Abstract
The lateral habenula (LHb) is bilaterally connected with serotoninergic raphe nuclei, and expresses high density of serotonin receptors. However, actions of serotonin on the excitatory synaptic transmission to LHb neurons have not been thoroughly investigated. The LHb contains two anatomically and functionally distinct regions: lateral (LHbl) and medial (LHbm) divisions. We compared serotonin’s effects on glutamatergic transmission across the LHb in rat brains. Serotonin bi-directionally and differentially modulated glutamatergic transmission. Serotonin inhibited glutamatergic transmission in higher percentage of LHbl neurons but potentiated in higher percentage of LHbm neurons. Magnitude of potentiation was greater in LHbm than in LHbl. Type 2 and 3 serotonin receptor antagonists attenuated serotonin’s potentiation. The serotonin reuptake blocker, and the type 2 and 3 receptor agonists facilitated glutamatergic transmission in both LHbl and LHbm neurons. Thus, serotonin via activating its type 2, 3 receptors, increased glutamate release at nerve terminals in some LHb neurons. Our data demonstrated that serotonin affects both LHbm and LHbl. Serotonin might play an important role in processing information between the LHb and its downstream-targeted structures during decision-making. It may also contribute to a homeostatic balance underlying the neural circuitry between the LHb and raphe nuclei.
The brainstem dorsal and medial raphe nuclei, cell groups B7 and B8, respectively1, are the main source of forebrain serotonergic innervation2,3,4. Serotonin (5-HT) exerts differential effects by a variety of receptor subtypes (5-HT1–7). It is implicated in a broad spectrum of functions, including the regulation of appetite, locomotion, stress response, emotional and social behavior, cognition and associative learning, the sleep-wake cycle, reward-related behaviors, and the etiology of psychiatric disorders, such as schizophrenia and major depression5,6.
The lateral habenula (LHb) has emerged as a key brain structure in the pathophysiology of depression7,8,9,10 and decision making11. The LHb is composed of lateral (LHbl) and medial (LHbm) divisions that are anatomically and functionally heterogeneous with different connectivity. The LHbl receives inputs mainly from the basal ganglia12 and sends outputs through the rostromedial tegmental nucleus (RMTg) mainly to the dopaminergic neurons in the ventral tegmental area and substantia nigra and the serotoninergic neurons in the raphe nuclei13,14. The LHbm receives inputs from the limbic areas and sends outputs mainly to the GABAergic interneurons in the raphe nuclei15,16. Hence, changes in the activity of LHb neurons in these subregions may lead to different reactions in their targeted brain areas.
Anatomical evidence has revealed a strong reciprocal innervation between the LHb and the raphe nuclei17,18,19,20, and a high density of 5-HT receptors are expressed in the LHb21,22,23,24,25,26 suggesting a candidate regulatory pathway consists of serotonergic afferents from the raphe nuclei to the LHb. Indeed, there is functional evidence that 5-HT12, as well as activation of 5-HT2C27 and 5-HT1B28 receptors affect glutamate transmission and/or activity of LHb neurons. Moreover, we recently demonstrated that 5-HT induces an excitatory inward current in the LHb. Interestingly, whereas this inward current in LHbl neurons is larger than that in LHbm neurons, 5-HT-induced increase in firing rate is similar in these two subregions29, suggesting that the postsynaptic excitatory effect of 5-HT may be counteracted by its effects on synaptic transmissions. However, actions of 5-HT on the excitatory synaptic transmission to LHb neurons have not been thoroughly investigated.
In this study, we examined the effects of 5-HT on the glutamate transmission in the LHb, and whether differences in the effects of 5-HT existed between the LHbm and LHbl neurons. We also examined the receptor subtypes that mediate 5-HT-induced facilitation of glutamate transmission.
Results
5-HT decreases glutamate transmission in a subset of LHbm and LHbl neurons
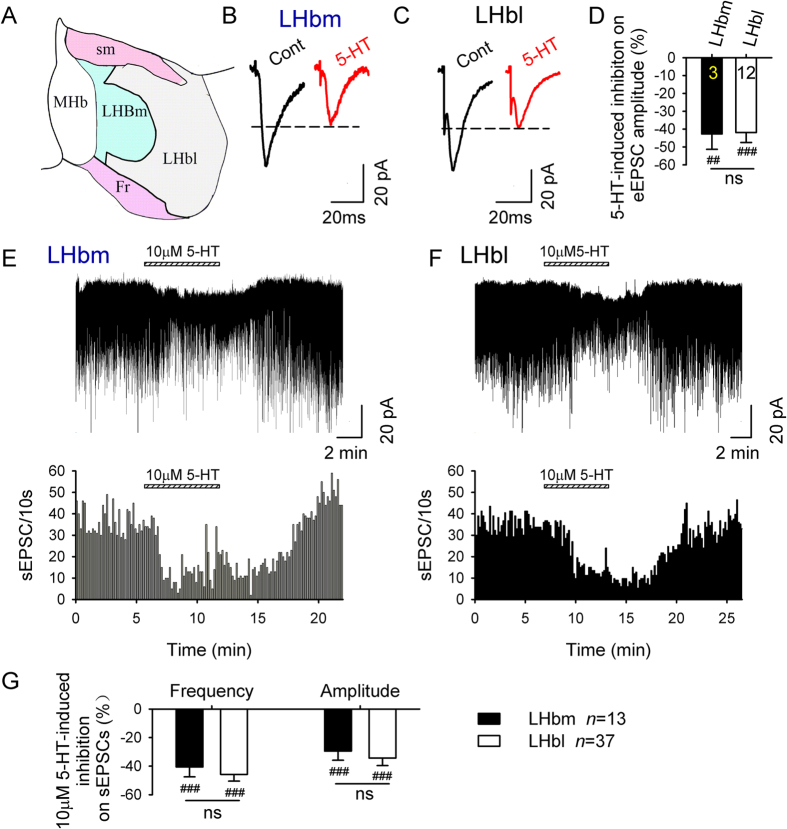
We identified the LHbm and LHbl according to previous reports30,31 (Fig. 1A). To compare the effects of 5-HT on glutamate transmission in LHbm and LHbl neurons, we first examined the effect of 5-HT on eEPSCs evoked by a local electrode. 5-HT (10 μM) markedly suppressed eEPSC amplitude in some neurons of the two subregions (Table 1, Fig. 1B–D). We then examined spontaneous EPSCs (sEPSCs). Neurons in the LHbl and LHbm had similar average basal sEPSC frequencies (LHbm, 1.9 ± 0.2 Hz, n = 141; LHbl, 2.1 ± 0.3 Hz, n = 97; LHbm vs LHbl: t = 0.78, p = 0.43) and amplitudes (LHbm, 16.8 ± 1.1 pA, n = 141; LHbl, 18.1 ± 1.2 pA, n = 97; LHbm vs LHbl: t = 0.8, p = 0.45). 5-HT inhibited sEPSCs in some neurons in these two subregions (Table 1, Fig. 1E–G).
Figure 1. 5-HT reduces glutamate transmission in some LHbm and LHbl neurons.
