Abstract
We previously reported that Rv1860 protein from Mycobacterium tuberculosis stimulated CD4+ and CD8+ T cells secreting gamma interferon (IFN-γ) in healthy purified protein derivative (PPD)-positive individuals and protected guinea pigs immunized with a DNA vaccine and a recombinant poxvirus expressing Rv1860 from a challenge with virulent M. tuberculosis. We now show Rv1860-specific polyfunctional T (PFT) cell responses in the blood of healthy latently M. tuberculosis-infected individuals dominated by CD8+ T cells, using a panel of 32 overlapping peptides spanning the length of Rv1860. Multiple subsets of CD8+ PFT cells were significantly more numerous in healthy latently infected volunteers (HV) than in tuberculosis (TB) patients (PAT). The responses of peripheral blood mononuclear cells (PBMC) from PAT to the peptides of Rv1860 were dominated by tumor necrosis factor alpha (TNF-α) and interleukin-10 (IL-10) secretions, the former coming predominantly from non-T cell sources. Notably, the pattern of the T cell response to Rv1860 was distinctly different from those of the widely studied M. tuberculosis antigens ESAT-6, CFP-10, Ag85A, and Ag85B, which elicited CD4+ T cell-dominated responses as previously reported in other cohorts. We further identified a peptide spanning amino acids 21 to 39 of the Rv1860 protein with the potential to distinguish latent TB infection from disease due to its ability to stimulate differential cytokine signatures in HV and PAT. We suggest that a TB vaccine carrying these and other CD8+ T-cell-stimulating antigens has the potential to prevent progression of latent M. tuberculosis infection to TB disease.
INTRODUCTION
Although only 5 to 10% of those infected with Mycobacterium tuberculosis progress to disease, depending upon their HIV status, the annual incidence of new cases of tuberculosis (TB) runs into millions due to more than 2 billion infected individuals worldwide (1). The lifetime risk of an M. tuberculosis-infected HIV-negative individual progressing to TB disease is a mere 10%, testifying to the success of immune mechanisms in containing the tubercle bacteria and maintaining them in a persistent state (2). The prophylactic management and control of tuberculosis have relied for the past several decades on the live attenuated strain, Mycobacterium bovis BCG, which showed variable efficacy in trials worldwide in the post-Second World War era (3). However, despite extensive investigations into the immunology of tuberculosis, the requirements for protective immunity in the host and the bacterial components that trigger such protective immune responses are poorly understood (4–6), which in turn has stalled the development of efficacious new vaccines.
The need to find improved vaccines for TB has become all the more pressing following the discouraging results from the human phase 2b trials of MVA85A (7–10), the most advanced among the 12 candidate vaccines that are undergoing human clinical trials, despite encouraging results in animal models. Development of effective TB vaccines is constrained by the lack of immune correlates of protection in humans and reliable animal models. While gamma interferon (IFN-γ) was long believed to be a correlate of protective immune responses against M. tuberculosis, based primarily on the increased susceptibility to TB of mice lacking IFN-γ (4, 11), several recent publications have demonstrated the absence of such a correlation (12–17). Polyfunctional T (PFT) cells capable of simultaneously secreting multiple TH1 cytokines were shown to confer protection against TB in the mouse model (18, 19); however, studies in humans revealed conflicting results. Several studies showed that human CD4+ PFT cells specific to the secreted antigens ESAT-6, CFP-10, 16-kDa protein, Ag85A, and Ag85B correlated with the bacterial antigen burdens (20–23); not only did these responses wane with chemotherapy (21, 23) but also T cell responses to Rv3873 and Rv3878 from within the RD1 locus in fact predicted progression to active disease (24). These observations led to the suggestion that PFT cells may at best serve as a useful biomarker of active tuberculosis and cure and may have no role in protection. However, in a South African cohort, PFT cells were more numerous in healthy latently infected volunteers (HV) than in TB patients (PAT) and 6 months of chemotherapy led to a resurgence of PFT cells (25). The contrasting results were attributed to differences in methodology, antigens tested, and cohort characteristics. The majority of these investigations with the exception of the South African study (25) queried only CD4+ and did not include CD8+ T cell responses. Clearly, correlates of protective immunity against TB in humans which are useful in reliably identifying potential vaccine candidates are yet to be discovered.
CD8+ T cells are increasingly being recognized for their contribution to TB control in mice (26–28), nonhuman primates (29), and humans (30–32). The increased susceptibility to TB of patients with rheumatoid arthritis and ankylosing spondylitis under infliximab treatment to neutralize tumor necrosis factor alpha (TNF-α) was shown to be due to the loss of the CD8+ TEMRA (effector memory T cells reexpressing CD45RA) cell subset which displayed antimicrobial activity against M. tuberculosis (33). This CD8+ TEMRA subset was in fact reported to be deficient in TB patients in contrast to latently infected healthy controls (34). However, very few antigens of M. tuberculosis with the ability to stimulate human CD8+ T cells have been identified (30). We initially identified Rv1860 from a screen of 24 recombinant proteins obtained from a genomic DNA expression library of M. tuberculosis (35) for its ability to elicit proliferation and IFN-γ secretion from both CD4+ and CD8+ T cells of healthy latently infected individuals and for its ability to protect guinea pigs against a challenge with a virulent field strain of M. tuberculosis (36; our unpublished observations). Rv1860 is a well-characterized secreted glycoprotein of M. tuberculosis and M. bovis BCG; the BCG homologue was first identified as a proline-rich culture filtrate protein (37, 38) that was immunogenic in infected guinea pigs. Elegant analyses of the glycosylation moieties of the purified 45-kDa culture filtrate-derived M. tuberculosis Rv1860 protein revealed that the threonine residues at positions 10, 18, 27, and 277 were glycosylated, and the attached carbohydrates were single mannose, mannobiose, or mannotriose units strung together by α-linkages (39, 40). We earlier reported that the glycosylated form of Rv1860 inhibited the T-cell-polarizing functions of mouse bone marrow-derived dendritic cells (41). In this study, we report that peptides derived from the sequence of Rv1860 stimulated human PFT cell responses, which were dominated by CD8+ T cells in contrast to the CD4+ T-cell-dominated responses to the well-studied antigens ESAT-6, CFP-10, Ag85A, and Ag85B. Several subsets of Rv1860-specific polyfunctional CD4+ and CD8+ T cells were significantly more numerous in HV than in PAT, in contrast to the reported superior CD4+ T cell responses to ESAT-6, CFP-10, Ag85A, and Ag85B in TB patients (21–23, 42). Our results suggest that Rv1860, by virtue of its capacity to stimulate CD8+ T cells, may serve as a useful candidate for inclusion in a TB vaccine with the potential for preventing the reactivation of latent M. tuberculosis infections which accounts for up to 80% of TB cases in some countries (43). We also identified a peptide spanning amino acids (aa) 21 to 39 of the Rv1860 protein sequence that gave rise to a mutually exclusive proliferation and cytokine signature from stimulated peripheral blood mononuclear cells (PBMC) of HV and PAT, revealing the potential for its use for evaluating new therapeutic agents and for monitoring progression from TB latency to disease.
MATERIALS AND METHODS
Study subjects.
Individuals presenting at the outpatient department of M. S. Ramiah Hospital, Bangalore, India, and diagnosed with pulmonary tuberculosis based on the presence of culturable acid-fast bacilli in sputum were recruited to participate in this study and included 17 males (mean age, 47 ± 8 years; mean weight, 54 ± 6 kg) and 3 females (mean age, 37 ± 6 years; mean weight, 45 ± 5 kg) (see Table S2 in the supplemental material). The diagnosis of pulmonary TB was routinely confirmed by sputum culture on Lowenstein-Jensen slants at the National Tuberculosis Institute. Blood samples were collected once from TB patients (PAT) before chemotherapy was initiated. Thirty healthy purified protein derivative (PPD)-positive volunteers, judged to be latently infected with M. tuberculosis (HV), based on the release of ≥0.7 IU of IFN-γ (1 IU of IFN-γ = 50 pg/ml) following 24 h of stimulation of whole blood with a pool of peptides covering the amino acid sequences of ESAT-6 and CFP-10, with a minimum induration reading of 9 mm in the Mantoux test (1 tuberculin unit of RT-23 from the Statens Serum Institute, Copenhagen, Denmark) (44), were recruited at the Indian Institute of Science and National Tuberculosis Institute, Bangalore, India. Unstimulated control blood samples did not produce detectable IFN-γ. Twenty-eight individuals completed the study (HV, 19 males [mean age of 41 ± 7 years and mean weight of 57 ± 7 kg] and 9 females [mean age of 39 ± 6 years and mean weight of 47 ± 4 kg]) (see Table S2 in the supplemental material). The exclusion criteria comprised fever, malaise, clinical symptoms of TB (verified by chest roentgenogram and sputum smears for acid-fast bacilli), and positive tests for HIV and hepatitis B. A group of 5 healthy PPD-negative (induration diameter of <5 mm) volunteers with no IFN-γ response to ESAT-6+ or CFP-10 peptides were available as controls. This study was approved by the Institutional Human Ethics Committees of the Indian Institute of Science (IISc) and the Ethical Review Board of M. S. Ramiah (MRS) Hospital. Signed informed consent was obtained from all individuals prior to enrollment in the study after the purpose and consequences of the study were fully explained.
