Abstract
Background:
Mucosal human papillomaviruses (HPV) are a major cause of cancers and papillomas of the anogenital and oropharyngeal tract. HPV-vaccination elicits neutralising antibodies in sera and cervicovaginal secretions and protects uninfected individuals from persistent anogenital infection and associated diseases caused by the vaccine-targeted HPV types. Whether immunisation can prevent oropharyngeal infection and diseases and whether neutralising antibodies represent the correlate of protection, is still unclear.
Methods:
We determined IgG and neutralising antibodies against low-risk HPV6 and high-risk HPV16/18 in sera and oral fluids from healthy females (n=20) before and after quadrivalent HPV-vaccination and compared the results with non-vaccinated controls.
Results:
HPV-vaccination induced type-specific antibodies in sera and oral fluids of the vaccinees. Importantly, the antibodies in oral fluids were capable of neutralising HPV pseudovirions in vitro, indicating protection from infection. The increased neutralising antibody levels against HPV16/18 in sera and oral fluids post-vaccination correlated significantly within an individual.
Conclusions:
We provide experimental proof that HPV-vaccination elicits neutralising antibodies to the vaccine-targeted types in oral fluids. Hence, immunisation may confer direct protection against type-specific HPV infection and associated diseases of the oropharyngeal tract. Measurement of antibodies in oral fluids represents a suitable tool to assess vaccine-induced protection within the mucosal milieu of the orophayrynx.
Keywords: human papillomavirus, HPV vaccination, neutralising antibodies, oral fluids, pseudovirion neutralisation assay, oropharyngeal disease
Human papillomaviruses (HPV) are small, double-stranded DNA viruses and, to date, more than 180 HPV (geno-)types have been identified (Bernard et al, 2010). These epitheliotropic viruses exclusively infect epithelial cells of skin or mucosa, allowing for their classification into cutaneous or mucosal HPV types, respectively. Depending on the virus type and host, HPV infection can be asymptomatic, can induce benign warts or papillomas, or lead to the development of anogenital malignancies—most notably cervical carcinomas—and head and neck cancers (HNSCC).
HNSCC represent a rising global burden with annually more than 500 000 incident cases and 300 000 deaths (Marur et al, 2010; Ferlay et al, 2015). These malignancies, predominantly squamous cell carcinomas, which arise from the mucosa of the oral cavity, the oro-, hypo-, nasopharnx, the larynx, and the sinonasal tract, can be divided into two distinct subsets according to their aetiology. The first subset is firmly associated with heavy tobacco and/or alcohol consumption, poor oral hygiene, or predisposing genetics (but is HPV-negative). In the past decade, a second entity was recognised as caused by infection with mucosal high-risk HPV, independently of the established risk factors (D'Souza et al, 2007; Gillison et al, 2008; Mehanna et al, 2013). In the latter the rate of detection of HPV DNA in the tumours overall was variable between the different anatomical sites, with the highest rate observed in cancers of the oropharynx (45.8% in particular of the tonsils and the base of tongue at 53.9%), the oral cavity (24.2%), and the larynx/hypopharynx (22.1%) (Ndiaye et al, 2014). The DNA was localised in the nuclei of cancerous cells at all stages—in preinvasive/invasive tumours and metastases—in integrated form and found to be transcriptionally active (Steenbergen et al, 1995; Gillison et al, 2000). The most dominant causative HPV type is mucosal high-risk HPV 16, which accounts for >90% of the carcinomas (Gillison et al, 2000; Kreimer et al, 2005; D'Souza et al, 2007; Marur et al, 2010). Second, high-risk HPV 18, and in rare cases other high-risk mucosal types and low-risk mucosal HPV 6 and 11 were detected in the HNSCC (Gillison et al, 2000; Kreimer et al, 2005). However, the latter two types predominantly cause papillomas of the respiratory and the laryngeal tract, including recurrent respiratory papillomatosis, a rare disease in children and adults that due to extensive warty growths in the upper airway and dissemination to bronchi, may cause significant airway obstruction and may undergo malignant transformation (Venkatesan et al, 2012; Taliercio et al, 2015).
In contrast to the well-established pathogenesis of anogenital, in particular cervical, HPV infection and associated malignancies, the natural history of HPV infection in the oropharyngeal tract is not as extensively investigated. Transmission of the virus most likely occurs via orogenital contact and oral HPV DNA prevalence was consistently associated with sexual behaviour. For instance, patients with HPV-positive oropharyngeal cancers were shown to have a higher number of lifetime sexual partners (as surrogate for oral HPV exposure) and presented oral HPV infection more frequently than controls (D'Souza et al, 2007; Heck et al, 2010). In the general population, oral HPV was reported at 4.5–6.9% and the prevalence of infections with one of the mucosal high-risk HPV types given at 3.5–3.7%, of which HPV 16 accounted for 1–1.3% (Kreimer et al, 2010; Chung et al, 2014).
Prophylactic HPV vaccination is highly immunogenic, leading to seroconversion in nearly all immunised individuals. In previously uninfected individuals, administration of the HPV vaccines prevented persistent infection and associated diseases of the anogenital tract that are caused by the HPV types included in the vaccine formulation with an efficacy of up to 100% (De Vincenzo et al, 2014; Herrero et al, 2015). This was convincingly demonstrated in multiple large, randomised, controlled, clinical trials and resulted in licensing and implementation of the vaccines in numerous national immunisation programmes (Markowitz et al, 2014; WHO, 2014). For vaccine-induced protection both, humoral and cellular, immune mechanisms are responsible. After vaccination, HPV type-specific antibodies capable of neutralising infectious virions were detectable in sera—at up to 4-log higher levels compared with titres induced by natural infection—and in cervicovaginal secretions (Harper et al, 2006; Schwarz et al, 2010; Petäjä et al, 2011). In addition, cell-mediated immune responses to the viral proteins are equally important for the success of prophylactic vaccination. Whereas T-helper responses are critically involved in the generation and maintenance of protective B-cell responses and the induction of neutralising antibodies, cytotoxic T-lymphocyte responses to the viral proteins are important for elimination of HPV-infected cells that escaped antibody neutralisation. While vaccine efficacy is firmly established for the prevention of anogenital infection and diseases, studies on the impact of vaccination on oral HPV infection and HPV-related oropharyngeal diseases are sparse.
