PPARγ.
Momordicac harantia also named “bitter gourd” had been found to be able to activate PPARα and PPARγ in vitro.[52] In our study, we also found it can activate PPARγ. Furthermore, extracts of Panax ginseng, Salvia miltiorrhiza, and Gynostemma pentaphyllum also can activate PPARγ.[23,53] The results were consistent with the previous study. However, among these chosen TCMs, only a few of them had been studied with regard to the PPARγ activation through a PPARγ transactivation assay in vitro. Our study firstly evaluated the PPARγ activation by different polarity extracts from 37 TCMs with a PPARγ transactivation assay, providing the basis of clarifying the specific mechanism, looking for other targets of activating extracts and screening the active compounds. Further studies will be focused on studying the specific mechanisms of active extracts on activating PPARγ and screening of the active compounds through bio-guided separation.
CONCLUSION
In summary, we have established that petroleum ether extracts of A. sroxburghii, P. hookeri, P. sibiricum, E. brevicornu, ethyl acetate extracts of root bark of L. barbarum, rhizome of P. saustralis and fruit of G. sinensis can significantly activate PPARγ as shown by specific activation of a PPARγ-LBD luciferase receptor assay. Hence, the anti-diabetic activity of them could in part be mediated by this nuclear receptor. Additional research will be necessary to further identify the effects of active extracts such as ethyl acetate extract of root bark of L. barbarum on PPARγ-mediated gene expression and biological responses. Moreover, further studies also will be done to identify the active compounds of PPARγ-LBD.
Financial support and sponsorship
Financial Support of this paper is provided by The National Natural Science Foundation of China (Grant No. 21175159, No. 21275169 and No. 81202886) and The International Cooperation Project of Ministry of Science and Technology of China (Grant No. 2010DFA32680).
Conflicts of interest
There are no conflicts of interest.
ABOUT AUTHOR

Zhining Xia
Zhining Xia, obtained his Ph. D, degree in University of Copenhagen. He is professor in the College of Chemistry and Chemical Engineering Chongqing University. Chongqing, China. His work mainly in: Medicinal plants, capillary electrophoresis, pharmaceutical analysis of Chinese medicines and nano-drug.
REFERENCES
- 1.Janani C, Ranjitha Kumari BD. PPAR gamma gene - A review. Diabetes Metab Syndr Clin Res Rev. 2014;438:1–5. doi: 10.1016/j.dsx.2014.09.015. [DOI] [PubMed] [Google Scholar]
- 2.Zephy D, Ahmad J. Type 2 diabetes mellitus: Role of melatonin and oxidative stress. Diabetes Metab Syndr. 2015;9:127–31. doi: 10.1016/j.dsx.2014.09.018. [DOI] [PubMed] [Google Scholar]
- 3.Wahli W, Michalik L. PPARs at the crossroads of lipid signaling and inflammation. Trends Endocrinol Metab. 2012;23:351–63. doi: 10.1016/j.tem.2012.05.001. [DOI] [PubMed] [Google Scholar]
- 4.Wang Yx. PPARs: Diverse regulators in energy metabolism and metabolic diseases. Cell Res. 2010;20:124–37. doi: 10.1038/cr.2010.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Abbas A, Blandon J, Rude J, Elfar A, Mukherjee D. PPAR-agonist in treatment of diabetes: Cardiovascular safety considerations. Cardiovasc Hematol Agents Med Chem. 2012;10:124–34. doi: 10.2174/187152512800388948. [DOI] [PubMed] [Google Scholar]
- 6.Montagner A, Rando G, Degueurce G, Leuenberger N, Michalik L, Wahli W. New insights into the role of PPARs. Prostaglandins Leukot Essent Fatty Acids. 2011;85:235–43. doi: 10.1016/j.plefa.2011.04.016. [DOI] [PubMed] [Google Scholar]
- 7.Dubois M, Pattou F, Kerr-Conte J, Gmyr V, Vandewalle B, Desreumaux P, et al. Expression of peroxisome proliferator-activated receptor gamma (PPAR gamma) in normal human pancreatic islet cells. Diabetologia. 2000;43:1165–9. doi: 10.1007/s001250051508. [DOI] [PubMed] [Google Scholar]
