Abstract
Hereditary angiopathy, nephropathy, aneurysms, and muscle cramps (HANAC) syndrome is an autosomal dominant syndrome caused by mutations in COL4A1 that encodes the α1 chain of collagen IV, a major component of basement membranes. Patients present with cerebral small vessel disease, retinal tortuosity, muscle cramps, and kidney disease consisting of multiple renal cysts, chronic kidney failure, and sometimes hematuria. Mutations producing HANAC syndrome localize within the integrin binding site containing CB3[IV] fragment of the COL4A1 protein. To investigate the pathophysiology of HANAC syndrome, we generated mice harboring the Col4a1 p.Gly498Val mutation identified in a family with the syndrome. Col4a1 G498V mutation resulted in delayed glomerulogenesis and podocyte differentiation without reduction of nephron number, causing albuminuria and hematuria in newborns. The glomerular defects resolved within the first month, but glomerular cysts developed in 3-month-old mutant mice. Abnormal structure of Bowman’s capsule was associated with metalloproteinase induction and activation of the glomerular parietal epithelial cells that abnormally expressed CD44, α-SMA, ILK, and DDR1. Inflammatory infiltrates were observed around glomeruli and arterioles. Homozygous Col4a1 G498V mutant mice additionally showed dysmorphic papillae and urinary concentration defects. These results reveal a developmental role for the α1α1α2 collagen IV molecule in the embryonic glomerular basement membrane, affecting podocyte differentiation. The observed association between molecular alteration of the collagenous network in Bowman’s capsule of the mature kidney and activation of parietal epithelial cells, matrix remodeling, and inflammation may account for glomerular cyst development and CKD in patients with COL4A1-related disorders.
Keywords: genetic renal disease, glomerular epithelial cells, molecular biology, kidney development
Type IV collagen is a major component of basement membranes (BMs). It consists of six homologous α chains designated α1(IV)–α6(IV) encoded by COL4A1–COL4A6 genes.1 Each α-chain contains a large collagenous domain composed of Gly-Xaa-Yaa repeats, a C-terminal noncollagenous region, and a small N-terminal domain. Three α-chains assemble to form three distinct triple-helical molecules.2 The α1α1α2(IV) trimer is ubiquitously expressed in embryonic BMs, including embryonic glomerular basement membranes (GBMs), and persists in most adult organs. In the mature kidney, it represents the main collagen IV molecule of the tubular, vascular, and Bowman’s capsule BM, whereas it remains only weakly expressed in the GBM.3 The α3α4α5(IV) and α5α5α6(IV) trimers display a more restricted distribution, including in the kidney, where the former is expressed in the mature GBM and distal tubule BM, and the latter is selectively found in the adult Bowman’s capsule.3
Collagen IV networks play a crucial structural role in BMs, but they are also involved in embryogenesis, cell migration and differentiation, angiogenesis, tumor growth, and metastasis. These functions are mediated through interaction with cell receptors, mainly integrins and discoidin domain receptors 1 (DDR1).4–6
Alport syndrome was the first collagen IV–inherited disease characterized with mutations affecting COL4A3, COL4A4, or COL4A5 genes.3 COL4A1 mutations were more recently identified in families affected by autosomal dominant porencephaly and cerebral small vessel disease.7–11 Eye abnormalities, such as cataract, anterior segment dysgenesis, and retinal tortuosity, were occasionally reported together with the brain defects.9–12 Concomitantly, several Col4a1 ENU mutant mice were analyzed, showing severe brain disease and eye alterations similar to the ones observed in patients.7,13–15
We have characterized a distinct systemic phenotype associated with COL4A1 mutations in six families, which we named hereditary angiopathy, nephropathy, aneurysms, and muscle cramps (HANAC).16 Affected patients usually show muscle cramps, mild cerebral small vessel disease, retinal arteriolar tortuosity, intracranial aneurysms, and a renal disease that associates multicystic kidneys, sometimes hematuria, and decreased GFR in older patients.17–19 HANAC COL4A1 mutations all substitute glycine residues between Gly498 and Gly528 within the cyanogen bromide–derived fragment CB3[IV] of the protein that contains integrin-binding sites.16–18 These data suggest that the pathogenic effects of HANAC mutations may occur through defective cell–BM interactions. Because COL4A1 mutations associated with the brain and eye phenotype are usually localized in the C-terminal half of the protein, phenotype-genotype correlations may occur among COL4A1-related diseases.
Because kidney disease, including cysts and hematuria, has not been reported in patients with brain-eye restricted disease and in ENU Col4a1 mice,7,14,15 we generated mice harboring the Col4a1 p.Gly498Val mutation previously identified in a family with HANAC to specifically investigate the pathophysiology of renal defects.
Results
Generation of HANAC Col4a1 G498V Mutant Mice
A targeting vector was designed to introduce the Col4a1 c.G1617T mutation in exon 25 leading to the p.Gly498Val substitution (G498V) (Supplemental Figure 1A). Electroporation of the construct into mouse embryonic stem (ES) cells resulted in 372 neomycin-resistant clones of which five were targeted (Supplemental Figure 1B). Three positive clones were injected into mouse blastocysts, and four chimeric mice were obtained. The targeted allele was transmitted through the germline (Supplemental Figure 1, C and D). Crossbreeding of Col4a1+/G498V mice generated 32% of Col4a1+/+, 51% of Col4a1+/G498V, and 17% of Col4a1G498V/G498V, indicating increased embryonic lethality of the homozygous mutants (expected Mendelian distribution 25%, 50%, and 25%, respectively). After birth however, heterozygous and homozygous mice showed normal viability. Brain hemorrhages were observed in Col4a1+/G498V and Col4a1G498V/G498V newborns that did not impair life span (data not shown), and retinal tortuosity and muscular dystrophy were present in adult animals (A. Trouillet et al., submitted; S. Guiraud et al., manuscript in preparation).
Col4a1 G498V Mutant Newborn Mice Showed Delayed Glomerulogenesis Resulting in Glomerular Hyperpermeability at Birth
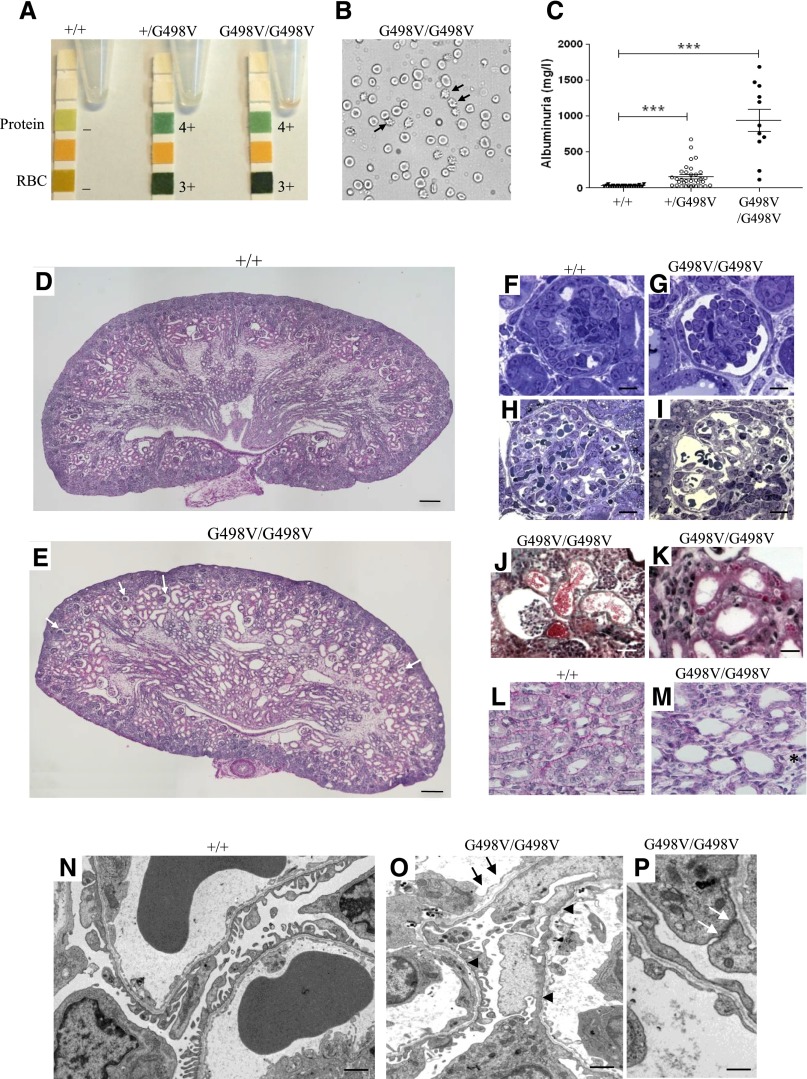
We previously reported that the COL4A1 G498V mutation is responsible for hematuria and multicystic kidney disease.16,18 Col4a1+/G498V and Col4a1G498V/G498V newborns at postnatal day 0 (P0) showed hematuria, with numerous urinary dysmorphic erythrocytes suggesting their glomerular origin (Figure 1, A and B). Albuminuria was also detected in mutant newborns, with mean urinary albumin concentrations of 34.6±7.8, 141.1±129.4, and 942.0±507.5 mg/l (P<0.001) in wild-type, heterozygous, and homozygous P0 mice, respectively (Figure 1C). Compared with the normal morphology of wild-type animals, homozygous kidneys showed alterations of glomeruli and tubules at P0 (Figure 1, D and E). Defective glomerulogenesis was observed after the formation of the first capillary loops, with podocytes arranging in multilayers instead of forming a regular layer (Figure 1, F and G). Additionally, in a subset of mature glomeruli, Bowman’s space appeared enlarged with retracted capillary tufts and widened capillary loops, without mesangiolysis (Figure 1, H–J). Proximal tubular sections in Col4a1+/G498V (not shown) and Col4a1G498V/G498V (Figure 1K) newborn kidneys showed cytoplasmic vacuoles that positively stained with anti-albumin antibody (data not shown), most likely linked to proteinuria. Col4a1G498V/G498V kidneys also showed focal dilation with flattened proximal tubule cells (Figure 1J). Collecting duct sections showed flattened epithelium with lumen widening, and ducts were focally dissociated by expansion of stroma (Figure 1, E and M).
