Abstract
Background
Chronic treatment with the dietary flavonoid quercetin is known to lower blood pressure and restore endothelial dysfunction in animal models of hypertension. This study investigated the direct effects of quercetin on vascular response in chronic 2-kidney, 1-clip (2K1C) renal hypertensive rats. The effects of antioxidant vitamin ascorbic acid on the vasoreactivity were also examined.
Methods
2K1C renal hypertension was induced by clipping the left renal artery; age-matched rats that received sham treatment served as controls. Thoracic aortae were mounted in tissue baths for the measurement of isometric tension.
Results
Relaxant responses to acetylcholine were significantly attenuated in 2K1C rats in comparison with sham rats. Quercetin or ascorbic acid augmented acetylcholine-induced relaxation in 2K1C rats, whereas no significant differences were noted in sham rats. The relaxation response to sodium nitroprusside was comparable between 2K1C and sham rats, and sodium nitroprusside–induced relaxation was not altered by quercetin or ascorbic acid in either group. The contractile response to phenylephrine was significantly enhanced in 2K1C rats compared with sham rats. Phenylephrine-induced contraction was inhibited by pretreatment with quercetin or ascorbic acid in 2K1C rats, whereas neither chemical affected responses in sham rats. Nw-nitro-L-arginine methyl ester markedly augmented the contractile response to phenylephrine in sham rats, whereas no significant differences were observed in 2K1C rats. Quercetin or ascorbic acid did not affect phenylephrine-induced contraction in the presence of Nw-nitro-L-arginine methyl ester in either 2K1C or sham rats.
Conclusion
Acute exposure to quercetin appears to improve endothelium-dependent relaxation and inhibit the contractile response, similar to the effect of ascorbic acid in 2K1C hypertension. These results partially explain the vascular beneficial effects of quercetin in renal hypertension.
Keywords: Antioxidant, Quercetin, Renal hypertension
Introduction
Hypertension, a typical risk factor of cardiovascular disease, involves endothelial dysfunction in large conduit and small resistance arteries [1]. Indeed, impaired endothelium-dependent vasodilation was observed in a number of experimental models of hypertension, including 2-kidney, 1-clip (2K1C) renal hypertension [2], [3], [4]. The biological activity of nitric oxide (NO), an effective endothelium-derived relaxing factor, is primarily associated with endothelial NO synthase activity or its interaction with the superoxide anion, which is produced in the vascular wall by free radical–generating enzymes [5]. Endothelium-derived NO may be scavenged by the superoxide anion, causing reduced bioavailability of NO and diminishing vasorelaxation [6]. It is now well established that endothelial dysfunction in hypertension is partially linked to the exaggerated production of superoxide anions [7], [8] and that oxidative stress is responsible for impaired endothelial modulation in 2K1C hypertension [9], [10]. Therefore, antioxidants may exert beneficial effects on the vascular complications associated with hypertension.
Quercetin is a commonly found flavonol-type flavonoid that is widely distributed in dietary vegetables, fruits, and wine [11]. Previous studies have demonstrated that the flavonoid quercetin has various physiological effects including antioxidant and antihypertensive effects as well as those associated with improvements in vascular reactivity [12], [13]. The vascular beneficial effects induced by quercetin are mainly due to its antioxidant properties, which might interact with the endothelium-derived NO system [14]. Accumulated evidence demonstrates that flavonoids increase the biological activity of NO by interacting with superoxide anions [15], and chronic oral treatment with quercetin in experimental hypertension was shown to protect against impairments in vascular endothelial function [16]. Apart from the chronic effects of flavonoids associated with endothelial dysfunction in hypertension, acute exposure to quercetin in vitro restores endothelium-dependent relaxation and inhibits the contractile responses to the agonist in genetically hypertensive rats [11], [17]. For this point of view, although it has been demonstrated that chronic treatment with quercetin exerts antihypertensive and antioxidant effects and improves endothelial function in renovascular hypertension [18], the acute effects of quercetin on vascular function in renal hypertension remain unclear.
The present study aimed to examine the direct effects of quercetin on vascular reactivity in chronic 2K1C hypertensive rats. The effects of quercetin on endothelium-dependent or endothelium-independent relaxation in response to acetylcholine or sodium nitroprusside (SNP) and the contractile responses to the α1-adrenergic agonist phenylephrine were investigated in isolated aortae from 2K1C hypertensive and sham-clipped control rats. The effects of the antioxidant vitamin ascorbic acid (vitamin C) on vascular reactivity were also examined.
Methods
Induction of 2K1C renal hypertension
Male Sprague-Dawley rats (Samtaco Inc., Osan, Korea), weighing 160–180 g, were anesthetized with intraperitoneal injection of sodium thiopental (40 mg/kg). Under antiseptic conditions, the left posterior side of the animal was shaved and sterilized with 70% ethanol. An incision was made on the left flank to provide access to the left renal artery, which was separated from the renal vein and cleaned of connective tissue. A silver clip with an internal diameter of 0.2 mm was applied on the exposed renal artery. The clip was then turned so that the slit opening faces the abdomen, resulting in partial occlusion of renal perfusion. The contralateral kidney was not disturbed. The muscles and skin were sutured immediately, and the rats were allowed to recover from anesthesia. Control rats received a sham treatment: they underwent the same surgical procedure as 2K1C rats, except for clip placement. All rats were maintained on standard chow with free access to drinking water. They were killed for examination at 10 weeks after clipping because endothelial dysfunction is associated with the duration of hypertension [19]. Hypertensive rats were selected by measuring systolic blood pressure in the conscious state using the tail cuff method; they were considered hypertensive when the systolic pressure was over 160 mmHg.
Tissue preparation
At the time of experimentation, the descending thoracic aorta between the aortic arch and diaphragm was excised through a ventral incision and placed in cold, standard physiological salt solution of the following composition (in mM): NaCl 118.3, KCl 4.7, NaHCO3 25.0, MgCl2 1.2, KH2PO4 1.2, CaCl2 2.5, and glucose 11.1. Vessels were cleaned of adherent fat and connective tissues and sectioned into cylindrical rings (2–3 mm in width) under a dissecting microscope. Care was taken not to stretch the artery or dislodge the vascular endothelium.
