Abstract
Filamentous bacteria of the Desulfobulbaceae family can conduct electrons over centimeter-long distances thereby coupling oxygen reduction at the surface of marine sediment to sulfide oxidation in deeper anoxic layers. The ability of these cable bacteria to use alternative electron acceptors is currently unknown. Here we show that these organisms can use also nitrate or nitrite as an electron acceptor thereby coupling the reduction of nitrate to distant oxidation of sulfide. Sulfidic marine sediment was incubated with overlying nitrate-amended anoxic seawater. Within 2 months, electric coupling of spatially segregated nitrate reduction and sulfide oxidation was evident from: (1) the formation of a 4–6-mm-deep zone separating sulfide oxidation from the associated nitrate reduction, and (2) the presence of pH signatures consistent with proton consumption by cathodic nitrate reduction, and proton production by anodic sulfide oxidation. Filamentous Desulfobulbaceae with the longitudinal structures characteristic of cable bacteria were detected in anoxic, nitrate-amended incubations but not in anoxic, nitrate-free controls. Nitrate reduction by cable bacteria using long-distance electron transport to get privileged access to distant electron donors is a hitherto unknown mechanism in nitrogen and sulfur transformations, and the quantitative importance for elements cycling remains to be addressed.
Keywords: nitrate, cable bacteria, electric current, cathodic reduction, sediment
Introduction
Electric currents can couple cathodic O2 reduction (O2+4H++4e−→2H2O) at the surface of marine sediment to anodic oxidation of sulfide (H2S+4H2O→SO42−+10H++8e−) over distances of more than 1 cm (Nielsen et al., 2010; Risgaard-Petersen et al., 2012). Evidence for electric coupling between these spatially segregated half-cell reactions includes (1) the formation of oxygen- and sulfide-depleted zones in the absence of reactive Mn and Fe oxides and mixing, and (2) the appearance of a distinct pH maximum in the oxic zone and a minimum in the anoxic zone, in accordance with proton consumption by cathodic O2 reduction and proton production by anodic sulfide oxidation, respectively. Recently, Pfeffer et al. (2012) showed that the electric coupling between spatially segregated half-cell reactions was mediated by filamentous, multicellular bacteria belonging to the family Desulfobulbaceae. These ‘cable bacteria' have uniform ridges formed by strings located inside a periplasmic space that is continuous between the individual cells. The strings have distinct electronic properties comparable to electron conductors, and they are proposed to be electric wires with the surrounding cytoplasmic and periplasmic membranes serving as insulation (Pfeffer et al., 2012).
The presence of cable bacteria that act as electron conductors and allow redox couples to interact far beyond their physical presence promotes a sediment geochemistry that cannot be understood with classical geochemical models (Risgaard-Petersen et al., 2012). However, the occurrence of these organisms and their impact on sediment geochemistry has been addressed only in the presence of O2, and to date, it remains unknown whether other electron acceptors such as NO3− can be used. Thermodynamically, NO3− is an electron acceptor almost as good as O2 and several prokaryotes are able to switch from O2 to NO3− respiration when anoxic conditions occur. Also, the closest cultured relative of cable bacteria, Desulfobulbus propionicus, can grow with NO3− as an electron acceptor and can couple sulfide oxidation to NO2− reduction (Dannenberg et al., 1992).
In this study we investigated whether cathodic NO3− reduction can sustain the distant oxidation of sulfide in marine sediment as previously described for O2 (Nielsen et al., 2010). In a first set of experiments, we incubated sediment from Aarhus Bay under anoxic seawater containing 200–230 μM NO3− and assessed the development of the traits typical of the electric coupling between spatially segregated half-cell reactions. Successively, we ran parallel incubations to compare NO3− and O2 effectiveness in sustaining the distant oxidation of sulfide. Finally, we addressed the nature of the conductors investigating whether cable bacteria can grow under anoxic conditions in the presence of NO3−.
Materials and methods
Sediment sampling and pre-treatment
Intact sediment samples were collected from Aarhus Bay (Denmark) at station M5 (56°06′20′′N, 10°27′48′′E; depth 30 m) using a box corer. On board, the upper 10–12 cm of sediment were discarded to minimize the presence of metal oxides and exclude large burrowing animals. The underlying sulfidic sediment was sealed in airtight bags, brought to the laboratory and stored at 15 °C. Within a few weeks, the bags were opened and the sediment was sieved (sieve mesh size 0.5 mm), homogenized and packed into glass liners or chambers before being incubated. Sediment exposure to air was minimized throughout the handling procedures.
