Abstract
Given the limited information available in this species, the aim of this study was to investigate the pharmacokinetic characteristics of enrofloxacin (ER) and its major metabolite ciprofloxacin (CP) in buffaloes, Bubalus bubalis. ER was administered intravenously (i.v.) or subcutaneously (s.c.) to buffaloes at doses of 5.0 and 7.5 mg/kg BW, and plasma, urine and fecal samples were collected until 48 hr post-administration. The concentrations of ER and CP in the plasma, urine and feces were analyzed using high-performance liquid chromatography equipped with a fluorescence detector. The plasma concentrations of ER and CP could be determined up to 24 hr and 32 hr after i.v. and s.c. administrations at doses of 5.0 and 7.5 mg/kg BW, respectively. CP concentrations were always lower than those of parental drug. The s.c. bioavailability of ER was 52.36 ± 4.24% and 72.12 ± 5.39% at doses of 5.0 and 7.5 mg/kg BW, respectively. Both ER and CP were detectable in urine and feces up to 24 hr. ER and CP were mainly excreted via the urine. Based on the pharmacokinetic data and PK-PD indices, s.c. administration of ER at doses of 5.0 and 7.5 mg/kg BW might be appropriate for the treatment of susceptible bacterial diseases in Thai swamp buffaloes.
Keywords: ciprofloxacin, enrofloxacin, pharmacokinetic, Thai swamp buffalo
Enrofloxacin (ER) is a synthetic bactericidal agent of the quinolones group, which acts by inhibiting the DNA gyrase enzyme, and it is predominantly used in veterinary practice for the treatment of respiratory and gastrointestinal infections. In both mammalian and non-mammalian species, it is partly de-ethylated to the main active metabolite ciprofloxacin (CP). Indeed, CP has been found as the metabolites that account almost 25–35% of the exposure to circulating parent drug [30]. It possesses antibacterial actions against a wide range of Gram-negative aerobes, a number of Gram-positive bacteria, mycoplasmas and some rickettsial micro-organisms [21]. ER has received growing attention, because of its potential efficacy and safety for the treatment of diseases in animals [28]. The dispositions of ER have been investigated in cattle [17, 18], sheep [25], horses [11, 13], pigs [3, 4, 24], rabbits [5], dogs [21], poultry [2] and exotic animals [12], and both intra- and inter-species differences in pharmacokinetics have been reported. For this reason, adequate information on the pharmacokinetics of ER in each species is pivotal for a successful therapeutic plan. Thus, the main aim of this study was to elucidate the disposition and s.c. bioavailability of ER in Thai swamp buffaloes, Bubalus bubalis, following i.v. and s.c. administration at two different doses.
MATERIALS AND METHODS
Standards and chemicals: Enrofloxacin (ER) (Baytril® 100, Bayer AG, Leverkuser, Germany) for injection was used. Enrofloxacin and ciprofloxacin standards were purchased from Sigma Chemical Co. (St. Louis, MO, U.S.A.). Other reagents and chemicals of analytical grade were purchased from Sigma Chemical Co. Purified water was produced using the Milli-Q water purification system from Millipore, Inc. (Bedford, MA, U.S.A.).
Animals: Ten female adult Thai swamp buffaloes between 5 and 10 years of age with an average body weight of 500 kg were selected. All ten buffaloes were housed together at the Animal Unit, Faculty of Natural Resources and Agro-industry, Chalermphrakiat Sakon Nakhon province campus, Kasetsart University, Sakon Nakhon province, Thailand. All experimental procedures were performed according to the Guidelines for Animal Experiments and approved by the Animal Ethics Research Committee of the Faculty of Veterinary Medicine, Kasetsart University. Prior to the study, each buffalo was assessed to be healthy based on a review of health history, physical examination, complete blood count and serum chemistry analysis.
Experimental design: To obtain the pharmacokinetic data, the ten Thai swamp buffaloes were randomly divided into two groups (n=5 per group). Each group was administered ER intravenously (i.v.) or subcutaneously (s.c.) (Baytril, 100 mg/ml) at a dose of 5.0 or 7.5 mg/kg BW. The experiments were performed in a two-phase, two-period, cross-over design with an interval of 3 weeks between the periods and the phases. Ten ml of blood was collected from the jugular vein of each animal with heparinized syringes at 0, 15 and 30 min and at 1, 2, 4, 6, 8, 12, 24, 32 and 48 hr after administration. The plasma was separated by centrifugation (1,986 × g) for 15 min. The urine and fecal samples were collected from 0 to 48 hr by natural collection and rectal palpation. All of the plasma, urine and fecal samples were frozen at −20°C until analysis.