The aortic rings were mounted between two triangle-shaped stainless steel holders in the vessel lumen in organ chambers containing 15 mL of physiological salt solution maintained at 37 ± 0.05°C, aerated with a mixture of 95% oxygen and 5% carbon dioxide to maintain a pH of 7.4 ± 0.01 throughout the experiment. One of the holders was fixed at the bottom of the chambers and the other was connected to a force displacement transducer (Grass FTO3, Quincy, Mass, USA) to measure isometric tension development. Before initiating specific experimental protocols, aortic rings were stretched to the point of their optimal length–tension relationship at 2 g, determined in similar preliminary experiments using repeated exposure to 60mM KCl solution (obtained by equimolar replacement of NaCl by KCl in the physiological solution), and allowed to equilibrate for a period of at least 90 minutes. During this period of stabilization, the bath solution was replaced every 15 minutes. After an equilibration period, aortic rings were stimulated with 60mM KCl to test their functional viability. All experimental procedures were performed in the presence of indomethacin (10–5M).
Protocols
Endothelium-dependent and endothelium-independent relaxation in response to acetylcholine and SNP were examined using aortic rings from 2K1C and sham-operated rats under submaximally precontracted with phenylephrine (2 × 10–7M in sham and 3 × 10–8M in 2K1C), based on doses obtained in preliminary experiments. The responses of the aortic rings to the cumulative addition of acetylcholine (from 10–9 to 10–5M) and SNP (from 10–10 to 10–6.5M) were examined in parallel rings, which had been incubated with the vehicle, quercetin (10–5M) [11], [17] or ascorbic acid (10–5M) 20 minutes before the addition of phenylephrine.
