Abstract
The endogenous signaling gasotransmitter, hydrosulfide (H2S), has been shown to exert cardioprotective effects against acute myocardial infarction (AMI) due to ischemic injury. However, the mechanisms responsible for these effects are not yet fully understood. In this study, we investigated whether sodium hydrogen sulfide (NaHS), an H2S donor, attenuates acute myocardial ischemic injury through glycogen synthase kinase-3β (GSK-3β)/β-catenin signaling. For this purpose, we utilized an in vivo rat model of AMI by occluding the left anterior descending coronary artery. NaHS (0.39, 0.78 or 1.56 mg/kg, intraperitoneally), the GSK-3β inhibitor, SB216763 (0.6 mg/kg, intravenously), or 1% dimethylsulfoxide (2 ml/kg, intravenously) were administered to the rats. The results demonstrated that the administration of medium- and high-dose NaHS and SB216763 significantly improved rat cardiac function, as evidenced by an increase in the mean arterial pressure, left ventricular developed pressure, contraction and relaxation rates, as well as a decrease in left ventricular end-diastolic pressure. In addition, the administration of NaHS and SB216763 attenuated myocardial injury as reflected by a decrease in apoptotic cell death and in the serum lactate dehydrogenase concentrations, and prevented myocardial structural changes. The administration of NaHS and SB216763 increased the concentrations of phosphorylated (p-)GSK-3β, the p-GSK-3β/t-GSK-3β ratio and downstream protein β-catenin. Moreover, western blot and immunohistochemical analyses of apoptotic signaling pathway proteins further established the cardioprotective potential of NaHS, as reflected by the upregulation of Bcl-2 expression, the downregulation of Bax expression, and a decrease in the number of TUNEL-positive stained cells. These findings suggest that hydrosulfide exerts cardioprotective effects against AMI-induced apoptosis through the GSK-3β/β-catenin signaling pathway.
Keywords: acute myocardial ischemia, hydrosulfide, glycogen synthase kinase-3β, SB216763, β-catenin, apoptosis
Introduction
It is well known that coronary disease is the leading cause of mortality in the modern world (1). It is characterized by transient or chronic myocardial ischemia that is caused by reduced coronary blood flow and tissue hypoxia. Ischemia plays a critical role in cardiomyocyte necrotic and apoptotic death in the border area close to a myocardial infarct area and greatly affects left ventricular remodeling, which causes heart failure and mortality (2). Apoptosis, which is determined by a balance between pro- and anti-apoptotic factors, is frequently detected in ischemic heart tissue (3). Attenuating the induction of apoptosis that is triggered by tissue hypoxia is a major strategy for treating ischemic heart disease.
One of the several signaling pathways that are involved in the apoptotic process is glycogen synthase kinase-3β (GSK-3β), which belongs to a family of conserved serine/threonine kinases present in eukaryotic cells. Its activity is regulated by various pathways, including the phosphatidylinositol-3-kinase (PI3K)/Akt, the extracellular signal-regulated kinase (ERK1/2) and the Wnt/wingless signaling pathways (4–6). GSK-3β phosphorylation at Ser9 causes its N-terminal protein tail to act as a pre-phosphorylated substrate, leading to GSK-3β inactivation, and thus it differs from other protein kinases (7). Moreover, the selective inhibition of GSK-3β has been shown to exert cardioprotective effects by maintaining mitochondrial function during ischemia/reperfusion injury (4). Together with adenomatous polyposis coli (APC) and Axin, GSK-3β forms a cytoplasmic multiprotein complex that can phosphorylate β-catenin, an important GSK-3β-regulated protein, leading to β-catenin degradation and limited β-catenin expression (8).
As with nitric oxide (NO) and carbon monoxide (CO), hydrosulfide (H2S) is the body's third gaseous signaling molecule (9–12). There is increasing evidence indicating that H2S exerts cardioprotective effects (13,14), such as the relaxation of vascular smooth muscle, the reduction of blood pressure (15), the inhibition of vascular smooth muscle cell proliferation, and the regulation of cardiac contractility (16). Sodium hydrogen sulfide (NaHS) is frequently used as an H2S donor (17). In addition, it has also been proposed that, in a model of myocardial ischemia, the protective effects of NaHS on myocardial tissues may be associated with its anti-inflammatory and anti-apoptotic properties (18,19); however, the underlying mechanisms remain unclear. This study was designed to determine whether NaHS exerts its cardioprotective effects through the activation of the GSK-3β/β-catenin signaling pathway. SB216763 was used as a GSK-3β inhibitor.
Materials and methods
Animals
Male Sprague-Dawley (SD) rats (n=96, provided by the Center of Experimental Animals of Hebei Province, China) with a body weight of 280–320 g were housed in a temperature-controlled environment, exposed to a natural photoperiod (light/dark cycle of 12:12 h), and were allowed access to food and water ad libitum. All rats were housed for 1 week to adapt to their environment prior to the initiation of the expriments. The study was approved and carried out in accordance with the guidelines of Hebei Medical University. The experimental protocols were performed in adherence with the Institutional Animal Care and Use Committee of Hebei Medical University.
Chemicals and reagents
NaHS was purchased from Sigma Chemical Co. (St. Louis, MO, USA). The antibodies against phosphorylated (p-)GSK-3β (#9323), total (t-)GSK-3β (#12456) and β-catenin (#8480) were obtained from Cell Signaling Technology (Beverly, MA, USA). Antibodies against Bax (sc-526), Bcl-2 (sc-492) and β-actin (sc-7210) were obtained from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA, USA). SB216763 (purity >98%) was purchased from Selleck Chemicals (Houston, TX, USA). The TUNEL apoptosis kit was obtained from Roche Diagnostics (Boehringer Mannheim, Germany). The lactate dehydrogenase (LDH) assay kit was purchased from Nanjing Jiancheng Biotechnology (Nanjing, China).
