Abstract
There is an increasing awareness of the role of macrophages in the regulation and maintenance of gastrointestinal function in health and disease. This work has proceeded in the context of an increased understanding of the complex phenotypic variation in macrophages throughout the body and has shown previously unidentified roles for macrophages in diseases such as gastroparesis, postoperative ileus, and inflammatory bowel disease. Opportunities for exploiting the phenotypic modulation of tissue resident macrophages have been identified as possible therapies for some of these diseases. In addition, macrophages are an established component of the innate immune system and can respond to variations and changes in the intestinal microbiome and potentially mediate part of the impact of the microbiota on intestinal health. We reviewed the latest work on novel concepts in defining macrophage phenotype, discuss possible mechanisms of action for tissue-resident macrophages in the gut, address the significance of microbiome effects on macrophage phenotype, and review the known and possible roles of macrophages in motility disorders of the gastrointestinal tract.
Keywords: Monocyte–Macrophage Precursor Cells, Gastrointestinal Motility, Enteric Nervous System, Interstitial Cells of Cajal
Abbreviations used in this paper: BMP, bone morphogenetic protein; CCR2, C-C chemokine receptor type 2; CD206, mannose receptor C, Type 1 aka MRC1; CSF1, colony-stimulating factor 1; CSF2, granulocyte monocyte colony stimulating factor; CX3CR1, chemokine receptor 1; GI, gastrointestinal; HO-1, heme oxygenase 1; IBD, inflammatory bowel disease; ICC, interstitial cells of Cajal; IL, interleukin; iNOS, inducible nitic oxide synthase; Ly6C, lymphocyte antigen 6C; MCP1, monocyte chemoattractive protein-1; NOD, nonobese diabetic mouse; op/op, osteopetrotic mouse; TGF, transforming growth factor; TGM2, transglutaminase 2; TNBS, 2,4,6-trinitrobenzene sulfonic acid; TNF, tumor necrosis factor
Summary.
This review focuses on the role of muscularis propria macrophages in gastrointestinal tract motility under both normal and pathologic conditions.
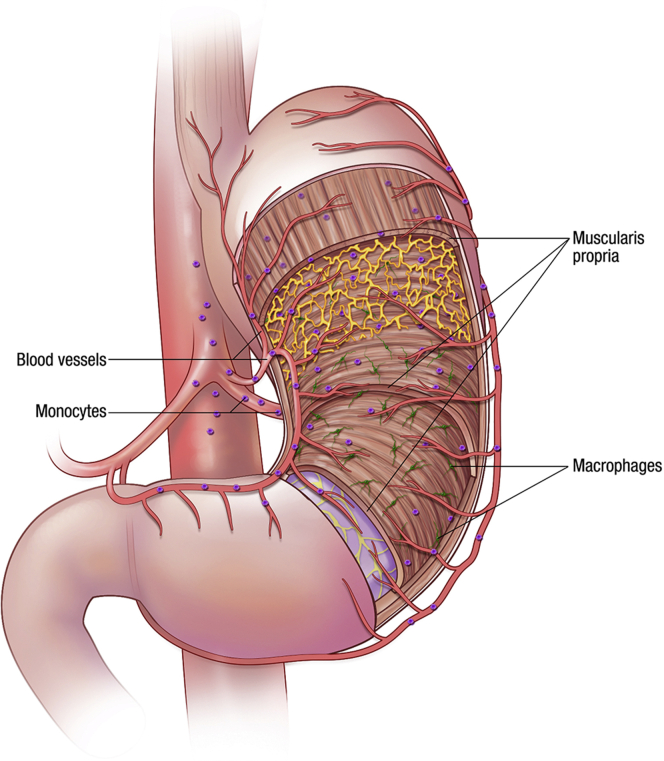
The extraordinary cellular complexity of the gastrointestinal (GI) tract is a reflection of the diverse roles of the organ. From an immune perspective, this means that there are complex interactions between multiple cell types that differ along the length as well as across the thickness of the wall of the GI tract. These interactions are constantly changing with development, age, alterations to the luminal content, and disease. This review is about one cell type, the macrophages, that within the GI tract represent the largest population of macrophages in the body1, 2 and can be involved in multiple GI functions. We review recent discoveries with respect to the complex phenotypic variations in macrophages and how these are modified in health, development, and disease. We refer to cells in all layers of the wall of the GI tract as shown in Figure 1, which shows the sources and destinations of macrophages in the GI tract. The primary focus is on macrophages residing in the muscularis propria and the consequences of their phenotypic changes on GI motility. Because there already are extensive reviews in the literature on the origins and roles of macrophages in the mucosa and submucosa,3, 4 the functions and properties of those macrophages are discussed in less detail and in the context of disturbances to GI motility or in comparison with the functions and properties of muscularis propria macrophages.
Figure 1.
Distribution of resident macrophages in the gastric muscularis propria. Circulating monocytes are recruited to the tissue where they become macrophages influenced by the local environment.
Macrophages in the Gastrointestinal Tract: Where Do They Come From?
Many types of cells of myeloid lineage including macrophages, but also microglia in the brain, Kupffer cells in the liver, and bone osteoclasts, are considered to be tissue resident.5 The source and maintenance of these cells are either ongoing population from circulating monocytes with varying rates of turnover or in some cases by population from yolk sac–derived progenitors and maintenance by self-renewal. Microglia represent a well-defined example of these self-renewing, stable, tissue resident cells.6 Renewal of resident macrophages is also from macrophage precursors that develop in the bone marrow, then enter into the blood stream as mature monocytes, and, finally, migrate into a specific tissue where they develop into a resident macrophage population.7 In many tissues they develop from primitive macrophages existing in the yolk sac or fetal liver and are maintained independently from bone marrow–derived monocytes in the steady-state condition.4, 8, 9, 10 However, by tracking macrophage development from birth to adulthood in the mucosa and lamina propria, it was observed that although primitive macrophages are present in the neonatal intestinal mucosa and lamina propria they are not maintained in adulthood.11, 12 This important finding suggests that in contrast to other tissues, macrophages that reside in the GI mucosa and lamina propria of adult mice are replaced continuously by blood monocytes and that yolk sac–derived macrophages are short-lived and/or rare. This is reasonable given that mucosal and lamina propria macrophages are thought to monitor and respond to multiple factors at the interface between the organism and the luminal environment, and that these factors and responses are variable and can change rapidly. The resident macrophages of the muscle layers have not been studied in as much detail. These cells are abundant throughout the GI tract and express a limited number of phenotypic markers of activation.13, 14, 15, 16, 17 It is clear that monocytes can be detected in the muscularis propria layers of control mice, an immune cell infiltrate derived from monocytes can be detected after inflammation in animal models,18, 19 and that varying numbers of macrophages can be identified by mannose receptor, C Type 1 (CD206, MRC1) immunoreactivity in human gastric muscularis propria.20 Our recent data have indicated that population of the gastric muscularis propria in macrophage-depleted, osteopetrotic (op/op) mice can occur by monocyte invasion and can produce both CD206-positive and CD206-negative macrophages. These studies all identified macrophages that express markers that are not present on resident macrophages in healthy, GI smooth muscle, such as those in the GI tract of nonobese diabetic (NOD) mice when they are not diabetic.21 Thus, muscularis propria macrophages are derived at least partially from monocytes and it remains to be determined whether some resident macrophages are the equivalent of microglia in the brain, derived from yolk sac progenitors and sustained by self-renewal. Determining the fate and source of all macrophages in the muscularis propria is an important research opportunity for the future.