In another set of experiments, aortic rings from 2K1C and sham rats were pretreated with the vehicle, quercetin (10–5M) or ascorbic acid (10–5M) for 20 minutes; then, the contractile responses to the cumulative addition of phenylephrine (from 10–9 to 10–5M) were recorded. The experiments were repeated in the presence and absence of the NO synthase inhibitor Nw-nitro-L-arginine methyl ester (L-NAME, 10–4M).
Drugs and chemicals
The drugs used in this study comprised acetylcholine, ascorbic acid, indomethacin, L-NAME, phenylephrine, quercetin, and SNP. They were purchased from Sigma Chemical Co. (St. Louis, MO, USA). All other chemicals were of reagent grade. Indomethacin and quercetin were dissolved in dimethylsulfoxide, while the others were prepared in distilled water. Final bath concentrations of dimethylsulfoxide were less than 0.05%, which did not alter the contraction or relaxation responses.
Statistical analysis
Values presented in the figures are expressed as the means and standard errors of the means for the number of rats or tissues indicated in legends. The relaxant responses are given as the percent change in phenylephrine-induced contractile tension; the contractile responses are expressed as a percentage of the contraction developed by exposure to 60mM KCl. Statistical comparisons were performed by Student's t test or analysis of variance with repeated measurements and Fisher's post hoc test. Differences were considered statistically significant when P < 0.05.
Results
The systolic blood pressures were significantly increased at 2 weeks after surgery in 2K1C hypertensive rats as compared with sham-clipped control rats (Table 1). The magnitude of KCl (60mM)–induced isometric tension development was comparable between the two groups (1.45 ± 0.12 g in control; 1.52 ± 0.14 g in 2K1C).