First evidence for a reduction of oral HPV infection after vaccination was obtained from the Costa Rica vaccine trial (Herrero et al, 2013). Prevalence of oral HPV 16/18 DNA was reduced in the recipients of the bivalent HPV vaccine 4 years post-vaccination compared with hepatitis A-vaccinated controls, with vaccine efficacy against HPV 16/18 infection estimated at 93.3% in this large cohort. Although this important study strongly indicates that HPV vaccination has the potential to confer protection against oral infection with the vaccine-targeted types, data on the baseline HPV prevalence are lacking and information on the presence of HPV-specific antibodies in the oral fluids of the vaccinees and their protective capacity is not available.
In this study, we determined the presence of type-specific IgG and neutralising antibodies in oral fluids after immunisation with the quadrivalent HPV vaccine and investigated a possible correlation between (neutralising) antibody titres in paired serum and oral fluids. We furthermore aimed at providing experimental proof that antibodies isolated from oral fluids are capable of preventing HPV infection, thus giving insight into one possible mechanism how oral HPV infection and possibly associated diseases can be prevented by systemic administration of an HPV vaccine.
Material and methods
Study population
The study protocol was approved by the ethics committee of the Medical University of Vienna before initiation (EK 690/2008) and informed consent was obtained from each participant.
Study participants (n=34) were healthy females between 18 and 26 years of age (mean age 23.2 years) with four or less lifetime sexual partners and no history of anogenital HPV infection, including condylomata and cervical dysplasia. Recipients of immunoglobulins, blood products, and immunomodifying drugs within 6 months before study initiation or pregnant or nursing women were excluded. Participants were recruited into two groups: 20 participants (age range 19–24 years, mean age 22.6 years) received the quadrivalent HPV vaccine against low-risk HPV 6/11 and high-risk HPV 16/18 intramuscularly according to the licensed schedule; 14 participants (age range 22–25 years, mean age 23.9 years) were included in the control group and not vaccinated. Medical and sexual histories were obtained at enrolment. All participants completed the compulsory four visits within the prespecified intervals.
Sample collection
Serum and oral fluids were collected from all participants before immunisation/study initiation (month 0), at each subsequent vaccination (months 2 and 6), and four weeks after the final immunisation/at end of study (month 7). Oral fluids were collected by inserting two pads (OraSure Technologies Inc., PA, USA) between buccal mucosa and gingiva for 4 min. To analyse the presence of HPV-specific total IgG antibodies by ELISA, one pad was subsequently inserted into the supplied vial (OraSure) containing a transportation buffer with preservative. To extract the oral fluid, vials were centrifuged at 2400 g for 20 min, the pad removed, and the remaining liquid collected. For detection of HPV-specific neutralising antibodies by pseudovirion (PsV)-based neutralisation assays, the second collection pad was repeatedly rinsed with 1 ml of phosphate-buffered saline (PBS) and the eluate collected by centrifugation at 2400 g for 15 min. This procedure was repeated with another millilitre of PBS, eluates were pooled and subjected to sterile filtration using Corning Costar Spin-X centrifuge tube filters with a 0.2-μm pore filter size at 13 000 r.p.m. for 10 min. Both, sera and oral fluids, were investigated for the presence of HPV-specific antibodies. HPV 6 served as representative for low-risk, HPV 16 and HPV 18 for high-risk mucosal HPV. All three types are targeted by the quadrivalent HPV vaccine.
Virus-like particle–ELISA
Recombinant baculoviruses expressing the major capsid protein L1 of HPV 6, 16, and 18 were described previously (Kirnbauer et al, 1993; Rose et al, 1994) and virus-like particles (VLP) of the respective HPV types generated and purified by standard methods (Kirnbauer et al, 1994; Handisurya et al, 2010). Sera and oral fluids were tested by VLP-ELISA for the presence of HPV-specific IgG antibodies to HPV types 6, 16 and 18, as described previously (Handisurya et al, 2010). Sera were fourfold serially diluted in PBS, ranging from 1 : 1000 to 1 : 64 000, oral fluid samples were used in dilutions of 1 : 10 and 1 : 100 (in PBS). Polyclonal rabbit sera, obtained after immunisation with VLP of the respective HPV types—either HPV 6, HPV 16, or HPV 18—served as positive controls, VLP-coated wells without addition of sample (PBS-only wells) as negative control in the assays. Optical density (OD) values were determined at 405 nm test wavelength/490 nm reference wavelength. Seropositivity was defined as mean OD values higher than twice the mean values of pooled sera obtained from non-infected, sexually naive controls.
Pseudovirion-based neutralisation assays
PsV of HPV types 6, 16, and 18 were generated by co-transfection of the human embryonic kidney cell line 293TT with a plasmid encoding for the respective viral capsid genes and a reporter plasmid encoding for secreted alkaline phosphatase (SEAP), according to standard protocols (Buck et al, 2005; Handisurya et al, 2010). For analysis by Pseudovirion-based neutralisation assays (PBNA), participants' sera were fourfold serially diluted from 1 : 400 to 1:25 600, oral fluids from 1 : 4 to 1 : 16, and both were independently tested for their ability to inhibit infection of 293TT cells by PsV of either HPV type 6, 16, or 18, as previously published. Rabbit polyclonal sera raised against either HPV 6, HPV 16, or HPV 18 VLP or a mixture of the analysed PsV and media without samples (PBS-only wells) were used as positive or negative controls, respectively. OD values were measured at 405 nm, as given in the manufacturer's instructions. Samples showing at least 50% reduction in SEAP activity were considered neutralising.
Statistical analyses
Statistical analyses were performed using the Statistical Package for the Social Sciences Version 15.0. (SPSS Inc., Chicago, IL, USA), P-values <0.05 (two-sided asymptotic significance) were considered statistically significant. Differences between the vaccinated and the non-vaccinated cohorts were calculated using the Mann–Whitney U-test, differences between samples obtained before and one month after completion of the vaccination series from the same study participant employing the Wilcoxon signed-rank test. Correlations between overall HPV-specific IgG antibody concentrations and neutralising antibodies against the same HPV type in samples obtained post-vaccination were determined using the Pearson's χ2-test.