(A) A scheme of the delineation of individual LHb subnuclei (modified from Fig. 3E30). Bath application of 10 μM 5-HT substantially suppressed EPSCs evoked by electrical stimulation in an LHbm (B) and an LHbl (C) neurons. Data are averages of 10 traces. (D) Summary of 10 μM 5-HT-induced inhibition on eEPSC amplitude in LHbm and LHbl neurons. 10 μM 5-HT greatly reduced sEPSCs in an LHbm (E) and an LHbl (F) cells. Horizontal bar above the current trace signals the application of the indicated drug. (G) Summary of 5-HT-induced % inhibitions on sEPSC frequency and amplitude. ##p < 0.01, ###p < 0.001, Student’s paired t-test for 5-HT vs baseline. ns means no significant difference between LHbm and LHbl neurons, unpaired t-test. Numbers of cells are indicated.
Table 1. 5-HT (10 μM) induced changes in glutamatergic transmission in the LHbm and LHbl neurons.
| Cases Distribution | p-Value (LHbm vs LHbl) | % Changes | p-Value (LHbm vs LHbl) | ||||
|---|---|---|---|---|---|---|---|
| EPSC | LHbm | LHbl | LHbm | LHbl | |||
| sEPSC frequency | ↓ | 13/67 | 37/70 | p < 0.001 | 40.6 ± 6.9### | 45.8 ± 4.7### | ns |
| ↑ | 50/67 | 27/70 | 153.4 ± 14.6### | 108.2 ± 13.5## | p = 0.029 | ||
| — | 4/67 | 6/70 | — | — | — | ||
| sEPSC amplitude | ↓ | 13/67 | 37/70 | p < 0.001 | 29.5 ± 6.4### | 34.4 ± 5.2### | ns |
| ↑ | 50/67 | 27/70 | 13.4 ± 1.9# | 12.5 ± 2.3 | ns | ||
| eEPSC amplitude | ↓ | 3/21 | 12/24 | p = 0.025 | 42.7 ± 8.6## | 41.9 ± 5.6### | ns |
| ↑ | 17/21 | 10/24 | 56.7 ± 8.5### | 32.3 ± 4.6### | p = 0.048 | ||
| — | 1/21 | 2/24 | — | — | — | ||
Values are expressed as mean ± SEM., #p < 0.05, ##p < 0.01, ###p < 0.001 vs baseline; ↓decrease; ↑increase; ─ no change. ns means no significant difference.
5-HT facilitates glutamate transmission in many LHb neurons
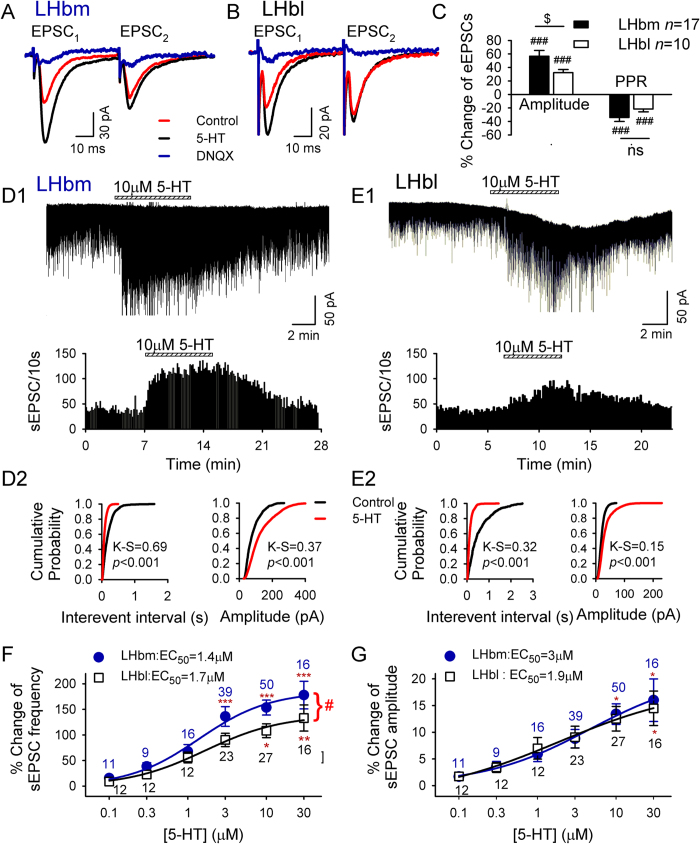
Interestingly, in contrast to the inhibition of EPSCs described above and by others12,28, we found 5-HT increased glutamate release in many LHb neurons, and the potentiation was greater in LHbm than LHbl neurons. Specifically, 10 μM 5-HT induced a significantly greater potentiation in eEPSC amplitude in 17/21 LHbm neurons than in 10/24 LHbl neurons (LHbm vs LHbl: t = 2.1, p = 0.048; Fig. 2A–C). In a similar manner, 10 μM 5-HT induced a significantly greater increase in sEPSC frequency in 50/67 LHbm neurons than in 27/70 LHbl neurons (LHbm vs LHbl: t = 2, p = 0.046; Fig. 2D–F, Table 1). In the remaining LHb neurons, 5-HT did not induce an observable response on eEPSCs (LHbm: n = 1/21; LHbl: n = 2/24; Table 1) or sEPSCs (LHbm: n = 4/67; LHbl: n = 6/70; Table 1).
Figure 2. 5-HT-induced potentiation of glutamate transmission is stronger in LHbm neurons than in LHbl neurons.
Two EPSCs were evoked by twin-pulses (50 msec apart) stimuli, and 5-HT increased the first (EPSC1) but not second (EPSC2) of each pair in LHbm (A) and LHbl (B) cells. 20 μM DNQX abolished the current. (C) Percent change in amplitude and paired-pulse ratio (PPR = EPSC2/EPSC1) of eEPSCs. ###p < 0.001, Student’s paired t-test for 5-HT vs baseline. $p < 0.05, unpaired t-test. (D1–E1) Upper: A representative example of increased sEPSCs by 10 μM 5-HT in an LHbm (D1) or an LHbl (E1) neuron. Lower: Time course of 5-HT’s effect on sEPSC frequency. Cumulative probability plots show higher incidence of shorter inter-sEPSC intervals and larger sEPSCs during 5-HT applications (red line) in the LHbm (D2) and LHbl (E2) neurons. Concentration-dependent increases in sEPSC frequency (F) and amplitude (G) in LHbm (•) and LHbl (▫) neurons. The smooth curve is the best fit to the data by the logistic equation. *p < 0.05, **p < 0.01, ***p < 0.001, two-way ANOVA followed by Tukey’s multiple comparisons test. #p < 0.05 between LHbm and LHbl.
Moreover, 5-HT potentiated EPSCs in the majority of the LHbm neurons (eEPSCs: n = 17/21; sEPSCs: n = 50/67), but in less than half of the LHbl neurons (eEPSCs: n = 10/24; sEPSCs: n = 27/70). The difference was significant (eEPSCs: Chi-square = 7.4 with df 2, p = 0.025; sEPSCs: Chi-square = 18.7 with df 2, p < 0.001). The deferential modulation of glutamate transmission by 5-HT (10 μΜ) in the LHbm and LHbl is summarized in Table 1. Notably, 5-HT inhibited EPSCs in significantly higher percentage of LHbl neurons than LHbm neurons (p < 0.001). By contrast, 5-HT potentiated EPSCs in significantly higher percentage of LHbm neurons than LHbl neurons (p < 0.001). Since our data of 5-HT inhibition of EPSCs generally agree with previous reports12,28, and there is no previous report on 5-HT potentiation of EPSCs, the present study was designed to investigate the mechanisms involved in the 5-HT-induced potentiation of EPSCs.