Table 1.
Changes in systolic blood pressure (mmHg) during 10 weeks in sham-clipped and 2K1C rats
| Weeks |
||||||
|---|---|---|---|---|---|---|
| 0 | 2 | 4 | 6 | 8 | 10 | |
| Sham (n = 43) | 132 (3.7) | 137 (3.2) | 136 (3.3) | 134 (3.0) | 136 (3.9) | 135 (3.0) |
| 2K1C (n = 42) | 134 (3.9) | 148 (3.4)∗ | 175 (3.5)∗ | 183 (4.1)∗ | 187 (3.8)∗ | 192 (3.0)∗ |
Results are means (SEs) for n experiments.
2K1C, 2-kidney, 1-clip; SE, standard error.
P < 0.05, compared with sham values.
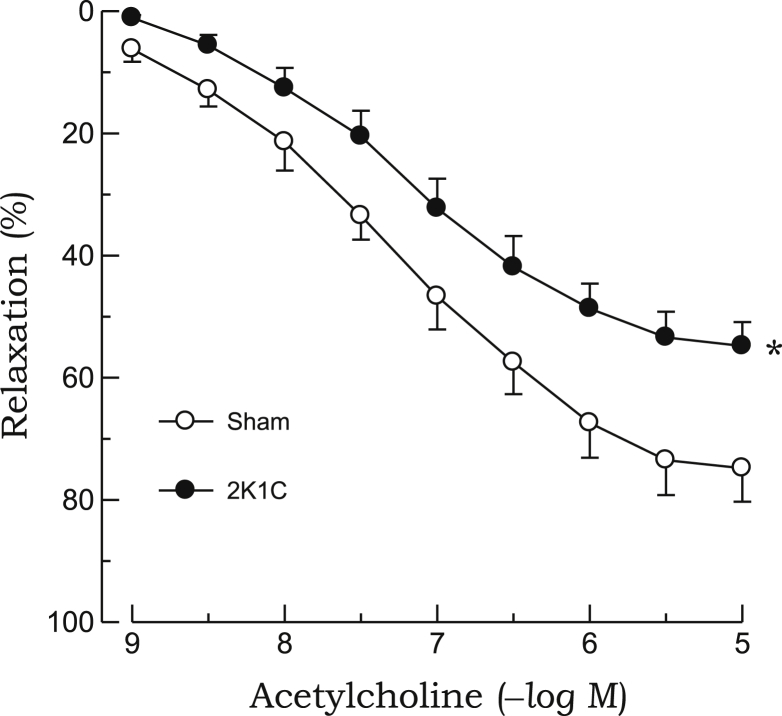
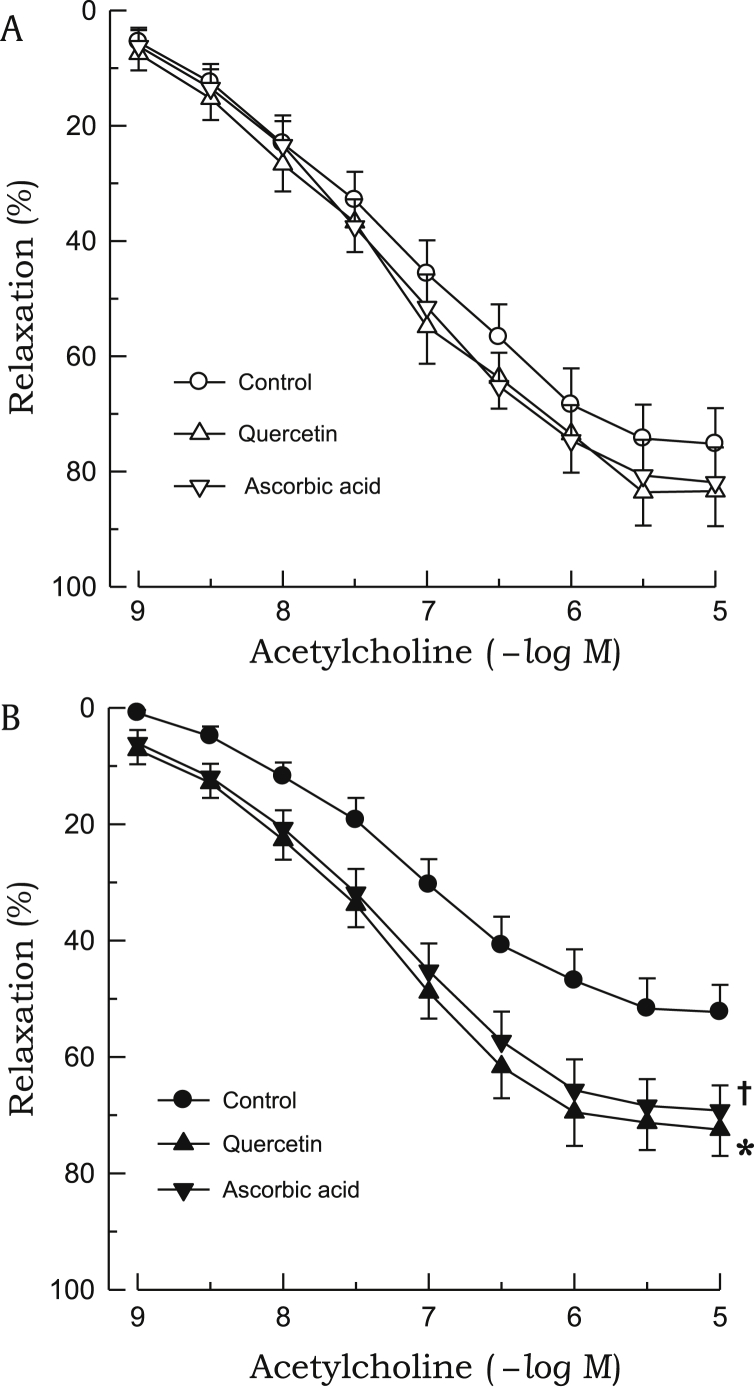
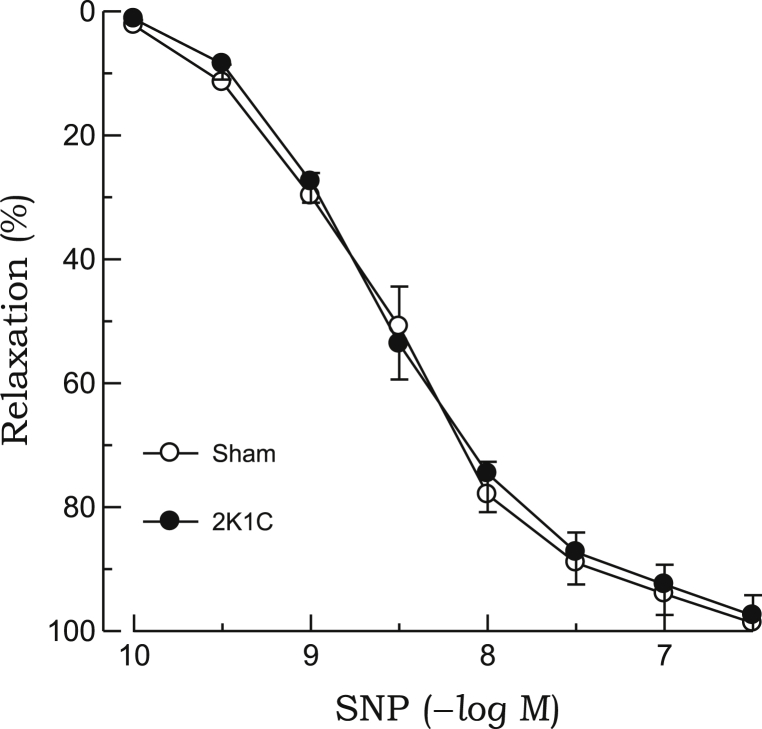
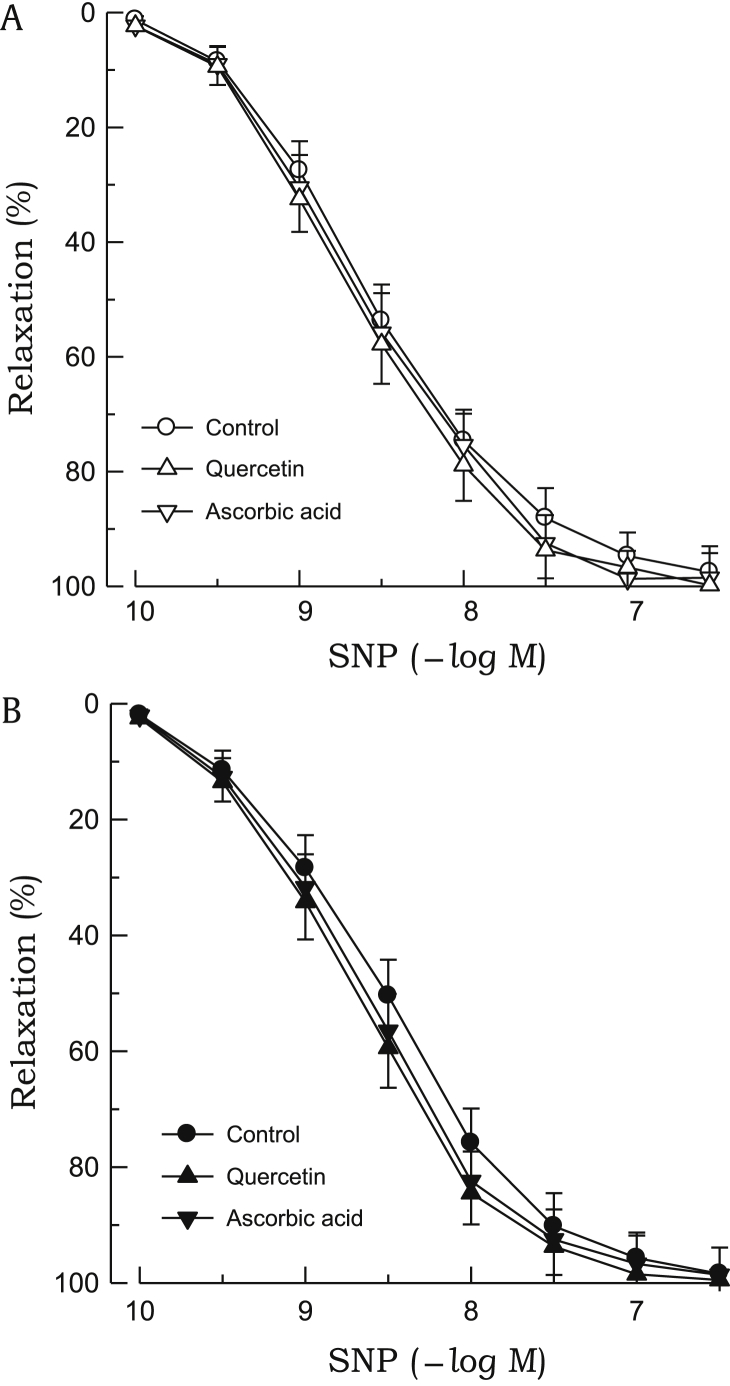
The relaxant response to acetylcholine in the aortic rings precontracted with phenylephrine was significantly attenuated in 2K1C rats as compared with sham rats (Fig. 1). Treatment with L-NAME (10–4M) completely blocked the acetylcholine-induced relaxation in the 2K1C and sham groups (data not shown). In aortic rings from 2K1C rats, impaired vasodilatory response to acetylcholine was improved by preincubation with quercetin or ascorbic acid. However, both chemicals did not affect the relaxant response to acetylcholine in sham rats (Fig. 2). The relaxant response to SNP was not altered in 2K1C rats (Fig. 3). Quercetin or ascorbic acid did not affect the SNP-induced relaxation in either 2K1C or sham rats (Fig. 4).
Figure 1.
Relaxant responses to acetylcholine in phenylephrine-precontracted aortic rings from 2K1C hypertensive and sham-clipped control rats. Points represent means ± SEs for 6–8 experiments.
∗P < 0.05, compared with sham values.2K1C, 2-kidney, 1-clip; SE, standard error.
