Abstract
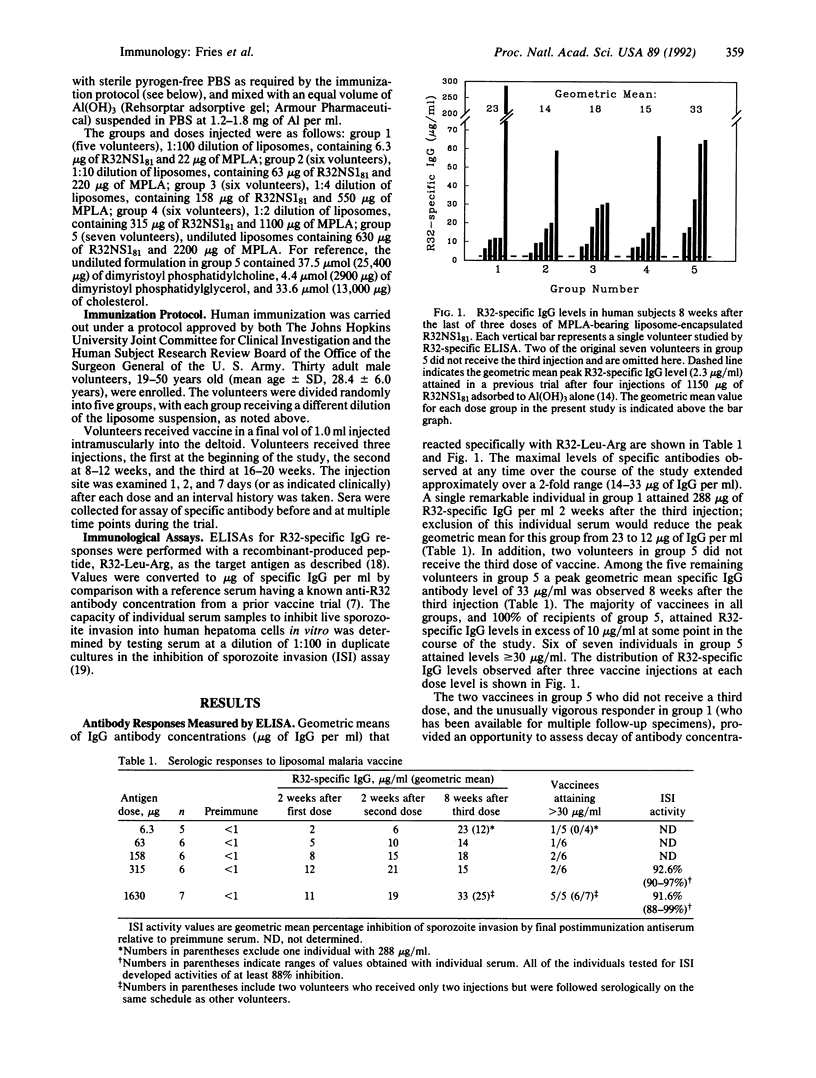
This study describes the safety and immunogenicity of a liposome-based vaccine injected into human subjects. Thirty healthy adult male volunteers were immunized with a liposome-encapsulated recombinant protein (R32NS181) containing epitopes from the repeat region of the circumsporozoite protein of Plasmodium falciparum. This antigen had previously been found to be poorly immunogenic in humans when it was adsorbed with Al(OH)3. In the present study, R32NS181 was encapsulated in liposomes containing monophosphoryl lipid A that were subsequently adsorbed to Al(OH)3. Increasing doses of liposomes containing antigen and monophosphoryl lipid A were used, but the liposomes were always adsorbed to the same dose of Al(OH)3. R32-specific serum IgG antibody responses to liposome-encapsulated R32NS181 were much higher than levels attained previously in humans with R32NS181 adsorbed to Al(OH)3. Geometric mean specific IgG levels after three doses ranged from 14 to 33 micrograms/ml. Sera from volunteers receiving the two highest doses inhibited P. falciparum sporozoite invasion of cultured hepatoma cells by an average of 92%, a result that was again superior to previously reported vaccines. Moderate but acceptable transient local reactogenicity was noted at high doses of the vaccine formulation, but little or no systemic toxicity was seen despite liposomal monophosphoryl lipid A doses up to 2200 micrograms. We conclude that encapsulation of poorly immunogenic circumsporozoite protein repeat peptides in monophosphoryl lipid A-containing liposomes is a successful adjuvant strategy in humans for inducing high levels of specific antibody production.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alving C. R. Liposomes as carriers of antigens and adjuvants. J Immunol Methods. 1991 Jun 24;140(1):1–13. doi: 10.1016/0022-1759(91)90120-5. [DOI] [PubMed] [Google Scholar]
- Alving C. R., Richards R. L. Liposomes containing lipid A: a potent nontoxic adjuvant for a human malaria sporozoite vaccine. Immunol Lett. 1990 Aug;25(1-3):275–279. doi: 10.1016/0165-2478(90)90127-c. [DOI] [PubMed] [Google Scholar]
- Alving C. R., Richards R. L., Moss J., Alving L. I., Clements J. D., Shiba T., Kotani S., Wirtz R. A., Hockmeyer W. T. Effectiveness of liposomes as potential carriers of vaccines: applications to cholera toxin and human malaria sporozoite antigen. Vaccine. 1986 Sep;4(3):166–172. doi: 10.1016/0264-410x(86)90005-8. [DOI] [PubMed] [Google Scholar]
- Aprile M. A., Wardlaw A. C. Aluminium compounds as adjuvants for vaccines and toxoids in man: a review. Can J Public Health. 1966 Aug;57(8):343–360. [PubMed] [Google Scholar]
- BARR M., GLENNY A. T., BUTLER N. R. Immunization of babies with diphtheriatetanus-pertusis prophylactic. Br Med J. 1955 Sep 10;2(4940):635–639. doi: 10.1136/bmj.2.4940.635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ballou W. R., Hoffman S. L., Sherwood J. A., Hollingdale M. R., Neva F. A., Hockmeyer W. T., Gordon D. M., Schneider I., Wirtz R. A., Young J. F. Safety and efficacy of a recombinant DNA Plasmodium falciparum sporozoite vaccine. Lancet. 1987 Jun 6;1(8545):1277–1281. doi: 10.1016/s0140-6736(87)90540-x. [DOI] [PubMed] [Google Scholar]
- Ballou W. R., Rothbard J., Wirtz R. A., Gordon D. M., Williams J. S., Gore R. W., Schneider I., Hollingdale M. R., Beaudoin R. L., Maloy W. L. Immunogenicity of synthetic peptides from circumsporozoite protein of Plasmodium falciparum. Science. 1985 May 24;228(4702):996–999. doi: 10.1126/science.2988126. [DOI] [PubMed] [Google Scholar]