- 8.Amin RH, Mathews ST, Camp HS, Ding L, Leff T. Selective activation of PPARγ in skeletal muscle induces endogenous production of adiponectin and protects mice from diet-induced insulin resistance. Am J Physiol Endocrinol Metab. 2010;298:E28–37. doi: 10.1152/ajpendo.00446.2009. [DOI] [PubMed] [Google Scholar]
- 9.Grygiel-Gorniak B. Peroxisome proliferator-activated receptors and their ligands: Nutritional and clinical implications – A review. Nut J. 2014;13:17. doi: 10.1186/1475-2891-13-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wang L, Waltenberger B, Pferschy-Wenzig EM, Blunder M, Liu X, Malainer C, et al. Natural product agonists of peroxisome proliferator-activated receptor gamma (PPARgamma): A review. Biochem Pharmacol. 2014;92:73–89. doi: 10.1016/j.bcp.2014.07.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Morales-Garcia JA, Luna-Medina R, Alfaro-Cervello C, Cortes-Canteli M, Santos A, Garcia-Verdugo JM, et al. Peroxisome proliferator-activated receptor?ligands regulate neural stem cell proliferation and differentiation in vitro and in vivo. Glia. 2011;59:293–307. doi: 10.1002/glia.21101. [DOI] [PubMed] [Google Scholar]
- 12.Grimaldi PA. The roles of PPARs in adipocyte differentiation. Prog Lipid Res. 2001;40:269–81. doi: 10.1016/s0163-7827(01)00005-4. [DOI] [PubMed] [Google Scholar]
- 13.Lin CY, Gurlo T, Haataja L, Hsueh WA, Butler PC. Activation of peroxisome proliferator-activated receptor-gamma by rosiglitazone protects human islet cells against human islet amyloid polypeptide toxicity by a phosphatidylinositol 3 ’-kinase-dependent pathway. J Clin Endocrinol Metab. 2005;90:6678–86. doi: 10.1210/jc.2005-0079. [DOI] [PubMed] [Google Scholar]
- 14.Kim HS, Hwang YC, Koo SH, Park KS, Lee MS, Kim KW, et al. PPAR-gamma activation increases insulin secretion through the up-regulation of the free fatty acid receptor GPR40 in pancreatic ß-cells. PLoS One. 2013;8:e50128. doi: 10.1371/journal.pone.0050128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kim HI, Kim JW, Kim SH, Cha JY, Kim KS, Ahn YH. Identification and functional characterization of the peroxisomal proliferator response element in rat GLUT2 promoter. Diabetes. 2000;49:1517–24. doi: 10.2337/diabetes.49.9.1517. [DOI] [PubMed] [Google Scholar]
- 16.Durbin RJ. Thiazolidinedione therapy in the prevention/delay of type 2 diabetes in patients with impaired glucose tolerance and insulin resistance. Diabetes Obes Metab. 2004;6:280–5. doi: 10.1111/j.1462-8902.2004.0348.x. [DOI] [PubMed] [Google Scholar]
- 17.Boyle PJ, King AB, Olansky L, Marchetti A, Lau H, Magar R, et al. Effects of pioglitazone and rosiglitazone on blood lipid levels and glycemic control in patients with type 2 diabetes mellitus: A retrospective review of randomly selected medical records. Clin Ther. 2002;24:378–96. doi: 10.1016/s0149-2918(02)85040-8. [DOI] [PubMed] [Google Scholar]
- 18.Shah P, Mudaliar S. Pioglitazone: Side effect and safety profile. Expert Opin Drug Saf. 2010;9:347–54. doi: 10.1517/14740331003623218. [DOI] [PubMed] [Google Scholar]
- 19.Dunn FL, Higgins LS, Fredrickson J, DePaoli AM. INT- study group. Selective modulation of PPARγ activity can lower plasma glucose without typical thiazolidinedione side-effects in patients with Type 2 diabetes. J Diabetes Complications. 2011;25:151–8. doi: 10.1016/j.jdiacomp.2010.06.006. [DOI] [PubMed] [Google Scholar]
- 20.Atanasov AG, Wang JN, Gu SP, Bu J, Kramer MP, Baumgartner L, et al. Honokiol: A non-adipogenic PPAR gamma agonist from nature. Biochim Biophys Acta. 2013;1830:4813–9. doi: 10.1016/j.bbagen.2013.06.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Xia ZN, Lin YX, Guo LX, Yang FQ, Xu P, Zhang YL, et al. Development of a cell-based high-throughput peroxisome proliferator-activated receptors (PPARs) screening model and its application for evaluation of the extracts from rhizoma Coptis. J Asian Nat Prod Res. 2013;15:225–34. doi: 10.1080/10286020.2012.761977. [DOI] [PubMed] [Google Scholar]