Figure 2.
Effects of quercetin or ascorbic acid on acetylcholine-induced relaxations. The aortic rings used were from sham-clipped control (A) and 2K1C hypertensive rats (B). Points represent means ± SEs for 6–8 experiments.
∗,✝P < 0.05, compared with corresponding control values.2K1C, 2-kidney, 1-clip; SE, standard error.
Figure 3.
Relaxant responses to SNP in phenylephrine-precontracted aortic rings from 2K1C hypertensive and sham-clipped control rats. Points represent means ± SEs for 6–8 experiments.2K1C, 2-kidney, 1-clip; SE, standard error; SNP, sodium nitroprusside.
Figure 4.
Effects of quercetin or ascorbic acid on SNP-induced relaxations. The aortic rings used were from sham-clipped control (A) and 2K1C hypertensive rats (B). Points represent means ± SEs for 6–8 experiments.2K1C, 2-kidney, 1-clip; SE, standard error; SNP, sodium nitroprusside.
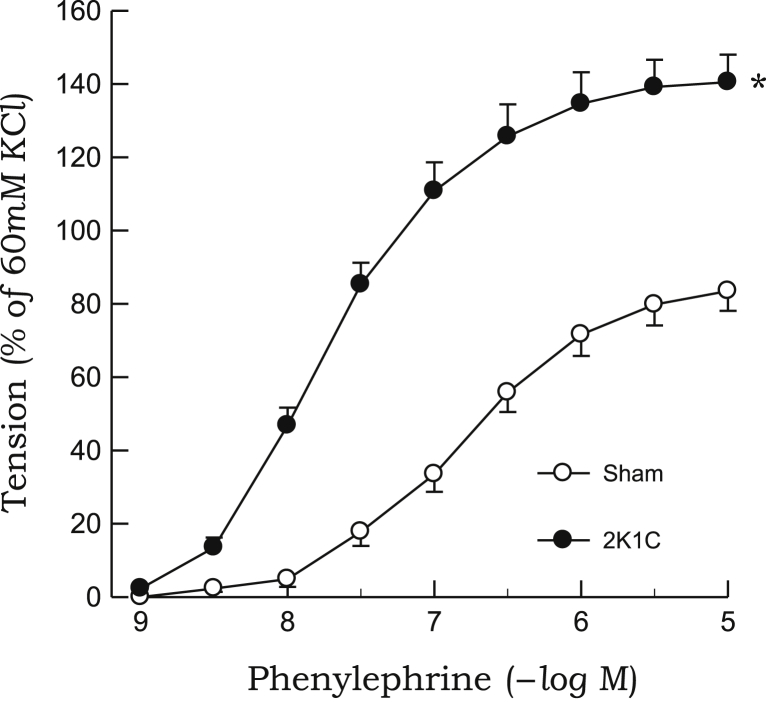
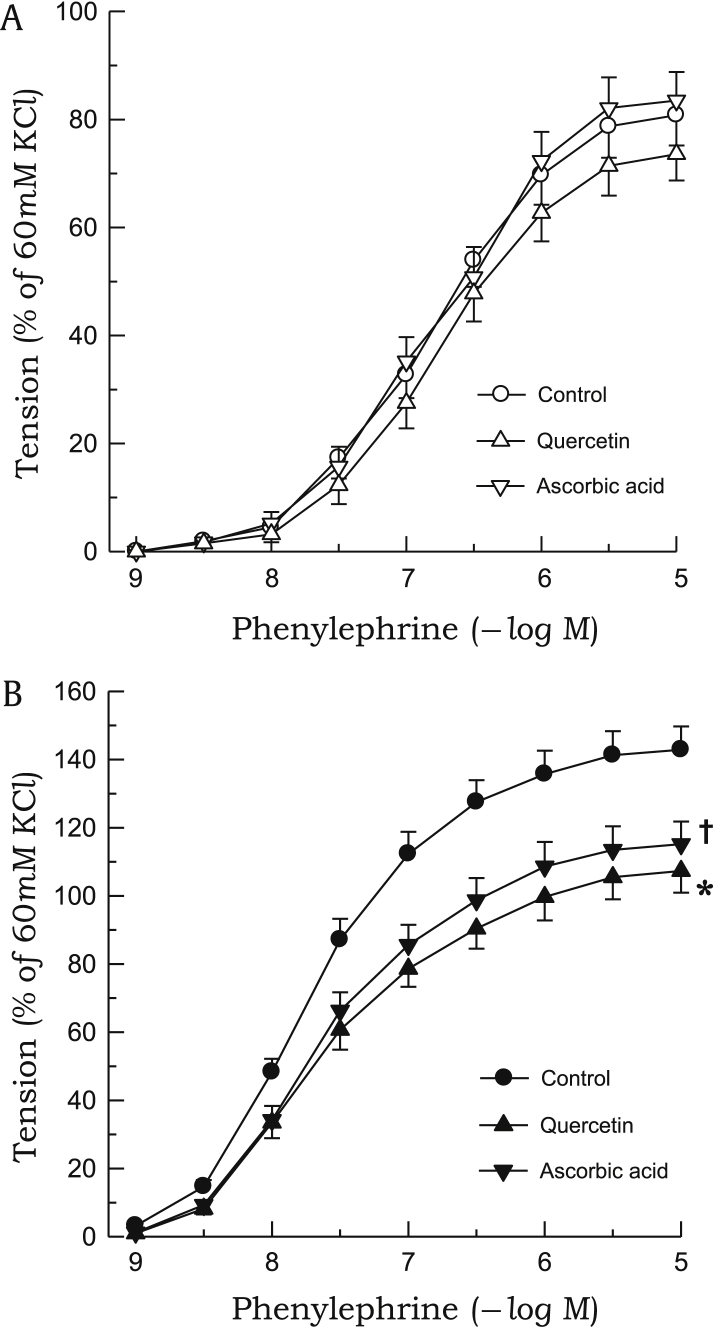
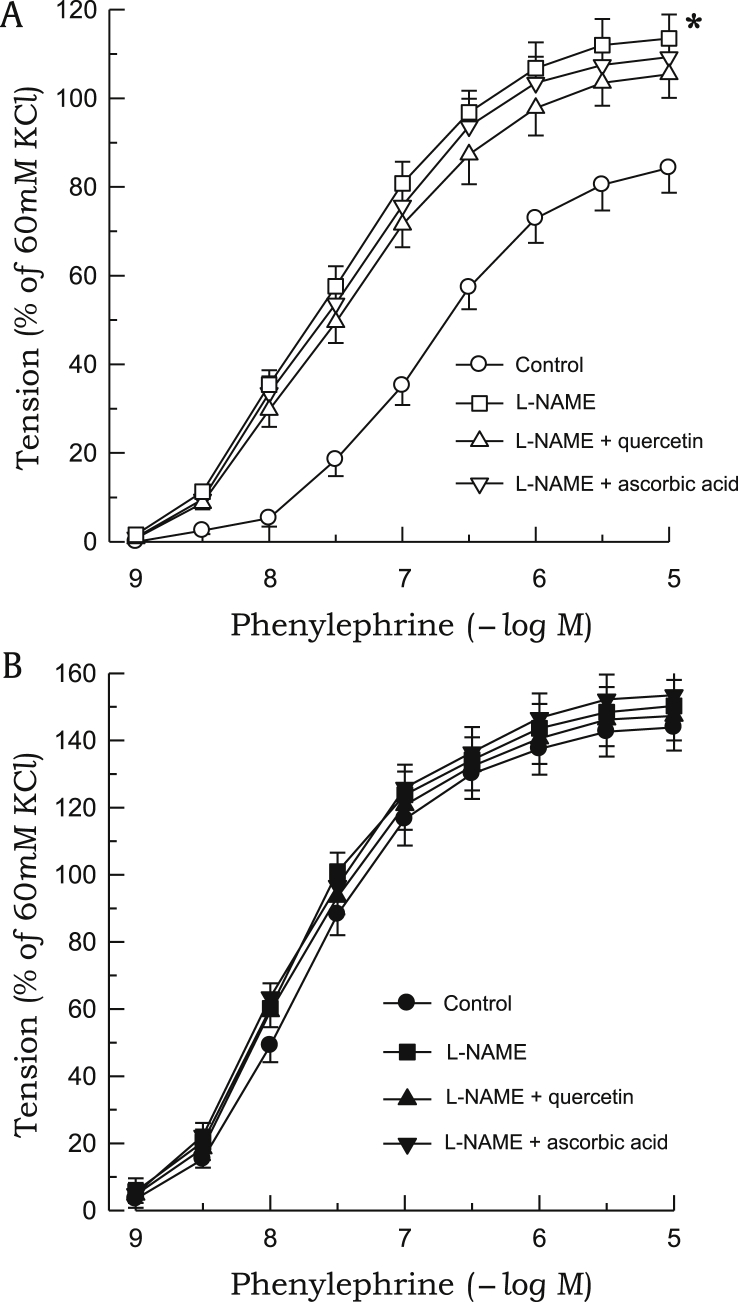
The contractile response to phenylephrine was significantly enhanced in aortic rings from 2K1C rats as compared with those from sham rats (Fig. 5). Phenylephrine-induced contraction was inhibited by pretreatment with quercetin or ascorbic acid in 2K1C rats, whereas neither compounds affected the contractile response to phenylephrine in sham rats (Fig. 6). L-NAME markedly augmented the contractile response to phenylephrine in aortic rings from sham rats, whereas no significant differences were noted in 2K1C rats. In the presence of L-NAME, neither quercetin nor ascorbic acid affected phenylephrine-induced contraction in either 2K1C or sham rats (Fig. 7). Quercetin or ascorbic acid did not affect L-NAME alone–induced vascular response.
