Abstract
Aim
The aim of this paper was to determine natural killer (NK) cytotoxic activity and if single nucleotide polymorphisms (SNPs) and genotypes in transient receptor potential (TRP) ion channels and acetylcholine receptors (AChRs) were present in isolated NK cells from previously identified myalgic encephalomyelitis (ME)/chronic fatigue syndrome (CFS) patients.
Subjects and methods
A total of 39 ME/CFS patients (51.69±2 years old) and 30 unfatigued controls (47.60±2.39 years old) were included in this study. Patients were defined according to the 1994 Centers for Disease Control and Prevention criteria. Flow cytometry protocols were used to examine NK cytotoxic activity. A total of 678 SNPs from isolated NK cells were examined for 21 mammalian TRP ion channel genes and for nine mammalian AChR genes via the Agena Bioscience iPlex Gold assay. SNP association and genotype was determined using analysis of variance and Plink software.
Results
ME/CFS patients had a significant reduction in NK percentage lysis of target cells (17%±4.68%) compared with the unfatigued control group (31%±6.78%). Of the 678 SNPs examined, eleven SNPs for TRP ion channel genes (TRPC4, TRPC2, TRPM3, and TRPM8) were identified in the ME/CFS group. Five of these SNPs were associated with TRPM3, while the remainder were associated with TRPM8, TRPC2, and TRPC4 (P<0.05). Fourteen SNPs were associated with nicotinic and muscarinic AChR genes: six with CHRNA3, while the remainder were associated with CHRNA2, CHRNB4, CHRNA5, and CHRNE (P<0.05). There were sixteen genotypes identified from SNPs in TRP ion channels and AChRs for TRPM3 (n=5), TRPM8 (n=2), TRPC4 (n=3), TRPC2 (n=1), CHRNE (n=1), CHRNA2 (n=2), CHRNA3 (n=1), and CHRNB4 (n=1) (P<0.05).
Conclusion
We identified a number of SNPs and genotypes for TRP ion channels and AChRs from isolated NK cells in patients with ME/CFS, suggesting these SNPs and genotypes may be involved in changes in NK cell function and the development of ME/CFS pathology. These anomalies suggest a role for dysregulation of Ca2+ in AChR and TRP ion channel signaling in the pathomechanism of ME/CFS.
Keywords: chronic fatigue syndrome, myalgic encephalomyelitis, transient receptor potential ion channels, acetylcholine receptors, genotypes
Introduction
Natural killer (NK) cells are granular lymphocytes found in peripheral blood, bone marrow, spleen, and lymph nodes.1–4 In peripheral blood, NK cells comprise 15% of lymphocytes and can be grouped into four subtypes according to the surface expression and density of CD56 (neural cell-adhesion molecule) and CD16 (FcγIII receptor, the low-affinity receptor for IgG).1–3,5,6 These phenotypes include CD56brightCD16−/dim, CD56dimCD16bright, CD56dimCD16−, and CD56−CD16bright.1–3 Approximately 90% of NK cells in peripheral blood are CD56dimCD16bright, and CD56bright comprise approximately 10%.2–4,7 NK cell cytotoxic activity requires a number of regulated processes to ensure apoptosis of the target cell.8
Though little is known about Ca2+ signaling in NK cells, it has been observed that the granule-dependent pathway of apoptosis is calcium-dependent, whereas the death receptor pathway is not.9,10 In apoptosis, lytic protein transport, exocytosis, and fusion have clearly shown calcium dependence.11–13 Ca2+ is also required for the reorientation of microtubules and the actin skeleton, as well as activation of cytokine gene transcription.13 Moreover, studies have demonstrated the relationship between calcium mobilization and the abrogation of degranulation in NK PLCγ2-deficient cells.13–15
Transient receptor potential (TRP) ion channels are expressed on almost all cells, and have a significant effect on physiological functions.16 Dysregulation in TRPs has been associated with pathological conditions and diseases, such as cancer, skeletal abnormalities, pain syndrome, glomerulo-sclerosis, Olmsted syndrome, mucolipidosis, and polycystic kidney disease.17–21 TRP ion channels are activated in the presence of irritants, inflammatory products, and xenobiotic toxins, and have an important role in Ca2+ signaling.
Acetylcholine (ACh) binds to two membrane proteins, namely the muscarinic receptor (mAChR) and nicotinic receptor (nAChR), of which there are multiple isoforms. ACh performs nonneuronal functions, termed the nonneuronal cholinergic system, comprising β-pancreatic cells, glial cells, lymphocytes, ocular lens cells, and brain vascular endothelium,17–26 which is mediated through Ca2+ signaling. AChRs transmit activation signals in a variety of human tissues, including skeletal and smooth muscle, all preganglionic autonomic nerve fibers, postganglionic autonomic parasympathetic nerves, and in many locations throughout the central nervous system.27–29
Myalgic encephalomyelitis (ME)/chronic fatigue syndrome (CFS) has an unknown etiology, and there is no specific diagnostic test. The illness is largely characterized by significant impairment in physical activity and debilitating fatigue, and can be accompanied by impairment in memory, cognition, and concentration, enhanced experience of muscle and joint pain, headaches, sore throat, and tender lymph nodes. It is further associated with dysregulation of the gastrointestinal, cardiovascular, and immune systems.30–42 Importantly, NK cell dysfunction, in particular reduced NK cell cytotoxic activity, is a common finding in ME/CFS patients.32–36,39,43 We have previously identified single-nucleotide polymorphisms (SNPs) in TRP ion channel genes and AChR genes, namely TRPM3, TRPA1, TRPC4,44 CHRM3, CHRNA10, CHRNA5, and CHRNA2 in peripheral blood mononuclear cells from ME/CFS patients.44 These SNP anomalies in genes for TRP ion channels and AChRs may produce altered receptor proteins, potentially changing TRP ion channel and AChR structures and also functions.
The aim of the present study was to determine NK cytotoxic activity, as well as whether SNPs and their genotypes were present in TRP ion channel and AChR genes in isolated NK cells from ME/CFS patients.
Subjects and methods
Subjects
ME/CFS patients were defined in accordance with the 1994 Centers for Disease Control and Prevention criteria for CFS.45 A total of 39 ME/CFS patients and 30 unfatigued controls recruited for this study reported no medical history, symptoms of prolonged fatigue, or illness of any kind, and were screened for major disease according to routine pathology tests. All participants provided informed written consent and all samples were deidentified for the purposes of analysis. This study was approved by the Griffith University Human Research Ethics Committee (MSC22/12HREC).
Sample preparation and measurements
A total volume of 80 mL of blood was collected from the antecubital vein of all participants into lithium-heparinized and ethylenediaminetetraacetic acid (EDTA) collection tubes between 9 am and 11 am. Routine blood samples were analyzed from 10 mL of blood within 6 hours of collection and analyzed for red blood cell counts, lymphocytes, granulocytes, and monocytes using an automated cell counter (ACT differential analyzer; Beckman Coulter, Brea, CA, USA) (Table 1).
Table 1.