5-HT’s enhancement of sEPSCs is greater in LHbm than in LHbl neurons
5-HT significantly increased the frequency and amplitude of sEPSCs in the LHbm and LHbl neurons (Fig. 2D1,E1). This effect, also indicated by the increased incidence of shorter inter-sEPSC intervals (Fig. 2D2,E2 left panels), was reversible by washout; and was significantly stronger in LHbm neurons than in LHbl neurons (main effect of subregions, F1, 231 = 5.2, p = 0.024; Fig. 2F), as revealed by Two way ANOVA. 5-HT’s action was concentration dependent (main effect of doses, F5, 231 = 13.5, p < 0.001), with the EC50s of 1.4 ± 0.4 μM for LHbm neurons and 1.7 ± 0.4 μM for LHbl neurons. Post-hoc tests showed that sEPSC frequencies were significantly increased from baseline following a moderate dose of 5-HT administration (LHbm ≥ 3 μM, LHbl ≥ 10 μM). The increase of sEPSC frequency induced by 3 μM (p = 0.042) or 10 μM (p = 0.029) 5-HT were significantly greater in LHbm neurons than in LHbl neurons (Fig. 2F). However, there was no significant difference on the subregions × doses interaction (F5, 231 = 0.4, p = 0.85).
5-HT-induced increase in sEPSC frequency was accompanied by a higher incidence of larger sEPSCs (K-S test, Fig. 2D2,E2 right panels) in a concentration-dependent manner, with EC50s of 3 ± 1.5 μM in LHbm cells and 1.9 ± 0.9 μM in LHbl cells (F5,231 = 7.4, p < 0.001;Two-way ANOVA; Fig. 2G). No significant difference was detected between these subregions (no effect of subregions, F1, 231 = 0.02, p = 0.9, or subregions × doses interaction, F5, 231 = 0.07, p = 0.99).
5-HT facilitates glutamate transmission via a presynaptic mechanism
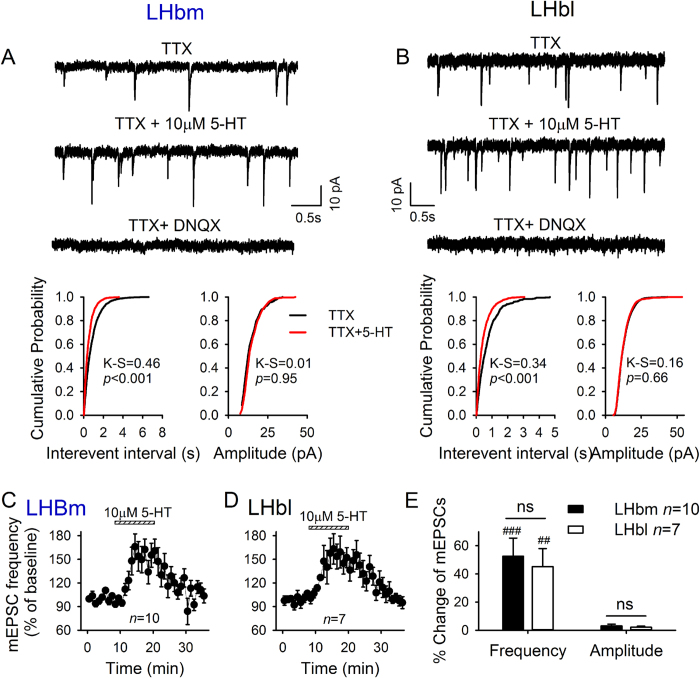
To investigate the mechanisms of 5-HT-induced potentiation of EPSCs, we measured the amplitude of EPSCs evoked by twin pulses of 50 milliseconds apart by a local electrode and the paired pulse ratio (PPR = EPSC2/EPSC1). 5-HT (10 μM) robustly enhanced the amplitude of the first EPSC (EPSC1) of each pair (Fig. 2A,B), but not the second EPSC (EPSC2). Thus, 5-HT significantly reduced the paired pulse ratio (LHbm: 34.1 ± 5.9% decrease relative to baseline, n = 17, p < 0.001; LHbl: 21.6 ± 4.1%, n = 10, p < 0.001; LHbm vs LHbl: t = 1.5, p = 0.15; Fig. 2C). These data suggested that 5-HT enhanced presynaptic glutamate release. This idea was further supported by the data of miniature EPSCs (mEPSCs) recorded in the presence of tetrodotoxin. 5-HT (10 μM) shifted the cumulative interevent interval distribution towards shorter intervals (p < 0.05, K-S test; Fig. 3A,B), thus significantly increasing the mean frequency (LHbm: p < 0.001, n = 10/12; LHbl: p < 0.01, n = 7/13; LHbm vs LHbl: p > 0.5, unpaired t-test; Fig. 3E). This effect was reversible by washout (Fig. 3C,D). 5-HT had no significant effect on mEPSC amplitude distribution (p > 0.05, K-S test; Fig. 3A,B) nor on mean mEPSC amplitude (both p > 0.05 vs baseline, paired t-test; LHbm vs LHbl: p > 0.5; Fig. 3E).
Figure 3. 5-HT enhances glutamate transmission on LHb neurons by presynaptic mechanisms.
Exemplar current traces showing facilitation of mEPSCs by10 μM 5-HT in a LHbm (A) and a LHbl (B) neuron in the presence of 0.5 μM TTX, 10 μM gabazine, and 1 μM strychnine. 20 μM DNQX completely abolished these mEPSCs. Cumulative probability plots show 5-HT increased incidence of shorter interval, but did not alter the amplitude of mEPSCs from the same neuron. Time course of 5-HT-induced changes in mEPSC frequency in the LHbm (C) and the LHbl (D) subnuclei. (E) Summary of 5-HT-induced changes (%) on mEPSCs.