Figure 1.
Col4a1 G498V mutant newborns show albuminuria and hematuria associated with podocyte and GBM defects at P0. (A) Urine dipstick tests revealed heavy proteinuria and hematuria in heterozygous (+/G498V) and homozygous (G498V/G498V) Col4a1 mutant mice at P0. (B) Phase-contrast microscopy analysis of spot urine sample of Col4a1 homozygous mutant P0 mice showed numerous dimorphic erythrocytes (arrow). (C) Quantification of albuminuria in wild-type (+/+), heterozygous and homozygous mutant newborns at P0. Values represent mean±SEM ***P<0.0001 compared with +/+. (D and E) Compared with wild-type animals (D), sagittal sections of newborn kidneys in Col4a1 homozygous mutant mice at P0 (E) showed enlarged Bowman’s spaces with retracted capillary tufts in a subset of glomeruli (white arrow) and dilated proximal tubular sections with flattened epithelial cells. Note also the widening of the collecting duct lumen in the medulla (periodic acid–Schiff). (F and G) Semi-thin sections of immature glomeruli after the formation of the first capillary loops in wild-type (F) and homozygous mutant (G) P0 kidneys showing podocyte disorganization in mutant glomeruli (toluidine blue stain). (H and I) Mature glomeruli in the deep cortex of wild-type (H) and homozygous mutant (I) P0 kidneys. Mutant mice showed dilated glomerular capillaries (toluidine blue stain). (J) Erythrocytes in dilated tubule sections with epithelial cell flattening, adjacent to a glomerulus with retracted capillary tuft and enlarged Bowman’s space in a homozygous mutant kidney (Masson’s trichrome). (K) Intracytoplasmic eosinophilic vacuoles in proximal tubular cells of homozygous mice (hematoxylin and eosin). (L and M) Collecting duct sections. Flattened epithelium and lumen widening of collecting ducts in homozygous kidneys (M) compared with wild-type animals (L). Note the expansion of the stroma component between ducts (*) (PAS). (M–O) EM analysis of mature glomeruli. Podocyte foot process effacement (arrowheads) and segmental duplication of GBM (arrows) in homozygous mutant newborn P0 kidneys (N) contrasting with normal architecture of podocytes and GBM in wild-type littermates. Note the presence of large intercellular junctions between podocytes in homozygous mutant kidneys (white arrow) (P). Scale bars: (D and E): 500 µm; (F–I, K): 50 µm; (L and M): 20 µm; (J) 100 µm; (N and O): 1 µm; and (O): 25 nm.
Electron microscopy (EM) of deep cortical glomeruli in homozygous kidneys at P0 revealed extensive foot process effacement with focal persistence of large cell cell-junction between podocytes and areas of splitting and duplication of the GBM (Figure 1, N–P). Although the glomerular architecture appeared intact in heterozygous kidneys, reduced podocyte foot process formation and GBM duplication were also observed by EM (data not shown).
During the first week, albuminuria decreased, with recovery of the morphologic glomerular and tubular lesions (Supplemental Figure 2, A and B). Erythrocyte casts remained present in tubular sections of 7-day-old mutant kidneys (Supplemental Figure 2C), but they were not observed at 1 month. In addition, neonate kidney defects did not result in a decreased glomerular density in adult Col4a1 G498V mutant animals (Supplemental Figure 2D).
Altogether, these results indicate that the Col4a1 G498V mutation induced delayed glomerulogenesis with defective GBM and podocyte maturation, resulting in glomerular hyperpermeability in newborn animals.
Col4a1 G498V Mutation Decreased α1α1α2(IV) Trimer Secretion and Altered Nephrin Expression in Newborn P0 Kidneys
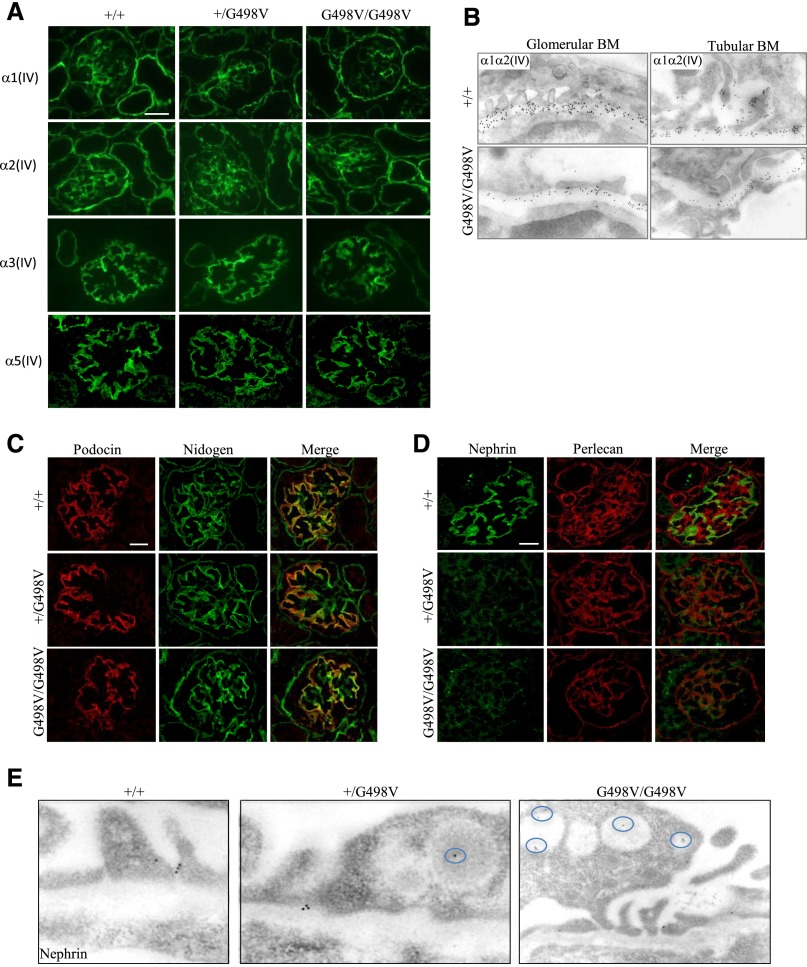
To assess the effect of the Col4a1 G498V mutation on type IV collagen expression, we analyzed the renal distribution of the α1α1α2(IV) trimer. In Col4a1G498V/G498V newborn kidneys, the expression of the mutant α1α1α2(IV) trimer was reduced compared with wild-type and heterozygous mice, but it remained detectable in the mesangium, Bowman’s capsule, tubular BM, and GBM (Figure 2, A and B), which indicated that the mutation only partially altered α1α1α2(IV) secretion. GBM expression of the α3(IV) and α5(IV) chains were comparable between genotypes (Figure 2A).
