Abstract
Hemodialysis vascular accesses are prone to recurrent stenosis and thrombosis after endovascular interventions. In vitro data suggest that indoxyl sulfate, a protein-bound uremic toxin, may induce vascular dysfunction and thrombosis. However, there is no clinical evidence regarding the role of indoxyl sulfate in hemodialysis vascular access. From January 2010 to June 2013, we prospectively enrolled patients undergoing angioplasty for dialysis access dysfunction. Patients were stratified into tertiles by baseline serum indoxyl sulfate levels. Study participants received clinical follow-up at 6-month intervals until June 2014. Primary end points were restenosis, thrombosis, and failure of vascular access. Median follow-up duration was 32 months. Of the 306 patients enrolled, 262 (86%) had symptomatic restenosis, 153 (50%) had access thrombosis, and 25 (8%) had access failure. In patients with graft access, free indoxyl sulfate tertiles showed a negative association with thrombosis-free patency (thrombosis-free patency rates of 54%, 38%, and 26% for low, middle, and high tertiles, respectively; P=0.001). Patients with graft thrombosis had higher free and total indoxyl sulfate levels. Using multivariate Cox regression analysis, graft thrombosis was independently predicted by absolute levels of free indoxyl sulfate (hazard ratio=1.14; P=0.01) and free indoxyl sulfate tertiles (high versus low, hazard ratio=2.41; P=0.001). Results of this study provide translational evidence that serum indoxyl sulfate is a novel risk factor for dialysis graft thrombosis after endovascular interventions.
Keywords: hemodialysis access, thrombosis, arteriovenous shunt
Hemodialysis vascular access dysfunction continues to be a major source of morbidity and mortality in patients with ESRD. After publication of the dialysis outcome quality initiative guidelines, endovascular interventions gradually replaced surgical revisions as the primary treatment of dysfunctional dialysis access.1 Although percutaneous transluminal angioplasty (PTA) can achieve a high success rate, recurrent stenosis and thrombosis are usually inevitable. As a result, repeated interventions are required and cause a substantial financial burden on the health care system.2
Intimal hyperplasia at the outflow vein is the most common cause of vascular access dysfunction.3 Thrombosis may develop secondary to outflow venous stenosis, but it can also develop without underlying anatomic abnormalities.4,5 Traditional cardiovascular risk factors are insufficient to explain such a high restenosis and thrombosis rate.6 Physiologic and anatomic differences between arteries and veins, hemodynamic stress, repeated puncture, thrombophilia, endothelial dysfunction, and uremic milieu have all been proposed as possible contributors.3
When renal failure develops, uremic retention solutes are retained. If these retention solutes exert biochemical or biologic activity, they are called uremic toxins.7 Indoxyl sulfate is one of the protein-bound uremic toxins that results from metabolism of dietary tryptophan. Recently, indoxyl sulfate has been shown to act as a vascular toxin through inhibition of endothelial cell repair, impairment of endothelial function, induction of inflammatory response, and stimulation of smooth muscle proliferation.8–11 In addition, uremic serum from hemodialysis patients has been shown to be profoundly thrombogenic and can increase the risk of postinterventional thrombosis. Recent clinical studies have also demonstrated that serum indoxyl sulfate was associated with vascular disease in patients with CKD and cardiovascular disease in hemodialysis patients.12–15
Despite evidence suggesting a critical role of indoxyl sulfate in vascular dysfunction, no clinical information is available concerning its effect on postangioplasty outcome of dialysis access. The principal aim of this prospective observational cohort study was to investigate the relation between serum levels of indoxyl sulfate and the outcome of dialysis access after PTA.
Results
Baseline Data
From January 2010 to June 2013, a total of 333 patients were prospectively enrolled. Twenty-seven patients were excluded because of arterial stenosis, central vein stenosis, failed PTA procedures, lack of significant stenosis on the basis of angiography, or recent acute illness. The final study group consisted of 306 patients (mean age, 69±12 years). The patients’ median shunt age was 39 months (interquartile range [IQR], 24–72 months), and 175 patients had graft access. Other baseline characteristics of the study participants and their shunts are provided in Table 1.
Table 1.
Baseline demographics and biochemical and vascular access characteristics
| Factor | Total (n=306) | Graft (n=175) | P Value | Native (n=131) | P Value | ||||
|---|---|---|---|---|---|---|---|---|---|
| IS/Low (n=59) | IS/Middle (n=58) | IS/High (n=58) | IS/Low (n=43) | IS/Middle (n=44) | IS/High (n=44) | ||||
| Clinical data | |||||||||
| Age (yr) | 69±12 | 68±13 | 70±11 | 69±12 | 0.79 | 68±15 | 67±11 | 69±12 | 0.88 |
| Sex (men/women) | 123/183 | 24/35 | 20/38 | 21/37 | 0.77 | 16/27 | 22/22 | 20/24 | 0.48 |
| Dialysis vintage (mo) | 48 (26–84) | 53 (34–72) | 55 (30–87) | 44 (24–84) | 0.41 | 36 (24–90) | 52 (26–84) | 55 (26–84) | 0.82 |
| Hypertension | 186 (61) | 34 (58) | 40 (69) | 34 (59) | 0.38 | 26 (61) | 24 (55) | 28 (64) | 0.68 |
| Diabetes | 157 (51) | 25 (42) | 31 (53) | 34 (5) | 0.20 | 17 (40) | 25 (57) | 25 (57) | 0.18 |
| Dyslipidemia | 60 (20) | 11 (19) | 9 (16) | 10 (17) | 0.90 | 11 (26) | 9 (21) | 10 (23) | 0.85 |
| Current smoker | 50 (16) | 7 (12) | 15 (26) | 10 (17) | 0.14 | 4 (9) | 9 (21) | 5 (11) | 0.27 |
| CVD | 138 (45) | 29 (49) | 19 (33) | 28 (48) | 0.13 | 25 (58) | 17 (39) | 20 (46) | 0.18 |
| Medication data | |||||||||
| Antiplatelet | 162(53) | 29 (49) | 26 (45) | 28 (48) | 0.89 | 28 (65) | 26 (59) | 25 (43) | 0.72 |
| Aspirin | 135 | 25 | 23 | 23 | 23 | 23 | 18 | ||
| Clopidogrel | 11 | 1 | 3 | 1 | 3 | 0 | 3 | ||
| Cilostazol | 30 | 7 | 5 | 3 | 7 | 6 | 2 | ||
| Anticoagulant | 12 (4) | 3 (5) | 3 (5) | 2 (3) | 0.88 | 1 (2) | 0 (0) | 3 (7) | 0.17 |
| INR>1.5 | 6 | 1 | 2 | 1 | 1 | 0 | 1 | ||
| INR<1.5 | 6 | 2 | 1 | 1 | 0 | 0 | 2 | ||
| Lipid-lowering agents | 25 (8) | 5 (10) | 5 (10) | 5 (10) | 0.99 | 4 (12) | 3 (8) | 3 (7) | 0.74 |
| Access/lesion data | |||||||||
