Abstract
Background
Stroke is a common and important adverse event after acute myocardial infarction (AMI) in the elderly. It is unclear whether the risk of stroke after AMI has changed with improvements in treatments and outcomes for AMI in the last decade.
Methods
To assess trends in risk of stroke after AMI, we used a national sample of Medicare data to identify Fee-for-Service patients (n = 2,305,441) aged ≥65 years who were discharged alive after hospitalization for AMI from 1999 to 2010.
Results
We identified 57,848 subsequent hospitalizations for ischemic stroke and 4,412 hospitalizations for hemorrhagic stroke within 1 year after AMI. The 1-year rate of ischemic stroke decreased from 3.4% (95% CI 3.3%-3.4%) to 2.6% (2.5%-2.7%; P < .001). The risk-adjusted annual decline was 3% (hazard ratio, 0.97; [0.97-0.98]) and was similar across all age and sex-race groups. The rate of hemorrhagic stroke remained stable at 0.2% and did not differ by subgroups. The 30-day mortality for patients admitted with ischemic stroke after AMI decreased from 19.9% (18.8%-20.9%) to 18.3% (17.1%-19.6%) and from 48.3% (43.0%-53.6%) to 45.7% (40.3%-51.2%) for those admitted with hemorrhagic stroke. We observed a decrease in 1-year mortality from 37.8% (36.5%-39.1%) to 35.3% (33.8%-36.8%) for ischemic stroke and from 66.6% (61.4%-71.5%) to 60.6% (55.1%-65.9%) for hemorrhagic stroke.
Conclusions
From 1999 to 2010, the 1-year risk for ischemic stroke after AMI declined, whereas the risk of hemorrhagic stroke remained unchanged. However, 30-day and 1-year mortality continued to be high.
Elderly patients have an elevated risk of acute myocardial infarction (AMI) and stroke. The vast majority of AMI and strokes occur in the Medicare population, and AMI is a key risk factor for subsequent stroke.1,2 As the population in the United States ages, the risk of stroke after AMI could be expected to increase. However, other factors may counterbalance the effect of this demographic change. Extensive national efforts to improve processes of care and outcomes for AMI, which resulted in reductions in AMI hospitalization and mortality rates,3,4 might also reduce the risk of stroke after AMI. Improved secondary prevention with statins and antiplatelet therapy to avoid adverse events after AMI could also contribute to risk reduction.5-12 Moreover, the trends in revascularization treatments, such as the increase in rates of percutaneous coronary intervention (PCI) and the decline in rates of coronary artery bypass graft (CABG) surgery,13,14 may impact the risk of stroke. Conversely, the greater use of antiplatelet agents recommended by recent guidelines intended to reduce the risk of ischemic stroke, such as clopidogrel and aspirin, could increase the risk of hemorrhagic stroke.15-18 Surveillance studies using contemporary national data are needed to evaluate whether the risk of stroke after AMI has changed over the last decade.19
To better understand changes in the incidence of stroke after AMI, we used 100% Medicare Fee-For-Service inpatient data from the Centers for Medicare & Medicaid Services (CMS) to characterize temporal trends in the risk for ischemic and hemorrhagic stroke within 1 year after hospitalization for AMI from 1999 to 2010. We also evaluated whether these trends varied by patient age, sex, race, and major surgical treatment subgroups (ie, PCI and CABG). Because of the remarkably high incidence of stroke in the southeastern United States, known as the Stroke Belt, we also examined whether patients who resided in that region also had high risk for strokes after AMI.
Methods
Study sample
We used CMS Medicare Provider Analysis and Review files to identify all Medicare Fee-For-Service patients, aged ≥65 years, who were discharged alive from acute care hospitals with a principal discharge diagnosis of AMI (International Classification of Diseases, Ninth Revision, Clinical Modification [ICD-9-CM] codes 410.xx except 410.x2) between January 1, 1999, and December 31, 2010. If a patient had ≥1 AMI hospitalization within a year, we randomly selected 1 of these hospitalizations. We used the Medicare denominator file to determine patients’ eligibility and enrollment status in Medicare and to obtain patients’ death information. We included patients who had participated for ≥12 months in Medicare Fee-For-Service after the index AMI discharge.
Outcomes
Our primary outcome was the 1-year rate of hospitalization for stroke among survivors of an AMI. The 1-year follow-up started from the date of discharge from the hospitalization for AMI. Consistent with prior studies, we classified strokes as ischemic (ICD-9-CM code 433.xx, 434.xx, or 436.xx) or hemorrhagic (ICD-9-CM codes 430. xx or 431.xx).20,21 Secondary outcomes were 30-day and 1-year all-cause mortality for patients hospitalized for stroke within a year of a hospitalization for AMI. We calculated rates of mortality for ischemic and hemorrhagic stroke separately, using the date of admission for stroke as “time zero.”