Figure 5.
Contractile responses to phenylephrine in aortic rings from 2K1C hypertensive and sham-clipped control rats. Points represent means ± SEs for 6–8 experiments.
∗P < 0.05, compared with sham values.2K1C, 2-kidney, 1-clip; SE, standard error.
Figure 6.
Effects of quercetin or ascorbic acid on phenylephrine-induced contractions. The aortic rings used were from sham-clipped control (A) and 2K1C hypertensive rats (B). Points represent means ± SEs for 6–8 experiments.
∗,✝P < 0.05, compared with corresponding control values.
2K1C, 2-kidney, 1-clip; SE, standard error.
Figure 7.
Effects of L-NAME on phenylephrine-induced contractions. The aortic rings used were from sham-clipped control (A) and 2K1C hypertensive rats (B). Effects of quercetin or ascorbic acid on phenylephrine-induced contractions in the presence of L-NAME are also shown. Points represent means ± SEs for 6–8 experiments.
∗P < 0.05, compared with corresponding control values.2K1C, 2-kidney, 1-clip; L-NAME, Nw-nitro-L-arginine methyl ester; SE, standard error.
Discussion
Dysfunctional endothelial modulation is commonly characterized by an alteration in the vasodilatory capacity of the vascular endothelium and plays a pivotal role in the pathophysiology of hypertension [20]. In the present study, endothelium-dependent relaxation in response to acetylcholine was markedly attenuated in 2K1C renal hypertensive rats as compared with that in sham-clipped control rats. Similar results were also demonstrated recently in our previous report [21]. The acetylcholine-induced vasodilatory effects were completely abolished in both 2K1C and sham rats by treatment with L-NAME, which is an NO synthase inhibitor, suggesting that acetylcholine-induced vasorelaxation mainly contributes to NO synthase–derived NO. As observed previously in genetically hypertensive rats [17], in this study, endothelium-dependent relaxation in response to acetylcholine was augmented by preincubation with quercetin or ascorbic acid in 2K1C rats, whereas the two chemicals did not affect the relaxant response to acetylcholine in sham rats. These results indicate that endothelium-derived NO-mediated relaxation responses induced by acetylcholine are promoted by quercetin and ascorbic acid in 2K1C hypertensive aortae, and such mechanisms are absent in normotensive tissues. In association with these observations, previous studies [22], [23] have proposed that an excessive production of endothelium-derived factors such as vasoconstrictor prostanoids or superoxide anions may take part in blunted acetylcholine-induced vasorelaxation in hypertensive arteries. However, the participation of vasoconstrictor prostanoids in acetylcholine-induced relaxation may be ruled out in 2K1C rats in this study because all the experimental procedures were performed in the presence of indomethacin, a cyclooxygenase inhibitor. Therefore, superoxide anions may be a possible candidate responsible for impairments in endothelium-dependent relaxation in hypertensive aortae. It may be postulated that the impaired relaxation in response to acetylcholine observed in the 2K1C aortae is caused by the harmful effects of superoxide anions on endothelium-derived NO. It has been demonstrated that NO can be scavenged by superoxide anions to form peroxynitrite (ONOO–) [24], thus reducing the bioavailability of endothelium-derived NO. Taken together, as previously observed in genetic hypertension [17], the enhanced acetylcholine-induced relaxation in 2K1C rats may be linked to superoxide anion scavenging properties by flavonoid quercetin or antioxidant vitamin.
The present study confirmed the earlier observation [21] that endothelium-independent relaxation in response to SNP, which is an NO donor, is comparable between aortic rings from 2K1C and sham rats. Quercetin or ascorbic acid did not affect SNP-induced relaxation in either 2K1C or sham rats, suggesting that NO-mediated vascular relaxation in hypertensive and normotensive rats is not altered by quercetin or ascorbic acid. It has been reported that flavonoids primarily exert their beneficial vascular effects by accumulating on the surfaces between endothelial and vascular smooth muscle cells and improving NO bioavailability by either enhancing endothelium-derived NO production or inhibiting its destruction by free radicals [15]. Thus, it may be hypothesized that the effects of quercetin or ascorbic acid are masked on which SNP induced fully NO-mediated vasorelaxation, as shown previously in genetically hypertensive rats [17].
It has been demonstrated that vascular reactivity to a contractile agonist is enhanced in disease states such as hypertension [25]. In accordance with our previous findings [26], the contractile response to phenylephrine was augmented in the aortic rings from 2K1C hypertensive rats as compared with those from sham-clipped control rats. In the present study, quercetin and ascorbic acid inhibited phenylephrine-induced contraction in the aortic rings from 2K1C rats, whereas these compounds had no effect on the aortic rings from sham rats. These results led us to hypothesize that improved biological activity of endothelium-derived NO may be responsible for reduction in phenylephrine-induced contraction by quercetin or ascorbic acid in 2K1C rats. To investigate this issue, the effects of quercetin or ascorbic acid on the contractile response to phenylephrine were examined in the presence of L-NAME. The contractile response to phenylephrine was markedly enhanced by L-NAME treatment in sham rats, whereas phenylephrine-induced contraction was comparable in 2K1C rats, suggesting that the role of endothelium-derived NO is impaired in hypertensive rats. Similar results were also found in our previous study [26], i.e., endothelium removal enhanced phenylephrine-induced contraction in 2K1C aortae, whereas it had no effect in sham tissues. These results imply that the endothelium may modulate not only vasorelaxation but also vascular contraction induced by vasoconstrictor agents. It has been reported that such vasoconstrictors interact with the endothelium to cause the release of vasorelaxation factors, which then have an inhibitory effect on vascular smooth muscle tone [3]. As expected, quercetin and ascorbic acid did not affect phenylephrine-induced contraction in the presence of L-NAME in either 2K1C or sham rats. The results strongly suggest that the reduction of the contractile response to phenylephrine in hypertensive rats by quercetin or ascorbic acid is due to alterations in the biological activity of endothelium-derived NO. On the other hand, our results are inconsistent with a previous study by Ajay et al [17], who observed that the magnitude of phenylephrine-induced contraction was similar between spontaneous hypertensive and normotensive Wistar rats, and the inhibitory effects of quercetin and ascorbic acid on the contractile response to phenylephrine in hypertensive rats were reversed in the presence of L-NAME. They also reported that the inhibitory effect of quercetin on phenylephrine-induced contraction in hypertensive aortae is more potent than that of ascorbic acid. These differences possibly arise from complex underlying mechanisms; a possible reason for this discrepancy may be differences in the method of induction of experimental hypertension, animal species, or the experimental methods used [25].