Sediment incubation with anoxic, NO3−-amended overlying water
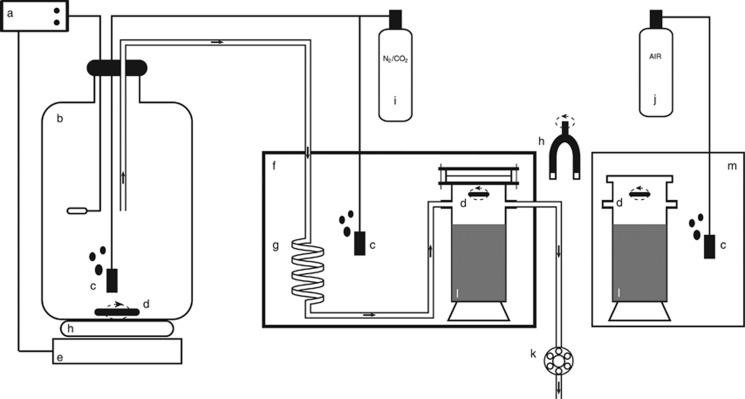
To address whether NO3− reduction can sustain distant oxidation of sulfide, the sediment was incubated in a modified version of the flow-through system described by Risgaard-Pedersen et al. (1994), where both the gas concentration and the supply of NO3− could be controlled (Figure 1). Preliminary attempts to incubate sediment in NO3−-amended anoxic seawater in batch mode failed due to substantial bubble formation in the sediment cores as a result of N2 production from denitrification. In November 2011, cylindrical glass chambers (inner diameter: 5.4 cm; height: 16 cm) were filled with sulfidic sediment up to ≈3 cm below the upper rim. The chambers were sealed with glass lids to be filled with water without leaving a gas phase, connected to the flow-through system and immersed into an aquarium containing anoxic water to assure that no O2 would diffuse through the sealing. Anoxic, NO3−-amended, artificial seawater (salinity: 30‰) based on MilliQ water (Millipore, Billerica, MA, USA) and Red Sea Salts (Red Sea Fish Pharm Ltd, Eilat, Israel) was pumped from a reservoir into the chambers at a constant rate of 190 μl min−1. Magnetic stir bars driven by an external rotating magnet maintained a homogeneous water column above the sediment in the chambers. The water in the reservoir was maintained at 30 °C and purged with N2 containing 0.04% CO2. Before entering the chambers, the water was cooled down to 13.6 °C to allow a constant exposure of the sediment to gas-undersaturated water, thereby preventing gas bubble formation. Throughout the 64 days of incubation, the NO3− concentration in the inflowing and outflowing water was regularly monitored. The NO3− concentration of the reservoir water remained constant within 266–273 μM.
Figure 1.
The flow-through system. Arrows indicate the direction of water flow. (a) Thermostat; (b) 20-liter water reservoir maintained at 30 °C; (c) gas-diffusing stones; (d) stirrer (teflon-coated magnetic bars); (e) heater; (f) sealed anoxic aquarium maintained at 13.6 °C; (g) coil for cooling of inflow water; (h) rotating magnet driving the stirrers; (i) N2 (99.96%) and CO2 (0.04%) gas mix tank; (j) air tank; (k) peristaltic pump; (l) sediment; (m) aerated aquarium.
Microprofiles of pH were measured in the sediment after 7, 18, 34, 53 and 64 days of incubation using pH microsensors. The depth distribution of NO3−+NO2− (NOx−), O2 and H2S was measured at the end of the experiment using in-house made sensors for NOx−, O2 and H2S. The sediment cores were then sliced in 3 mm sections down to 18 mm depth. Each section was homogenously mixed and sediment samples (approx. vol. 1 ml) were collected in triplicates, transferred into polypropylene centrifuge tubes and frozen at −20 °C for later analysis of the intracellular+porewater NO3− pool. This analysis was performed to address the presence of NO3−-storing organisms (for example, Foraminifera and Beggiatoa) in the sediment.
Comparison of NO3− and O2 effectiveness in sustaining distant sulfide oxidation
In March 2012, freshly collected sediment was pre-treated as described above and incubated in three treatments where the overlying artificial seawater was maintained aerated, anoxic (NO3−-free) or anoxic in the presence of 200 μM NO3−. The three parallel incubations lasted for 27, 24 and 28 days, respectively. In the anoxic incubations in presence of NO3− (hereafter referred to as the NO3− treatment), the sediment was packed into glass chambers and incubated in the flow-through system as described above. Additional sediment chambers (prepared as above, but not sealed at the top) were used for the oxic treatment. These chambers were immersed into an aquarium filled with artificial seawater, constantly flushed with air and maintained at 13.6 °C (Figure 1). Homogeneous water chemistry was maintained by a water pump placed in the aquarium and a magnetic stirrer suspended at about 2 cm above the sediment surface. In the anoxic (NO3−-free) control, the sediment was packed into glass liners (inner diameter: 1.8 cm; height: 10 cm) and placed into a sealed aquarium filled with artificial seawater at 13.6 °C. The water was kept stirred by means of an aquarium pump. Anoxic conditions (O2<0.1 μM) were maintained by constantly flushing the water with a gas mixture of N2 (99.96%) and CO2 (0.04%), and monitored throughout the entire incubation with the sensitive STOX O2 sensor (Revsbech et al., 2009) inserted into the aquaria and connected to a strip chart recorder.
To evaluate whether cable bacteria can alternate between O2 and NO3− as a terminal electron acceptor for driving distant sulfide oxidation, sediment cores previously incubated in the oxic treatment were exposed to NO3−-amended anoxic water. Hence, at the end of the oxic incubation the chambers were sealed at the top and connected to the flow-through system described above for the following 9 days. At the end of each incubation the vertical microdistribution of O2, NOx−, H2S and pH in the sediment was measured with microsensors.
Microsensor measurements
Sediment microprofiles of H2S, O2 and pH were measured with microsensors (Revsbech and Jorgensen, 1986; Revsbech, 1989; Jeroschewski et al., 1996), whereas microscale biosensors were prepared according to Revsbech and Glud (2009) for NOx− (NO3−+NO2−). Total hydrogen sulfide (ΣH2S=[H2S]+[HS−]+[S2−]) concentrations were calculated at each depth from the measured H2S and pH values (Jeroschewski et al., 1996). Microprofiles were measured by mounting single sensors on a computer-controlled microprofiler as described by Nielsen et al. (2009). Sensor tips were manually positioned at the sediment surface while observing them through a horizontal dissection microscope. Before measuring microprofiles in the NO3− treatment, the water flow was stopped and the chambers removed from the anoxic aquarium. Microsensors were then inserted through openings (diameter: 1 cm) previously drilled into the glass lid (and maintained sealed during the incubation with rubber stoppers). To prevent O2 diffusion into the glass chamber, the openings were flushed with N2 during measurements. Consumption rates of the measured parameters were estimated by modeling the concentration microprofiles with the algorithm developed by Berg et al. (1998). Porosity (vol/vol) was determined from density and water content of 3-mm-thick sediment slides. The diffusion coefficients of O2, NO3− and HS− were calculated according to Boudreau (1997).