Isolation of PBMC and T cell proliferation assays.
PBMC isolated from heparinized blood by the Ficoll-Hypaque density gradient method (45) were cultured (1.5 × 105 cells per well) in triplicate with peptides at a concentration of 2 μg/ml for 5 days. Stimulation with the mitogen phytohemagglutinin A (PHA) (Sigma, St. Louis, MO) at 10 μg/ml was carried out for 3 days. Lymphocyte proliferation was measured by the incorporation of [3H]thymidine (DuPont NEN, Boston, MA) added at 0.5 μCi per well for the final 18 to 20 h of culture. Incorporated radiolabel in harvested cells was quantitated, and the proliferative response was expressed as the stimulation index, SI = (mean counts per minute [46] of test antigen-stimulated cultures)/(mean counts per minute of unstimulated cultures in triplicate wells), as described earlier (36).
A positive response to the Rv1860 peptides was scored on the criterion that the SI was ≥2.5 and the average counts per minute obtained on stimulation with recombinant antigen was ≥500 along with a significant increase in the counts per minute induced by the test antigen over that of the unstimulated control as ascertained by the Student t test. We recovered comparable numbers (1 to 2 × 106 PBMC per ml) of PBMC from the blood samples of TB patients and PPD-positive healthy donors.
Antigens.
Peptides of 20-mer length spanning the entire length of the Rv1860 protein and overlapping with each other by 10 amino acids were obtained from Genemed Synthesis Inc., TX, USA (see Table S1 in the supplemental material). ESAT-6 (NR-14868), CFP-10 (NR-14869), Ag85A (NR-14871), and Ag85B (NR-14870) were obtained from the Biodefense and Emerging Infections (BEI) Resources of the American Type Culture Collection.
Quantitation of cytokines by ELISA.
IFN-γ, interleukin-2 (IL-2), IL-10, and TNF-α were quantitated by enzyme-linked immunosorbent assays (ELISAs) using commercially available antibody pairs (R&D Systems) according to the manufacturer's instructions. IFN-γ was measured in 72-h PBMC culture supernatants, whereas all other cytokines were measured using 48-h culture supernatants. The lower limit of detection for all the cytokines was 15 pg/ml.
Intracellular cytokine detection.
Whole, heparinized (sodium heparin) blood samples were diluted 1:1 with RPMI 1640, and 1-ml aliquots were stimulated with peptides, each at a concentration of 2.5 μg/ml in 13-ml tubes for 18 h as described previously (47). Data were acquired on a BD FACSCanto II flow cytometer (Becton Dickinson, San Jose, CA). For details of the protocol and analysis, refer to the supplemental material.
Small lymphocytes were collected by gating on forward scatter versus side scatter (SSC) and then gated on singlet cells, followed by CD3high T lymphocytes (see Fig. S1A in the supplemental material). We excluded dead cells (verified by separate live/dead staining to be <0.01% of CD3+ T cells) by avoiding cells high on SSC and those close to the y axis, which was feasible as all staining was done on fresh PBMC. CD3high/CD8− cells were considered CD4+ T cells. For each analysis, a minimum of 100,000 CD4+/CD8+ T cell subsets were acquired, and data were analyzed using FlowJo (Treestar), PESTLE, and SPICE (Mario Roederer, NIH, USA) software. Positive staining was affirmed by comparing the dot plots of the antibody-stained unstimulated and antigen-stimulated cells and a minimum number of 50 events was used as the cutoff for a positive response. Gates were positioned to ensure that the percentages of fully stained unstimulated cells were ≤0.01% of total CD4+ or CD8+ T cells for IFN-γ-, TNF-α-, and IL-2-secreting T cells, while it was ≤0.05 for MIP-1β-secreting T cells.
HLA restriction analysis.
The alleles of HLA-A, HLA-B, and HLA-DRB1 loci were studied using PCRs followed by hybridization with sequence-specific oligonucleotide probes (SSOPs) as described earlier (48), using a bead-based technology (Luminex, Austin, TX) and following the manufacturer's instructions (LABType SSO kit from One Lambda, Canoga Park, CA, USA). The latest nomenclature for the HLA system was used to designate the alleles of the three loci studied (49). The data acquired were analyzed using LABType software (One Lambda) for analysis of HLA alleles.
Statistical analysis.
The results of the lymphoproliferative assays with human PBMC are presented as mean SI values from triplicate wells of a 96-well plate. The results of the ELISAs are expressed as means of duplicate wells in picograms per milliliter. Cytokine responses (IL-2, IL-6, IL-10, IFN-γ, and TNF-α) and SI values (see Fig. 2) were compared between HV and PAT with the Student t test and the Mann-Whitney test using GraphPad Prism, version 4.0 for Windows (GraphPad Software, San Diego, CA, USA). Bonferroni's correction was applied for these six comparisons and gave a critical value for each test of 0.05/6 = 0.0083. Where significant differences are shown, both tests confirmed this result and the more conservative values are indicated. The age, weight, and sex trends for the SI values and cytokine responses were analyzed using logistic regression and were found not to significantly influence the outcome. The ages and weights of males and females were compared using a 2-tailed Student t test (see Table S2 in the supplemental material). For comparing the percentages of polyfunctional T cell subsets between HV and PAT, we used the nonparametric Wilcoxon test available within SPICE on log-transformed data. We then applied Bonferroni's correction for multiple comparisons between HV and PAT or between CD4+ and CD8+ T cells of the 15 different CD4+ or CD8+ T cell subsets secreting different combinations of four different cytokines (IL-2, IFN-γ, MIP-1β, and TNF-α), giving a calculated critical value for each individual comparison of 0.05/15 = 0.0033; P values less than this were considered significant. For analysis of the association of HLA alleles and cytokine gene polymorphisms, Fisher's exact test or the chi-square test was used wherever suitable. The central limit theorem for proportions and Fisher's exact test were used to compare the proportions of men and women between the HV and PAT groups (see Table S2 in the supplemental material) and to compare the frequency of specific HLA alleles between the two groups.
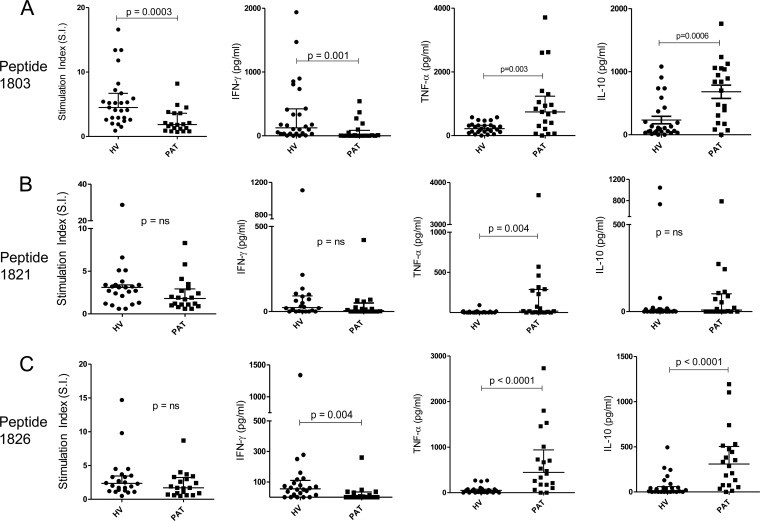
FIG 2.
Proliferation and cytokine secretions by PBMC in response to immunodominant peptides from Rv1860. (A) Peptide 1803; (B) peptide 1821; (C) peptide 1826. Twenty-eight HV and 20 PAT were compared. The P values for significant differences between the two groups were computed using Mann-Whitney tests; the P values that did not remain significant after Bonferroni's correction are indicated as ns (not significant).
RESULTS
Rv1860 peptides stimulate recall T cell responses in M. tuberculosis-infected individuals.