Figure 2.
Expression of IV collagen and podocyte slit diaphragm proteins in Col4a1 G498V mutant newborn P0 kidneys. (A and B) Collagen IV expression in newborn kidneys at P0. (A) Expression of the mutant α1(IV) chain was decreased in homozygous (G498V/G498V) mice compared with heterozygous (+/G498V) and wild-type (+/+) littermates, but it remained detectable in the mesangium, GBM, Bowman’s capsule, and tubular BM. A comparable decreased expression of the α2(IV) chain was observed in Col4a1G498V/G498V mutant kidneys. The α3(IV) and α5(IV) chains were normally expressed in the GBM and in the BM of distal tubules in Col4a1 G498V mutant animals. Scale bar: 25 μm (B) Immunoelectron microscopy using α1α2(IV) antibodies demonstrated a decreased but persistent secretion of the mutant α1α1α2(IV) molecule in the GBM (left column) and tubular BM (right column) in homozygous animals when compared with controls. (C–E) Expression of podocyte slit diaphragm proteins in Col4a1 G498V mutant newborn P0 kidneys. Costaining for podocin and nidogen showed normal expression of podocin in the basal region of the podocytes along the GBM in mutant kidneys (C), whereas nephrin expression was dramatically reduced in both heterozygous and homozygous P0 mice (D). Note the normal detection of perlecan in the mesangium and in extraglomerular BMs in mutant kidneys. Scale bar: 25 μm. (E) Immunoelectron microscopy using antinephrin antibody occasionally detected gold particles in the slit diaphragm of podocytes in Col4a1 mutant P0 animals, but nephrin was abnormally localized in vesicles within the podocytes (circle) in heterozygous and homozygous kidneys. Scale bars for (A), (C), and (D): 50 µm.
Given the podocyte defects observed in the mutant newborns at P0, we investigated the expression of slit diaphragm proteins. Although the expression of podocin remained normal (Figure 2C), that of nephrin was dramatically reduced in Col4a1+/G498V and Col4a1G498V/G498V glomeruli (Figure 2D). By immunogold EM, nephrin was occasionally detected in slit diaphragms while it was accumulated in cytoplasmic vacuoles (Figure 2E). Defective nephrin expression was consistent with the dramatic reduction of foot process formation and albuminuria in P0 Col4a1 mutants.
Col4a1 G498V Mutant Adult Mice Developed Glomerulocystic Kidney Disease and Dysmorphic Papillae with Mild Renal Function Alteration
Although tubular sections were normal in 1-month mutant kidneys, careful analysis disclosed abnormal morphology of the glomerular parietal epithelial cells (PECs) in approximately 15% of mutant glomeruli. PECs were cube-shaped with enlarged nuclei, instead of forming a monolayer of thin epithelial cells with flat nuclei (Supplemental Figure 3A). At 3 months of age, glomerular cysts and inflammatory infiltrates first developed in Col4a1+/G498V and Col4a1G498V/G498V kidneys. Glomerular cysts were distributed throughout the cortex area and were characterized by enlarged Bowman's space lined by flattened PECs and retracted capillary tufts (Figure 3, A and B). Glomerulotubular junction was preserved in normal glomeruli and in glomeruli undergoing cystic transformation (Figure 3C). The percentage of glomeruli with cystic changes significantly increased at 6 months in both heterozygous and homozygous mice, and also at 12 months in homozygous mutants (Figure 3D). Cystic surfaces were comparable between ages and genotypes, except at 12 months where glomerular cysts appeared slightly but significantly larger in heterozygous kidneys (Figure 3D). Cuboidal PECs lining the Bowman’s capsule were present in about one-third of noncystic glomeruli in 6- and 12-month animals (Figure 3A). Large infiltrates mostly composed of CD3-positive lymphocytes surrounded by F4/80-positive macrophages occasionally developed around cystic and noncystic glomeruli and around arterioles or venules with intact vessel walls (Figure 3A, Supplemental Figure 3, B and C). No significant fibrosis developed except in the inflammatory areas, and tubules remained normal at 12 months.
Figure 3.
Morphologic and functional characteristics of Col4a1 G498V mutant adult kidney. (A) Histologic analysis of 6-month adult kidney. Heterozygous (+/G498V) and homozygous (G498V/G498V) mice developed cystic glomeruli with dilation of Bowman’s space and retracted capillary tuft (first column, Masson’s trichrome). Scale bars: 200 µm. Noncystic glomeruli showed cuboidal PECs (black arrows) (second column, Jones’ methenamine silver). Nodular inflammatory infiltrates were seen in both heterozygous and homozygous mutant kidneys around glomeruli (third column) and arterioles (fourth column) (Masson’s trichrome). Scale bar: 50 µm. (B) Transverse sections of 12-month heterozygous and homozygous kidneys showing distribution of glomerular cysts throughout the cortex area. Note the dysmorphic papilla in homozygous kidney (Masson’s trichrome). Scale bar: 1 mm. (C) Glomeruli with cystic change (cyst surface, left panel: 11,283 μm2; cyst surface, right panel: 14,900 μm2) showing a normal glomerulotubular junction (black arrow) in +/G498V and G498V/G498V 6-month kidneys (periodic acid–Schiff). Scale bar: 20 μm. (D) Percentage of glomeruli with cystic changes (left panel) and glomerular cyst area (right panel) in heterozygous and homozygous kidneys at 3 months (+/G498V, n=4; G498V/G498V, n=4), 6 months (+/G498V, n=8; G498V/G498V, n=6) and 12 months (+/G498V, n=7; G498V/G498V, n=7) of age. Values represent mean±SEM. *P<0.05, ***P<0.001.
Ultrastructural analysis of adult mutant kidneys performed at 6 months disclosed morphologic PEC alterations, with thickening and multilamination of the Bowman’s capsule (Supplemental Figure 3D), whereas the GBM and the tubular BM remained normal (not shown).
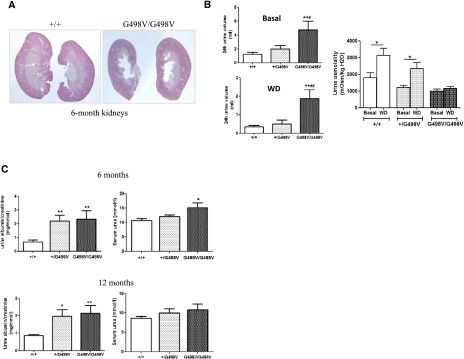
Beside glomerular and vascular alterations, homozygous mice showed dysmorphic papillae (Figure 4A), which were associated with polyuria and defects in urine concentration under water deprivation (Figure 4B).
Figure 4.
Dysmorphic papilla and functional defects in adult Col4a1 G498V mutant mice. (A) Longitudinal section of 6-month adult kidneys showing dysmorphic papilla in homozygous animals (Masson’s trichrome). (B) Measurements of 24-hour urine volume and urine osmolality in 6-month animals with free access to water (Basal) (left upper panel) or under water deprivation (WD) (left lower panel) showed polyuria in homozygous mice in both conditions and no significant increase in urine osmolality under water deprivation in homozygous animals (right panel) (+/+: n=8; +/G498V: n=10; G498V/G498V: n=5). Values represent mean±SEM. *P<0.05, **P<0.01 compared with +/+, #P<0.05, ##P<0.01 compared with +/G498V. (C) Urine albumin/creatinine ratio and serum urea at 6 and 12 months of age (6 months: +/+: n=11; +/G498V: n=10; G498V/G498V: n=7; 12 months: +/+: n=8; +/G498V: n=9; G498V/G498V: n=6). Values represent mean±SEM. *P<0.05, **P<0.01 compared with +/+.
As previously observed in newborn kidneys, α1(IV) and α2(IV) chain staining analyzed at 6 months was decreased in the mesangium and tubular BM of homozygous adults and in the Bowman’s capsule where the expression of the α5α5α6(IV) trimers remained normal (Supplemental Figure 4A). Intermediate staining was observed in heterozygous mutants. Expression of the BM proteins, nidogen/entactin, laminin α2, and laminin β1, was normal, as was that of fibronectin and collagen I (Supplemental Figure 4B).