Analysis of the intracellular+porewater NO3− pool
The frozen sediment samples were thawed in boiling water to promote cell lysis (Risgaard-Petersen et al., 2006) and then centrifuged at 2000 g for 10 min. The concentration of pooled intracellular and porewater-dissolved NO3− was determined in the supernatant on a chemiluminescence detector (CLD 86, Eco Physics, Duernten, Switzerland) after being reduced to NO by the VCl3 method (Braman and Hendrix, 1989).
Estimation of the cathodic O2 and NO3− reduction rates and associated current densities
Minimum estimates of the cathodic O2 reduction and equivalent current density were calculated from the electron–proton–oxygen mass balance proposed by Nielsen et al. (2010).
Minimum estimates of the cathodic NO3− reduction and equivalent current density were estimated from an electron–proton–nitrate mass balance. We considered two alternative cathodic NO3− reductions that lead either to N2 (1) or to NH4+ (2) production:
 |
 |
As main non-cathodic NO3− reducing processes we considered organotrophic denitrification (3) and FeS oxidation with NO3− (4).
 |
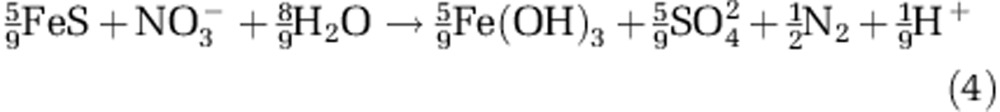 |
The net proton consumption in the NO3− reduction zones (JH+) equals the proton consumption by cathodic NO3− reduction minus the proton production due to the non-cathodic consumption of NO3−. This can be expressed as follows:
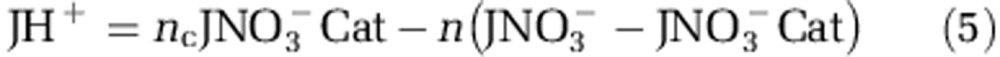 |
Here  is the total NO3− consumption rate,
is the total NO3− consumption rate,  is the rate of cathodic NO3− reduction, nc is the number of moles of protons consumed by one mole of NO3− reduced cathodically and can be either 6 or 10 (Equations 1 and 2), n is the number of moles of protons produced by the non-cathodic reduction of one mole of NO3− and can be either 1/4 or 1/9 (Equations 3 and 4). Rearranging Equation 5 gives the following expression for the rate of cathodic NO3− reduction:
is the rate of cathodic NO3− reduction, nc is the number of moles of protons consumed by one mole of NO3− reduced cathodically and can be either 6 or 10 (Equations 1 and 2), n is the number of moles of protons produced by the non-cathodic reduction of one mole of NO3− and can be either 1/4 or 1/9 (Equations 3 and 4). Rearranging Equation 5 gives the following expression for the rate of cathodic NO3− reduction:
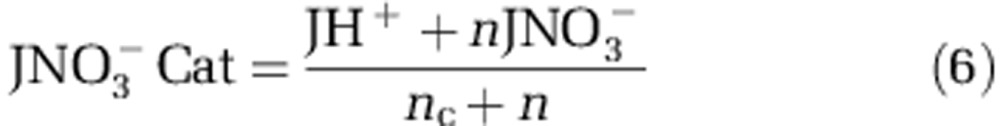 |
The cathodic NO3− reduction rate was calculated for each of the four possible scenarios in which one of the two cathodic NO3− reductions was alternatively assumed to compete with one of the two non-cathodic NO3− reductions by substituting nc and n according to the considered stoichiometry. JH+ in the NO3− reduction zone was calculated on the basis of the pH microprofile and the dissolved inorganic carbon concentration in the sediment porewater according to Nielsen et al. (2010). Dissolved inorganic carbon was measured as CO2 after acidification on a gas chromatograph equipped with a thermal conductivity detector (ML GC 82, Mikrolab, Aarhus, Denmark).  was calculated as the net NO3− flux across the water–sediment interface in the flow-through incubations by means of the following equation:
was calculated as the net NO3− flux across the water–sediment interface in the flow-through incubations by means of the following equation:
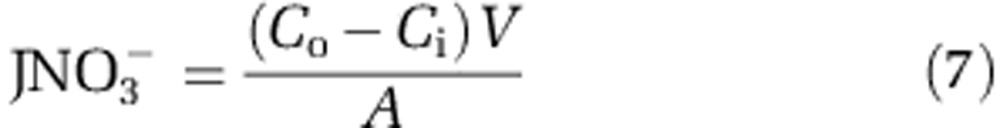 |
where Co and Ci are the NO3− concentrations in the water at the outlet and inlet of the chamber, respectively; V is the water flow rate and A is the surface area of the sediment core. The current density (Je−) needed to sustain the cathodic NO3− reduction was calculated as follows:
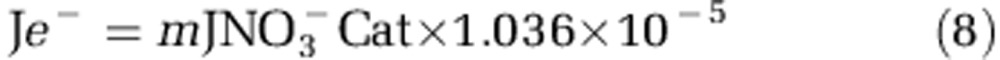 |
where m is the number of moles of electrons consumed by one mole of NO3− reduced cathodically and can be either 5 or 8 (Equations 1 and 2), and 1.036 × 10−5 is the conversion factor from mol e− s−1 to Ampere.