Participant characteristics for ME/CFS and unfatigued controls
| Variable | ME/CFS, n=39 | Unfatigued controls, n=30 | P-value |
|---|---|---|---|
| Sex (% female) | 71.79% | 56.67% | 0.228 |
| Mean age (years) | 51.69±2 | 47.6±2.39 | 0.191 |
| Hemoglobin (g/L) | 136.05±2.07 | 138.8±2.24 | 0.375 |
| Hematocrit (%) | 0.41±0.01 | 0.41±0.01 | 0.702 |
| Red-cell count (×1012/L) | 4.54±0.07 | 4.58±0.08 | 0.697 |
| Mean corpuscular volume (fL) | 89.97±0.55 | 90.07±0.7 | 0.917 |
| White-cell count (×109/L) | 5.95±0.26 | 6.38±0.31 | 0.747 |
| Neutrophils (×109/L) | 3.53±0.19 | 3.96±0.26 | 0.173 |
| Lymphocytes (×109/L) | 1.91±0.1 | 1.97±0.08 | 0.64 |
| Monocytes (×109/L) | 0.34±0.02 | 0.32±0.02 | 0.41 |
| Eosinophils (×109/L) | 0.33±0.18 | 0.37±0.23 | 0.892 |
| Basophils (×109/L) | 0.20±0.18 | 0.02±0 | 0.385 |
| Platelets (×109/L) | 262.56±8.41 | 256.79±9.58 | 0.653 |
Note: Data presented as mean ± standard deviation.
Abbreviations: ME, myalgic encephalomyelitis; CFS, chronic fatigue syndrome.
NK cell isolation
NK cells were isolated from 40 mL of whole blood using Ficoll-Paque (GE Healthcare Bio-Sciences AB, Uppsala, Sweden) and performed using an NK isolation kit (Miltenyi Biotech, Bergisch Gladbach, Germany) according to the manufacturer’s instructions. NK cell purity was measured after staining with CD56 and CD3 antibodies and analyzed using a FACSCalibur flow cytometer (BD Biosciences, San Diego, CA, USA). NK cells were reported as the percentage of positive CD56+CD3− cells. Isolated NK cells were either used to measure NK cell cytotoxicity activity or snap-frozen in liquid nitrogen and stored at −80°C until further SNP assessment.
NK cell cytotoxicity
NK cytotoxic activity was conducted as previously described.36,39 Briefly, following NK lymphocyte isolation using density-gradient centrifugation and labeling with 0.4% PKH-26 (Sigma-Aldrich, St Louis, MO, USA), NK cells were incubated with K562 cells for 4 hours at 37°C in 95% air, 5% CO2 at an effector to target (E:T) ratio of 25 (NK cells):1 (K562). An E:T ratio of 25:1 has been previously shown by us and other researchers to be the most optimal ratio for assessing cytotoxic activity.36,39 NK cell lysis activity was determined after incubating K562 cells for 4 hours, and was calculated to determine induced tumor-cell death or apoptosis.1 FACSCalibur flow cytometry using annexin V–fluorescein isothiocyanate and 7-aminoactinomycin reagents (BD Biosciences) was employed. NK cytotoxic activity was performed within 2–4 hours upon receipt of all blood samples.
DNA extraction
A volume of 40 mL was collected into EDTA tubes for SNP analysis. Genomic DNA was extracted from isolated NK cell samples using the Qiagen DNA blood minikit as per manufacturer’s instructions (Qiagen NV, Venlo, the Netherlands). SNP-genotyping studies were performed as previously described.44,46
SNP analysis
A total of 678 SNPs from isolated NK cells were examined for 21 mammalian TRP ion channel genes (TRPA1, TRPC1, TRPC2, TRPC3, TRPC4, TRPC6, TRPC7, TRPM1, TRPM2, TRPM3, TRPM4, TRPM5, TRPM6, TRPM7, TRPM8, TRPV1, TRPV2, TRPV3, TRPV4, TRPV5, and TRPV6) and for nine mammalian AChR genes (muscarinic M1, M2, M3, M4, M5, nicotinic α2, α3, α5, α7, α10, and ε), and were examined using a MassArray iPlex Gold assay (Sequenom Inc, San Diego, CA, USA). Quality and quantity of the DNA extracted was determined by a NanoDrop, where approximately 2 µg of genomic DNA was used to perform the SNP analysis. SNP analysis was performed as previously described.44,46 Briefly, the MassArray (matrix-assisted laser desorption/ionization time-of-flight mass-spectrometry platform) was employed to discriminate alleles based on single-base extension of an extension primer of known mass that was designed to attach directly next to the SNP site of interest. Custom multiplexed wells were designed in silico using Agena’s Assay Design Suite. The designed multiplexes were then built using custom synthesized oligonucleotides that were pooled together for sample processing. The iPlex Gold chemistry utilized two multiplexed oligopools for each genotyping well. These were pooled and balanced prior to running against DNA samples. First, a multiplexed polymerase chain reaction (PCR) pool was utilized to generate short amplicons that included all the genomic markers of interest in that particular well. After PCR and cleanup steps were undertaken, a secondary PCR “extension” step was undertaken, utilizing pools of extension primers that were designed to attach directly next to the SNP sites of interest. A termination mix was added to the extension phase, which allowed these extension primers to be extended by a single base only. As the molecular weight of the extension primer was known, discrimination of the allele was able to be measured using the peak heights of the unextended primer (UEP) and this primer plus the possible single-base extension possibilities for the SNP.
TRP ion channel and AChR SNP assays
For each SNP, two PCR primers and one extension primer were created using the Assay Designer (Sequenom) according to the manufacturer’s instructions. A tag was added to the 5′-end of the sequence for each primer. This was done to increase their mass so they were not detected by MassArray when analyzing the extension primers. DNA was amplified via PCR under the following conditions: 94°C for 2 minutes, 94°C for 30 seconds, 56°C for 30 seconds, and 72°C for 1 minute. Amplification products were then treated with shrimp alkaline phosphatase at 37°C for 40 minutes, 85°C for 5 minutes’ reaction, and a final incubation at 4°C. Extension primers were optimized to control the signal-to-noise ratio, where UEPs are examined on the SpectroChip and evaluated in Typer 4.0 to enable the division into low-mass UEP, medium-mass UEP, and high-mass UEP. To perform the iPlex extension reaction, a mixture containing iPlex Gold reaction was prepared using iPlex Gold Buffer Plus, iPlex termination mix, iPlex enzyme, and primer mix. The iPlex reaction was cycled at an initial denaturation of 94°C for 30 seconds, annealing at 52°C for 5 minutes, extension at 80°C for 5 minutes (five cycles of annealing and extension were performed, but the whole reaction was performed in 40 cycles), and extension again at 72°C for 3 minutes. Resin beads were used to rinse all iPlex Gold reaction products. Following the iPlex Gold reaction, spectrometry was performed using the MassArray mass spectrometer, and the data generated were analyzed using the TyperAnalyzer software.