| Shunt age (mo) | 39 (24–72) | 42 (24–63) | 40 (24–74) | 36 (20–57) | 0.41 | 36 (23–93) | 48 (26–80) | 48 (25–81) | 0.82 |
| Lower/upper arm | 236/70 | 40/19 | 45/13 | 46/12 | 0.30 | 37/6 | 33/11 | 35/9 | 0.60 |
| Left/right arm | 237/69 | 47/12 | 45/13 | 41/17 | 0.50 | 36/7 | 33/11 | 35/9 | 0.60 |
| Restenosis lesion | 278 (91) | 55 (93) | 56 (97) | 51 (88) | 0.20 | 37 (86) | 38 (86) | 41 (93) | 0.50 |
| Pre-PTA stenosis (%) | 76±15 | 77±15 | 76±17 | 74±14 | 0.54 | 78±15 | 75±16 | 76±12 | 0.71 |
| Post-PTA stenosis (%) | 5±9 | 7±10 | 7±10 | 3±7 | 0.04 | 6±10 | 5±9 | 5±8 | 0.75 |
| Previous PTA (time) | 6.1±6.8 | 8.0±7.4 | 9.0±8.5 | 6.3±7.4 | 0.18 | 3.8±3.7 | 3.7±4.6 | 4.4±4.5 | 0.69 |
| Operator experience (yr) | 9.0±4.5 | 9.1±4.3 | 8.9±4.5 | 9.1±4.8 | 0.96 | 9.2±4.9 | 9.0±4.6 | 8.6±4.2 | 0.82 |
| Biochemical data | |||||||||
| Cholesterol (mg/dl) | 164±38 | 168±36 | 164±39 | 157±36 | 0.36 | 171±39 | 161±46 | 162±32 | 0.50 |
| Triglycerides (mg/dl) | 147 (95–223) | 138 (94–216) | 160 (89–228) | 158 (114– 243) | 0.71 | 147 (97–208) | 134 (93–212) | 139 (97–212) | 0.83 |
| Albumin (g/dl) | 3.80±0.38 | 3.73±0.30 | 3.82±0.43 | 3.75±0.43 | 0.41 | 3.91±0.33 | 3.77±0.41 | 3.80±0.30 | 0.14 |
| Hemoglobin (g/dl) | 10.6±1.4 | 10.3±1.5 | 10.9±1.2 | 10.4±1.3 | 0.04 | 11.0±1.3 | 10.8±1.3 | 10.4±1.4 | 0.12 |
| Calcium (mg/dl) | 9.4±1.0 | 9.5±0.9 | 9.2±0.8 | 9.6±1.0 | 0.08 | 9.4±1.3 | 9.2±0.9 | 9.3±1.0 | 0.65 |
| Phosphate (mg/dl) | 4.7±1.5 | 5.0±1.9 | 4.5±1.2 | 4.6±1.7 | 0.23 | 4.8±1.3 | 4.8±1.2 | 4.8±1.6 | 0.99 |
| Ca×P (mg2/dl2) | 50±16 | 48±20 | 42±12 | 45±18 | 0.19 | 46±14 | 45±11 | 44±15 | 0.84 |
| Creatinine (mg/dl)a | 8.5±2.4 | 8.2±1.9 | 8.8±2.3 | 8.6±2.5 | 0.35 | 8.5±2.8 | 8.5±2.1 | 8.6±2.7 | 0.97 |
| Kt/V | 1.47±0.32 | 1.47±0.35 | 1.49±0.25 | 1.49±0.26 | 0.90 | 1.49±0.29 | 1.46±0.44 | 1.43±0.36 | 0.72 |
| RKt/V | 0.08±0.18 | 0.06±0.16 | 0.05±0.17 | 0.03±0.13 | 0.75 | 0.11±0.21 | 0.05±0.14 | 0.06±0.15 | 0.19 |
| hs-CRP (mg/L) | 2.13±3.36 | 2.21±3.27 | 2.79±5.48 | 2.20±3.96 | 0.70 | 1.82±1.24 | 1.71±0.77 | 1.80±1.40 | 0.90 |
| Homocysteine (μM) | 20±11 | 19±4 | 21±7 | 20±5 | 0.14 | 22±16 | 20±3 | 20±4 | 0.53 |
| IS, total (μg/ml) | 7.0 (4.4–9.2) | 3.8 (2.7–6.2) | 6.3 (4.3–8.2) | 9.1 (7.3–11.2) | <0.001 | 5.0 (3.4–7.7) | 6.3 (4.3–8.2) | 9.4 (6.5–12.3) | <0.001 |
| IS, free (μg/ml) | 2.2 (1.2–3.5) | 1.0 (0.6–1.3) | 2.1 (1.8–2.5) | 4.2 (3.4–5.8) | <0.001 | 0.9 (0.6–1.2) | 2.1 (1.8–2.5) | 4.0 (3.4–5.7) | <0.001 |
Values are mean±SD, median (interquartile range, 25%–75%), n (%), or as otherwise indicated. CVD, cardiovascular disease; INR, international normalize ratio; Ca×P, calcium×phosphate; RKt/V, residual urea clearance; IS, indoxyl sulfate.
Predialysis creatinine.
When participants were stratified into three groups according to their baseline free indoxyl sulfate levels, there were no differences in clinical, medication, access, procedural, or biochemical data. When the indoxyl sulfate level was treated as a continuous variable, it correlated significantly with diabetes only.
Follow-Up
During the study period, two patients received kidney transplants. One had an access thrombosis event before transplantation, and the other was censored. No patient was transferred to peritoneal dialysis. Eight patients were transferred to other nonstudy dialysis centers: five had access events before, and the others were censored. The median follow-up duration was 32 months (IQR, 19–47 months).
Nonaccess Events
Of the 306 patients, 53 died, and 18 of them died of cardiac diseases. Table 2 shows the clinical and access outcomes stratified by the tertiles of free indoxyl sulfate. The tertiles did not correlate with the incidence of all-cause or cardiac-cause death. Fifty-five patients experienced nonaccess thrombotic events, with a nonsignificant trend parallel to the indoxyl sulfate tertiles. Details of respective thrombotic events are listed in Table 2. When nonaccess thrombosis events were pooled with cardiac death, a significant correlation with the free indoxyl sulfate tertiles was observed (low, 14%; middle, 22%; high, 28%; P=0.04).
Table 2.
Access and nonaccess outcomes stratified by the tertile of free indoxyl sulfate
| Factor | Tertile of Free Indoxyl Sulfate | |||
|---|---|---|---|---|
| Low (n=102) | Medium (n=102) | High (n=102) | P Value | |
| Access events | ||||
| Restenosis | 90 (88) | 86 (84) | 86 (84) | 0.65 |
| Thrombosis | 40 (39) | 50 (49) | 63 (62) | 0.01 |
| Failure | 6 (6) | 12 (12) | 7 (7) | 0.26 |
| Nonaccess events | ||||
| All vascular thrombosis | 12 (12) | 19 (19) | 24 (24) | 0.09 |
| Coronary artery thrombosis | 9 (9) | 10 (10) | 14 (14) | |
| Cerebral artery thrombosis | 1 (1) | 5 (5) | 4 (4) | |
| Peripheral artery thrombosis | 1 (1) | 3 (3) | 3 (3) | |
| DVT/PE | 1 (1) | 1 (1) | 3 (3) | |
| Death | ||||
| Any cause | 16 (16) | 16 (16) | 21 (21) | 0.55 |
| Cardiac cause | 4 (4) | 4 (4) | 10 (10) | 0.12 |
| Composite outcomes | ||||
| Access or nonaccess thrombosis | 42 (41) | 53 (52) | 68 (67) | 0.001 |
| Access thrombosis or death | 49 (48) | 59 (58) | 72 (71) | 0.01 |
| Nonaccess thrombosis or cardiac death | 14 (14) | 22 (22) | 29 (28) | 0.04 |
| Nonaccess thrombosis or death | 25 (25) | 31 (30) | 35 (34) | 0.31 |
Values are n (%) or as otherwise indicated. DVT, deep vein thrombosis; PE, pulmonary embolism.
Access Restenosis
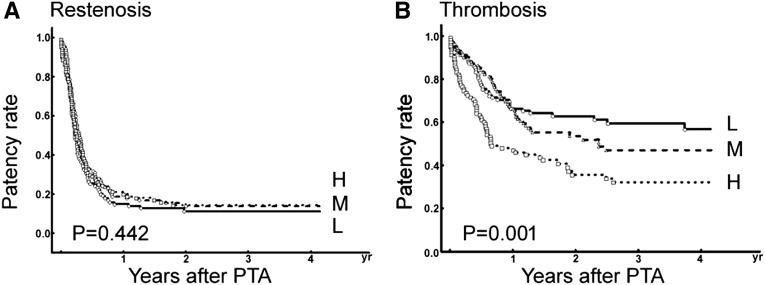
Of the 306 patients, 262 had restenosis and received reinterventions. The restenosis rate was not related to the free indoxyl sulfate tertiles (Table 2). Kaplan–Meier plots showed that free indoxyl sulfate tertiles were not associated with restenosis (Figure 1). On univariate Cox proportional hazard model, neither absolute nor tertiles of indoxyl sulfate was associated with restenosis-free patency. When restenosis were evaluated by quantitative parameters, serum free indoxyl sulfate levels were not correlated with pre-PTA stenosis (r=0.03, P=0.57), relative late loss (r=0.02, P=0.69), absolute late loss (r=–0.01, P=0.82), relative late loss rate (r=0.05, P=0.33), or absolute late loss rate (r=–0.08, P=0.18). Twenty-five patients had access failure, which was not related to serum indoxyl sulfate levels.
Figure 1.
Kaplan–Meier curves of access patency. (A) Restenosis-free patency and (B) thrombosis-free patency stratified by the tertiles of baseline free indoxyl sulfate levels. P values were tested by log-rank test. H, high; L, low; M, middle.