Macrophage Phenotypes in Healthy Tissues
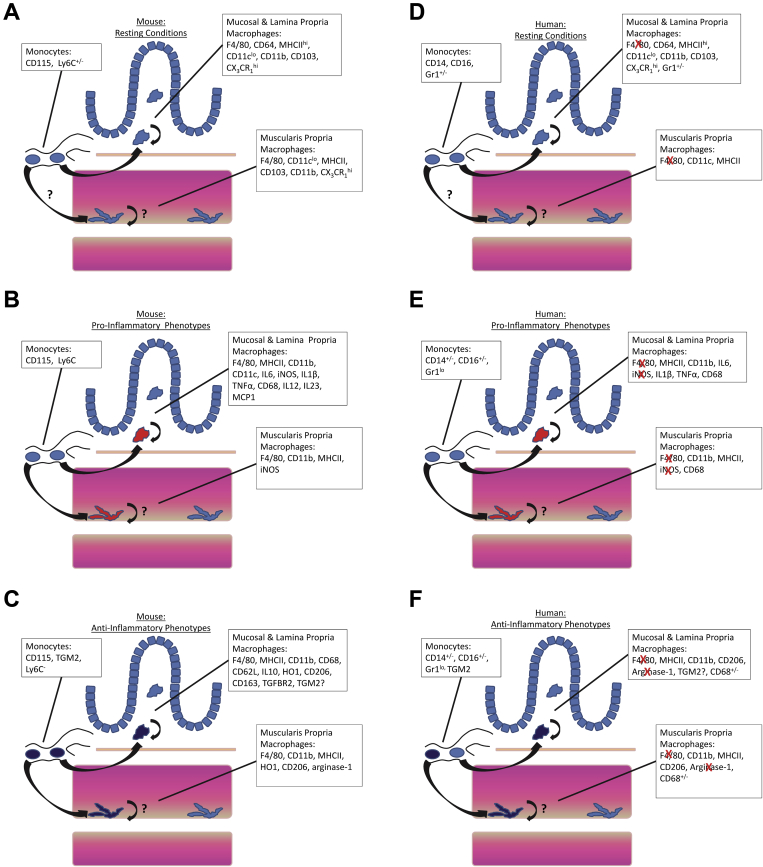
The phenotype of macrophages in healthy tissues appears to be determined in part by the original monocyte progenitor from which the macrophages were derived as shown in Figure 2 and Supplemental Poster. This is shown most clearly in op/op mice, which show reduced numbers of macrophages in most tissues. Monocyte development and survival depends on colony-stimulating factor 1 (macrophage) (CSF1), and op/op mice have an inactivating mutation in the gene encoding CSF1.15, 22
Figure 2.
Summary of the published markers of macrophage and monocyte phenotype in (A–C) mouse and (D–F) human gastrointestinal tract. Note that differing expression patterns likely are dependent on the disease state, stimulus, or genetic background of the tissue under study (see text and Table 1 for more detail). Red “X” indicates genes that are expressed in mouse but not human macrophages.
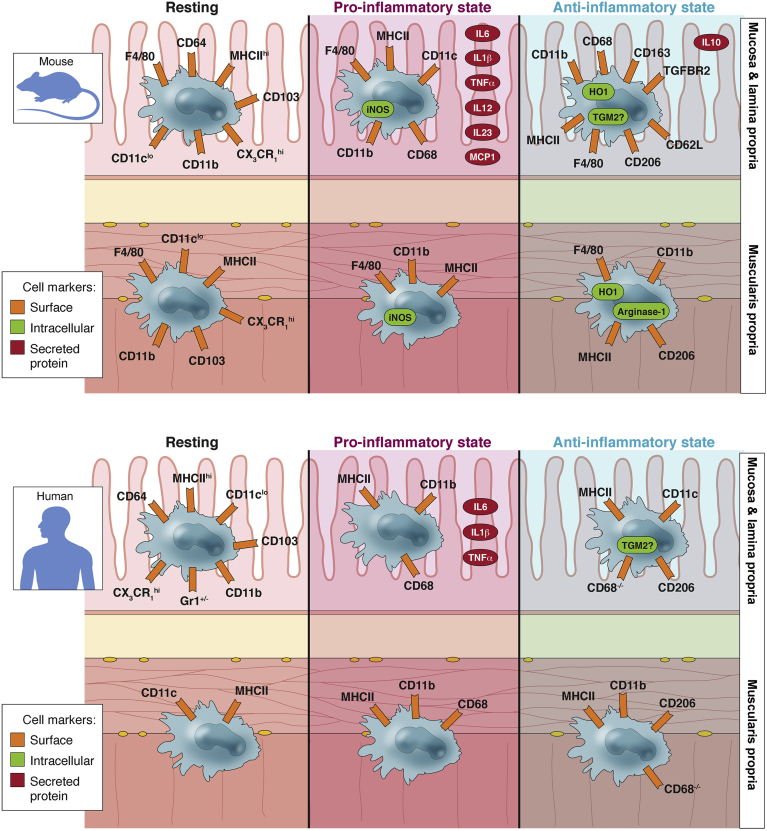
Supplemental Poster.
Tissue resident macrophages in the gastrointestinal wall express a variety of cell-surface and intracellular markers as well as secreted cytokines and chemokines. These markers change in response to injury or disease. If the macrophages polarize towards a pro-inflammatory state then the cells contribute towards tissue injury and death and increase inflammation, if the macrophages polarize towards to an anti-inflammatory state then the cells release molecules that can reverse inflammation and promote tissue healing and repair. Phenotypic characterization of macrophages is more thorough in the mucosa and lamina propria of both humans and rodents. Data on the source and mechanism of activation of macrophages in the muscularis propria is less well characterized. It should also be noted that certain proteins including iNOS and arginase-1 are expressed in rodent macrophages but not in human macrophages. (Cell Mol Gastroenterol Hepatol 2016;2:120–130;http://dx.doi.org/10.1016/j.jcmgh.2016.01.003)
In mice, monocytes can be identified by CD115 immunoreactivity and can be divided into 2 different groups according to differences in lymphocyte antigen 6C (Ly6C) expression. CD115+ Ly6C+ monocytes are associated with an inflammatory phenotype and differentiation into conventionally activated, proinflammatory M1 macrophages or inflammatory dendritic cells when they enter tissues. In the GI mucosa and lamina propria, Ly6chi monocytes develop an intermediate phenotype once inside the tissue, defined as chemokine receptor 1int (CX3CR1int) before they differentiate into mature macrophages.12, 23 It takes 4–5 days for monocytes to acquire the phenotype of resident mucosal and lamina propria macrophages characterized by the expression of F4/80, CD64, major histocompatibility complex II, CD11c, and CX3CR1, and are associated with a slower rate of cellular turnover.12 CX3CR1 expression is associated with a signature cytokine profile. CX3CR1- macrophages are characterized by high levels of proinflammatory cytokines such as interleukin (IL)6, inducible nitric oxide synthase (iNOS, NOS2), IL1β, and tumor necrosis factor (TNF)α.12, 24 In contrast, CD115+, Ly6C- monocytes do not express inflammatory markers, are long-lived, and contribute to the resident macrophage population in GI mucosa.25, 26 Ly6Clo monocytes appear to be a precursor of a subset of resident CX3CR1+ macrophages that are characterized by lower levels of C-C chemokine receptor type 2 (CCR2) and CD62L (L-selectin), and have an anti-inflammatory signature with increased levels of IL10 and heme oxygenase-1 (HO-1) and CD206.12 These Ly6Clo monocytes are a possible alternative to yolk sac progenitors as a source of resident macrophages in GI muscularis propria, but this has not been tested and it should be noted that the resident macrophages in the healthy muscularis propria express very low levels of HO-1 and CD206 in, for example, nondiabetic NOD mice.21 Furthermore, flow cytometry and immunohistochemistry indicate that macrophages in healthy mouse intestinal muscularis propria are a homogeneous population of major histocompatibility complex IIhi, CD11cloCD103+ CD11b+ expressing high levels of CX3CR1.27 Further research is necessary in understanding the source and fate of muscularis propria macrophages, with close attention to the strain of the mice under study. The immune systems in mice do vary widely by strain28 and some of the differences in macrophage phenotype and markers identified by different studies likely can be ascribed to differences in the genetic background strains used.
In human beings, 3 monocyte populations have been defined based on CD14 and CD16 expression with distinct differences from mice in the markers and likely fate of those cells. For example, the equivalent of the mouse marker Ly6C is the Gr1 epitope in human monocytes, but although mouse Ly6C+ monocytes are proinflammatory, Gr1+ human monocytes release IL10 and do not express proinflammatory markers.26 These differences are of great importance because to translate the large body of work performed in mice to understanding human health and disease, it is necessary to fully characterize human macrophages. One effort to achieve this was a comprehensive survey of markers conserved on macrophages cultured and differentiated from human and mouse blood monocytes and then validated in human lung tissue. An 87-gene transcriptome was identified that can be used as a basis for characterizing human macrophages in other tissues23 including the GI mucosa, lamina propria, and muscularis propria. In particular, transglutaminase 2 (TGM2) is identified as a novel marker of alternatively activated, anti-inflammatory M2 macrophages that is as reliable as CD206 for tissue macrophages, but that also labels circulating monocytes with an anti-inflammatory phenotype, unlike CD206/mannose receptor C1. It will be very interesting to see how TGM2 expression correlates with disorders of gastrointestinal function, especially if it proves that the levels of circulating TGM2-positive monocytes correlate with the numbers of TGM2 or CD206-positive tissue macrophages.