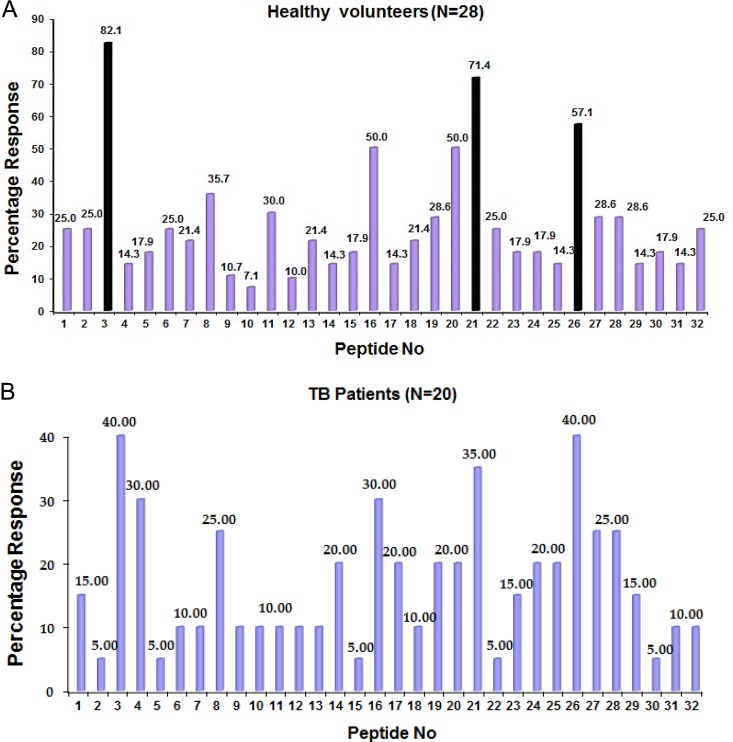
In our initial screen of 24 recombinant M. tuberculosis proteins, we observed proliferation and IFN-γ secretion by human PBMC in response to Rv1860 (36). We therefore investigated in detail the proliferative and cytokine responses of human PBMC to a panel of 32 20-mer peptides spanning the complete sequence of Rv1860 (shown at the bottom of Table S1 in the supplemental material) and overlapping by 10 amino acids (see Table S1). Twenty-eight healthy latently infected individuals (see Table S2 in the supplemental material) out of the 30 volunteers recruited were tested for proliferative responses of PBMC to the 32 overlapping peptides from Rv1860 (Fig. 1A). Two volunteers did not show a proliferative response to any of the peptides. One individual responded to only 3 peptides, while 2 responded to only 4 peptides. PBMC from all other latently infected healthy individuals showed proliferative responses to multiple peptides, while one individual unusually responded to 23 out of the 32 overlapping peptides from Rv1860. Of the total 203 proliferative responses obtained, the majority had SI values between 2.5 and 5, while 38 responses had SI values between 5 and 20, 13 of which were to peptide 1803. Out of 20 PAT (see Table S2) tested, 16 responded by proliferation of PBMC to at least 3 peptides each. Only two patients had SI values between 5 and 10 to 8 different peptides, while all other responses fell between 2.5 and 4.5; however, all 20 patients produced cytokines. Figure 1 summarizes the frequency of the proliferative responses to each peptide among the 28 HV and 20 PAT tested. PBMC from all volunteers secreted substantial levels (between 100 and 2,000 pg/ml) of one or more cytokines (IFN-γ, TNF-α, IL-10, or IL-6) in response to at least one peptide, measured by ELISAs; the cytokine profiles in patients were dominated by TNF-α and IL-10 (Fig. 2).
FIG 1.
Percentages of volunteers whose PBMC showed proliferative responses to individual 20-mer peptides derived from Rv1860 among 28 latently M. tuberculosis-infected healthy volunteers (HV) (A) and 20 TB patients (PAT) (B). The three most immunodominant peptides, 1803, 1821, and 1826, are highlighted in panel A.
Peptides 1803, 1821, and 1826 (see Table S1 in the supplemental material) elicited the largest frequency of proliferative responses in both HV and PAT (Fig. 1). The analysis of the cytokine responses to these 3 peptides revealed a distinct pattern of response to each (Fig. 2). While 1803 stimulated significantly greater proliferative (5.511 ± 0.7699 for HV and 2.390 ± 0.4215 for PAT; P = 0.0003) and IFN-γ responses in HV (336.6 ± 90.43 for HV and 77.78 ± 33.72 for PAT; P = 0.001), it elicited significantly more TNF-α (245.2 ± 31.92 for HV and 954.7 ± 220.6 for PAT; P = 0.003) and IL-10 (232.7 ± 59.02 for HV and 680.9 ± 105.8 for PAT; P = 0.0006) responses in PAT (Fig. 2A). Peptide 1821 stimulated significantly more TNF-α secretion from PAT (Fig. 2B). In response to 1826, however, HV secreted significantly greater amounts of IFN-γ, while PAT secreted significantly greater amounts of TNF-α and IL-10 (Fig. 2C). PPD-negative controls did not secrete measurable levels of cytokines, although we detected low levels of IL-10 in one sample in response to peptide 1803 (data not shown). IL-2 and IL-6 did not display significant differences between HV and PAT (data not shown). Thus, the responses to peptides 1803 and 1826 were capable of distinguishing latently infected healthy individuals from TB patients.
Rv1860 peptides stimulate human polyfunctional T cells.
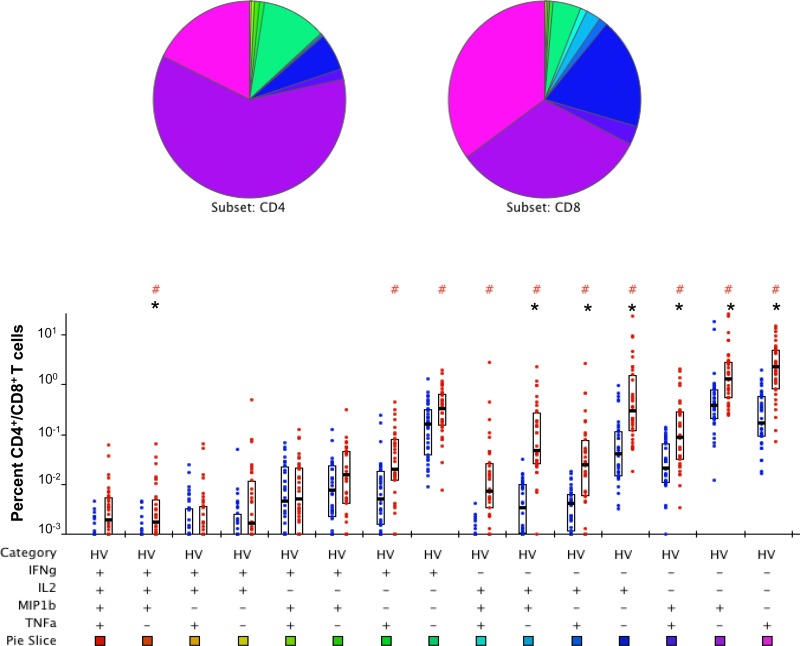
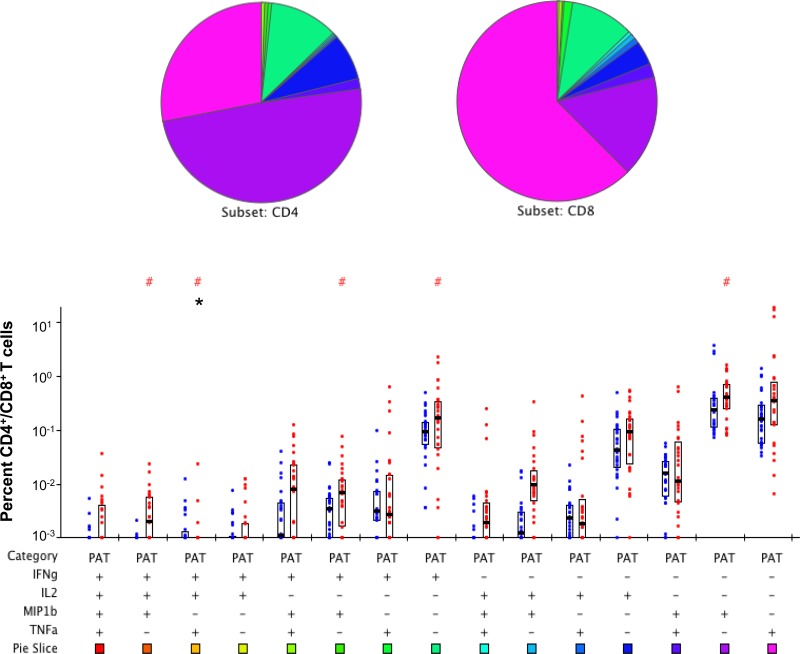
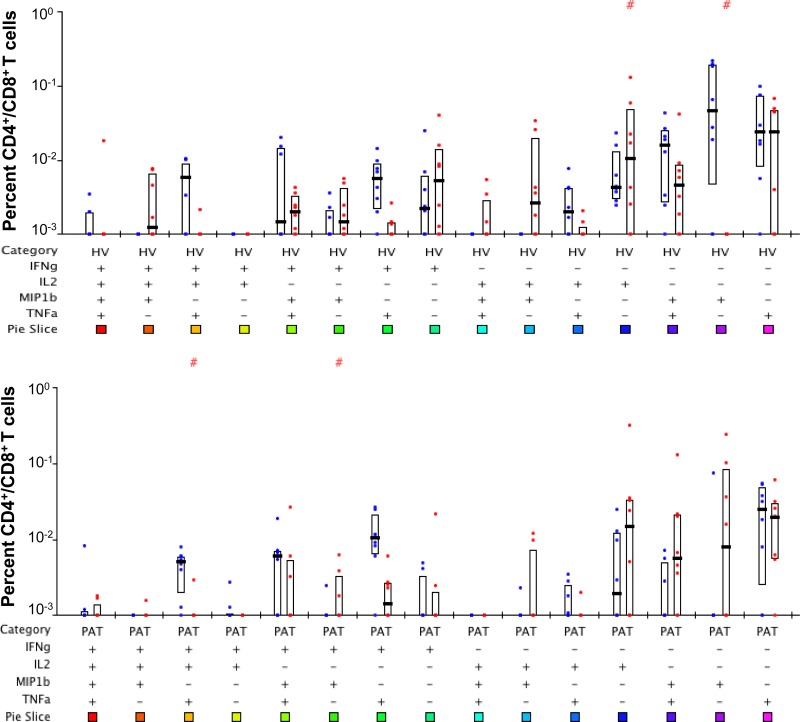
We then queried the subset of T cells responsible for the secretion of the cytokines observed above in HV and PAT using the gating strategy for flow cytometry shown in Fig. S1A in the supplemental material. A mixture of the three immunodominant peptides (1803, 1821, and 1826) from Rv1860 stimulated robust PFT cells secreting all combinations of IFN-γ, IL-2, TNF-α, and MIP-1β in HV (Fig. 3; see also Fig. S1B in the supplemental material) and also in PAT (Fig. 4). The striking feature of the Rv1860 T cell response was the dominant role for CD8+ T cells (right bar in each pair), which secreted a variety of cytokine combinations in both HV (Fig. 3) and PAT (Fig. 4), with several subsets reaching statistical significance. After Bonferroni's correction, while the difference in seven different T cell subsets remained statistically significant in HV (Fig. 3, asterisks), only the subset secreting a combination of IL-2, IFN-γ, and TNF-α showed a significantly greater frequency of CD4+ T cells in PAT (Fig. 4, asterisks), perhaps due to the overall lower responses observed. The pie chart (Fig. 3) revealed that in HV, Rv1860-specific memory T cells secreting IL-2 alone made up a greater proportion of the CD8+ than the CD4+ populations, a feature missing in PAT (Fig. 4). This feature is also evident in the pie chart comparing the CD8+ T cell subsets between HV and PAT (see Fig. S3 in the supplemental material). Single TNF-α and IFN-γ secretors made up a larger proportion of CD8+ T cells in PAT than in HV, while single IL-2 and MIP-1β secretors made up a larger proportion of CD8+ T cells in HV than in PAT (see the pie chart in Fig. S3). However, with the exception of IFN-γ, the frequencies of the other three single-cytokine-secreting CD8+ T cells along with those secreting a combination of IL-2 and MIP-1β were significantly greater in HV (see Fig. S3, asterisks). Overall, several cytokine-secreting combinations were significantly overrepresented in HV compared to those in PAT in both CD4+ and CD8+ T cell subsets (see Fig. S2 and S3, respectively) to support our previously reported predominance of T cell responses specific to Rv1860 in healthy latently infected individuals compared to those in TB patients (36). It is to be noted that the high levels of TNF-α in stimulated PBMC culture supernatants from PAT as detected by ELISAs were not confirmed by the flow cytometric analysis of T cells, pointing to myeloid cells, presumably neutrophils and monocyte/macrophages, as the major source of this inflammatory cytokine in PAT, which corroborates the neutrophil-dependent inflammatory signature reported in TB patients (50).
FIG 3.