Cable bacteria identification and density estimation
To investigate whether cable bacteria can grow by respiring NO3− under anoxic conditions, freshly collected sediment was pre-treated as described above, packed into glass liners (inner diameter: 3.5 cm; height: 5.4 cm) and incubated in batch mode in anoxic, NO3−-amended seawater. Anoxic incubations without NO3− served as controls. The O2 concentration in the overlying water (monitored with an O2 optode; Lumos, Graz University of Technology) was kept below 15 nM by continuous bubbling with N2/CO2 as described above, except for the short profiling periods, when O2 was up to 2.5 μM. Nitrate concentrations were kept at 80–350 μM (monitored as described above) by regularly adding NO3− to the overlying water. Microprofiles of pH and H2S were recorded after 5, 10 and 12 days to confirm the establishment of electric coupling between NO3− reduction and sulfide oxidation in the NO3−-amended cores. After 12 and 14 days, sediment cores were sectioned in 2 mm intervals down to 10 mm; sections were fixed in 50% ethanol (final concentration) and stored at −20 °C for fluorescence in situ hybridization (FISH), or snap frozen in liquid nitrogen and stored at −80 °C for DNA analysis. Control cores were sampled after 28 days.
DNA extraction (using PowerSoil DNA Isolation Kit (MO BIO Laboratories, Carlsbad, CA, USA)), PCR, cloning and sequence analysis were done as previously described (Pfeffer et al., 2012), except that two different primer combinations were applied for PCR: (i) 8F (Loy et al., 2002)/DSBB+1297R (Kjeldsen et al., 2007); and (ii) ELF645F (5′-CTTGGCTTGAGTATCAGAGG-3′)/DSBB+1297R. The annealing temperature was 58 °C in both reactions. Sequences have been deposited at Genbank under accession number KJ021894 to KJ021926.
Identification and quantification of cable bacteria by FISH were done as previously described (Pfeffer et al., 2012). Probes DSB706 (specific for Desulfobulbaceae) and ELF645 (specific for a Desulfobulbaceae lineage confirmed as cable bacteria) were applied to 6 × -diluted subsamples from 4–6 mm depth from NO3−-amended and control sediment of triplicate cores. Filament length density (that is, meters of filament per cubic centimeter of sediment) was determined for probe DSB706-positive filaments using the line-intercept method (Newman, 1966) as described for filamentous bacteria (Hogslund et al., 2010). For comparison, filament length was directly measured in triplicates in digital images of at least 300 microscopic fields by microscope digital photography using imaging software (AxioVision, Carl Zeiss, Göttingen, Germany) (Schauer et al., 2014). The fraction of ELF645-positive filaments was determined relative to all Desulfobulbaceae (probe DSB706-positive) filaments in triplicates by checking at least 1000 cells after double hybridizations with both probes, labeled in CY3 or FITC. Analyses were carried out on an Axiovert 200 M epifluorescence microscope (Carl Zeiss).
Atomic force microscopy
The outer surface of cable bacteria was investigated by a combination of FISH and atomic force microscopy (AFM) imaging. Single filaments were picked with a sterile glass hook under microscopic guidance (Pfeffer et al., 2012), cleaned in MilliQ water and transferred to gelatine-coated cover slides. Samples were dehydrated, and FISH was performed as described above with probe DSB706. Optical and AFM imaging were performed on a Zeiss Axiovert 200 M fluorescence microscope combined with a Nanowizard II AFM (JPK Instruments, Berlin, Germany). The coverslip was placed on the inverted microscope, and fluorescence images were obtained on the dry cells without any anti-bleaching agent using Zeiss filterset 43 to detect the CY3-labeled FISH probe. AFM images were then obtained from the same cells in ambient conditions using Olympus OMCL-AC160TS silicon cantilevers with a nominal spring constant of 26 N m−1 in intermittent contact mode at a target frequency of 332 Hz, target amplitude of 1.5 V, set-point value of 0.95 V and a scan rate of 1 Hz.