In the present study, the effects of the flavonoid quercetin on vascular function resembled those of the antioxidant vitamin ascorbic acid, suggesting that the quercetin-induced improved vascular reactivity in 2K1C hypertensive rats may be attributed to the antioxidant effect of the flavonoid. To our knowledge, this is the first report to show that acute exposure to quercetin or ascorbic acid promotes endothelium-dependent relaxation and inhibits contractile responses in 2K1C rats. In addition, the enhanced acetylcholine-induced relaxation and diminished phenylephrine-induced contraction caused by quercetin or ascorbic acid in hypertensive rats are associated with alterations in the biological activity of endothelium-derived NO. Previous studies [27], [28] have demonstrated elevated superoxide anion levels in renovascular hypertensive rats. Accumulating evidence suggests that maintenance of 2K1C hypertension involves a systemic increase in oxidative stress [9], [10]; furthermore, we recently reported that vascular oxidative stress might be involved in chronic 2K1C renal hypertension [29]. Therefore, as discussed previously, the possibility that the improved vascular reactivity by quercetin in 2K1C hypertensive rats may be linked, at least in part, to the flavonoid's protective effects via a biological interaction between vascular endothelium-derived NO and superoxide anions cannot be ruled out; confirmation of this theory would require the direct measurement of NO and superoxide anion levels, which we were unable to perform in this study.
In summary, acute exposure to quercetin improves endothelium-dependent relaxation and inhibits α1-adrenoceptor–mediated contractions in aortic rings from 2K1C hypertensive rats, similar to the effects of the antioxidant vitamin ascorbic acid. The effects of quercetin may be linked, at least in part, to its protective effects via the biological actions of vascular endothelial NO in chronic renal hypertension.
Conflicts of interest
All authors have no conflicts of interest to declare.
Acknowledgments
This study was supported by research funds from Chosun University, 2014.
References
- 1.Zhong M.F., Shen W.L., Wang J., Yang J., Yuan W.J., He J., Wu P.P., Wang Y., Zhang L., Higashino H., Chen H. Paradoxical effects of streptozotocin-induced diabetes on endothelial dysfunction in stroke-prone spontaneously hypertensive rats. J Physiol. 2011;589:5153–5165. doi: 10.1113/jphysiol.2011.213686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lüscher T.F., Raij L., Vanhoutte P.M. Endothelium-dependent responses in normotensive and hypertensive Dahl rats. Hypertension. 1987;9:157–163. doi: 10.1161/01.hyp.9.2.157. [DOI] [PubMed] [Google Scholar]
- 3.Dohi Y., Kojima M., Sato K. Endothelial modulation of contractile responses in arteries from hypertensive rats. Hypertension. 1996;28:732–737. doi: 10.1161/01.hyp.28.5.732. [DOI] [PubMed] [Google Scholar]
- 4.Callera G.E., Varanda W.A., Bendhack L.M. Impaired relaxation to acetylcholine in 2K-1C hypertensive rat aortas involves an abnormal contribution of endothelial factors. Gen Pharmacol. 2000;34:379–389. doi: 10.1016/s0306-3623(01)00075-1. [DOI] [PubMed] [Google Scholar]
- 5.Cai H., Harrison D.G. Endothelial dysfunction in cardiovascular diseases: the role of oxidant stress. Circ Res. 2000;87:840–844. doi: 10.1161/01.res.87.10.840. [DOI] [PubMed] [Google Scholar]
- 6.Griendling K.K., Minieri C.A., Ollerenshaw J.D., Alexander R.W. Angiotensin II stimulates NADH and NADPH oxidase activity in cultured vascular smooth muscle cells. Circ Res. 1994;74:1141–1148. doi: 10.1161/01.res.74.6.1141. [DOI] [PubMed] [Google Scholar]
- 7.Toba H., Kojima Y., Wang J., Noda K., Tian W., Kobara M., Nakata T. Erythropoietin attenuated vascular dysfunction and inflammation by inhibiting NADPH oxidase-derived superoxide production in nitric oxide synthase-inhibited hypertensive rat aorta. Eur J Pharmacol. 2012;691:190–197. doi: 10.1016/j.ejphar.2012.07.018. [DOI] [PubMed] [Google Scholar]
- 8.Arnalich-Montiel A., González M.C., Delgado-Baeza E., Delgado-Martos M.J., Condezo-Hoyos L., Martos-Rodríguez A., Rodríguez-Rodríguez P., Quintana-Villamandos B. Short-term esmolol improves coronary artery remodeling in spontaneously hypertensive rats through increased nitric oxide bioavailability and superoxide dismutase activity. Biomed Res Int. 2014;531087:1–9. doi: 10.1155/2014/531087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lerman L.O., Nath K.A., Rodriguez-Porcel M., Krier J.D., Schwartz R.S., Napoli C., Romero J.C. Increased oxidative stress in experimental renovascular hypertension. Hypertension. 2001;37:541–546. doi: 10.1161/01.hyp.37.2.541. [DOI] [PubMed] [Google Scholar]
- 10.Oliveira-Sales E.B., Nishi E.E., Carillo B.A., Boim M.A., Dolnikoff M.S., Bergamaschi C.T., Campos R.R. Oxidative stress in the sympathetic premotor neurons contributes to sympathetic activation in renovascular hypertension. Am J Hypertens. 2009;22:484–492. doi: 10.1038/ajh.2009.17. [DOI] [PubMed] [Google Scholar]