Patient characteristics
We used Medicare Provider Analysis and Review data to identify covariates that reflect patient demographic characteristics (age, sex, and race [white, black, and other races]), comorbidities, revascularization, and region of residence. Comorbidities were based on the principal and secondary diagnoses during the 12 months before the index hospitalization using the CMS Hierarchical Condition Categories (online Appendix Supplementary).7,22-27 We also identified whether patients received revascularization during the index AMI hospitalization with PCI only, CABG only, or both PCI and CABG. We defined residence in the Stroke Belt region as primary residence in Alabama, Arkansas, Georgia, Indiana, Kentucky, Louisiana, Mississippi, North Carolina, South Carolina, Tennessee, or Virginia, applying the definition used by the Stroke Belt initiative of the National Heart, Lung, and Blood Institute.28
Statistical analysis
We described the baseline characteristics of patients hospitalized for AMI for each year from 1999 through 2010 using the Cochran-Armitage trend test to examine the significance of trends. We then fit a Cox proportional hazards model to assess the annual trends in the 1-year rate of stroke after AMI, adjusting for age, sex, race, and comorbidities. We fit separate Cox models for age and gender-race subgroups. All Cox models included an ordinal time variable from 0 to 11, corresponding to years 1999 (time = 0) to 2010 (time = 11), to represent the risk-adjusted annual trend in 1-year rates of stroke after AMI. We compared the rates of stroke in AMI patients residing in the Stroke Belt region with those of patients in other regions by including a dummy variable (1 = Stroke Belt and 0 = non–Stroke Belt) in a Cox model. We used SAS version 9.3 64-bit (SAS Institute Inc, Cary, NC) to perform all analyses and set the significance level of all statistical tests at .05. To facilitate data presentation, we report data for patient characteristics in the beginning (1999 and 2000), middle (2004 and 2005), and end (2009 and 2010) of the study period.
This work was supported by grant 1 U01 HL105270-05 (Center for Cardiovascular Outcomes Research at Yale University) from the National Heart, Lung, and Blood Institute. Dr Ross is supported by the National Institute on Aging (K08 AG032886) and by the American Federation for Aging Research through the Paul B. Beeson Career Development Award Program. The authors are solely responsible for the design and conduct of this study, all study analyses, the drafting and editing of the paper, and its final contents. The Yale University Human Investigation Committee approved the study and waived the requirement for participant informed consent.
Results
Patient characteristics
Our study sample included 2,305,441 patients discharged alive from a hospitalization for AMI. The mean age (SD) remained stable from 78.6 (7.8) years in 1999 to 78.8 (8.6) years in 2010. Several comorbidities increased by ≥5 absolute percentage points, including atherosclerosis, respiratory failure, hypertension, and renal failure (Table I and online Appendix Supplementary Table I). The 3 most common comorbidities in 2010 were atherosclerosis, hypertension, and diabetes. The mean length of stay (SD) decreased from 7.2 (6.2) to 5.6 (5.3) days. The proportion of patients with AMI who received postdischarge home care, care at an intermediate care or a skilled nursing facility, or hospice services increased, with the proportion receiving hospice services demonstrating the most change (0.1% in 1999 to 3.4% in 2010, Table I).
Table I.
Patient characteristics, 1999 to 2010
| 1999* | 2000 | 2004 | 2005 | 2009 | 2010 | |
|---|---|---|---|---|---|---|
| Total, no. | 190,745 | 197,916 | 207,584 | 200,092 | 169,716 | 169,863 |
| Age, mean (SD) | 78.6 (7.8) | 78.8 (7.8) | 78.8 (8.1) | 78.9 (8.2) | 78.8 (8.6) | 78.8 (8.6) |
| Female, n (%) | 97398 (51.1) | 102061 (51.6) | 105022 (50.6) | 100186 (50.1) | 83214 (49.0) | 82915 (48.8) |
| White, n (%) | 169698 (89.0) | 175784 (88.8) | 182244 (87.8) | 175552 (87.7) | 148338 (87.4) | 148211 (87.3) |
| Black, n (%) | 13451 (7.1) | 14148 (7.1) | 15583 (7.5) | 15097 (7.5) | 13409 (7.9) | 13441 (7.9) |
| Other, n (%) | 7596 (4.0) | 7984 (4.0) | 9757 (4.7) | 9443 (4.7) | 7969 (4.7) | 8211 (4.8) |
| Atherosclerosis, n (%) | 130343 (68.3) | 138158 (69.8) | 152343 (73.4) | 147186 (73.6) | 125652 (74.0) | 125929 (74.1) |
| Respiratory failure, n (%) | 4798 (2.5) | 5333 (2.7) | 6029 (2.9) | 6049 (3.0) | 8126 (4.8) | 8466 (5.0) |
| Hypertension, n (%) | 101361 (53.1) | 108999 (55.1) | 124539 (60.0) | 120195 (60.1) | 112716 (66.4) | 113563 (66.9) |
| Renal failure, n (%) | 8049 (4.2) | 9335 (4.7) | 13988 (6.7) | 14767 (7.4) | 21803 (12.8) | 22986 (13.5) |
| Home, n (%) | 119406 (62.6) | 122708 (62.0) | 115209 (55.5) | 111251 (55.6) | 94702 (55.8) | 95123 (56.0) |
| Homecare, n (%) | 25560 (13.4) | 25729 (13.0) | 33006 (15.9) | 31214 (15.6) | 26645 (15.7) | 27008 (15.9) |
| ICF/SNF, n (%) | 36242 (19.0) | 38198 (19.3) | 42555 (20.5) | 41419 (20.7) | 33434 (19.7) | 32953 (19.4) |
| Hospice, n (%) | 191 (0.1) | 396 (0.2) | 4152 (2.0) | 4602 (2.3) | 5770 (3.4) | 5775 (3.4) |
| Length of stay, mean (SD) | 7.2 (6.2) | 7.1 (6.2) | 6.7 (6.2) | 6.5 (6.0) | 5.8 (5.4) | 5.6 (5.3) |
| Days to readmission, Mean (SD) | 142 (108) | 141 (109) | 138 (108) | 139 (109) | 138 (109) | 142 (110) |
Abbreviation: ICF/SNF, intermediate care facility/skilled nursing facility.
Data and the comorbidities that increased by ≥5 absolute percentage points over the study period are reported for 1999, 2000, 2004, 2005, 2009, and 2010 for display purposes (see online Appendix Supplementary Table I for all comorbidities).