Experimental design and surgical procedures
A total of 40 rats were randomly assigned to 5 groups as follows: i) the sham-operated group; ii) the group subjected to ischemia for 2 h (Is2h group); iii) the group subjected to ischemia for 3 h (Is3h group); iv) the group subjected to ischemia for 6 h (Is6h group); and v) the group subjected to ischemia for 9 h (Is9h group). The protein expression levels of GSK-3β and β-catenin were then determined by western blot analysis as described below.
The remaining 56 rats were randomly assigned to one of the following treatment groups (n=8/group) as follows: i) the sham-operated group; ii) the group subjected to acute myocardial infarction (AMI; ischemia) and not treated with any agents (AMI group); iii) the group subjected to AMI and treated with low-dose NaHS (AMI + NaHS-L group); iv) the group subjected to AMI and treated with medium-dose NaHS (AMI + NaHS-M group); v) the group subjected to AMI and treated with high-dose NaHS (AMI + NaHS-H group); vi) the group subjected to AMI and treated with SB216763 (AMI + SB group); and vii) the group subjected to AMI and treated with the vehicle (AMI + vehicle group).
The rats were subjected to ligation of the anterior descending artery as previously described (20,21), resulting in acute myocardial ischemic injury. In brief, the rats were anesthetized with ketamine-xylazine (90 and 9 mg/kg, respectively, intraperitoneally), and intubated with a 14-gauge polyethylene catheter and ventilated with room air using a small animal ventilator. The heart was exposed via a left-sided thoracotomy, and the anterior descending artery was ligated using a 6–0 silk suture between the pulmonary outflow tract and the left atrium. The sham-operated rats underwent the same procedure except that the suture was passed under the coronary artery and then removed. The rats in the Is2h, Is3h, Is6h and Is9h groups were sacrificed at 2, 3, 6 and 9 h after ischemia, respectively. The rats in the treatment groups were sacrificed at 6 h after ischemia.
NaHS (0.78, 1.56, 3.12 mg/kg, intraperitoneally) was respectively administered to the rats in the AMI + low-, medium-, and high-dose NaHS groups, SB216763 (0.6 mg/kg, dissolved in 1% dimethylsulfoxide, intravenously) was administered to the rats in the AMI + SB group, and 1% dimethyl sulfoxide (DMSO; 2 ml/kg, intravenously) was administered to the rats in the AMI + vehicle group. The doses of NaHS and SB216763 were established from prior investigations (22–26).
Determination of cardiac function
The rats were anesthetized, and a polyethylene Millar catheter was inserted into the right common carotid artery and then further advanced into the left ventricular chamber, and the cannula was connected to a pressure transducer. The mean arterial pressure (MAP), left ventricular developed pressure (LVDP), the left ventricular end-diastolic pressure (LVEDP) and the rate of contraction and relaxation (dp/dtmax and dp/dtmin) were recorded using an 8-channel polygraph system (Powerlab 8s/30 ADInstruments, Castle Hill, NSW, Australia).
Measurement of serum LDH levels
Myocardial cellular damage and necrosis were evaluated by measuring the serum LDH levels. Blood samples were collected after the measurement of cardiac function. The LDH levels were measured in a blinded manner using a spectrophotometer (BioTek, Instruments, Inc., Winooski, VT, USA) in duplicate.
Morphological changes in the myocardium
The rats were sacrificed by exsanguination (blood was taken from rats via the right carotid artery). The thoracic cavity was opened and rat hearts were obtained and washed with cold physiological saline. The non-infarcted left ventricular tissue was separated for determination. The non-infarcted left ventricular tissues were fixed in 4% paraformaldehyde and then dehydrated in ascending grades of alcohol and embedded in paraffin, and sliced into 5-µm-thick sections using a microtome (RM2245; Leica Biosystems, Buffalo Grove, IL, USA). The myocardial sections (5-µm-thick) were stained with hematoxylin and eosin (H&E) to analyze the morphological changes in the myocardium.
Terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick-end labeling (TUNEL) assay
The apoptosis of cardiomyocytes was detected by TUNEL assay. Briefly, the apoptotic cells from the heart tissues were identified using an in situ cell death detection kit according to the manufacturer's instructions. Nuclei with brown staining indicated TUNEL-positive cells. For each group, 20 randomly selected fields (5 hearts/group, 4 fields/heart) were observed under a microscope. The apoptotic index (AI), namely the percentage of apoptotic nuclei (TUNEL-positive) vs. the total number of nuclei was measured.
Western blot analysis of p-GSK-3β, total GSK-3β, β-catenin, Bax, Bcl-2 and β-actin
The hearts were excised and the left ventricles were immediately frozen in liquid nitrogen before being stored at −80°C. Total protein from cardiomyocytes was extracted and the concentrations were determined by using the BCA protein assay kit (Boster Biotechnology, Wuhan, China). Proteins were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and then transferred onto a polyvinylidene difluoride (PVDF) membrane, blocked, and probed with primary antibodies against p-GSK-3β, t-GSK-3β, β-catenin, Bax, Bcl-2 and β-actin. The membrane was then incubated with horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG. Finally, the bound antibody complexes were detected using the ECL Western blotting detection kit (Beyotime Institute of Biotechnology, Nanjing, China).
Immunohistochemical staining
Immunohistochemical staining for β-catenin, Bcl-2 and Bax was performed on the myocardial tissue. The non-infarcted left ventricular tissue was deparaffinized, rehydrated in a graded series of alcohol solutions, and washed twice with distilled water. The sections were incubated with endogenous peroxidase blocked in 50 ml of 3% H2O2 at room temperature for 10 min and then washed with phosphate-buffered saline (PBS) (pH 7.4) 3 times for 5 min each. Following incubation in 2% BSA in PBS at room temperature for 30 min, the sections were washed once with PBS. The β-catenin, Bcl-2 or Bax antibodies were added, and the sections were incubated at 4°C overnight. The β-catenin, Bcl-2 and Bax proteins were assayed with an UltraSensitive SP kit (ZSGB-BIO, Beijing, China). The sections were counterstained with hematoxylin. The incubation of the tissue sections with normal rabbit IgG served as the negative control. The cardiomyocytes were seeded on slides, and then counterstained with hematoxylin, mounted with DPX (Solarbio, Beijing, China) and visualized under a microscope (Olympus BX3-CBH, Olympus, Tokyo, Japan).