Results
HPV immunisation induced total IgG and neutralising antibodies to the vaccine-targeted types in sera of the vaccinees
Sera of all study participants were analysed for the presence of type-specific IgG and neutralising antibodies against HPV 6, HPV 16 and HPV 18 by VLP-ELISA and PBNA, respectively. These three types are the most common and important representatives of low- and high-risk mucosal HPV and are included in the quadrivalent vaccine formulation.
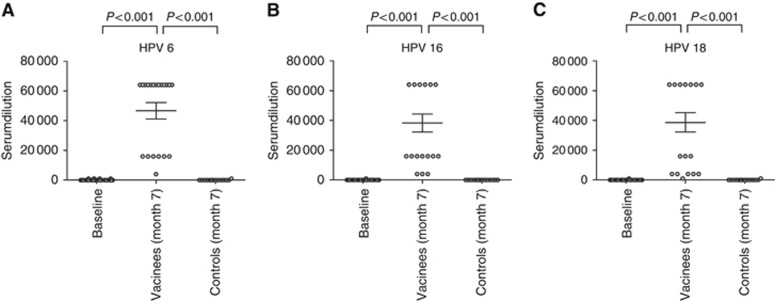
At month 7 (1 month after the third and final immunisation of the vaccination cohort) all participants of the vaccine group had seroconverted, showing high titres ranging from 4000 to 64 000 for HPV 6, 4000 to 64 000 for HPV 16, and 1000 to 64 000 for HPV 18 (Figure 1A–C). Compared with the baseline levels (at month 0) of the total study cohort and to the results obtained in controls at month 7, the elevation of immunisation-induced IgG antibodies in sera of vaccinees against the three tested HPV types was statistically significant (P<0.001 for all; Figure 1A–C).
Figure 1.
Total HPV-specific IgG antibodies in sera of the study cohort. IgG antibodies to (A) HPV 6, (B) HPV 16, and (C) HPV 18 were determined by virus-like particle ELISA at month 0 (baseline) in all study participants (n=34) and at month 7 in the vaccine (n=20) and control (n=14) cohort. The scatter plots display the serumdilutions, which gave a positive signal. The horizontal bars show the mean value for each group ±s.d.
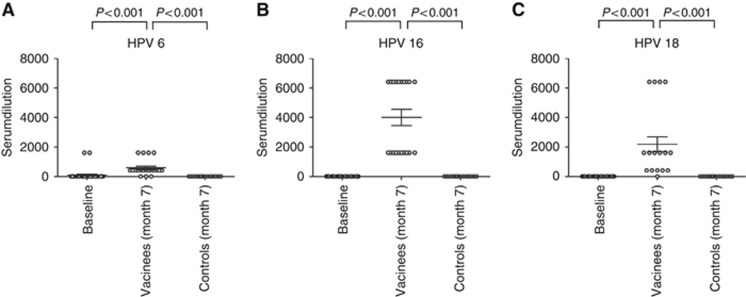
Neutralising antibodies are thought to represent the correlate of protection against HPV infection. In the vaccination cohort, neutralising antibodies against HPV 6 were detected in 75% (15/20) of the participants (titres ranging from 400 to 1600), anti-HPV 16 neutralising antibodies in all vaccinees (titres ranging from 1600 to 6400), and anti-HPV 18 neutralising antibodies in 95% (19/20) of the vaccinated participants (titres ranging from 400 to 6400) at month 7. The neutralising antibody titres of all three types were significantly higher in the vaccinees than the baseline titres (at month 0) of the total study cohort (P<0.001; Figure 2A–C). However, at study enrolment neutralising antibodies against HPV 6 were found in two study participants, both with titres of 1600, (Figure 2A). The two individuals belonged to the vaccine cohort and after vaccination the titres remained positive at the same dilution. Surprisingly, overall IgG against HPV 6, as determined by VLP-ELISA, were not detectable in these particular subjects at baseline and post-vaccination the anti-HPV 6 IgG levels increased to titres of 64 000. At month 7 type-specific neutralising antibodies to HPV 6, 16, and 18 in the sera of the vaccinated group were significantly elevated compared with non-immunised controls (P<0.001 for all three HPV types tested; Figure 2A–C). The type-specific levels of total IgG and neutralising antibodies in sera showed a significant correlation (P<0.001) for each tested HPV type, 6, 16, and 18 (not shown).
Figure 2.
Neutralising serum antibodies in sera of the study participants. Pseudovirion-based neutralisation assays were employed to determine the presence of neutralising antibodies to (A) HPV 6, (B) HPV 16, and (C) HPV 18 in the study cohort. Neutralising antibodies were evaluated in sera obtained at month 0 (baseline) from the total cohort (n=34) and at month 7 from the vaccinees (n=20) and the controls (n=14). The scatter plots display the serumdilutions, which gave a positive signal. The horizontal bars show the mean value for each group ±s.d.
After vaccination type-specific IgG antibodies to the vaccine types were detectable in oral fluids
To determine whether type-specific IgG antibodies are elicited after vaccination in oral fluids, samples were analysed by ELISA using VLP of HPV 6, 16, and 18 as the antigen.