Statistical analysis
Statistical analysis was performed using SPSS software version 22 (IBM Corporation, Armonk, NY, USA). The experimental data represented in this study are reported as mean ± standard error of the mean, while all the clinical data are reported as mean ± standard deviation. Comparative assessments among participants (ME/CFS and unfatigued controls) were performed with analysis of variance, and the criterion for significance was set at P<0.05.
The Plink version 1.07 (http://pngu.mgh.harvard.edu/purcell/plink) whole-genome-analysis tool set was used to determine associations between the ME/CFS patients and the unfatigued control group. A two-column χ2 test was used to examine differences where P<0.05 was determined to be significant, and the resulting variants and their consequences can be found in Table 2 for TRP and AChR, respectively. Further genotype analysis for differences between CFS and the unfatigued group was also completed according to a two-column χ2 test with significance set at P<0.05. Analyses were performed at the Australian Genome Research Facility Ltd, Walter and Eliza Hall Institute, Melbourne, Australia.
Table 2.
Analysis of frequency, distribution, and significance of SNPs in genes for TRP ion channels and AChRs in ME/CFS patients and unfatigued controls in rank order of significance
| Gene | CL | SNP | BPs | A1 | FM | FC | A2 | χ2 | OR | P-value |
|---|---|---|---|---|---|---|---|---|---|---|
| TRPM8 | 2 | rs17865678 | 2.348 | A | 0.4595 | 0.1667 | G | 12.88 | 4.25 | 0.000332 |
| TRPM8 | 2 | rs11563204 | 2.348 | A | 0.3553 | 0.1167 | G | 10.18 | 4.172 | 0.00142 |
| CHRNB4 | 15 | rs12441088 | 78,635,922 | G | 0.1795 | 0.3793 | T | 6.824 | 0.358 | 0.008993 |
| TRPC4 | 13 | rs2985167 | 37,656,405 | G | 0.2821 | 0.5 | A | 6.742 | 0.3929 | 0.009418 |
| TRPM3 | 9 | rs6560200 | 71,365,306 | T | 0.3974 | 0.6207 | C | 6.633 | 0.4031 | 0.01001 |
| TRPM3 | 9 | rs1106948 | 71,402,258 | C | 0.3974 | 0.6167 | T | 6.521 | 0.41 | 0.01066 |
| TRPM8 | 2 | rs6758653 | 2.348 | A | 0.2436 | 0.45 | G | 6.502 | 0.3936 | 0.01078 |
| CHRNA3 | 15 | rs12914385 | 78,606,381 | T | 0.4872 | 0.2833 | C | 5.879 | 2.403 | 0.01532 |
| CHRNA2 | 8 | rs891398 | 27,467,305 | C | 0.5526 | 0.3448 | T | 5.714 | 2.347 | 0.01683 |
| TRPM3 | 9 | rs12350232 | 71,417,232 | G | 0.3974 | 0.6 | T | 5.571 | 0.4397 | 0.01826 |
| CHRNA2 | 8 | rs2741343 | 27,468,610 | C | 0.5526 | 0.35 | T | 5.537 | 2.294 | 0.01862 |
| TRPM3 | 9 | rs11142822 | 71,427,327 | T | 0.03846 | 0.15 | G | 5.314 | 0.2267 | 0.02115 |
| CHRNA3 | 15 | rs2869546 | 78,615,003 | C | 0.2763 | 0.4667 | T | 5.271 | 0.4364 | 0.02168 |
| TRPM3 | 9 | rs1891301 | 71,403,580 | T | 0.5769 | 0.3833 | C | 5.085 | 2.194 | 0.02414 |
| CHRNA3 | 15 | rs951266 | 78,586,199 | T | 0.4359 | 0.2586 | C | 4.536 | 2.215 | 0.03319 |
| TRPC2 | 11 | rs7108612 | 3,628,856 | T | 0.1923 | 0.06667 | G | 4.509 | 3.333 | 0.03372 |
| TRPC2 | 11 | rs6578398 | 3,616,831 | A | 0.3462 | 0.1833 | G | 4.506 | 2.358 | 0.03378 |
| CHRM1 | 11 | rs6578398 | 62,920,797 | A | 0.2436 | 0.1034 | G | 4.354 | 2.791 | 0.03691 |
| CHRM3 | 1 | rs4620530 | 2.48 | T | 0.4744 | 0.3 | G | 4.301 | 2.106 | 0.03809 |
| CHRM1 | 11 | rs11823728 | 62,909,330 | T | 0.05263 | 0.1607 | C | 4.242 | 0.2901 | 0.03943 |
| CHRNA3 | 15 | rs4243084 | 78,619,330 | G | 0.4359 | 0.2667 | C | 4.204 | 2.125 | 0.04034 |
| CHRNA3 | 15 | rs3743075 | 78,617,110 | A | 0.2821 | 0.45 | G | 4.177 | 0.4802 | 0.04097 |
| CHRNA3 | 15 | rs3743074 | 78,617,138 | C | 0.2821 | 0.45 | T | 4.177 | 0.4802 | 0.04097 |
| CHRNE | 17 | rs33970119 | 4,901,607 | A | 0.03846 | 0.1333 | G | 4.161 | 0.26 | 0.04136 |
| CHRNA5 | 15 | rs7180002 | 78,581,651 | T | 0.4342 | 0.2667 | A | 4.084 | 2.11 | 0.0433 |
Notes: SNPs of 39 CFS/ME patients and 30 unfatigued controls. Data presented all P<0.05.
Abbreviations: SNPs, single-nucleotide polymorphisms; TRP, transient receptor potential; AChRs, acetylcholine receptors; ME, myalgic encephalomyelitis; CFS, chronic fatigue syndrome; CL, chromosome location; BPs, base pairs; FM, frequency of A1/A2 allele in ME/CFS cases; FC, frequency of A1/A2 allele in controls; OR, odds ratio.
Results
Participants
There were 39 CFS patients (age 51.69±2 years), of which 71.79% were females. There were 30 unfatigued controls (age 47.60±2.39 years), of which 56.67% were females. All participants in both groups were of European descent and were residents of Australia at the time of blood collection. There were no significant changes in white blood-cell counts between ME/CFS patients and the unfatigued control group. Table 1 outlines participants’ characteristics.
NK cell purity
There was no significant difference between groups for levels of NK purity. Figure 1 outlines the high levels of purity (>93%) of NK cells following isolation and enrichment.
Figure 1.
NK cell purity.
Notes: The purity of NK cells represents minimal contamination from other cell types. Data shown for ME/CFS (n=38), and unfatigued controls (n=30), and presented as mean ± standard error of mean.
Abbreviations: NK, natural killer; ME, myalgic encephalomyelitis; CFS, chronic fatigue syndrome.
NK cell cytotoxic activity
There was a significant difference for NK cytotoxic activity between groups at the E:T ratio of 25:1. ME/CFS patients had a significant reduction in NK percentage lysis of target cells (17%±4.68%) compared with the control group (31%±6.78%) (Figure 2).
Figure 2.