Statistical analysis
All data are presented as the means ± standard error of mean (SEM). Statistical analyses were performed using the SPSS statistical package (SPSS, version 16.0; SPSS Inc., Chicago, IL, USA). Differences between the groups of rats were analyzed by one-way ANOVA. Differences between 2 groups were analyzed using the Student-Newman-Keuls test. Probability values <0.05 were considered to indicate statistically significant differences.
Results
Protein expression of GSK-3β and β-catenin in rats with AMI
As shown by western blot analysis, no significant difference was observed in the total GSK-3β expression levels between the rats in the sham-operated group and the rats subjected to ishemia for various periods of time (Fig. 1A and B). However, the levels of p-GSK-3β and the p-GSK-3β/t-GSK-3β ratio were significantly decreased in the rats subjected to ischemia for 2, 3, 6 and 9 h compared with the rats in the sham-operated group when they were normalized to the GSK-3β total protein level, as illustrated in Fig. 1B and C (p<0.01). The levels of β-catenin, a downstream target of GSK-3β, were also significantly decreased in the rats subjected to ischemia for 2, 3, 6 and 9 h compared with those of the rats in the sham-operated group (p<0.05 and p<0.01; Fig. 1D). In addition, H&E staining of the rat hearts from the Is2h, Is3h, Is6h and Is9h groups showed that cardiomyocyte mecrosis, edema and inflammatory cell infiltration were observed in the Is6h and Is9h groups (data not shown). Thus, in the following experiments, the rats were subjected to ischemia for 3 h and sacrificed at 6 h after ischemia.
Figure 1.
Expression of phosphorylated (p-)glycogen synthase kinase-3β (GSK-3β), total (t-)GSK-3β and β-catenin in rats with acute myocardial infarction. Rats were subjected to ischemia for 2, 3, 6 or 9 h (Is2h, Is3h, Is6h and Is9h, respectively) as described in the Materials and methods. (A) Western blot analysis of the indicated proteins. Quantification of (B) p-GSK-3β and t-GSK-3β, (C) p-GSK-3β/t-GSK-3β ratio, and (D) β-catenin expression relative to actin. Data are expressed as the means ± SEM. *p<0.05 and **p<0.01 vs. sham-operated group.
Effect of hydrosulfide on cardiac function in rats with AMI
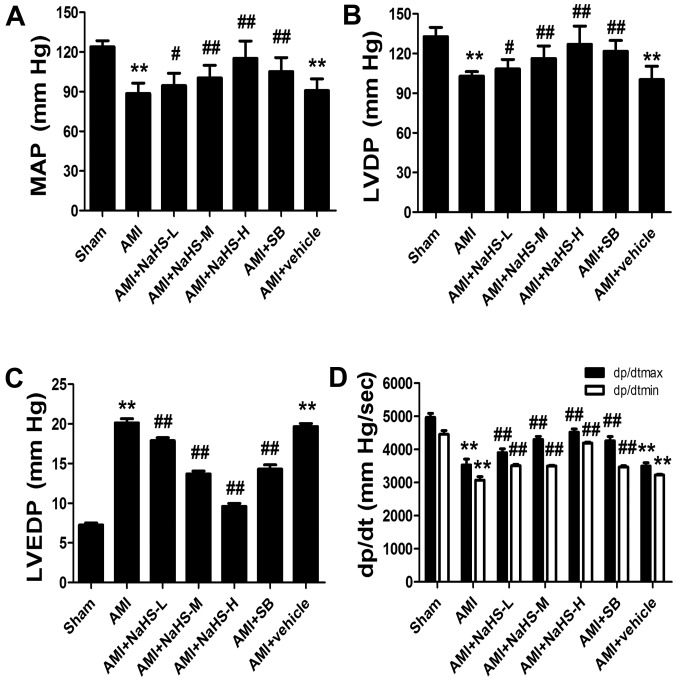
The MAP, LVDP and maximal positive and negative rate of left ventricular pressure (dp/dtmax, dp/dtmin) were decreased, and LVEDP was increased in the rats in the AMI group compared with the rats in the sham-operated group. However, the MAP, LVDP, dp/dtmax and dp/dtmin were significantly increased, and LVEDP was significantly decreased in the rats in the AMI + low-, medium- and high-dose NaHS groups compared with the rats in the AMI group (p<0.01; Fig. 2). Treatment with SB216763 had similar effects, increasing MAP, LVDP and dp/dtmax and dp/dtmin, and decreasing LVEDP.
Figure 2.
Effect of sodium hydrosulfide on cardiac function in rats with acute myocardial infarction (AMI). (A) Mean arterial pressure (MAP), (B) left ventricular developed pressure (LVDP), (C) left ventricular end diastolic pressure (LVEDP), and (D) maximal positive and negative rate of left ventricular pressure (dp/dtmax, dp/dtmin). Data are expressed as the means ± SEM. (n=8 rats/group). **p<0.01 vs. sham-operated group; #p<0.05 and ##p<0.01 vs. AMI group. Sham, sham-operated group; AMI, rats subjected to AMI (ischemia) and not treated with any agents; AMI + NaHS-L, rats subjected to AMI and treated with low-dose (0.78 mg/kg) NaHS; AMI + NaHS-M, rats subjected to AMI and treated with medium-dose (1.56 mg/kg) NaHS; AMI + NaHS-H, rats subjected to AMI and treated with high-dose (03.12 mg/kg) NaHS; AMI + SB, rats subjected to AMI and treated with the glycogen synthase kinase-3β (GSK-3β) inhibitor, SB216763 (0.6 mg/kg); AMI + vehicle, rats subjected to AMI and treated with the vehicle (DMSO, 2 ml/kg).