Macrophage Phenotypes: What Happens in Disease and Injury?
Changes in macrophage phenotype or activation of macrophages frequently are evident in response to disease or injury as discussed later for GI disorders. A variety of factors initiate signaling cascades that alter the expression of phenotypic markers in tissue macrophages in human beings and in animal models of disease. Recent position pieces and reviews have moved beyond a strict division of macrophages into defined inflammatory vs anti-inflammatory, M1 vs M2, or conventionally vs alternatively activated phenotypes.29, 30 It is clear that the tissue, strain, and species under study and the injury or disease that activated the macrophages can produce many different outcomes. In the GI tract, specific enzymes and cytokines have been identified when the tissues are challenged by injury or disease; these include iNOS, TNFα, various interleukins and cytokines, HO-1, arginase-1, or CD206. The association of these molecules with inflammation, repair, or suppression of inflammation is summarized in Table 1. Some of these markers are reliable when studied in some species but not others; for example, iNOS is expressed robustly in mouse macrophages in inflammation but is repressed epigenetically in human macrophages.31 Arginase-1 and Ym1 are examples of other proteins that are expressed in mouse but not human M2 macrophages.32 Many of the markers are autocrine factors that feed back to increase or decrease the level of inflammation in injured tissues, or paracrine factors that support or repress the invasion and differentiation of cells in the affected tissue. IL10 is an example of an autocrine factor that is released from and promotes CD206-positive, anti-inflammatory macrophages, while suppressing proinflammatory markers in other macrophages and immune cells.33, 34 For example, in Toxoplasma gondii ileitis in the mouse, prostaglandin E2–dependent IL10 production after accumulation of Ly6Chi monocytes inhibits production of tissue-damaging TNFα by neutrophils.35 We discuss later the changing balance in proinflammatory and anti-inflammatory markers in macrophages of the mouse gastric muscularis propria that leads to discrete temporal patterns of cellular injury during diabetes and the development of gastroparesis.
Table 1.
Protein Markers Used to Study and Characterize Macrophages, Monocytes, and Dendritic Cells in the GI Tract, Whether Associated With Proinflammatory or Anti-inflammatory States of the Cells or as Markers of Phenotype and Cell Type
| Proinflammatory markers |
Anti-inflammatory markers |
Other markers |
|||
|---|---|---|---|---|---|
| Marker | Notes | Marker | Notes | Marker | Notes |
| iNOS | Not in human | CD206 | Classic M2 marker | MCP1 | Monocyte recruitment |
| IL1β | HO-1 | Produces CO | CSF1 | Monocyte differentiation | |
| IL6 | Arginase-1 | Lowers substrate for iNOS activity, not human | BMP2 | Cellular signaling | |
| IL12 | TGFBR2 | Receptor of TGFβ | CD115 | CSF1 receptor | |
| IL23 | CD163 | Heme transport | CX3CR1 | Also on lymphocytes | |
| TNFα | Target of infliximab, adalimumab, and so forth | IL10 | MHCII | Antigen-presenting cells | |
| CCR2 | Receptor for MCP1 | 5-HT2B | Receptor for serotonin | CD68 | Phagocytic cells |
| Ly6C | High on proinflammatory monocytes | 5-HT7 | Receptor for serotonin | F4/80 | Epitope on EMR1 (mouse only) |
| CD11B | Also known as integrin αM, MAC1, CR3 | Nicotinic α7R | Receptor for acetylcholine | CD64 | Also known as FcγR1 |
| SMAD7 | Suppressor of TGFβ signaling | CD103 | Dendritic cells | CD11c | High on dendritic cells |
| E-cadherin | Dendritic cells | CD62L | Also known as L-selectin. Also in lymphocytes | ||
| Gr1 | Epitope on human Ly6G | ||||
| MyD88 | Toll-like receptor adapter protein | ||||
NOTE. For details and references see text. Figure 2 provides additional information.
CD62L, L-selectin; MHCII, major histocompatibility complex II; MyD88, myeloid differentiation primary response gene 88; TGFBR2, transforming growth factor-β receptor II.
The eventual phenotype of activated macrophages in disease also is determined significantly by the cellular origin of the cells. The underlying mechanisms can involve activation of monocyte invasion, expansion of the resulting macrophages in situ after monocyte invasion, or expansion of resident macrophages. For muscularis propria macrophages, these mechanisms have not been studied extensively. On the other hand, there have been many studies on mucosal and lamina propria macrophages in animal models of inflammation and human tissues. In the mucosa and lamina propria of C57Bl6 mouse colon, dextran sulfate sodium–induced inflammation initiates a tissue invasion of Ly6Chi monocytes that expand into CX3CR1int macrophages, characterized by high levels of IL6, TNFα, CCR2, IL1β, IL12, and IL23.12, 36 At the same time, the CX3CR1hi population, representing the resident macrophages, does not change because it is the infiltrating monocytes that develop into a CX3CR1int phenotype. In fact, during the same period of time, the CX3CR1hi population maintains its anti-inflammatory phenotype, characterized by high production of CD206, CD163, and transforming growth factor-β receptor II.12 CCR2 activation by monocyte chemoattractive protein-1 (MCP-1) has an important role in Ly6Chi monocyte accumulation in inflamed tissues as indicated in studies on the CCR2 knockout mouse showing reduced systemic inflammation37 and low levels of monocyte invasion in atherosclerosis,38 type II diabetes, and obesity.39 In the GI tract, CCR2 knockout mice have reduced mucosal inflammation and damage after dextran sulfate sodium colitis40 and in rats, 2,4,6-trinitrobenzene sulfonic acid–induced injury to the muscularis propria is reduced by MCP-1–neutralizing antibodies.18 In surveying the complexities of macrophage activation, it is evident that the mechanisms and regulation of activation are not worked out in sufficient detail and certainly not for the muscularis propria. However, there are real opportunities for understanding and treating disease by examining these important cellular processes.
Potential Roles of Macrophages
Regulation of Excitable Cells in the Muscularis Propria
Macrophages have been observed in the muscularis propria in proximity to neuronal processes and interstitial cells of Cajal (ICC), cells that are fundamental for the maintenance of normal GI function.13, 16, 41 In this context, these muscularis macrophages have been proposed not only to be immune regulators, but also to play a housekeeping role and be involved in the maintenance of normal motility in the healthy gut. With respect to ICCs, muscularis macrophages are observed to be enveloped by and in close association with ICC in healthy mouse and human GI tract,42 suggesting a possible role for them in the regulation of ICC network organization and integrity. In disease, the expression of proinflammatory markers such as iNOS is associated with ICC network damage, whereas expression of anti-inflammatory markers is associated with preservation of ICC networks in diabetic mice with delayed gastric emptying21 (see later). In disease, although macrophages clearly are activated or polarized in some manner, it is not clear whether unpolarized, resident macrophages also have a role in ICC network maintenance. The interaction of macrophages with neurons is indicated by the variety of neurotransmitter receptors that are present on macrophages that functionally regulate macrophage phenotype including neurokinin receptors, glycine receptors, nicotinic α7 acetylcholine receptors, and P2 purine receptors.43, 44, 45, 46 The converse also is true, neurons express receptors for macrophage-derived signaling molecules that alter neuronal activity and survival including bone morphogenetic protein (BMP)227 and TNFα.47 Nitric oxide produced by iNOS in mouse (but not human) macrophages31 suppresses neuronal48 and smooth muscle excitability.49 Thus, neuronal excitability can be impaired significantly when adjacent macrophages in the muscularis propria are activated, especially in disease; as discussed in the section below on known roles of macrophages in gastrointestinal function and disease.
