Abstract
Hair-follicle-associated pluripotent (HAP) stem cells can differentiate into many cell types, including neurons and heart muscle cells, and have been shown to repair peripheral nerves and the spinal cord in mice. HAP stem cells can be obtained from each individual patient for regenerative medicine which overcomes problems with immune rejection. Previously, we have demonstrated that genetically-encoded protein markers such as GFP in transgenic mice can be used to visualize HAP stem cells in vivo by multiphoton tomography. Detection and visualization of stem cells in vivo without exogenous labels such as GFP would be important for human application. In the present report, we demonstrate label-free visualization of hair follicle stem cells in mouse whiskers by multiphoton tomography due to the intrinsic fluorophores such as NAD(P)H/flavins. We compared multiphoton tomography of GFP-labeled HAP stem cells and unlabeled stem cells in isolated mouse whiskers. We show that observation of HAP stem cells by label-free multiphoton tomography is comparable to detection using GFP-labeled stem cells. The results described here have important implications for detection and isolation of human HAP stem cells for regenerative medicine.
Keywords: GFP, hair follicles, multiphoton imaging, non-invasive imaging, nestin, pluripotent, skin, stem cells, tomography
Introduction
Nestin-expressing multipotent stem cells are present in the bulge area and dermal papilla of the hair follicle.1,2 We have termed these cells hair follicle-associated pluripotent (HAP) stem cells. HAP stem cells in the hair follicle are positive for CD34 and negative for keratin 15.1,3 The in situ role of the hair-follicle nestin-expressing stem cells is probably to form the hair follicle sensory nerve.4,5 HAP stem cells can differentiate into neurons,3 cardiac muscle cells.6 and other cell types as well as into hair follicle cell types.1,7 HAP stem cells can also enhance nerve and spinal cord regeneration upon transplantation.7-9 HAP stem cells can also form blood vessels and smooth muscle cells and participate in wound healing.3,8-11
We previously demonstrated non-invasive high-resolution multiphoton tomography (MPT) of HAP stem cells in living transgenic nude mice in which the HAP stem cells expressed GFP. The tomograph is comprised of a tunable NIR femtosecond laser system with 360° scan/detector head, mounted on a flexible mechano-optical articulated arm for simultaneous intra-tissue fluorescence and second-harmonic generation (SHG) detection. Non-invasive MPT enables in vivo long-term tracking of intra-tissue stem cells in living mice. Multiphoton imaging with subcellular resolution can visualize the real-time behavior of single stem cells in their native tissue microenvironment.12
Label-free observation of proliferation, migration, and differentiation of stem cells is of high interest. For example, in vivo studies of human stem cells with GFP is not feasible. Multiphoton imaging can overcome this problem because it is based on 2-photon excitation of intrinsic fluorophores such as NAD(P)H, flavins, porphyrins, elastin, and melanin. In addition, SHG images can be obtained from certain biomolecule structures such as collagen and myosin.13
In the present study, HAP stem cells in isolated mouse whiskers were imaged by high-resolution MPT, with and without GFP expression, in order to determine the potential of label-free imaging of HAP stem cells in situ.
Results and Discussion
Non-destructive multiphoton tomography (Fig. 1) was performed on isolated whisker hair follicles expressing nestin-driven GFP (ND-GFP),1,14 as well as whiskers from non-transgenic mice, not expressing GFP.15,16 Optical sections based on the simultaneous detection of 2-photon excited GFP or autofluorescence, as well as SHG of collagen were obtained with a laser wavelength at 760 nm using 2 photomultiplier detectors (Fig. 1).
Figure 1.