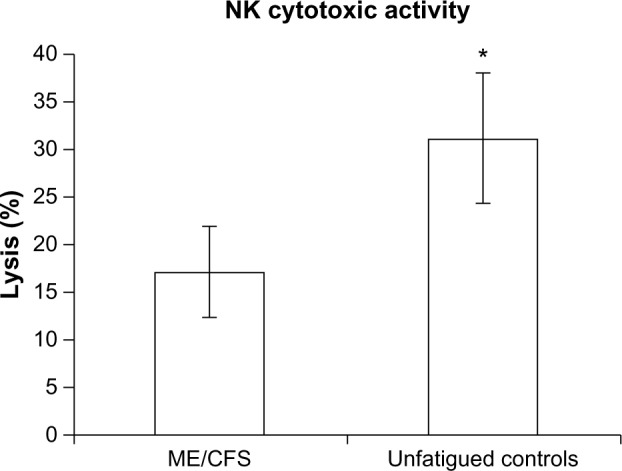
Reduced NK cytotoxic activity in CFS/ME.
Notes: In vivo assessment of NK (cytotoxic activity) of tumor-cell lines K562 in ME/CFS (n=39) and unfatigued controls (n=30). Lytic activity represented by percentage lysis of target cells on the y-axis. Data presented as mean ± standard error of mean. *P<0.05.
Abbreviations: NK, natural killer; ME, myalgic encephalomyelitis; CFS, chronic fatigue syndrome.
SNP analysis
Of 678 SNPs identified in TRP ion channel and AChR genes from isolated NK cells, there were eleven SNPs for TRP ion channel genes (TRPC4, TRPC2, TRPM3, and TRPM8) that were significantly associated in the ME/CFS group. Five of these SNPs were associated with TRPM3 (rs6560200, P=0.01; rs1106948, P=0.01; rs12350232, P=0.018; rs11142822, P=0.021; rs1891301, P=0.024) while the remainder were associated with TRPM8 (rs17865678, P=0; rs1156320, P=0.001), TRPC2 (rs7108612, P=0.034; rs6578398, P=0.0334), and TRPC4 (rs2985167, P=0.001; rs655207, P=0.018).
Fourteen SNPs were associated with nicotinic and muscarinic AChR genes, where six were CHRNA3 (rs12914385, P=0.015; rs2869546, P=0.021; rs951266, P=0.033; rs4243084, P=0.040; rs3743075, P=0.041; rs3743074, P=0.041), while the remainder were associated with CHRNA2 (rs891398, P=0.017; rs2741343, P=0.019), CHRNB4 (rs12441088, P=0.009), CHRNA5 (rs7180002, P=0.043), and CHRNE (rs33970119, P=0.041). Table 2 presents the SNPs for TRP ion channel and AChR genes isolated from NK cells.
Genotype analysis
There were 16 genotypes identified from SNPs that were reported significant for TRPM3 (n=5), TRPM8 (n=2), TRPC4 (n=3), TRPC2 (n=1), CHRNE (n=1), CHRNA2 (n=2), CHRNA3 (n=1), and CHRNB4 (n=1). Of the significant SNPs identified in Table 2, we further analyzed whether genotypes differed between groups. Table 3 presents the genotypes for SNPs in TRP and AChR genes from isolated NK cells that were reported as statistically significant between groups. The odds ratios for specific genotypes for SNPs in TRP and AChR genes from isolated NK cells ranged between 3.13 and 11.39 for ME/CFS compared with the unfatigued control group.
Table 3.
Analysis of genotype, ORs, and significance of SNPs in genes for TRP ion channels and AChRs in ME/CFS patients and unfatigued controls in rank order of significance
| Gene | CL | SNP | Genotype | ME/CFS, n (%) | Unfatigued controls, n (%) | χ2 | OR | P-value |
|---|---|---|---|---|---|---|---|---|
| TRPM8 | 2 | rs11563204 | GA | 23 (82.1) | 5 (17.9) | 12.59 | 7.19 | 0 |
| CHRNA2 | 8 | rs891398 | CC | 11 (91.7) | 1 (8.3) | 7.31 | 11.39 | 0.007 |
| CHRNA2 | 8 | rs2741343 | CC | 11 (91.7) | 1 (8.3) | 7.3 | 11.39 | 0.007 |
| TRPC4 | 13 | rs2985167 | AA | 20 (76.9) | 6 (23.1) | 7.07 | 4.21 | 0.008 |
| TRPM3 | 9 | rs6560200 | CC | 15 (83.3) | 3 (16.7) | 7.12 | 5.63 | 0.008 |
| TRPC4 | 13 | rs1570612 | GG | 30 (68.2) | 14 (31.8) | 6.72 | 3.81 | 0.01 |
| CHRNB4 | 15 | rs12441088 | TT | 25 (71.4) | 10 (28.6) | 6.42 | 3.57 | 0.011 |
| TRPM8 | 2 | rs17865678 | AG | 22 (73.3) | 8 (26.7) | 6.1 | 3.56 | 0.013 |
| TRPC4 | 13 | rs655207 | GG | 12 (85.7) | 2 (14.3) | 6.09 | 6.22 | 0.014 |
| CHRNA3 | 15 | rs12914385 | TT | 12 (85.7) | 2 (14.3) | 6.09 | 6.22 | 0.014 |
| TRPM3 | 9 | rs11142822 | GG | 36 (63.2) | 21 (36.8) | 5.87 | 5.14 | 0.015 |
| TRPM3 | 9 | rs1106948 | TT | 15 (78.9) | 4 (21.1) | 5.37 | 4.06 | 0.021 |
| TRPC2 | 11 | rs7108612 | GT | 15 (78.9) | 4 (21.1) | 5.37 | 4.06 | 0.021 |
| CHRNE | 17 | rs33970119 | GG | 36 (62.1) | 22 (37.9) | 4.56 | 4.36 | 0.033 |
| TRPM3 | 9 | rs1891301 | TT | 14 (77.8) | 4 (22.2) | 4.48 | 3.64 | 0.034 |
| TRPM3 | 9 | rs12350232 | TT | 15 (75) | 5 (25) | 3.91 | 3.13 | 0.048 |
Notes: Genotypes from 39 ME/CFS patients and 30 unfatigued controls. Data presented all P<0.05; df=1 for basic allelic test.
Abbreviations: ORs, odds ratios; SNPs, single-nucleotide polymorphisms; TRP, transient receptor potential; AChRs, acetylcholine receptors; ME, myalgic encephalomyelitis; CFS, chronic fatigue syndrome; CL, chromosome location.
Discussion
Reduced NK cell cytotoxic activity has previously been reported in ME/CFS, and the current investigation supports those findings. The current investigation reports novel findings for a number of SNPs in genes for AChR and TRP variants and genotypes from isolated NK cells from ME/CFS patients. A further novel finding from this investigation is the identification of SNPs in TRPM3 and TRPM8 from isolated NK cells, suggesting TRPM3 and TRPM8 receptors are located on NK cells.