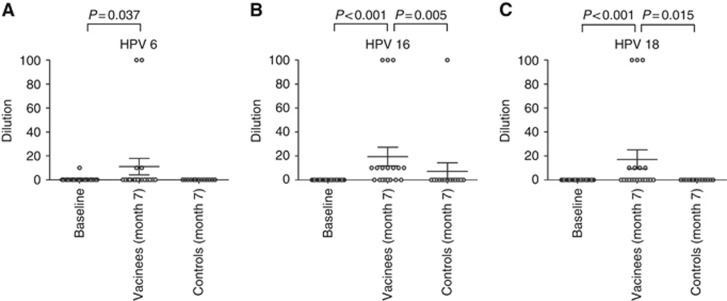
At month 0, antibodies against HPV 6 were found in one participant at a dilution of 10. All other participants did not reveal detectable antibodies in the oral fluids to any of the tested types at this time point. One month after completion of the vaccination series, antibodies against HPV 6 were found in 15% (3/20) of the vaccinees at dilutions of 10 to 100. The one individual that had detectable IgG against HPV 6 at baseline belonged to this cohort and remained anti-HPV 6 positive at the same dilution after vaccination. In the vaccine recipients IgG antibodies against HPV 16 were detectable in a majority (60% 12/20) and against HPV 18 in 35% (7/20) with detectable titres ranging from 10 to 100. Compared with baseline, titres were statistically significantly elevated at month 7 (P=0.037 for HPV 6, P<0.001 for HPV 16, P<0.001 for HPV 18; Figure 3A–C). In the control arm, one participant had antibodies against HPV 16 at a dilution of 100 at the 7-month time point. Neither anti-HPV 6 nor anti-HPV 18 antibodies were found in this individual. All other controls remained negative to the tested HPV types 6, 16, and 18 in the oral fluids throughout the study period (Figure 3A–C). HPV-specific IgG antibody levels in oral fluids of the vaccine group were significantly higher for HPV 16 (P=0.005) and HPV 18 (P=0.015) compared with the non-vaccinated group. Although antibody titres for HPV 6 were increased in vaccinated compared with non-vaccinated participants, differences were not statistically significant (P=0.080).
Figure 3.
Total HPV-specific IgG antibodies in oral fluids of the study cohort. By virus-like particle ELISA IgG antibodies to (A) HPV 6, (B) HPV 16, and (C) HPV 18 were detected in oral fluids. Oral fluid samples were obtained at month 0 (baseline; n=34) and at month 7 from the vaccine (n=20) and the control (n=14) groups. The plots display the dilutions of the oral fluids, at which a positive signal was obtained. The horizontal bars show the mean value for each group ±s.d.
Anti-HPV 16 IgG antibody levels in sera and oral fluids after vaccination are significantly correlated
Next, we compared the levels of IgG antibodies in sera and oral fluids against the same HPV type within the same vaccinated individual to determine whether evaluation of antibodies in oral fluids can be used as surrogate for measuring serum antibody titres. No statistically significant correlations were found for HPV 6 (P=0.052) and HPV 18 (P=0.418). However, this may be due to the low numbers of positives, especially when results were obtained from oral fluids. In contrast, anti-HPV 16 IgG antibodies in sera and in oral fluids correlated significantly (P=0.042; Table 1).
Table 1. Summary of IgG antibodies by virus-like particle ELISA against HPV 6, HPV 16, and HPV 18 in paired sera and oral fluids after HPV vaccination.
|
Vaccinees oral fluid titre |
Controls oral fluid titre |
|||||||
|---|---|---|---|---|---|---|---|---|
| Serum titre | 100 | 10 | 0 | Total | 100 | 10 | 0 | Total |
|
HPV 6 | ||||||||
| 64 000 | 0 | 1 | 12 | 13 | 0 | 0 | 0 | 0 |
| 16 000 | 2 | 1 | 3 | 6 | 0 | 0 | 0 | 0 |
| 4000 | 0 | 0 | 1 | 1 | 0 | 0 | 0 | 0 |
| 1000 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 1 |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 13 | 13 |
| Total | 2 | 2 | 16 | 20 | 0 | 0 | 14 | 14 |
|
HPV 16 | ||||||||
| 64 000 | 1 | 5 | 4 | 10 | 0 | 0 | 0 | 0 |
| 16 000 | 1 | 3 | 3 | 7 | 0 | 0 | 0 | 0 |
| 4000 | 2 | 0 | 1 | 3 | 0 | 0 | 0 | 0 |
| 1000 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 0 | 0 | 0 | 0 | 0 | 1 | 0 | 13 | 14 |
| Total | 4 | 8 | 8 | 20 | 1 | 0 | 13 | 14 |
|
HPV 18 | ||||||||
| 64 000 | 0 | 5 | 7 | 12 | 0 | 0 | 0 | 0 |
| 16 000 | 0 | 0 | 3 | 3 | 0 | 0 | 0 | 0 |
| 4000 | 0 | 0 | 4 | 4 | 0 | 0 | 0 | 0 |
| 1000 | 0 | 0 | 1 | 1 | 0 | 0 | 1 | 1 |
| 0 | 0 | 5 | 0 | 0 | 0 | 0 | 14 | 14 |
| Total | 0 | 5 | 15 | 20 | 0 | 0 | 14 | 14 |
HPV-specific antibodies present in oral fluids after HPV vaccination were neutralising
The important correlate for protection against HPV infection are thought to be neutralising antibodies, thus the proportion of neutralising antibodies in oral fluids were determined using PBNA assays. Of note, in contrast to serum samples, to our knowledge, these assays are not validated for oral fluids.
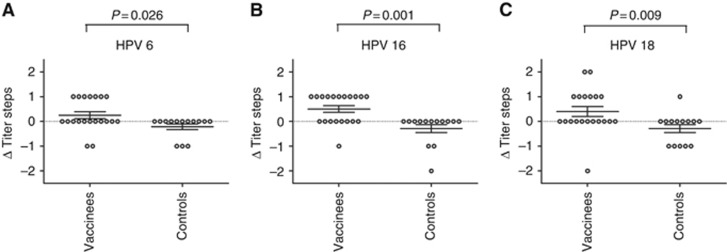
Neutralising antibody titres specific for HPV 6, 16 or 18 in the oral fluids were determined in vaccinees and controls at months 0 and 7, respectively. Both samples of an individual were analysed within the same experiment and on the same plate. To rule out any possible bias due to factors such as individual differences in composition or viscosity of oral fluids, results are given as differences in the titres between months 0 and 7 (Δ titre steps). In the vaccine cohort, 35% (7/20) of participants showed a one-step increase in neutralisation titres to HPV 6 after completion of the immunisation series compared with baseline. For HPV 16, 55% (11/20) of the vaccinees had an increase of one titre step at month 7. The highest increase of two titre steps were observed in 10% (2/20) of the vaccinated participants against HPV 18. A one-step increase against the same type 18 was noted for 30% (6/20) of this cohort. In the control population neutralising antibody titres were not elevated after the 7-month study period to any tested HPV types, with the exception of one participant, who had a one-step increase in the anti-HPV 18 neutralising titres at month 7 compared with baseline. However, anti-HPV 18 IgG were undetectable by VLP-ELISA in the oral fluid of this individual at any time point tested. Surprisingly, a decrease in the titres was noted for some study participants. In the vaccine population decreased titres were observed in 10% (2/20) for HPV 6, 5% (1/20) for HPV 16, and 5% (1/20) for HPV 18, and in the non-vaccinated population in 21.4% (3/14) for HPV 6 and HPV 16 and 35.7% (5/14) for HPV 18. Although the relevance of the observed decrease in titre steps is unclear, nevertheless, the negative differences were included in subsequent statistical calculations. Analyses on the differences in titre steps of the neutralising antibodies in oral fluids between the vaccine and the control groups at month 7 revealed statistically significant differences between changes in titre steps for all three tested types. For vaccinees the increase in the titres steps at month 7 was significantly higher compared with controls with P=0.026 for HPV 6, P=0.001 for HPV 16, and P=0.009 for HPV 18 (Figure 4A–C).
