Abstract
The interaction of nanomaterials with biomolecules, cells, and organisms is an enormously vital area of current research, with applications in nanoenabled diagnostics, imaging agents, therapeutics, and contaminant removal technologies. Yet the potential for adverse biological and environmental impacts of nanomaterial exposure is considerable and needs to be addressed to ensure sustainable development of nanomaterials. In this Outlook four research needs for the next decade are outlined: (i) measurement of the chemical nature of nanomaterials in dynamic, complex aqueous environments; (ii) real-time measurements of nanomaterial–biological interactions with chemical specificity; (iii) delineation of molecular modes of action for nanomaterial effects on living systems as functions of nanomaterial properties; and (iv) an integrated systems approach that includes computation and simulation across orders of magnitude in time and space.
Short abstract
The interaction of nanomaterials with biosystems is a vital area of research. Yet the potential for adverse impacts is a concern. Research needs are outlined to enable sustainable nanotechnology.
Engineered nanomaterials began to be created in earnest by chemists and materials scientists at the dawn of the 21st century due to government investments such as the U.S. National Nanotechnology Initiative (NNI).1 Most fundamentally interesting are materials with quantitatively and qualitatively unique behaviors that emerge at the 1–100 nm length scales. For instance, semiconductors exhibit quantum confinement effects in the ∼1–10 nm range, and metals display plasmon resonances at optical frequencies on the ∼10–100 nm scale.2−4 Even small organic molecules, when formulated into nanoscale rather than micrometer-scale particles, possess remarkable properties. For example, nanoscale pharmaceutical formulations have faster dissolution rates than earlier micrometer-scale technologies, enhancing bioavailability.5
The physical properties of nanoscale materials are fascinating on many levels. The nexus of these materials with biology is currently an area of intense study, driven largely by the promise in biomedical applications (Figure 1): diagnostic chemical sensing, cellular imaging, drug delivery, therapeutics, and tissue engineering. Yet, notions of peril temper this promise with concerns about biological or environmental exposures that may lead to unintended adverse consequences. The managers of the NNI have been aware of these environmental, health, and safety (EHS) concerns regarding nanotechnology since its inception and have supported programs to study and address EHS issues.1,6 This is a welcome development in the chemical sciences compared to the past, in which new chemicals or materials entered commerce with little thought for their potential EHS impacts (e.g., asbestos, DDT, CFCs, PCBs).
Figure 1.
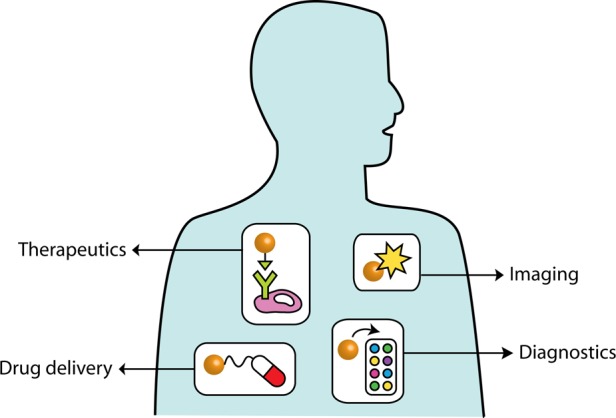
The promise of nanotechnology to improve human health includes diagnostics, drug delivery, imaging, and therapy.
The promise of nanotechnology to improve human health is clear. Nanoparticles are of the right size to circulate through the body, be taken up by living cells, or passively accumulate near tumors (according to many, but not all, studies).7−10 Therefore, drug delivery has emerged as a potential application for many organic and inorganic nanoparticles. If nanoparticles can be made active—optically, thermally, or magnetically—then they can be employed as bioimaging and contrast agents that go beyond the current standards in biology, for instance, to enable both diagnosis and therapy from a single engineered nanoparticle platform.11−16 These formulations include examples such as near-infrared-absorbing inorganic nanoparticles that upon illumination at the proper wavelengths produce enough heat to kill cancer cells11,12 and silver nanoparticles embedded in wound dressings that slowly oxidize to produce a steady stream of antimicrobial silver ions.15 However, nanomaterials that have been approved by the U.S. Food and Drug Administration for drug delivery consist largely of organic nanoformulations: liposomes, lipoplexes, polyethylene glycol–drug nanocomplexes, or albumin nanosphere conjugates.16−19 Superparamagnetic iron oxide nanoparticles have been the only clinically approved metal oxide nanoparticles,20 and of the metal nanoparticles, gold nanoparticles have entered clinical trials as drug delivery vehicles or as photothermal therapeutics for light-induced ablation of tumors.21
Beyond direct use of engineered nanomaterials for applications in health and medicine, large quantities (tons, in some cases) of nanomaterials are now constituents of mass consumer products, with more coming online every day. Many of these nanomaterials are components in emerging clean energy and clean water systems that benefit both human and environmental health.22,23 For example, an electric vehicle (e.g., the 2015/2016 Nissan Leaf) will contain approximately 50 kg of nanostructured metal oxide electrodes in its cathodes;22 as electric vehicles replace conventional vehicles, automobile emissions will decline. Nanomaterials can adsorb environmental contaminants and are being actively studied for drinking water treatment.24 Nanoscale semiconductor quantum dots are now being integrated into plastic films to enhance color saturation in displays for consumer electronics, including laptop computers, electronic readers, and cell phones.25 Concerns regarding the biological, environmental, and ultimately human health consequences of inadvertent release of such engineered nanomaterials into the environment through a variety of pathways need to be taken seriously, given our past history and experience with introducing supposedly benign materials into the environment. From a fundamental chemistry perspective, then, we need to ask: what will we need to know about new nanomaterials that are to be produced on a large scale if we wish to avoid negative outcomes, while at the same time take advantage of what these new materials offer?