- Butler N. R., Voyce M. A., Burland W. L., Hilton M. L. Advantages of aluminium hydroxide adsorbed combined diphtheria, tetanus, and pertussis vaccines for the immunization of infants. Br Med J. 1969 Mar 15;1(5645):663–666. doi: 10.1136/bmj.1.5645.663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dame J. B., Williams J. L., McCutchan T. F., Weber J. L., Wirtz R. A., Hockmeyer W. T., Maloy W. L., Haynes J. D., Schneider I., Roberts D. Structure of the gene encoding the immunodominant surface antigen on the sporozoite of the human malaria parasite Plasmodium falciparum. Science. 1984 Aug 10;225(4662):593–599. doi: 10.1126/science.6204383. [DOI] [PubMed] [Google Scholar]
- Del Giudice G., Engers H. D., Tougne C., Biro S. S., Weiss N., Verdini A. S., Pessi A., Degremont A. A., Freyvogel T. A., Lambert P. H. Antibodies to the repetitive epitope of Plasmodium falciparum circumsporozoite protein in a rural Tanzanian community: a longitudinal study of 132 children. Am J Trop Med Hyg. 1987 Mar;36(2):203–212. doi: 10.4269/ajtmh.1987.36.203. [DOI] [PubMed] [Google Scholar]
- Edelman R. Vaccine adjuvants. Rev Infect Dis. 1980 May-Jun;2(3):370–383. doi: 10.1093/clinids/2.3.370. [DOI] [PubMed] [Google Scholar]
- Etlinger H. M., Felix A. M., Gillessen D., Heimer E. P., Just M., Pink J. R., Sinigaglia F., Stürchler D., Takacs B., Trzeciak A. Assessment in humans of a synthetic peptide-based vaccine against the sporozoite stage of the human malaria parasite, Plasmodium falciparum. J Immunol. 1988 Jan 15;140(2):626–633. [PubMed] [Google Scholar]
- Good M. F., Berzofsky J. A., Maloy W. L., Hayashi Y., Fujii N., Hockmeyer W. T., Miller L. H. Genetic control of the immune response in mice to a Plasmodium falciparum sporozoite vaccine. Widespread nonresponsiveness to single malaria T epitope in highly repetitive vaccine. J Exp Med. 1986 Aug 1;164(2):655–660. doi: 10.1084/jem.164.2.655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gordon D. M., Davis D. R., Lee M., Lambros C., Harrison B. A., Samuel R., Campbell G. H., Jegathesan M., Selvarajan K., Lewis G. E., Jr Significance of circumsporozoite-specific antibody in the natural transmission of Plasmodium falciparum, Plasmodium vivax, and Plasmodium malariae in an aboriginal (Orang Asli) population of central peninsula Malaysia. Am J Trop Med Hyg. 1991 Jul;45(1):49–56. doi: 10.4269/ajtmh.1991.45.49. [DOI] [PubMed] [Google Scholar]
- Herrington D. A., Clyde D. F., Losonsky G., Cortesia M., Murphy J. R., Davis J., Baqar S., Felix A. M., Heimer E. P., Gillessen D. Safety and immunogenicity in man of a synthetic peptide malaria vaccine against Plasmodium falciparum sporozoites. Nature. 1987 Jul 16;328(6127):257–259. doi: 10.1038/328257a0. [DOI] [PubMed] [Google Scholar]
- Hollingdale M. R., Appiah A., Leland P., do Rosario V. E., Mazier D., Pied S., Herrington D. A., Chulay J. D., Ballou W. R., Derks T. Activity of human volunteer sera to candidate Plasmodium falciparum circumsporozoite protein vaccines in the inhibition of sporozoite invasion assay of human hepatoma cells and hepatocytes. Trans R Soc Trop Med Hyg. 1990 May-Jun;84(3):325–329. doi: 10.1016/0035-9203(90)90303-v. [DOI] [PubMed] [Google Scholar]
- Hollingdale M. R., Nardin E. H., Tharavanij S., Schwartz A. L., Nussenzweig R. S. Inhibition of entry of Plasmodium falciparum and P. vivax sporozoites into cultured cells; an in vitro assay of protective antibodies. J Immunol. 1984 Feb;132(2):909–913. [PubMed] [Google Scholar]