Figure 4.
Differences in neutralising antibody titres (Δ titre steps) in oral fluids between vaccinated and non-vaccinated participants. Neutralising antibody titres to (A) HPV 6, (B) HPV 16, and (C) HPV 18 were measured by pseudovirion-based neutralisation assays in oral fluids in all participants (n=34) at month 0 and month 7. The differences in titres between these time points are given as Δ titre steps. The plots display the Δ titre steps of the neutralising antibodies in oral fluids of the vaccinees and the controls. The horizontal bars show the mean value for each group ±s.d.
We next determined the number of immunisations that were necessary to elicit detectable neutralising antibodies in the oral fluids. However, to date the threshold of antibody titres that are sufficient for protection is not known. As prime example we chose high-risk HPV 16, as this type induces a robust immune response and represents the most important virus type. In our population, the majority (63% 7/11) mounted detectable anti-HPV 16 neutralising antibodies in the oral fluids only after the third and final vaccination (data not shown).
The increased titres of neutralising antibodies against high-risk HPV 16 found in sera and oral fluids of vaccinees at month 7 correlated significantly
We next investigated whether the HPV-vaccine-induced neutralising antibodies in sera correlated with neutralising antibodies in oral fluids within the same individual. As mentioned above, we regarded the differences in titre steps (Δ titre steps) of neutralising antibodies in oral fluids as more relevant than the absolute titres. Therefore, to allow for an appropriate comparison, we additionally determined the increases in titre steps for neutralising antibodies in sera. Similar to the results of type-specific total IgG antibodies between sera and oral fluids, no statistically significant correlations could be observed between neutralising antibody titres of sera and oral fluids for HPV 6 and HPV 18 (P=0.883 and P=0.806, respectively). In contrast, for high-risk mucosal HPV 16 the differences in titre steps of neutralising antibodies correlated significantly between corresponding sera and oral fluids (P=0.023) (Table 2).
Table 2. Summary of differences in titre steps (Δ titre steps) of neutralising antibodies against HPV 6, HPV 16, and HPV 18 in paired sera and oral fluids after HPV vaccination.
| Vaccinees Δ titre steps (oral fluids) | Controls Δ titre steps (oral fluids) | |||||||
|---|---|---|---|---|---|---|---|---|
|
HPV 6 | ||||||||
| Δ Titre steps (sera) | 1 | 0 | −1 | Total | 1 | 0 | −1 | Total |
| 0 | 2 | 0 | 0 | 2 | 0 | 11 | 3 | 14 |
| 1 | 4 | 7 | 2 | 13 | 0 | 0 | 0 | 0 |
| 2 | 1 | 1 | 0 | 2 | 0 | 0 | 0 | 0 |
| 3 | 0 | 3 | 0 | 3 | 0 | 0 | 1 | 1 |
| Total | 7 | 11 | 2 | 20 | 0 | 11 | 3 | 14 |
|
HPV 16 | ||||||||
| Δ Titre steps (sera) | 2 | 1 | 0 | Total | 2 | 1 | 0 | Total |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 | 14 | 14 |
| 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 2 | 0 | 3 | 7 | 10 | 0 | 0 | 0 | 0 |
| 3 | 0 | 8 | 2 | 10 | 0 | 0 | 0 | 0 |
| Total | 0 | 11 | 9 | 20 | 0 | 0 | 14 | 14 |
|
HPV 18 | ||||||||
| Δ Titre steps (sera) | 2 | 1 | 0 | Total | 2 | 1 | 0 | Total |
| 0 | 0 | 0 | 1 | 1 | 0 | 1 | 13 | 14 |
| 1 | 1 | 1 | 3 | 5 | 0 | 0 | 0 | 0 |
| 2 | 0 | 4 | 6 | 10 | 0 | 0 | 0 | 0 |
| 3 | 1 | 1 | 2 | 4 | 0 | 0 | 1 | 1 |
| Total | 2 | 6 | 12 | 20 | 0 | 1 | 13 | 14 |
Discussion
In this study, we found that HPV-vaccination induced specific antibodies to the vaccine-targeted types in blood and oral fluids. Importantly, we demonstrated that the elicited antibodies in oral fluids were capable of neutralising infectious PsV of the homologous HPV type in vitro and therefore may represent one protective correlate against infection of the oropharyngeal tract with the vaccine-targeted mucosal HPV types. Furthermore, we showed that the increased titres of neutralising antibodies against high-risk HPV 16—which causes the vast majority of HPV-attributable HNSCC—correlated significantly between sera and oral fluids of vaccinees. Therefore, (non-invasive) measurements of antibodies in oral fluids can be used in the future to assess vaccine-induced protection within the mucosal milieu of the orophayrynx, at least for this important type.
Equivalent work in the female genital tract has been published previously, demonstrating HPV-specific antibodies in sera and at the disease-relevant sites of the anogenital tract. HPV 16 and 18 antibodies were detected in cervicovaginal secretions of vaccinated girls and women and the measured levels strongly correlated with the respective titres in sera (Kemp et al, 2008; Schwarz et al, 2010; Petäjä et al, 2011).