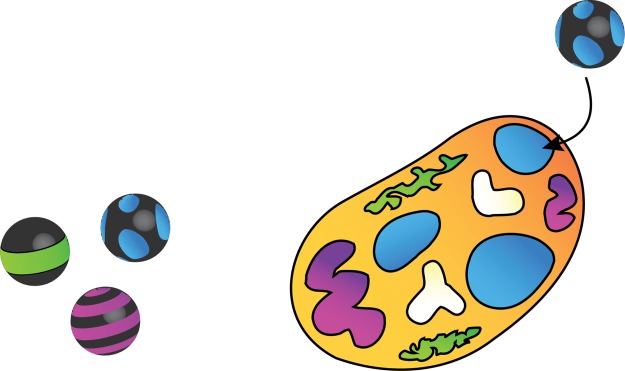
The EHS aspect of nanomaterials research has led to a number of studies aimed at correlating the physicochemical characteristics of nanomaterials with biological or environmental outcomes. For example, substantial evidence exists that cationic nanoparticles lead to more deleterious effects than their anionic counterparts at the cellular level.26 Existing large-scale efforts to screen nanomaterials for biological effects, while valuable, focus on identifying exposure levels that cause death or induce physiological changes or on the development of structure–toxicity relationships.27 Such empirical studies provide few mechanistic details about the chemical and physical processes occurring where a nanoparticle interfaces with its environment: its “skin,” the nanoparticle surface. Molecular-scale insight into the nanomaterial interactions with biological systems (Figure 2) is essential for effective design of nanomaterials with minimal detrimental biological effects while maintaining their function. Here, we argue that establishing causality—rather than correlations—in how nanomaterial properties and behavior impact biological outcomes is a key challenge that can be addressed by specifically drawing on recent advances and developments in the chemical sciences. Previous work in the nano-EHS area has generally focused on single-component nanoparticles such as TiO2, Au, and carbon nanotubes. Looking ahead, it becomes apparent that the nanoparticles used in technology will often be multicomponent systems or nanocomposites; thus, increasingly complex and technologically relevant nanomaterial products need to be examined in as great detail, if not greater, than the constituent nanomaterials for EHS concerns.
Figure 2.
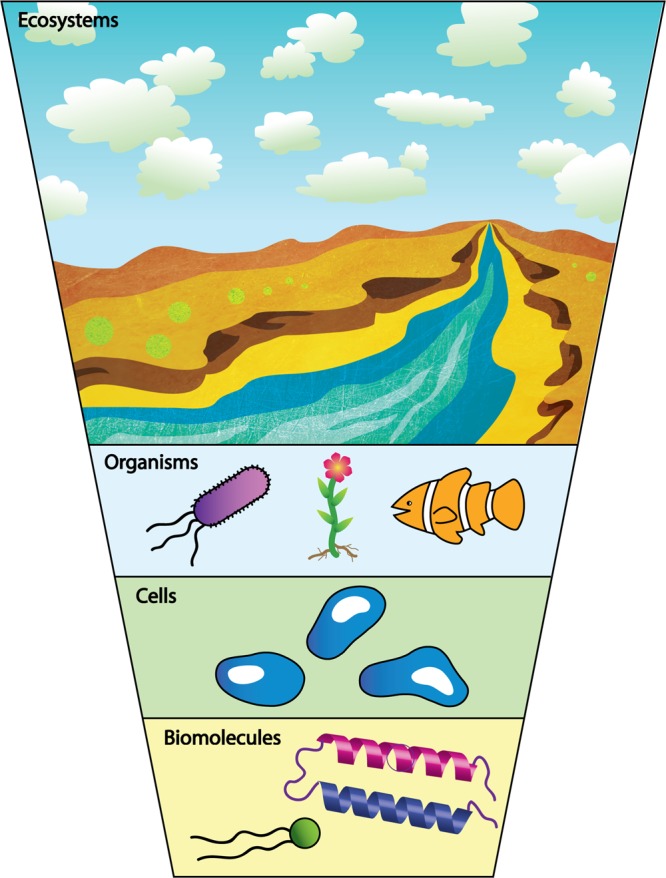
Nanoparticles interact with biological systems at the molecular, cellular, organismal, and ecosystem levels.