Muscularis propria macrophages seem to interact preferentially with only specific subtypes of neurons, namely those expressing choline acetyltransferase and neuronal NO.41 Because those subtypes of neurons appear to be reduced with age,50, 51, 52 as well as in particular pathologies affecting GI function, it has been proposed that macrophages play a potential role in the regulation of the enteric nervous system plasticity and development.27 In support of this conclusion, op/op mice that lack muscularis macrophages have more total enteric neurons compared with wild-type littermates,27 although it has been not reported whether this phenomenon affects all neurons or only specific subtypes. It will be important to further elucidate the role of macrophages in pathologies affecting the GI tract and characterized by neuronal loss.53
Reciprocal Interaction With Microbiome
Gut microbiota and their products can affect GI motility by interacting with key cells that regulate GI motility including enteric neurons, glial cells, enterochromaffin cells, and muscularis propria macrophages.27, 54, 55 The ability of different microbes and their products to modulate immune responses in the lamina propria of the gut has been studied extensively.56 Intestinal macrophages play an important role in sensing microbial signals and produce IL1β, required for RAR-related orphan receptor γ-positive innate lymphoid cell–driven granulocyte monocyte colony stimulating factor (aka CSF2) production, which in turn regulate regulatory T-cell numbers.57 In addition to the effect on immune homeostasis, recently the mechanisms by which gut microbiota interact with muscularis propria macrophages and potentially influence gastrointestinal motility have been identified.27 Muscularis macrophages secrete BMP2, which signals constitutively to enteric neurons through the BMP receptor. In turn, enteric neurons contribute to the maintenance of muscularis macrophages through secretion of the macrophage growth factor, CSF1. Antibiotic treatment diminishes the expression of BMP2 and the number of macrophages, as well as signaling via the BMP receptor and expression of CSF1 in neurons, and results in changes in GI transit.27 These results strongly suggest a regulatory role for gut microbiota on the enteric neurons by affecting the muscularis macrophages, however, further work is needed to elucidate the specific signaling pathways by which microbes or their specific products mediate this effect.
Gut microbiota also can influence gastrointestinal motility by signaling the host to increase serotonin (5-hydroxytryptamine [5-HT]) biosynthesis in enterochromaffin cells via fermentative end products such as short-chain fatty acids and bile acids.55, 58 5-HT is a neurotransmitter that activates a large variety of receptors on cells in the muscularis propria and plays an important role in gastrointestinal motility. Macrophages as a target of serotonin have not been explored in the GI tract but human alveolar macrophages express both 5-HT2B and 5-HT7 serotonin receptors and activation of those receptors increases the expression of anti-inflammatory markers and reduces the expression of proinflammatory markers.59 We are not aware of any data indicating expression of serotonin receptors on GI tract macrophages and this serves to emphasize that a detailed transcriptional profiling of subsets of muscularis propria macrophages such as that performed by Martinez et al23 is necessary and would be revealing. Although the earlier-described studies highlight the effect of gut microbiota on intestinal macrophages, future work will shed light on the role of gut microbiota/microbial products on potential cross-talk between macrophages and other key cell types involved in gastrointestinal motility such as enteric glia and ICCs.
Known Roles of Macrophages in Gastrointestinal Function and Disease
Studies on a variety of GI diseases and disorders have indicated that the activation of specific macrophage-mediated pathways in different subsets of cells results in either sustained impairment of tissue function and morphology or activation of potential mechanisms for repair and recovery of function. The extreme heterogeneity of tissue-resident macrophages during homeostasis and inflammation shows that a macrophage often is not just M1 or M2 when residing in a specific tissue,29, 60, 61 therefore it is important to characterize these signatures, determine the important effector molecules, their mechanism of effect, and the principal cellular targets. Progress has been made by the study of several conditions in which a contribution of the innate immune system is either a part of, or the major mediator of, alterations in function. In this section we discuss those conditions in pathologies affecting the GI tract.
Ileus
Ileus is a prolonged decrease in the rate of movement of intestinal contents that can be induced after sepsis or after abdominal surgery. The bacterial product, lipopolysaccharide, is sufficient to reproduce the symptoms of sepsis-induced ileus including macrophage activation and reduced GI transit and motility.62, 63 Cannon and Murphy64 identified that it is handling of the intestine that results in postoperative ileus, and although modern surgical techniques have reduced the likelihood of the development of severe ileus, it continues to be a complication of intestinal surgery that significantly extends hospital stays for the affected patients and can be fatal.65, 66 The underlying mechanisms for the development of ileus have been studied extensively because of the existence of reliable animal models that recapitulate the pathophysiological changes observed for ileus in human beings. For postoperative ileus, these animal studies have shown that an initial, neuronally mediated inhibition of motility67, 68 and also mast cell degranulation is followed by a late inflammatory phase caused by activation of resident macrophages in the muscularis externa, which release chemoattractant molecules that in turn stimulate the population of the tissue with large numbers of monocyte-derived macrophages.14 TNFα, IL6, MCP-1 CCL2, IL1β, and other cytokines and chemokines produced by these macrophages suppress motility by effects on smooth muscle and both intrinsic and extrinsic nerves.69
Neurons are both the targets and the mediators of the macrophage response in postoperative ileus. Stimulation of the vagal nerve after induction of ileus has been shown to reduce the release of proinflammatory cytokines and, consequently, inflammation as a result of the effect of released acetylcholine on α7 nicotinic receptors expressed on macrophages.70, 71 This effect can be reproduced by using selective α7 nicotinic receptors agonists to treat gastric ileus.72 Because vagal innervation is most dense in the stomach and proximal small intestine,73 vagal nerve activity is most likely to regulate inflammation in those regions. In the distal GI tract, the interaction of other cholinergic nerves with macrophages, including possibly intrinsic enteric motor neurons, is plausible but has not been shown. It also is not clear whether release of anti-inflammatory cytokines from M2-like macrophages are affected by vagal nerve stimulation in the GI tract, and it is also not clear whether all macrophages express α7 nicotinic receptors. This requires further research because it was reported that IL10, a potent cytokine derived from M2-like macrophages, which suppresses expression of inflammatory cytokines by M1-like macrophages,33 is necessary for recovery from postoperative ileus.74 Thus, parallel mechanisms may be exploited for suppression of proinflammatory macrophages and up-regulation of anti-inflammatory macrophages in treatment of ileus. Up-regulation of HO-1 in macrophages of the intestinal muscularis externa is one effective treatment for ileus74 that exploits the potent cytoprotective effects of HO-1 and its active product, carbon monoxide.75 Indeed, inhaled carbon monoxide is effective in the treatment of postoperative ileus in mice.76 Activation of glycine receptors to reduce proinflammatory cytokine production by macrophages and the resulting impairment of smooth muscle contractility is another effective way to reduce the degree of ileus after surgery in rats.77
The animal studies have shown that macrophage activation is central in the cascade of cellular and molecular events that result in ileus, and that modifying macrophage phenotype can ameliorate or treat the symptoms of ileus. Furthermore, the process of studying postoperative ileus in animal models has identified interactions between macrophages and the other cells in the GI tract that are relevant to other gastroenteropathies as well as normal physiology and the development of the GI tract. These observations in animal models should be translatable to treatment of ileus in affected patients, but at present this has not yet been exploited fully.
Inflammatory Bowel Disease
In inflammatory bowel disease (IBD), macrophages and dendritic cells have been identified as homeostatic regulators of mucosal integrity and function by interacting with T cells and responding to T-cell activation.78 Because inflammation in the intestinal mucosa represents one of the defining pathologies of IBD, the mucosa is the focus of research into understanding the causes and opportunities for treatment of these diseases. Genetic risk factors that impair the mucosal barrier and exposure to microbes appear to determine whether inflammation occurs at the mucosal level, but how this results in changes of the macrophage activation in the muscularis, as well as changes in motility, have received limited attention.18
Inflammatory changes in the mucosa are a consequence of activation of T cells with lamina propria dendritic cells and CX3CR1-positive macrophages playing a central role in these responses.79 The effectiveness of TNFα-neutralizing antibodies (infliximab, adalimumab, dolimumab, and so forth) in the treatment of IBD80 indicates that proinflammatory cytokines released from macrophages, such as TNFα, contribute to the disease. In addition, TNFα neutralization induces CD206-positive, regulatory macrophages,81 which can increase T-cell apoptosis82 and contribute to mucosal healing.83 Several powerful anti-inflammatory molecules are generated by regulatory or alternatively activated macrophages including transforming growth factor (TGF)-β and IL10.84, 85 TGF-β signaling is tightly linked to the development of colitis. For example, in patients with IBD, the absence of an anti-inflammatory response to TGF-β was associated with up-regulation of SMAD7, a suppressor of TGF-β signaling.86 IL10 promotes immune tolerance, suppresses Th1 immune responses, and appears to be a central determinant or gatekeeper of the pattern of immune responses.85 The importance of IL10 signaling in the mucosa is indicated clearly by the severe colitis that develops in IL10-receptor knockout mice.87 However, administration of recombinant IL10 to patients with inflammatory bowel disease has not proved effective in treating the disease,88, 89 possibly because IL10 signaling is most important in preventing the initiation of inflammation rather than resolving established responses. In IL10-receptor knockout mice, knockout of Toll-like receptor signaling in myeloid differentiation primary response gene 88–deficient mice prevents colitis,90 which supports the suggestion that IL10 mediates initiation of inflammation in response to Toll-like receptors. Interactions between dendritic cells and T cells also depend on which subtypes of dendritic cells are examined because, similar to macrophages, dendritic cells can show diverse phenotypes. CD103-positive dendritic cells suppress colitis,91 whereas E-cadherin–positive dendritic cells appear to promote inflammation via increased Th17 responses.92
The persistence of GI dysmotility during and after infection or inflammation is a well recognized but incompletely understood phenomenon.93 It may be mediated partly by molecules generated by immune cells. In this respect, persistent symptoms during remission from IBD are similar to postinfectious irritable bowel syndromes in which symptoms persist after an infection and the accompanying macroinflammation has resolved.94, 95 Changes to enteric neurons, glia, ICC, and smooth muscle have been reported,96, 97 as well as effects on extrinsic nerves.98 However, although macrophage activation in the muscularis is known to occur after immune cell infiltration in both type 1 and type 2 immune responses,99 the role of cytokines that are derived from macrophages and persistently up-regulated postinflammation is not well defined. For example, there may be a residual maintenance of changes to cellular function and activity mediated by macrophage-derived factors.