The available prophylactic HPV vaccines are safe and highly efficacious in preventing persistent infection and associated diseases of the anogenital tract, caused by the vaccine-targeted HPV types. So far, two mechanisms seem to be responsible for the local protective effects at the infection sites by the circulating antibodies that are elicited by intramuscular immunisation. First, the circulating antibodies are thought to transudate from sera into the cervical mucus and, second, to exudate at sites of microtrauma or abrasions, which are a prerequisite for infection, into the surrounding tissues (Roden and Wu, 2006; Kemp et al, 2008). Albeit conceivable, so far it was not clear whether the protective mechanism(s) established for the anogenital tract are also applicable to HPV-induced diseases of the oropharynx. IgG antibodies against HPV 6, 11, and 16 were reported to be present in oral fluids of HIV-positive participants for whom the genital HPV status was unknown and in women with cervical dysplasia and carcinomas (Marais et al, 2001; Cameron et al, 2003; Marais et al, 2006). While after natural HPV infection antibodies are induced in about 70–80% of individuals with generally low and not very robust titres (Ho et al, 1998), the serological response after vaccination is up to four logs higher. Although the presence of HPV-specific IgG antibodies was reported in oral fluids after parenteral immunisation of a large cohort, the functionality, that is, the neutralisation potential, of the detected antibodies was unknown (Rowhani-Rahbar et al, 2009). Herein, we provide experimental proof for the presence of HPV-specific neutralising antibodies that are elicited by vaccination at oral sites and in oral fluids in humans and thus may be capable of preventing persisting oral HPV infection, similar to the anogenital site.
Oral fluid represents the liquid in the oral cavity, which consists of saliva—secreted by the salivary glands—and oral mucosal transudate, the serous liquid that, originating from the capillaries, passes the buccal and gingival mucosa to enter the mouth. Two major antibody classes are present in oral fluids: secretory IgA and IgG. Whereas IgA is synthesised by plasma cells in salivary glands, most IgG are derived from serum, although some IgG is locally produced. The translocation of IgG from blood to extracellular fluid occurs most notably in the dental capillary bed. Systemic vaccine administration strongly induces serum IgG antibodies, however, generally poorly stimulates secretory IgA at mucosal sites, as shown for instance for influenza (Moldoveanu et al, 1995; Belshe et al, 2000). As in this study we addressed the question whether intramuscular immunisation elicits neutralising IgG antibodies in sera and oral fluids, we focused on the detection of IgG antibodies. Evaluation of HPV-specific IgA in oral fluid to the vaccine-targeted types will be of interest for future investigations.
This study was not designed to examine whether elicited neutralising antibodies in the oral fluids can effectively prevent persistent infection and/or the development of oropharyngeal HPV-associated diseases in our study population. However, prior epidemiological studies provided evidence that HPV vaccination leads to a decrease in oral HPV infection (Herrero et al, 2013; Grün et al, 2015). Albeit limited by the small sample size of the study population, the findings presented herein indicate one possible mechanism how systemic HPV vaccination may exert protection from oral infection. In addition, owing to the fact that oropharyngeal HPV infection is largely associated with sexual behaviour, the decrease in the prevalence of anogenital HPV infection due to vaccination might indirectly reduce the incidence of oral HPV infection. This indicates the feasibility of HPV vaccination for protection against oropharyngeal HPV infection and associated diseases.
In addition to humoral immune responses, T-cell responses contribute to the overall protection against HPV infection and diseases. Vaccination-induced CD8+-driven T-cell response may represent the key mechanism whereby virus-infected cells are eliminated from both the oral cavity and the anogenital tract, presumably before clinical lesions are established. This is supported by previous observations on vaccinees who remained protected against HPV infection after waning of detectable antibody titres (Joura et al, 2008). To this end, L1-specific T-cell responses were measured after vaccination by proliferation of both CD4+ and CD8+ T cells and efficient cytokine responses inducing both, Th1 and Th2, patterns of cellular immune responses were detected in the vaccinees (Pinto et al, 2003; Pinto et al, 2005; Weinberg et al, 2012). However, further studies to elucidate the role of cytotoxic T cells in mucosal HPV (re)infection need to be performed.
Acknowledgments
We thank Viktoria Gruber for excellent technical help. The work was supported by a grant (LS11–006) to RK from the Vienna Science and Technology Fund (WWTF).
Footnotes
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License.
RK is a coinventor on HPV16 L1 patents licensed to GlaxoSmithKline (GSK) and Merck (MSD). The remaining authors declare no conflict of interest.