Given the wide variety of nanomaterial–biological studies with so many variables, some broad needs clearly exist at the basic science level:
1. Chemically Driven Understanding of the Molecular Nature of Engineered Nanoparticles in Complex, Realistic Environments
The problem is rooted in complexity: nanomaterials come in a variety of sizes, shapes, and initial surface coatings, all of which affect their bioactivity. Many commercial nanomaterials are delivered to users as agglomerates of smaller primary particles; determination of the actual particle size under relevant conditions can be very challenging. Well-documented cases in which apparent adverse nanomaterial effects were due to leftover reagents from the synthesis28,29 highlight the importance in this field for careful controls and attention to nanoparticle purification methods. Similarly, the low quality of nanomaterials in some studies (e.g., mixtures of different sizes and shapes of particles) complicates the extraction of nanosize-specific information in terms of biological impact. New methods, therefore, are needed to assess nanomaterial quality during synthesis and when in use. In the U.S., the Nanotechnology Characterization Laboratory (NCL) of the National Cancer Institute currently performs a well-regarded set of biological assays on submitted nanomaterials, at no charge; but the NCL rejects samples that are deemed too variable in their physical and chemical parameters such as size, shape, composition, and solubility. The U.S. National Institute of Standards and Technology (NIST) has only a few standard reference nanomaterials available, including 10, 30, and 60 nm diameter gold nanoparticles (SRM 8011, 8012, 8013 respectively) and, as of March 2015, polymer-coated silver nanoparticles with a nominal diameter of 75 nm (SRM 8017). The fact that the main U.S. agency charged with providing chemical and materials standards to the scientific community has taken so long to produce very basic nanomaterials speaks to the difficulty of developing reproducible and scalable syntheses. The current “best” colloidal nanomaterials with respect to polydispersity have dimensions within 5% of the mean length across one or more axes.
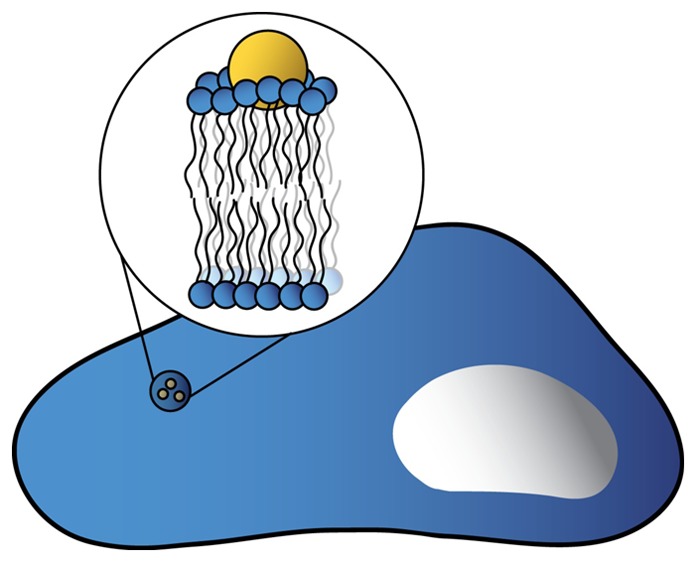
The initial surface chemistry of the nanomaterials clearly influences their fate and distribution in biological systems, despite the well-known biomolecular “coronas” that overcoat the nanomaterials upon immersion into biological fluids.30 It is not yet clear what combination of initial nanomaterial properties (e.g., charge, size, surface functionalization, aggregation state) can be used to predict the composition of the biomolecular coronas they acquire, how this corona changes over time, or the ultimate biological outcome of nanoparticles bearing biomolecular coronas; yet all of these initial nanomaterial properties have been reported to generally affect biological outcomes in one form or another. In situ measurements of the dynamic transformations of nanomaterials and their surfaces, in biological environments, represents a key need for the next decade.
2. Real-Time Measurements of Nanomaterial Interaction with Living Cells and Organisms That Provide Chemical Information at Nanometer Length Scales To Yield Invaluable Mechanistic Insight and Improve Predictive Understanding of the Nano–Bio Interface
While nanomaterials can be large enough to visualize in many different microscopy experiments, doing so in aquo and in real time while simultaneously acquiring molecular information (as opposed to the localization of fluorescent spots) represents a major challenge. In many cases, the first biological interface nanomaterials encounter is a cell membrane. The lipid bilayer–nanomaterial interface has therefore emerged as an important focus of experimental and theoretical interest over the last several years.31−34 Our recent paper demonstrated that a large assembly of physical and analytical measurements was needed to quantitatively assess the thermodynamics and electrostatics of nanoparticle–membrane interactions.35 To date, most such model membrane systems are quite simple, consisting of single phospholipids or binary or ternary mixtures of lipids. Intact cellular membranes contain a large variety of components in addition to phosphoplipids (e.g., peripheral and transmembrane proteins, proteoglycans, glycolipids) that may also influence nanomaterial interaction with cell surfaces. From an imaging perspective, tracking nanomaterials as they interact with living cells or tissues36,37 using a combination of standard and super-resolution fluorescence microscopies,38 as well as nonlinear optical microscopies with video-rate tracking,39,40 is a burgeoning area of interest. Super-resolution vibrational imaging would provide nanoscale spatial and chemical information. Additional innovative approaches combining existing and novel instrumentation to access the nano–bio interface need to be developed to enable these fascinating molecular interactions to be understood, controlled, and predicted.