Gastroparesis
Gastroparesis is defined as delayed gastric emptying that is not accompanied by mechanical obstruction. Symptoms usually include early satiety, nausea, vomiting, and abdominal pain.100, 101, 102 Gastroparesis affects people with diabetes, but also can be idiopathic or iatrogenic. Because of the development of animal models of diabetic gastroparesis53, 103 in which delayed gastric emptying developed in a subset of diabetic mice,104 it has been possible to explore the cellular changes in gastroparesis and translate those observations into studies on human tissues. As a result, there is a growing body of evidence that macrophages are involved in the cellular changes that are evident in gastroparesis.20, 21, 105 Changes in CD206+ macrophage populations have been correlated directly with the development of delayed gastric emptying in diabetic NOD mice21 with the expression of HO-1 in those cells appearing to provide a mechanism for protecting ICC when diabetes develops and neuronal NO activity from neurons has been down-regulated.104 In human subjects with diabetic gastroparesis, CD206+ cell numbers in the muscularis propria of the gastric body correlated with ICC numbers,20, 106 and ICC loss is the cellular defect that most strongly associates with delayed gastric emptying.106 Loss of the anti-inflammatory CD206+ cells causes the cellular injury in diabetic gastroparesis because the direct damaging effects of M1-like macrophage cytokines, oxidative stress, high glucose, and insulin resistance were not being negated by the cytoprotective effects of anti-inflammatory cytokines and the HO-1 metabolite carbon monoxide generated by the CD206+ cells.104, 107 However, studies in the op/op, macrophage-depleted mouse also have determined that in the absence of any macrophages, diabetic op/op mice do not develop delayed gastric emptying and ICC networks are preserved.108 Thus, the CD206- macrophages that appear to express iNOS and are abundant in diabetic NOD mice with delayed gastric emptying104 must be up-regulated to result in cellular damage and delayed gastric emptying. The source of these cells and mechanisms for up-regulation require further investigation and the results will need translating into studies in human gastroparesis.
There are few effective treatments for gastroparesis but the evidence that active regulation of macrophage phenotype contributes to the development of delayed gastric emptying in mice and ICC loss in human beings suggests that targeting the polarization of macrophages might be an effective therapy. Indeed, in mice, up-regulating HO-1 in CD206+ gastric muscularis macrophages reverses delayed gastric emptying in diabetic NOD mice104 and delivering the product of HO-1 activity, which is CO, has the same effect.107 IL10 also has shown promise in this animal model by increasing the expression of HO-1 in the gastric body of diabetic NOD mice and returning delayed gastric emptying, electrical slow-wave abnormalities, and ICC network changes to normal.109 This is consistent with the known effects of IL10 in suppressing proinflammatory macrophages and increasing anti-inflammatory macrophages.33 IL10 is known to be safe for use in human beings and could be more effective in treating a disorder such as diabetic gastroparesis, in which macrophage changes appear to be a cause of the damage and functional changes to the tissue as opposed to inflammatory bowel diseases such as colitis in which a large variety of immune responses are causing the disease and IL10 did not produce a clear clinical response.110 The recent demonstration that proinflammatory macrophages are necessary for the development of delayed gastric emptying in diabetic mice108 also suggests that neutralizing the proinflammatory cytokines generated by conventionally activated, M1-like macrophages may be another opportunity for treatment.
In conclusion, macrophages are involved in many processes that control gastrointestinal motility in health and disease. In addition to being the mediators of responses to injury and disease, they appear to have roles in the development and regulation of cells in the healthy GI muscularis propria. This diversity of roles is reflected in many mechanisms for macrophage activation and up-regulation of various phenotypic markers. The responses and phenotypes are tissue- and disease-dependent and further understanding of these intricacies represents opportunities for better understanding of GI motility disorders and potentially treating the disorders.
Footnotes
Conflicts of interest The authors disclose no conflicts.
Funding The work of GC, SJG, PCK, and GF was supported by a National Institutes of Health grant (P01 DK 68055).
Supplementary Material
References
- 1.Hume D.A., Perry V.H., Gordon S. The mononuclear phagocyte system of the mouse defined by immunohistochemical localisation of antigen F4/80: macrophages associated with epithelia. Anat Rec. 1984;210:503–512. doi: 10.1002/ar.1092100311. [DOI] [PubMed] [Google Scholar]
- 2.Lee S.H., Starkey P.M., Gordon S. Quantitative analysis of total macrophage content in adult mouse tissues. Immunochemical studies with monoclonal antibody F4/80. J Exp Med. 1985;161:475–489. doi: 10.1084/jem.161.3.475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bain C.C., Mowat A.M. Macrophages in intestinal homeostasis and inflammation. Immunol Rev. 2014;260:102–117. doi: 10.1111/imr.12192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Smythies L.E., Sellers M., Clements R.H. Human intestinal macrophages display profound inflammatory anergy despite avid phagocytic and bacteriocidal activity. J Clin Invest. 2005;115:66–75. doi: 10.1172/JCI19229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gordon S., Taylor P.R. Monocyte and macrophage heterogeneity. Nat Rev Immunol. 2005;5:953–964. doi: 10.1038/nri1733. [DOI] [PubMed] [Google Scholar]
- 6.Ginhoux F., Greter M., Leboeuf M. Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science. 2010;330:841–845. doi: 10.1126/science.1194637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.van Furth R., Cohn Z.A. The origin and kinetics of mononuclear phagocytes. J Exp Med. 1968;128:415–435. doi: 10.1084/jem.128.3.415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hashimoto D., Chow A., Noizat C. Tissue-resident macrophages self-maintain locally throughout adult life with minimal contribution from circulating monocytes. Immunity. 2013;38:792–804. doi: 10.1016/j.immuni.2013.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ajami B., Bennett J.L., Krieger C. Local self-renewal can sustain CNS microglia maintenance and function throughout adult life. Nat Neurosci. 2007;10:1538–1543. doi: 10.1038/nn2014. [DOI] [PubMed] [Google Scholar]
- 10.Rivollier A., He J., Kole A. Inflammation switches the differentiation program of Ly6Chi monocytes from antiinflammatory macrophages to inflammatory dendritic cells in the colon. J Exp Med. 2012;209:139–155. doi: 10.1084/jem.20101387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bain C.C., Bravo-Blas A., Scott C.L. Constant replenishment from circulating monocytes maintains the macrophage pool in the intestine of adult mice. Nat Immunol. 2014;15:929–937. doi: 10.1038/ni.2967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bain C.C., Scott C.L., Uronen-Hansson H. Resident and pro-inflammatory macrophages in the colon represent alternative context-dependent fates of the same Ly6Chi monocyte precursors. Mucosal Immunol. 2013;6:498–510. doi: 10.1038/mi.2012.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mikkelsen H.B. Macrophages in the external muscle layers of mammalian intestines. Histol Histopathol. 1995;10:719–736. [PubMed] [Google Scholar]