Access Thrombosis
During the follow-up period, 153 of the 306 patients experienced vascular access thrombosis. In Table 2, a significant association between free indoxyl sulfate tertile and vascular access thrombosis was observed (low, 39%; middle, 49%; high, 62%; P=0.01). Nonetheless, the association was significant in patients with graft (low, 46%; middle, 62%; high, 74%; P=0.01) but not in those with native accesses (low, 28%; middle, 34%; high, 38%; P=0.58).
In Kaplan–Meier analysis, thrombosis free patency of vascular access was associated with serum indoxyl sulfate tertiles with a graded effect demonstrated (low, 61%; middle, 51%; high, 38%, P=0.001) (Figure 1).
Subgroup Analysis
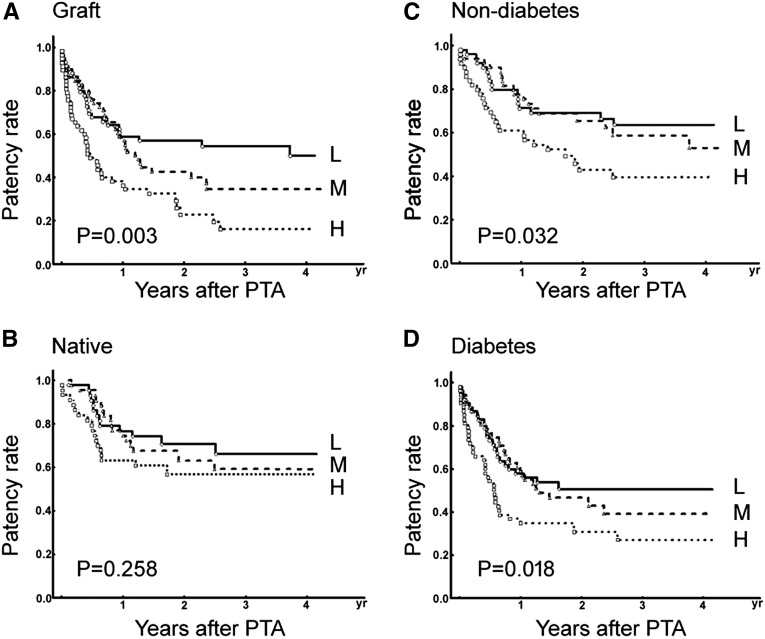
When patients was stratified by access type, free indoxyl sulfate tertiles revealed an increasingly negative effect on thrombosis-free patency in patients with graft accesses (low, 54%; middle, 38%; high, 26%, P=0.001) but not patients with native access (low, 70%; middle, 64%; high, 59%, P=0.43) (Figure 2). We divided all study subjects by diabetes, a homogenously negative effect in both the diabetic group (low, 51%; middle, 39%; high, 27%, P=0.018) and nondiabetic group (low, 64%; middle, 53%; high, 40%, P=0.032) was observed (Figure 2).
Figure 2.
Kaplan–Meier curves of thrombosis-free patency by groups. (A) Graft access, (B) native access, (C) nondiabetic patients, and (D) diabetic patients stratified by the tertiles of baseline free indoxyl sulfate levels. P values were tested by log-rank test. H, high; L, low; M, middle; PTA, percutaneous transluminal angioplasty.
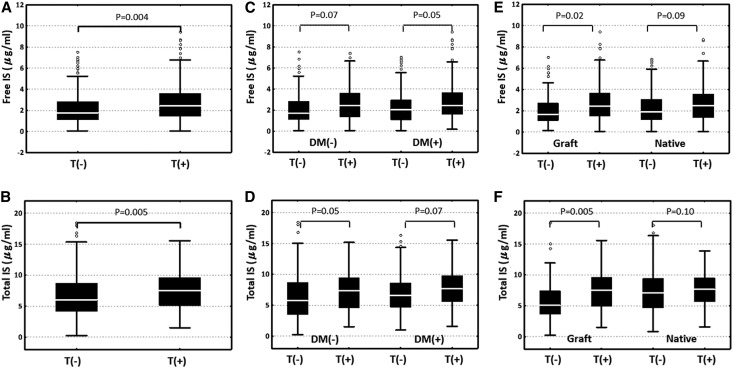
Patients with access thrombosis had higher median free and total indoxyl sulfate levels compared with those without thrombosis. Nonetheless, the trend of difference remained when patients were stratified by the status of diabetes, but they were only significant for graft accesses when stratified by access type (Figure 3).
Figure 3.
Comparisons of indoxyl sulfate levels between patients with and without access thrombosis. (A and B) All participants, (C and D) stratified by diabetes or nondiabetes, and (E and F) stratified by graft or native accesses. Upper row (A, C, and E): free form; and lower row (B, D, and F): total form. Whisker plots show the 10th, 25th, 50th, 75th, and 90th percentile distributions in each panel. P values were tested by Mann–Whitney U test. DM, diabetes; IS, indoxyl sulfate; T, thrombosis.
Univariate and Multivariate Cox Models
On univariate Cox proportional hazards analysis, diabetes, current smoker, cardiovascular disease history, graft access, previous PTA, post-PTA stenosis, high sensitive C-reactive protein (hs-CRP), and indoxyl sulfate were significantly associated with vascular access thrombosis (Table 3). On the basis of the findings from univariate analysis, the following parameters were entered into the multivariate analysis: diabetes, current smoker, cardiovascular disease history, type and side of access, previous PTA, post-PTA stenosis, Kt/V, hs-CRP, and free indoxyl sulfate. In multivariate analysis, graft access, previous PTA frequency, and free indoxyl sulfate were consistently retained as independent risk factors (Table 4).
Table 3.
Univariate Cox regression analysis for factors predicting access thrombosis
| Factor | Unit of Increase | All Access | Graft Access | ||
|---|---|---|---|---|---|
| HR (95% CI) | P Value | HR (95% CI) | P Value | ||
| Age | 1 yr | 1.01 (0.99 to 1.02) | 0.45 | 1.00 (0.99 to 1.02) | 0.82 |
| Sex | Men | 0.98 (0.71 to 1.36) | 0.90 | 0.72 (0.48 to 1.09) | 0.12 |
| Dialysis vintage | 1 mo | 1.00 (0.99 to 1.01) | 0.71 | 1.00 (0.99 to 1.00) | 0.70 |
| Hypertension | Yes versus no | 1.20 (0.86 to 1.67) | 0.28 | 1.18 (0.79 to 1.75) | 0.43 |
| Diabetes | Yes versus no | 1.50 (1.08 to 2.06) | 0.02 | 2.21 (1.48 to 3.28) | <0.001 |
| Dyslipidemia | Yes versus no | 1.08 (0.64 to 1.83) | 0.76 | 1.10 (0.68 to 1.79) | 0.70 |
| Current smoker | Yes versus no | 1.83 (1.24 to 2.63) | 0.002 | 1.68 (1.07 to 2.63) | 0.02 |
| Cardiovascular disease | Yes versus no | 1.47 (1.07 to 2.02) | 0.02 | 1.56 (1.07 to 2.09) | 0.02 |
| Antiplatelet | Yes versus no | 0.77 (0.56 to 1.05) | 0.10 | 0.85 (0.58 to 1.25) | 0.41 |
| Anticoagulant | Yes versus no | 1.78 (0.83 to 3.79) | 0.14 | 1.70 (0.74 to 3.88) | 0.21 |
| Lipid-lowering agent | Yes versus no | 1.50 (0.88 to 2.52) | 0.13 | 1.22 (0.63 to 2.35) | 0.56 |
| Shunt age | 1 mo | 0.99 (1.00 to 1.00) | 0.22 | 0.99 (0.99 to 1.00) | 0.06 |
| Type of access | Graft | 2.12 (1.51 to 2.99) | <0.001 | — | — |
| Location of access | Upper | 1.08 (0.73 to 1.59) | 0.70 | 0.80 (0.50 to 1.26) | 0.33 |
| Side of access | Right | 0.73 (0.51 to 1.04) | 0.09 | 1.22 (0.79 to 1.88) | 0.36 |
| Restenosis lesion | Yes versus no | 1.42 (0.77 to 2.63) | 0.26 | 1.04 (0.50 to 2.11) | 0.92 |
| Pre-PTA stenosis | 1% | 1.00 (1.00 to 1.01) | 0.71 | 1.00 (0.99 to 1.01) | 0.95 |
| Post-PTA stenosis | 1% | 1.02 (1.00 to 1.03) | 0.04 | 1.03 (1.01 to 1.05) | 0.01 |
| Previous PTA | 1 time | 1.18 (1.12 to 1.24) | <0.001 | 1.14 (1.08 to 1.21) | <0.001 |
| Operator experience | 1 yr | 1.01 (0.97 to 1.04) | 0.80 | 1.01 (0.97 to 1.05) | 0.68 |
| Total cholesterol | 1 mg/dl | 1.00 (1.00 to 1.01) | 0.29 | 1.00 (0.99 to 1.00) | 0.71 |
| Triglycerides | 1 mg/dl | 1.00 (1.00 to 1.00) | 0.44 | 1.00 (1.00 to 1.00) | 0.31 |
| Albumin | 1 g/dl | 0.78 (0.47 to 1.10) | 0.13 | 0.79 (0.47 to 1.34) | 0.38 |
| Hemoglobin | 1 g/dl | 0.96 (0.85 to 1.08) | 0.46 | 1.09 (0.94 to 1.27) | 0.26 |
| Calcium | 1 mg/dl | 0.93 (0.78 to 1.10) | 0.39 | 0.98 (0.78 to 1.24) | 0.88 |
| Phosphate | 1 mg/dl | 1.00 (0.89 to 1.12) | 0.99 | 0.95 (0.84 to 1.08) | 0.44 |
| Calcium × phosphate | 1 mg2/dl2 | 0.64 (0.99 to 1.00) | 0.99 | 1.00 (0.98 to 1.01) | 0.47 |
| Predialysis creatinine | 1 md/dl | 0.98 (0.16 to 1.05) | 0.56 | 0.93 (0.86 to 1.20) | 0.13 |
| Kt/V | 1 unit | 0.63 (0.39 to 1.03) | 0.07 | 0.59 (0.31 to 1.14) | 0.11 |
| RKt/V | 1 unit | 0.55 (0.21 to 1.43) | 0.22 | 0.43 (0.11 to 1.66) | 0.22 |
| Homocysteine | 1 μM | 0.98 (0.95 to 1.01) | 0.23 | 0.99 (0.95 to 1.02) | 0.51 |