Effect of hydrosulfide on myocardial injury (histopathology, necrosis and apoptosis) in rats with AMI
The disorganization of cell structures, the loss of cell-cell adherence between cardiomyocytes and abnormally shaped cell nuclei were observed in the rat hearts from the AMI group compared with those of the rats in the sham-operated group. Compared with the hearts of the rats in the AMI group, cardiomyocyte necrosis, edema and inflammatory cell infiltration into the myocardium were significantly decreased in the rats in the AMI + low-, medium-, high-dose NaHS groups (Fig. 3A). Treatment with SB216763 had similar effects to NaHS, attenuating myocardial injury.
Figure 3.
Effect of sodium hydrosulfide on rat myocardial tissue necrosis and cell apoptosis. (A) Hematoxylin and eosin (H&E) staining to determine histopathological changes under a light microscope (×20 magnification; scale bar, 100 µm). Circles indicate the disorganization of cell structures and inflammatory cell infiltration. (B) Serum lactate dehydrogenase (LDH) concentration, (C) cardiomyocyte apoptosis evaluated by TUNEL staining (×20 magnification; scale bar, 100 µm). (D) Quantification of TUNEL-positive nuclei. Data are expressed as the means ± SEM. **p<0.01 vs. sham-operated group; #p<0.05 and ##p<0.01 vs. acute myocardial infarction (AMI) group. Sham, sham-operated group; AMI, rats subjected to AMI (ischemia) and not treated with any agents; AMI + NaHS-L, rats subjected to AMI and treated with low-dose (0.78 mg/kg) NaHS; AMI + NaHS-M, rats subjected to AMI and treated with medium-dose (1.56 mg/kg) NaHS; AMI + NaHS-H, rats subjected to AMI and treated with high-dose (03.12 mg/kg) NaHS; AMI + SB, rats subjected to AMI and treated with the glycogen synthase kinase-3β (GSK-3β) inhibitor, SB216763 (0.6 mg/kg); AMI + vehicle, rats subjected to AMI and treated with the vehicle (DMSO, 2 ml/kg).
The serum LDH level was significantly elevated in the rats in the AMI group compared with the rats in the sham-operated group (P<0.01). Compared with the rats in the AMI group, the serum LDH level was significantly decreased in the rats in the AMI + low-, medium- and high-dose NaHS groups (P<0.05 and P<0.01; Fig. 3B). Therefore, treatment with the H2S donor, NaHS, decreased myocardial necrosis in vivo post-AMI. Treatment with SB216763 had similar effects to NaHS, significantly decreasing the serum LDH level (P<0.01) compared to the rats in the AMI group (Fig. 3B).
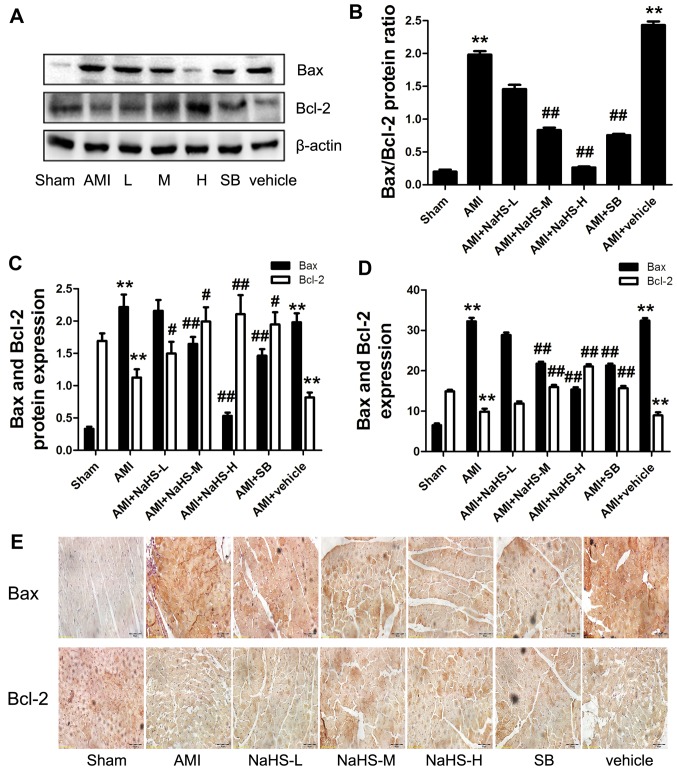
The expression of Bcl-2 was significantly increased and the Bax and Bax/Bcl-2 ratio was significantly decreased in the rats in the AMI + low-, medium-, high-dose NaHS groups compared with the rats in the AMI group (P<0.01; Fig. 4A–D). Moreover, the same results were shown by immunohistochemical analysis for Bax and Bcl-2 (Fig. 4E). Again, similar effects were observed following treatment with SB216763 (Fig. 4). In addition, as regards apoptosis, the number of TUNEL-positive cells was significantly increased in the AMI group compared with that of the sham-operated group. However, compared with the AMI group, the number of TUNEL-positive cardiac cells was significantly decreased in the AMI + low-, medium- high-dose NaHS groups and in the SB group (P<0.01; Fig. 3C and D). Taken together, these data suggest that treatment with the H2S donor, NaHS, decreases AMI-induced cardiomyocyte apoptosis in vivo.
Figure 4.
Effect of sodium hydrosulfide on apoptotic protein in rats with acute myocardial infarctiom (AMI). (A) Western blot analysis of the indicated proteins. Quantification of (B) Bax/Bcl-2 ratio, (C) Bax and Bcl-2. (D) Quantification of Bax and Bcl-2 immunohistochemical analysis, (E) immunohistochemical analysis of Bax and Bcl-2 (×40 magnification; scale bar, 50 µm). Data are expressed as the means ± SEM. **p<0.01 vs. sham-operated group; #p<0.05 and ##p<0.01 vs. AMI group. Sham, sham-operated group; AMI, rats subjected to AMI (ischemia) and not treated with any agents; AMI + NaHS-L, rats subjected to AMI and treated with low-dose (0.78 mg/kg) NaHS; AMI + NaHS-M, rats subjected to AMI and treated with medium-dose (1.56 mg/kg) NaHS; AMI + NaHS-H, rats subjected to AMI and treated with high-dose (03.12 mg/kg) NaHS; AMI + SB, rats subjected to AMI and treated with the glycogen synthase kinase-3β (GSK-3β) inhibitor, SB216763 (0.6 mg/kg); AMI + vehicle, rats subjected to AMI and treated with the vehicle (DMSO, 2 ml/kg).