- 22.Cookman CJ, Belcher SM. Classical nuclear hormone receptor activity as a mediator of complex concentration response relationships for endocrine active compounds. Curr Opin Pharmacol. 2014;19:112–9. doi: 10.1016/j.coph.2014.09.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Li L, Bonneton F, Chen XY, Laudet V. Botanical compounds and their regulation of nuclear receptor action: The case of traditional Chinese medicine. Mol Cell Endocrinol. 2015;401:221–37. doi: 10.1016/j.mce.2014.10.028. [DOI] [PubMed] [Google Scholar]
- 24.Gonzalez-Sanchez E, Firrincieli D, Housset C, Chignard N. Nuclear receptors in acute and chronic cholestasis. Dig Dis. 2015;33:357–66. doi: 10.1159/000371688. [DOI] [PubMed] [Google Scholar]
- 25.Moschetta A. Nuclear receptors and cholesterol metabolism in the intestine. Atheroscler Suppl. 2015;17:9–11. doi: 10.1016/S1567-5688(15)50003-2. [DOI] [PubMed] [Google Scholar]
- 26.Polvani S, Tarocchi M, Tempesti S, Galli A. Nuclear receptors and pathogenesis of pancreatic cancer. World J Gastroenterol. 2014;20:12062–81. doi: 10.3748/wjg.v20.i34.12062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Vincent ZA, Aurelien GA, Michielin O. Peroxisome proliferator-activated receptor structures: Ligand specificity, molecular switch and interactions with regulators. Biochim Biophys Acta Mol Cell Biol Lipids. 2007;1771:915–25. doi: 10.1016/j.bbalip.2007.01.007. [DOI] [PubMed] [Google Scholar]
- 28.Kojetin DJ, Burris TP. Small molecule modulation of nuclear receptor conformational dynamics: Implications for function and drug discovery. Mol Pharmacol. 2013;83:1–8. doi: 10.1124/mol.112.079285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rochel N, Moras D. Ligand binding domain of Vitamin D receptors. Curr Top Med Chem. 2006;6:1229–41. doi: 10.2174/156802606777864926. [DOI] [PubMed] [Google Scholar]
- 30.Weatherman RV, Fletterick RJ, Scanlan TS. Nuclear-receptor ligands and ligand-binding domains. Annu Rev Biochem. 1999;68:559–81. doi: 10.1146/annurev.biochem.68.1.559. [DOI] [PubMed] [Google Scholar]
- 31.Feige JN, Gelman L, Michalik L, Desvergne B, Wahli W. From molecular action to physiological outputs: Peroxisome proliferator-activated receptors are nuclear receptors at the crossroads of key cellular functions. Prog Lipid Res. 2006;45:120–59. doi: 10.1016/j.plipres.2005.12.002. [DOI] [PubMed] [Google Scholar]
- 32.Gronemeyer H, Gustafsson JA, Laudet V. Principles for modulation of the nuclear receptor superfamily. Nat Rev Drug Discov. 2004;3:950–64. doi: 10.1038/nrd1551. [DOI] [PubMed] [Google Scholar]
- 33.Guo D, Wang J, Wang X, Luo H, Zhang H, Cao D, et al. Double directional adjusting estrogenic effect of naringin from Rhizoma drynariae (Gusuibu) J Ethnopharmacol. 2011;138:451–7. doi: 10.1016/j.jep.2011.09.034. [DOI] [PubMed] [Google Scholar]
- 34.Pulgaron ER, Delamater AM. Obesity and type 2 diabetes in children: Epidemiology and treatment. Curr Diab Rep. 2014;14:508. doi: 10.1007/s11892-014-0508-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Van Buren DJ, Tibbs TL. Lifestyle interventions to reduce diabetes and cardiovascular disease risk among children. Curr Diab Rep. 2014;14:557. doi: 10.1007/s11892-014-0557-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chawla A, Repa JJ, Evans RM, Mangelsdorf DJ. Nuclear receptors and lipid physiology: Opening the X-files. Science. 2001;294:1866–70. doi: 10.1126/science.294.5548.1866. [DOI] [PubMed] [Google Scholar]