Renal functional parameters were assessed at 6 and 12 months of age. Urine albumin/creatinine was mildly but significantly increased in 6- and 12-month-old mutant animals compared with control littermates (Figure 4C). Only homozygous mutants showed an increase of serum urea at 6 months (10.7±0.66, 12.1±0.51, and 15.1±0.16 mmol/l in Col4a1+/+, Col4a1+/G498V, and Col4a1G498V/G498V, respectively, P<0.05; Col4a1G498V/G498V compared with controls), whereas at 12 months, urea was nonsignificantly increased in this genotype (Figure 4C). Because body weights at 6 and 12 months were significantly lower in Col4a1G498V/G498V animals (Supplemental Figure 5), these results suggest that alteration of kidney function was likely underestimated in homozygous mutant animals.
Col4a1 G498V Mutation Led to PEC Activation and Induction of DDR1 and Integrin-Linked Kinase Pathway
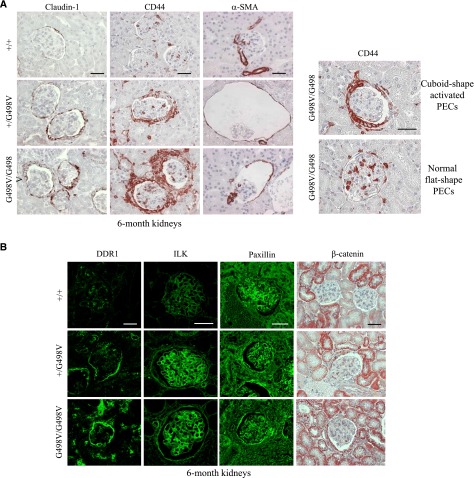
We focused on the alterations of the PECs in 6-month animals with overt cystic renal disease. The tight junction protein claudin-1, a specific marker of PECs, was strongly expressed at the apical and lateral surface of PECs in mutant mice, whereas it was observed at a lower level in control kidneys (Figure 5A). More importantly, cuboidal PECs expressed CD44, a marker of activated parietal epithelial cells (aPECs), not detected in normal glomeruli or in normal flat-shape PECs in mutant kidneys (Figure 5A). PECs lining cystic glomeruli showed de novo expression of α-smooth muscle actin (α-SMA), whereas cube-shaped PECs were rarely stained for α-SMA (Figure 5A). Claudin-1 upregulation and CD44 induction in PECs were actually first detected in 1-month-old mice before the development of the glomerular cysts and periglomerular inflammation (Supplemental Figure 6A). CD44, an endogenous marker of T lymphocytes and macrophages, also labeled periglomerular and periarteriolar infiltrates (Figure 5A). Col4a1 G498V mutant kidneys did not show any glomerular crescent or sclerotic segment of the glomerular tuft at any age.
Figure 5.
Phenotypic alterations of glomerular PECs in Col4a1 G498V mutant adult mice. (A) Immunohistochemical staining of a 6-month adult kidney with antibodies to claudin-1, CD44, and α-SMA (left panels). In heterozygous (+/G498V) and homozygous (G498V/G498V) mutants, cuboid shape PECs in noncystic glomeruli showed claudin-1 upregulation and induction of CD44 expression. PECs lining cystic glomeruli stained positive for α-SMA. Right panels illustrate the selective induction of CD44 marker in cuboid-shape PECs (upper right panel) and absence of CD44 expression in normal flat-shape PECs (lower right panel) in 6-month mutant glomeruli. Note the presence of periglomerular inflammatory cells that also are stained for CD44. Scale bar: 50 μm. (B) Expression of the DDR1, ILK, paxillin, and β-catenin in a 6-month adult kidney. DDR1 expression was induced in the Bowman’s capsule and the small vessel wall of Col4a1+/G498V and Col4a1G498V/G498V mice. ILK and ILK downstream partners, paxillin, and β-catenin were induced in PECs. In addition, ILK expression was strongly enhanced in the mesangium and podocytes in both mutant genotypes compared with controls (+/+). Scale bars: 50 µm.
We next investigated whether decreased BM expression of the mutant α1α1α2(IV) trimer modulated collagen receptors. In Col4a1 mutant kidneys, induction of DDR1 expression was observed in PECs and in small vessels at 1 month and persisted at 6 months, whereas no DDR1 staining in the Bowman’s capsule was observed in control kidneys (Figure 5B, Supplemental Figure 6B). Expression of integrin-linked kinase (ILK), an integrin interacting protein involved in cell-matrix adhesion and signaling, was also strongly induced in PECs and markedly enhanced in podocytes and mesangium at 1 and 6 months of age in mutant kidneys (Figure 5B, Supplemental Figure 6B). Similarly, paxillin and β-catenin, two elements downstream of the integrin/ILK signaling pathway, were strongly upregulated in glomerular PECs of mutant mice, whereas their expression was absent in controls (Figure 5B, Supplemental Figure 6B).
Overall, these results indicated that alteration of collagen IV network in Bowman’s capsule was associated with early changes in PECs that showed an activated phenotype, induction of DDR1, and activation of ILK pathway preceding the development of glomerular cysts and periglomerular inflammation.
Col4a1 G498V Mutation Is Responsible for Matrix Metalloproteinase Induction
To investigate whether the mutant α1α1α2(IV) network enhanced matrix remodeling, we investigated activity and expression of metalloproteinase-2 (MMP2) and MMP9, two matrix MMPs specific for collagen IV. Increased MMP2 and MMP9 activity was detected in both heterozygous and homozygous kidney homogenates by SDS-PAGE gelatin zymography at 6 months (Figure 6A). RT-PCR experiments showed that mRNA of MMP2 and MT1-MMP involved in MMP2 activation was upregulated in 6-month Col4a1 mutant kidneys, whereas MMP9 mRNA was only significantly increased in heterozygous mice (Figure 6B). Likewise, mRNA levels of tissue inhibitors of MMPs TIMP1 and TIMP2 were increased (Figure 5B). We then performed in situ zymography, which localized increased gelatinase activity to the Bowman’s capsule and the tubular BM in 6-month mutant kidneys. The enzymatic activity was inhibited by EDTA, indicating that MMP2 and/or MMP9 carried gelatinolytic activity (Figure 6C). These results showed that despite increased MMP inhibitor mRNAs, the resulting gelatinolytic activity was increased in mutant kidney tissue, particularly in the Bowman’s capsule.
Figure 6.
Matrix MMP induction in Col4a1 G498V mutant adult kidneys. (A) SDS-PAGE zymography of protein extracts from 6-month kidneys showed induction of pro-MMP9 and upregulation of pro-MMP2 and MMP2 enzymatic activities in Col4a1+/G498V and Col4a1G498V/G498V mutant kidneys compared with control littermates (+/+). (B) Renal mRNA level of MMP-2, MMP-9, MT1-MMP, TIMP1, and TIMP2 (+/+: n=5; +/G498V: n=5; G498V/G498V: n=3) in 6-month adult kidneys. Values represent mean±SEM. *P<0.05 compared with +/+. (C) In situ gelatin zymography revealed enhanced MMP enzymatic activity in the periglomerular region and around the renal tubules in 6-month-old Col4a1+/G498V and Col4a1G498V/G498V kidneys. Pretreatment of the renal tissue with EDTA, a specific inhibitor of MMP2 and MMP9, blocked gelanolytic activity. Scale bar: 100 μm.
Discussion
Generation of the first animal model of HANAC syndrome provided us the opportunity to analyze the pathophysiology of organ defects in this multisystemic syndrome and to approach the pleiotropic role of the ubiquitously expressed α1 isoform of collagen IV.
Several Col4a1 ENU mutant mice have been analyzed.7,9,13–15,20 They usually showed severe brain disease and eye abnormalities, but no consistent kidney alteration has been reported so far, especially renal cysts, a cardinal phenotypic characteristic of HANAC.16,18 Because HANAC COL4A1 mutations are localized within a 30 amino acid segment containing integrin-binding sites, alteration of this protein domain might trigger specific pathogenic pathways.
Contrasting with previous ENU Col4a1 animals, Col4a1 G498V homozygous newborns are viable. The detection of the mutant α1α1α2(IV) trimer in BMs of homozygous animals may account for the less severe phenotype observed in our Col4a1 mutant animals. Indeed, in homozygous ENU Col4a1 mice with exon 40 deletion, the α1(IV) chain mutation results in the absence of the collagen IV network in embryonic BMs responsible for embryonic lethality.7,13 Therefore, as previously demonstrated in Alport syndrome,21–23 the type of mutation and location of glycine substitution may influence the severity of the phenotype in COL4A1-related diseases.