- 14.Kalff J.C., Schwarz N.T., Walgenbach K.J. Leukocytes of the intestinal muscularis: their phenotype and isolation. J Leukoc Biol. 1998;63:683–691. doi: 10.1002/jlb.63.6.683. [DOI] [PubMed] [Google Scholar]
- 15.Mikkelsen H.B., Thuneberg L. Op/op mice defective in production of functional colony-stimulating factor-1 lack macrophages in muscularis externa of the small intestine. Cell Tissue Res. 1999;295:485–493. doi: 10.1007/s004410051254. [DOI] [PubMed] [Google Scholar]
- 16.Mikkelsen H.B., Larsen J.O., Hadberg H. The macrophage system in the intestinal muscularis externa during inflammation: an immunohistochemical and quantitative study of osteopetrotic mice. Histochem Cell Biol. 2008;130:363–373. doi: 10.1007/s00418-008-0423-x. [DOI] [PubMed] [Google Scholar]
- 17.Mikkelsen H.B., Thuneberg L., Rumessen J.J. Macrophage-like cells in the muscularis externa of mouse small intestine. Anat Rec. 1985;213:77–86. doi: 10.1002/ar.1092130111. [DOI] [PubMed] [Google Scholar]
- 18.Hori M., Nobe H., Horiguchi K. MCP-1 targeting inhibits muscularis macrophage recruitment and intestinal smooth muscle dysfunction in colonic inflammation. Am J Physiol Cell Physiol. 2008;294:C391–C401. doi: 10.1152/ajpcell.00056.2007. [DOI] [PubMed] [Google Scholar]
- 19.Linden D.R., Couvrette J.M., Ciolino A. Indiscriminate loss of myenteric neurones in the TNBS-inflamed guinea-pig distal colon. Neurogastroenterol Motil. 2005;17:751–760. doi: 10.1111/j.1365-2982.2005.00703.x. [DOI] [PubMed] [Google Scholar]
- 20.Bernard C.E., Gibbons S.J., Mann I.S. Association of low numbers of CD206-positive cells with loss of ICC in the gastric body of patients with diabetic gastroparesis. Neurogastroenterol Motil. 2014;26:1275–1284. doi: 10.1111/nmo.12389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Choi K.M., Kashyap P.C., Dutta N. CD206-positive M2 macrophages that express heme oxygenase-1 protect against diabetic gastroparesis in mice. Gastroenterology. 2010;138:2399–2409. doi: 10.1053/j.gastro.2010.02.014. 409 e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yoshida H., Hayashi S., Kunisada T. The murine mutation osteopetrosis is in the coding region of the macrophage colony stimulating factor gene. Nature. 1990;345:442–444. doi: 10.1038/345442a0. [DOI] [PubMed] [Google Scholar]
- 23.Martinez F.O., Helming L., Milde R. Genetic programs expressed in resting and IL-4 alternatively activated mouse and human macrophages: similarities and differences. Blood. 2013;121:e57–e69. doi: 10.1182/blood-2012-06-436212. [DOI] [PubMed] [Google Scholar]
- 24.Tamoutounour S., Henri S., Lelouard H. CD64 distinguishes macrophages from dendritic cells in the gut and reveals the Th1-inducing role of mesenteric lymph node macrophages during colitis. Eur J Immunol. 2012;42:3150–3166. doi: 10.1002/eji.201242847. [DOI] [PubMed] [Google Scholar]
- 25.Geissmann F., Jung S., Littman D.R. Blood monocytes consist of two principal subsets with distinct migratory properties. Immunity. 2003;19:71–82. doi: 10.1016/s1074-7613(03)00174-2. [DOI] [PubMed] [Google Scholar]
- 26.Auffray C., Sieweke M.H., Geissmann F. Blood monocytes: development, heterogeneity, and relationship with dendritic cells. Annu Rev Immunol. 2009;27:669–692. doi: 10.1146/annurev.immunol.021908.132557. [DOI] [PubMed] [Google Scholar]
- 27.Muller P.A., Koscso B., Rajani G.M. Crosstalk between muscularis macrophages and enteric neurons regulates gastrointestinal motility. Cell. 2014;158:300–313. doi: 10.1016/j.cell.2014.04.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mills C.D., Kincaid K., Alt J.M. M-1/M-2 macrophages and the Th1/Th2 paradigm. J Immunol. 2000;164:6166–6173. doi: 10.4049/jimmunol.164.12.6166. [DOI] [PubMed] [Google Scholar]
- 29.Martinez F.O., Gordon S. The M1 and M2 paradigm of macrophage activation: time for reassessment. F1000Prime Rep. 2014;6:13. doi: 10.12703/P6-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Murray P.J., Allen J.E., Biswas S.K. Macrophage activation and polarization: nomenclature and experimental guidelines. Immunity. 2014;41:14–20. doi: 10.1016/j.immuni.2014.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gross T.J., Kremens K., Powers L.S. Epigenetic silencing of the human NOS2 gene: rethinking the role of nitric oxide in human macrophage inflammatory responses. J Immunol. 2014;192:2326–2338. doi: 10.4049/jimmunol.1301758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Raes G., Van den Bergh R., De Baetselier P. Arginase-1 and Ym1 are markers for murine, but not human, alternatively activated myeloid cells. J Immunol. 2005;174:6561. doi: 10.4049/jimmunol.174.11.6561. [DOI] [PubMed] [Google Scholar]
- 33.Deng B., Wehling-Henricks M., Villalta S.A. IL-10 triggers changes in macrophage phenotype that promote muscle growth and regeneration. J Immunol. 2012;189:3669–3680. doi: 10.4049/jimmunol.1103180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lee T.S., Chau L.Y. Heme oxygenase-1 mediates the anti-inflammatory effect of interleukin-10 in mice. Nat Med. 2002;8:240–246. doi: 10.1038/nm0302-240. [DOI] [PubMed] [Google Scholar]
- 35.Grainger J.R., Wohlfert E.A., Fuss I.J. Inflammatory monocytes regulate pathologic responses to commensals during acute gastrointestinal infection. Nat Med. 2013;19:713–721. doi: 10.1038/nm.3189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zigmond E., Varol C., Farache J. Ly6C hi monocytes in the inflamed colon give rise to proinflammatory effector cells and migratory antigen-presenting cells. Immunity. 2012;37:1076–1090. doi: 10.1016/j.immuni.2012.08.026. [DOI] [PubMed] [Google Scholar]
- 37.Kurihara T., Warr G., Loy J. Defects in macrophage recruitment and host defense in mice lacking the CCR2 chemokine receptor. J Exp Med. 1997;186:1757–1762. doi: 10.1084/jem.186.10.1757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Boring L., Gosling J., Cleary M. Decreased lesion formation in CCR2-/- mice reveals a role for chemokines in the initiation of atherosclerosis. Nature. 1998;394:894–897. doi: 10.1038/29788. [DOI] [PubMed] [Google Scholar]
- 39.Kanda H., Tateya S., Tamori Y. MCP-1 contributes to macrophage infiltration into adipose tissue, insulin resistance, and hepatic steatosis in obesity. J Clin Invest. 2006;116:1494–1505. doi: 10.1172/JCI26498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Platt A.M., Bain C.C., Bordon Y. An independent subset of TLR expressing CCR2-dependent macrophages promotes colonic inflammation. J Immunol. 2010;184:6843–6854. doi: 10.4049/jimmunol.0903987. [DOI] [PubMed] [Google Scholar]
- 41.Phillips R.J., Powley T.L. Macrophages associated with the intrinsic and extrinsic autonomic innervation of the rat gastrointestinal tract. Auton Neurosci. 2012;169:12–27. doi: 10.1016/j.autneu.2012.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mikkelsen H.B. Interstitial cells of Cajal, macrophages and mast cells in the gut musculature: morphology, distribution, spatial and possible functional interactions. J Cell Mol Med. 2010;14:818–832. doi: 10.1111/j.1582-4934.2010.01025.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Nemethova A., Michel K., Gomez-Pinilla P.J. Nicotine attenuates activation of tissue resident macrophages in the mouse stomach through the beta2 nicotinic acetylcholine receptor. PLoS One. 2013;8:e79264. doi: 10.1371/journal.pone.0079264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ho W.Z., Lai J.P., Zhu X.H. Human monocytes and macrophages express substance P and neurokinin-1 receptor. J Immunol. 1997;159:5654–5660. [PubMed] [Google Scholar]