Results
Sediment geochemistry in the presence of NO3− in overlying water
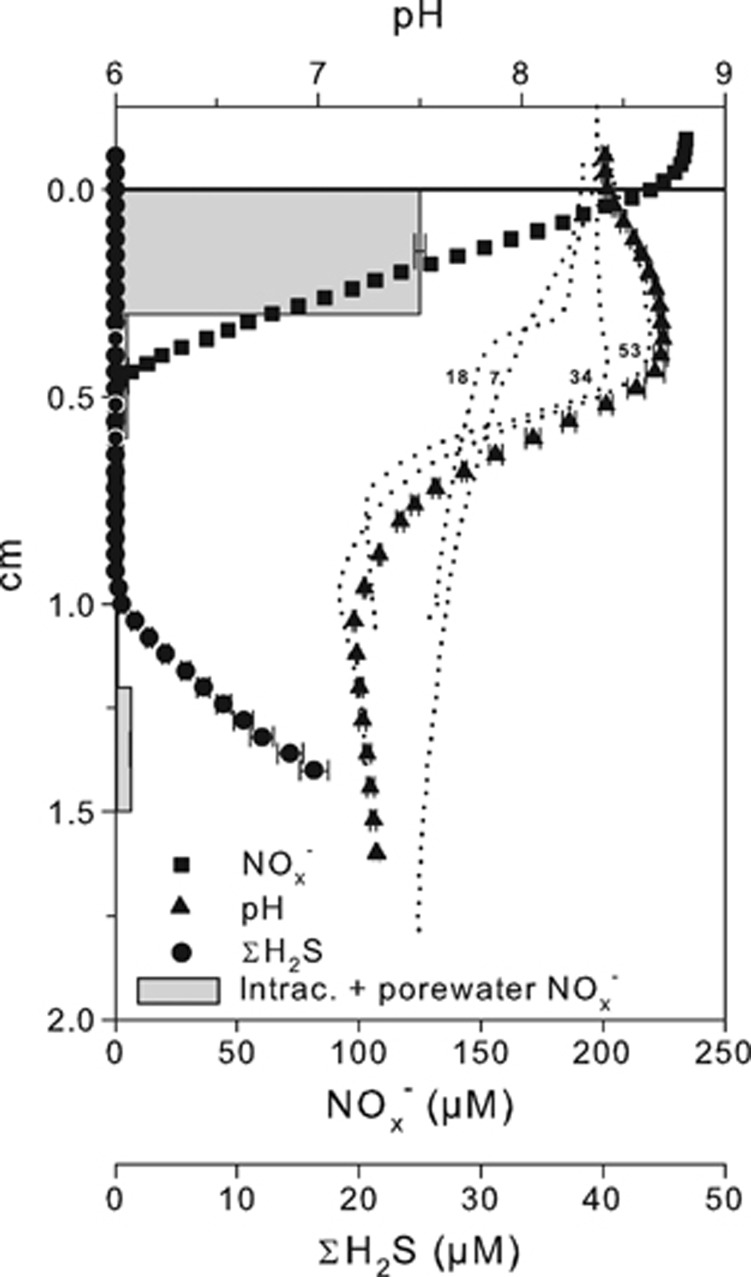
Microdistributions of NOx−, ΣH2S and pH in sediment exposed to anoxic NO3−-amended seawater are shown in Figure 2. At the end of the incubation (Day 64), NOx− penetrated 4–5 mm into the sediment, whereas ΣH2S was detectable from a depth of 9–10 mm, resulting in a 4–6-mm-thick zone devoid from both NOx− and ΣH2S. Within the incubation time, the pH profile developed to a maximum at 4.2 mm and a minimum at 1 cm depth. The pH maximum and minimum indicated intense proton consumption and production at depths of NOx− and ΣH2S consumption, respectively. Throughout the incubation, the pH minimum always coincided with the sulfide front (data not shown). The areal consumption rate of NOx− was 118 and 104 μmol m−2 h−1 when calculated from microprofile modeling and from Equation 7, respectively. The ΣH2S oxidation rate derived from microprofile modeling was 11.9 μmol m−2 h−1.
Figure 2.
Microprofiles of pH, ΣH2S and NOx− measured in sediment incubated for 64 days under anoxic overlying water in the presence of 235 μM NO3− in November 2011. Data are shown as mean±s.e.m. (n=3). Dotted lines represent single pH profiles measured after 7, 18, 34 and 53 days of incubation. Gray bars represent pooled intracellular and free NO3− extracted from frozen and thawed sediment samples. Data are shown as mean±s.e.m. (n=3).
Pooled intracellular and porewater NO3− profile showed concentrations comparable to those measured with the NOx− microsensor in the depth interval 0–3 mm. Below 3 mm depth, the concentration decreased to insignificant values, indicating the absence of NO3−-storing organisms.
Sediment geochemistry in the presence or absence of O2 and NO3− in overlying water
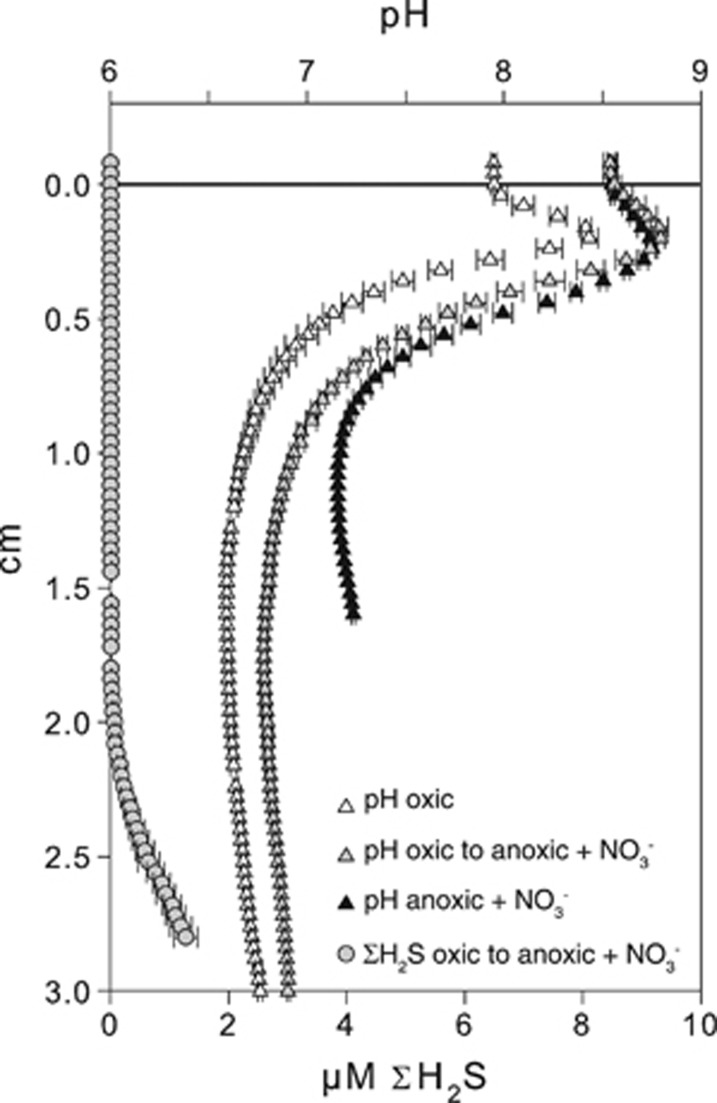
Microdistribution of O2, ΣH2S, NOx− and pH in three parallel incubations where the sediment was exposed to oxic, anoxic NO3−-amended and anoxic NO3−-free seawater are shown in Figure 3. Sediment incubated with anoxic NO3−-free overlying water remained fully sulfidic and the pH decreased along with depth after 24 days of incubation. In the NO3− treatment, ΣH2S was progressively consumed from the surface sediment, and at the end of the incubation (Day 28) the sulfidic front was detected at a depth of 11 mm. Nitrate penetrated 3.8 mm into the sediment and a distinct pH peak indicated proton consumption in the NO3− reduction zone. The estimated current density in the NO3− treatment varied between 6.6 and 8.8 mA m−2 depending on whether N2 or NH4+ was considered as the cathodic end product and whether organic carbon or FeS was the major electron donor for non-cathodic NO3− reduction. The estimated cathodic NO3− reduction represented only 12 and 19% of the total NO3− consumption. In the oxic treatment, ΣH2S was detected at 21 mm depth after 27 days of incubation. Oxygen penetrated 1.8 mm into the sediment resulting in a ≈19-mm-thick suboxic zone. The pH microprofile showed a peak at a depth of 1.8 mm indicating net proton consumption. The cathodic O2 reduction accounted for 34% of the total O2 consumption supporting a current density of 28 mA m−2. Both the pH peak and the sulfide-free zone remained 9 days after the overlying oxic water was replaced with NO3−-amended anoxic water (Figure 4).
Figure 3.
Sediment microprofiles of pH, ΣH2S, O2 and NOx− measured in sediment cores incubated in March 2012 under oxic (left panel), anoxic 200 μM NO3−-amended (center panel) and anoxic NO3−-free (left panel) overlying water.
Figure 4.