References
- Belshe RB, Gruber WC, Mendelman PM, Mehta HB, Mahmood K, Reisinger K, Treanor J, Zangwill K, Hayden FG, Bernstein DI, Kotloff K, King J, Piedra PA, Block SL, Yan L, Wolff M (2000) Correlates of immune protection induced by live, attenuated, cold-adapted, trivalent, intranasal influenza virus vaccine. J Infect Dis 181: 1133–1137. [DOI] [PubMed] [Google Scholar]
- Bernard HU, Burk RD, Chen Z, van Doorslaer K, zur Hausen H, de Villiers EM (2010) Classification of papillomaviruses (PVs) based on 189 PV types and proposal of taxonomic amendments. Virology 401: 70–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buck CB, Pastrana DV, Lowy DR, Schiller JT (2005) Generation of HPV pseudovirions using transfection and their use in neutralization assays. Methods Mol Med 119: 445–462. [DOI] [PubMed] [Google Scholar]
- Cameron JE, Snowhite IV, Chaturvedi AK, Hagensee ME (2003) Human papillomavirus-specific antibody status in oral fluids modestly reflects serum status in human immunodeficiency virus-positive individuals. Clin Diagn Lab Immunol 10: 431–438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung CH, Bagheri A, D'Souza G (2014) Epidemiology of oral human papillomavirus infection. Oral Oncol 50: 364–369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Vincenzo R, Conte C, Ricci C, Scambia G, Capelli G (2014) Long-term efficacy and safety of human papillomavirus vaccination. Int J Womens Health 6: 999–1010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D'Souza G, Kreimer AR, Viscidi R, Pawlita M, Fakhry C, Koch WM, Westra WH, Gillison ML (2007) Case-control study of human papillomavirus and oropharyngeal cancer. N Engl J Med 356: 1944–1956. [DOI] [PubMed] [Google Scholar]
- Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F (2015) Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 136: E359–E386. [DOI] [PubMed] [Google Scholar]
- Gillison ML, Koch WM, Capone RB, Spafford M, Westra WH, Wu L, Zahurak ML, Daniel RW, Viglione M, Symer DE, Shah KV, Sidransky D (2000) Evidence for a causal association between human papillomavirus and a subset of head and neck cancers. J Natl Cancer Inst 92: 709–720. [DOI] [PubMed] [Google Scholar]
- Gillison ML, D'Souza G, Westra W, Sugar E, Xiao W, Begum S, Viscidi R (2008) Distinct risk factor profiles for human papillomavirus type 16-positive and human papillomavirus type 16-negative head and neck cancers. J Natl Cancer Inst 100: 407–420. [DOI] [PubMed] [Google Scholar]
- Grün N, Ährlund-Richter A, Franzén J, Mirzaie L, Marions L, Ramqvist T, Dalianis T (2015) Oral human papillomavirus (HPV) prevalence in youth and cervical HPV prevalence in women attending a youth clinic in Sweden, a follow up-study 2013-2014 after gradual introduction of public HPV vaccination. Infect Dis (Lond) 47: 57–61. [DOI] [PubMed] [Google Scholar]
- Handisurya A, Schellenbacher C, Reininger B, Koszik F, Vyhnanek P, Heitger A, Kirnbauer R, Förster-Waldl E (2010) A quadrivalent HPV vaccine induces humoral and cellular immune responses in WHIM immunodeficiency syndrome. Vaccine 28: 4837–4841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harper DM, Franco EL, Wheeler CM, Moscicki AB, Romanowski B, Roteli-Martins CM, Jenkins D, Schuind A, Costa Clemens SA, Dubin G HPV Vaccine Study group (2006) Sustained efficacy up to 4.5 years of a bivalent L1 virus-like particle vaccine against human papillomavirus types 16 and 18: follow-up from a randomised control trial. Lancet 367: 1247–1255. [DOI] [PubMed] [Google Scholar]
- Heck JE, Berthiller J, Vaccarella S, Winn DM, Smith EM, Shan'gina O, Schwartz SM, Purdue MP, Pilarska A, Eluf-Neto J, Menezes A, McClean MD, Matos E, Koifman S, Kelsey KT, Herrero R, Hayes RB, Franceschi S, Wünsch-Filho V, Fernández L, Daudt AW, Curado MP, Chen C, Castellsagué X, Ferro G, Brennan P, Boffetta P, Hashibe M (2010) Sexual behaviours and the risk of head and neck cancers: a pooled analysis in the International Head and Neck Cancer Epidemiology (INHANCE) consortium. Int J Epidemiol 39: 166–181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herrero R, Quint W, Hildesheim A, Gonzalez P, Struijk L, Katki HA, Porras C, Schiffman M, Rodriguez AC, Solomon D, Jimenez S, Schiller JT, Lowy DR, van Doorn LJ, Wacholder S, Kreimer AR CVT Vaccine Group (2013) Reduced prevalence of oral human papillomavirus (HPV) 4 years after bivalent HPV vaccination in a randomized clinical trial in Costa Rica. PLoS One 8: e68329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herrero R, González P, Markowitz LE (2015) Present status of human papillomavirus vaccine development and implementation. Lancet Oncol 16: e206–e216. [DOI] [PubMed] [Google Scholar]
- Ho GY, Bierman R, Beardsley L, Chang CJ, Burk RD (1998) Natural history of cervicovaginal papillomavirus infection in young women. N Engl J Med 338: 423–428. [DOI] [PubMed] [Google Scholar]
- Joura EA, Kjaer SK, Wheeler CM, Sigurdsson K, Iversen OE, Hernandez-Avila M, Perez G, Brown DR, Koutsky LA, Tay EH, García P, Ault KA, Garland SM, Leodolter S, Olsson SE, Tang GW, Ferris DG, Paavonen J, Lehtinen M, Steben M, Bosch X, Dillner J, Kurman RJ, Majewski S, Muñoz N, Myers ER, Villa LL, Taddeo FJ, Roberts C, Tadesse A, Bryan J, Lupinacci LC, Giacoletti KE, Lu S, Vuocolo S, Hesley TM, Haupt RM, Barr E (2008) HPV antibody levels and clinical efficacy following administration of a prophylactic quadrivalent HPV vaccine. Vaccine 26: 6844–6851. [DOI] [PubMed] [Google Scholar]