Col4a1 G498V mutant mice are a relevant animal model for HANAC, reproducing the organs defects observed in patients, including retinal tortuosity and muscular dystrophy (A. Trouillet et al., manuscript submitted; S. Guiraud et al., manuscript in preparation), brain hemorrhages, and renal cysts in adults. However, we also unexpectedly observed renal alterations in Col4a1 G498V mutant newborns. Increased glomerular permeability observed in these animals is related to delayed podocyte differentiation, nephrin mislocalization, and GBM disorganization that resolve during the first weeks after birth. The α1α1α2(IV) trimer constitutes the first collagen IV network of the embryonic GBM,24 and we provide the demonstration that it contributes to podocyte maturation. Interestingly, podocyte and GBM alterations are reminiscent of the findings in mice lacking the α3 integrin and in those with podocyte-specific deletion of the β1 integrin.25,26 Immature collecting ducts in Col4a1 G498V mutant mice at birth were also observed in α3 integrin defective mice.25 During nephrogenesis, the α3β1 integrin is expressed in the ureteric bud and in podocyte precursors.27,28 It specifically localizes in the podocyte foot processes and contributes to podocyte cytoskeleton stabilization through interaction with GBM components. The GBM ligand for the α3β1 integrin in mature glomerulus is laminin 521,29 but the ligand is unknown in earlier stage of glomerulogenesis. Data in our mutant mice with delayed podocyte maturation suggest that the ligand might be α1α1α2(IV). This hypothesis is supported by the site of the Col4a1 G498V substitution, which alters an integrin-binding motif.18 Additionally, reduced integration of the normal α1(IV) chain and/or expression of the mutant α1(IV) isoform in the collagen IV network might weaken the GBM. The replacement of embryonic isoforms of collagen IV and laminin, by α3α4α5(IV) and laminin 521, respectively,30 may account for morphologic and functional recovery of the glomerular filtration barrier in 1-month animals. Finally, the delayed glomerulogenesis observed in the mutant newborns did not affect the nephronic mass because glomerular density and kidney weights were preserved in adult kidneys.
Analysis of the adult Col4a1 G498V mutant kidneys provided new insights into the cystic kidney phenotype observed in patients with HANAC. Actually, computed tomography scan imaging did not allow identification of the origin of the renal cysts, which showed a broad cortical distribution in patients with HANAC syndrome,16 a pattern that remains compatible with a glomerulocystic kidney disease.31 Kidney biopsy specimens were analyzed in two young patients with normal renal imaging and did not identify cyst formations.16,17 Histologic observations in adult mutant animals now clearly show that cysts arise from glomeruli with Bowman’s space dilatation and they distribute through the cortex area. As observed in several patients, the number of cysts increased with age in mutant mice. Therefore, HANAC potentially represents a new inherited form of glomerulocystic kidney disease, which adds to autosomal dominant or recessive polycystic kidney disease and uromodulin and HNF1β/TCF2 mutation–related disorders.31
Complex alterations of the Bowman’s capsule and the PECs precede glomerular cysts in mutant adult kidneys. PECs feature morphologic characteristics of activated PECs with coexpression of claudin-1 and CD44.32–34 These alterations were seen as early as 1 month before appearance of the glomerular cysts and periglomerular inflammation. PEC phenotypical changes were not associated either with glomerular crescent or pseudocrescent formations, as observed in crescentic glomerulonephritis and collapsing glomerulopathy, respectively, or lesions of segmental glomerulosclerosis.32–34
In glomerular diseases associated with the aPEC phenotype, triggers of PEC activation remain poorly defined. Interestingly, aPECs in Col4a1 G498V mutant mice show induction of DDR1 and the ILK signaling pathway. DDR1 links collagen IV and plays an important role in cell response to matrix remodeling and proinflammatory response.35–38 In our mice, DDR1 induction may be the consequence of the enhanced matrix turnover observed in the Bowman’s capsule, as shown by increased collagenase activity and ultrastructural alterations. Periglomerular inflammation may result from DDR1 overexpression, which may also account for perivascular inflammation because upregulation of DDR1 was observed in the vascular wall.
Although activation of ILK pathway is well established in podocytes,39–41 it was not previously reported in PECs. ILK regulates extracellular matrix-induced signaling pathway through binding of β1 and β3 integrins.42 In our mouse model, induction of ILK in PECs may result from an enhanced, integrin-mediated signaling pathway triggered by Col4a1 mutation and/or collagen IV network alteration. In turn, ILK activation may account for β-catenin overexpression through inhibition of GSK3β, which promotes β-catenin degradation in normal conditions.43 We have shown upregulation of claudin-1 and induction of CD44, which are both target genes for β-catenin/Tcf(Lef-1) pathways.44,45
Mechanisms of glomerular cyst formation have not been previously addressed. We have not observed morphologic defects that would support the hypothesis of obstructive nephropathy as explanation for the glomerulocystic features. In particular, glomerulotubular junction was preserved in glomeruli with cystic change. The mutated Col4a1 protein, together with decreased α1α1α2(IV) trimer secretion and enhanced matrix remodeling, may alter the mechanical properties of the Bowman’s capsule and may therefore inefficiently counteract the glomerular filtration pressure. In addition, there are dramatic alterations of PECs that show signs of activation at 1 month and later on show signs of mesenchymal transition in PECs lining glomerular cysts, with expression of α-SMA. These alterations might be related to activation of ILK, which has been shown to promote epithelial mesenchymal transition46–48 and enhance MMP2 and MMP9 activity.49,50
In addition to the complex glomerular alterations observed in Col4a1 G498V mutant mice, dysmorphic papillae and urine concentration defects were selectively observed in adult homozygous animals, as were collecting duct anomalies in homozygous neonates. Such functional alterations were not recorded in patients with HANAC, which carry however the COL4A1 mutation in the heterozygous state.
In summary, our study reveals the previously unrecognized role during glomerulogenesis of α1α1α2(IV), the first collagen IV network of the GBM, and its contribution to podocyte differentiation. In mature kidneys, α1α1α2(IV) collagen network integrity is crucial for maintenance of PEC differentiation. Primary molecular alterations of the Bowman’s capsule matrix trigger PEC activation and mesenchymal transition. From a clinical point of view the Col4a1 G498V mutant mice provide new insight into the pathomechanisms of glomerular cystic kidney disease.
Concise Methods
Generation of Col4a1 G498V Mutant Mice
The Col4a1 G498V mutant mouse line was established at the MCI/ICS (Mouse Clinical Institute, Illkirch, France; http://www-mci.u-strasbg.fr). A 4.8-kb fragment encompassing introns 22–26 and exons 22–25 of the Col4a1 gene was amplified by PCR using mus musculus DNA as a template and subcloned in an MCI proprietary vector, resulting in step1 plasmid. This MCI vector has a floxed neomycin resistance cassette driven by a phosphoglycerate kinase 1 promoter. A 3.1-kb fragment encompassing intron 26 was amplified by PCR and subcloned in step1 plasmid to generate the final targeting construct. The Col4a1 c.G1617T mutation was introduced in exon 25 via site-directed mutagenesis. Sequencing of the final targeting vector was performed. The linearized targeting construct was electroporated in 129Sv/Pas mouse ES cells. After selection, five targeted clones were identified by PCR using external primers and confirmed by Southern blot using 5′ and 3′ external probes (Supplemental Figure 1). Two positive ES clones were injected into C57BL/6N blastocysts, and the derived male chimeras gave germline transmission. Tail biopsies from F1 mice were genotyped with primers flanking neomycin cassette. Col4a1+/G498V offsprings were crossed with CMV-Cre mice. Offsprings were assessed by PCR for the excision of the cassette, and several heterozygous Col4a1+/G4984V were obtained. Col4a1+/G498V mice on mixed background were crossed with wild-type mice from C57BL/6 background for two generations. Genotyping was performed by PCR using Ef/Er primers flanking the residual Lox site, and presence of the p.Gly498Val was confirmed by sequencing of genomic DNA from Col4a1+/G498V animals (Supplemental Figure 1). Sex of newborns was determined by PCR amplification of genomic DNA sequence of the Kdm5c gene.51 Newborn mice were euthanized. Adult mice were euthanized at 7 days, 1 month, 3 months, 6 months, and 1 year with intraperitoneal administration of pentobarbital (150 mg/kg). Mice were maintained in a specific pathogen-free environment, and experiments were conducted in accordance with French government policies.