3. Delineation of Molecular Modes of Action for Nanomaterial Effects on Living Systems as Functions of Nanomaterial Properties
Over 50,000 studies have now been published on the interaction of nanomaterials, both organic and inorganic, with cultured cells and whole organisms (Web of Science, searching topic of (nanomaterials OR nanoparticles) AND biol* AND (cell* OR organ*), accessed May 22, 2015). These studies provide a plethora of data that inform us of potential mechanistic interactions of nanoparticles with cells.41−45 However, these studies have covered dozens of different cell lines, either established or primary; the time of nanomaterial exposure ranges from minutes to days; the nanomaterial doses applied can differ by 6 orders of magnitude, which can dramatically impact the molecular responses instigated in a system and the ultimate consequence of the exposure. Therefore, stating general conclusions about chemical—let alone nanomaterial—effects on living cells becomes difficult, complicating attempts to extrapolate from cellular data to whole organisms.46 Indeed, it is rare that a clear molecular pathway from nanomaterial to cellular response can be constructed. Yet, such molecular pathways, if properly understood, could serve as a means to predict the future impact of nanomaterials on living systems, as evidenced by the groundswell of papers that tell us that cells and organisms can up- or downregulate the expression of specific genes upon exposure to nanomaterials.44,47−51 Other molecular signals have been found: nearly 7000 papers point to reactive oxygen species as a “smoking gun” in cell–nanomaterial toxicity experiments (although the ability of cells and organisms to adapt to oxidative stress is well-known). Moreover, nanomaterial interference with specific enzymes has been observed.52 Even nanomaterial alteration of the extracellular matrix that surrounds cells can lead, indirectly, to nano–bio interactions.47,53 Given this context, the chemical nature of the biological “receptor” (cell membrane, tissue lining, etc.) needs to be understood at the molecular level in time and space, as it is an equal partner in nano–bio interactions (Figure 3).
Figure 3.
Interactions between nanoparticles that are chemically complex and biological “receptors” need to be understood at a molecular level.
Compounding the problems associated with the delineation of molecular modes of action for nanomaterial effects on living systems is that many of the available studies used acute (short-term) exposures, and we know from other classes of chemicals (e.g., pesticides, endocrine disruptors) that extending measures of biological response beyond mortality and short time points is needed to fully assess the potential unintended consequences of exposure. Numerous investigations have shown that aspects of the chemical nature of nanomaterials (e.g., initial surface charge, ligand composition, potential for dissolution) influence their organism-level impact.54−57 However, studies are difficult to compare in that different organisms respond differently to a given chemical, let alone nanomaterial.41−46,58 The difficulty of understanding and predicting bioactivity of nanomaterials is exacerbated when one considers that very small metallic nanomaterials (≤3 nm) are catalytically active in a size-dependent manner.59,60 Thus, biological impacts from size alone can be convolved with new intrinsic reactivity if the studies include samples that span the “unreactive” and “reactive” regimes.
4. Computation and Simulation of the Nano–Bio Interface
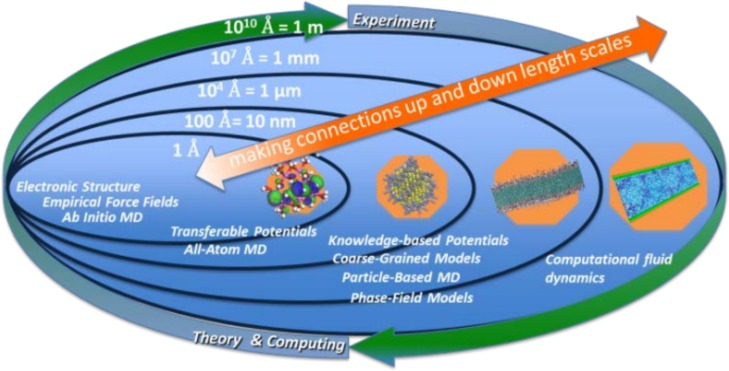
A need exists to develop multiscale algorithms and models that provide direct and correct structure and dynamics of nanoparticles in complex environments including those within organisms. At the smallest scale, such multiscale computational tools must address molecular-scale interactions that can be specifically tailored through chemical control of a nanoparticle in the initial design and are modified through its life cycle in products and in the environment. At the largest scales, such tools must address the changing behavior and transport of nanoparticles inside organisms. The span of these scales, illustrated in Figure 4, is on the order of 1010. Establishing explicit connections to experimental observables at all these scales is essential for validation and refinement of computational methodologies.
Figure 4.
Computational approaches to understanding the nano–bio interface span orders of magnitude in time and space.
To this end, computational frameworks that enable prediction of molecular-level interactions between nanoparticles and their environment are vitally needed. These frameworks may integrate state-of-the-art computational methods spanning multiple length scales, from angstrom using atomistic quantum-mechanical calculations through particle and continuum levels using coarse-grained modeling. Reliable atomistic force fields need to be developed based on accurate quantum chemistry computations, including advanced density functional theory61,62 and embedding methods that integrate correlated ab initio and DFT calculations. To efficiently characterize the binding of biomolecules to nanoparticles under complex environmental conditions,63 novel equilibrium64,65 and nonequilibrium66 sampling techniques are also required. Effective coarse-grained (CG)67−70 and dynamic models71 guided by atomistic simulations to model nanoparticle assembly and their interaction with complex membranes72 are likely to play increasingly important roles as well because they can deal with longer time scales and larger length scales than their atomistic counterparts. Ideally, computational frameworks that feature intimate coupling of CG dynamics, kinetic Monte Carlo, and local atomistic computations will be outcomes from such endeavors, along with the ability to use such new modeling frameworks for computationally driven, rationally designed nanomaterials that are also sustainable. A validated multiscale framework providing a connection between molecular-scale tunability and meso- to macroscale structure and function would be useful beyond the applications of interest to studying nanomaterials, potentially including emergent catalytic behavior and transport of various species within organisms.