Polychromatic flow cytometry analysis of cytokine production profiles of Rv1860-derived peptide-specific T cells from latently M. tuberculosis infected volunteers (HV). Whole-blood ICC samples stimulated with a mixture of peptides 1803, 1821, and 1826 were stained and analyzed as described in Materials and Methods. Percentages of CD4+ (left bar in each pair) were compared with those of CD8+ (right bar in each pair) T cells secreting the different combinations of cytokines IFN-γ, IL-2, TNF-α, and MIP-1β indicated below the pairs of bars as + or − signs. The boxes represent the 25th and 75th percentile values, and the bars denote medians. Dots represent individual values. #P values computed by the nonparametric Wilcoxon test for significant differences between CD4+ and CD8+; *significant P values below the Bonferroni-corrected critical value of 0.05/15 = 0.0033.
FIG 4.
Polychromatic flow cytometry analysis of cytokine production profiles of Rv1860-derived peptide-specific T cells in TB patients (PAT). Whole-blood ICC samples stimulated with a mixture of peptides 1803, 1821, and 1826 were stained and analyzed as described in Materials and Methods. Percentages of CD4+ (left bar in each pair) were compared with those of CD8+ (right bar in each pair) T cells secreting the different combinations of cytokines IFN-γ, IL-2, TNF-α, and MIP-1β indicated below the pairs of bars as + or −. The boxes represent the 25th and 75th percentile values, and the bars denote medians. Dots represent individual values. #P values computed by the nonparametric Wilcoxon test for significant differences between CD4+ and CD8+; *significant P values below the Bonferroni-corrected critical value of 0.05/15 = 0.0033.
As a comparison, we also performed PFT cell analysis in our cohort in response to the well-studied antigens ESAT-6, CFP-10, Ag85A and Ag85B of M. tuberculosis. As evident from Fig. 5 (see also Fig. S4, S5, and S6 in the supplemental material, the latter two showing average values from all four antigens), for the subsets where the cytokine-secreting pattern differed significantly between CD4+ and CD8+ T cells, both T cell types were equally represented, in contrast to the CD8+ T-cell-dominated response to Rv1860 peptides. However, following Bonferroni's correction for multiple comparisons, only three of these P values remained significant.
FIG 5.
Polyfunctional human T cell response to secreted antigen ESAT-6. The plots show comparisons of frequencies of CD4+ (left bar in each pair) with CD8+ (right bar in each pair) T cells in latently infected HV (top panel) and PAT (bottom panel) secreting different combinations of the cytokines IFN-γ, IL-2, TNF-α, and MIP-1β indicated below the pairs of bars as + or −. #P values for significant differences between HV and PAT computed by the nonparametric Wilcoxon test available within SPICE. None of the values were significant following Bonferroni's correction for multiple comparisons.
HLA alleles associated with response to Rv1860.
We carried out typing for the alleles of HLA-A, HLA-B, and HLA-DRB1 loci for all HV and PAT (see Tables S3A and S3B in the supplemental material). We found the HLA-DRB1*15:02 allele in 9 of the 30 HV (0.3) initially recruited for the study, while only 1 of the 20 PAT (0.05) carried this allele (highlighted in Tables S3A and S3B; P = 0.03). No data on the epitopes from Rv1860, validated by immunoassays for binding to HLA alleles, were available in the Immune Epitope Database (IEDB) (http://www.iedb.org/); however, there were several epitopes predicted to bind several HLA class I and II alleles with low 50% inhibitory concentration (IC50) (nanomolar) values. We additionally used the epitope prediction software programs SYFPEITHI and BIMAS to predict the epitopes binding to HLA class I and II alleles from the 3 immunodominant peptides (1803, 1821, and 1826) and from the full-length Rv1860 protein. All three prediction methods identified the same epitopes from both the 20-mer peptide and the full-length Rv1860 protein sequences, and the predictions broadly concurred with one another. As expected, additional 15-mer major histocompatibility complex (MHC) class II epitopes that were not available in the peptides were predicted from the full-length protein, since the overlap between any two consecutive peptides was only 10 amino acids.
We compared the predicted scores for HLA allele binding to the epitopes from the 3 immunodominant peptides to the experimentally detected PBMC responses. We saw no correlation with proliferation (SI values), but strong correlations with the secreted cytokines were evident. SYFPEITHI scores of <20 did not show strong correlations to the experimentally observed responses and were not used for analysis. While MHC molecules are extremely polymorphic, alleles with similar peptide binding pockets may be clustered in one group called supertypes, such that HLA molecules in one supertype may present similar peptides (51). The A02 supertype (51) correlated with a strong response to peptide 1803 (amino acid positions 21 to 40 of Rv1860), to which a very high proportion of HV (22/28) responded as shown by either the SI and/or cytokine secretion. The decamer AMASASLVTV (amino acid positions 22 to 31 of Rv1860) with a predicted SYFPEITHI score of 30 for binding to HLA-A*02:01 along with other nonamers overlapping with this sequence and predicted to bind this HLA allele with scores of 21 and 22 seems to have contributed to this immunodominant response. Additionally, HLA-B*51:01 (B07 supertype) was predicted to bind the nonamer MASASLVTV in peptide 1803 (amino acid positions 23 to 31 of Rv1860) with a score of 25 along with 2 other epitopes with scores of 21. Nineteen of the 22 HV for whom complete data from proliferation and cytokine ELISAs for each of the 32 individual peptides, along with intracellular cytokine (ICC) data for the 3 immunodominant peptides, were available responded to peptide 1803. Ten of these 19 HV carried the HLA-A02 supertype, and 13 HV carried the HLA-B07 supertype, while 8 HV carried both supertype alleles. The levels of cytokines IFN-γ (400 to 2,900 pg/ml) and TNF-α (200 to 700 pg/ml) were 7- to 15-fold higher in the 10 individuals who carried a HLA-A02 supertype allele than in those with HLA-B*51:01 and other B07 supertype alleles (80 to 200 pg/ml IFN-γ and 50 to 100 pg/ml TNF-α). Interestingly, BIMAS scores for HLA A02 binding were between 5 and 10, while those for HLA-B*51:01 binding were between 110 and 170 for nonamers from peptide 1803.