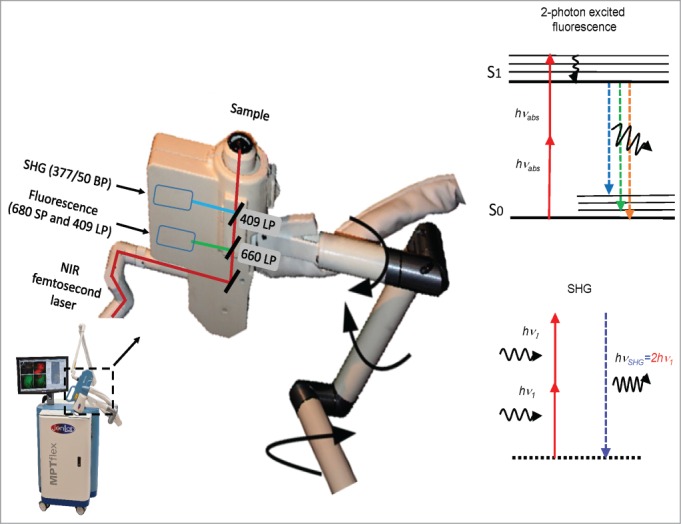
Multiphoton tomograph MPTflex with 360° scan/detector head containing a dual-photon detector unit for the measurement of autofluorescence and SHG. BP, bandpass filter, LP, longpass filter, SP, shortpass filter, NIR, near infrared, SHG, second harmonic generation.
Nestin-GFP-expressing HAP stem cells in the bulge area of mouse whiskers imaged with MPT
Nestin-GFP-expressing HAP stem cells were monitored with 2-photon excitation in the bulge area of the whisker at a depth of 5–200 μm along with SHG detection. The nestin GFP-expressing stem cells occurred in clusters of different numbers of cells per bulge. The typical size of the nestin-expressing stem cells was approximately 7 μm in diameter when they were spherical and less than 5 μm in width when they were elongated (Fig. 2).
Figure 2.
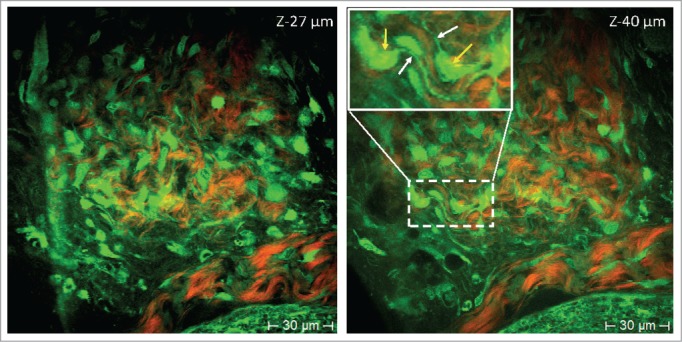
Nestin-GFP expressing hair follicle associated pluripotent (HAP) stem cells in the bulge imaged with multiphoton tomography at different depths (z-27 µm and z-40 µm). As shown in these 2-photon- (760 nm excitation) induced fluorescence images, nestin-GFP HAP stem cells within the whisker appear to have a specific morphology with a round-shaped body and typically 2–3 extrusions (GFP-green-colored cells). Extracellular matrix protein–collagen was imaged without external labeling based on a non-linear effect, termed the second-harmonic generation (red color [pseudo color coded]). 3D optical sectioning demonstrated that nestin-GFP HAP stem cells are distributed in the bulge area within a niche in perpendicular and horizontal sections (position) and well aligned within the collagen fibrillar structures. High intensity GFP fluorescence is detected either in the cell body, including the nucleus and extrusions, or only in the cytoplasm and extrusions but not in the nucleus. Yellow arrows indicate body and white arrows indicate extrusions of HAP stem cells.
Nestin-GFP-expressing HAP stem cells within the whisker appear to have a specific morphology with a round-shaped body and typically 2–3 extrusions. Extracellular matrix protein–collagen was imaged simultaneously based on SHG (red color [pseudo color coded]). 3D optical sectioning demonstrated that nestin-GFP HAP stem cells are distributed in the bulge area within a niche in perpendicular and horizontal sections (position) and well aligned within the collagen fibrillar structures. Furthermore, high intensity GFP-fluorescence was detected either in the cell body, including the nucleus and extrusions, or only in the cytoplasm and extrusions but not in the nucleus (Fig. 2).
Label-free HAP stem cells in the bulge area of the mouse whisker imaged with MPT
Label-free HAP stem cell images were obtained with 2-photon induced autofluorescence of cells and SHG of collagen fibrillar structures. Autofluorescence was detected mainly in the cytoplasm of the HAP stem cells, including extrusions, but not in the nucleus when non-labeled whiskers are excited at 760 nm. The unlabeled HAP stem cells within the hair follicle bulge have the same morphology and size as the nestin-GFP hair follicle stem cells with a round-shaped body and with typically 2–3 extrusions (green color) (Fig. 3).