Histologic and Ultrastructural Analysis
For light microscopy analysis, kidneys were fixed with alcohol-formalin-acetic acid (AFA) solution, dehydrated, and embedded in paraffin. There were 4-µm sections that were deparaffinized, rehydrated, and stained with Masson’s trichrome, hematoxylin and eosin, and periodic acid–Schiff. For quantification of glomerular density, glomerular cyst number, and size, longitudinal kidney sections were scanned at a high resolution using a slide scanner (NanoZoomer 2.0-HT; Hamamatsu, Massy, France). Size of glomerular cysts was measured on at least two different section levels/kidneys using Image J software. Glomeruli were counted one by one, using the NanoZoomer software, within cortical areas of 1-mm depth from the renal capsule that covered the kidney sections; glomerular density was evaluated as the average number of glomeruli per square millimeter. Transmission EM was performed on kidney samples fixed in 2.5% glutaraldehyde dissolved in 0.1 mol/L cacodylate buffer (pH 7.4) at 4°C. The tissue fragments were postfixed in 1% OsO4, dehydrated, and embedded in epoxy resin. Semi-thin sections (0.5 μm) were stained by toluidine blue, and 60-nm ultra-thin sections were contrasted with electron-opaque solution uranyl acetate and then lead citrate; they were then examined using a JEOL 1010 electron microscope (JEOL, Tokyo, Japan) and a MegaView III camera (Olympus Soft Imaging Systems, Münster, Germany).
Immunohistochemistry, Immunofluorescence, and Immunoelectron Microscopy
AFA-fixed, paraffin-embedded sections were used for immunohistochemistry. Antigen retrieval was performed by heat in citrate buffer (pH 6.0) or with proteinase K/PBS solution. Tissue sections were probed using primary antibodies, followed by incubation with Histofine immunohistochemical staining reagent (Nichirei Biosciences, Japan). Immunofluorescence experiments were carried out with 4-µm cryosections or AFA-fixed, paraffin-embedded sections of kidney, depending on the primary antibody affinity. Following blockade by 10% BSA/PBS solution, the sections were incubated with primary antibodies overnight. The slides were then incubated with appropriate AlexaFluor 488– or AlexaFluor 555–labeled secondary antibodies (Invitrogen) for 1 hour at room temperature. Immunofluorescence micrographs were obtained using a Leica TCS SP2 confocal microscope. Primary antibodies used for immunostaining are listed in Supplemental Table 1. Immunogold EM was performed on ultrathin frozen sections of kidney tissue as previously described.52 Sections were processed for indirect immunogold labeling with the use of rabbit anti–α1α2(IV) collagen (Rockland) and guinea pig antinephrin (PROGEN).
Urine and Blood Samples Analysis
Urine samples were collected using metabolic cages in adult mice with free access to water or after 24 hours of water deprivation. Peripheral blood was collected after puncture of the retro-orbital sinus in mice anesthetized using pentobarbital 10%. Urine osmolality was measured using a Vapro 5520 vapor pressure osmometer (Wescor). Serum urea, urine creatinine, and albuminuria were measured by photometry using an Olympus AU 400 apparatus (IFR02; Hôpital Bichat, Paris, France). Urine of newborn mice (P0–P7) was collected by spot urine sampling. For SDS-PAGE analysis, 1 μl of urine sample was loaded on polyacrylamide gel electrophoresis, and the gels then were stained with Coomassie Brilliant Blue solution.
Detection of Matrix MMP Activity
Gelatinolytic activity was determined by in situ zymography on 8-µm unfixed cryostat sections using quenched fluorogenic substrate DQ gelatin (Molecular Probes). Then 1 mg/ml of DQ gelatin solution was prepared with water and was 1:10 diluted in 1% (w/v) low gelling temperature agarose (Sigma-Aldrich) in PBS. The 50-µl samples of mixture were applied on top of air-dried cryosections and covered with a coverslip and gelled after incubation at 4°C.53 The slides were incubated for 3 hours at 37°C, and fluorescence was analyzed under confocal microscopy. To determine the specificity of gelatinolytic activity, kidney cryosections were preincubated for 1 hour at room temperature with 20-mM EDTA in PBS, which inhibits Mmp2 and Mmp9 activity, and incubation with DQ gelatin was performed as previously described.
SDS-PAGE gelatin zymography was performed with renal tissue homogenized in RIPA lysis buffer supplemented with PMSF, sodium orthovanadate, and protease inhibitor cocktail solution (sc-24948; Santa Cruz Biotechnology) using a T10 Basic Ultra-Turrax disperser (IKA, Germany). Gelatin zymography was performed with tissue homogenates using 8% polyacrylamide gel containing 1 mg/ml of gelatin. After electrophoresis, gels were washed in 2.5% Triton X-100 and then incubated in ZnCl2/CaCl2 containing Tris buffer. As a negative control, identical gels were incubated in EDTA-containing Tris buffer. Gels were then stained in Coomassie Brilliant Blue solution.
Quantitative Real-Time PCR
Total RNAs were prepared from kidney tissue samples using the TRIzol Reagent (Life Technologies) and the T10 Basic Ultra-Turrax disperser. RNA quality was checked by NanoDrop, and residual genomic DNA was removed by DNase I treatment (Fermentas). For quantitative reverse-transcription polymerase chain reaction, the mRNA was converted into cDNA using the Revert Aid H minus First Strand DNA Synthesis kit (Fermentas). cDNA was amplified by PCR using specific primers, FastStart DNA Master SYBR Green I kit (Roche Diagnostic) and the LightCycler 480 System (Roche Diagnostic). For the normalization of gene expression data, the GeNorm was used to determine the two most stable references among five candidate housekeeping genes. Results are presented as normalized relative expression levels.
Statistical Analyses
All data are presented as individual measurements with mean±SEM. Statistical significance was determined by using either nonparametric tests, including Mann–Whitney and Kruskal–Wallis tests, or the paired t test, when appropriate. All statistical tests were calculated using GrapPad Prism 5.00 (GraphPad Software, San Diago, CA). Statistical significance was defined by P<0.05, P<0.01, and P<0.001.
Study Approval
All of the experimental procedures were approved by the ethics committee at the University Pierre et Marie Curie and the French Ministry of Research (agreement 01108.01) and were performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and the French Government animal welfare policy.
Disclosures
None.
Supplementary Material
Acknowledgments
We thank Chantal Jouanneau, Souhila Ouchelouche, Rémi Piedagnel, Sandra Onifarasoaniaina, (Institut National de la Santé et de la Recherche Médicale [INSERM], Unité Mixte de Recherche UMR_S 1155, Paris, France), Perrine Frere, and Philippe Fontanges (Platerforme d’Imagerie, Sorbonne Universités, Université Pierre et Marie Curie (UPMC) Univ Paris 06, Institut Fédératif de Recherche 65, Paris, France) for technical assistance. We thank Nicolas Sohraindo (Institut Fédératif de Recherche 02, Bichat hospital, Paris, France) for biochemical analysis of urine and serum sample.
This work was supported by grants from INSERM, Université Pierre et Marie Curie–Paris 6 (Bonus Qualité Recherche and legs Tessier), Assistance Publique-Hôpitaux de Paris (Délégation à la Recherche Clinique, Contract CRC06032/P061017), Association pour l’Utilisation du Rein Artificiel, Agence Nationale de la Recherche (ANR-08-Genopath-018-02), Délégation Générale à la Santé, through Coordination Theme 1 (Health) of the European Community’s 7th Framework Programme EUNEFRON (grant agreement no. HEALTH-F2-2007- 201590), Association pour l’Information et la Recherche sur les Maladies Rénales Génétiques (AIRG-France), and AMGEN Company.
Footnotes
Published online ahead of print. Publication date available at www.jasn.org.
This article contains supplemental material online at http://jasn.asnjournals.org/lookup/suppl/doi:10.1681/ASN.2014121217/-/DCSupplemental.