- 37.Senese R, Lasala P, Lseanza C, de Lange P. New avenues for regulation of lipid metabolism by thyroid hormones and analogs. Front Physiol. 2014;5:475. doi: 10.3389/fphys.2014.00475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Houseknecht KL, Cole BM, Steele PJ. Peroxisome proliferator-activated receptor gamma (PPAR gamma) and its ligands: A review. Domest Anim Endocrinol. 2002;22:1–23. doi: 10.1016/s0739-7240(01)00117-5. [DOI] [PubMed] [Google Scholar]
- 39.Kliewer SA, Xu HE, Lambert MH, Willson TM. Peroxisome proliferator-activated receptors: From genes to physiology. Recent Prog Horm Res. 2001;56:239–63. doi: 10.1210/rp.56.1.239. [DOI] [PubMed] [Google Scholar]
- 40.Kung J, Henry RR. Thiazolidinedione safety. Expert Opin Drug Saf. 2012;11:565–79. doi: 10.1517/14740338.2012.691963. [DOI] [PubMed] [Google Scholar]
- 41.Rubenstrunk A, Hanf R, Hum DW, Fruchart JC, Staels B. Safety issues and prospects for future generations of PPAR modulators. Biochim Biophys Acta. 2007;1771:1065–81. doi: 10.1016/j.bbalip.2007.02.003. [DOI] [PubMed] [Google Scholar]
- 42.Sood V, Colleran K, Burge MR. Thiazolidinediones: A comparative review of approved uses. Diabetes Technol Ther. 2000;2:429–40. doi: 10.1089/15209150050194297. [DOI] [PubMed] [Google Scholar]
- 43.Higgins LS, DePaoli AM. Selective peroxisome proliferator-activated receptor gamma (PPAR gamma) modulation as a strategy for safer therapeutic PPAR gamma activation. Am J Clin Nutr. 2010;91:267S–72S. doi: 10.3945/ajcn.2009.28449E. [DOI] [PubMed] [Google Scholar]
- 44.Gupta RK, Kesari AN, Murthy PS, Chandra R, Tandon V, Watal G. Hypoglycemic and antidiabetic effect of ethanolic extract of leaves of Annona squamosa L. in experimental animals. J Ethnopharmacol. 2005;99:75–81. doi: 10.1016/j.jep.2005.01.048. [DOI] [PubMed] [Google Scholar]
- 45.Modak M, Dixit P, Londhe J, Ghaskadbi S, Devasagayam TP. Indian herbs and herbal drugs used for the treatment of diabetes. J Clin Biochem Nutr. 2007;40:163–73. doi: 10.3164/jcbn.40.163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Malviya N, Jain S, Malviya S. Antidiabetic potential of medicinal plants. Acta Pol Pharm. 2010;67:113–8. [PubMed] [Google Scholar]
- 47.Xia ZN, Lin YX, Guo LX, Yang FQ, Xu P, Zhang YL, et al. Development of a cell-based high-throughput peroxisome proliferator-activated receptors (PPARs) screening model and its application for evaluation of the extracts from rhizoma Coptis. J Asian Nat Prod Res. 2013;15:225–34. doi: 10.1080/10286020.2012.761977. [DOI] [PubMed] [Google Scholar]
- 48.Zhang L, Hu JJ, Lin JW, Fang WS, Du GH. Anti-inflammatory and analgesic effects of ethanol and aqueous extracts of Pterocephalus hookeri (C.B Clarke) H#eck. J Ethnopharmacol. 2009;123:510–4. doi: 10.1016/j.jep.2009.01.039. [DOI] [PubMed] [Google Scholar]
- 49.Donath MY, Shoelson SE. Type 2 diabetes as an inflammatory disease. Nat Rev Immunol. 2011;11:98–107. doi: 10.1038/nri2925. [DOI] [PubMed] [Google Scholar]
- 50.Varga T, Czimmerer Z, Nagy L. PPARs are a unique set of fatty acid regulated transcription factors controlling both lipid metabolism and inflammation. Biochim Biophys Acta. 2011;1812:1007–22. doi: 10.1016/j.bbadis.2011.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Li WL, Zheng HC, Bukuru J, De Kimpe N. Natural medicines used in the traditional Chinese medical system for therapy of diabetes mellitus. J Ethnopharmacol. 2004;92:1–21. doi: 10.1016/j.jep.2003.12.031. [DOI] [PubMed] [Google Scholar]
- 52.Chao CY, Huang CJ. Bitter gourd (Momordica charantia) extract activates peroxisome proliferator-activated receptors and upregulates the expression of the acyl CoA oxidase gene in H4IIEC3 hepatoma cells. J Biomed Sci. 2003;10(6 Pt 2):782–91. doi: 10.1159/000073966. [DOI] [PubMed] [Google Scholar]
- 53.Gong Z, Huang C, Sheng X, Zhang Y, Li Q, Wang MW, et al. The role of tanshinone IIA in the treatment of obesity through peroxisome proliferator-activated receptor gamma antagonism. Endocrinology. 2009;150:104–13. doi: 10.1210/en.2008-0322. [DOI] [PubMed] [Google Scholar]