- 45.Froh M., Thurman R.G., Wheeler M.D. Molecular evidence for a glycine-gated chloride channel in macrophages and leukocytes. Am J Physiol Gastrointest Liver Physiol. 2002;283:G856–G863. doi: 10.1152/ajpgi.00503.2001. [DOI] [PubMed] [Google Scholar]
- 46.Marques-da-Silva C., Burnstock G., Ojcius D.M. Purinergic receptor agonists modulate phagocytosis and clearance of apoptotic cells in macrophages. Immunobiology. 2011;216:1–11. doi: 10.1016/j.imbio.2010.03.010. [DOI] [PubMed] [Google Scholar]
- 47.Coquenlorge S., Duchalais E., Chevalier J. Modulation of lipopolysaccharide-induced neuronal response by activation of the enteric nervous system. J Neuroinflammation. 2014;11:202. doi: 10.1186/s12974-014-0202-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Green C.L., Ho W., Sharkey K.A. Dextran sodium sulfate-induced colitis reveals nicotinic modulation of ion transport via iNOS-derived NO. Am J Physiol Gastrointest Liver Physiol. 2004;287:G706–G714. doi: 10.1152/ajpgi.00076.2004. [DOI] [PubMed] [Google Scholar]
- 49.Hori M., Kita M., Torihashi S. Upregulation of iNOS by COX-2 in muscularis resident macrophage of rat intestine stimulated with LPS. Am J Physiol Gastrointest Liver Physiol. 2001;280:G930–G938. doi: 10.1152/ajpgi.2001.280.5.G930. [DOI] [PubMed] [Google Scholar]
- 50.Cowen T., Johnson R.J., Soubeyre V. Restricted diet rescues rat enteric motor neurones from age related cell death. Gut. 2000;47:653–660. doi: 10.1136/gut.47.5.653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wu M., Van Nassauw L., Kroese A.B. Myenteric nitrergic neurons along the rat esophagus: evidence for regional and strain differences in age-related changes. Histochem Cell Biol. 2003;119:395–403. doi: 10.1007/s00418-003-0526-3. [DOI] [PubMed] [Google Scholar]
- 52.Takahashi T., Qoubaitary A., Owyang C. Decreased expression of nitric oxide synthase in the colonic myenteric plexus of aged rats. Brain Res. 2000;883:15–21. doi: 10.1016/s0006-8993(00)02867-5. [DOI] [PubMed] [Google Scholar]
- 53.Watkins C.C., Sawa A., Jaffrey S. Insulin restores neuronal nitric oxide synthase expression and function that is lost in diabetic gastropathy. J Clin Invest. 2000;106:803. doi: 10.1172/jci8273c1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kabouridis P.S., Lasrado R., McCallum S. Microbiota controls the homeostasis of glial cells in the gut lamina propria. Neuron. 2015;85:289–295. doi: 10.1016/j.neuron.2014.12.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Reigstad C.S., Salmonson C.E., Rainey J.F., 3rd Gut microbes promote colonic serotonin production through an effect of short-chain fatty acids on enterochromaffin cells. FASEB J. 2015;29:1395–1403. doi: 10.1096/fj.14-259598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Round J.L., Mazmanian S.K. The gut microbiota shapes intestinal immune responses during health and disease. Nat Rev Immunol. 2009;9:313–323. doi: 10.1038/nri2515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Mortha A., Chudnovskiy A., Hashimoto D. Microbiota-dependent crosstalk between macrophages and ILC3 promotes intestinal homeostasis. Science. 2014;343:1249288. doi: 10.1126/science.1249288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Yano J.M., Yu K., Donaldson G.P. Indigenous bacteria from the gut microbiota regulate host serotonin biosynthesis. Cell. 2015;161:264–276. doi: 10.1016/j.cell.2015.02.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.de las Casas-Engel M., Dominguez-Soto A., Sierra-Filardi E. Serotonin skews human macrophage polarization through HTR2B and HTR7. J Immunol. 2013;190:2301–2310. doi: 10.4049/jimmunol.1201133. [DOI] [PubMed] [Google Scholar]
- 60.Davies L.C., Jenkins S.J., Allen J.E. Tissue-resident macrophages. Nat Immunol. 2013;14:986–995. doi: 10.1038/ni.2705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Xue J., Schmidt S.V., Sander J. Transcriptome-based network analysis reveals a spectrum model of human macrophage activation. Immunity. 2014;40:274–288. doi: 10.1016/j.immuni.2014.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Eskandari M.K., Kalff J.C., Billiar T.R. LPS-induced muscularis macrophage nitric oxide suppresses rat jejunal circular muscle activity. Am J Physiol. 1999;277:G478–G486. doi: 10.1152/ajpgi.1999.277.2.G478. [DOI] [PubMed] [Google Scholar]
- 63.Lodato R.F., Khan A.R., Zembowicz M.J. Roles of IL-1 and TNF in the decreased ileal muscle contractility induced by lipopolysaccharide. Am J Physiol. 1999;276:G1356–G1362. doi: 10.1152/ajpgi.1999.276.6.G1356. [DOI] [PubMed] [Google Scholar]
- 64.Cannon W.B., Murphy F.T. IV. The movements of the stomach and intestines in some surgical conditions. Ann Surg. 1906;43:512–536. doi: 10.1097/00000658-190604000-00004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Bauer A.J., Schwarz N.T., Moore B.A. Ileus in critical illness: mechanisms and management. Curr Opin Crit Care. 2002;8:152–157. doi: 10.1097/00075198-200204000-00011. [DOI] [PubMed] [Google Scholar]
- 66.Boeckxstaens G.E., de Jonge W.J. Neuroimmune mechanisms in postoperative ileus. Gut. 2009;58:1300–1311. doi: 10.1136/gut.2008.169250. [DOI] [PubMed] [Google Scholar]
- 67.Bueno L., Fioramonti J., Ruckebusch Y. Postoperative intestinal motility in dogs and sheep. Am J Dig Dis. 1978;23:682–689. doi: 10.1007/BF01072352. [DOI] [PubMed] [Google Scholar]
- 68.Tache Y., Yoneda M. Central action of TRH to induce vagally mediated gastric cytoprotection and ulcer formation in rats. J Clin Gastroenterol. 1993;17(Suppl 1):S58–S63. doi: 10.1097/00004836-199312001-00013. [DOI] [PubMed] [Google Scholar]
- 69.Turler A., Schwarz N.T., Turler E. MCP-1 causes leukocyte recruitment and subsequently endotoxemic ileus in rat. Am J Physiol Gastrointest Liver Physiol. 2002;282:G145–G155. doi: 10.1152/ajpgi.00263.2001. [DOI] [PubMed] [Google Scholar]
- 70.Tracey K.J. Physiology and immunology of the cholinergic antiinflammatory pathway. J Clin Invest. 2007;117:289–296. doi: 10.1172/JCI30555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Matteoli G., Gomez-Pinilla P.J., Nemethova A. A distinct vagal anti-inflammatory pathway modulates intestinal muscularis resident macrophages independent of the spleen. Gut. 2014;63:938–948. doi: 10.1136/gutjnl-2013-304676. [DOI] [PubMed] [Google Scholar]
- 72.The F.O., Boeckxstaens G.E., Snoek S.A. Activation of the cholinergic anti-inflammatory pathway ameliorates postoperative ileus in mice. Gastroenterology. 2007;133:1219–1228. doi: 10.1053/j.gastro.2007.07.022. [DOI] [PubMed] [Google Scholar]
- 73.Berthoud H.R., Carlson N.R., Powley T.L. Topography of efferent vagal innervation of the rat gastrointestinal tract. Am J Physiol. 1991;260:R200–R207. doi: 10.1152/ajpregu.1991.260.1.R200. [DOI] [PubMed] [Google Scholar]
- 74.Stoffels B., Schmidt J., Nakao A. Role of interleukin 10 in murine postoperative ileus. Gut. 2009;58:648–660. doi: 10.1136/gut.2008.153288. [DOI] [PubMed] [Google Scholar]
- 75.Motterlini R., Otterbein L.E. The therapeutic potential of carbon monoxide. Nat Rev Drug Discov. 2010;9:728–743. doi: 10.1038/nrd3228. [DOI] [PubMed] [Google Scholar]
- 76.Moore B.A., Otterbein L.E., Turler A. Inhaled carbon monoxide suppresses the development of postoperative ileus in the murine small intestine. Gastroenterology. 2003;124:377–391. doi: 10.1053/gast.2003.50060. [DOI] [PubMed] [Google Scholar]
- 77.Stoffels B., Turler A., Schmidt J. Anti-inflammatory role of glycine in reducing rodent postoperative inflammatory ileus. Neurogastroenterol Motil. 2011;23:76–87. doi: 10.1111/j.1365-2982.2010.01603.x. e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.MacDonald T.T., Monteleone I., Fantini M.C. Regulation of homeostasis and inflammation in the intestine. Gastroenterology. 2011;140:1768–1775. doi: 10.1053/j.gastro.2011.02.047. [DOI] [PubMed] [Google Scholar]