Our investigation reports a significant reduction in NK lysis in ME/CFS patients compared with the unfatigued controls. The role of TRP ion channels in Ca2+ signaling and innate and adaptive immune cells is being increasingly documented.47 TRPs have been reported to express TRP cation channel and TRP melastatin subfamilies, mainly TRPC1, TRPC3, TRPC5, TRPM2, TRPM4, and TRPM7.48 These channels are nonselective and permeable to calcium. In NK cells, Ca2+ plays a key role in lytic granule fusion,11,12,49 as well as ensuring lytic granules mobilize to the immune synapse to release perforin and granzymes to kill target cells.11,12,49 The Rho GTPase Miro provides a link between the mitochondria and the microtubules, where it mediates the Ca2+-dependent arrest of mitochondrial motility.50 As Miro modifies mitochondrial polarization, it may also alter lytic granule trans port to the immune synapse as well as lytic function, due to modulation by cytosolic Ca2+ concentration through TRP melastatin and AChR genes. Clearly, mitochondria play a key role in NK cell function. A recent discovery that mitochondria express a range of AChR subtypes, including nicotinic α3, although differentially expressed according to tissue type,51 suggests that nAChRs may impact mitochondrial function and regulate oxidant stress. Interestingly, we have previously reported a significant decrease in respiratory bust function of neutrophils from ME/CFS patients.34
TRPM2 and TRPM3 mobilize Ca2+, where the latter has been shown to mediate Ca2+ signaling for cytolytic granule polarization and degranulation.52 Further, NK cells treated with an adenosine diphosphate-ribose antagonist had reduced tumor-induced granule polarization, degranulation, granzyme B secretion, and cytotoxicity of NK cells. Interestingly, similar findings for NK cell functions have been reported from previous ME/CFS research,32–36 potentially suggesting that the genotype changes reported in this present study for TRPM3 may also play a similar role in cytolytic granule polarization and degranulation.
Of the 678 SNPs examined, eleven variants for TRP ion channels and 14 variants for AChRs were found to be significantly associated with ME/CFS patients compared with the unfatigued controls. The variant TRP SNPs were located in the gene sequence of two of the canonical TRP ion channels (TRPC2 and TRPC4) and two melastatin TRP ion channels (TRPM3 and TRPM8). We also report variant SNPs in genes for two of the CHRM3 receptors, two CHRM1 receptors, six CHRNA3 receptors, three CHRNA2 receptors, and one CHRNA5 receptor, as well as one CHRNB4 receptor and one CHRNE receptor.
Our current research reports significant SNP associations of genotypes for AChRs in isolated NK cells from ME/CFS patients. Lymphocytes expressed both muscarinic and nicotinic AChRs, where T- and B-cells and monocytes expressed all five subtypes of mAChRs (M1–M5), while nAChRs were found for α2–α7, α9, α10, β2, and β10 subunits.53–56 Lymphocytes constitute a cholinergic system that is independent of cholinergic nerves, resulting in the regulation of immune function.53,54 AChR agonists have been shown to enhance lymphocyte cytotoxicity and increase their intracellular cyclic guanosine monophosphate and inositol-1,4,5-triphosphate,53,57 suggesting the lymphocytic cholinergic system is involved in the regulation of immune function via AChRs coupled to PLC via changes in Ca2+.58–63 Previous research has highlighted the importance of variants in affecting gene transcripts by causing alternative splicing, resulting in anomalies in messenger RNA and translation products.64 We have also identified SNPs and genotype in CHRNE in ME/CFS patients. Interestingly, this SNP is located in the 3′-untranslated region, which is a binding site for regulatory proteins as well as microRNAs (miRNAs).65 Binding to specific sites within the 3′-untranslated region, miRNAs can decrease gene expression of various messenger RNAs by either inhibiting translation or directly causing degradation of the transcript. Our previous research has found significant differences in NK cytotoxic activity, as well as in miRNAs from isolated NK cells from ME/CFS patients.32
Previous investigations have suggested that alternate splicing in the coding and also in the noncoding sequences may have significant unexpected outcomes on the splicing mechanism of the gene transcripts.64,66 Splicing genetic variants located in the exons, introns, and the assembly of the spliceosome all contribute to the splicing mechanism for the correct coding of a protein sequence. Moreover, silencers and enhancers located either in the exons or introns are integral in recognition of the correct exon sequence.67 Importantly, introns are able to generate active spliceosomes, giving rise to alternative splicing events.68,69 Gene 80036 (TRPM3) is associated with calcium entry and calcium-store depletion via different isoforms that have been identified through alternative splicing.70 A region that is indispensable for channel function is an 18-amino acid-residue region whose absence renders the channels functionally unable to mediate calcium entry, and is found devoid in a TRPM3 variant.71 Coexpression of these TRPM3 ICF variants with functional TRPM3 ion channels additionally shows impaired calcium mobilization.71 As TRPM3 ICF variants show ubiquitous expression in many tissues and cell types and constitute 15% of all TRPM3 isoforms, expression on NK cells may provide a potential explanation for reduced cytotoxic activity in ME/CFS patients. Particular splice variants, such as TRPM3α1, may potentially be favored, culminating in a diminished NK cell cytotoxic response and heat detection, including dysregulation of thermoregulatory responses, nociception, and transmission of pain, such as central and peripheral pain perception. Moreover, TRPM8 has also been identified to be activated by cold and noxious stimuli,72–74 suggesting that the genotype changes reported in this investigation align with the clinical presentation of thermoregulatory responses, nociception, and transmission of central and peripheral pain perception seen in ME/CFS patients.75
Our results suggest that SNP variants and genotypes reported in NK cells may not be exclusive to this immune-cell type. AChRs and TRP ion channel receptors are located ubiquitously on multiple cell types and control other functions in body systems. Ca2+ signaling in the context of TRP ion channels and AChR function is vital for the function of the central nervous system, and there is wide variety in the nicotinic receptors expressed in animal and human immune cells.56 Of note, nAChRs are reported to be involved in arousal, sleep, and fatigue, as well as those functions that are responsible for processing of pain, memory, and cognition, all of which are clinical symptoms reported in ME/CFS patients.76–78
Conclusion
In this study, we identified, for the first time, SNPs in genes for TRPM3 and TRPM8 ion channels on isolated NK cells. We also identified numerous SNPs of nAChRs along with other TRP channels on isolated NK cells, which may explain the important role of the nonneuronal ACh system in NK cell function. Anomalies in genotypes for TRP ion channels and AChRs suggest that altered calcium would be an important functional consequence not only for NK cells but also depending upon tissue type, susceptibility, or predisposition to ME/CFS.
Acknowledgments
The authors would like to thank Dr Lavinia Gordon, Australian Genome Research Facility, Melbourne, Australia, for completing the bioinformatics SNP analysis. This study was supported by funding from the Alison Hunter Memorial Foundation, Stafford Fox Medical Research Foundation, Mason Foundation, and Queensland Co-Investment Program.