One individual (LAT3) with HLA-A*02:11 and -68:01 and HLA-B*51:01 and -40:04 (see Table S3A in the supplemental material) responded strongly to peptide 1803 and weakly to peptide 1826, with no response whatsoever to any other peptide, confirming that the HLA-A02 supertype restricted a strong response to peptide 1803. LAT4 with HLA-A*11:01 (A03 supertype) but lacking both A02 and B07 supertype alleles had a weak response to peptide 1803, in keeping with the predicted score of 20 for HLA-A*11:01 binding to decamer epitope ASLVTVAVPA (aa 26 to 35 of Rv1860) within peptide 1803. The above predictions matched with alleles predicted by the IEDB; all top 20 alleles with percentile ranks of 0.2 to 3.0 predicted to bind 10 or 9 mers from the peptide 1803 sequence belonged to either supertype A02 or B07, similar to results with SYFPEITHI and BIMAS. Eleven out of the top 17 had IC50 values between 3 and 49 nM, while 6 values were between 55 and 957 nM. Thus, peptides from 1803 had very high predicted binding affinity for their cognate alleles; the IC50 (nanomolar) values predicted for peptides 1821 and 1826 were higher and reflected the much lower levels of IFN-γ, IL-2, TNF-α, and IL-6 secreted by PBMC in response to peptides 1821 and 1826.
The response to peptide 1821 (amino acids 201 to 220 of Rv1860) was also dominated by the HLA-A02 supertype alleles. HLA-A*02:01 was predicted to bind the nonamer RINQETVSL (amino acid 204 to 212 of Rv1860) with a SYFPEITHI score of 25 but had poor scores in BIMAS, whereas for binding of HLA-B*27:05 to the decamer TRINQETVSL (amino acid 203 to 212 of Rv1860), with a score of 24 in SYFPEITHI, the BIMAS score was 200. The HLA-B27 supertype was poorly represented in our cohort with only one individual (LAT1) who carried a B27 supertype allele and not experimentally respond to peptide 1821. Peptide 1821-specific responses were weaker than those to peptides 1803 and 1826, reflecting the lower SYFPEITHI scores for binding of A02 supertype alleles to epitopes from the sequence of 1821. In the IEDB predictions, HLA-A*68:02 (A02 supertype) and HLA-A*32:01 (A01 supertype) occupied the top 3 percentile ranks of 0.15, 0.4, and 0.8 with IC50 values between 4 and 33 nM; neither of these alleles was present in our cohort. Although 5 HLA-B alleles were in the top 10, all except one were poor binders with IC50 values between 2,000 and 14,000 nM.
The response to peptide 1826 (amino acids 251 to 270 of Rv1860) was predicted to be solely due to presentation by HLA-B alleles. The predicted binding of HLA-B*07:02 to the decamer PPQRWFVVWL (aa 255 to 264) with score 23, of HLA-B*51:01 (B07 supertype) to the nonamer GPPQRWFVV (aa 254 to 262) with score 22 and octamer PPQRWFVV (aa 255 to 262) with score 23, of HLA-B*35:01 (B07 supertype) binding to the decamer PPQRWFVVWL (aa 255 to 264) with score 22, and of HLA-B*53:01 binding to the decamer APDAGPPQRW (aa 250 to 259) with score 25 explained the experimentally observed response to 1826. However, the secreted cytokine levels were higher in volunteers with HLA-B*35 and HLA-B*07 alleles than in those with HLA-B*51 alleles.
Of the 13 HV with B07 supertype alleles, 12 showed cytokine responses to peptide 1826 (30 to 350 pg/ml of IFN-γ and TNF-α) as predicted by the 3 different methods used. However, the average levels of cytokines were 5- to 10-fold less than those seen for peptide 1803. LAT12 and LAT21 with HLA-B*07:05 secreted the highest levels of IFN-γ and TNF-α (500 to 1200 pg/ml) in response to peptide 1826; LAT9 with HLA-B*35:03 had a much stronger cytokine response to peptide 1826 than LAT11 and LAT30 with HLA-B*35:01, whereas LAT17 with HLA-B*35:04 had no detectable response to this peptide. LAT25 with HLA-B*07:18 and -51:04 (both alleles with one exact and one key residue pocket match in the B07 supertype) (51) had a weak TNF-α response to peptide 1826. LAT10 with HLA B*51:06 (one exact and one key residue pocket match in the B07 supertype) (51) showed no response whatsoever to peptide 1826. Thus, we observed graded cytokine responses, depending on the exact B07 supertype allele present in the volunteer. Three HV who did not carry any HLA-B07 supertype allele also responded weakly to peptide 1826. The HLA-B*53:01 allele (B07 supertype) which scored the highest for binding epitopes from peptide 1826 using all 3 prediction methods was not represented in our cohort. Thus, broadly for class I alleles, the prediction scores correlated with the magnitudes of the experimentally observed responses. Scrutiny of the flow cytometry data further confirmed that in keeping with the predictions, cytokines were secreted by CD8+ T cells. Thus, Rv1860 appears to sport multiple stretches with HLA class I binding propensity, consistent with its observed strong human CD8+ T-cell-stimulating property.
In contrast, we observed poor correlation between prediction scores and observed responses for HLA class II alleles. None of the 4 volunteers with HLA DRB1-0301 responded with an SI or cytokines to peptides 1816 or 1819, which carried 15-mer epitopes with predicted high-binding SYFPEITHI scores of 34 and 29 (PPPVANDTRIVLGRL [amino acids 156 to 170 of Rv1860] and AARLGSDMGEFYMPY [amino acids 186 to 200 of Rv1860]), respectively. A thorough analysis of the flow cytometry data revealed distinct, but low, cytokine secretion by CD4+ T cells in response to peptide 1826 in individuals carrying the HLA-DRB1*0701 allele, which was, however, not supported by prediction scores.
DISCUSSION
A meaningful understanding of the immunology of tuberculosis has the potential to favorably impact global health through the development of an efficacious vaccine superior to BCG, the sole vaccine currently available, and through informed use of immune parameters as surrogates for monitoring new prophylactics and therapies. To this end, we have attempted to identify the proteins of M. tuberculosis that stimulate stronger TH1-dominated CD4+ and CD8+ T cell responses in healthy latently infected individuals than in TB patients with the simple premise that a measurable response that is greater in healthy people living in areas where TB is endemic but absent or lower in TB patients is highly likely to contribute to preventing the latently infected state from progressing to disease. We show in this study that peptides derived from the sequence of Rv1860, a secreted antigen of M. tuberculosis, which we previously reported to differentially stimulate IFN-γ-secreting CD4+ and CD8+ T cells in healthy M. tuberculosis-exposed volunteers better than in TB patients (36), stimulated human PFT cells secreting various combinations of the four cytokines, IFN-γ, IL-2, TNF-α, and MIP-1β, predominantly by CD8+ T cell subsets. In striking contrast, the human PFT cell responses to the well-studied secreted proteins ESAT-6, CFP-10, Ag85A, and Ag85B of M. tuberculosis were primarily from CD4+ T cells in multiple studies of cohorts from several continents (21–23, 25, 42), a feature common to our cohort also. Additionally, these responses were significantly greater in HV, in contrast to the superior PFT cell responses in TB patients to the secreted proteins ESAT-6, CFP-10, Ag85A, and Ag85B of M. tuberculosis that waned with chemotherapy (21, 23). Interestingly, soluble cytokine measurements from stimulated PBMC revealed an abundance of TNF-α and IL-10 in TB patients, the former not corroborated by the PFT cell analysis, in keeping with the reported neutrophil-dependent inflammatory signature in TB patients (50). The simultaneous secretion of IL-10 by PAT results in a much smaller TH1/TH2 ratio in PAT as reported by other investigators (52).
To the best of our knowledge, Rv1860 represents the first reported protein of M. tuberculosis capable of eliciting a dominant CD8+ human T cell response and constitutes a welcome addition to the highly restricted panel of CD4+ T-cell-eliciting proteins that include ESAT-6, CFP-10, Ag85A and Ag85B, from which all conclusions about the contribution of M. tuberculosis-specific T cells to protection/exacerbation/inflammation in TB disease were hitherto drawn. As a secreted protein of M. tuberculosis, Rv1860 perhaps gains efficient access to the infected cell cytosol from the M. tuberculosis-containing phagosome, allowing for efficient cross-presentation on MHC class I to CD8+ T cells (53, 54). The loss of antituberculosis immunity in rhesus macaques depleted of CD8+ T cells suggests an important role for this T cell subset in human immunity to TB (29), facilitated by the expanded repertoire of CD1 molecules in primates, which present epitopes along with MHC class I to this subset of T cells. We first identified Rv1860 as reactive to TB patient sera from a genomic DNA expression library of a field strain of M. tuberculosis (35), and it represented one of only two human T cell stimulatory proteins among 24 random proteins that we screened. Rv1860, also called APA due to its repeating alanine-proline-alanine motifs, was first reported as a secreted culture filtrate protein of BCG, capable of stimulating both a delayed-type hypersensitive (DTH) response (37) and an antibody response in guinea pigs immunized with live but not killed Mycobacterium bovis BCG (38). The M. tuberculosis homolog coding for a 50- to 55-kDa, 325-amino-acid-long Rv1860 protein (55) was subsequently cloned and expressed in both Mycobacterium smegmatis and Escherichia coli (56).