| hs-CRP | 1 mg/L | 1.04 (1.01 to 1.08) | 0.003 | 1.03 (1.00 to 1.07) | 0.03 |
| Free indoxyl sulfate | 1 μg/ml | 1.14 (1.06 to 1.23) | 0.001 | 1.16 (1.05 to 1.27) | 0.002 |
| Middle versus low | 1.25 (0.82 to 1.89) | 0.39 | 1.40 (0.84 to 2.30) | 0.19 | |
| High versus low | 2.07 (1.39 to 3.08) | <0.001 | 2.37 (1.46 to 3.85) | <0.001 | |
| Total indoxyl sulfate | 1 μg/ml | 1.01 (1.00 to 1.02) | 0.04 | 1.06 (1.02 to 1.09) | 0.001 |
| Middle versus low | 1.62 (1.07 to 2.44) | 0.02 | 1.81 (1.11 to 2.94) | 0.02 | |
| High versus low | 1.93 (1.28 to 2.89) | 0.002 | 2.63 (1.65 to 4.33) | <0.001 | |
RKt/V, residual urea clearance; HR, hazard ratio; 95% CI, 95% confidence interval.
Table 4.
Multivariate Cox regression analysis for factors predicting access thrombosis
| Factor | Unit of Increase | All Access | Graft Access | ||
|---|---|---|---|---|---|
| HR (95% CI) | P Value | HR (95% CI) | P Value | ||
| Model A (free indoxyl sulfate entered as continuous variable) | |||||
| Diabetes | Yes versus no | 1.39 (1.98 to 1.97) | 0.06 | 1.74 (1.15 to 2.64) | 0.01 |
| Current smoker | Yes versus no | 1.38 (0.92 to 2.06) | 0.12 | 1.30 (0.80 to 2.11) | 0.30 |
| Cardiovascular disease | Yes versus no | 1.20 (0.85 to 1.69) | 0.30 | 1.23 (0.81 to 1-85) | 0.34 |
| Shunt age | 1 mo | — | — | 0.99 (0.99 to 1.00) | 0.04 |
| Type of access | Graft | 2.12 (1.51 to 2.99) | <0.001 | — | — |
| Side of access | Right | 1.35 (0.92 to 1.97) | 0.13 | — | — |
| Post-PTA stenosis | 1% | 1.02 (1.00 to 1.03) | 0.11 | 1.03 (1.00 to 1.05) | 0.03 |
| Previous PTA | 1 time | 1.12 (1.06 to 1.18) | <0.001 | 1.10 (1.04 to 1.17) | 0.001 |
| Kt/V | 1 unit | 0.68 (0.39 to 1.20) | 0.18 | — | — |
| hs-CRP | 1 mg/L | 1.03 (1.00 to 1.01) | 0.10 | 1.02 (0.99 to 1.05) | 0.29 |
| Free indoxyl sulfate | 1 μg/ml | 1.11 (1.03 to 1.21) | 0.01 | 1.14 (1.03 to 1.25) | 0.01 |
| Model B (free indoxyl sulfate entered as categorical variable) | |||||
| Diabetes | Yes versus no | 1.36 (0.96 to 1.92) | 0.08 | 1.67 (1.10 to 2.54) | 0.02 |
| Current smoker | Yes versus no | 1.33 (0.89 to 1.99) | 0.17 | 1.20 (0.73 to 1.98) | 0.47 |
| Cardiovascular disease | Yes versus no | 1.24 (0.87 to 1.75) | 0.24 | 1.29 (0.84 to 1.97) | 0.24 |
| Shunt age | 1 month | — | — | 0.99 (0.99 to 1.00) | 0.03 |
| Type of access | Graft | 2.07 (1.44 to 2.96) | <0.001 | — | — |
| Side of access | Right | 1.32 (0.90 to 1.93) | 0.15 | — | — |
| Post-PTA stenosis | 1% | 1.02 (1.00 to 1.03) | 0.11 | 1.03 (1.00 to 1.05) | 0.03 |
| Previous PTA | 1 time | 1.13 (1.07 to 1.19) | <0.001 | 1.12 (1.06 to 1.20) | <0.001 |
| Kt/V | 1 unit | 0.69 (0.39 to 1.20) | 0.19 | — | — |
| hs-CRP | 1 mg/L | 1.02 (0.99 to 1.06) | 0.11 | 1.02 (0.99 to 1.05) | 0.31 |
| Free indoxyl sulfate | Middle versus low | 1.27 (0.82 to 1.98) | 0.28 | 1.37 (0.80 to 2.37) | 0.25 |
| High versus low | 2.04 (1.35 to 3.10) | 0.001 | 2.41 (1.45 to 4.01) | 0.001 | |
HR, hazard ratio; 95% CI, 95% confidence interval.
When patients with graft access were analyzed separately, diabetes, current smoker, cardiovascular disease history, previous PTA frequency, post-PTA stenosis, hs-CRP, and free indoxyl sulfate were significantly associated with graft thrombosis. In multivariate analysis, free indoxyl sulfate retained as an independent predictor (Table 4). When either residual renal function or dialysis vintage was forced into multivariate analysis, free indoxyl sulfate remained a significant predictor of thrombosis, both for all accesses or graft accesses only (Supplemental Tables 1 and 2).
Discussion
Main Findings
This prospective cohort study evaluated the independent role of indoxyl sulfate and other important clinical variables on the outcome of hemodialysis access after PTA. We found that high baseline free indoxyl sulfate levels were independently associated with risk of postangioplasty dialysis graft thrombosis. This association was not affected by other known clinical or biochemical factors, such as diabetes, active smoker, frequency of PTA, or hs-CRP levels.
Relevance of Restenosis and Thrombosis after Angioplasty
PTA is a well established therapy for dialysis access dysfunction. Nonetheless, at 1 year, only 26%–58% of native fistulas remained functional without subsequent intervention.16 Postangioplasty results were even worse for graft access. At 6 months, only 40%–50% remained functional without further intervention. If thrombosis developed, the outcome was even poorer, with a 3-month unassisted patency rate, which ranged from 30% to 40%.17 The postangioplasty outcomes from dialysis access in our study were similar to the previous data. Therefore, repeated interventions are usually required, which result in a substantial financial burden on the health care system.2
Vascular Access Thrombosis
Vascular thrombosis involves the interplay among blood stasis, vessel injury, and hypercoagulability.18 A major predisposing factor for thrombosis is anatomic stenosis, which causes blood stasis at the outflow veins.19 Nonetheless, access thrombosis may occur without outflow venous stenosis, and it remains unclear why some patients experience more thrombosis. Currently, there is paucity of strategies to prevent thrombosis. Both antiplatelet and anticoagulant agents showed inconclusive or negative results.20,21
Uremic patients have an elevated risk of both arterial and venous thrombosis.22 Previous studies have shown a prothrombotic state in dialysis patients.23–27 However, only limited data are available concerning the role of thrombophilia in vascular access thrombosis, and the results are inconsistent.20,28–31 Although we have shown only a mild association between indoxyl sulfate levels and vascular thrombosis, it was strengthened by a demonstrated biologic gradient. The association was supported by a similar trend in nonaccess thrombosis. The effect was more prominent in graft access, however, which usually showed a worse outcome. The discrepancy between graft and fistula may come from the unfavorable vascular anatomy of graft recipients, secondary to the access-creation policy.