Figure 3.
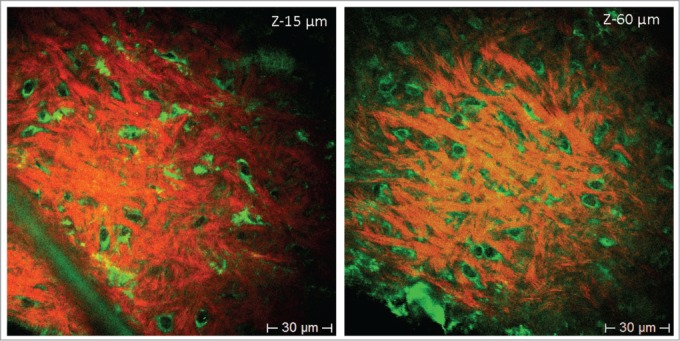
Label-free HAP stem cells in the bulge of the mouse whisker imaged with multiphoton tomography. Images are produced due to the 2-photon-excitation-induced autofluorescence of cells and SHG of collagen fibrillar structures at different depths (z-15 µm and z-60 µm, 760 nm excitation). In these autofluorescence images, fluorescence is detected mainly in the cytoplasm including extrusions, but not in the nucleus. Label-free HAP stem cells within the hair follicle bulge have the same morphology and size as the ND-GFP HAP stem cells with a round-shaped body and with typically 2–3 extrusions (green color). Collagen is imaged by SHG (red [pseudo color coded]).
Nestin-GFP-expressing and label-free HAP stem cells in the dermal papilla imaged with MPT
HAP stem cells in the dermal papilla have migrated from the bulge area.17,18 The nestin-GFP-expressing HAP stem cells in the dermal papilla have ND-GFP expression in the cytoplasm and nucleus (Fig. 4). Label-free stem cells in whiskers from non-GFP-expressing mice were visualized by 2-photon excited autofluorescence at 760 nm and showed identical morphology to ND-GFP expressing stem cells. Autofluorescence arises mainly from mitochondria which contain the endogenous fluorophores NAD(P)H and flavins (Fig. 4). The nuclei are non-fluorescent (Fig. 4).
Figure 4.
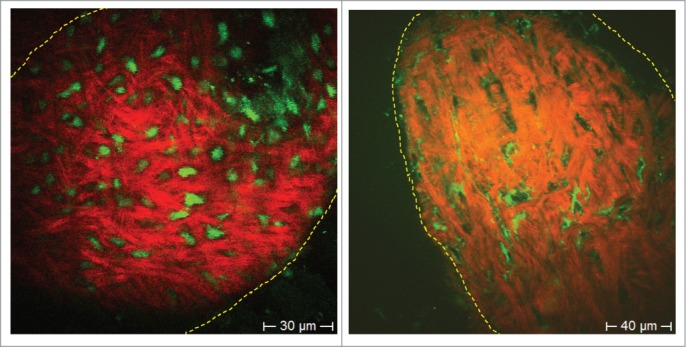
ND-GFP-expressing (left) and label-free HAP stem cells (non-GFP, right) in the dermal papilla. HAP stem cells were imaged with 2-photon induced GFP or by autofluorescence, respectively. Extracellular matrix protein collagen was imaged due to SHG. In the GFP fluorescence image the entire HAP stem cell fluoresced bright green (left panel). In the autofluorescence image (right panel), the cell nucleus appeared dark due to the lack of endogenous fluorophores. Bright autofluorescent structures are in the mitochondria within the cytoplasm, which are rich in intrinsic fluorescent NAD(P)H and flavins. Collagen fibers visualized by SHG are well aligned structures (red [pseudo color coded]).
Materials and Methods
Mice
Transgenic mice, with GFP expression driven by the nestin promoter [nestin-driven GFP (ND-GFP)],1,13,14 were used as a source of whiskers which contain ND-GFP-expressing HAP stem cells. Non-transgenic Balb-C mice were used as a source of whiskers which contain non-GFP-expressing HAP stem cells. Mice were anesthetized with a 30 μl ketamine solution (20 mg/kg ketamine, 15.2 mg/kg xylazine and 0.48 mg/kg Acepromazine maleate). The average age of mice varied from 4 weeks to 4 months. All animal studies were conducted in accordance with the principles of and procedures outlined in the NIH guide for the care and use of laboratory animals under assurance number A3873-1.