5-HT2 and 5-HT3 receptors mediate 5-HT’s potentiation of glutamate transmission in LHb neurons
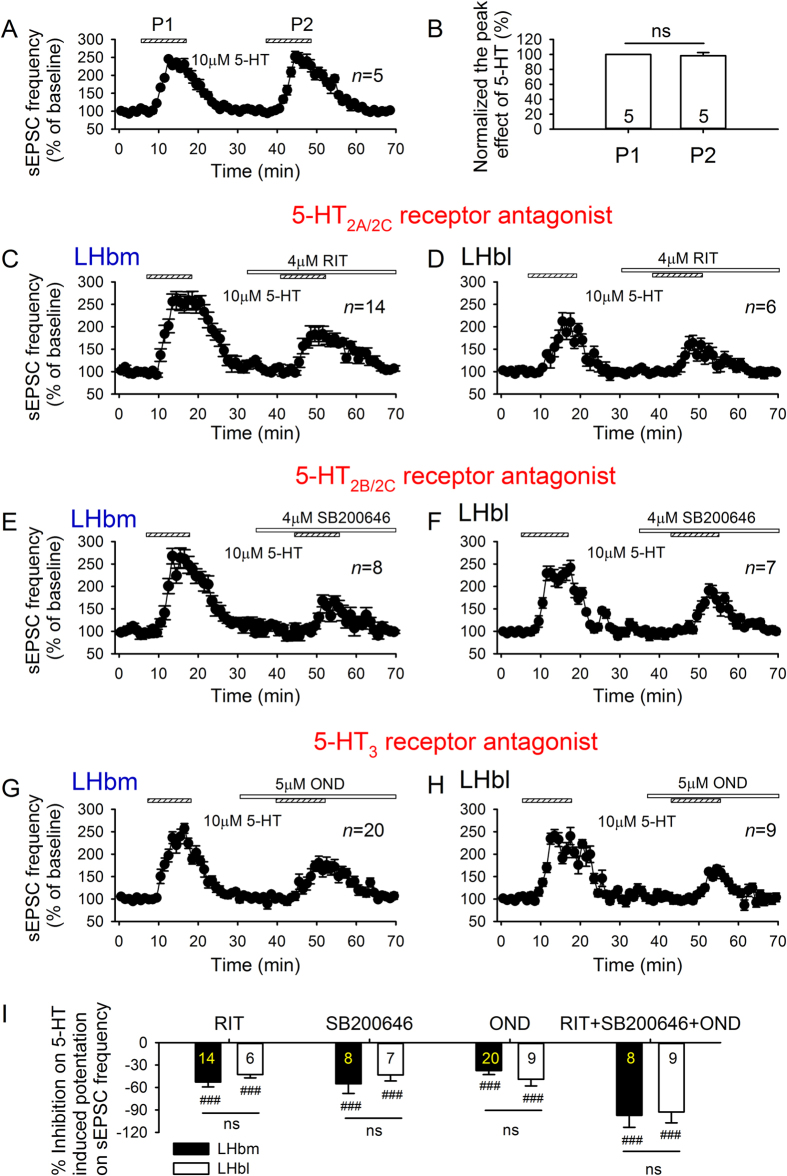
5-HT2 and 5-HT3 receptors mediate 5-HT’s excitatory effects in many brain areas, and histological and molecular evidence indicates that 5-HT226 and 5-HT3 receptors24,25 exist in the LHb. The second application of 5-HT to the same neuron was equally effective in facilitating sEPSC frequency (Fig. 4A,B). Thus, 5-HT produced a reliable and pronounced increase in sEPSC frequency in the majority of LHb neurons. We compared the effects of 5-HT on sEPSC frequency in the absence and presence of antagonists of 5-HT2 and 5-HT3 receptors. After recovery from the facilitation of sEPSCs induced by the first 5-HT application, an antagonist of 5-HT2A/C receptor (10 μM ritanserin (RIT)), 5-HT2B/C receptor (4 μM SB200646), 5-HT3 receptor (5 μM ondansetron (OND)), or a combination of these (RIT + SB200646 + OND) were bath applied for 6–8 min before the second 5-HT application. These antagonists, when applied alone, did not significantly alter the basal sEPSCs (data not shown), but their presence substantially attenuated 5-HT-induced facilitation of sEPSCs (all p < 0.001 vs 5-HT alone, paired t-test; Fig. 4C–H). The attenuation in LHbm was similar to that in LHbl (all p > 0.25, unpaired t-test; Fig. 4I). Notably, 5-HT-induced facilitation was completely eliminated by the combination of 5-HT2 and 5-HT3 antagonists (Fig. 5l), indicating that it was mediated by 5-HT2 and 5-HT3 receptors.
Figure 4. 5-HT2 and 5-HT3 receptor mediate 5-HT-induced increase in sEPSC frequency.
(A,B) Repeated applications of 5-HT (10 μM) induced similar facilitation of sEPSC frequency. Time courses of 5-HT-induced acceleration of sEPSCs in the absence and presence of 5HT2A/2CR antagonist RIT (C,D), 5-HT2B/2CR antagonist SB200646 (E,F), and 5-HT3R antagonist OND (G,H) in LHbm (C,E,G) and LHbl (D,F,H) neurons. (I) Summary of inhibition (%) by RIT, SB200646, OND or the cocktail containing RIT, SB200646 plus OND on the increase of sEPSC frequency induced by10 μM 5-HT in LHbm and LHbl cells. ###p < 0.001 vs 5-HT alone, Student’s paired t-test.
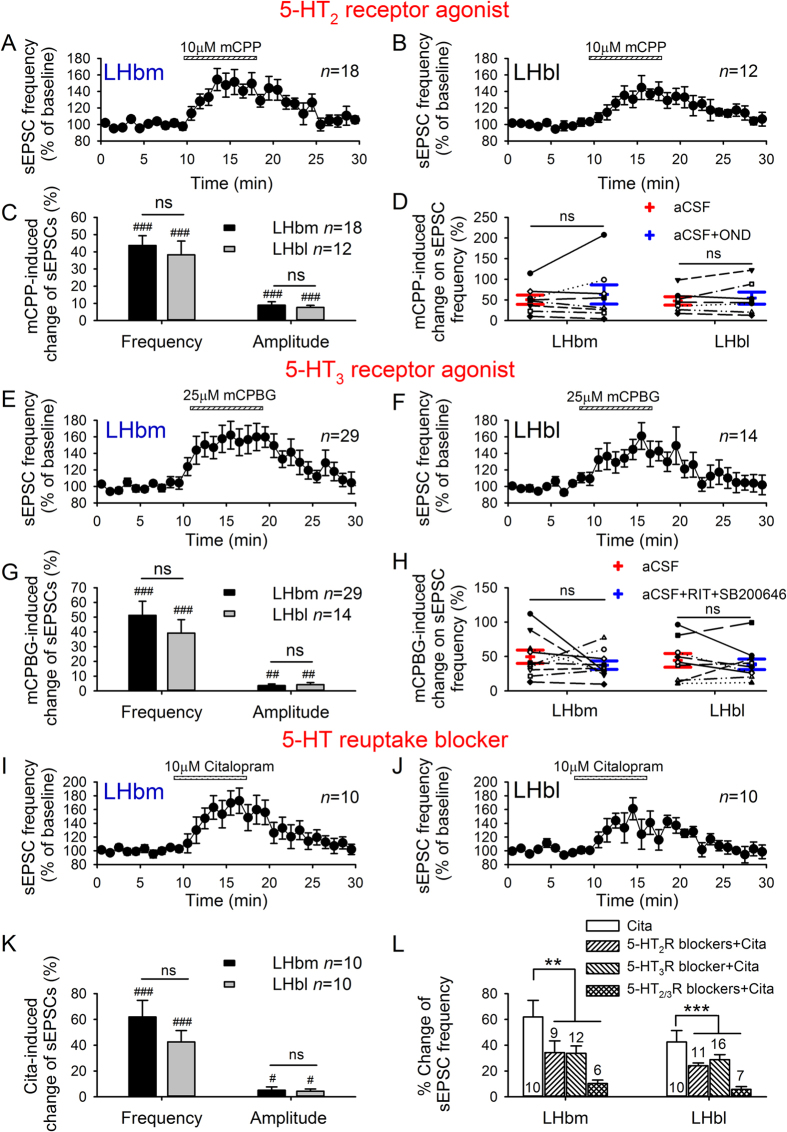
Figure 5. 5-HT2 and 5-HT3 receptor agonists, and 5-HT reuptake blocker potentiate glutamate transmission in the LHb.