Effect of hydrosulfide on the phosphorylation of GSK-3β and the concentration of the downstream target, β-catenin
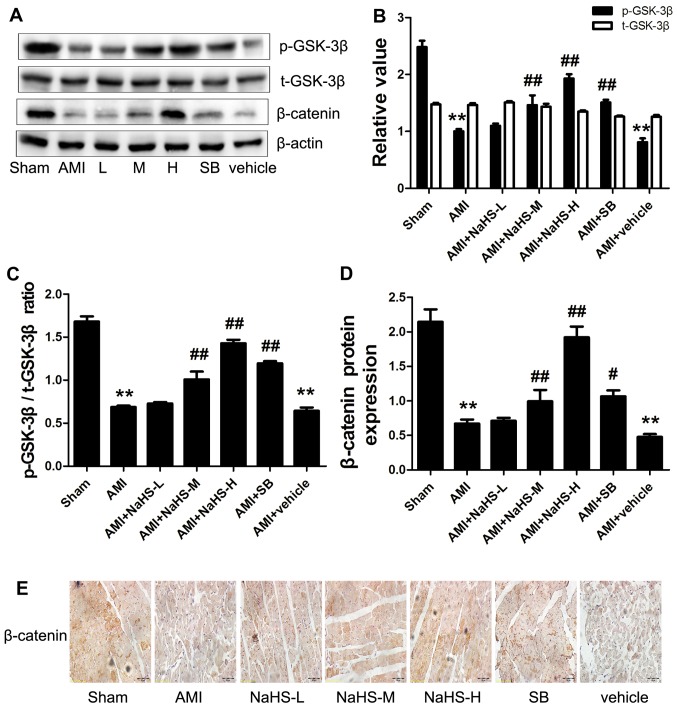
The p-GSK-3β/t-GSK-3β ratio and the content of β-catenin were significantly decreased in the AMI group compared with the sham-operated group. However, compared with the AMI group, the p-GSK-3β/t-GSK-3β ratio and the content of β-catenin were significantly increased in the AMI + low-, medium- and high-dose NaHS groups (P<0.01; Fig. 5). It should be noted that treatment with SB216763 had similar effects to NaHS (Fig. 5).
Figure 5.
Effect of sodium hydrosulfide on phosphorylation of glycogen synthase kinase-3β (GSK-3β) and β-catenin expression in rats with acute myocardial infarction (AMI). (A) Western blot analysis of the indicated proteins. Quantification of (B) p-GSK-3β and t-GSK-3β, (C) p-GSK-3β/t-GSK-3β ratio and (D) β-catenin relative to actin. (E) Immunohistochemical detection of β-catenin (×40 magnification; scale bar, 50 µm). All values are expressed as the means ± SEM. **p<0.01 vs. sham-operated group; #p<0.05 and ##p<0.01 vs. AMI group. Sham, sham-operated group; AMI, rats subjected to AMI (ischemia) and not treated with any agents; AMI + NaHS-L, rats subjected to AMI and treated with low-dose (0.78 mg/kg) NaHS; AMI + NaHS-M, rats subjected to AMI and treated with medium-dose (1.56 mg/kg) NaHS; AMI + NaHS-H, rats subjected to AMI and treated with high-dose (03.12 mg/kg) NaHS; AMI + SB, rats subjected to AMI and treated with the GSK-3β inhibitor, SB216763 (0.6 mg/kg); AMI + vehicle, rats subjected to AMI and treated with the vehicle (DMSO, 2 ml/kg).
Role of GSK-3β/β-catenin signaling in hydrosulfide-induced cardioprotection
To investigate the molecular mechanisms underlying NaHS-mediated cardioprotection, we measured the levels of p-GSK-3β and t-GSK-3β, and the p-GSK-3β/t-GSK-3β ratio and the content of β-catenin by western blot analysis. Compared with the AMI group, the phosphorylation levels of GSK-3β, the p-GSK-3β/t-GSK-3β ratio, and the content of β-catenin were significantly increased in the AMI + SB group (P<0.05 and P<0.01; Fig. 5). In addition, the MAP, LVDP, dp/dtmax and dp/dtmin were significantly increased, and the LVEDP, serum LDH level, the Bax/Bcl-2 ratio, and the number of apoptotic cells were significantly decreased in the AMI + SB group compared with the AMI group, as shown by our above-mentioned results (Figs. 2–4).
Discussion
In the present study, our data demonstrated that NaHS attenuated AMI-induced injury in vivo through the activation of the GSK-3β/β-catenin signaling pathway and exerted anti-apoptotic effects.
Evidence indicates that H2S plays an important role in vasodilation in different disease states (27–30), functions as a neuromodulator (31), and is also a novel mediator of lipopolysaccharide-induced inflammation (19,32). Furthermore, it is postulated that this gaseous signaling molecule acts as a regulator of physiological cardiac function by exerting antioxidant and anti-apoptotic effects in cardiovascular and neurological diseases (33–35). Cardiomyocyte apoptosis is a major pathogenic mechanism underlying AMI-induced injury; thus, inhibiting cardiomyocyte apoptosis may effectively minimize cardiac injury (36). As part of the intrinsic apoptosis pathway, the Bcl-2 protein family is an important regulator during cardiomyocyte apoptosis (37). The anti-apoptotic protein, Bcl-2, and survivin (38) have been shown to block the release of cytochrome c from the mitochondria, and to inhibit caspase activity and decrease cell apoptosis (5,37,39). Therefore, the balance in apoptotic signaling is influenced by the Bcl-2/Bax ratio. In this study, we demonstrated that the administration of NaHS and SB216763 (GSK-3β inhibitor) increased the expression of Bcl-2, decreased the expression of Bax, and decreased TUNEL-positive staining in rat heart tissue. The administration of NaHS significantly ameliorated cardiac function, as reflected by an increase in MAP, LVDP and dp/dtmax and dp/dtmin, and a decrease in LVEDP and the serum LDH level. Treatment with NaHS preserved myocardial structural integrity, thereby effectively restricting myocardial enzyme leakage.