- Kemp TJ, García-Piñeres A, Falk RT, Poncelet S, Dessy F, Giannini SL, Rodriguez AC, Porras C, Herrero R, Hildesheim A, Pinto LA Costa Rica Vaccine Trial (CVT) Group (2008) Evaluation of systemic and mucosal anti-HPV16 and anti-HPV18 antibody responses from vaccinated women. Vaccine 26: 3608–3616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirnbauer R, Taub J, Greenstone H, Roden R, Dürst M, Gissmann L, Lowy DR, Schiller JT (1993) Efficient self-assembly of human papillomavirus type 16 L1 and L1-L2 into virus-like particles. J Virol 67: 6929–6936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirnbauer R, Hubbert NL, Wheeler CM, Becker TM, Lowy DR, Schiller JT (1994) A virus-like particle enzyme-linked immunosorbent assay detects serum antibodies in a majority of women infected with human papillomavirus type 16. J Natl Cancer Inst 86: 494–499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kreimer AR, Clifford GM, Boyle P, Franceschi S (2005) Human papillomavirus types in head and neck squamous cell carcinomas worldwide: a systematic review. Cancer Epidemiol Biomarkers Prev 14: 467–475. [DOI] [PubMed] [Google Scholar]
- Kreimer AR, Bhatia RK, Messeguer AL, González P, Herrero R, Giuliano AR (2010) Oral human papillomavirus in healthy individuals: a systematic review of the literature. Sex Transm Dis 37: 386–391. [DOI] [PubMed] [Google Scholar]
- Marais DJ, Best JM, Rose RC, Keating P, Soeters R, Denny L, Dehaeck CM, Nevin J, Kay P, Passmore JA, Williamson AL (2001) Oral antibodies to human papillomavirus type 16 in women with cervical neoplasia. J Med Virol 65: 149–154. [PubMed] [Google Scholar]
- Marais DJ, Sampson C, Jeftha A, Dhaya D, Passmore JA, Denny L, Rybicki EP, Van Der Walt E, Stephen LX, Williamson AL (2006) More men than women make mucosal IgA antibodies to Human papillomavirus type 16 (HPV-16) and HPV-18: a study of oral HPV and oral HPV antibodies in a normal healthy population. BMC Infect Dis 6: 95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markowitz LE, Dunne EF, Saraiya M, Chesson HW, Curtis CR, Gee J, Bocchini JA Jr, Unger ER Centers for Disease Control and Prevention (CDC) (2014) Human papillomavirus vaccination: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 63: 1–30. [PubMed] [Google Scholar]
- Marur S, D'Souza G, Westra WH, Forastiere AA (2010) HPV-associated head and neck cancer: a virus-related cancer epidemic. Lancet Oncol 11: 781–789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mehanna H, Beech T, Nicholson T, El-Hariry I, McConkey C, Paleri V, Roberts S (2013) Prevalence of human papillomavirus in oropharyngeal and nonoropharyngeal head and neck cancer—systematic review and meta-analysis of trends by time and region. Head Neck 35: 747–755. [DOI] [PubMed] [Google Scholar]
- Moldoveanu Z, Clements ML, Prince SJ, Murphy BR, Mestecky J (1995) Human immune responses to influenza virus vaccines administered by systemic or mucosal routes. Vaccine 13: 1006–1012. [DOI] [PubMed] [Google Scholar]
- Ndiaye C, Mena M, Alemany L, Arbyn M, Castellsagué X, Laporte L, Bosch FX, de Sanjosé S, Trottier H (2014) HPV DNA, E6/E7 mRNA, and p16INK4a detection in head and neck cancers: a systematic review and meta-analysis. Lancet Oncol 15: 1319–1331. [DOI] [PubMed] [Google Scholar]
- Petäjä T, Pedersen C, Poder A, Strauss G, Catteau G, Thomas F, Lehtinen M, Descamps D (2011) Long-term persistence of systemic and mucosal immune response to HPV-16/18 AS04-adjuvanted vaccine in preteen/adolescent girls and young women. Int J Cancer 129: 2147–2157. [DOI] [PubMed] [Google Scholar]
- Pinto LA, Castle PE, Roden RB, Harro CD, Lowy DR, Schiller JT, Wallace D, Williams M, Kopp W, Frazer IH, Berzofsky JA, Hildesheim A (2005) HPV-16 L1 VLP vaccine elicits a broad-spectrum of cytokine responses in whole blood. Vaccine 23: 3555–3564. [DOI] [PubMed] [Google Scholar]
- Pinto LA, Edwards J, Castle PE, Harro CD, Lowy DR, Schiller JT, Wallace D, Kopp W, Adelsberger JW, Baseler MW, Berzofsky JA, Hildesheim A (2003) Cellular immune responses to human papillomavirus (HPV)-16 L1 in healthy volunteers immunized with recombinant HPV-16 L1 virus-like particles. J Infect Dis 188: 327–338. [DOI] [PubMed] [Google Scholar]
- Roden R, Wu TC (2006) How will HPV vaccines affect cervical cancer? Nat Rev Cancer 6: 753–763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rose RC, Bonnez W, Da Rin C, McCance DJ, Reichman RC (1994) Serological differentiation of human papillomavirus types 11, 16 and 18 using recombinant virus-like particles. J Gen Virol 75: 2445–2449. [DOI] [PubMed] [Google Scholar]
- Rowhani-Rahbar A, Carter JJ, Hawes SE, Hughes JP, Weiss NS, Galloway DA, Koutsky LA (2009) Antibody responses in oral fluid after administration of prophylactic human papillomavirus vaccines. J Infect Dis 200: 1452–1455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwarz TF, Kocken M, Petäjä T, Einstein MH, Spaczynski M, Louwers JA, Pedersen C, Levin M, Zahaf T, Poncelet S, Hardt K, Descamps D, Dubin G (2010) Correlation between levels of human papillomavirus (HPV)-16 and 18 antibodies in serum and cervicovaginal secretions in girls and women vaccinated with the HPV-16/18 AS04-adjuvanted vaccine. Hum Vaccin 6: 1054–1061. [DOI] [PubMed] [Google Scholar]
- Steenbergen RD, Hermsen MA, Walboomers JM, Joenje H, Arwert F, Meijer CJ, Snijders PJ (1995) Integrated human papillomavirus type 16 and loss of heterozygosity at 11q22 and 18q21 in an oral carcinoma and its derivative cell line. Cancer Res 55: 5465–5471. [PubMed] [Google Scholar]
- Taliercio S, Cespedes M, Born H, Ruiz R, Roof S, Amin MR, Branski RC (2015) Adult-onset recurrent respiratory papillomatosis: a review of disease pathogenesis and implications for patient counseling. JAMA Otolaryngol Head Neck Surg 141: 78–83. [DOI] [PubMed] [Google Scholar]
- Venkatesan NN, Pine HS, Underbrink MP (2012) Recurrent respiratory papillomatosis. Otolaryngol Clin North Am 45: 671–694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weinberg A, Song LY, Saah A, Brown M, Moscicki AB, Meyer WA 3rd, Bryan J, Levin MJ IMPAACT/PACTG P1047 Team (2012) Humoral, mucosal, and cell-mediated immunity against vaccine and nonvaccine genotypes after administration of quadrivalent human papillomavirus vaccine to HIV-infected children. J Infect Dis 206: 1309–1318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WHO (2014) Human papillomavirus vaccines: WHO position paper, October 2014-Recommendations. Vaccine 33(36): 4383–4384. [DOI] [PubMed] [Google Scholar]