The multiple subsets of predominantly CD8+ and some subsets of CD4+ T cells specific to Rv1860 were significantly overrepresented in HV than in PAT. CD8+ T cells were shown to be required for controlling the latent phase of M. tuberculosis in mice (28), and their depletion resulted in a 10-fold increase in lung bacterial burden during the latent phase of infection, while having no effect on the acute phase. We may therefore surmise that human Rv1860-specific CD8+ T cells (along with other subsets of CD8+ T cells with other specificities) perhaps contributed to preventing reactivation of latent M. tuberculosis in the latently infected volunteers. In fact, CD8+ T cells specific to M. tuberculosis antigens were also previously reported to be significantly greater in BCG vaccinees relative to TB patients (57). Several studies have reported the protective potential of human M. tuberculosis-specific CD8+ T cells (30–33) by virtue of killing M. tuberculosis-infected cells or M. tuberculosis directly. Despite this, neither their role in protective immunity against TB nor the identities of M. tuberculosis antigens that elicit CD8+ T cells have been investigated in depth. One study (34), which again used the well-studied proteins ESAT-6 and CFP-10, reported that CD8+ TEMRA cells were significantly overrepresented in latently infected human volunteers relative to TB patients whose CD8+ T cells were effector memory T cells (TEM). CD4+ T cells with the dual IFN-γ plus IL-2 cytokine profile were earlier shown in an exhaustive and elegant longitudinal study to progressively increase during TB chemotherapy and dominate the immune response postcure (58). It has been suggested that T cells of this phenotype belong to the effector memory population (59–61). We found that Rv1860-specific CD8+ memory T cells with this dual cytokine secretion profile (along with other IL-2- and IFN-γ-secreting profiles) were more abundant in HV than in PAT (see Fig. S3 in the supplemental material). The ability of these CD8+ T cells to secrete IL-2, a well-established T cell growth factor, would help in their self-perpetuation, and IFN-γ, a cytokine that activates macrophages to eliminate M. tuberculosis would be crucial for protection against TB (4, 11, 62). The Rv1860-specific memory CD8+ T cells secreting only IL-2 also occupied a higher proportion of the total CD8+ response in HV than in PAT (Fig. S3). The critical role of T regulatory (Treg) cells elicited by IL-2 in TB resistance in the rhesus macaque model of pulmonary TB (63) further reinforces the importance of IL-2-secreting T cells in TB control. Of the 4 subsets of CD8+ T cells significantly overrepresented in HV than in PAT, the 2 detected at reasonably high frequency were single cytokine producers (see Fig. S3 in the supplemental material, asterisks).
Several reports have pointed to the role of T cells in TB pathogenesis (for a review, see reference 64). The lung tissue destruction leading to enhanced TB transmission associated with cavitary TB has been directly correlated with CD4+ T cell numbers in HIV-TB-coinfected individuals (65). Again, the exacerbated pulmonary inflammation in PD1 knockout mice infected with M. tuberculosis was alleviated by CD4+ T cell depletion (66). However, unlike those for CD4+ T cells, we have not encountered reports of deleterious effects of CD8+ T cells in TB pathology.
All volunteers, both HV and PAT, responded to at least one peptide by both proliferation and secretion of at least one cytokine. We observed strong correlations between the predicted binding of epitopes from the three immunodominant peptides to HLA class I A and B alleles and the measured levels of secreted cytokines from PBMC of volunteers carrying those alleles. Scrutiny of the flow cytometry data further confirmed cytokine secretions from CD8+ T cells. Thus, peptides from Rv1860 must be presented by a wide array of HLA alleles, as predicted by high scores with epitope prediction software programs such as IEDB, SYFPEITHI, and BIMAS, suggesting that a vaccine carrying Rv1860 would have wide population coverage. The flow cytometry analysis of single peptide-stimulated whole-blood samples demonstrated both CD4+ and CD8+ T cell responses in several individuals, revealing both MHC class I- and II-binding epitopes within single peptides.
The results from the recently concluded human phase 2b trial of MVA85A (7–10) revealed that the currently used mouse and guinea pig animal models cannot reliably predict vaccine efficacy in humans. They also suggest that IFN-γ secretion and the ability to stimulate PFT cells in humans do not correlate with protective efficacy against TB. Transcriptional signatures have been recently suggested as a potentially more reliable biomarker of efficacy for vaccines and therapies (67). While our observations need validation from a larger human cohort study, the capacity of Rv1860 to elicit a memory CD8+ T cell-dominated PFT cell profile, combined with the significantly greater response in HV over PAT that we observed in this pilot study, suggests that vaccines, including this antigen may have the ability to protect against progression of latent M. tuberculosis infection to TB disease. Additionally, the significantly superior proliferation along with the significantly reduced IL-10 and TNF-α secretions by PBMC in response to the Rv1860-derived peptide 1803 in HV over PAT suggests a potential use for this peptide in monitoring the efficacy of TB vaccines and TB therapy as well as progression of latently infected individuals to TB disease.
Supplementary Material
ACKNOWLEDGMENTS
The M. tuberculosis antigens ESAT-6, CFP-10, Ag85A, and Ag85B were obtained from the Biodefense and Emerging Infections (BEI) Resources of the American Type Culture Collection.
This work was supported in part by grant BT/PR4047/Med/14/504/2003 to V.S. and R.R. from the Department of Biotechnology, Government of India.
Funding Statement
This work was funded in part by the Department of Biotechnology, Ministry of Science and Technology (DBT). The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Footnotes
Supplemental material for this article may be found at http://dx.doi.org/10.1128/CVI.00554-15.
REFERENCES
- 1.Lawn SD, Zumla AI. 2011. Tuberculosis. Lancet 378:57–72. doi: 10.1016/S0140-6736(10)62173-3. [DOI] [PubMed] [Google Scholar]
- 2.Hernández-Pando R, Jeyanathan M, Mengistu G, Aguilar D, Orozco H, Harboe M, Rook GAW, Bjune G. 2000. Persistence of DNA from Mycobacterium tuberculosis in superficially normal lung tissue during latent infection. Lancet 356:2133–2138. doi: 10.1016/S0140-6736(00)03493-0. [DOI] [PubMed] [Google Scholar]
- 3.Fine PEM. 1995. Variation in protection by BCG: implications of and for heterologous immunity. Lancet 346:1339–1345. doi: 10.1016/S0140-6736(95)92348-9. [DOI] [PubMed] [Google Scholar]
- 4.Cooper AM, Dalton DK, Stewart TA, Griffin JP, Russell DG, Orme IM. 1993. Disseminated tuberculosis in interferon gamma gene-disrupted mice. J Exp Med 178:2243–2247. doi: 10.1084/jem.178.6.2243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.O'Garra A, Redford PS, McNab FW, Bloom CI, Wilkinson RJ, Berry MPR. 2013. The immune response in tuberculosis. Annu Rev Immunol 31:475–527. doi: 10.1146/annurev-immunol-032712-095939. [DOI] [PubMed] [Google Scholar]
- 6.Torrado E, Cooper AM. 2011. What do we really know about how CD4 T cells control Mycobacterium tuberculosis? PLoS Pathog 7:e1002196. doi: 10.1371/journal.ppat.1002196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Baleta A. 2013. Profile: SATVI—a leading light in tuberculosis vaccine research. Lancet 381:984. doi: 10.1016/S0140-6736(13)60698-4. [DOI] [PubMed] [Google Scholar]
- 8.Bishai W, Sullivan Z, Andersen BRBP. 2013. Bettering BCG: a tough task for a TB vaccine? Nat Med 19:410–411. doi: 10.1038/nm.3153. [DOI] [PubMed] [Google Scholar]
- 9.Gulland A. 2013. Tuberculosis vaccine trial shows disappointing results. BMJ 346:f765. doi: 10.1136/bmj.f765. [DOI] [PubMed] [Google Scholar]