Time course of facilitation of sEPSC frequency induced by 5-HT2R agonist (10 μM mCPP) in LHbm (A) and LHbl (B) neurons, and by 5-HT3R agonist (25 μM mCPBG) in LHbm (E) and LHbl (F) neurons. Mean (± SEM) of mCPP (C) or mCPBG (G)-induced increase on sEPSC frequency and amplitude. Mean ± SEM and individual values before and after 5-HT antagonists demonstrated that 5-HT3R antagonist (OND) or 5-HT2R antagonists (RIT plus SB200646) did not alter facilitation of sEPSC frequency induced by mCPP (D) or mCPBG (H). Time course of facilitation of sEPSC frequency induced by the 5-HT transport inhibitor citalopram (10 μM) in LHbm (I) and LHbl (J) neurons. (K) Mean (±SEM) of citalopram-induced increase on sEPSC frequency and amplitude. #p < 0.05, ##p < 0.01, ###p < 0.001 vs baseline, Student’s paired t-test. ns, no significant difference between LHbm and LHbl neurons, unpaired t-test. (L) Mean (±SEM) potentiation of sEPSC frequency induced by citalopram in the absence and presence of 5-HT2 receptor antagonists (RIT plus SB200646), 5-HT3 receptor antagonist (OND) or the cocktail of 5-HT2/3 receptor antagonists. **p < 0.01, ***p < 0.001, One-way ANOVA followed by Tukey’s multiple comparisons test.
5-HT2 and 5-HT3 receptor agonists facilitate glutamate transmission in LHb neurons
We next investigated the effects of the 5-HT2 receptor agonist (10 μM mCPP, preferentially activates 5-HT2C receptors), and the 5-HT3 receptor agonist (25 μM mCPBG), on sEPSCs. mCPP significantly increased the frequency and the amplitude of sEPSCs in all neurons tested in the LHbm (18/18, p < 0.001; Fig. 5A,C) and the majority of LHbl neurons (12/14, t = 4.8, p < 0.001; Fig. 5B,C). 5-HT3 receptor antagonist OND did not alter mCPP-induced increase of sEPSC frequency (LHbm: n = 8, p = 0.38; LHbl: n = 7, p = 0.33; Fig. 5D). Furthermore, mCPBG robustly potentiated sEPSCs in the majority of neurons in the LHbm (n = 30/32, frequency: p < 0.001; amplitude: p < 0.01; Fig. 5E,G) and LHbl (n = 13/15, frequency: p < 0.001; amplitude: p < 0.01; Fig. 5F,G). 5-HT2 antagonists (RIT plus SB200646) did not alter mCPBG-induced facilitation of sEPSC frequency (LHbm: n = 10, p = 0.34; LHbl: n = 9, p = 0.48; Fig. 5H). Notably, the potentiation induced by mCPP or mCPBG was similar in the LHbm and LHbl (all p > 0.5, unpaired t-test; Fig. 5C,G).
5-HT reuptake blocker increases glutamate transmission in LHb neurons
Given that LHb neurons are innervated by serotonergic fibers, we assessed whether endogenous 5-HT could facilitate sEPSCs in LHb neurons. A 10 min bath application of 10 μM citalopram, a highly selective 5-HT reuptake blocker, substantially increased sEPSC frequency in 10/11 LHbm neurons (t = 4.8, p < 0.001; Fig. 5I) and 10/16 LHbl neurons (p < 0.001; Fig. 5J). This effect was accompanied by an increased occurrence of larger sEPSCs (LHbm: by 5.2 ± 2.4%, p = 0.047; LHbl: by 4.3 ± 1.7%, p = 0.018; Fig. 5K). There was no significant difference in citalopram-induced augmentation on sEPSCs between the LHbm and LHbl (frequency: t = 1.24, p = 0.23; amplitude: t = 0.29, p = 0.77; Fig. 5K). In addition, citalopram-induced facilitation of sEPSC frequency in neurons in both the LHbm (F3,33 = 4.6, p = 0.009) and LHbl (F3,40 = 6.8, p < 0.001) was substantially attenuated by the 5-HT2 antagonists (RIT plus SB200646; LHbm: p = 0.038; LHbl: p = 0.032), and the 5-HT3 antagonist (OND; LHbm: p = 0.047; LHbl: p = 0.049), and almost completely abolished by the cocktail (RIT, SB200646 plus OND; LHbm: p = 0.003; LHbl: p < 0.001) (Fig. 5L).
Discussion
We provide here the first electrophysiological evidence that 5-HT bi-directionally and differentially regulates glutamate transmission in the LHb. Furthermore, 5-HT’s facilitation of glutamate transmission is mediated by 5-HT2 and 5-HT3 receptors probably at the glutamatergic terminals. Finally, blockade of 5-HT reuptake facilitates glutamate transmission in most of the LHbm and LHbl neurons, suggesting that 5-HT may regulate glutamate transmission in the LHb under physiological conditions. Via altering glutamate transmission that regulates activity of LHb neurons, 5-HT may in turn alter the activity of raphe nuclei. Thus, 5-HT’s effects in the LHb may provide a feedback loop to the raphe nuclei, and may help maintain homeostatic balance of serotonergic function.
5-HT bi-directionally and differentially modulates glutamate transmission in the LHbm and LHbl neurons. Our result of 5-HT inhibition of glutamate transmission in LHbl neurons generally agreed with previous reports showing that 5-HT suppressed EPSCs in LHbl neurons in slices of rats12, and that 5-HT1B agonist inhibited glutamate transmission in the LHb28. Our finding of 5-HT facilitation of glutamate transmission supports a recent in vivo study showing that intra-LHb injection of selective 5-HT2C agonist Ro60-0175 increased the expression of depressive-like behaviors in rats, suggesting an excitatory effect of 5-HT in the LHb27. We extend this finding by demonstrating that 5-HT facilitated glutamate transmission in the majority of the LHbm neurons and a minority of the LHbl neurons.
A future study is needed to determine whether the different 5-HT effects are due to the difference in the neuronal phenotype. Using morphological analysis Weiss and Veh31 revealed four main categories of projection neurons randomly distributed throughout the LHb. However, electrophysiological characterization of neurons within the different categories demonstrated no significant differences between groups. Based on the pattern of spontaneous activity, neurons were classified as silent, tonic or bursting. The occurrence of distinctive firing modes was not related to topographic allocation. These investigators thus concluded that the formation of functional neuronal entities within the LHb may be achieved through defined synaptic inputs to particular neurons, rather than by individual neuronal morphologies and intrinsic membrane properties. By analyzing the gene expressions of neurotransmitter markers in the habenula, Aizawa, et al.21 showed that neurons in the LHb were almost uniformly glutamatergic. The current study revealed that EPSCs were recorded in almost all LHb neurons tested, and were sensitive to 5-HT. Thus, the different responses to 5-HT may be resulted from the heterogeneous expression of 5-HT receptors on the glutamatergic terminals in LHb neurons.
In the current study, we identified presynaptic 5-HT2A/B/C and 5-HT3 receptors mediate 5-HT-induced facilitation of glutamate transmission. 5-HT potentiated the frequency and amplitude of sEPSCs. Tetrodotoxin completely abolished the effect on sEPSC amplitude, indicating that 5-HT’s effect on sEPSC amplitude depends on the action potentials. Action potential firing may increase the proportion of multiquantal events, thus skewing the amplitude distribution of synaptic currents towards larger size classes. 5-HT significantly increased the frequency but not the amplitude of mEPSCs, suggesting that 5-HT increases the probability of glutamate release, probably at the nerve terminals. In support, 5-HT increased the amplitude, and decreased the paired pulse ratio of the eEPSCs.