Isolation of vibrissa hair follicles
To isolate the vibrissa follicles from GFP-transgenic and non-transgenic mice, the upper lip containing the vibrissa pad was cut under anesthesia and the inner surface was exposed. Entire vibrissa hair follicles were dissected under a binocular microscope and plucked from the pad by pulling them gently by the neck with fine forceps.4
High resolution multiphoton tomography
A multiphoton tomograph MPTflexTM (JenLab GmbH, Jena, Germany and MultiPhoton Laser Technologies Inc., Irvine, CA, Fig. 1) was used. A sealed turn-key tunable 80 MHz titanium:sapphire femtosecond laser (710–920 nm) is mounted on an optical breadboard. The optical unit consists of an active optical power attenuator to regulate the in situ power of the laser, an active beam-stabilization device, a safety unit and a flexible articulated mirror-arm with a compact scan head.19 The scan head consists of a fast galvo-scanning device to generate 2D (XY) scans, a piezodriven z-scanner and high NA focusing optics (NA 1.3). The optical arm is stabilized with a mechanical arm and can be positioned in upright, inverted or in any other position. The 360° scan/detector head contains a dual-photon detector unit for the measurement of autofluorescence and SHG. The overall field-of-view of the optical system covers 350 × 350 μm2. A lateral resolution of 300 nm and an axial resolution of 1–2 µm were determined by the measurement of the point-spread-function of 50 nm fluorescent nanobeads. Optical sections can be generated as deep as 300 μm. The acquisition time for one optical section is typically 6 seconds. Multiphoton imaging is achieved by focusing femtosecond laser radiation at low picojoule pulse energy into the whisker.12
Disclosure of Potential Conflicts of Interest
No potential conflicts of interest were disclosed.
Dedication
This paper is dedicated to the memory of A. R. Moossa, M.D.
Funding
This study was supported in part by the National Institute of Neurological Disorders and Stroke grant NS086217.
References
- 1.Li L, Mignone J, Yang M, Matic M, Penman S, Enikolopov G, Hoffman RM. Nestin expression in hair follicle sheath progenitor cells. Proc Natl Acad Sci USA 2003; 100:9958-61; PMID:12904579; http://dx.doi.org/ 10.1073/pnas.1733025100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hoffman RM. Nestin-expressing hair follicle-accessible pluripotent stem cells for nerve and spinal cord repair. Cells Tissues Organs 2015; 200:42-7; http://dx.doi.org/ 10.1159/000366098 [DOI] [PubMed] [Google Scholar]
- 3.Amoh Y, Li L, Katsuoka K, Penman S, Hoffman RM. Multipotent nestin-positive, keratin-negative hair-follicle-bulge stem cells can form neurons. Proc Natl Acad Sci USA 2005; 102:5530-4; PMID:15802470; http://dx.doi.org/ 10.1073/pnas.0501263102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mii S, Duong J, Tome Y, Uchugonova A, Liu F, Amoh Y, Saito N, Katsuoka K, Hoffman RM. The role of hair follicle nestin-expressing stem cells during whisker sensory-nerve growth in long-term 3D culture. J Cell Biochem 2013; 114:1674-84; PMID:23444061; http://dx.doi.org/ 10.1002/jcb.24509 [DOI] [PubMed] [Google Scholar]