Footnotes
Author contributions
All authors contributed toward data analysis, drafting and revising the paper and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Caligiuri MA. Human natural killer cells. Blood. 2008;112:461–469. doi: 10.1182/blood-2007-09-077438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cooper MA, Fehniger TA, Caligiuri MA. The biology of human natural killer-cell subsets. Trends Immunol. 2001;22:633–640. doi: 10.1016/s1471-4906(01)02060-9. [DOI] [PubMed] [Google Scholar]
- 3.Sun JC, Lanier LL. NK cell development, homeostasis and function: parallels with CD8+ T cells. Nat Rev Immunol. 2011;11:645–657. doi: 10.1038/nri3044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Möller MJ, Kammerer R, von Kleist S. A distinct distribution of natural killer cell subgroups in human tissues and blood. Int J Cancer. 1998;78:533–538. doi: 10.1002/(sici)1097-0215(19981123)78:5<533::aid-ijc1>3.0.co;2-2. [DOI] [PubMed] [Google Scholar]
- 5.Bryceson YT, March ME, Ljunggren HG, Long EO. Activation, coactivation, and costimulation of resting human natural killer cells. Immunol Rev. 2006;214:73–91. doi: 10.1111/j.1600-065X.2006.00457.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fan YY, Yang BY, Wu CY. Phenotypically and functionally distinct subsets of natural killer cells in human PBMCs. Cell Biol Int. 2008;32:188–197. doi: 10.1016/j.cellbi.2007.08.025. [DOI] [PubMed] [Google Scholar]
- 7.Cooper MA, Fehniger TA, Turner SC, et al. Human natural killer cells: a unique innate immunoregulatory role for the CD56bright subset. Blood. 2001;97:3146–3151. doi: 10.1182/blood.v97.10.3146. [DOI] [PubMed] [Google Scholar]
- 8.Bryceson YT, March ME, Ljunggren HG, Long EO. Synergy among receptors on resting NK cells for the activation of natural cytotoxicity and cytokine secretion. Blood. 2006;107:159–166. doi: 10.1182/blood-2005-04-1351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dustin ML, Long EO. Cytotoxic immunological synapses. Immunol Rev. 2010;235:24–34. doi: 10.1111/j.0105-2896.2010.00904.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rouvier E, Luciani M, Golstein P. Fas involvement in Ca(2+)-independent T cell-mediated cytotoxicity. J Exp Med. 1993;177:195–200. doi: 10.1084/jem.177.1.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Maul-Pavicic A, Chiang SC, Rensing-Ehl A, et al. ORAI1-mediated calcium influx is required for human cytotoxic lymphocyte degranulation and target cell lysis. Proc Natl Acad Sci U S A. 2011;108:3324–3329. doi: 10.1073/pnas.1013285108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Pores-Fernando AT, Zweifach A. Calcium influx and signaling in cytotoxic T-lymphocyte lytic granule exocytosis. Immunol Rev. 2009;231:160–173. doi: 10.1111/j.1600-065X.2009.00809.x. [DOI] [PubMed] [Google Scholar]
- 13.Tassi I, Presti R, Kim S, Yokoyama WM, Gilfillan S, Colonna M. Phospholipase C-γ2 is a critical signaling mediator for murine NK cell activating receptors. J Immunol. 2005;175:749–754. doi: 10.4049/jimmunol.175.2.749. [DOI] [PubMed] [Google Scholar]
- 14.Caraux A, Kim N, Bell SE, et al. Phospholipase C-γ2 is essential for NK cell cytotoxicity and innate immunity to malignant and virally infected cells. Blood. 2006;107:994–1002. doi: 10.1182/blood-2005-06-2428. [DOI] [PubMed] [Google Scholar]
- 15.Zanovello P, Rosato A, Bronte V, et al. Interaction of lymphokine-activated killer cells with susceptible targets does not induce second messenger generation and cytolytic granule exocytosis. J Exp Med. 1989;170:665–677. doi: 10.1084/jem.170.3.665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nilius B, Owsianik G. The transient receptor potential family of ion channels. Genome Biol. 2011;12:218. doi: 10.1186/gb-2011-12-3-218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nilius B, Biro T. TRPV3: a ‘more than skinny’ channel. Exp Dermatol. 2013;22:447–452. doi: 10.1111/exd.12163. [DOI] [PubMed] [Google Scholar]
- 18.Nilius B, Biro T, Owsianik G. TRPV3: time to decipher a poorly understood family member! J Physiol. 2014;592:295–304. doi: 10.1113/jphysiol.2013.255968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Nilius B, Szallasi A. Transient receptor potential channels as drug targets: from the science of basic research to the art of medicine. Pharmacol Rev. 2014;66:676–814. doi: 10.1124/pr.113.008268. [DOI] [PubMed] [Google Scholar]
- 20.Nilius B, Voets T. The puzzle of TRPV4 channelopathies. EMBO Rep. 2013;14:152–163. doi: 10.1038/embor.2012.219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Vennekens R, Menigoz A, Nilius B. TRPs in the brain. Rev Physiol Biochem Pharmacol. 2012;163:27–64. doi: 10.1007/112_2012_8. [DOI] [PubMed] [Google Scholar]
- 22.Elhusseiny A, Hamel E. Muscarinic – but not nicotinic – acetylcholine receptors mediate a nitric oxide-dependent dilation in brain cortical arterioles: a possible role for the M5 receptor subtype. J Cereb Blood Flow Metab. 2000;20:298–305. doi: 10.1097/00004647-200002000-00011. [DOI] [PubMed] [Google Scholar]
- 23.Felder CC, Bymaster FP, Ward J, DeLapp N. Therapeutic opportunities for muscarinic receptors in the central nervous system. J Med Chem. 2000;43:4333–4353. doi: 10.1021/jm990607u. [DOI] [PubMed] [Google Scholar]
- 24.Oki T, Takagi Y, Inagaki S, et al. Quantitative analysis of binding parameters of [3H]N-methylscopolamine in central nervous system of muscarinic acetylcholine receptor knockout mice. Brain Res Mol Brain Res. 2005;133:6–11. doi: 10.1016/j.molbrainres.2004.09.012. [DOI] [PubMed] [Google Scholar]
- 25.Sharma G, Vijayaraghavan S. Nicotinic cholinergic signaling in hippocampal astrocytes involves calcium-induced calcium release from intracellular stores. Proc Natl Acad Sci U S A. 2001;98:4148–4153. doi: 10.1073/pnas.071540198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wess J, Duttaroy A, Zhang W, et al. M1-M5 muscarinic receptor knockout mice as novel tools to study the physiological roles of the muscarinic cholinergic system. Receptors Channels. 2003;9:279–290. [PubMed] [Google Scholar]
- 27.Beckmann J, Lips KS. The non-neuronal cholinergic system in health and disease. Pharmacology. 2013;92:286–302. doi: 10.1159/000355835. [DOI] [PubMed] [Google Scholar]