The serine/threonine survival kinase GSK-3β is a point of downstream convergence for PI3K/Akt and Wnt signaling. It is known that ischemic pre- and post-conditioning activate PI3K/Akt (25,40–42), and promote the phosphorylation of downstream target molecules, such as GSK-3β, downstream of insulin receptor substrates (25,43,44). In contrast to other protein kinases, GSK-3β phosphorylation inhibits GSK-3β activity, thus inhibiting cardiomyocyte apoptosis, exerting cardioprotective effects (7,26). Kaga et al (7) reported that the activation of GSK-3β/β-catenin further increases Bcl-2 and survivin expression, thereby providing enhanced anti-apoptotic effects in the ischemic preconditioned myocardium. Therefore, we propose that the administration of NaHS exerts cardioprotective effects through the GSK-3β signaling pathway.
In the present study, our results revealed that AMI-induced injury led to decreased levels of p-GSK-3β and β-catenin. However, the administration of NaHS increased the phosphorylation levels of GSK-3β and the expression of β-catenin, and reduced cardiomyocyte apoptosis by regulating the expression of the Bcl-2 family proteins. A similar effect was also noted following the administration of SB216763, a potent inhibitor of the GSK-3β ATP binding site (45).
In conclusion, our data provide strong supportive evidence that the phosphorylation of GSK-3β by NaHS and its inhibitor, SB216763, exert cardioprotective and anti-apoptotic effects against AMI-induced injury through the activation of the GSK-3β/β-catenin signaling pathway. However, the specific subcellular mechanisms responsible for the NaHS-induced anti-apoptotic effects remain unknown, and this warrants further investigation.
Acknowledgments
This study was supported by the Basic Research Project of Hebei Province, China (no. 13967602D) and the Natural Science Foundation of Hebei Province, China (no. C2009001458).
References
- 1.Thom T, Haase N, Rosamond W, Howard VJ, Rumsfeld J, Manolio T, Zheng ZJ, Flegal K, O'Donnell C, Kittner S, et al. American Heart Association Statistics Committee and Stroke Statistics Subcommittee: Heart disease and stroke statistics - 2006 update: A report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2006;113:e85–e151. doi: 10.1161/CIRCULATIONAHA.105.171600. [DOI] [PubMed] [Google Scholar]
- 2.Nabel EG, Braunwald E. A tale of coronary artery disease and myocardial infarction. N Engl J Med. 2012;366:54–63. doi: 10.1056/NEJMra1112570. [DOI] [PubMed] [Google Scholar]
- 3.Chiong M, Wang ZV, Pedrozo Z, Cao DJ, Troncoso R, Ibacache M, Criollo A, Nemchenko A, Hill JA, Lavandero S. Cardiomyocyte death: Mechanisms and translational implications. Cell Death Dis. 2011;2:e244. doi: 10.1038/cddis.2011.130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Juhaszova M, Zorov DB, Kim SH, Pepe S, Fu Q, Fishbein KW, Ziman BD, Wang S, Ytrehus K, Antos CL, et al. Glycogen synthase kinase-3beta mediates convergence of protection signaling to inhibit the mitochondrial permeability transition pore. J Clin Invest. 2004;113:1535–1549. doi: 10.1172/JCI19906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nishihara M, Miura T, Miki T, Tanno M, Yano T, Naitoh K, Ohori K, Hotta H, Terashima Y, Shimamoto K. Modulation of the mitochondrial permeability transition pore complex in GSK-3beta-mediated myocardial protection. J Mol Cell Cardiol. 2007;43:564–570. doi: 10.1016/j.yjmcc.2007.08.010. [DOI] [PubMed] [Google Scholar]
- 6.Steinbrecher KA, Wilson W, III, Cogswell PC, Baldwin AS. Glycogen synthase kinase 3beta functions to specify gene-specific, NF-kappaB-dependent transcription. Mol Cell Biol. 2005;25:8444–8455. doi: 10.1128/MCB.25.19.8444-8455.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kaga S, Zhan L, Altaf E, Maulik N. Glycogen synthase kinase-3beta/beta-catenin promotes angiogenic and anti-apoptotic signaling through the induction of VEGF, Bcl-2 and survivin expression in rat ischemic preconditioned myocardium. J Mol Cell Cardiol. 2006;40:138–147. doi: 10.1016/j.yjmcc.2005.09.009. [DOI] [PubMed] [Google Scholar]
- 8.Liu J, Stevens J, Matsunami N, White RL. Targeted degradation of beta-catenin by chimeric F-box fusion proteins. Biochem Biophys Res Commun. 2004;313:1023–1029. doi: 10.1016/j.bbrc.2003.12.035. [DOI] [PubMed] [Google Scholar]
- 9.Abe K, Kimura H. The possible role of hydrogen sulfide as an endogenous neuromodulator. J Neurosci. 1996;16:1066–1071. doi: 10.1523/JNEUROSCI.16-03-01066.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wang R. Two's company, three's a crowd: Can H2S be the third endogenous gaseous transmitter? FASEB J. 2002;16:1792–1798. doi: 10.1096/fj.02-0211hyp. [DOI] [PubMed] [Google Scholar]