Overall, this is an exciting time to be studying the nanomaterial–biological interface. Nanomaterial syntheses have improved to the point where very monodisperse and well-characterized samples can be prepared at reasonably large scales; computation and simulation have improved to the point where an entire virus can be simulated with all-atom molecular dynamics;73 super-resolution fluorescence microscopy approaches have been developed in the past 10 years that enable 10–20 nm resolution imaging in intact hydrated cells;74,75 and genomics, proteomics, and metabolomics greatly speed the measurement of biological end points and uncovering of pathways associated with cellular response and toxicity.76,77 Taken together, this confluence of scientific advances will enable true molecular-level understanding of the nanomaterial–biology interface in the next decade and beyond.
Acknowledgments
The authors acknowledge support from the National Science Foundation, Center for Sustainable Nanotechnology (NSF CHE-1240151).
The authors declare no competing financial interest.
References
- Roco M., Bainbridge W. S., Eds. Nanotechnology: Societal implications I; Maximizing benefits for humanity; Springer: Dordrecht, The Netherlands, 2007. [Google Scholar]
- Alivisatos A. P. Semiconductor clusters, nanocrystals, and quantum dots. Science 1996, 271, 933–937. [Google Scholar]
- Kelly K. L.; Coronado E.; Zhao L. L.; Schatz G. C. The optical properties of metal nanoparticles: the influence of size, shape, and dielectric environment. J. Phys. Chem. B 2003, 107, 668–677. [Google Scholar]
- Burda C.; Chen X.; Narayanan R.; El-Sayed M. A. Chemistry and properties of nanocrystals of different shapes. Chem. Rev. 2005, 105, 1025–1102. [DOI] [PubMed] [Google Scholar]
- Liversidge G. G.; Cundy K. C. Particle-size reduction for improvement of oral bioavailability of hydrophobic drugs. 1. Absolute oral bioavailability of nanocrystalline danazol in beagle dogs. Int. J. Pharm. 1995, 125, 91–97. [Google Scholar]
- Guzman K. A. D.; Taylor M. R.; Banfield J. F. Environmental risks of nanotechnology: National nanotechnology initiative funding, 2000–2004. Environ. Sci. Technol. 2006, 40, 1401–1407. [DOI] [PubMed] [Google Scholar]
- Maeda H.; Wu J.; Sawa T.; Matsumura Y.; Hori K. Tumor vascular permeability and the EPR effect in macromolecular therapeutics: a review. J. Controlled Release 2000, 65, 271–284. [DOI] [PubMed] [Google Scholar]
- Davis M. E.; Chen Z.; Shin D. M. Nanoparticle therapeutics: an emerging treatment modality for cancer. Nat. Rev. Drug Discovery 2008, 7, 771–782. [DOI] [PubMed] [Google Scholar]
- Bertrand N.; Wu J.; Xu X.; Kamaly N.; Farokhzad O. C. Cancer nanotechnology: the impact of passive and active targeting in the era of modern cancer biology. Adv. Drug Delivery Rev. 2014, 66, 2–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu X. S.; Li H.; Chen Y. J.; Jin Q.; Ren K. F.; Ji J. Mixed-charge nanoparticles for long circulation, low reticuloendothelial system clearance, and high tumor accumulation. Adv. Healthcare Mater. 2014, 3, 1439–1447. [DOI] [PubMed] [Google Scholar]
- Loo C.; Lowery A.; Hala N. J.; West J. Immunotargeted nanoshells for integrated cancer imaging and therapy. Nano Lett. 2005, 5, 709–711. [DOI] [PubMed] [Google Scholar]
- Huang X.; El-Sayed E. H.; Qian W.; El-Sayed M. A. Cancer cell imaging and photothermal therapy in the near-infrared region by using gold nanorods. J. Am. Chem. Soc. 2006, 128, 2115–2120. [DOI] [PubMed] [Google Scholar]
- Gao X. H.; Cui Y. Y.; Leveson R. M.; Chung L. W. K.; Nie S. In vivo cancer targeting and imaging with semiconductor quantum dots. Nat. Biotechnol. 2004, 22, 969–976. [DOI] [PubMed] [Google Scholar]
- McCarthy J. R.; Weissleder R. Multifunctional magnetic nanoparticles for targeted imaging and therapy. Adv. Drug Delivery Rev. 2008, 60, 1241–1251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chaloupka K.; Malam Y.; Seifalian A. M. Nanosilver as a new generation of nanoproduct in biomedical applications. Trends Biotechnol. 2010, 28, 580–588. [DOI] [PubMed] [Google Scholar]
- Ryu J. H.; Lee S.; Son S.; Kim S. H.; Leary J. F.; Choi K.; Kwon I. C. Theranostic nanoparticles for future personalized medicine. J. Controlled Release 2014, 190, 477–484. [DOI] [PubMed] [Google Scholar]
- Gabizon A. A. PEGylated liposomal doxorubicin: metamorphosis of an old drug into a new form of chemotherapy. Cancer Invest. 2001, 19, 424–436. [DOI] [PubMed] [Google Scholar]
- Rehman Z.; Hoekstra D.; Zuhorn I. Mechanism of polylplex- and lipoplex-mediated delivery of nucleic acids: real-time visualization of transient membrane destabilization without endosomal lysis. ACS Nano 2013, 7, 3767–3777. [DOI] [PubMed] [Google Scholar]