One-year stroke rate after AMI
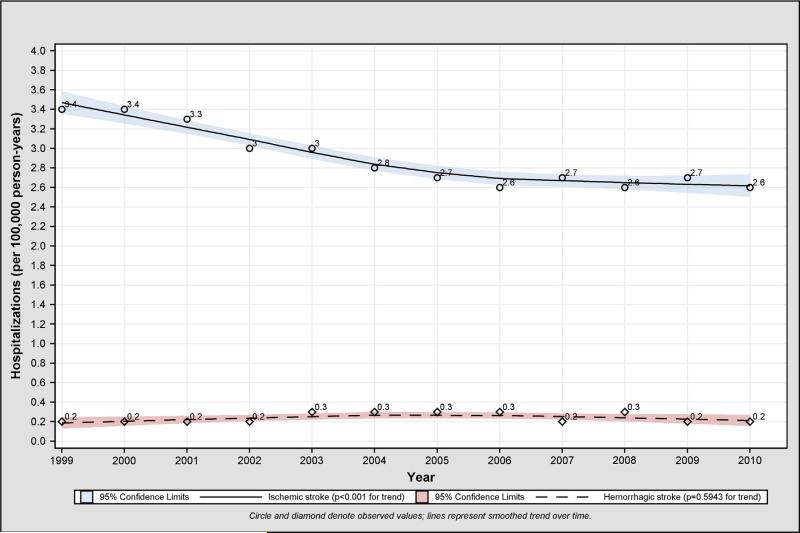
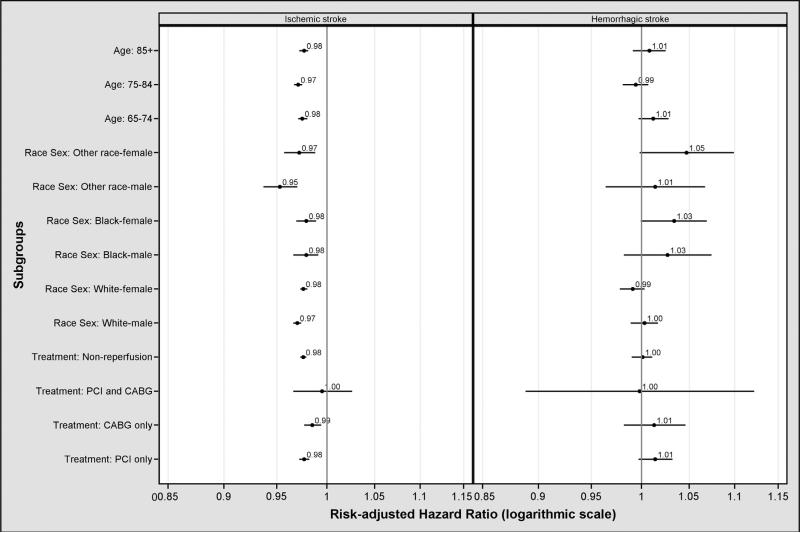
The 1-year rate of ischemic stroke hospitalization after AMI decreased from 3.4% (95% CI 3.3%-3.4%; n = 5,528) in 1999 to 2.6% (95% CI 2.5%-2.7%; n = 3,803) in 2010, a relative decline of 23.5% (P < .001; Figure 1). This decline was similar across all age and sex-race groups (Table II). The median time from discharge from the hospitalization for AMI to subsequent hospitalization for ischemic stroke was 142 days, which remained consistent in a comparison between 1999 and 2010. The rate of ischemic stroke after AMI decreased for patients in the PCI as well as the CABG group but not for patients who had undergone both PCI and CABG (Table II). These findings remained unchanged after adjustment for patient characteristics and for between- and within-hospital variance. The risk-adjusted hazard ratio that represents the annual decline in the 1-year rate of ischemic stroke after AMI was 0.97 (95% CI 0.97-0.98) (risk-adjusted annual trends by subgroups are shown in Figure 2 and online Appendix Supplementary Table II). The 1-year hemorrhagic stroke rate remained stable at 0.2% (Figure 1). This finding was similar across all age and sex-race groups (Table II). No significant annual trends were observed for 1-year rates of hemorrhagic stroke after risk adjustment (hazard ratio, 1.00; [95% CI 0.99-1.01]) (risk-adjusted annual trends in rates of hemorrhagic stroke after AMI by subgroup are shown in Figure 2 and online Appendix Supplementary Table III).
Figure 1.
Observed annual trend in 1-year ischemic and hemorrhagic stroke rates after AMI, 1999 to 2010.
Table II.