- 10.Tameris MD, Hatherill M, Landry BS, Scriba TJ, Snowden MA, Lockhart S, Shea JE, McClain JB, Hussey GD, Hanekom WA, Mahomed H, McShane H, MVA85A 020 Trial Study Team. 2013. Safety and efficacy of MVA85A, a new tuberculosis vaccine, in infants previously vaccinated with BCG: a randomised, placebo-controlled phase 2b trial. Lancet 381:1021–1028. doi: 10.1016/S0140-6736(13)60177-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Flynn JL, Chan J, Triebold KJ, Dalton DK, Stewart TA, Bloom BR. 1993. An essential role for interferon gamma in resistance to Mycobacterium tuberculosis infection. J Exp Med 178:2249–2254. doi: 10.1084/jem.178.6.2249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Aagaard CS, Hoang TTKT, Vingsbo-Lundberg C, Dietrich J, Andersen P. 2009. Quality and vaccine efficacy of CD4+ T cell responses directed to dominant and subdominant epitopes in ESAT-6 from Mycobacterium tuberculosis. J Immunol 183:2659–2668. doi: 10.4049/jimmunol.0900947. [DOI] [PubMed] [Google Scholar]
- 13.Gallegos AM, van Heijst JWJ, Samstein M, Su X, Pamer EG, Glickman MS. 2011. A gamma interferon independent mechanism of CD4 T cell mediated control of M. tuberculosis infection in vivo. PLoS Pathog 7:e1002052. doi: 10.1371/journal.ppat.1002052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hovav A-H, Mullerad J, Davidovitch L, Fishman Y, Bigi F, Cataldi A, Bercovier H. 2003. The Mycobacterium tuberculosis recombinant 27-kilodalton lipoprotein induces a strong Th1-type immune response deleterious to protection. Infect Immun 71:3146–3154. doi: 10.1128/IAI.71.6.3146-3154.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Leal IS, Smedegård B, Andersen P, Appelberg R. 2001. Failure to induce enhanced protection against tuberculosis by increasing T-cell-dependent interferon-γ generation. Immunology 104:157–161. doi: 10.1046/j.1365-2567.2001.01305.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Majlessi L, Simsova M, Jarvis Z, Brodin P, Rojas M-J, Bauche C, Nouzé C, Ladant D, Cole ST, Sebo P, Leclerc C. 2006. An increase in antimycobacterial Th1-cell responses by prime-boost protocols of immunization does not enhance protection against tuberculosis. Infect Immun 74:2128–2137. doi: 10.1128/IAI.74.4.2128-2137.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Olsen AW, Hansen PR, Holm A, Andersen P. 2000. Efficient protection against Mycobacterium tuberculosis by vaccination with a single subdominant epitope from the ESAT-6 antigen. Eur J Immunol 30:1724–1732. doi:. [DOI] [PubMed] [Google Scholar]
- 18.Derrick SC, Yabe IM, Yang A, Morris SL. 2011. Vaccine-induced anti-tuberculosis protective immunity in mice correlates with the magnitude and quality of multifunctional CD4 T cells. Vaccine 29:2902–2909. doi: 10.1016/j.vaccine.2011.02.010. [DOI] [PubMed] [Google Scholar]
- 19.Lindenstrøm T, Agger EM, Korsholm KS, Darrah PA, Aagaard C, Seder RA, Rosenkrands I, Andersen P. 2009. Tuberculosis subunit vaccination provides long-term protective immunity characterized by multifunctional CD4 memory T cells. J Immunol 182:8047–8055. doi: 10.4049/jimmunol.0801592. [DOI] [PubMed] [Google Scholar]
- 20.Beveridge NER, Price DA, Casazza JP, Pathan AA, Sander CR, Asher TE, Ambrozak DR, Precopio ML, Scheinberg P, Alder NC, Roederer M, Koup RA, Douek DC, Hill AVS, McShane H. 2007. Immunisation with BCG and recombinant MVA85A induces long-lasting, polyfunctional Mycobacterium tuberculosis-specific CD4+ memory T lymphocyte populations. Eur J Immunol 37:3089–3100. doi: 10.1002/eji.200737504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Caccamo N, Guggino G, Joosten SA, Gelsomino G, Di Carlo P, Titone L, Galati D, Bocchino M, Matarese A, Salerno A, Sanduzzi A, Franken WPJ, Ottenhoff THM, Dieli F. 2010. Multifunctional CD4+ T cells correlate with active Mycobacterium tuberculosis infection. Eur J Immunol 40:2211–2220. doi: 10.1002/eji.201040455. [DOI] [PubMed] [Google Scholar]
- 22.Sutherland JS, Adetifa IM, Hill PC, Adegbola RA, Ota MOC. 2009. Pattern and diversity of cytokine production differentiates between Mycobacterium tuberculosis infection and disease. Eur J Immunol 39:723–729. doi: 10.1002/eji.200838693. [DOI] [PubMed] [Google Scholar]
- 23.Young JM, Adetifa IMO, Ota MOC, Sutherland JS. 2010. Expanded polyfunctional T cell response to mycobacterial antigens in TB disease and contraction post-treatment. PLoS One 5:e11237. doi: 10.1371/journal.pone.0011237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dosanjh DPS, Bakir M, Millington KA, Soysal A, Aslan Y, Efee S, Deeks JJ, Lalvani A. 2011. Novel M tuberculosis antigen-specific T-cells are early markers of infection and disease progression. PLoS One 6:e28754. doi: 10.1371/journal.pone.0028754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Day CL, Abrahams DA, Lerumo L, Janse van Rensburg E, Stone L, O'rie T, Pienaar B, de Kock M, Kaplan G, Mahomed H, Dheda K, Hanekom WA. 2011. Functional capacity of Mycobacterium tuberculosis-specific T cell responses in humans is associated with mycobacterial load. J Immunol 187:2222–2232. doi: 10.4049/jimmunol.1101122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mogues T, Goodrich ME, Ryan L, LaCourse R, North RJ. 2001. The relative importance of t cell subsets in immunity and immunopathology of airborne Mycobacterium tuberculosis infection in mice. J Exp Med 193:271–280. doi: 10.1084/jem.193.3.271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Orme IM, Collins FM. 1984. Adoptive protection of the Mycobacterium tuberculosis-infected lung: dissociation between cells that passively transfer protective immunity and those that transfer delayed-type hypersensitivity to tuberculin. Cell Immunol 84:113–120. doi: 10.1016/0008-8749(84)90082-0. [DOI] [PubMed] [Google Scholar]
- 28.van Pinxteren LAH, Cassidy JP, Smedegaard BHC, Agger EM, Andersen P. 2000. Control of latent Mycobacterium tuberculosis infection is dependent on CD8 T cells. Eur J Immunol 30:3689–3698. doi:. [DOI] [PubMed] [Google Scholar]
- 29.Chen CY, Huang D, Wang RC, Shen L, Zeng G, Yao S, Shen Y, Halliday L, Fortman J, McAllister M, Estep J, Hunt R, Vasconcelos D, Du G, Porcelli SA, Larsen MH, Jacobs WR Jr, Haynes BF, Letvin NL, Chen ZW. 2009. A critical role for CD8 T cells in a nonhuman primate model of tuberculosis. PLoS Pathog 5:e1000392. doi: 10.1371/journal.ppat.1000392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cho S, Mehra V, Thoma-Uszynski S, Stenger S, Serbina N, Mazzaccaro RJ, Flynn JL, Barnes PF, Southwood S, Celis E, Bloom BR, Modlin RL, Sette A. 2000. Antimicrobial activity of MHC class I-restricted CD8+ T cells in human tuberculosis. Proc Natl Acad Sci U S A 97:12210–12215. doi: 10.1073/pnas.210391497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Stenger S, Hanson DA, Teitelbaum R, Dewan P, Niazi KR, Froelich CJ, Ganz T, Thoma-Uszynski S, Melian A, Bogdan C, Porcelli SA, Bloom BR, Krensky AM, Modlin RL. 1998. An antimicrobial activity of cytolytic T cells mediated by granulysin. Science 282:121–125. doi: 10.1126/science.282.5386.121. [DOI] [PubMed] [Google Scholar]
- 32.Stenger S, Mazzaccaro RJ, Uyemura K, Cho S, Barnes PF, Rosat JP, Sette A, Brenner MB, Porcelli SA, Bloom BR, Modlin RL. 1997. Differential effects of cytolytic T cell subsets on intracellular infection. Science 276:1684–1687. doi: 10.1126/science.276.5319.1684. [DOI] [PubMed] [Google Scholar]
- 33.Bruns H, Meinken C, Schauenberg P, Härter G, Kern P, Modlin RL, Antoni C, Stenger S. 2009. Anti-TNF immunotherapy reduces CD8+ T cell-mediated antimicrobial activity against Mycobacterium tuberculosis in humans. J Clin Invest 119:1167–1177. doi: 10.1172/JCI38482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rozot V, Vigano S, Mazza-Stalder J, Idrizi E, Day CL, Perreau M, Lazor-Blanchet C, Petruccioli E, Hanekom W, Goletti D, Bart P-A, Nicod L, Pantaleo G, Harari A. 2013. Mycobacterium tuberculosis-specific CD8+ T cells are functionally and phenotypically different between latent infection and active disease. Eur J Immunol 43:1568–1577. doi: 10.1002/eji.201243262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Amara RR, Satchidanandam V. 1996. Analysis of a genomic DNA expression library of Mycobacterium tuberculosis using tuberculosis patient sera: evidence for modulation of host immune response. Infect Immun 64:3765–3771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kumar P, Amara RR, Challu VK, Chadda VK, Satchidanandam V. 2003. The Apa protein of Mycobacterium tuberculosis stimulates gamma interferon-secreting CD4+ and CD8+ T cells from purified protein derivative-positive individuals and affords protection in a guinea pig model. Infect Immun 71:1929–1937. doi: 10.1128/IAI.71.4.1929-1937.