- Stinchcombe T. E. Nanoparticle albumin-bound paclitazel: a novel Cremphor-EL(R) −free formulation of paclitaxel. Nanomedicine 2007, 2, 415–423. [DOI] [PubMed] [Google Scholar]
- Yildirimer L.; Thanh N. T. K.; Loizidou M.; Seifalian A. M. Toxicological considerations of clinically applicable nanoparticles. Nano Today 2011, 6, 586–607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jain S.; Bch M. B.; Hirst D. G.; O’Sullivan J. M. Gold nanoparticles as novel agents for cancer therapy. Br. J. Radiol. 2012, 85, 101–113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Samarasingha P. B.; Wijayasinghe A.; Behm M.; Dissanayake L.; Lindbergh G. Development of cathode materials for lithium ion rechargeable batteries based on the system Li(Ni1/3Mn1/3Co(1/3-x)Mx)O2, (M = Mg, Fe, Al and x=0.00 to 0.33). Solid State Ionics 2014, 268, 226–230. [Google Scholar]
- Shannon M. A.; Bohn P. W.; Elimelech M.; Georgiadis J. G.; Marinas B. J.; Mayes A. M. Science and technology for water purification in the coming decades. Nature 2008, 452, 301–301. [DOI] [PubMed] [Google Scholar]
- Khin M. M.; Nair S. A.; Babu V. J.; Murugan R.; Ramakrishna S. A review on nanomaterials for environmental remediation. Energy Environ. Sci. 2012, 5, 8075–8109. [Google Scholar]
- Talapin D. V.; Steckel J. Quantum-dot light-emitting devices. MRS Bull. 2013, 38, 685–691. [Google Scholar]
- Frohlich E. The role of surface charge in cellular uptake and cytotoxicity of medical nanoparticles. Int. J. Nanomed. 2012, 7, 5577–5591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nel A. E. Implementation of alternative test strategies for the safety assessment of nanomaterials. J. Int. Med. 2013, 274, 561–577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pulskamp K.; Diabate S.; Krug H. F. Carbon nanotubes show no sign of acute toxicity but induce intracellular reactive oxygen species in dependence on contaminants. Toxicol. Lett. 2007, 168, 58–74. [DOI] [PubMed] [Google Scholar]
- Alkilany A. M.; Nagaria P. K.; Hexel C. R.; Shaw T. J.; Murphy C. J.; Wyatt M. D. Cellular uptake and cytotoxicity of gold nanorods: Molecular origin of cytotoxicity and surface effects. Small 2009, 5, 701–708. [DOI] [PubMed] [Google Scholar]
- Lundqvist M.; Stigler J.; Elia G.; Lynch I.; Cedervall T.; Dawson K. A. Nanoparticle size and surface properties determine the protein corona with possible implications for biological impacts. Proc. Natl. Acad. Sci. U.S.A. 2008, 105, 14265–14270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang B.; Zhang L.; Bae S. C.; Granick S. Nanoparticle-induced surface reconstruction of phospholipid membranes. Proc. Natl. Acad. Sci. U.S.A. 2008, 105, 18171–18175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moghadam B. Y.; Hou W.-C.; Corredor C.; Westerhoff P.; Posner J. D. Role of nanoparticle surface functionality in the disruption of model cell membranes. Langmuir 2012, 28, 16319–16326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carney R. P.; Astier Y.; Carney T. M.; Voitchovsky K.; Silva P. H. J.; Stellacci F. Electrical method to quantify nanoparticle interaction with lipid bilayers. ACS Nano 2013, 7, 932–942. [DOI] [PubMed] [Google Scholar]
- Van Lehn R. C.; Alexander-Katz A. Free energy change for insertion of charged, monolayer-protected nanoparticles into lipid bilayers. Soft Matter 2014, 10, 648–658. [DOI] [PubMed] [Google Scholar]
- Troiano J. M.; Olenick L. L.; Kuech T. R.; Melby E. S.; Hu D.; Lohse S. E.; Mensch A. C.; Dogangun M.; Vartanian A. M.; Torelli M. D.; Ehimiaghe E.; Walter S. R.; Fu L.; Anderton C. R.; Zhu Z.; Wang H.; Orr G.; Murphy C. J.; Hamers R. J.; Pedersen J. A.; Geiger F. M. Direct probes of 4-nm diameter gold nanoparticles interacting with supported lipid bilayers. J. Phys. Chem. C 2015, 119, 534–546. [Google Scholar]
- Tadu H.; Higuchi H.; Wanatabe T. M.; Ohucji N. In vivo real-time tracking of single quantum dots conjugated with monoclonal anti-HER2 antibody in tumors of mice. Cancer Res. 2007, 67, 1138–1144. [DOI] [PubMed] [Google Scholar]
- Gu Y.; Wang G.; Fang N. Simultaneous single-particle superlocalization and rotational tracking. ACS Nano 2013, 7, 1658–1665. [DOI] [PubMed] [Google Scholar]
- Chien M. P.; Carlini A. S.; Hu D. S.; Barback C. V.; Rush A. M.; Hall D. J.; Orr G.; Gianneschi N. C. Enzyme-directed assembly of nanoparticles in tumors monitored by in vivo whole animal imaging and ex vivo super-resolution fluorescence imaging. J. Am. Chem. Soc. 2013, 135, 18710–18713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macias-Romero C.; Didier M. E. P.; Zubkovs V.; Delannoy L.; Dutto F.; Radenovic A.; Roke S. Probing rotational and translational diffusion of nanodoublers in living cells on microsecond time scales. Nano Lett. 2014, 14, 2552–2557. [DOI] [PubMed] [Google Scholar]