One-year ischemic and hemorrhagic stroke rates after AMI, 1999 to 2010
| 1999* | 2000 | 2004 | 2005 | 2009 | 2010 | |
|---|---|---|---|---|---|---|
| Ischemic stroke | ||||||
| Total hospitalizations, no. | 5528 | 5721 | 5073 | 4717 | 3905 | 3803 |
| Rate, % (95% CI) | 3.4 (3.26-3.44) | 3.4 (3.26-3.43) | 2.8 (2.76-2.91) | 2.7 (2.66-2.81) | 2.7 (2.58-2.75) | 2.6 (2.51-2.67) |
| By age group, % (95% CI) | ||||||
| 65-74 | 2.9 (2.76-3.03) | 2.8 (2.67-2.93) | 2.5 (2.37-2.61) | 2.3 (2.20-2.44) | 2.3 (2.19-2.44) | 2.3 (2.16-2.40) |
| 75-84 | 3.5 (3.32-3.59) | 3.6 (3.45-3.73) | 3.0 (2.85-3.10) | 2.9 (2.77-3.02) | 2.8 (2.62-2.90) | 2.6 (2.51-2.78) |
| ≥85 | 3.9 (3.71-4.11) | 3.8 (3.57-3.96) | 3.2 (2.98-3.33) | 3.1 (2.90-3.24) | 3.1 (2.88-3.24) | 3.0 (2.78-3.13) |
| By race-sex group, % (95% CI) | ||||||
| White female | 3.5 (3.35-3.62) | 3.5 (3.34-3.60) | 2.9 (2.74-2.98) | 2.9 (2.74-2.98) | 2.9 (2.73-3.00) | 2.8 (2.67-2.93) |
| White male | 3.0 (2.92-3.17) | 3.0 (2.91-3.16) | 2.6 (2.44-2.65) | 2.5 (2.36-2.58) | 2.3 (2.17-2.40) | 2.3 (2.20-2.42) |
| Black female | 4.7 (4.21-5.22) | 5.0 (4.51-5.53) | 4.5 (4.00-4.92) | 4.5 (4.04-4.98) | 3.9 (3.41-4.34) | 3.7 (3.19-4.10) |
| Black male | 3.6 (3.10-4.18) | 3.9 (3.33-4.42) | 3.6 (3.13-4.13) | 2.7 (2.27-3.14) | 3.3 (2.82-3.84) | 3.0 (2.53-3.49) |
| Other race female | 4.0 (3.30-4.62) | 3.4 (2.77-3.96) | 3.8 (3.22-4.36) | 2.5 (2.03-2.98) | 3.4 (2.80-4.03) | 2.4 (1.91-2.96) |
| Other race male | 3.2 (2.59-3.80) | 3.2 (2.62-3.80) | 2.9 (2.39-3.40) | 2.4 (1.92-2.85) | 2.5 (2.03-3.05) | 1.9 (1.47-2.35) |
| By history of revascularization, % (95% CI) | ||||||
| PCI | 2.5 (2.35-2.67) | 2.4 (2.27-2.57) | 2.3 (2.13-2.36) | 2.1 (1.97-2.20) | 2.1 (2.03-2.26) | 2.1 (2.00-2.23) |
| CABG | 3.0 (2.76-3.29) | 3.1 (2.79-3.31) | 2.8 (2.56-3.06) | 2.7 (2.44-2.95) | 2.7 (2.43-3.02) | 2.7 (2.42-3.01) |
| PCI and CABG | 3.1 (2.10-4.09) | 2.9 (1.89-3.90) | 2.9 (2.05-3.80) | 2.0 (1.25-2.70) | 3.1 (2.10-4.06) | 2.8 (1.88-3.78) |
| Non-reperfusion | 3.7 (3.58-3.80) | 3.8 (3.65-3.87) | 3.2 (3.11-3.33) | 3.2 (3.09-3.32) | 3.1 (2.93-3.18) | 3.0 (2.84-3.09) |
| Hemorrhagic stroke | ||||||
| Total hospitalizations, no. | 356 | 400 | 451 | 435 | 353 | 335 |
| Rate, % (95% CI) | 0.2 (0.19-0.24) | 0.2 (0.21-0.26) | 0.3 (0.23-0.28) | 0.3 (0.23-0.28) | 0.2 (0.22-0.27) | 0.2 (0.20-0.25) |
| By age group, % (95% CI) | ||||||
| 65-74 | 0.2 (0.12-0.18) | 0.2 (0.15-0.22) | 0.2 (0.16-0.23) | 0.2 (0.19-0.27) | 0.2 (0.16-0.23) | 0.2 (0.18-0.25) |
| 75-84 | 0.3 (0.22-0.30) | 0.3 (0.23-0.30) | 0.3 (0.24-0.31) | 0.3 (0.21-0.29) | 0.2 (0.20-0.29) | 0.2 (0.18-0.26) |
| ≥85 | 0.2 (0.19-0.29) | 0.3 (0.21-0.31) | 0.3 (0.26-0.37) | 0.3 (0.24-0.35) | 0.3 (0.25-0.37) | 0.3 (0.21-0.32) |
| By race-sex group, % (95% CI) | ||||||
| White female | 0.3 (0.22-0.29) | 0.3 (0.22-0.29) | 0.3 (0.25-0.32) | 0.3 (0.25-0.32) | 0.3 (0.21-0.29) | 0.2 (0.20-0.28) |
| White male | 0.2 (0.15-0.21) | 0.2 (0.17-0.24) | 0.2 (0.18-0.24) | 0.2 (0.18-0.25) | 0.2 (0.17-0.23) | 0.2 (0.16-0.22) |
| Black female | 0.3 (0.14-0.37) | 0.3 (0.18-0.44) | 0.3 (0.17-0.41) | 0.4 (0.26-0.55) | 0.4 (0.26-0.57) | 0.4 (0.23-0.52) |
| Black male | 0.2 (0.08-0.35) | 0.3 (0.12-0.42) | 0.3 (0.15-0.44) | 0.3 (0.12-0.40) | 0.4 (0.20-0.54) | 0.3 (0.14-0.43) |
| Other race female | 0.1 (0.00-0.24) | 0.4 (0.15-0.54) | 0.4 (0.22-0.61) | 0.1 (0.00-0.19) | 0.4 (0.18-0.60) | 0.4 (0.15-0.55) |
| Other race male | 0.4 (0.14-0.56) | 0.3 (0.09-0.43) | 0.2 (0.04-0.28) | 0.3 (0.13-0.46) | 0.4 (0.17-0.56) | 0.4 (0.18-0.57) |
| By history of revascularization, % (95% CI) | ||||||
| PCI | 0.1 (0.09-0.16) | 0.1 (0.09-0.16) | 0.2 (0.16-0.23) | 0.2 (0.18-0.25) | 0.2 (0.16-0.23) | 0.2 (0.15-0.22) |
| CABG | 0.2 (0.10-0.23) | 0.2 (0.15-0.30) | 0.2 (0.15-0.29) | 0.2 (0.11-0.24) | 0.2 (0.12-0.29) | 0.3 (0.16-0.34) |
| PCI and CABG | 0.2 (−0.07-0.41) | 0.3 (−0.04-0.59) | 0.0 (0.00-0.00) | 0.3 (0.01-0.57) | 0.0 (0.00-0.00) | 0.2 (−0.07-0.42) |
| Non-reperfusion | 0.3 (0.23-0.29) | 0.3 (0.25-0.31) | 0.3 (0.27-0.34) | 0.3 (0.26-0.33) | 0.3 (0.26-0.34) | 0.3 (0.23-0.30) |
For display purposes, we only report data for 1999, 2000, 2004, 2005, 2009, and 2010.