- 11.Ibarra M., Moreno L., Vera R., Cogolludo A., Duarte J., Tamargo J., Perez-Vizcaino F. Effects of the flavonoid quercetin and its methylated metabolite isorhamnetin in isolated arteries from spontaneously hypertensive rats. Planta Med. 2003;69:995–1000. doi: 10.1055/s-2003-45144. [DOI] [PubMed] [Google Scholar]
- 12.García-Saura M.F., Galisteo M., Villar I.C., Bermejo A., Zarzuelo A., Vargas F., Duarte J. Effects of chronic quercetin treatment in experimental renovascular hypertension. Mol Cell Biochem. 2005;270:147–155. doi: 10.1007/s11010-005-4503-0. [DOI] [PubMed] [Google Scholar]
- 13.Mackraj I., Govender T., Ramesar S. The antihypertensive effects of quercetin in a salt-sensitive model of hypertension. J Cardiovasc Pharmacol. 2008;51:239–245. doi: 10.1097/FJC.0b013e318162011f. [DOI] [PubMed] [Google Scholar]
- 14.Cook N.C., Samman S. Flavonoids–chemistry, metabolism, cardioprotective effects, and dietary sources. Nutr Biochem. 1996;7:66–76. [Google Scholar]
- 15.Nijveldt R.J., van Nood E., van Hoorn D.E., Boelens P.G., van Norren K., van Leeuwen P.A. Flavonoids: a review of probable mechanisms of action and potential applications. Am J Clin Nutr. 2001;74:418–425. doi: 10.1093/ajcn/74.4.418. [DOI] [PubMed] [Google Scholar]
- 16.Galisteo M., García-Saura M.F., Jiménez R., Villar I.C., Wangensteen R., Zarzuelo A., Vargas F., Duarte J. Effects of quercetin treatment on vascular function in deoxycorticosterone acetate-salt hypertensive rats. Comparative study with verapamil. Planta Med. 2004;70:334–341. doi: 10.1055/s-2004-818945. [DOI] [PubMed] [Google Scholar]
- 17.Ajay M., Achike F.I., Mustafa A.M., Mustafa M.R. Direct effects of quercetin on impaired reactivity of spontaneously hypertensive rat aortae: comparative study with ascorbic acid. Clin Exp Pharmacol Physiol. 2006;33:345–350. doi: 10.1111/j.1440-1681.2006.04373.x. [DOI] [PubMed] [Google Scholar]
- 18.Montenegro M.F., Neto-Neves E.M., Dias-Junior C.A., Ceron C.S., Castro M.M., Gomes V.A., Kanashiro A., Tanus-Santos J.E. Quercetin restores plasma nitrite and nitroso species levels in renovascular hypertension. Naunyn Schmiedebergs Arch Pharmacol. 2010;382:293–301. doi: 10.1007/s00210-010-0546-1. [DOI] [PubMed] [Google Scholar]
- 19.Sigmon D.H., Beierwaltes W.H. Influence of nitric oxide in the chronic phase of two-kidney one clip renovascular hypertension. Hypertension. 1998;31:649–656. doi: 10.1161/01.hyp.31.2.649. [DOI] [PubMed] [Google Scholar]
- 20.Vanhoutte P.M. Endothelial dysfunction in hypertension. J Hypertens Suppl. 1996;14:S83–S93. [PubMed] [Google Scholar]
- 21.Choi S., Jung W.S., Cho N.S., Ryu K.H., Jun J.Y., Shin B.C., Chung J.H., Yeum C.H. Mechanisms of phytoestrogen biochanin A-induced vasorelaxation in renovascular hypertensive rats. Kidney Res Clin Pract. 2014;33:181–186. doi: 10.1016/j.krcp.2014.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Diederich D., Yang Z.H., Bühler F.R., Lüscher T.F. Impaired endothelium-dependent relaxations in hypertensive resistance arteries involve cyclooxygenase pathway. Am J Physiol. 1990;258:H445–H451. doi: 10.1152/ajpheart.1990.258.2.H445. [DOI] [PubMed] [Google Scholar]
- 23.Cuzzocrea S., Mazzon E., Dugo L., Di Paola R., Caputi A.P., Salvemini D. Superoxide: a key player in hypertension. FASEB J. 2004;18:94–101. doi: 10.1096/fj.03-0428com. [DOI] [PubMed] [Google Scholar]
- 24.Touyz R.M. Reactive oxygen species, vascular oxidative stress, and redox signaling in hypertension: what is the clinical significance? Hypertension. 2004;44:248–252. doi: 10.1161/01.HYP.0000138070.47616.9d. [DOI] [PubMed] [Google Scholar]
- 25.Bohr D.F., Dominiczak A.F., Webb R.C. Pathophysiology of the vasculature in hypertension. Hypertension. 1991;18(Suppl 5):III69–III75. doi: 10.1161/01.hyp.18.5_suppl.iii69. [DOI] [PubMed] [Google Scholar]
- 26.Jun J.Y., Yeum C.H., Moon S.H., Cho C.H., Jun K.B., Chung J.H., Yoon P.J. Altered endothelial modulation of vasoconstriction in chronic two-kidney, one clip hypertensive rats. Kor J Nephrol. 2001;20:381–392. [Google Scholar]
- 27.Oliveira-Sales E.B., Colombari D.S., Davisson R.L., Kasparov S., Hirata A.E., Campos R.R., Paton J.F. Kidney-induced hypertension depends on superoxide signaling in the rostral ventrolateral medulla. Hypertension. 2010;56:290–296. doi: 10.1161/HYPERTENSIONAHA.110.150425. [DOI] [PubMed] [Google Scholar]
- 28.Han Y., Fan Z.D., Yuan N., Xie G.Q., Gao J., De W., Gao X.Y., Zhu G.Q. Superoxide anions in the paraventricular nucleus mediate the enhanced cardiac sympathetic afferent reflex and sympathetic activity in renovascular hypertensive rats. J Appl Physiol. 2011;110:646–652. doi: 10.1152/japplphysiol.00908.2010. [DOI] [PubMed] [Google Scholar]
- 29.Choi S., Shin H.R., Kim S.H., Lee M.J., Jun J.Y., Kim H.L., Chung J.H., Yeum C.H. Effects of oxidative stress on endothelial modulation of contractions in aorta from renal hypertensive rats. Kidney Res Clin Pract. 2014;33:19–25. doi: 10.1016/j.krcp.2013.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]