Possible Pathogenic Mechanisms
Several pathogenic mechanisms may contribute to the association between indoxyl sulfate and vascular access thrombosis. Uremic patients have been shown to have higher concentrations of circulating tissue factors, which correlate with serum levels of indoxyl sulfate and indole-3-acetic acid but not with other uremic toxins. The increased levels of circulating tissue factors are associated with higher procoagulant activity. In an in vitro study, indoxyl sulfate and indole-3-acetic acid increased the expression of tissue factors in endothelial cells and peripheral blood mononuclear cells, but the free indoxyl sulfate level in that study may be too high because of absence of albumin in the medium.32 In a model of a de-endothelialized, postinterventional state, uremic serum obtained from patients with ESRD caused greater clot formation. Relevant uremic solutes, such as indoxyl sulfate and indole-3-acetic acid, recapitulated these effects in cell culture and a flow loop model. Indoxyl sulfate significant prolonged the half-lives of tissue factors by inhibiting their ubiquination.33 These observations suggest indoxyl sulfate itself may increase thrombosis risk through altered expression and stability of tissue factors at circulating tissue levels.
A second possible mechanism concerns re-endothelialization after injury. An intact endothelium possesses both antiplatelet and antithrombotic properties. Re-endothelialization is a critical factor attributed to vascular thrombosis after angioplasty or other kinds of injury. Indoxyl sulfate inhibits endothelial proliferation and impairs wound repair.34 Endothelial progenitor cells have been shown to mobilize and incorporate into the denuded vessel wall after balloon injury, a process associated with accelerated endothelial regeneration.35 In patients with AKI, indoxyl sulfate negatively correlated with endothelial progenitor cell levels and had a deleterious effect on progenitor cell function in vitro.36 The inhibitory role of indoxyl sulfate on progenitor cells was also demonstrated using another uremic solute, p-cresyl sulfate.8 Human studies have shown that deficiency of endothelial progenitor cells was related to restenosis and thrombosis after endovascular intervention.37,38 The effect of indoxyl sulfate on endothelial repair and progenitor cells may contribute to the development of vascular thrombosis.
Finally, indoxyl sulfate has been shown to induce endothelial dysfunction in patients with CKD through induction of oxidative stress, which is ameliorated by oral charcoal adsorbent, AST-120 administration.10 Moreover, indoxyl sulfate directly stimulates smooth muscle cell proliferation by mechanisms of inflammatory activation and profibrotic cytokine activation.11,15,39 The multifactorial effects of indoxyl sulfate on inflammation, reactive oxygen species production, and vascular smooth muscle cell proliferation may potentiate the development of vascular thrombosis.
Other Contributors to Thrombosis
For the first time, to our knowledge, we showed that a current smoking history associated with access thrombosis. Cigarette smoking is a well established risk factor for atherosclerotic disease, but its role in venous thromboembolism remains controversial.40 In addition to its atherosclerotic effect, exposure to cigarette smoke alters the hemostatic process via multiple mechanisms, including alterations of endothelial function, platelets, fibrinogen, and coagulation factors. These mechanisms may create an imbalance between thrombosis and fibrinolytic factors that precipitates thrombosis.41
Although diabetes is a leading cause of vascular disease, previous studies revealed inconsistent results regarding its role in dialysis access thrombosis. In the univariate model we found that diabetes increased the risk of thrombosis but not the risk of restenosis. Several mechanisms contribute to a prothrombotic state in patients with diabetes, including endothelial dysfunction, coagulation activation, and platelet hyper-reactivity, which result from the interaction among hyperglycemia, insulin resistance, inflammation, and oxidative stress on the vessels.42
Thrombosis within coronary arteries and deep veins is related to vessel inflammation. In hemodialysis patients, CRP has been shown to be associated with stenosis and thrombosis of fistulas.43 Pathologic examination revealed preferential expression of matrix metalloproteinase-9 near the vascular lumen of thrombosed arteriovenous fistulas, which may cause disruption of the neointimal, with subsequent exposure of the highly procoagulant subendothelial tissues to circulating blood.44
Restenosis
Indoxyl sulfate has been shown to play an important role in renal fibrosis by the mechanisms of inflammatory reaction and expression of profibrotic cytokines.45 In animal models, indoxyl sulfate has been shown to induce endothelial dysfunction and stimulate smooth muscle cell proliferation.11,15,39 These findings have been translated into vascular disease and cardiovascular mortality in patients with CKD.15 Our analysis, either clinical or angiographic restenosis, did not support a pivotal role in intimal hyperplasia of dialysis accesses. In this study, less than one-tenth of participants were treated for de novo lesions. In consequence, a high percentage of restenotic lesions may have blunted the opportunity to show their effect on intimal hyperplasia. In addition, a variety of uremic solutes have deleterious effect on vascular function, but only indoxyl sulfate was investigated in this study. The sources of intimal hyperplasia may be multifactorial, and indoxyl sulfate seems not to play a dominant role.
Nonaccess Events
Serum indoxyl sulfate was associated with both overall mortality and cardiovascular mortality at different stages of CKD, which only partially depended on vascular calcification or stiffness.15 Our results suggested that enhanced thrombotic risk could be a possible underlying mechanism. However, the effect of serum indoxyl sulfate on nonaccess events was not as prominent as its effect on access events. Repeated puncture and hemostasis, frequent interventions, uncommon use of stents, and inadequate administration of antiplatelet agents may have contributed to the disparity between access and nonaccess thrombosis.
In contrast with patients with CKD, indoxyl sulfate was not associated with either overall mortality or cardiovascular mortality in this hemodialysis cohort, in accordance with previous observations.46,47 This result may have been secondary to advanced alternation in cardiovascular systems in hemodialysis patients compared with nondialysis CKD patients. Alternatively, only one-third of hemodialysis patients died from cardiovascular causes in this cohort, therefore reducing the role played by indoxyl sulfate in these patients’ mortality.
Study Limitations
Some limitations should be addressed. First, this is an observational study where the causality between indoxyl sulfate and thrombosis could not be proved. Nevertheless, our findings corroborate both the in vitro and in vivo evidence that indoxyl sulfate plays an important role in thrombosis.32,33 Second, the study population was composed of a single population from a single center; therefore, the results may not be applicable to other ethnic groups. Third, the study did not start at the initiation of dialysis; therefore, confounding biases, such as dialysis vintage, previous interventions, and residual renal function, could not be avoided. Our focus was on the determinants of postangioplasty outcomes rather than primary prevention after access creation. In consequence, evaluation and follow-up after PTA were more reasonable and practical for secondary prevention. Finally, serum levels of indoxyl sulfate may have varied during the long-term follow-up period. However, a single baseline measurement was widely used in studies investigating post-PTA outcomes. Baseline sampling is nearest to the critical period of restenosis or thrombosis, free from the interference of intervention, and more applicable in clinical practice. In addition, by taking measurements in the eight patients before and 4 weeks after PTA, the variability in serum indoxyl sulfate levels over time was found to be acceptable.
Clinical Implications
Patients with dysfunctional vascular accesses are at higher risk of thrombotic events after endovascular interventions.48 Our study provided the first translational evidence that indoxyl sulfate, a uremic toxin, is associated with graft access thrombosis after endovascular interventions. Currently, a variety of strategies to remove protein-bound uremic toxins are under investigation, including enhancement of solute removal, devices for extracorporeal absorption, reduction of gastrointestinal absorption, and maintenance of residual renal function.32 Clinical trials using preventive or therapeutic strategies are warranted to clarify the role of indoxyl sulfate in secondary prevention of graft thrombosis after PTA.
The results from this study suggest that a protein-bound uremic solute, indoxyl sulfate, is associated with thrombosis of dialysis grafts after angioplasty. This association was independent of clinical, biochemical, and access factors. Whether indoxyl sulfate can be modified to prevent thrombosis after endovascular interventions deserves further investigations.