Figure 2.
Risk-adjusted annual trend in 1-year ischemic stroke rates after AMI, 1999 to 2010 (left panel); risk-adjusted annual trend in 1-year hemorrhagic stroke rate after AMI, 1999 to 2010 (right panel).
Stroke Belt versus other regions
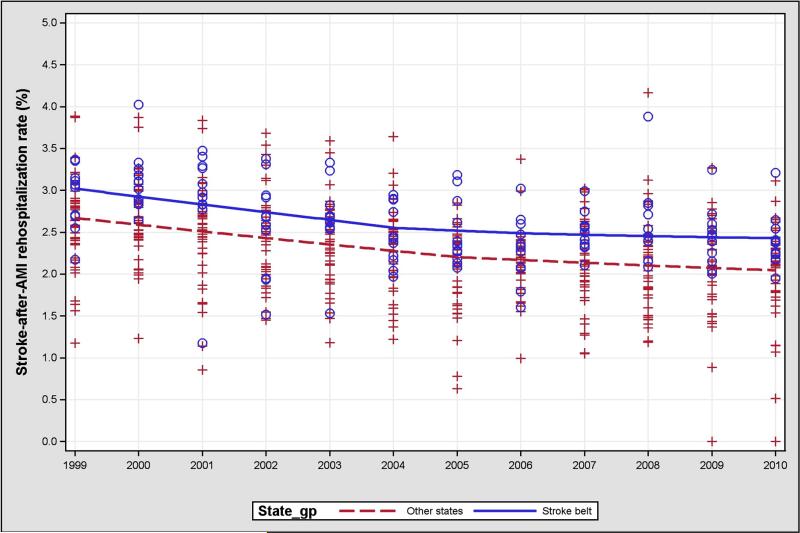
Overall, the risk-adjusted hazard ratio for the rate of ischemic stroke in Stroke Belt regions compared with other regions was 1.06 (95% CI 1.02-1.10). There was no significant interaction between the Stroke Belt region and time. The interaction of Stroke Belt and the time variable was 1.00 (95% CI 0.99-1.01). The rates of ischemic stroke in the Stroke Belt region and non–Stroke Belt regions decreased to a similar degree (Figure 3). Rates of hemorrhagic stroke were not significantly higher in the Stroke Belt region (risk-adjusted hazard ratio, 0.90; [95% CI 0.79-1.03]) and did not significantly change over time. There was no significant interaction between the Stroke Belt region and time (hazard ratio, 1.00; [95% CI 0.98-1.02]).
Figure 3.
One-year ischemic stroke rates by Stroke Belt and non–Stroke Belt regions. Blue dots = Stroke Belt regions; red crosses = non–Stroke Belt regions; lines = mean hospitalization rates of ischemic stroke from 1999 to 2010.
Thirty-day and 1-year all-cause mortality
For patients who survived an AMI and had an ischemic stroke within 1 year of a hospitalization for AMI, the observed rate of 30-day mortality decreased from 19.9% (95% CI 18.8%-20.9%) in 1999 to 18.3% (95% CI 17.1%-19.6%) in 2010. The rate of 1-year mortality also decreased from 37.8% (95% CI 36.5%-39.1%) in 1999 to 35.3% (95% CI 33.8%-36.8%) in 2010 (Table III). For patients who had a hemorrhagic stroke within 1 year of a hospitalization for AMI, the 30-day mortality decreased from 48.3% (95% CI 43.0%-53.6%) in 1999 to 45.7% (95% CI 40.3%-51.2%) in 2010, and the 1-year mortality decreased from 66.6% (95% CI 61.4%-71.5%) in 1999 to 60.6% (95% CI 55.1%-65.9%) in 2010 (Table III). We observed notable variances in rates of mortality across age, sex, and race subgroups for both ischemic and hemorrhagic strokes after AMI (online Appendix Supplementary Tables IV and V).
Table III.
Thirty-day and 1-year mortality rates for ischemic stroke and hemorrhagic stroke after AMI, 1999 to 2010
| 1999* | 2000 | 2004 | 2005 | 2009 | 2010 | |
|---|---|---|---|---|---|---|
| Total ischemic stroke, no. | 5528 | 5721 | 5073 | 4717 | 3905 | 3803 |
| 30-day mortality, % (95% CI) | 19.9 (18.8-20.9) | 20.0 (19.0-21.1) | 18.9 (17.9-20.0) | 20.3 (19.1-21.4) | 19.0 (17.8-20.2) | 18.3(17.1-19.6) |
| 1-year mortality, % (95% CI) | 37.8 (36.5-39.1) | 38.4 (37.2-39.7) | 36.8 (35.5-38.2) | 37.7 (36.3-39.1) | 35.4 (33.9-36.9) | 35.3 (33.8-36.8) |
| Total hemorrhagic stroke, no. | 356 | 400 | 451 | 435 | 423 | 335 |
| 30-day mortality, % (95% CI) | 48.3 (43.0-53.6) | 53.0 (48.0-58.0) | 53.4 (48.7-58.1) | 48.7 (44.0-53.5) | 47.8 (42.9-52.6) | 45.7 (40.3-51.2) |
| 1-year mortality, % (95% CI) | 66.6 (61.4-71.5) | 67.5 (62.7-72.1) | 66.3 (61.7-70.7) | 64.1 (59.4-68.7) | 62.2 (57.4-66.8) | 60.6 (55.1-65.9) |
For display purposes, we only report data for 1999, 2000, 2004, 2005, 2009, and 2010.