- Chung C.-Y.; Boik J.; Potma E. O. Biomolecular imaging with coherent nonlinear vibrational microscopy. Annu. Rev. Phys. Chem. 2013, 64, 77–99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alkilany A. M.; Murphy C. J. Toxicity and cellular uptake of gold nanoparticles: What have we learned so far?. J. Nanopart. Res. 2010, 12, 2313–2333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khlebtsov N.; Dykman L. Biodistribution and toxicity of gold nanoparticles: a review of in vitro and in vivo studies. Chem. Soc. Rev. 2011, 40, 1647–1671. [DOI] [PubMed] [Google Scholar]
- Bondarenko O.; Juganson K.; Ivask A.; Kasemets K.; Mortimer M.; Kahur A. Toxicity of Ag, CuO and ZnO nanoparticles to selected environmentally relevant test organisms and mammalian cells in vitro: a critical review. Arch. Toxicol. 2013, 87, 1181–1200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klaper R.; Arndt D.; Bozich J.; Dominguez G. Molecular interactions of nanomaterials and organisms: defining biomarkers for toxicity and high-throughput screening using traditional and next-generation sequencing approaches. Analyst 2014, 139, 882–895. [DOI] [PubMed] [Google Scholar]
- Xia T.; Hamilton R. F. Jr.; Bonner J. C.; Crandall E. D.; Elder A.; Fazlollahi F.; Girtsman T. A.; Kim K.; Mitra S.; Ntim S. A.; Orr G.; TAgmount M.; Taylor A. J.; Telesca D.; Tolic A.; Vulpe C. D.; Walker A. J.; Wang X.; Witzmann F. A.; Wu N.; Xie Y.; Zink J. I.; Nel A. E.; Holian A. Interlaboratory evaluation of in vitro cytotoxicity and inflammatory responses to engineering nanomaterials: the NIEHS Nano GO consortium. Environ. Health Perspect. 2013, 121, 683–690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsoi K. M.; Dai Q.; Alman B. A.; Chan W. C. W. Are quantum dots toxic? Exploring the discrepancy between cell culture and animal studies. Acc. Chem. Res. 2013, 46, 662–671. [DOI] [PubMed] [Google Scholar]
- Sisco P. N.; Wilson C. G.; Chernak D.; Clark J. C.; Grzinic E. M.; Ako-Asare K.; Goldsmith E. C.; Murphy C. J. Adsorption of cellular proteins to polyelectrolyte-functionalized gold nanorods: a mechanism for nanoparticle regulation of cell phenotype?. PLoS One 2014, 9, e86670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kong L. J.; Tuomela S.; Hahne L.; Ahlfors H.; Yli-Harja O.; Fadeel B.; Lahesmaa R.; Autio R. NanoMiner—integrative human transcriptomics data resource for nanoparticle research. PLoS One 2013, 8, e68414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grzincic E. M.; Yang J. A.; Drnevich J.; Falagan-Lotsch P.; Murphy C. J. Global transcriptomic analysis of model human cell lines exposed to surface-modified gold nanoparticles: the effect of surface chemistry. Nanoscale 2015, 7, 1349–1362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hauck T. S.; Ghazani A. A.; Chan W. C. W. Assessing the effect of surface chemistry on gold nanorod uptake, toxicity, and gene expression in mammalian cells. Small 2008, 4, 153–159. [DOI] [PubMed] [Google Scholar]
- Lin S.; Zhao Y.; Ji Z.; Ear J.; Chang C. H.; Zhang H.; Low-Kam C.; Yamada K.; Meng H.; Wang X.; Liu R.; Pokhrel S.; Maedler L.; Damoiseaux R.; Xia T.; Godwin H. A.; Lin S.; Nel A. E. Zebrafish high-throughput screening to study the impact of dissolvable metal oxide nanoparticles on the hatching enzyme ZHE1. Small 2013, 9, 1776–1785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu R.; France B.; George S.; Rallo R.; Zhang H.; Xia T.; Nel A. E.; Bradley K.; Cohen Y. Association rule mining of cellular responses induced by metal and metal oxide nanoparticles. Analyst 2014, 139, 943–953. [DOI] [PubMed] [Google Scholar]
- Albanese A.; Walkey C. D.; Olsen J. B.; Guo H. B.; Emil A.; Chan W. C. W. Secreted biomolecules alter the biological identity and cellular interactions of nanoparticles. ACS Nano 2014, 8, 5515–5526. [DOI] [PubMed] [Google Scholar]
- Liu Y.; Zhao Y.; Sun B.; Chen C. Understanding the toxicity of carbon nanotubes. Acc. Chem. Res. 2013, 46, 702–713. [DOI] [PubMed] [Google Scholar]
- Arndt D. A.; Moua M.; Chen J.; Klaper R. D. Core structure and surface functionalization of carbon nanomaterials alter impacts to daphnid mortality, reproduction, and growth: Acute assays to do not predict chronic exposure impacts. Environ. Sci. Technol. 2013, 47, 9444–9452. [DOI] [PubMed] [Google Scholar]