Concise Methods
Study Population
Patients referred for PTA because of dysfunctional hemodialysis vascular access were prospectively enrolled. Patients were referred for PTA on the basis of one or more of the following criteria: (1) clinical signs suggesting vascular access dysfunction (decreased thrill, increased pulsatility, development of collateral veins, and prolonged bleeding from puncture sites); (2) a reduction in flow rate of >25% from baseline; (3) total access blood flow rate <500 ml/min by ultrasound dilution method (Transonic Flow-QC; Transonic Systems, Ithaca, NY); and (4) increased venous pressure during dialysis (dynamic venous pressure exceeding threshold level measured three consecutive times).
Exclusion criteria were the following: (1) patients who received regular dialysis for <6 months; (2) patients with acute or chronic infection disease, decompensated heart failure, recent myocardial infarction, or unstable angina requiring hospitalization in previous 3 months; (3) patients with an insignificant lesion (diameter stenosis <50%), arterial stenosis, or central vein stenosis; and (4) patients with failed PTA procedures.
After enrollment, clinical data, access characteristics, and details of the procedure were obtained from review of medical records, angioplasty reports, and hemodialysis records. Cardiovascular diseases were defined as coronary, peripheral, or cerebral artery diseases. All eligible patients received 4 hours of hemodialysis three times a week using a synthetic dialysis membrane (polyamide or polysulfone). The dialysate in all patients was a standard ionic composition and bicarbonate-based buffer, and dialyzers were not reused. The adequacy of dialysis was accessed monthly using the single pool Kt/V of urea nitrogen. In Taiwan, a native arteriovenous fistula is the preferred access for placement, and synthetic grafts are reserved for patients whose vascular anatomy is not suitable for fistula creation.
The study was on the basis of the Declaration of Helsinki (edition 6, revised 2000). Informed consent was obtained from all study participants, and this study was approved by the institutional research board of our hospital.
Angiography and Angioplasty Procedures
Diagnostic angiography was performed on a midweek nondialysis day. After diagnostic angiography, angioplasty was performed according to the National Kidney Foundation’s Disease Outcomes Quality Initiative guidelines (i.e., only patients with a clinical indicator of access dysfunction coupled with a minimum diameter stenosis of at least 50% underwent angioplasty).49
The stenosis was treated with standard balloon angioplasty techniques, as previously described.50 Medication for underlying cardiovascular disease or CKD was continued. A computer-based system was used for quantitative angiographic analysis. Diagnostic angiography and angiograms of all PTA procedures were independently reviewed by a physician who was unaware of the patients’ clinical and biochemical data.
Anatomic measurements were made using a calibrated reference marker with computer-assisted edge detection software provided by the angiographic imaging system. The reference vessel was defined as an adjacent segment of normal vein located upstream from the target lesion. The degree of stenosis was reported as a reduction in maximum diameter compared with the reference vessel diameter.
Laboratory Methods
After a 12-hour overnight fast and cessation of medications before the diagnostic procedure, 10 ml of blood was drawn from each patient on the morning of the procedure. The blood sample was drawn directly from the vascular access before PTA, which was arranged on a midweek nondialysis day. They were centrifuged at 3000 rpm for 10 minutes at 4°C immediately after collection. The plasma samples were then stored at –80°C until use.
Plasma biochemical parameters, including cholesterol, triglycerides, calcium, phosphate, and albumin, were analyzed by standard laboratory procedures. Single-pool Kt/V of urea nitrogen was calculated after enrollment using the second-generation logarithmic formula of Daugirdas. Residual renal function was estimated from an interdialytic urine collection and expressed as weekly renal Kt/V. Plasma hs-CRP levels were measured with a commercially available kit (Dade Behring, Marburg, Germany) and homocysteine by enzyme immunoassay (Axis Homocysteine EIA; Axis-Shield AS, Oslo, Norway). Total indoxyl sulfate (i.e., both free and protein-bound fractions) was obtained via protein precipitation of the serum sample. The free form was obtained from serum ultrafiltrate using a Microcon YM-30 separator (EMD Millipore, Billerica, MA). Indoxyl sulfate levels in serum were measured using an HPLC-fluorescence method. The analyst was separated by a Luna phenyl-hexyl column (150×4.6 mm, 5 μm, Phenomenex). The mobile phase consisting of 10 mM phosphate buffer at pH 6.0 and acetonitrile (75:25, volume/volume) was eluted at a flow rate of 1.0 ml/min. The fluorescence detector was set at λex 280 nm/λem 346 nm. Intra- and interassay coefficients of variation of indoxyl sulfate in serum were 7% and 10% for free form and 5% and 8% for total form, respectively. Intraindividual variability of serum indoxyl sulfate over time was assessed by two measurements before PTA and 4 weeks after PTA in eight patients. The intraclass correlation coefficient for free indoxyl sulfate was 0.72 (P=0.01), and the intraclass correlation coefficient for total indoxyl sulfate was 0.78 (P=0.01) (Bland–Altman plots in Supplemental Figure 1).
End Point Evaluation
Follow-up was extended until June 30, 2014. After PTA, all patients received prospective clinical follow-up of their hemodialysis vascular access under the same protocol at all respective hemodialysis centers. The follow-up data were obtained by our vascular access coordinator via telephone contact with referring hemodialysis centers. Follow-up surveillance of vascular access patients included physical examination, dynamic venous pressure monitoring at each hemodialysis session, and transonic examination of access blood flow rate immediately after intervention, and then monthly, if available. When abnormal clinical or hemodynamic parameters fulfilling the original referring criteria were detected, patients were referred for fistulography and intervention, as appropriate. Clinical events, including cardiovascular events and cause of death, were identified by review of medical records by a coding physician blinded to the measured biomarkers. Whenever there was any doubt about a given event, the coding physicians conferred with the referring nephrologists or the attending physician.
Access restenosis was defined as >50% diameter stenosis anywhere within the outflow vein associated with clinical evidence of dysfunction. Relative late loss was defined as the difference of percentage stenosis after angioplasty at baseline and before angioplasty at next intervention. Absolute late loss was the difference of minimal luminal diameter after angioplasty at baseline and before angioplasty at next intervention. Rate of late loss was defined as late loss divided by the patency days. Access thrombosis was defined as a sudden cessation of access function rendering hemodialysis impossible and requiring thrombectomy or placement of another hemodialysis access. Access failure was defined as abandonment or surgical revision of the access.
Cardiovascular death included death because of myocardial infarction, heart failure, ischemic stroke, or sudden death. Cases of unobserved sudden death were considered cardiovascular death only when other potential causes could be excluded. Nonaccess thrombotic events were defined as hospitalizations because of coronary, cerebral, peripheral arterial, or deep venous thrombosis. Coronary thrombosis was diagnosed on the basis of elevated cardiac enzymes with/without the typical electrocardiographic changes or coronary intervention. Cerebral arterial thrombosis was defined as ischemic stroke with neurologic deficit lasting >24 hours, confirmed by computed tomography or magnetic resonance imaging. Peripheral arterial thrombosis was defined as acute or critical ischemia with the lower-limb arteries. Deep venous thrombosis was confirmed by compression ultrasound and evidence of pulmonary embolism on computed tomography.
Statistical Analyses
Continuous variables were expressed as mean±SD for normally distributed variables or median (IQR) for non-normally distributed variables. Differences between groups stratified by tertile of free indoxyl sulfate levels were assessed using the one-way ANOVA test for normally distributed continuous variables, the Kruskal–Wallis test for non-normally distributed continuous variables, and the Pearson chi-squared test for dichotomous variables. The association between indoxyl sulfate levels, baseline variables, and late loss was analyzed using a Spearman rank correlation matrix for continuous variables and Pearson chi-squared test for dichotomous variables.
Cumulative access patency without thrombosis was generated by the Kaplan–Meier method and compared by the log-rank test. In each approach, data were censored at death, kidney transplantation, transfer to nonstudy dialysis centers, or at the end of follow-up unless a vascular event developed before. The Cox proportional hazards model was used for univariate analysis of predictors of dialysis access thrombosis. The assumption of proportionality was checked graphically using the log-log plot and was found to be acceptable for the risk factors of interest. To compare the strength of the independent association between indoxyl sulfate and access thrombosis relative to other factors, indoxyl sulfate was tested using multivariate analysis, including all risk factors significant at the P<0.1 level at univariate analysis. Statistical analyses were performed using the computer software SPSS version 20.0. (SPSS Inc., Chicago, IL). All P values were two-tailed, and P<0.05 was considered statistically significant.