Discussion
In this study of contemporary trends in the occurrence and outcomes of stroke after AMI from 1999 to 2010, we found that the rates of ischemic stroke had a relative decline of 23.5%, whereas rates of hemorrhagic stroke remained stable. The improvements represent a nontrivial decline, suggesting 1 fewer subsequent ischemic stroke for every 125 hospitalizations for AMI.
Our findings are consistent with those from European studies.29 In addition, a meta-analysis by Witt et al 30that found the 1-year rate of ischemic stroke after AMI was 2.1%, smaller than the lowest rate in our study. This could possibly be attributed to the younger mean age of the participants in the meta-analysis compared with those in our study.
Our observations extend previous work surrounding trends in rates of stroke hospitalization that were based on the general elderly population, which has a much lower risk of stroke compared with our cohort of elderly patients who have had an AMI. Hall at el 31reported that, from 1999 to 2009, the stroke rate decreased 20% for those aged 65 to 74 and ≥85 years and decreased 24% for those aged 75 to 84. Our study showed that the declines in rates of stroke for AMI patients aged 65 to 74 or ≥85 years were 20.7% and 20.5%, respectively. The decline in the rate of stroke for AMI patients aged 75 to 84 years was 24%. Thus, our study demonstrates a significant reduction in risk of stroke after AMI over the last decade.
Our findings reflect improvements in treatments and outcomes for patients with AMI over the past decade, all of which were associated with reducing risk of stroke. During that period, patients were increasingly treated with procedures and antithrombotic therapies in the acute setting as well as secondary prevention strategies, such as statins and antihypertensive medications, and more patients were recommended to receive dual antiplatelet therapy.15,32-36 Although we cannot establish a direct association between the decline in rates of stroke and improvements in care for AMI, our observations suggest the likelihood of a modest benefit from improvements in treatment and indicate that reductions in rates of ischemic stroke did not appear to be offset by increases in rates of, or morbidity from, hemorrhagic stroke.
The decline in the rates of hospitalization for stroke after AMI may also reflect national efforts focused on risk reduction for this condition. Lifestyle changes, such as smoking cessation, decline in the consumption of red meat, and improvements in the management of diabetes,37-39 could have contributed to the reduced risk for stroke. The observed decline in the rate of ischemic stroke appeared to plateau after 2006, consistent with trends in the rates of stroke in the general population.40 This may suggest that the decline in rates of stroke after AMI could also be attributed to better overall preventive strategies.
Our study has limitations that merit mention. We were limited to the Medicare Fee-For-Service population, the best available database that can provide information about post-AMI outcomes at the national level. Our findings, however, may not reflect changes in the Medicare Managed Care population, a group that tends to be healthier. We were unable to include strokes that did not result in hospitalization, including those that were fatal as well as those that were less severe. We determined diagnosis of AMI and comorbidities through administrative codes rather than medical records. The use of administrative data also precludes the consideration of some clinically relevant prognostic factors as well as the evaluation of the quality of care. Nevertheless, previous studies show that the performance of administrative models is comparable with that of the medical chart abstract-based models.22-24 Furthermore, we could not assess the use of troponin testing, but it had already been adopted during most of our study period. Moreover, there is evidence that the additional individuals diagnosed by a positive troponin test (ie, those who are troponin positive by creatine kinase–MB negative) actually have a higher risk profile than those who are creatine kinase–MB positive.41 Furthermore, during this period, the number of AMI hospitalizations actually decreased.3
Conclusions
Among Medicare beneficiaries from 1999 to 2010, we observed a marked reduction in hospitalizations for ischemic stroke after AMI, possibly related to improved treatment and outcomes associated with AMI. Hemorrhagic stroke after AMI remained rare with persistent rates of high mortality despite the availability of more potent antiplatelet regimens.
Supplementary Material
Footnotes
Disclosures
Drs Ross and Krumholz work under contract with the CMS to develop and maintain performance measures; they are also recipients of research grants from Medtronic and from Johnson & Johnson, through Yale University, to develop methods of clinical trial data sharing. Dr Krumholz is the chair of a cardiac scientific advisory board for UnitedHealth, and Dr Ross is a member of a scientific advisory board for FAIR Health. Dr Masoudi has contracts with the Oklahoma Foundation for Medical Quality and the American College of Cardiology Foundation.