- 11.Chen YH, Yao WZ, Geng B, Ding YL, Lu M, Zhao MW, Tang CS. Endogenous hydrogen sulfide in patients with COPD. Chest. 2005;128:3205–3211. doi: 10.1378/chest.128.5.3205. [DOI] [PubMed] [Google Scholar]
- 12.Porokhya MV, Abramochkin DV, Abramov AA, Kuzmin VS, Sukhova GS. Inotropic effects of gaseous transmitters in isolated rat heart preparation. Bull Exp Biol Med. 2012;153:855–857. doi: 10.1007/s10517-012-1843-0. [DOI] [PubMed] [Google Scholar]
- 13.Hu X, Li T, Bi S, Jin Z, Zhou G, Bai C, Li L, Cui Q, Liu W. Possible role of hydrogen sulfide on the preservation of donor rat hearts. Transplant Proc. 2007;39:3024–3029. doi: 10.1016/j.transproceed.2007.05.086. [DOI] [PubMed] [Google Scholar]
- 14.Lavu M, Bhushan S, Lefer DJ. Hydrogen sulfide-mediated cardioprotection: Mechanisms and therapeutic potential. Clin Sci (Lond) 2011;120:219–229. doi: 10.1042/CS20100462. [DOI] [PubMed] [Google Scholar]
- 15.Sikora M, Drapala A, Ufnal M. Exogenous hydrogen sulfide causes different hemodynamic effects in normotensive and hypertensive rats via neurogenic mechanisms. Pharmacological Rep. 2014;66:751–758. doi: 10.1016/j.pharep.2014.04.004. [DOI] [PubMed] [Google Scholar]
- 16.Li L, Liu D, Bu D, Chen S, Wu J, Tang C, Du J, Jin H. Brg1-dependent epigenetic control of vascular smooth muscle cell proliferation by hydrogen sulfide. Biochim Biophys Acta. 18332013:1347–1355. doi: 10.1016/j.bbamcr.2013.03.002. [DOI] [PubMed] [Google Scholar]
- 17.Mostofa MG, Saegusa D, Fujita M, Tran LS. Hydrogen sulfide regulates salt tolerance in rice by maintaining Na(+)/K(+) balance, mineral homeostasis and oxidative metabolism under excessive salt stress. Front Plant Sci. 2015;6(1055) doi: 10.3389/fpls.2015.01055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sivarajah A, Collino M, Yasin M, Benetti E, Gallicchio M, Mazzon E, Cuzzocrea S, Fantozzi R, Thiemermann C. Anti-apoptotic and anti-inflammatory effects of hydrogen sulfide in a rat model of regional myocardial I/R. Shock. 2009;31:267–274. doi: 10.1097/SHK.0b013e318180ff89. [DOI] [PubMed] [Google Scholar]
- 19.Rios EC, Szczesny B, Soriano FG, Olah G, Szabo C. Hydrogen sulfide attenuates cytokine production through the modulation of chromatin remodeling. Int J Mol Med. 2015;35:1741–1746. doi: 10.3892/ijmm.2015.2176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bhindi R, Witting PK, McMahon AC, Khachigian LM, Lowe HC. Rat models of myocardial infarction. Pathogenetic insights and clinical relevance. Thromb Haemost. 2006;96:602–610. [PubMed] [Google Scholar]
- 21.Lee TM, Lin MS, Chang NC. Effect of ATP-sensitive potassium channel agonists on ventricular remodeling in healed rat infarcts. J Am Coll Cardiol. 2008;51:1309–1318. doi: 10.1016/j.jacc.2007.11.067. [DOI] [PubMed] [Google Scholar]
- 22.Liu F, Liu GJ, Liu N, Zhang G, Zhang JX, Li LF. Effect of hydrogen sulfide on inflammatory cytokines in acute myocardial ischemia injury in rats. Exp Ther Med. 2015;9:1068–1074. doi: 10.3892/etm.2015.2218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Johansen D, Ytrehus K, Baxter GF. Exogenous hydrogen sulfide (H2S) protects against regional myocardial ischemia-reperfusion injury - Evidence for a role of K ATP channels. Basic Res Cardiol. 2006;101:53–60. doi: 10.1007/s00395-005-0569-9. [DOI] [PubMed] [Google Scholar]
- 24.Chen QL, Gu EW, Zhang L, Cao YY, Zhu Y, Fang WP. Diabetes mellitus abrogates the cardioprotection of sufentanil against ischaemia/reperfusion injury by altering glycogen synthase kinase-3β. Acta Anaesthesiol Scand. 2013;57:236–242. doi: 10.1111/j.1399-6576.2012.02748.x. [DOI] [PubMed] [Google Scholar]
- 25.Gross ER, Hsu AK, Gross GJ. Diabetes abolishes morphine-induced cardioprotection via multiple pathways upstream of glycogen synthase kinase-3beta. Diabetes. 2007;56:127–136. doi: 10.2337/db06-0907. [DOI] [PubMed] [Google Scholar]
- 26.Obame FN, Plin-Mercier C, Assaly R, Zini R, Dubois-Randé JL, Berdeaux A, Morin D. Cardioprotective effect of morphine and a blocker of glycogen synthase kinase 3 beta, SB216763 [3-(2,4-dichlorophenyl)-4(1-methyl-1H-indol-3-yl)-1H-pyrrole-2,5-dione], via inhibition of the mitochondrial permeability transition pore. J Pharmacol Exp Ther. 2008;326:252–258. doi: 10.1124/jpet.108.138008. [DOI] [PubMed] [Google Scholar]
- 27.Zhao W, Zhang J, Lu Y, Wang R. The vasorelaxant effect of H(2)S as a novel endogenous gaseous K(ATP) channel opener. EMBO J. 2001;20:6008–6016. doi: 10.1093/emboj/20.21.6008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zhang Z, Huang H, Liu P, Tang C, Wang J. Hydrogen sulfide contributes to cardioprotection during ischemia-reperfusion injury by opening K ATP channels. Can J Physiol Pharmacol. 2007;85:1248–1253. doi: 10.1139/Y07-120. [DOI] [PubMed] [Google Scholar]