RNA labeling evidence indicates the existence of 5-HT2 receptors in the LHb32. Pharmacological evidence has confirmed the existence of 5-HT1B28 and 5-HT2C27 receptors in rat LHb. In both the LHbl and LHbm, we showed that 5-HT2A/C antagonist ritanserin and 5-HT2B/C antagonist SB200646 substantially attenuated 5-HT-induced facilitation of sEPSCs. Conversely, the 5-HT2C agonist mCPP increased sEPSCs, in general agreement with a recent in vivo study27. Our data suggest that activation of 5-HT2 receptors increases glutamate release probability at the nerve terminals. The existence of 5-HT3 receptors was revealed by the application of ondansetron, which significantly attenuated 5-HT-induced facilitation of sEPSCs. Accordingly, bath application of the selective 5-HT3 agonist mCPBG increased sEPSCs. These data suggest that activation of 5-HT3 receptors increases glutamate release probability and partly mediates 5-HT’s facilitation of glutamate transmission in the LHb. Notably, the cocktail containing 5-HT2 and 5-HT3 receptor antagonists completely abolished 5-HT-induced potentiation of glutamate transmission. Our results thus revealed the presence of functional 5-HT2A/B/C and 5-HT3 receptors that mediate 5-HT-induced facilitation of glutamate transmission in the LHb.
5-HT may modulate glutamate transmission in the LHb under physiological conditions. There is recent evidence33 that the 5-HT reuptake blocker citalopram affected the activity of the synapse connecting the basal ganglia with the LHb neuron. In general agreement with their finding, we found that citalopram potentiated glutamate transmission in LHb neurons, and the antagonists of 5-HT2A/C, 5-HT2B/C, and 5-HT3 receptors significantly attenuated this potentiation, and the cocktail containing 5-HT2 and 5-HT3 receptor antagonists almost completely abolished this potentiation. These results suggest that citalopram, via the accumulated extracellular 5-HT, activates presynaptic 5-HT2 and 5-HT3 receptors, and increases probability of glutamate release.
Medial and lateral subdivisions of the LHb were recognized in an early rat study34. Ultrastructural35 and immunohistological36 studies in rats have defined as many as four medial and five lateral LHb regions. A corresponding subnuclear structure has been described in the mouse37. As mentioned, the neurons in the LHbl and LHbm are heterogeneous with different connectivity31. The subregions of the medial and lateral nuclei give rise to distinct projections to midbrain areas15, both in rat14,30 and mouse38. Since the LHbm sends glutamatergic projections mainly to interneurons in the raphe nucli15,16, LHbm activation may suppress raphe nuclei and 5-HT release. Conversely, since the LHbl mainly projects to the RMTg, which in turn sends GABAergic projections to the midbrain dopaminergic neurons and raphe serotoninergic neurons13,14, LHbl activation may reduce the activity of dopaminergic and serotoninergic neurons.
Notably, although 5-HT inhibited glutamate transmission in a subset of LHbl neurons, we have recently shown that 5-HT increases firing of the majority of the LHbl neurons by activating the postsynaptic 5-HT receptors29. We therefore speculate that the net effect of 5-HT may increase the activity of LHb neurons, which may in turn inhibit dopaminergic and serotoninergic neurons. This may provide an additional explanation at the cellular level for the previous functional discoveries; changes of the LHb activity lead to an opposite reaction of raphe cell activity, i.e., lesion of the LHb is followed by an increase in 5-HT in the dorsal raphe nucleus20,39,40,41; where electrical42,43 as well as chemical16,25 stimulation of the habenula markedly suppressed serotonergic neurons in the raphe nuclei.
The LHb itself is a hub in the forebrain, which plays critical roles in a variety of brain functions, such as depression, addiction, and sleep cycle disorders, as well as decision making11,44,45,46,47. At the neuronal level, integration of synaptic inputs and intrinsic properties sets the frequency and pattern of neuronal firing activity. Through the influence over glutamate transmission in the LHb, 5-HT may change the LHb output to the downstream regions. In view of the extensive innervation on midbrain monoaminergic nuclei by LHb neurons, these dual actions of 5-HT may have a profound effect on the operation of the entire midbrain network. Taken together, our data suggest a feedback loop between the LHb and the raphe nuclei, which may have a role in the balance of the reciprocal neural activity. Disruption of this fine natural balance may be involved in many neuropathology like drug abuse and mood disorders: depression and anxiety.
Methods
Animals
The Animal Care and Utilization Committee of Rutgers, the State University of New Jersey, in accordance with National Institutes of Health guidelines, approved all procedures, minimizing the number of animals used and their suffering. Sprague-Dawley (SD) rats (n = 200) at postnatal days 25–35 of both sexes were housed under standard conditions at 22–24 °C, 50–60% humidity, and a 12 h light/dark cycle. Food and water are available to all rats ad libidum unless otherwise indicated. Since the data from juvenile male rats did not differ significantly from those from female rats, the data were pooled.
Brain slice preparation and electrophysiology
Coronal epithalamic slices (250 μm) were cut in ice-cold glycerol-based artificial cerebrospinal fluid (GaCSF) containing (in mM): 252 glycerol, 2.5 KCl, 1.25 NaH2PO4, 1 MgCl2, 2 CaCl2, 25 NaHCO3, 0.3 L-ascorbate, and 11 glucose, and saturated with 95%O2/5%CO2 (carbogen). Slices were incubated for >1-hr at 24–25 °C in carbogenated aCSF of similar composition as GaCSF, but with 126 mM NaCl replacing glycerol. Electrophysiological recordings (from ~700 LHb neurons) were performed at ~33 °C aCSF perfused at 1.5–2 ml/min, as described48. Patch pipettes (6–8 MΩ) were filled with internal solutions containing (in mM) 140 cesium methanesulfonate, 5 KCl, 2 MgCl2, 10 HEPES, 2 MgATP, 0.2 GTP for recordings under voltage-clamp. Both evoked and spontaneous events were recorded at a holding potential (VH) of −70 mV in the presence of gabazine (10 μM) and strychnine (1 μM), which block GABAA and glycine receptors, respectively. These events were blocked by 6,7-dinitroquinoxaline-2,3-dione (DNQX), an antagonist of α-amino-3-hydroxy-5-methylisoxazole-4-propionic acid (AMPA) receptors; indicating that they were excitatory postsynaptic currents (EPSCs), mediated by AMPA receptors. Electrical stimuli (100–200 μs in duration, 0.05 Hz) elicited EPSCs via a nichrome wire bipolar electrode positioned within 200 μm of the soma. Near the start of the recording an input/output curve was obtained and the stimulation was then set to 20–30% of the maximum, an intensity that resulted in stable responses with no failures. Paired eEPSCs were elicited with a pair of identical stimuli separated by an interval of 50 milliseconds.