- 5.Mii S, Uehara F, Yano S, Tran B, Miwa S, Hiroshima Y, Amoh Y, Katsuoka K, Hoffman RM. Nestin-expressing stem cells promote nerve growth in long-term 3-dimensional Gelfoam®-supported histoculture. PLoS One 2013; 8:e67153; PMID:23840607; http://dx.doi.org/ 10.1371/journal.pone.0067153 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yashiro M, Mii S, Aki R, Hamada Y, Arakawa N, Kawahara K, Hoffman RM, Amoh Y. From hair to heart: nestin-expressing hair-follicle-associated pluripotent (HAP) stem cells differentiate to beating cardiac muscle cells. Cell Cycle 2015; 14(14):2362-66; PMID:25970547 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Amoh Y, Li L, Campillo R, Kawahara K, Katsuoka K, Penman S, Hoffman RM. Implanted hair follicle stem cells form Schwann cells that support repair of severed peripheral nerves. Proc Natl Acad Sci USA 2005; 102:17734-8; PMID:16314569; http://dx.doi.org/ 10.1073/pnas.0508440102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Liu F, Uchugonova A, Kimura H, Zhang C, Zhao M, Zhang L, Koenig K, Duong J, Aki R, Saito N, et al.. The bulge area is the major hair follicle source of nestin-expressing pluripotent stem cells which can repair the spinal cord compared to the dermal papilla. Cell Cycle 2011; 10:830-9; PMID:21330787; http://dx.doi.org/ 10.4161/cc.10.5.14969 [DOI] [PubMed] [Google Scholar]
- 9.Amoh Y, Li L, Katsuoka K, Hoffman RM. Multipotent hair follicle stem cells promote repair of spinal cord injury and recovery of walking function. Cell Cycle 2008; 7:1865-9; PMID:18583926; http://dx.doi.org/ 10.4161/cc.7.12.6056 [DOI] [PubMed] [Google Scholar]
- 10.Amoh Y, Kanoh M, Niiyama S, Hamada Y, Kawahara K, Sato Y, Hoffman RM, Katsuoka K. Human hair follicle pluripotent stem (hfPS) cells promote regeneration of peripheral-nerve injury: An advantageous alternative to ES and iPS cells. J Cell Biochem 2009; 107:1016-20; PMID:19507228; http://dx.doi.org/ 10.1002/jcb.22204 [DOI] [PubMed] [Google Scholar]
- 11.Amoh Y, Hamada Y, Aki R, Kawahara K, Hoffman RM, Katsuoka K. Direct transplantation of uncultured hair-follicle pluripotent stem (hfPS) cells promotes the recovery of peripheral nerve injury. J Cell Biochem 2010; 110:272-7; PMID:20411592 [DOI] [PubMed] [Google Scholar]
- 12.Uchugonova A, Hoffman RM, Weinigel M, Koenig K. Watching stem cells in the skin of living mice noninvasively. Cell Cycle 2011; 10:2017-20; PMID:21558804; http://dx.doi.org/ 10.4161/cc.10.12.15895 [DOI] [PubMed] [Google Scholar]
- 13.Uchugonova A, Hoffman RM, Koenig K. Stem cell imaging in living animals: stem cells in hairs tracked by by multiphoton tomography. Imaging Microscopy 2013; 3:44-6. [Google Scholar]
- 14.Mignone JL, Kukekov V, Chiang AS, Steindler D, Enikolopov G. Neural stem and progenitor cells in nestin-GFP transgenic mice. J Comp Neurol 2004; 469:311-24; PMID:14730584; http://dx.doi.org/ 10.1002/cne.10964 [DOI] [PubMed] [Google Scholar]
- 15.Li L, Margolis LB, Hoffman RM. Skin toxicity determined in vitro by three-dimensional, native-state histoculture. Proc Natl Acad Sci USA 1991; 88:1908-12; PMID:1848016; http://dx.doi.org/ 10.1073/pnas.88.5.1908 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hoffman RM. The pluripotency of hair follicle stem cells. Cell Cycle 2006; 5:232-3; PMID:16410728; http://dx.doi.org/ 10.4161/cc.5.3.2397 [DOI] [PubMed] [Google Scholar]
- 17.Duong J, Mii S, Uchugonova A, Liu F, Moossa AR, Hoffman RM. Real-time confocal imaging of trafficking of nestin-expressing multipotent stem cells in mouse whiskers in long-term 3-D histoculture. In Vitro Cell Dev Biol Anim 2012; 48:301-5; PMID:22580909; http://dx.doi.org/ 10.1007/s11626-012-9514-z [DOI] [PubMed] [Google Scholar]
- 18.Uchugonova A, Duong J, Zhang N, König K, Hoffman RM. The bulge area is the origin of nestin-expressing pluripotent stem cells of the hair follicle. J Cell Biochem 2011; 112:2046-50; PMID:21465525; http://dx.doi.org/ 10.1002/jcb.23122 [DOI] [PubMed] [Google Scholar]
- 19.König K. Hybrid multiphoton tomography of in vivo human skin. Intravital 2012; 1:11-26; http://dx.doi.org/ 10.4161/intv.21938 [DOI] [Google Scholar]