- 29.Chen SS, Tang CS, Jin HF, Du JB. Sulfur dioxide acts as a novel endogenous gaseous signaling molecule in the cardiovascular system. Chin Med J (Engl) 2011;124:1901–1905. [PubMed] [Google Scholar]
- 30.Yang G, Sun X, Wang R. Hydrogen sulfide-induced apoptosis of human aorta smooth muscle cells via the activation of mitogen-activated protein kinases and caspase-3. FASEB J. 2004;18:1782–1784. doi: 10.1096/fj.04-2279fje. [DOI] [PubMed] [Google Scholar]
- 31.Whiteman M, Cheung NS, Zhu YZ, Chu SH, Siau JL, Wong BS, Armstrong JS, Moore PK. Hydrogen sulphide: A novel inhibitor of hypochlorous acid-mediated oxidative damage in the brain? Biochem Biophys Res Commun. 2005;326:794–798. doi: 10.1016/j.bbrc.2004.11.110. [DOI] [PubMed] [Google Scholar]
- 32.Li L, Bhatia M, Zhu YZ, Zhu YC, Ramnath RD, Wang ZJ, Anuar FB, Whiteman M, Salto-Tellez M, Moore PK. Hydrogen sulfide is a novel mediator of lipopolysaccharide-induced inflammation in the mouse. FASEB J. 2005;19:1196–1198. doi: 10.1096/fj.04-3583fje. [DOI] [PubMed] [Google Scholar]
- 33.Tokuda K, Kida K, Marutani E, Crimi E, Bougaki M, Khatri A, Kimura H, Ichinose F. Inhaled hydrogen sulfide prevents endotoxin-induced systemic inflammation and improves survival by altering sulfide metabolism in mice. Antioxid Redox Signal. 2012;17:11–21. doi: 10.1089/ars.2011.4363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jiang LH, Luo X, He W, Huang XX, Cheng TT. Effects of exogenous hydrogen sulfide on apoptosis proteins and oxidative stress in the hippocampus of rats undergoing heroin withdrawal. Arch Pharm Res. 2011;34:2155–2162. doi: 10.1007/s12272-011-1220-y. [DOI] [PubMed] [Google Scholar]
- 35.Sodha NR1, Clements RT, Feng J, Liu Y, Bianchi C, Horvath EM, Szabo C, Sellke FW. The effects of therapeutic sulfide on myocardial apoptosis in response to ischemia-reperfusion injury. Eur J Cardiothorac Surg. 2008;33:906–913. doi: 10.1016/j.ejcts.2008.01.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Tian Y, Zhang W, Xia D, Modi P, Liang D, Wei M. Postconditioning inhibits myocardial apoptosis during prolonged reperfusion via a JAK2-STAT3-Bcl-2 pathway. J Biomed Sci. 2011;18(53) doi: 10.1186/1423-0127-18-53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kumar D, Jugdutt BI. Apoptosis and oxidants in the heart. J Lab Clin Med. 2003;142:288–297. doi: 10.1016/S0022-2143(03)00148-3. [DOI] [PubMed] [Google Scholar]
- 38.Lu F, Xing J, Zhang X, Dong S, Zhao Y, Wang L, Li H, Yang F, Xu C, Zhang W. Exogenous hydrogen sulfide prevents cardiomyocyte apoptosis from cardiac hypertrophy induced by isoproterenol. Mol Cell Biochem. 2013;381:41–50. doi: 10.1007/s11010-013-1686-7. [DOI] [PubMed] [Google Scholar]
- 39.Cook SA, Sugden PH, Clerk A. Regulation of bcl-2 family proteins during development and in response to oxidative stress in cardiac myocytes: Association with changes in mitochondrial membrane potential. Circ Res. 1999;85:940–949. doi: 10.1161/01.RES.85.10.940. [DOI] [PubMed] [Google Scholar]
- 40.Bharti S, Golechha M, Kumari S, Siddiqui KM, Arya DS. Akt/GSK-3β/eNOS phosphorylation arbitrates safranal-induced myocardial protection against ischemia-reperfusion injury in rats. Eur J Nutr. 2012;51:719–727. doi: 10.1007/s00394-011-0251-y. [DOI] [PubMed] [Google Scholar]
- 41.Bharti S, Singh R, Chauhan SS, Hussain T, Al-Attas OS, Arya DS. Phosphorylation of Akt/GSK-3β/eNOS amplifies 5-HT2B receptor blockade mediated anti-hypertrophic effect in rats. FEBS Lett. 2012;586:180–185. doi: 10.1016/j.febslet.2011.12.015. [DOI] [PubMed] [Google Scholar]
- 42.Lu Y, Zhou J, Xu C, Lin H, Xiao J, Wang Z, Yang B. JAK/STAT and PI3K/AKT pathways form a mutual transactivation loop and afford resistance to oxidative stress-induced apoptosis in cardiomyocytes. Cell Physiol Biochem. 2008;21:305–314. doi: 10.1159/000129389. [DOI] [PubMed] [Google Scholar]
- 43.Yadav HN, Singh M, Sharma PL. Involvement of GSK-3β in attenuation of the cardioprotective effect of ischemic preconditioning in diabetic rat heart. Mol Cell Biochem. 2010;343:75–81. doi: 10.1007/s11010-010-0500-z. [DOI] [PubMed] [Google Scholar]
- 44.Desrois M, Sidell RJ, Gauguier D, King LM, Radda GK, Clarke K. Initial steps of insulin signaling and glucose transport are defective in the type 2 diabetic rat heart. Cardiovasc Res. 2004;61:288–296. doi: 10.1016/j.cardiores.2003.11.021. [DOI] [PubMed] [Google Scholar]
- 45.Coghlan MP, Culbert AA, Cross DA, Corcoran SL, Yates JW, Pearce NJ, Rausch OL, Murphy GJ, Carter PS, Roxbee Cox L, et al. Selective small molecule inhibitors of glycogen synthase kinase-3 modulate glycogen metabolism and gene transcription. Chem Biol. 2000;7:793–803. doi: 10.1016/S1074-5521(00)00025-9. [DOI] [PubMed] [Google Scholar]