Drugs
We purchased common salts and 1-(3-Chlorophenyl) biguanide hydrochloride (mCPBG); gabazine; 1,2-bis(2-aminophenoxy) ethane-N,N,N,N-tetraacetic acid (BAPTA); SCH50911; 6,7-dinitroquinoxaline-2,3-dione (DNQX); tetrodotoxin (TTX); strychnine; SB200646 hydrochloride (SB200646); ritanserin (RIT); ondansetron hydrochloride dihydrate (OND); and 5-HT hydrochloride (5-HT) from Sigma-Aldrich Chemical Company (St Louis, MO, USA). Citalopram hydrobromide, WAY100635, and 1-(3-Chlorophenyl) piperazine hydrochloride (mCPP) from Tocris Bioscience (Ellisville, MO, USA).
Data analysis and statistics
All data are expressed as means ± SEM. Baseline electrophysiological data were recorded for 10 min, before drug superfusion, and during the washout. To calculate the percent change in EPSC frequency/amplitude for a given cell, recordings during the initial control period (baseline) were averaged and normalized to 100%. Comparisons between the LHbm and LHbl were made using two-tailed unpaired Student’s t-tests. Possible significant differences in the percent distribution of EPSCs were compared by Chi-square test. The different concentrations of 5-HT on sEPSCs were analyzed using two-way ANOVA with “subregions” (LHbm vs LHbl) as between-group factors and “dose” (from 0.1 to 30 μM) as within subject factor. Tukey’s post hoc test was used for multiple dose comparisons. The effects of 5-HT antagonists on changes in sEPSCs induced by 5-HT/citalopram were assessed by paired t-test or one-way ANOVA. Dose-response data were fitted to the logistic equation: y = 100xα/(xα + xoα), where y is the percentage change, x is the concentration of 5-HT, α the slope parameter, and xo the 5-HT concentration which induces a half-maximal change. Values of p < 0.05 were considered significant.
Additional Information
How to cite this article: Xie, G. et al. Serotonin modulates glutamatergic transmission to neurons in the lateral habenula. Sci. Rep. 6, 23798; doi: 10.1038/srep23798 (2016).
Acknowledgments
This research is supported by NIH-NIAAA AA021657, AA022292 (JHY).
Footnotes
Author Contributions J.H.Y. and A.B. elaborated the study design. J.H.Y., G.X. and W.Z. drafted the article. G.X., W.Z., L.W., W.L. and W.W. collected data. All authors critically reviewed content and approved final version for publication.
References
- Dahlstrom A. & Fuxe K. Localization of monoamines in the lower brain stem. Experientia 20, 398–399 (1964). [DOI] [PubMed] [Google Scholar]
- Michelsen K. A., Prickaerts J. & Steinbusch H. W. The dorsal raphe nucleus and serotonin: implications for neuroplasticity linked to major depression and Alzheimer’s disease. Prog Brain Res 172, 233–264, 10.1016/S0079-6123(08)00912-6 (2008). [DOI] [PubMed] [Google Scholar]
- Jahanshahi A. et al. Altered expression of neuronal tryptophan hydroxylase-2 mRNA in the dorsal and median raphe nuclei of three genetically modified mouse models relevant to depression and anxiety. J Chem Neuroanat 41, 227–233, 10.1016/j.jchemneu.2011.05.015 (2011). [DOI] [PubMed] [Google Scholar]
- Steinbusch H. W. & Nieuwenhuys R. Localization of serotonin-like immunoreactivity in the central nervous system and pituitary of the rat, with special references to the innervation of the hypothalamus. Adv Exp Med Biol 133, 7–35 (1981). [DOI] [PubMed] [Google Scholar]
- Hayes D. J. & Greenshaw A. J. 5-HT receptors and reward-related behaviour: a review. Neurosci Biobehav Rev 35, 1419–1449, 10.1016/j.neubiorev.2011.03.005 (2011). [DOI] [PubMed] [Google Scholar]
- Maier S. F. & Watkins L. R. Stressor controllability and learned helplessness: the roles of the dorsal raphe nucleus, serotonin, and corticotropin-releasing factor. Neurosci Biobehav Rev 29, 829–841, 10.1016/j.neubiorev.2005.03.021 (2005). [DOI] [PubMed] [Google Scholar]
- Li B. et al. Synaptic potentiation onto habenula neurons in the learned helplessness model of depression. Nature 470, 535–539, 10.1038/nature09742 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li K. et al. betaCaMKII in lateral habenula mediates core symptoms of depression. Science 341, 1016–1020, 10.1126/science.1240729 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lecca S. et al. Rescue of GABA and GIRK function in the lateral habenula by protein phosphatase 2A inhibition ameliorates depression-like phenotypes in mice. Nat Med, 10.1038/nm.4037 (2016). [DOI] [PubMed] [Google Scholar]
- Sachs B. D., Ni J. R. & Caron M. G. Brain 5-HT deficiency increases stress vulnerability and impairs antidepressant responses following psychosocial stress. Proc Natl Acad Sci USA 112, 2557–2562, 10.1073/pnas.1416866112 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hikosaka O. The habenula: from stress evasion to value-based decision-making. Nat Rev Neurosci 11, 503–513, 10.1038/nrn2866 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shabel S. J., Proulx C. D., Trias A., Murphy R. T. & Malinow R. Input to the lateral habenula from the basal ganglia is excitatory, aversive, and suppressed by serotonin. Neuron 74, 475–481, 10.1016/j.neuron.2012.02.037 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Proulx C. D., Hikosaka O. & Malinow R. Reward processing by the lateral habenula in normal and depressive behaviors. Nat Neurosci 17, 1146–1152, 10.1038/nn.3779 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sego C. et al. Lateral habenula and the rostromedial tegmental nucleus innervate neurochemically distinct subdivisions of the dorsal raphe nucleus in the rat. J Comp Neurol 522, 1454–1484, 10.1002/cne.23533 (2014). [DOI] [PubMed] [Google Scholar]
- Kim U. Topographic commissural and descending projections of the habenula in the rat. J Comp Neurol 513, 173–187, 10.1002/cne.21951 (2009). [DOI] [PubMed] [Google Scholar]
- Varga V., Kocsis B. & Sharp T. Electrophysiological evidence for convergence of inputs from the medial prefrontal cortex and lateral habenula on single neurons in the dorsal raphe nucleus. Eur J Neurosci 17, 280–286 (2003). [DOI] [PubMed] [Google Scholar]
- Hikosaka O., Sesack S. R., Lecourtier L. & Shepard P. D. Habenula: crossroad between the basal ganglia and the limbic system. J Neurosci 28, 11825–11829, 10.1523/JNEUROSCI.3463-08.2008 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pobbe R. L. & Zangrossi H. Jr. The lateral habenula regulates defensive behaviors through changes in 5-HT-mediated neurotransmission in the dorsal periaqueductal gray matter. Neurosci Lett 479, 87–91, 10.1016/j.neulet.2010.05.021 (2010). [DOI] [PubMed] [Google Scholar]
- Vertes R. P., Fortin W. J. & Crane A. M. Projections of the median raphe nucleus in the rat. J Comp Neurol 407, 555–582 (1999). [PubMed] [Google Scholar]