(Gray) Microprofiles of pH (triangle) and ΣH2S (circles) measured in cores incubated under oxic overlying water for 27 days and successively exposed to anoxic overlying water containing 200 μM NO3− for 9 days (oxic to anoxic+NO3−). For comparison, pH microprofiles measured in the same core before the change in overlying water conditions (open triangles) (oxic) and pH microprofiles measured in cores only exposed to the NO3− treatment (black triangles) (anoxic+NO3−) are shown (these profiles correspond to the profiles shown in Figure 3). Data are shown as mean±s.e.m. (n=3).
Filaments identification and density
In the batch incubation with NO3−, the ΣH2S front retracted from 2 mm (Day 5) to 4.7 mm (Day 10) to 7.6 mm (Day 12), and a pH peak appeared at 1.6 mm depth at day 12. The controls remained fully sulfidic and no pH peak developed (data not shown). Filamentous Desulfobulbaceae similar to the previously described cable bacteria (Pfeffer et al., 2012) were detected by FISH with probe DSB706 (Figure 5a). Filament length density at 4–6 mm depth was 30±7 m cm−3 (mean of triplicate cores±s.d.) by the line-intercept method and 85±22 m cm−3 by direct measurement in the NO3−-amended sediment. Filament diameters ranged from 0.4 to 1.0 μm, with an average of 0.6 μm. Only a few and rather short filaments were detected in the controls, with a length density <3 m cm−3. A filament subpopulation hybridized with the more specific probe ELF645 (Figure 5a); these cable bacteria accounted for 21.2% (±12.6%) of all DSB706-positive filamentous Desulfobulbaceae. Likewise, only 5 out of 33 Desulfobulbaceae-like 16S rRNA gene sequences clustered with the previously described ‘cable bacteria lineage' (Pfeffer et al., 2012) (Supplementary Figure S1).
Figure 5.
(a) Fluorescence in situ hybridization micrograph of filamentous Desulfobulbaceae identified by probe DSB706. (b) The characteristic ‘cable-like' structure of these bacteria is confirmed in the AFM amplitude image.
Longitudinal structures along the filaments similar to those observed by Pfeffer et al. (2012) were identified for filamentous bacteria from NO3−-treated sediment, using combined optical and AFM imaging (Figure 5b). These filaments were first hybridized with probe DSB706 to confirm their affiliation with Desulfobulbaceae (Figure 5a).
Discussion
Electric coupling between NO3− reduction and ΣH2S oxidation
The results of this study show that NO3− reduction can sustain the distant oxidation of ΣH2S in marine sediment as previously described for O2 (Nielsen et al., 2010). Exposing the sediment to NO3−-amended anoxic overlying water resulted in the development of geochemical traits typical of electric coupling between distant half-cell reactions such as: (1) development of a 4–7-mm-wide zone devoid of both NO3− and ΣH2S, consistent with the separation of ΣH2S oxidation from the associated NO3− reduction; (2) consumption of protons in the NO3− reduction zone and proton production at the ΣH2S oxidation depth consistent with the presence of cathodic NO3− reduction and anodic ΣH2S oxidation, respectively. Furthermore, sediment that showed the geochemical traits typical of electric coupling between distant half-cell reactions in the oxic treatment maintained those characteristics after O2 was replaced with NO3− (Figure 4). Previous investigations of similar sediment showed a fast rising of the ΣH2S front (up to 5 mm d−1) as a consequence of the interruption of the electric current either by physically disturbing the sediment (Pfeffer et al., 2012) or by removing O2 from the overlying water (Nielsen et al., 2010). The stability of the ΣH2S front combined with the presence of marked proton consumption in the upper sediment indicates that NO3− can efficiently drive the distant oxidation of ΣH2S when O2 is no longer available.