References
- 1.Lichtman JH, Krumholz HM, Wang Y, et al. Risk and predictors of stroke after myocardial infarction among the elderly: results from the Cooperative Cardiovascular Project. Circulation. 2002;105:1082–7. doi: 10.1161/hc0902.104708. [DOI] [PubMed] [Google Scholar]
- 2.Witt BJ, Brown RD, Jr, Jacobsen SJ, et al. A community-based study of stroke incidence after myocardial infarction. Ann Intern Med. 2005;143:785–92. doi: 10.7326/0003-4819-143-11-200512060-00006. [DOI] [PubMed] [Google Scholar]
- 3.Chen J, Normand SL, Wang Y, et al. Recent declines in hospitalizations for acute myocardial infarction for Medicare fee-for-service beneficiaries: progress and continuing challenges. Circulation. 2010;121:1322–8. doi: 10.1161/CIRCULATIONAHA.109.862094. [DOI] [PubMed] [Google Scholar]
- 4.Krumholz HM, Wang Y, Chen J, et al. Reduction in acute myocardial infarction mortality in the United States: risk-standardized mortality rates from 1995-2006. JAMA. 2009;302:767–73. doi: 10.1001/jama.2009.1178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hurlen M, Abdelnoor M, Smith P, et al. Warfarin, aspirin, or both after myocardial infarction. N Engl J Med. 2002;347:969–74. doi: 10.1056/NEJMoa020496. [DOI] [PubMed] [Google Scholar]
- 6.ISIS-2 (Second International Study of Infarct Survival) Collaborative Group Randomised trial of intravenous streptokinase, oral aspirin, both, or neither among 17,187 cases of suspected acute myocardial infarction: ISIS-2. Lancet. 1988;2:349–60. [PubMed] [Google Scholar]
- 7.Keenan PS, Normand SL, Lin Z, et al. An administrative claims measure suitable for profiling hospital performance on the basis of 30-day all-cause readmission rates among patients with heart failure. Circ Cardiovasc Qual Outcomes. 2008;1:29–37. doi: 10.1161/CIRCOUTCOMES.108.802686. [DOI] [PubMed] [Google Scholar]
- 8.Loh E, Sutton MS, Wun CC, et al. Ventricular dysfunction and the risk of stroke after myocardial infarction. N Engl J Med. 1997;336:251–7. doi: 10.1056/NEJM199701233360403. [DOI] [PubMed] [Google Scholar]
- 9.Nayak D, Aronow WS, Sukhija R, et al. Comparison of frequency of left ventricular thrombi in patients with anterior wall versus non-anterior wall acute myocardial infarction treated with antithrombotic and antiplatelet therapy with or without coronary revascularization. Am J Cardiol. 2004;93:1529–30. doi: 10.1016/j.amjcard.2004.02.066. [DOI] [PubMed] [Google Scholar]
- 10.Setoguchi S, Glynn RJ, Avorn J, et al. Improvements in long-term mortality after myocardial infarction and increased use of cardiovascular drugs after discharge: a 10-year trend analysis. J Am Coll Cardiol. 2008;51:1247–54. doi: 10.1016/j.jacc.2007.10.063. [DOI] [PubMed] [Google Scholar]
- 11.Smith P, Arnesen H, Holme I. The effect of warfarin on mortality and reinfarction after myocardial infarction. N Engl J Med. 1990;323:147–52. doi: 10.1056/NEJM199007193230302. [DOI] [PubMed] [Google Scholar]
- 12.Udell JA, Wang JT, Gladstone DJ, et al. Anticoagulation after anterior myocardial infarction and the risk of stroke. PLoS One. 2010;5:e12150. doi: 10.1371/journal.pone.0012150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Epstein AJ, Polsky D, Yang F, et al. Coronary revascularization trends in the United States, 2001-2008. JAMA. 2011;305:1769–76. doi: 10.1001/jama.2011.551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nallamothu BK, Young J, Gurm HS, et al. Recent trends in hospital utilization for acute myocardial infarction and coronary revascularization in the United States. Am J Cardiol. 2007;99:749–53. doi: 10.1016/j.amjcard.2006.10.029. [DOI] [PubMed] [Google Scholar]
- 15.Antman EM, Hand M, Armstrong PW, et al. 2007 Focused Update of the ACC/AHA 2004 Guidelines for the Management of Patients With ST-Elevation Myocardial Infarction: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines: developed in collaboration with the Canadian Cardiovascular Society endorsed by the American Academy of Family Physicians: 2007 Writing Group to Review New Evidence and Update the ACC/AHA 2004 Guidelines for the Management of Patients With ST-Elevation Myocardial Infarction, Writing on Behalf of the 2004 Writing Committee. Circulation. 2008;117:296–329. doi: 10.1161/CIRCULATIONAHA.107.188209. [DOI] [PubMed] [Google Scholar]
- 16.Awtry EH, Loscalzo J. Aspirin. Circulation. 2000;101:1206–18. doi: 10.1161/01.cir.101.10.1206. [DOI] [PubMed] [Google Scholar]
- 17.He J, Whelton PK, Vu B, et al. Aspirin and risk of hemorrhagic stroke: a meta-analysis of randomized controlled trials. JAMA. 1998;280:1930–5. doi: 10.1001/jama.280.22.1930. [DOI] [PubMed] [Google Scholar]
- 18.Van de Werf F, Bax J, Betriu A, et al. Management of acute myocardial infarction in patients presenting with persistent ST-segment elevation: the Task Force on the Management of ST-Segment Elevation Acute Myocardial Infarction of the European Society of Cardiology. Eur Heart J. 2008;29:2909–45. doi: 10.1093/eurheartj/ehn416. [DOI] [PubMed] [Google Scholar]
- 19.Goff DC, Jr, Brass L, Braun LT, et al. Essential features of a surveillance system to support the prevention and management of heart disease and stroke: a scientific statement from the American Heart Association Councils on Epidemiology and Prevention, Stroke, and Cardiovascular Nursing and the Interdisciplinary Working Groups on Quality of Care and Outcomes Research and Atherosclerotic Peripheral Vascular Disease. Circulation. 2007;115:127–55. doi: 10.1161/CIRCULATIONAHA.106.179904. [DOI] [PubMed] [Google Scholar]