Disclosures
None.
Supplementary Material
Acknowledgments
This study was supported in part to Dr. C.C. Wu by grants from the National Taiwan University Hospital, Hsinchu Branch (HCH101-13, HCH102-16, and HCH103-14) and the National Science Council (NSC103-2314-B-002-183), to Dr. P.H. Huang by grants from the Taipei Veterans General Hospital (VGH-V102B-016 and VGH-V102E2-002) and the National Science Council (UST-UCSD International Centre of Excellence in Advanced Bio-engineering NSC-100-2911-I-009-101-A2), and to Dr. D.C. Tarng by grants from the National Science Council (NSC99-2314-B-010-002-MY3 and NSC 102-2314-B-010-004-MY3), Taipei Veterans General Hospital (V102-C-129), Taipei City Hospital (10101-62-028), Foundation for Poison Control, and Ministry of Education’s aim for the Top University Plan in National Yang-Ming University.
Footnotes
Published online ahead of print. Publication date available at www.jasn.org.
See related editorial, “Dialysis Vascular Access Intervention and the Search for Biomarkers,” on pages 970–972.
This article contains supplemental material online at http://jasn.asnjournals.org/lookup/suppl/doi:10.1681/ASN.2015010068/-/DCSupplemental.
References
- 1.National Kidney Foundation-Dialysis Outcomes Quality Initiative : NKF-DOQI clinical practice guidelines for vascular access. Am J Kidney Dis 30[Suppl 3]: S150–S191, 1997 [PubMed] [Google Scholar]
- 2.US Renal Data System: USRDS 2010 Annual Data Report: Atlas of Chronic Kidney Disease and End-Stage Renal Disease in the United States, Bethesda, MD, National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, 2010 [Google Scholar]
- 3.Roy-Chaudhury P, Sukhatme VP, Cheung AK: Hemodialysis vascular access dysfunction: A cellular and molecular viewpoint. J Am Soc Nephrol 17: 1112–1127, 2006 [DOI] [PubMed] [Google Scholar]
- 4.Schwab SJ, Oliver MJ, Suhocki P, McCann R: Hemodialysis arteriovenous access: Detection of stenosis and response to treatment by vascular access blood flow. Kidney Int 59: 358–362, 2001 [DOI] [PubMed] [Google Scholar]
- 5.Moist LM, Churchill DN, House AA, Millward SF, Elliott JE, Kribs SW, DeYoung WJ, Blythe L, Stitt LW, Lindsay RM: Regular monitoring of access flow compared with monitoring of venous pressure fails to improve graft survival. J Am Soc Nephrol 14: 2645–2653, 2003 [DOI] [PubMed] [Google Scholar]
- 6.Neuen BL, Gunnarsson R, Webster AC, Baer RA, Golledge J, Mantha ML: Predictors of patency after balloon angioplasty in hemodialysis fistulas: A systematic review. J Vasc Interv Radiol 25: 917–924, 2014 [DOI] [PubMed] [Google Scholar]
- 7.Vanholder R, Baurmeister U, Brunet P, Cohen G, Glorieux G, Jankowski J, European Uremic Toxin Work Group : A bench to bedside view of uremic toxins. J Am Soc Nephrol 19: 863–870, 2008 [DOI] [PubMed] [Google Scholar]
- 8.Dou L, Jourde-Chiche N, Faure V, Cerini C, Berland Y, Dignat-George F, Brunet P: The uremic solute indoxyl sulfate induces oxidative stress in endothelial cells. J Thromb Haemost 5: 1302–1308, 2007 [DOI] [PubMed] [Google Scholar]
- 9.Dou L, Bertrand E, Cerini C, Faure V, Sampol J, Vanholder R, Berland Y, Brunet P: The uremic solutes p-cresol and indoxyl sulfate inhibit endothelial proliferation and wound repair. Kidney Int 65: 442–451, 2004 [DOI] [PubMed] [Google Scholar]
- 10.Yu M, Kim YJ, Kang DH: Indoxyl sulfate-induced endothelial dysfunction in patients with chronic kidney disease via an induction of oxidative stress. Clin J Am Soc Nephrol 6: 30–39, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yamamoto H, Tsuruoka S, Ioka T, Ando H, Ito C, Akimoto T, Fujimura A, Asano Y, Kusano E: Indoxyl sulfate stimulates proliferation of rat vascular smooth muscle cells. Kidney Int 69: 1780–1785, 2006 [DOI] [PubMed] [Google Scholar]
- 12.Vanholder R, Schepers E, Pletinck A, Nagler EV, Glorieux G: The uremic toxicity of indoxyl sulfate and p-cresyl sulfate: A systematic review. J Am Soc Nephrol 25: 1897–1907, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bammens B, Evenepoel P, Keuleers H, Verbeke K, Vanrenterghem Y: Free serum concentrations of the protein-bound retention solute p-cresol predict mortality in hemodialysis patients. Kidney Int 69: 1081–1087, 2006 [DOI] [PubMed] [Google Scholar]
- 14.Liabeuf S, Barreto DV, Barreto FC, Meert N, Glorieux G, Schepers E, Temmar M, Choukroun G, Vanholder R, Massy ZA, European Uraemic Toxin Work Group (EUTox) : Free p-cresylsulphate is a predictor of mortality in patients at different stages of chronic kidney disease. Nephrol Dial Transplant 25: 1183–1191, 2010 [DOI] [PubMed] [Google Scholar]
- 15.Barreto FC, Barreto DV, Liabeuf S, Meert N, Glorieux G, Temmar M, Choukroun G, Vanholder R, Massy ZA, European Uremic Toxin Work Group (EUTox) : Serum indoxyl sulfate is associated with vascular disease and mortality in chronic kidney disease patients. Clin J Am Soc Nephrol 4: 1551–1558, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Neuen BL, Gunnarsson R, Baer RA, Tosenovsky P, Green SJ, Golledge J, Mantha ML: Factors associated with patency following angioplasty of hemodialysis fistulae. J Vasc Interv Radiol 25: 1419–1426, 2014 [DOI] [PubMed] [Google Scholar]
- 17.Lilly RZ, Carlton D, Barker J, Saddekni S, Hamrick K, Oser R, Westfall AO, Allon M: Predictors of arteriovenous graft patency after radiologic intervention in hemodialysis patients. Am J Kidney Dis 37: 945–953, 2001 [DOI] [PubMed] [Google Scholar]
- 18.Crowther MA, Kelton JG: Congenital thrombophilic states associated with venous thrombosis: A qualitative overview and proposed classification system. Ann Intern Med 138: 128–134, 2003 [DOI] [PubMed] [Google Scholar]
- 19.Schwab SJ, Harrington JT, Singh A, Roher R, Shohaib SA, Perrone RD, Meyer K, Beasley D: Vascular access for hemodialysis. Kidney Int 55: 2078–2090, 1999 [DOI] [PubMed] [Google Scholar]
- 20.Crowther MA, Clase CM, Margetts PJ, Julian J, Lambert K, Sneath D, Nagai R, Wilson S, Ingram AJ: Low-intensity warfarin is ineffective for the prevention of PTFE graft failure in patients on hemodialysis: A randomized controlled trial. J Am Soc Nephrol 13: 2331–2337, 2002 [DOI] [PubMed] [Google Scholar]
- 21.Kaufman JS, O’Connor TZ, Zhang JH, Cronin RE, Fiore LD, Ganz MB, Goldfarb DS, Peduzzi PN, Veterans Affairs Cooperative Study Group on Hemodialysis Access Graft Thrombosis : Randomized controlled trial of clopidogrel plus aspirin to prevent hemodialysis access graft thrombosis. J Am Soc Nephrol 14: 2313–2321, 2003 [DOI] [PubMed] [Google Scholar]
- 22.Ocak G, Vossen CY, Rotmans JI, Lijfering WM, Rosendaal FR, Parlevliet KJ, Krediet RT, Boeschoten EW, Dekker FW, Verduijn M: Venous and arterial thrombosis in dialysis patients. Thromb Haemost 106: 1046–1052, 2011 [DOI] [PubMed] [Google Scholar]
- 23.Casserly LF, Dember LM: Thrombosis in end-stage renal disease. Semin Dial 16: 245–256, 2003 [DOI] [PubMed] [Google Scholar]