- Richards R. L., Hayre M. D., Hockmeyer W. T., Alving C. R. Liposomes, lipid A, and aluminum hydroxide enhance the immune response to a synthetic malaria sporozoite antigen. Infect Immun. 1988 Mar;56(3):682–686. doi: 10.1128/iai.56.3.682-686.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richards R. L., Swartz G. M., Jr, Schultz C., Hayre M. D., Ward G. S., Ballou W. R., Chulay J. D., Hockmeyer W. T., Berman S. L., Alving C. R. Immunogenicity of liposomal malaria sporozoite antigen in monkeys: adjuvant effects of aluminium hydroxide and non-pyrogenic liposomal lipid A. Vaccine. 1989 Dec;7(6):506–512. doi: 10.1016/0264-410x(89)90274-0. [DOI] [PubMed] [Google Scholar]
- Rickman L. S., Gordon D. M., Wistar R., Jr, Krzych U., Gross M., Hollingdale M. R., Egan J. E., Chulay J. D., Hoffman S. L. Use of adjuvant containing mycobacterial cell-wall skeleton, monophosphoryl lipid A, and squalane in malaria circumsporozoite protein vaccine. Lancet. 1991 Apr 27;337(8748):998–1001. doi: 10.1016/0140-6736(91)92659-p. [DOI] [PubMed] [Google Scholar]
- Verma J. N., Wassef N. M., Wirtz R. A., Atkinson C. T., Aikawa M., Loomis L. D., Alving C. R. Phagocytosis of liposomes by macrophages: intracellular fate of liposomal malaria antigen. Biochim Biophys Acta. 1991 Jul 22;1066(2):229–238. doi: 10.1016/0005-2736(91)90191-a. [DOI] [PubMed] [Google Scholar]
- Vosika G. J., Barr C., Gilbertson D. Phase-I study of intravenous modified lipid A. Cancer Immunol Immunother. 1984;18(2):107–112. doi: 10.1007/BF00205743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Webster H. K., Boudreau E. F., Pang L. W., Permpanich B., Sookto P., Wirtz R. A. Development of immunity in natural Plasmodium falciparum malaria: antibodies to the falciparum sporozoite vaccine 1 antigen (R32tet32). J Clin Microbiol. 1987 Jun;25(6):1002–1008. doi: 10.1128/jcm.25.6.1002-1008.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wirtz R. A., Ballou W. R., Schneider I., Chedid L., Gross M. J., Young J. F., Hollingdale M., Diggs C. L., Hockmeyer W. T. Plasmodium falciparum: immunogenicity of circumsporozoite protein constructs produced in Escherichia coli. Exp Parasitol. 1987 Apr;63(2):166–172. doi: 10.1016/0014-4894(87)90158-5. [DOI] [PubMed] [Google Scholar]
- Yamada A., Ziese M. R., Young J. F., Yamada Y. K., Ennis F. A. Influenza virus hemagglutinin-specific cytotoxic T cell response induced by polypeptide produced in Escherichia coli. J Exp Med. 1985 Aug 1;162(2):663–674. doi: 10.1084/jem.162.2.663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young J. F., Desselberger U., Palese P., Ferguson B., Shatzman A. R., Rosenberg M. Efficient expression of influenza virus NS1 nonstructural proteins in Escherichia coli. Proc Natl Acad Sci U S A. 1983 Oct;80(19):6105–6109. doi: 10.1073/pnas.80.19.6105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young J. F., Hockmeyer W. T., Gross M., Ballou W. R., Wirtz R. A., Trosper J. H., Beaudoin R. L., Hollingdale M. R., Miller L. H., Diggs C. L. Expression of Plasmodium falciparum circumsporozoite proteins in Escherichia coli for potential use in a human malaria vaccine. Science. 1985 May 24;228(4702):958–962. doi: 10.1126/science.2988125. [DOI] [PubMed] [Google Scholar]
- Zavala F., Tam J. P., Hollingdale M. R., Cochrane A. H., Quakyi I., Nussenzweig R. S., Nussenzweig V. Rationale for development of a synthetic vaccine against Plasmodium falciparum malaria. Science. 1985 Jun 21;228(4706):1436–1440. doi: 10.1126/science.2409595. [DOI] [PubMed] [Google Scholar]