- 20.Lichtman JH, Jones SB, Leifheit-Limson EC, et al. 30-day mortality and readmission after hemorrhagic stroke among Medicare beneficiaries in Joint Commission primary stroke center-certified and noncertified hospitals. Stroke. 2011;42:3387–91. doi: 10.1161/STROKEAHA.111.622613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lichtman JH, Jones SB, Wang Y, et al. Outcomes after ischemic stroke for hospitals with and without Joint Commission-certified primary stroke centers. Neurology. 2011;76:1976–82. doi: 10.1212/WNL.0b013e31821e54f3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bratzler DW, Normand SL, Wang Y, et al. An administrative claims model for profiling hospital 30-day mortality rates for pneumonia patients. PLoS One. 2011;6:e17401. doi: 10.1371/journal.pone.0017401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Krumholz HM, Lin Z, Drye EE, et al. An administrative claims measure suitable for profiling hospital performance based on 30-day all-cause readmission rates among patients with acute myocardial infarction. Circ Cardiovasc Qual Outcomes. 2011;4:243–52. doi: 10.1161/CIRCOUTCOMES.110.957498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Krumholz HM, Wang Y, Mattera JA, et al. An administrative claims model suitable for profiling hospital performance based on 30-day mortality rates among patients with an acute myocardial infarction. Circulation. 2006;113:1683–92. doi: 10.1161/CIRCULATIONAHA.105.611186. [DOI] [PubMed] [Google Scholar]
- 25.Krumholz HM, Wang Y, Mattera JA, et al. An administrative claims model suitable for profiling hospital performance based on 30-day mortality rates among patients with heart failure. Circulation. 2006;113:1693–701. doi: 10.1161/CIRCULATIONAHA.105.611194. [DOI] [PubMed] [Google Scholar]
- 26.Lindenauer PK, Normand SL, Drye EE, et al. Development, validation, and results of a measure of 30-day readmission following hospitalization for pneumonia. J Hosp Med. 2011;6:142–50. doi: 10.1002/jhm.890. [DOI] [PubMed] [Google Scholar]
- 27.Pope GC, Kautter J, Ellis RP, et al. Risk adjustment of Medicare capitation payments using the CMS-HCC model. Health Care Financ Rev. 2004;25:119–41. [PMC free article] [PubMed] [Google Scholar]
- 28.National Heart, Lung, and Blood Institute [March 25, 2014];Stroke belt initiative. wwwnhlbinihgov/health/prof/heart/other/sb_specpdf.
- 29.Jakobsson S, Bergstrom L, Bjorklund F, et al. The risk of ischemic stroke after an acute myocardial infarction in diabetic subjects (abstract). Eur Heart J. 2013;34(Suppl 1) [Google Scholar]
- 30.Witt BJ, Ballman KV, Brown RD, Jr, et al. The incidence of stroke after myocardial infarction: a meta-analysis. Am J Med. 2006;119:354. [e1–9]. doi: 10.1016/j.amjmed.2005.10.058. [DOI] [PubMed] [Google Scholar]
- 31.Hall MJ, Levant S, DeFrances CJ. Hospitalization for stroke in U.S. hospitals, 1989-2009. NCHS Data Brief. 2012:1–8. [PubMed] [Google Scholar]
- 32.Holmes JS, Kozak LJ, Owings MF. Use and in-hospital mortality associated with two cardiac procedures, by sex and age: national trends, 1990-2004. Health Aff (Millwood) 2007;26:169–77. doi: 10.1377/hlthaff.26.1.169. [DOI] [PubMed] [Google Scholar]
- 33.Lichtman JH, Leifheit-Limson EC, Jones SB, et al. 30-day risk-standardized mortality and readmission rates after ischemic stroke in critical access hospitals. Stroke. 2012;43:2741–7. doi: 10.1161/STROKEAHA.112.665646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mann D, Reynolds K, Smith D, et al. Trends in statin use and low-density lipoprotein cholesterol levels among US adults: impact of the 2001 National Cholesterol Education Program guidelines. Ann Pharmacother. 2008;42:1208–15. doi: 10.1345/aph.1L181. [DOI] [PubMed] [Google Scholar]
- 35.Rich MM, Colman H, Lichtman JW. In vivo imaging shows loss of synaptic sites from neuromuscular junctions in a model of myasthenia gravis. Neurology. 1994;44:2138–45. doi: 10.1212/wnl.44.11.2138. [DOI] [PubMed] [Google Scholar]
- 36.Tanne D, Koton S, Molshazki N, et al. Trends in management and outcome of hospitalized patients with acute stroke and transient ischemic attack: the National Acute Stroke Israeli (NASIS) registry. Stroke. 2012;43:2136–41. doi: 10.1161/STROKEAHA.111.647610. [DOI] [PubMed] [Google Scholar]
- 37.Daniel CR, Cross AJ, Koebnick C, et al. Trends in meat consumption in the USA. Public Health Nutr. 2011;14:575–83. doi: 10.1017/S1368980010002077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Levy DT, Nikolayev L, Mumford E. Recent trends in smoking and the role of public policies: results from the SimSmoke tobacco control policy simulation model. Addiction. 2005;100:1526–36. doi: 10.1111/j.1360-0443.2005.01205.x. [DOI] [PubMed] [Google Scholar]
- 39.Looker HC, Krakoff J, Andre V, et al. Secular trends in treatment and control of type 2 diabetes in an American Indian population: a 30-year longitudinal study. Diabetes Care. 2010;33:2383–9. doi: 10.2337/dc10-0678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Mitka M. Heart disease and stroke deaths fall, but some fear a reverse in the trend. JAMA. 2012;307(550):2. doi: 10.1001/jama.2012.86. [DOI] [PubMed] [Google Scholar]
- 41.Kontos MC, de Lemos JA, Ou FS, et al. Troponin-positive, MB-negative patients with non-ST-elevation myocardial infarction: An undertreated but high-risk patient group: results from the National Cardiovascular Data Registry Acute Coronary Treatment and Intervention Outcomes Network-Get With The Guidelines (NCDR ACTION-GWTG) Registry. Am Heart J. 2010;160:819–25. doi: 10.1016/j.ahj.2010.07.022. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.