Abstract
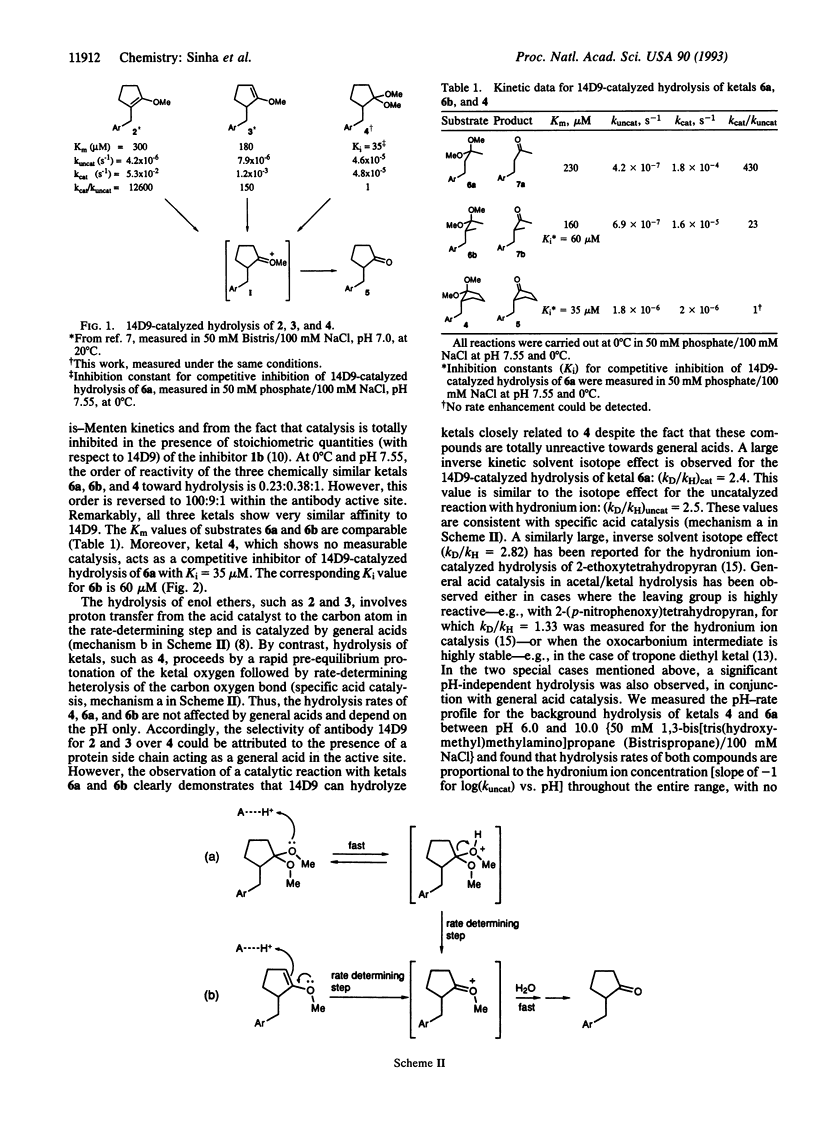
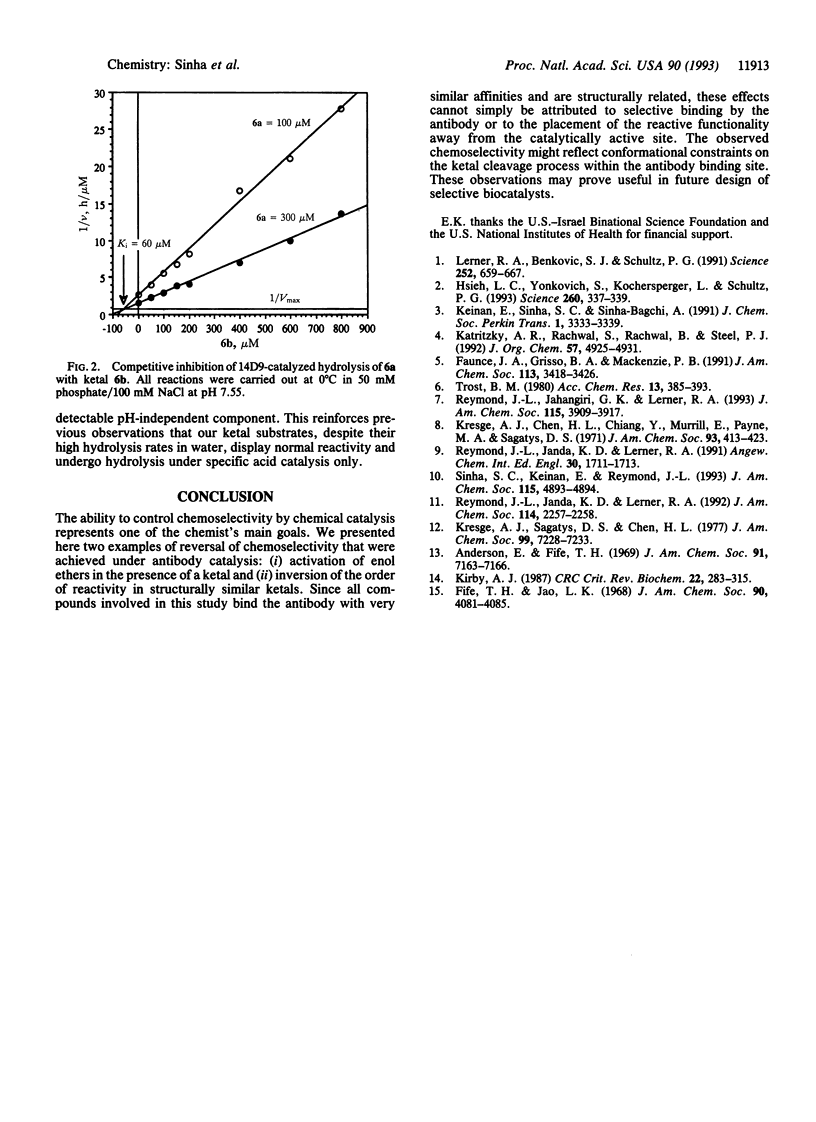
A monoclonal antibody, 14D9, which has been elicited against a cationic hapten, N-alkyl-N-methyl-3-glutarylamidomethyl piperidinium, in which alkyl = [4-(2-hydroxyethylamido)carbonyl]phenylmethyl, is capable of inverting the intrinsic order of reactivity in a series of structurally related enol ethers and ketals towards hydrolysis. The order of reactivity of compounds 2 (1-methoxy-2-alkylcyclopent-1-ene), 3 (1-methoxy-5-alkylcylopent-1-ene), and 4 (1,1-dimethoxy-2-alkylcyclopentane) has changed from 0.09:0.17:1 in the uncatalyzed reaction to 1100:25:1 under antibody catalysis. Also, the order of reactivity of the three chemically similar ketals, 6a (1-alkyl-2,2-dimethoxypropane), 6b (1-alkyl-1-methyl-2,2-dimethoxypropane), and 4, has changed from 0.23:0.38:1 in the uncatalyzed hydrolysis to 100:9:1 within the antibody active site. As all compounds bind the antibody with very similar affinities, these effects cannot be simply attributed to selective binding by the antibody. In fact, ketal 4, which shows no measurable catalysis, acts as a competitive inhibitor of 14D9-catalyzed hydrolysis of 6a. Both the solution and the antibody-catalyzed hydrolysis of the ketal substrates are shown to be specific acid catalyzed, involving the unimolecular cleavage of the protonated substrate or antibody-substrate complex in the rate-determining step. Reactivity effects from the acid catalyst itself on ketal hydrolysis (reagent-controlled reactivity) are ruled out under this mechanistic scheme.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Hsieh L. C., Yonkovich S., Kochersperger L., Schultz P. G. Controlling chemical reactivity with antibodies. Science. 1993 Apr 16;260(5106):337–339. doi: 10.1126/science.10049109. [DOI] [PubMed] [Google Scholar]
- Kirby A. J. Mechanism and stereoelectronic effects in the lysozyme reaction. CRC Crit Rev Biochem. 1987;22(4):283–315. doi: 10.3109/10409238709086959. [DOI] [PubMed] [Google Scholar]
- Lerner R. A., Benkovic S. J., Schultz P. G. At the crossroads of chemistry and immunology: catalytic antibodies. Science. 1991 May 3;252(5006):659–667. doi: 10.1126/science.2024118. [DOI] [PubMed] [Google Scholar]