- 28.Lanzafame AA, Christopoulos A, Mitchelson F. Cellular signaling mechanisms for muscarinic acetylcholine receptors. Receptors Channels. 2003;9:241–260. [PubMed] [Google Scholar]
- 29.Wess J. Molecular biology of muscarinic acetylcholine receptors. Crit Rev Neurobiol. 1996;10:69–99. doi: 10.1615/critrevneurobiol.v10.i1.40. [DOI] [PubMed] [Google Scholar]
- 30.Aaron LA, Burke MM, Buchwald D. Overlapping conditions among patients with chronic fatigue syndrome, fibromyalgia, and temporomandibular disorder. Arch Intern Med. 2000;160:221–227. doi: 10.1001/archinte.160.2.221. [DOI] [PubMed] [Google Scholar]
- 31.Allen J, Murray A, Di Maria C, Newton JL. Chronic fatigue syndrome and impaired peripheral pulse characteristics on orthostasis – a new potential diagnostic biomarker. Physiol Meas. 2012;33:231–241. doi: 10.1088/0967-3334/33/2/231. [DOI] [PubMed] [Google Scholar]
- 32.Brenu EW, Ashton KJ, van Driel M, et al. Cytotoxic lymphocyte microRNAs as prospective biomarkers for chronic fatigue syndrome/myalgic encephalomyelitis. J Affect Disord. 2012;141:261–269. doi: 10.1016/j.jad.2012.03.037. [DOI] [PubMed] [Google Scholar]
- 33.Brenu EW, Ashton KJ, Batovska J, Staines DR, Marshall-Gradisnik SM. High-throughput sequencing of plasma microRNA in chronic fatigue syndrome/myalgic encephalomyelitis. PloS One. 2014;9:e102783. doi: 10.1371/journal.pone.0102783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Brenu EW, Staines DR, Baskurt OK, et al. Immune and hemorheological changes in chronic fatigue syndrome. J Transl Med. 2010;8:1. doi: 10.1186/1479-5876-8-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Brenu EW, van Driel ML, Staines DR, et al. Longitudinal investigation of natural killer cells and cytokines in chronic fatigue syndrome/myalgic encephalomyelitis. J Transl Med. 2012;10:88. doi: 10.1186/1479-5876-10-88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Brenu EW, van Driel ML, Staines DR, et al. Immunological abnormalities as potential biomarkers in chronic fatigue syndrome/myalgic encephalomyelitis. J Transl Med. 2011;9:81. doi: 10.1186/1479-5876-9-81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.DeLuca J, Johnson SK, Beldowicz D, Natelson BH. Neuropsychological impairments in chronic fatigue syndrome, multiple sclerosis, and depression. J Neurol Neurosurg Psychiatry. 1995;58:38–43. doi: 10.1136/jnnp.58.1.38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hoad A, Spickett G, Elliott J, Newton J. Postural orthostatic tachycardia syndrome is an under-recognized condition in chronic fatigue syndrome. QJM. 2008;101:961–965. doi: 10.1093/qjmed/hcn123. [DOI] [PubMed] [Google Scholar]
- 39.Klimas NG, Salvato FR, Morgan R, Fletcher MA. Immunologic abnormalities in chronic fatigue syndrome. J Clin Microbiol. 1990;28:1403–1410. doi: 10.1128/jcm.28.6.1403-1410.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lewis I, Pairman J, Spickett G, Newton JL. Clinical characteristics of a novel subgroup of chronic fatigue syndrome patients with postural orthostatic tachycardia syndrome. J Intern Med. 2013;273:501–510. doi: 10.1111/joim.12022. [DOI] [PubMed] [Google Scholar]
- 41.Medow MS, Stewart JM. The postural tachycardia syndrome. Cardiol Rev. 2007;15:67–75. doi: 10.1097/01.crd.0000233768.68421.40. [DOI] [PubMed] [Google Scholar]
- 42.Nisenbaum R, Reyes M, Mawle AC, Reeves WC. Factor analysis of unexplained severe fatigue and interrelated symptoms: overlap with criteria for chronic fatigue syndrome. Am J Epidemiol. 1998;148:72–77. doi: 10.1093/oxfordjournals.aje.a009562. [DOI] [PubMed] [Google Scholar]
- 43.Maher KJ, Klimas NG, Fletcher MA. Chronic fatigue syndrome is associated with diminished intracellular perforin. Clin Exp Immunol. 2005;142:505–511. doi: 10.1111/j.1365-2249.2005.02935.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Marshall-Gradisnik S, Smith P, Nilius B, Staines DR. Examination of single nucleotide polymorphisms in acetylcholine receptors in chronic fatigue syndrome patients. Immunol Immunogenet Insights. 2015;7:7–20. [Google Scholar]
- 45.Fukuda K, Straus SE, Hickie I, Sharpe MC, Dobbins JG, Komaroff A. The chronic fatigue syndrome: a comprehensive approach to its definition and study. International Chronic Fatigue Syndrome Study Group. Ann Intern Med. 1994;121:953–959. doi: 10.7326/0003-4819-121-12-199412150-00009. [DOI] [PubMed] [Google Scholar]
- 46.Marshall-Gradisnik SM, Smith P, Brenu EW, Nilius B, Ramos SB, Staines DR. Examination of single nucleotide polymorphisms (SNPs) in transient receptor potential (TRP) ion channels in chronic fatigue syndrome patients. Immunol Immunogenet Insights. 2015;2015:1–6. [Google Scholar]
- 47.Billeter AT, Hellmann JL, Bhatnagar A, Polk HC., Jr Transient receptor potential ion channels: powerful regulators of cell function. Ann Surg. 2014;259:229–235. doi: 10.1097/SLA.0b013e3182a6359c. [DOI] [PubMed] [Google Scholar]
- 48.Nilius B, Owsianik G, Voets T, Peters JA. Transient receptor potential cation channels in disease. Physiol Rev. 2007;87:165–217. doi: 10.1152/physrev.00021.2006. [DOI] [PubMed] [Google Scholar]
- 49.Lyubchenko TA, Wurth GA, Zweifach A. Role of calcium influx in cytotoxic T lymphocyte lytic granule exocytosis during target cell killing. Immunity. 2001;15:847–859. doi: 10.1016/s1074-7613(01)00233-3. [DOI] [PubMed] [Google Scholar]
- 50.Wang X, Schwarz TL. The mechanism of Ca2+-dependent regulation of kinesin-mediated mitochondrial motility. Cell. 2009;136:163–174. doi: 10.1016/j.cell.2008.11.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lykhmus O, Gergalova G, Koval L, Zhmak M, Komisarenko S, Skok M. Mitochondria express several nicotinic acetylcholine receptor subtypes to control various pathways of apoptosis induction. Int J Biochem Cell Biol. 2014;53:246–252. doi: 10.1016/j.biocel.2014.05.030. [DOI] [PubMed] [Google Scholar]
- 52.Rah SY, Kwak JY, Chung YJ, Kim UH. ADP-ribose/TRPM2-mediated Ca2+ signaling is essential for cytolytic degranulation and antitumor activity of natural killer cells. Sci Rep. 2015;5:9482. doi: 10.1038/srep09482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kawashima K, Fujii T. Extraneuronal cholinergic system in lymphocytes. Pharmacol Ther. 2000;86:29–48. doi: 10.1016/s0163-7258(99)00071-6. [DOI] [PubMed] [Google Scholar]