- 24.Baskin E, Duman O, Beşbaş N, Ozen S: Hypercoagulopathy in a hemodialysis patient: Are elevations in factors VII and VIII effective? Nephron 83: 180, 1999 [DOI] [PubMed] [Google Scholar]
- 25.Segarra A, Chacón P, Martinez-Eyarre C, Argelaguer X, Vila J, Ruiz P, Fort J, Bartolomé J, Camps J, Moliner E, Pelegrí A, Marco F, Olmos A, Piera L: Circulating levels of plasminogen activator inhibitor type-1, tissue plasminogen activator, and thrombomodulin in hemodialysis patients: Biochemical correlations and role as independent predictors of coronary artery stenosis. J Am Soc Nephrol 12: 1255–1263, 2001 [DOI] [PubMed] [Google Scholar]
- 26.Małyszko J, Małyszko JS, Hryszko T, Myśliwiec M: Thrombin activatable fibrinolysis inhibitor (TAFI) and markers of endothelial cell injury in dialyzed patients with diabetic nephropathy. Thromb Haemost 91: 480–486, 2004 [DOI] [PubMed] [Google Scholar]
- 27.Mercier E, Branger B, Vecina F, Al-Sabadani B, Berlan J, Dauzat M, Fourcade J, Gris JC: Tissue factor coagulation pathway and blood cells activation state in renal insufficiency. Hematol J 2: 18–25, 2001 [DOI] [PubMed] [Google Scholar]
- 28.Knoll GA, Wells PS, Young D, Perkins SL, Pilkey RM, Clinch JJ, Rodger MA: Thrombophilia and the risk for hemodialysis vascular access thrombosis. J Am Soc Nephrol 16: 1108–1114, 2005 [DOI] [PubMed] [Google Scholar]
- 29.Nampoory MR, Das KC, Johny KV, Al-Hilali N, Abraham M, Easow S, Saed T, Al-Muzeirei IA, Sugathan TN, Al Mousawi M: Hypercoagulability, a serious problem in patients with ESRD on maintenance hemodialysis, and its correction after kidney transplantation. Am J Kidney Dis 42: 797–805, 2003 [DOI] [PubMed] [Google Scholar]
- 30.O’shea SI, Lawson JH, Reddan D, Murphy M, Ortel TL: Hypercoagulable states and antithrombotic strategies in recurrent vascular access site thrombosis. J Vasc Surg 38: 541–548, 2003 [DOI] [PubMed] [Google Scholar]
- 31.LeSar CJ, Merrick HW, Smith MR: Thrombotic complications resulting from hypercoagulable states in chronic hemodialysis vascular access. J Am Coll Surg 189: 73–79, discussion 79–81, 1999 [DOI] [PubMed] [Google Scholar]
- 32.Gondouin B, Cerini C, Dou L, Sallée M, Duval-Sabatier A, Pletinck A, Calaf R, Lacroix R, Jourde-Chiche N, Poitevin S, Arnaud L, Vanholder R, Brunet P, Dignat-George F, Burtey S: Indolic uremic solutes increase tissue factor production in endothelial cells by the aryl hydrocarbon receptor pathway. Kidney Int 84: 733–744, 2013 [DOI] [PubMed] [Google Scholar]
- 33.Chitalia VC, Shivanna S, Martorell J, Balcells M, Bosch I, Kolandaivelu K, Edelman ER: Uremic serum and solutes increase post-vascular interventional thrombotic risk through altered stability of smooth muscle cell tissue factor. Circulation 127: 365–376, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.de Groot K, Bahlmann FH, Sowa J, Koenig J, Menne J, Haller H, Fliser D: Uremia causes endothelial progenitor cell deficiency. Kidney Int 66: 641–646, 2004 [DOI] [PubMed] [Google Scholar]
- 35.Sata M, Saiura A, Kunisato A, Tojo A, Okada S, Tokuhisa T, Hirai H, Makuuchi M, Hirata Y, Nagai R: Hematopoietic stem cells differentiate into vascular cells that participate in the pathogenesis of atherosclerosis. Nat Med 8: 403–409, 2002 [DOI] [PubMed] [Google Scholar]
- 36.Wu VC, Young GH, Huang PH, Lo SC, Wang KC, Sun CY, Liang CJ, Huang TM, Chen JH, Chang FC, Chen YL, Kuo YS, Chen JB, Chen JW, Chen YM, Ko WJ, Wu KD, NSARF group : In acute kidney injury, indoxyl sulfate impairs human endothelial progenitor cells: Modulation by statin. Angiogenesis 16: 609–624, 2013 [DOI] [PubMed] [Google Scholar]
- 37.Wu CC, Huang PH, Lai CL, Leu HB, Chen JW, Lin SJ: The impact of endothelial progenitor cells on restenosis after percutaneous angioplasty of hemodialysis vascular access. PLoS One 9: e101058, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lev EI, Leshem-Lev D, Mager A, Vaknin-Assa H, Harel N, Zimra Y, Bental T, Greenberg G, Dvir D, Solodky A, Assali A, Battler A, Kornowski R: Circulating endothelial progenitor cell levels and function in patients who experienced late coronary stent thrombosis. Eur Heart J 31: 2625–2632, 2010 [DOI] [PubMed] [Google Scholar]
- 39.Yisireyili M, Saito S, Abudureyimu S, Adelibieke Y, Ng HY, Nishijima F, Takeshita K, Murohara T, Niwa T: Indoxyl sulfate-induced activation of (pro)renin receptor promotes cell proliferation and tissue factor expression in vascular smooth muscle cells. PLoS One 9: e109268, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Cheng YJ, Liu ZH, Yao FJ, Zeng WT, Zheng DD, Dong YG, Wu SH: Current and former smoking and risk for venous thromboembolism: A systematic review and meta-analysis. PLoS Med 10: e1001515, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Barua RS, Ambrose JA: Mechanisms of coronary thrombosis in cigarette smoke exposure. Arterioscler Thromb Vasc Biol 33: 1460–1467, 2013 [DOI] [PubMed] [Google Scholar]
- 42.Vazzana N, Ranalli P, Cuccurullo C, Davì G: Diabetes mellitus and thrombosis. Thromb Res 129: 371–377, 2012 [DOI] [PubMed] [Google Scholar]
- 43.Chou CY, Kuo HL, Yung YF, Liu YL, Huang CC: C-reactive protein predicts vascular access thrombosis in hemodialysis patients. Blood Purif 24: 342–346, 2006 [DOI] [PubMed] [Google Scholar]
- 44.Chang CJ, Ko YS, Ko PJ, Hsu LA, Chen CF, Yang CW, Hsu TS, Pang JH: Thrombosed arteriovenous fistula for hemodialysis access is characterized by a marked inflammatory activity. Kidney Int 68: 1312–1319, 2005 [DOI] [PubMed] [Google Scholar]
- 45.Miyazaki T, Ise M, Hirata M, Endo K, Ito Y, Seo H, Niwa T: Indoxyl sulfate stimulates renal synthesis of transforming growth factor-beta 1 and progression of renal failure. Kidney Int Suppl 63: S211–S214, 1997 [PubMed] [Google Scholar]
- 46.Lin CJ, Wu CJ, Pan CF, Chen YC, Sun FJ, Chen HH: Serum protein-bound uraemic toxins and clinical outcomes in haemodialysis patients. Nephrol Dial Transplant 25: 3693–3700, 2010 [DOI] [PubMed] [Google Scholar]
- 47.Wu IW, Hsu KH, Hsu HJ, Lee CC, Sun CY, Tsai CJ, Wu MS: Serum free p-cresyl sulfate levels predict cardiovascular and all-cause mortality in elderly hemodialysis patients--a prospective cohort study. Nephrol Dial Transplant 27: 1169–1175, 2012 [DOI] [PubMed] [Google Scholar]
- 48.Lambert ND, Sacrinty MT, Ketch TR, Turner SJ, Santos RM, Daniel KR, Applegate RJ, Kutcher MA, Sane DC: Chronic kidney disease and dipstick proteinuria are risk factors for stent thrombosis in patients with myocardial infarction. Am Heart J 157: 688–694, 2009 [DOI] [PubMed] [Google Scholar]
- 49.Vascular Access 2006 Work Group : Clinical practice guidelines for vascular access. Am J Kidney Dis 48[Suppl 1]: S176–S247, 2006 [DOI] [PubMed] [Google Scholar]
- 50.Wu CC, Lin MC, Pu SY, Tsai KC, Wen SC: Comparison of cutting balloon versus high-pressure balloon angioplasty for resistant venous stenoses of native hemodialysis fistulas. J Vasc Interv Radiol 19: 877–883, 2008 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.