Large sulfur bacteria of genus Beggiatoa are able to transport NO3− from the sediment surface and use it to oxidize the ΣH2S, with the resultant formation of a suboxic zone in marine sediments (for example, Sayama et al. (2005)). We can exclude that Beggiatoa played a significant role in the separation of NO3− and ΣH2S in our incubations. Because of their ability to accumulate NO3− intracellularly to millimolar concentrations (Jørgensen and Nelson, 2004), intracellular NO3− measured in sediment inhabited by these organisms largely exceed the concentration of NO3− dissolved in the porewater (Sayama, 2001; Preisler et al., 2007). In our incubation, the pooled intracellular and porewater NOx− profile agreed with the porewater NOx− microprofile measured by the biomicrosensor, indicating that intracellular NO3− storage was insignificant (the low NO3− concentration values detected below 6 mm depth were likely due to analytical errors or contaminations). Moreover, the activity of Beggiatoa would result in a characteristic pH microprofile with a minimum in the upper sediment and a maximum at the ΣH2S front (Sayama et al., 2005) and our pH microprofiles showed opposite trends.
The reduction of solid Fe and Mn oxides coupled to oxidation of ΣH2S can also result in the development of suboxic zones in sediments. Reactive fractions of Fe and Mn oxides are not expected to be present in our incubations, as the batch sediment was highly sulfidic and bioturbating infauna possibly mediating their formation was absent. In addition, in the anoxic NO3−-free treatment the sediment remained fully sulfidic and the pH did not show defined peaks, confirming that the background concentrations of solid electron acceptors were neither sufficient to generate a ΣH2S-free zone nor to sustain an intense proton-consuming process.
Filamentous Desulfobulbaceae
The presence of filamentous Desulfobulbaceae resembling the cable bacteria described by Pfeffer et al. (2012) in NO3−-amended anoxic sediment and their absence in the NO3−-free anoxic incubations indicates that these organisms can grow using NO3− as an electron acceptor in the absence of O2. The presence of geochemical traits indicative of electric coupling between NO3− reduction and ΣH2S oxidation in NO3−-amended cores with cable bacteria further suggests that these organisms can perform cathodic NO3− reduction and mediate an electric coupling between distant NO3− reduction and ΣH2S oxidation in the absence of O2. However, NO3− seemed to be a much less effective electron acceptor as compared with O2. The current density generated with NO3− acting as electron acceptor was less than 9 mA m−2, which was less than one-third of the current density estimated in parallel oxic incubations (28 mA m−2). Interestingly, only a fraction of all filamentous Desulfobulbaceae (detected by probe DSB706) hybridized with probe ELF645 specifically designed for cable bacteria (Pfeffer et al., 2012). This result indicates that probe ELF645 does not target all cable bacteria and suggests a broader diversity of cable bacteria within the Desulfobulbaceae. This conclusion is supported by the 16S rRNA gene sequence analysis that showed a dominance of diverse Desulfobulbaceae sequences outside the previously described cable bacteria cluster (Supplementary Figure S1). The true extent of the cable phenotype within Desulfobulbaceae remains yet to be defined.
In the present study we have demonstrated that cathodic NO3− reduction can sustain the distant oxidation of ΣH2S in marine sediment as previously described for O2, and that cable bacteria are involved. The ability of microorganisms to perform cathodic NO3− reduction has been previously invoked to explain electric current generation in microbial fuel cells (for example, Gregory et al. (2004)) and syntrophic relations in pure cultures in the presence of magnetite (Kato et al., 2012). For these organisms, cathodic NO3− reduction implies electron transfer from a cell to an external electrode or a conductive mineral. For cable bacteria, cathodic NO3− reduction implies the transfer of electrons from distant donors through internal biological structures over millimeter to centimeter distances. The importance of this hitherto unknown mechanism for elemental cycling remains to be addressed.
Acknowledgments
We are grateful to Preben Sørensen, Jesper Voetmann and Jens Kondrup for the technical assistance. Regina Schauer helped with FISH analysis, and Jesper Bjerg helped to extract the filamentous bacteria analyzed by AFM. Financial support was given by SENSEnet ITN (EC Framework Programme 7, PITN-GA-2009-237868) (UM and NPR), the European Research Council (267233 and 291650) (UM, NPR and LPN), the Danish Council for Independent Research (RLM and LPN), the Danish National Research Foundation (NRP) and the German Max Planck Society (NRP).