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Romain F, Augier J, Pescher P, Marchal G. 1993. Isolation of a proline-rich mycobacterial protein eliciting delayed-type hypersensitivity reactions only in guinea pigs immunized with living mycobacteria. Proc Natl Acad Sci U S A 90:5322–5326. doi: 10.1073/pnas.90.11.5322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Romain F, Laqueyrerie A, Militzer P, Pescher P, Chavarot P, Lagranderie M, Auregan G, Gheorghiu M, Marchal G. 1993. Identification of a Mycobacterium bovis BCG 45/47-kilodalton antigen complex, an immunodominant target for antibody response after immunization with living bacteria. Infect Immun 61:742–750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Dobos KM, Khoo KH, Swiderek KM, Brennan PJ, Belisle JT. 1996. Definition of the full extent of glycosylation of the 45-kilodalton glycoprotein of Mycobacterium tuberculosis. J Bacteriol 178:2498–2506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Dobos KM, Swiderek K, Khoo KH, Brennan PJ, Belisle JT. 1995. Evidence for glycosylation sites on the 45-kilodalton glycoprotein of Mycobacterium tuberculosis. Infect Immun 63:2846–2853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Satchidanandam V, Kumar N, Jumani RS, Challu V, Elangovan S, Khan NA. 2014. The glycosylated Rv1860 protein of Mycobacterium tuberculosis inhibits dendritic cell mediated TH1 and TH17 polarization of T cells and abrogates protective immunity conferred by BCG. PLoS Pathog 10:e1004176. doi: 10.1371/journal.ppat.1004176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wilkinson KA, Wilkinson RJ. 2010. Polyfunctional T cells in human tuberculosis. Eur J Immunol 40:2139–2142. doi: 10.1002/eji.201040731. [DOI] [PubMed] [Google Scholar]
- 43.Shea KM, Kammerer JS, Winston CA, Navin TR, Horsburgh Robert C. 2014. Estimated rate of reactivation of latent tuberculosis infection in the United States, overall and by population subgroup. Am J Epidemiol 179:216–225. doi: 10.1093/aje/kwt246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Chadha V, Challu VK. 2009. The tuberculin skin test, p 176–188. In Tuberculosis, 2nd ed Jaypee Brothers Medical Publishers, New Delhi, India. [Google Scholar]
- 45.Fuss IJ, Kanof ME, Smith PD, Zola H. 2001. Isolation of whole mononuclear cells from peripheral blood and cord blood. Curr Protoc Immunol Chapter 7:Unit 7.1. [DOI] [PubMed] [Google Scholar]
- 46.Mohagheghpour N, Gammon D, Kawamura LM, van Vollenhoven A, Benike CJ, Engleman EG. 1998. CTL response to Mycobacterium tuberculosis: identification of an immunogenic epitope in the 19-kDa lipoprotein. J Immunol 161:2400–2406. [PubMed] [Google Scholar]
- 47.Hanekom WA, Hughes J, Mavinkurve M, Mendillo M, Watkins M, Gamieldien H, Gelderbloem SJ, Sidibana M, Mansoor N, Davids V, Murray RA, Hawkridge A, Haslett PAJ, Ress S, Hussey GD, Kaplan G. 2004. Novel application of a whole blood intracellular cytokine detection assay to quantitate specific T-cell frequency in field studies. J Immunol Methods 291:185–195. doi: 10.1016/j.jim.2004.06.010. [DOI] [PubMed] [Google Scholar]
- 48.Singh A, Sharma P, Kar HK, Sharma VK, Tembhre MK, Gupta S, Laddha NC, Dwivedi M, Begum R, Gokhale RS, Rani R. 2012. HLA alleles and amino-acid signatures of the peptide-binding pockets of HLA molecules in vitiligo. J Investig Dermatol 132:124–134. doi: 10.1038/jid.2011.240. [DOI] [PubMed] [Google Scholar]
- 49.Marsh SGE, Albert ED, Bodmer WF, Bontrop RE, Dupont B, Erlich HA, Fernández-Viña M, Geraghty DE, Holdsworth R, Hurley CK, Lau M, Lee KW, Mach B, Maiers M, Mayr WR, Müller CR, Parham P, Petersdorf EW, Sasazuki T, Strominger JL, Svejgaard A, Terasaki PI, Tiercy JM, Trowsdale J. 2010. Nomenclature for factors of the HLA system, 2010. Tissue Antigens 75:291–455. doi: 10.1111/j.1399-0039.2010.01466.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Berry MPR, Graham CM, McNab FW, Xu Z, Bloch SAA, Oni T, Wilkinson KA, Banchereau R, Skinner J, Wilkinson RJ, Quinn C, Blankenship D, Dhawan R, Cush JJ, Mejias A, Ramilo O, Kon OM, Pascual V, Banchereau J, Chaussabel D, O'Garra A. 2010. An interferon-inducible neutrophil-driven blood transcriptional signature in human tuberculosis. Nature 466:973–977. doi: 10.1038/nature09247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Sidney J, Peters B, Frahm N, Brander C, Sette A. 2008. HLA class I supertypes: a revised and updated classification. BMC Immunol 9:1. doi: 10.1186/1471-2172-9-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Tan YJ, Zhang YB, Feng DY, Xie B, Lin ZY, Chen HG, Li YG. 2004. The change and the clinical significance of peripheral blood Th1/Th2 cells in patients with pulmonary tuberculosis. Zhonghua Jie He He Hu Xi Za Zhi 27:385–389. (In Chinese.) [PubMed] [Google Scholar]
- 53.Mazzaccaro RJ, Gedde M, Jensen ER, van_ Santen HM, Ploegh HL, Rock KL, Bloom BR. 1996. Major histocompatibility class I presentation of soluble antigen facilitated by Mycobacterium tuberculosis infection. Proc Natl Acad Sci U S A 93:11786–11791. doi: 10.1073/pnas.93.21.11786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Schaible UE, Winau F, Sieling PA, Fischer K, Collins HL, Hagens K, Modlin RL, Brinkmann V, Kaufmann SHE. 2003. Apoptosis facilitates antigen presentation to T lymphocytes through MHC-I and CD1 in tuberculosis. Nat Med 9:1039–1046. doi: 10.1038/nm906. [DOI] [PubMed] [Google Scholar]
- 55.Espitia C, Espinosa R, Saavedra R, Mancilla R, Romain F, Laqueyrerie A, Moreno C. 1995. Antigenic and structural similarities between Mycobacterium tuberculosis 50- to 55-kilodalton and Mycobacterium bovis BCG 45- to 47-kilodalton antigens. Infect Immun 63:580–584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Laqueyrerie A, Militzer P, Romain F, Eiglmeier K, Cole S, Marchal G. 1995. Cloning, sequencing, and expression of the apa gene coding for the Mycobacterium tuberculosis 45/47-kilodalton secreted antigen complex. Infect Immun 63:4003–4010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Smith SM, Malin AS, Pauline T, Lukey Atkinson SE, Content J, Huygen K, Dockrell HM. 1999. Characterization of human Mycobacterium bovis bacille Calmette-Guérin-reactive CD8+ T cells. Infect Immun 67:5223–5230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Millington KA, Innes JA, Hackforth S, Hinks TSC, Deeks JJ, Dosanjh DPS, Guyot-Revol V, Gunatheesan R, Klenerman P, Lalvani A. 2007. Dynamic relationship between IFN-γ and IL-2 profile of Mycobacterium tuberculosis-specific T cells and antigen load. J Immunol 178:5217–5226. doi: 10.4049/jimmunol.178.8.5217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Harari A, Vallelian F, Meylan PR, Pantaleo G. 2005. Functional heterogeneity of memory CD4 T cell responses in different conditions of antigen exposure and persistence. J Immunol 174:1037–1045. doi: 10.4049/jimmunol.174.2.1037. [DOI] [PubMed] [Google Scholar]
- 60.Harari A, Vallelian F, Pantaleo G. 2004. Phenotypic heterogeneity of antigen-specific CD4 T cells under different conditions of antigen persistence and antigen load. Eur J Immunol 34:3525–3533. doi: 10.1002/eji.200425324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Sallusto F, Lenig D, Forster R, Lipp M, Lanzavecchia A. 1999. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 401:708–712. doi: 10.1038/44385. [DOI] [PubMed] [Google Scholar]
- 62.Ottenhoff T, Kumararante D, Casanova J. 1998. Novel human immunodeficiencies reveal the essential role of type-1 cytokines in immunity to intracellular bacteria. Immunol Today 19:491–494. doi: 10.1016/S0167-5699(98)01321-8. [DOI] [PubMed] [Google Scholar]
- 63.Chen CY, Huang D, Yao S, Halliday L, Zeng G, Wang RC, Chen ZW. 2012. IL-2 Simultaneously expands Foxp3+ T regulatory and T effector cells and confers resistance to severe tuberculosis (TB): implicative Treg–T effector cooperation in immunity to TB. J Immunol 188:4278–4288. doi: 10.4049/jimmunol.1101291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ernst JD. 2012. The immunological life cycle of tuberculosis. Nat Rev Immunol 12:581–591. doi: 10.1038/nri3259. [DOI] [PubMed] [Google Scholar]
- 65.Kwan CK, Ernst JD. 2011. HIV and tuberculosis: a deadly human syndemic. Clin Microbiol Rev 24:351–376. doi: 10.1128/CMR.00042-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Barber DL, Mayer-Barber KD, Feng CG, Sharpe AH, Sher A. 2011. CD4 T cells promote rather than control tuberculosis in the absence of PD-1-mediated inhibition. J Immunol 186:1598–1607. doi: 10.4049/jimmunol.1003304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Fletcher HA. 2015. Profiling the host immune response to tuberculosis vaccines. Vaccine 33:5313–5315. doi: 10.1016/j.vaccine.2015.07.090. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.