- Bozich J. S.; Lohse S. E.; Torelli M. D.; Murphy C. J.; Hamers R. J.; Klaper R. D. Surface chemistry, charge and ligand type impact the toxicity of gold nanoparticles to Daphnia magna. Environ. Sci.: Nano 2014, 1, 260–270. [Google Scholar]
- Yang X. Y.; Gondikas A. P.; Marinakos S. M.; Auffan M.; Liu J.; Hsu-Kim H.; Meyer J. N. Mechanism of silver nanoparticle toxicity is dependent on dissolved silver and surface coating in Caenorhabditis elegans. Environ. Sci. Technol. 2012, 46, 1119–1127. [DOI] [PubMed] [Google Scholar]
- Bour A.; Mouchet F.; Verneuil L.; Evariste L.; Silvestro J.; Pinelli E.; Gauthier L. Toxicity of CeO2 nanoparticles at different trophic levels: effects on diatoms, chironomids, and amphibians. Chemosphere 2015, 120, 230–236. [DOI] [PubMed] [Google Scholar]
- Leifert A.; Pan-Bartnek Y.; Simon U.; Jahnen-Dechent W. Molecularly stabilized ultrasmall gold nanoparticles: synthesis, characterization and bioactivity. Nanoscale 2013, 6, 6224–6242. [DOI] [PubMed] [Google Scholar]
- Gao D.; Zhou H.; Wang J.; Miao S.; Yang F.; Wang G.; Wang J.; Bao X. Size-dependent electrocatalytic reduction of CO2 over Pd nanoparticles. J. Am. Chem. Soc. 2015, 137, 4288–4291. [DOI] [PubMed] [Google Scholar]
- Hafner J. Ab initio simulations of materials using VASP: Density-functional theory and beyond. J. Comput. Chem. 2008, 29, 2044–2078. [DOI] [PubMed] [Google Scholar]
- Huang P.; Carter E. A. Advances in correlated electronic structure methods for solids, surfaces and nanostructures. Annu. Rev. Phys. Chem. 2008, 59, 261–290. [DOI] [PubMed] [Google Scholar]
- Donnini S.; Tegeler F.; Groenhof G.; Grubmuller H. Constant pH molecular dynamics in explicit solvent with lambda-dynamics. J. Chem. Theory Comput. 2011, 7, 1962–1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mori T.; Hamers R. J.; Pedersen J. A.; Cui Q. An explicit consideration of desolvation is critical to binding free energy calculations of charged molecules at ionic surfaces. J. Chem. Theory Comput. 2013, 9, 5059–5069. [DOI] [PubMed] [Google Scholar]
- Zuckerman D. M. Equilibrium sampling in biomolecular simulations. Annu. Rev. Biophys. 2011, 40, 41–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozer G.; Keyes T.; Quirk S.; Hernandez R. Multiple branched adaptive steered molecular dynamics. J. Chem. Phys. 2014, 141, 064101. [DOI] [PubMed] [Google Scholar]
- Ozer G.; Valeev E. F.; Quirk S.; Hernandez R. Adaptive steered molecular dynamics of the long-distance unfolding of neuropeptide y. J. Chem. Theory Comput. 2010, 6, 3026–3038. [DOI] [PubMed] [Google Scholar]
- Wu Z.; Cui Q.; Yethiraj A. A new coarse-grained force field for membrane-peptide simulations. J. Chem. Theory Comput. 2011, 7, 3793–3802. [DOI] [PubMed] [Google Scholar]
- Marrink S. J.; Tieleman D. P. Perspective on the Martini model. Chem. Soc. Rev. 2013, 42, 6801–6822. [DOI] [PubMed] [Google Scholar]
- Van Lehn R. C.; Alexander-Katz A. Penetration of lipid bilayers by nanoparticles with environmentally-responsive surfaces: simulations and theory. Soft Matter 2011, 7, 11392–11404. [Google Scholar]
- Hernandez R.; Popov A. V. Molecular dynamics out of equilibrium: mechanics and measurables. Wiley Interdiscip. Rev.: Comput. Mol. Sci. 2014, 4, 541–561. [Google Scholar]
- Heikkilä E.; Martinez-Seara H.; Gurtovenko A. A.; Javanainen M.; Häkkinen H.; Vattulainen I.; Akola J. Cationic Au nanoparticle binding with plasma membrane-like lipid bilayers: potential mechanism for spontaneous permeation to cells revealed by atomistic simulations. J. Phys. Chem. C 2014, 118, 11131–11141. [Google Scholar]
- Zhao G.; Perilla J. R.; Yufenyuy E. L.; Meng X.; Chen B.; Ning J.; Ahn J.; Gronenborn A. M.; Schulten K.; Aiken C.; Zhang P. Mature HIV-1 capsid structure by cryo-electron microscopy and all-atom molecular dynamics. Nature 2013, 497, 643–646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mockl L.; Lamb D. C.; Brauchle C. Super-resolved fluorescence microscopy: Nobel Prize in Chemistry 2014 for Eric Betzig, Stefan Hell, and William E. Moerner. Angew. Chem., Int. Ed. 2014, 53, 13972–13977. [DOI] [PubMed] [Google Scholar]
- Jones S. A.; Shim S. H.; He J.; Zhuang X. W. Fast, three-dimensional super-resolution imaging of live cells. Nat. Methods 2011, 8, 499–505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai Z. W.; Yan Y.; Caruso F.; Nice E. C. Emerging techniques in proteomics for probing nano–bio interactions. ACS Nano 2012, 6, 10438–10448. [DOI] [PubMed] [Google Scholar]
- Jia L.; Lu J.; Shao J.; Liang X.; Xu Y. Nanoproteomics: a new sprout from emerging links between nanotechnology and proteomics. Trends Biotechnol. 2013, 31, 99–107. [DOI] [PubMed] [Google Scholar]