- 54.Kawashima K, Fujii T. The lymphocytic cholinergic system and its biological function. Life Sci. 2003;72:2101–2109. doi: 10.1016/s0024-3205(03)00068-7. [DOI] [PubMed] [Google Scholar]
- 55.Kawashima K, Fujii T. Basic and clinical aspects of non-neuronal acetylcholine: overview of non-neuronal cholinergic systems and their biological significance. J Pharmacol Sci. 2008;106:167–173. doi: 10.1254/jphs.fm0070073. [DOI] [PubMed] [Google Scholar]
- 56.Kawashima K, Fujii T, Moriwaki Y, Misawa H. Critical roles of acetylcholine and the muscarinic and nicotinic acetylcholine receptors in the regulation of immune function. Life Sci. 2012;91:1027–1032. doi: 10.1016/j.lfs.2012.05.006. [DOI] [PubMed] [Google Scholar]
- 57.Maśliński W. Cholinergic receptors of lymphocytes. Brain Behav Immun. 1989;3:1–14. doi: 10.1016/0889-1591(89)90001-9. [DOI] [PubMed] [Google Scholar]
- 58.Fujii T, Kawashima K. Calcium signaling and c-Fos gene expression via M3 muscarinic acetylcholine receptors in human T- and B-cells. Jpn J Pharmacol. 2000;84:124–132. doi: 10.1254/jjp.84.124. [DOI] [PubMed] [Google Scholar]
- 59.Fujii T, Kawashima K. YM905, a novel M3 antagonist, inhibits Ca2+ signaling and c-Fos gene expression mediated via muscarinic receptors in human T cells. Gen Pharmacol. 2000;35:71–75. doi: 10.1016/s0306-3623(01)00093-3. [DOI] [PubMed] [Google Scholar]
- 60.Fujii T, Harada H, Koyama T, Nakajima Y, Kawashima K. Effects of physostigmine and calcium on acetylcholine efflux from the hippocampus of freely moving rats as determined by in vivo microdialysis and a radioimmunoassay. Neurosci Lett. 2000;289:181–184. doi: 10.1016/s0304-3940(00)01308-2. [DOI] [PubMed] [Google Scholar]
- 61.Fujii T, Kawashima K. An independent non-neuronal cholinergic system in lymphocytes. Jpn J Pharmacol. 2001;85:11–15. doi: 10.1254/jjp.85.11. [DOI] [PubMed] [Google Scholar]
- 62.Kimura R, Ushiyama N, Fujii T, Kawashima K. Nicotine-induced Ca2+ signaling and down-regulation of nicotinic acetylcholine receptor subunit expression in the CEM human leukemic T-cell line. Life Sci. 2003;72:2155–2158. doi: 10.1016/s0024-3205(03)00077-8. [DOI] [PubMed] [Google Scholar]
- 63.Kawashima K, Fujii T. Expression of non-neuronal acetylcholine in lymphocytes and its contribution to the regulation of immune function. Front Biosci. 2004;9:2063–2085. doi: 10.2741/1390. [DOI] [PubMed] [Google Scholar]
- 64.Pagani F, Baralle FE. Genomic variants in exons and introns: identifying the splicing spoilers. Nat Rev Genet. 2004;5:389–396. doi: 10.1038/nrg1327. [DOI] [PubMed] [Google Scholar]
- 65.Kuersten S, Goodwin EB. The power of the 3′ UTR: translational control and development. Nat Rev Genet. 2003;4:626–637. doi: 10.1038/nrg1125. [DOI] [PubMed] [Google Scholar]
- 66.Cartegni L, Krainer AR. Correction of disease-associated exon skipping by synthetic exon-specific activators. Nat Struct Biol. 2003;10:120–125. doi: 10.1038/nsb887. [DOI] [PubMed] [Google Scholar]
- 67.Fedor MJ. Alternative splicing minireview series: combinatorial control facilitates splicing regulation of gene expression and enhances genome diversity. J Biol Chem. 2008;283:1209–1210. doi: 10.1074/jbc.R700046200. [DOI] [PubMed] [Google Scholar]
- 68.Crawford MH, Banerjee P, Demarchi DA, et al. Applications of pooled DNA samples to the assessment of population affinities: short tandem repeats. Hum Biol. 2005;77:723–733. doi: 10.1353/hub.2006.0012. [DOI] [PubMed] [Google Scholar]
- 69.Shcherbakova I, Hoskins AA, Friedman LJ, et al. Alternative spliceosome assembly pathways revealed by single-molecule fluorescence microscopy. Cell Rep. 2013;5:151–165. doi: 10.1016/j.celrep.2013.08.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Lesch A, Hui X, Lipp P, Thiel G. Transient receptor potential melastatin-3 (TRPM3)-induced activation of AP-1 requires Ca2+ ions and the transcription factors c-Jun, ATF2, and ternary complex factor. Mol Pharmacol. 2015;87:617–628. doi: 10.1124/mol.114.095695. [DOI] [PubMed] [Google Scholar]
- 71.Frühwald J, Camacho Londoño J, Dembla S, et al. Alternative splicing of a protein domain indispensable for function of transient receptor potential melastatin 3 (TRPM3) ion channels. J Biol Chem. 2012;287:36663–36672. doi: 10.1074/jbc.M112.396663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Liu B, Qin F. Functional control of cold- and menthol-sensitive TRPM8 ion channels by phosphatidylinositol 4,5-bisphosphate. J Neurosci. 2005;25:1674–1681. doi: 10.1523/JNEUROSCI.3632-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.McKemy DD. How cold is it? TRPM8 and TRPA1 in the molecular logic of cold sensation. Mol Pain. 2005;1:16. doi: 10.1186/1744-8069-1-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Park CK, Kim MS, Fang Z, et al. Functional expression of thermo-transient receptor potential channels in dental primary afferent neurons: implication for tooth pain. J Biol Chem. 2006;281:17304–17311. doi: 10.1074/jbc.M511072200. [DOI] [PubMed] [Google Scholar]
- 75.Meeus M, Nijs J. Central sensitization: a biopsychosocial explanation for chronic widespread pain in patients with fibromyalgia and chronic fatigue syndrome. Clin Rheumatol. 2007;26:465–473. doi: 10.1007/s10067-006-0433-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Caseras X, Mataix-Cols D, Giampietro V, et al. Probing the working memory system in chronic fatigue syndrome: a functional magnetic resonance imaging study using the n-back task. Psychosom Med. 2006;68:947–955. doi: 10.1097/01.psy.0000242770.50979.5f. [DOI] [PubMed] [Google Scholar]
- 77.Caseras X, Mataix-Cols D, Rimes KA, et al. The neural correlates of fatigue: an exploratory imaginal fatigue provocation study in chronic fatigue syndrome. Psychol Med. 2008;38:941–951. doi: 10.1017/S0033291708003450. [DOI] [PubMed] [Google Scholar]
- 78.Nakatomi Y, Mizuno K, Ishii A, et al. Neuroinflammation in patients with chronic fatigue syndrome/myalgic encephalomyelitis: an 11C-(R)-PK11195 PET study. J Nucl Med. 2014;55:945–950. doi: 10.2967/jnumed.113.131045. [DOI] [PubMed] [Google Scholar]