References
- 1.Khoshnoodi J, Pedchenko V, Hudson BG: Mammalian collagen IV. Microsc Res Tech 71: 357–370, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Boutaud A, Borza DB, Bondar O, Gunwar S, Netzer KO, Singh N, Ninomiya Y, Sado Y, Noelken ME, Hudson BG: Type IV collagen of the glomerular basement membrane. Evidence that the chain specificity of network assembly is encoded by the noncollagenous NC1 domains. J Biol Chem 275: 30716–30724, 2000 [DOI] [PubMed] [Google Scholar]
- 3.Hudson BG, Tryggvason K, Sundaramoorthy M, Neilson EG: Alport’s syndrome, Goodpasture’s syndrome, and type IV collagen. N Engl J Med 348: 2543–2556, 2003 [DOI] [PubMed] [Google Scholar]
- 4.Heino J, Huhtala M, Käpylä J, Johnson MS: Evolution of collagen-based adhesion systems. Int J Biochem Cell Biol 41: 341–348, 2009 [DOI] [PubMed] [Google Scholar]
- 5.Vogel W, Gish GD, Alves F, Pawson T: The discoidin domain receptor tyrosine kinases are activated by collagen. Mol Cell 1: 13–23, 1997 [DOI] [PubMed] [Google Scholar]
- 6.Leitinger B, Hohenester E: Mammalian collagen receptors. Matrix Biol 26: 146–155, 2007 [DOI] [PubMed] [Google Scholar]
- 7.Gould DB, Phalan FC, Breedveld GJ, van Mil SE, Smith RS, Schimenti JC, Aguglia U, van der Knaap MS, Heutink P, John SW: Mutations in Col4a1 cause perinatal cerebral hemorrhage and porencephaly. Science 308: 1167–1171, 2005 [DOI] [PubMed] [Google Scholar]
- 8.van der Knaap MS, Smit LM, Barkhof F, Pijnenburg YA, Zweegman S, Niessen HW, Imhof S, Heutink P: Neonatal porencephaly and adult stroke related to mutations in collagen IV A1. Ann Neurol 59: 504–511, 2006 [DOI] [PubMed] [Google Scholar]
- 9.Gould DB, Phalan FC, van Mil SE, Sundberg JP, Vahedi K, Massin P, Bousser MG, Heutink P, Miner JH, Tournier-Lasserve E, John SW: Role of COL4A1 in small-vessel disease and hemorrhagic stroke. N Engl J Med 354: 1489–1496, 2006 [DOI] [PubMed] [Google Scholar]
- 10.Lanfranconi S, Markus HS: COL4A1 mutations as a monogenic cause of cerebral small vessel disease: A systematic review. Stroke 41: e513–e518, 2010 [DOI] [PubMed] [Google Scholar]
- 11.Yoneda Y, Haginoya K, Kato M, Osaka H, Yokochi K, Arai H, Kakita A, Yamamoto T, Otsuki Y, Shimizu S, Wada T, Koyama N, Mino Y, Kondo N, Takahashi S, Hirabayashi S, Takanashi J, Okumura A, Kumagai T, Hirai S, Nabetani M, Saitoh S, Hattori A, Yamasaki M, Kumakura A, Sugo Y, Nishiyama K, Miyatake S, Tsurusaki Y, Doi H, Miyake N, Matsumoto N, Saitsu H: Phenotypic spectrum of COL4A1 mutations: Porencephaly to schizencephaly. Ann Neurol 73: 48–57, 2013 [DOI] [PubMed] [Google Scholar]
- 12.Coupry I, Sibon I, Mortemousque B, Rouanet F, Mine M, Goizet C: Ophthalmological features associated with COL4A1 mutations. Arch Ophthalmol 128: 483–489, 2010 [DOI] [PubMed] [Google Scholar]
- 13.Gould DB, Marchant JK, Savinova OV, Smith RS, John SW: Col4a1 mutation causes endoplasmic reticulum stress and genetically modifiable ocular dysgenesis. Hum Mol Genet 16: 798–807, 2007 [DOI] [PubMed] [Google Scholar]
- 14.Van Agtmael T, Schlötzer-Schrehardt U, McKie L, Brownstein DG, Lee AW, Cross SH, Sado Y, Mullins JJ, Pöschl E, Jackson IJ: Dominant mutations of Col4a1 result in basement membrane defects which lead to anterior segment dysgenesis and glomerulopathy. Hum Mol Genet 14: 3161–3168, 2005 [DOI] [PubMed] [Google Scholar]
- 15.Favor J, Gloeckner CJ, Janik D, Klempt M, Neuhäuser-Klaus A, Pretsch W, Schmahl W, Quintanilla-Fend L: Type IV procollagen missense mutations associated with defects of the eye, vascular stability, the brain, kidney function and embryonic or postnatal viability in the mouse, Mus musculus: An extension of the Col4a1 allelic series and the identification of the first two Col4a2 mutant alleles. Genetics 175: 725–736, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Plaisier E, Gribouval O, Alamowitch S, Mougenot B, Prost C, Verpont MC, Marro B, Desmettre T, Cohen SY, Roullet E, Dracon M, Fardeau M, Van Agtmael T, Kerjaschki D, Antignac C, Ronco P: COL4A1 mutations and hereditary angiopathy, nephropathy, aneurysms, and muscle cramps. N Engl J Med 357: 2687–2695, 2007 [DOI] [PubMed] [Google Scholar]
- 17.Plaisier E, Alamowitch S, Gribouval O, Mougenot B, Gaudric A, Antignac C, Roullet E, Ronco P: Autosomal-dominant familial hematuria with retinal arteriolar tortuosity and contractures: A novel syndrome. Kidney Int 67: 2354–2360, 2005 [DOI] [PubMed] [Google Scholar]
- 18.Plaisier E, Chen Z, Gekeler F, Benhassine S, Dahan K, Marro B, Alamowitch S, Paques M, Ronco P: Novel COL4A1 mutations associated with HANAC syndrome: A role for the triple helical CB3[IV] domain. Am J Med Genet A 152A: 2550–2555, 2010 [DOI] [PubMed] [Google Scholar]
- 19.Alamowitch S, Plaisier E, Favrole P, Prost C, Chen Z, Van Agtmael T, Marro B, Ronco P: Cerebrovascular disease related to COL4A1 mutations in HANAC syndrome. Neurology 73: 1873–1882, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kuo DS, Labelle-Dumais C, Mao M, Jeanne M, Kauffman WB, Allen J, Favor J, Gould DB: Allelic heterogeneity contributes to variability in ocular dysgenesis, myopathy and brain malformations caused by Col4a1 and Col4a2 mutations. Hum Mol Genet 23: 1709–1722, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jais JP, Knebelmann B, Giatras I, De Marchi M, Rizzoni G, Renieri A, Weber M, Gross O, Netzer KO, Flinter F, Pirson Y, Verellen C, Wieslander J, Persson U, Tryggvason K, Martin P, Hertz JM, Schröder C, Sanak M, Krejcova S, Carvalho MF, Saus J, Antignac C, Smeets H, Gubler MC: X-linked Alport syndrome: Natural history in 195 families and genotype- phenotype correlations in males. J Am Soc Nephrol 11: 649–657, 2000 [DOI] [PubMed] [Google Scholar]
- 22.Bekheirnia MR, Reed B, Gregory MC, McFann K, Shamshirsaz AA, Masoumi A, Schrier RW: Genotype-phenotype correlation in X-linked Alport syndrome. J Am Soc Nephrol 21: 876–883, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Storey H, Savige J, Sivakumar V, Abbs S, Flinter FA: COL4A3/COL4A4 mutations and features in individuals with autosomal recessive Alport syndrome. J Am Soc Nephrol 24: 1945–1954, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Miner JH: Developmental biology of glomerular basement membrane components. Curr Opin Nephrol Hypertens 7: 13–19, 1998 [DOI] [PubMed] [Google Scholar]
- 25.Kreidberg JA, Donovan MJ, Goldstein SL, Rennke H, Shepherd K, Jones RC, Jaenisch R: Alpha 3 beta 1 integrin has a crucial role in kidney and lung organogenesis. Development 122: 3537–3547, 1996 [DOI] [PubMed] [Google Scholar]
- 26.Kanasaki K, Kanda Y, Palmsten K, Tanjore H, Lee SB, Lebleu VS, Gattone VH, Jr, Kalluri R: Integrin beta1-mediated matrix assembly and signaling are critical for the normal development and function of the kidney glomerulus. Dev Biol 313: 584–593, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Korhonen M, Ylänne J, Laitinen L, Virtanen I: The α 1-α 6 subunits of integrins are characteristically expressed in distinct segments of developing and adult human nephron. J Cell Biol 111: 1245–1254, 1990 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zent R, Bush KT, Pohl ML, Quaranta V, Koshikawa N, Wang Z, Kreidberg JA, Sakurai H, Stuart RO, Nigám SK: Involvement of laminin binding integrins and laminin-5 in branching morphogenesis of the ureteric bud during kidney development. Dev Biol 238: 289–302, 2001 [DOI] [PubMed] [Google Scholar]