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies this paper on The ISME Journal website (http://www.nature.com/ismej)
Supplementary Material
References
- Berg P, Risgaard-Petersen N, Rysgaard S. (1998). Interpretation of measured concentration profiles in sediment pore water. Limnol Oceanogr 43: 1500–1510. [Google Scholar]
- Boudreau BD. (1997) Diagenetic Models and their Implementation. Springer-Verlag: Berlin. [Google Scholar]
- Braman RS, Hendrix SA. (1989). Nanogram nitrite and nitrate determination in environmental and biological-materials by vanadium(III) reduction with chemi-luminescence detection. Anal Chem 61: 2715–2718. [DOI] [PubMed] [Google Scholar]
- Dannenberg S, Kroder M, Dilling W, Cypionka H. (1992). Oxidation of H2, organic-compounds and inorganic sulfur-compounds coupled to reduction of O2 or nitrate by sulfate-reducing bacteria. Arch Microbiol 158: 93–99. [Google Scholar]
- Gregory KB, Bond DR, Lovley DR. (2004). Graphite electrodes as electron donors for anaerobic respiration. Environ Microbiol 6: 596–604. [DOI] [PubMed] [Google Scholar]
- Hogslund S, Nielsen JL, Nielsen LP. (2010). Distribution, ecology and molecular identification of Thioploca from Danish brackish water sediments. FEMS Microbiol Ecol 73: 110–120. [DOI] [PubMed] [Google Scholar]
- Jeroschewski P, Steuckart C, Kuhl M. (1996). An amperometric microsensor for the determination of H2S in aquatic environments. Anal Chem 68: 4351–4357. [Google Scholar]
- Jørgensen BB, Nelson DC. (2004). Sulfide oxidation in marine sediments: geochemistry meets microbiology. Geol Soc Am Spec Pap 379: 63–81. [Google Scholar]
- Kato S, Hashimoto K, Watanabe K. (2012). Microbial interspecies electron transfer via electric currents through conductive minerals. Proc Natl Acad Sci USA 109: 10042–10046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kjeldsen KU, Loy A, Jakobsen TF, Thomsen TR, Wagner M, Ingvorsen K. (2007). Diversity of sulfate-reducing bacteria from an extreme hypersaline sediment, Great Salt Lake (Utah). FEMS Microbiol Ecol 60: 287–298. [DOI] [PubMed] [Google Scholar]
- Loy A, Lehner A, Lee N, Adamczyk J, Meier H, Ernst J et al. (2002). Oligonucleotide microarray for 16S rRNA gene-based detection of all recognized lineages of sulfate-reducing prokaryotes in the environment. Appl Environ Microbiol 68: 5064–5081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newman EI. (1966). A method of estimating the total length of root in a sample. J Appl Ecol 3: 139–145. [Google Scholar]
- Nielsen LP, Risgaard-Petersen N, Fossing H, Christensen PB, Sayama M. (2010). Electric currents couple spatially separated biogeochemical processes in marine sediment. Nature 463: 1071–1074. [DOI] [PubMed] [Google Scholar]
- Nielsen M, Gieseke A, de Beer D, Revsbech NP. (2009). Nitrate, nitrite, and nitrous oxide transformations in sediments along a salinity gradient in the Weser Estuary. Aquat Microb Ecol 55: 39–52. [Google Scholar]
- Pfeffer C, Larsen S, Song J, Dong MD, Besenbacher F, Meyer RL et al. (2012). Filamentous bacteria transport electrons over centimetre distances. Nature 491: 218–221. [DOI] [PubMed] [Google Scholar]
- Preisler A, de Beer D, Lichtschlag A, Lavik G, Boetius A, Jorgensen BB. (2007). Biological and chemical sulfide oxidation in a Beggiatoa inhabited marine sediment. ISME J 1: 341–353. [DOI] [PubMed] [Google Scholar]
- Revsbech NP. (1989). An oxygen microsensor with a guard cathode. Limnol Oceanogr 34: 474–478. [Google Scholar]
- Revsbech NP, Glud RN. (2009). Biosensor for laboratory and lander-based analysis of benthic nitrate plus nitrite distribution in marine environments. Limnol Oceanogr Methods 7: 761–770. [Google Scholar]
- Revsbech NP, Jorgensen BB. (1986). Microelectrodes—their use in microbial ecology. Adv Microb Ecol 9: 293–352. [Google Scholar]
- Revsbech NP, Larsen LH, Gundersen J, Dalsgaard T, Ulloa O, Thamdrup B. (2009). Determination of ultra-low oxygen concentrations in oxygen minimum zones by the STOX sensor. Limnol Oceanogr Methods 7: 371–381. [Google Scholar]
- Risgaard-Pedersen N, Rysgaard S, Nielsen LP, Revsbech NP. (1994). Diurnal-variation of denitrification and nitrification in sediments colonized by benthic microphytes. Limnol Oceanogr 39: 573–579. [Google Scholar]
- Risgaard-Petersen N, Langezaal AM, Ingvardsen S, Schmid MC, Jetten MSM, Op den Camp HJM et al. (2006). Evidence for complete denitrification in a benthic foraminifer. Nature 443: 93–96. [DOI] [PubMed] [Google Scholar]
- Risgaard-Petersen N, Revil A, Meister P, Nielsen LP. (2012). Sulfur, iron-, and calcium cycling associated with natural electric currents running through marine sediment. Geochim Cosmochim Ac 92: 1–13. [Google Scholar]
- Sayama M. (2001). Presence of nitrate-accumulating sulfur bacteria and their influence on nitrogen cycling in a shallow coastal marine sediment. Appl Environ Microbiol 67: 3481–3487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sayama M, Risgaard-Petersen N, Nielsen LP, Fossing H, Christensen PB. (2005). Impact of bacterial NO3- transport on sediment biogeochemistry. Appl Environ Microbiol 71: 7575–7577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schauer R, Risgaard-Pedersen N, Kjeldsen KU, Tataru Bjerg JJ, Jorgensen BB, Schramm A et al. (2014). Succession of cable bacteria and electric currents in marine sediment. ISME J e-pub ahead of print 23 January 2014; doi:10.1038/ismej.2013.239. [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.