- Yang L. M., Hu B., Xia Y. H., Zhang B. L. & Zhao H. Lateral habenula lesions improve the behavioral response in depressed rats via increasing the serotonin level in dorsal raphe nucleus. Behav Brain Res 188, 84–90, 10.1016/j.bbr.2007.10.022 (2008). [DOI] [PubMed] [Google Scholar]
- Aizawa H., Kobayashi M., Tanaka S., Fukai T. & Okamoto H. Molecular characterization of the subnuclei in rat habenula. J Comp Neurol 520, 4051–4066, 10.1002/cne.23167 (2012). [DOI] [PubMed] [Google Scholar]
- Bull E. J., Porkess V., Rigby M., Hutson P. H. & Fone K. C. Pre-treatment with 3,4-methylenedioxymethamphetamine (MDMA) causes long-lasting changes in 5-HT2A receptor-mediated glucose utilization in the rat brain. J Psychopharmacol 20, 272–280, 10.1177/0269881106059583 (2006). [DOI] [PubMed] [Google Scholar]
- Cornide-Petronio M. E., Anadon R., Barreiro-Iglesias A. & Rodicio M. C. Serotonin 1A receptor (5-HT1A) of the sea lamprey: cDNA cloning and expression in the central nervous system. Brain Struct Funct 218, 1317–1335, 10.1007/s00429-012-0461-y (2013). [DOI] [PubMed] [Google Scholar]
- Mitchell E. A. & Pratt J. A. Neuroanatomical structures involved in the action of the 5-HT3 antagonist ondansetron: a 2-deoxyglucose autoradiographic study in the rat. Brain Res 538, 289–294 (1991). [DOI] [PubMed] [Google Scholar]
- Jones D. N. et al. The distribution of 5-HT3 recognition sites in the marmoset brain. Eur J Pharmacol 215, 63–67 (1992). [DOI] [PubMed] [Google Scholar]
- Pompeiano M., Palacios J. M. & Mengod G. Distribution of the serotonin 5-HT2 receptor family mRNAs: comparison between 5-HT2A and 5-HT2C receptors. Brain Res Mol Brain Res 23, 163–178 (1994). [DOI] [PubMed] [Google Scholar]
- Han L. N. et al. Activation of serotonin receptors in the lateral habenular nucleus increases the expression of depression-related behaviors in the hemiparkinsonian rat. Neuropharmacology 93C, 68–79, 10.1016/j.neuropharm.2015.01.024 (2015). [DOI] [PubMed] [Google Scholar]
- Hwang E. K. & Chung J. M. 5HT(1B) receptor-mediated pre-synaptic depression of excitatory inputs to the rat lateral habenula. Neuropharmacology 81, 153–165, 10.1016/j.neuropharm.2014.01.046 (2014). [DOI] [PubMed] [Google Scholar]
- Zuo W. et al. Serotonin stimulates lateral habenula via activation of the post-synaptic serotonin 2/3 receptors and transient receptor potential channels. Neuropharmacology 101, 449–459, 10.1016/j.neuropharm.2015.10.012 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goncalves L., Sego C. & Metzger M. Differential projections from the lateral habenula to the rostromedial tegmental nucleus and ventral tegmental area in the rat. J Comp Neurol 520, 1278–1300, 10.1002/cne.22787 (2012). [DOI] [PubMed] [Google Scholar]
- Weiss T. & Veh R. W. Morphological and electrophysiological characteristics of neurons within identified subnuclei of the lateral habenula in rat brain slices. Neuroscience 172, 74–93, 10.1016/j.neuroscience.2010.10.047 (2011). [DOI] [PubMed] [Google Scholar]
- Tohda M. & Watanabe H. Imipramine-induced increase in 5-HT2C receptor mRNA level in the rat brain. Neurosci Res 24, 189–193, 10.1016/0168-0102(95)00992-2 (1996). [DOI] [PubMed] [Google Scholar]
- Shabel S. J., Proulx C. D., Piriz J. & Malinow R. Mood regulation. GABA/glutamate co-release controls habenula output and is modified by antidepressant treatment. Science 345, 1494–1498, 10.1126/science.1250469 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herkenham M. & Nauta W. J. Efferent connections of the habenular nuclei in the rat. J Comp Neurol 187, 19–47, 10.1002/cne.901870103 (1979). [DOI] [PubMed] [Google Scholar]
- Andres K. H., von During M. & Veh R. W. Subnuclear organization of the rat habenular complexes. J Comp Neurol 407, 130–150 (1999). [DOI] [PubMed] [Google Scholar]
- Geisler S., Andres K. H. & Veh R. W. Morphologic and cytochemical criteria for the identification and delineation of individual subnuclei within the lateral habenular complex of the rat. J Comp Neurol 458, 78–97, 10.1002/cne.10566 (2003). [DOI] [PubMed] [Google Scholar]
- Wagner F., Stroh T. & Veh R. W. Correlating habenular subnuclei in rat and mouse by using topographic, morphological, and cytochemical criteria. J Comp Neurol 522, 2650–2662, 10.1002/cne.23554 (2014). [DOI] [PubMed] [Google Scholar]
- Quina L. A. et al. Efferent pathways of the mouse lateral habenula. J Comp Neurol 523, 32–60, 10.1002/cne.23662 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalen P., Pritzel M., Nieoullon A. & Wiklund L. Further evidence for excitatory amino acid transmission in the lateral habenular projection to the rostral raphe nuclei: lesion-induced decrease of high affinity glutamate uptake. Neurosci Lett 68, 35–40 (1986). [DOI] [PubMed] [Google Scholar]
- Kalen P., Strecker R. E., Rosengren E. & Bjorklund A. Regulation of striatal serotonin release by the lateral habenula-dorsal raphe pathway in the rat as demonstrated by in vivo microdialysis: role of excitatory amino acids and GABA. Brain Res 492, 187–202 (1989). [DOI] [PubMed] [Google Scholar]
- Luo X. F. et al. Lateral habenula as a link between dopaminergic and serotonergic systems contributes to depressive symptoms in Parkinson’s disease. Brain Res Bull 110, 40–46, 10.1016/j.brainresbull.2014.11.006 (2015). [DOI] [PubMed] [Google Scholar]
- Wang R. Y. & Aghajanian G. K. Physiological evidence for habenula as major link between forebrain and midbrain raphe. Science 197, 89–91 (1977). [DOI] [PubMed] [Google Scholar]
- Lim L. W. et al. Electrical stimulation alleviates depressive-like behaviors of rats: investigation of brain targets and potential mechanisms. Transl Psychiatry 5, e535, 10.1038/tp.2015.24 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lecourtier L. et al. Habenula lesions alter synaptic plasticity within the fimbria-accumbens pathway in the rat. Neuroscience 141, 1025–1032, 10.1016/j.neuroscience.2006.04.018 (2006). [DOI] [PubMed] [Google Scholar]
- Matsumoto M. & Hikosaka O. Lateral habenula as a source of negative reward signals in dopamine neurons. Nature 447, 1111–1115, 10.1038/nature05860 (2007). [DOI] [PubMed] [Google Scholar]
- Salaberry N. L. & Mendoza J. Insights into the Role of the Habenular Circadian Clock in Addiction. Front Psychiatry 6, 179, 10.3389/fpsyt.2015.00179 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zuo W. et al. Ethanol drives aversive conditioning through dopamine 1 receptor and glutamate receptor-mediated activation of lateral habenula neurons. Addict Biol, 10.1111/adb.12298 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zuo W., Chen L., Wang L. & Ye J. H. Cocaine facilitates glutamatergic transmission and activates lateral habenular neurons. Neuropharmacology 70, 180–189, 10.1016/j.neuropharm.2013.01.008 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]