- 29.Delwel GO, de Melker AA, Hogervorst F, Jaspars LH, Fles DL, Kuikman I, Lindblom A, Paulsson M, Timpl R, Sonnenberg A: Distinct and overlapping ligand specificities of the alpha 3A beta 1 and alpha 6A beta 1 integrins: Recognition of laminin isoforms. Mol Biol Cell 5: 203–215, 1994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Miner JH, Sanes JR: Collagen IV alpha 3, alpha 4, and alpha 5 chains in rodent basal laminae: Sequence, distribution, association with laminins, and developmental switches. J Cell Biol 127: 879–891, 1994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lennerz JK, Spence DC, Iskandar SS, Dehner LP, Liapis H: Glomerulocystic kidney: One hundred-year perspective. Arch Pathol Lab Med 134: 583–605, 2010 [DOI] [PubMed] [Google Scholar]
- 32.Smeets B, Uhlig S, Fuss A, Mooren F, Wetzels JF, Floege J, Moeller MJ: Tracing the origin of glomerular extracapillary lesions from parietal epithelial cells. J Am Soc Nephrol 20: 2604–2615, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Smeets B, Kuppe C, Sicking EM, Fuss A, Jirak P, van Kuppevelt TH, Endlich K, Wetzels JF, Gröne HJ, Floege J, Moeller MJ: Parietal epithelial cells participate in the formation of sclerotic lesions in focal segmental glomerulosclerosis. J Am Soc Nephrol 22: 1262–1274, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Shankland SJ, Smeets B, Pippin JW, Moeller MJ: The emergence of the glomerular parietal epithelial cell. Nat Rev Nephrol 10: 158–173, 2014 [DOI] [PubMed] [Google Scholar]
- 35.Shrivastava A, Radziejewski C, Campbell E, Kovac L, McGlynn M, Ryan TE, Davis S, Goldfarb MP, Glass DJ, Lemke G, Yancopoulos GD: An orphan receptor tyrosine kinase family whose members serve as nonintegrin collagen receptors. Mol Cell 1: 25–34, 1997 [DOI] [PubMed] [Google Scholar]
- 36.Fu HL, Valiathan RR, Arkwright R, Sohail A, Mihai C, Kumarasiri M, Mahasenan KV, Mobashery S, Huang P, Agarwal G, Fridman R: Discoidin domain receptors: Unique receptor tyrosine kinases in collagen-mediated signaling. J Biol Chem 288: 7430–7437, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kerroch M, Guerrot D, Vandermeersch S, Placier S, Mesnard L, Jouanneau C, Rondeau E, Ronco P, Boffa JJ, Chatziantoniou C, Dussaule JC: Genetic inhibition of discoidin domain receptor 1 protects mice against crescentic glomerulonephritis. FASEB J 26: 4079–4091, 2012 [DOI] [PubMed] [Google Scholar]
- 38.Guerrot D, Kerroch M, Placier S, Vandermeersch S, Trivin C, Mael-Ainin M, Chatziantoniou C, Dussaule JC: Discoidin domain receptor 1 is a major mediator of inflammation and fibrosis in obstructive nephropathy. Am J Pathol 179: 83–91, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Teixeira VP, Blattner SM, Li M, Anders HJ, Cohen CD, Edenhofer I, Calvaresi N, Merkle M, Rastaldi MP, Kretzler M: Functional consequences of integrin-linked kinase activation in podocyte damage. Kidney Int 67: 514–523, 2005 [DOI] [PubMed] [Google Scholar]
- 40.El-Aouni C, Herbach N, Blattner SM, Henger A, Rastaldi MP, Jarad G, Miner JH, Moeller MJ, St-Arnaud R, Dedhar S, Holzman LB, Wanke R, Kretzler M: Podocyte-specific deletion of integrin-linked kinase results in severe glomerular basement membrane alterations and progressive glomerulosclerosis. J Am Soc Nephrol 17: 1334–1344, 2006 [DOI] [PubMed] [Google Scholar]
- 41.Dai C, Stolz DB, Bastacky SI, St-Arnaud R, Wu C, Dedhar S, Liu Y: Essential role of integrin-linked kinase in podocyte biology: Bridging the integrin and slit diaphragm signaling. J Am Soc Nephrol 17: 2164–2175, 2006 [DOI] [PubMed] [Google Scholar]
- 42.Wickström SA, Lange A, Montanez E, Fässler R: The ILK/PINCH/parvin complex: The kinase is dead, long live the pseudokinase! EMBO J 29: 281–291, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Joshi MB, Ivanov D, Philippova M, Erne P, Resink TJ: Integrin-linked kinase is an essential mediator for T-cadherin-dependent signaling via Akt and GSK3beta in endothelial cells. FASEB J 21: 3083–3095, 2007 [DOI] [PubMed] [Google Scholar]
- 44.Miwa N, Furuse M, Tsukita S, Niikawa N, Nakamura Y, Furukawa Y: Involvement of claudin-1 in the beta-catenin/Tcf signaling pathway and its frequent upregulation in human colorectal cancers. Oncol Res 12: 469–476, 2001 [DOI] [PubMed] [Google Scholar]
- 45.Wielenga VJ, Smits R, Korinek V, Smit L, Kielman M, Fodde R, Clevers H, Pals ST: Expression of CD44 in Apc and Tcf mutant mice implies regulation by the WNT pathway. Am J Pathol 154: 515–523, 1999 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Novak A, Hsu SC, Leung-Hagesteijn C, Radeva G, Papkoff J, Montesano R, Roskelley C, Grosschedl R, Dedhar S: Cell adhesion and the integrin-linked kinase regulate the LEF-1 and beta-catenin signaling pathways. Proc Natl Acad Sci U S A 95: 4374–4379, 1998 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Somasiri A, Howarth A, Goswami D, Dedhar S, Roskelley CD: Overexpression of the integrin-linked kinase mesenchymally transforms mammary epithelial cells. J Cell Sci 114: 1125–1136, 2001 [DOI] [PubMed] [Google Scholar]
- 48.Gil D, Ciołczyk-Wierzbicka D, Dulińska-Litewka J, Zwawa K, McCubrey JA, Laidler P: The mechanism of contribution of integrin linked kinase (ILK) to epithelial-mesenchymal transition (EMT). Adv Enzyme Regul 51: 195–207, 2011 [DOI] [PubMed] [Google Scholar]
- 49.Troussard AA, Costello P, Yoganathan TN, Kumagai S, Roskelley CD, Dedhar S: The integrin linked kinase (ILK) induces an invasive phenotype via AP-1 transcription factor-dependent upregulation of matrix metalloproteinase 9 (MMP-9). Oncogene 19: 5444–5452, 2000 [DOI] [PubMed] [Google Scholar]
- 50.Li Y, Yang J, Dai C, Wu C, Liu Y: Role for integrin-linked kinase in mediating tubular epithelial to mesenchymal transition and renal interstitial fibrogenesis. J Clin Invest 112: 503–516, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Zhang YH, Huang BL, Eastman K, McCabe LL, MacLennan NK, McCabe ER: Mouth cell collection device for newborn mice. Mol Genet Metab 89: 164–167, 2006 [DOI] [PubMed] [Google Scholar]
- 52.Regele HM, Fillipovic E, Langer B, Poczewki H, Kraxberger I, Bittner RE, Kerjaschki D: Glomerular expression of dystroglycans is reduced in minimal change nephrosis but not in focal segmental glomerulosclerosis. J Am Soc Nephrol 11: 403–412, 2000 [DOI] [PubMed] [Google Scholar]
- 53.Mook OR, Van Overbeek C, Ackema EG, Van Maldegem F, Frederiks WM: In situ localization of gelatinolytic activity in the extracellular matrix of metastases of colon cancer in rat liver using quenched fluorogenic DQ-gelatin. J Histochem Cytochem 51: 821–829, 2003 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.