- 79.Cader M.Z., Kaser A. Recent advances in inflammatory bowel disease: mucosal immune cells in intestinal inflammation. Gut. 2013;62:1653–1664. doi: 10.1136/gutjnl-2012-303955. [DOI] [PubMed] [Google Scholar]
- 80.Rutgeerts P., Vermeire S., Van Assche G. Biological therapies for inflammatory bowel diseases. Gastroenterology. 2009;136:1182–1197. doi: 10.1053/j.gastro.2009.02.001. [DOI] [PubMed] [Google Scholar]
- 81.Vos A.C., Wildenberg M.E., Duijvestein M. Anti-tumor necrosis factor-alpha antibodies induce regulatory macrophages in an Fc region-dependent manner. Gastroenterology. 2011;140:221–230. doi: 10.1053/j.gastro.2010.10.008. [DOI] [PubMed] [Google Scholar]
- 82.Van den Brande J.M., Braat H., van den Brink G.R. Infliximab but not etanercept induces apoptosis in lamina propria T-lymphocytes from patients with Crohn's disease. Gastroenterology. 2003;124:1774–1785. doi: 10.1016/s0016-5085(03)00382-2. [DOI] [PubMed] [Google Scholar]
- 83.Vos A.C., Wildenberg M.E., Arijs I. Regulatory macrophages induced by infliximab are involved in healing in vivo and in vitro. Inflamm Bowel Dis. 2012;18:401–408. doi: 10.1002/ibd.21818. [DOI] [PubMed] [Google Scholar]
- 84.Maloy K.J., Powrie F. Intestinal homeostasis and its breakdown in inflammatory bowel disease. Nature. 2011;474:298–306. doi: 10.1038/nature10208. [DOI] [PubMed] [Google Scholar]
- 85.Li M.O., Flavell R.A. Contextual regulation of inflammation: a duet by transforming growth factor-beta and interleukin-10. Immunity. 2008;28:468–476. doi: 10.1016/j.immuni.2008.03.003. [DOI] [PubMed] [Google Scholar]
- 86.Fantini M.C., Rizzo A., Fina D. Smad7 controls resistance of colitogenic T cells to regulatory T cell-mediated suppression. Gastroenterology. 2009;136:1308–1316. doi: 10.1053/j.gastro.2008.12.053. e1-3. [DOI] [PubMed] [Google Scholar]
- 87.Kuhn R., Lohler J., Rennick D. Interleukin-10-deficient mice develop chronic enterocolitis. Cell. 1993;75:263–274. doi: 10.1016/0092-8674(93)80068-p. [DOI] [PubMed] [Google Scholar]
- 88.Huhn R.D., Radwanski E., Gallo J. Pharmacodynamics of subcutaneous recombinant human interleukin-10 in healthy volunteers. Clin Pharmacol Ther. 1997;62:171–180. doi: 10.1016/S0009-9236(97)90065-5. [DOI] [PubMed] [Google Scholar]
- 89.Moore K.W., de Waal Malefyt R., Coffman R.L. Interleukin-10 and the interleukin-10 receptor. Annu Rev Immunol. 2001;19:683–765. doi: 10.1146/annurev.immunol.19.1.683. [DOI] [PubMed] [Google Scholar]
- 90.Rakoff-Nahoum S., Hao L., Medzhitov R. Role of toll-like receptors in spontaneous commensal-dependent colitis. Immunity. 2006;25:319–329. doi: 10.1016/j.immuni.2006.06.010. [DOI] [PubMed] [Google Scholar]
- 91.Hammer G.E., Turer E.E., Taylor K.E. Expression of A20 by dendritic cells preserves immune homeostasis and prevents colitis and spondyloarthritis. Nat Immunol. 2011;12:1184–1193. doi: 10.1038/ni.2135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Siddiqui K.R., Laffont S., Powrie F. E-cadherin marks a subset of inflammatory dendritic cells that promote T cell-mediated colitis. Immunity. 2010;32:557–567. doi: 10.1016/j.immuni.2010.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Brierley S.M., Linden D.R. Neuroplasticity and dysfunction after gastrointestinal inflammation. Nat Rev Gastroenterol Hepatol. 2014;11:611–627. doi: 10.1038/nrgastro.2014.103. [DOI] [PubMed] [Google Scholar]
- 94.Crosthwaite A.I., Huizinga J.D., Fox J.A. Jejunal circular muscle motility is decreased in nematode-infected rat. Gastroenterology. 1990;98:59–65. doi: 10.1016/0016-5085(90)91291-d. [DOI] [PubMed] [Google Scholar]
- 95.Hosseini J.M., Goldhill J.M., Bossone C. Progressive alterations in circular smooth muscle contractility in TNBS-induced colitis in rats. Neurogastroenterol Motil. 1999;11:347–356. doi: 10.1046/j.1365-2982.1999.00165.x. [DOI] [PubMed] [Google Scholar]
- 96.Bercik P., Wang L., Verdu E.F. Visceral hyperalgesia and intestinal dysmotility in a mouse model of postinfective gut dysfunction. Gastroenterology. 2004;127:179–187. doi: 10.1053/j.gastro.2004.04.006. [DOI] [PubMed] [Google Scholar]
- 97.Shea-Donohue T., Notari L., Sun R. Mechanisms of smooth muscle responses to inflammation. Neurogastroenterol Motil. 2012;24:802–811. doi: 10.1111/j.1365-2982.2012.01986.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Coldwell J.R., Phillis B.D., Sutherland K. Increased responsiveness of rat colonic splanchnic afferents to 5-HT after inflammation and recovery. J Physiol. 2007;579:203–213. doi: 10.1113/jphysiol.2006.123158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Zhao A., Urban J.F., Jr., Anthony R.M. Th2 cytokine-induced alterations in intestinal smooth muscle function depend on alternatively activated macrophages. Gastroenterology. 2008;135:217–225 e1. doi: 10.1053/j.gastro.2008.03.077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Parkman H.P., Yates K., Hasler W.L. Similarities and differences between diabetic and idiopathic gastroparesis. Clin Gastroenterol Hepatol. 2011;9:1056–1064. doi: 10.1016/j.cgh.2011.08.013. quiz e133–e134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Hasler W.L., Wilson L.A., Parkman H.P. Factors related to abdominal pain in gastroparesis: contrast to patients with predominant nausea and vomiting. Neurogastroenterol Motil. 2013;25:427–438. doi: 10.1111/nmo.12091. e300–e301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Parkman H.P., Camilleri M., Farrugia G. Gastroparesis and functional dyspepsia: excerpts from the AGA/ANMS meeting. Neurogastroenterol Motil. 2010;22:113–133. doi: 10.1111/j.1365-2982.2009.01434.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Ordog T., Takayama I., Cheung W.K. Remodeling of networks of interstitial cells of Cajal in a murine model of diabetic gastroparesis. Diabetes. 2000;49:1731–1739. doi: 10.2337/diabetes.49.10.1731. [DOI] [PubMed] [Google Scholar]
- 104.Choi K.M., Gibbons S.J., Nguyen T.V. Heme oxygenase-1 protects interstitial cells of Cajal from oxidative stress and reverses diabetic gastroparesis. Gastroenterology. 2008;135:2055–2064. doi: 10.1053/j.gastro.2008.09.003. 64 e1–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Neshatian L., Gibbons S.J., Farrugia G. Macrophages in diabetic gastroparesis–the missing link? Neurogastroenterol Motil. 2015;27:7–18. doi: 10.1111/nmo.12418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Grover M., Farrugia G., Lurken M.S. Cellular changes in diabetic and idiopathic gastroparesis. Gastroenterology. 2011;140:1575–1585 e8. doi: 10.1053/j.gastro.2011.01.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Kashyap P.C., Choi K.M., Dutta N. Carbon monoxide reverses diabetic gastroparesis in NOD mice. Am J Physiol Gastrointest Liver Physiol. 2010;298:G1013–G1019. doi: 10.1152/ajpgi.00069.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Cipriani G., Gibbons S.J., Verhulst P.-J. Diabetic Csf1op/op mice lacking macrophages are protected against the development of delayed gastric emptying. Cell Mol Gastroenterol Hepatol. 2016;2:40–47. doi: 10.1016/j.jcmgh.2015.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Choi K.M., Sha L., Verhulst P.-J. Treatment with IL-10 reverses gastroparesis in diabetic NOD/Shiltj mice. Gastroenterology. 2012;142:S-66. [Google Scholar]
- 110.Asadullah K., Sterry W., Volk H.D. Interleukin-10 therapy–review of a new approach. Pharmacol Rev. 2003;55:241–269. doi: 10.1124/pr.55.2.4. [DOI] [PubMed] [Google Scholar]