Sample extraction: The extraction method of ER and CP in plasma, urine and feces was performed as described previously [19, 26, 36], after revalidation. Briefly, plasma samples were extracted using liquid-liquid extraction. 300 µl of acetonitrile (ACN) was added to 200 µl plasma in a tube and then shaken for 15 sec. The mixture was centrifuged at 4,500 × g and 4°C for 10 min. A 150 µl sample of the supernatant was transferred to a new tube and mixed with 300 µl of water. After passing it through a Ministart® RC filter (pore size 0.45 µm, Sartorius AG, Göttingen, Germany), an aliquot of 200 µl of this mixture was injected for high performance liquid chromatography (HPLC).
One hundred µl of urine was diluted with 900 µl of distilled water in a microcentrifuge tube. The solution was vortex-mixed and filtered using a Ministart® RC filter (pore size 0.45 µm, Sartorius AG); then, the sample (50 µl) was injected onto HPLC.
Fecal samples (100 mg) were homogenized with 400 µl of distilled water in a motor driven glass homogenizer for 2–3 min. To each sample, 2 ml of extraction solution was added, and the samples were mixed. Samples were stored at room temperature, in the dark, for 45 min before 1.5 ml of 0.07 M ortho-phosphoric acid were added. Samples were then vortexed and allowed to stand for a further 10 min before being centrifuged at 5,800 × g at 4°C for 10 min. The solution was filtered with a Ministart® RC filter (pore size 0.45 µm, Sartorius AG); then, the sample (50 µl) was injected onto HPLC.
HPLC conditions: The analytical method was performed in accordance with Otero et al. [27]. The HPLC analysis was performed using an Agilent 1260 series system consisting of a binary pump, an automatic sample injector, a column thermostat and a fluorescence detector (Agilent Technologies, Waldbronn, Germany) with excitation and emission wavelength of 294 and 500 nm, respectively. Separation was achieved using a PLRP-S column (5 µm, 4.6 × 150 mm) with an RP18-E guard column (5 µm, 4 × 40 mm) (Polymer Laboratories Inc., Church Stretton, U.K.). The column was maintained at a temperature of 35°C using a column oven. The mobile phase consisted of 14% ACN and 86% 0.07 M ortho-phosphoric acid. The flow rate was 1.2 ml/min. The retention times of ER and CP were 7.93 and 5.45 min, respectively. The limit of quantification (LOQ) of ER and CP in plasma was 0.1 and 0.05 µg/ml, respectively.
Fortification and method validation: The calibration curves were obtained by spiking the working standard solution into buffalo blank plasma, urine and feces to yield final concentrations of 0.1, 0.5, 1, 5 and 10 µg/ml of ER and CP. Buffalo blank plasma, urine and feces (for quality control samples) were obtained some days before the treatment protocol commenced. Linearity of the regression curve in the range 0.1–10 µg/ml was assessed on the basis of the residual plot, the lack of fit test and the back calculation (within 20% of known amount). Five duplicates of the quality control samples at concentrations of 0.5, 5 and 10 µg/ml were prepared and used to determine the recoveries, intra- and inter-day precision, and accuracy of the method. The procedure was repeated five times within the same day to gain intra-day run precision and accuracy and five times for each concentration over five consecutive days to obtain inter-day run precision and accuracy. The extraction recoveries of ER from plasma were 85.19 ± 2.52, 87.12 ± 1.95 and 86.24 ± 3.11% for 0.5, 5 and 10 µg/ml, respectively. The extraction recoveries were 84.22 ± 4.52, 83.26 ± 2.9 and 83.24 ± 3.33% for urine, and 82.63 ± 3.32, 81.64 ± 3.68 and 82.45 ± 4.31% for feces, respectively. The intra- and inter-day precision values in plasma, urine and feces were lower than 7% and accuracy was more than 95%, respectively. The variation coefficient (r2) value of the ER and CP calibration curves was >0.9997.
Pharmacokinetic calculation and statistical analysis: The concentrations of ER and CP in Thai swamp buffaloes versus time were pharmacokinetically analyzed by applying a two-compartment and non-compartmental model, respectively, using the PK Solution 2.0TM Program (Summit Research Services, Montrose, CO, U.S.A.). The absolute subcutaneous bioavailability (F) was calculated using the following equation:
| (%) F(S.C.)=(AUCS.C.) / (AUCI.V.) × 100 |
The statistical analyses were evaluated using the student-ttest. The results were presented as mean ± standard deviation (SD). All the analyses were conducted using GraphPad InStat (GraphPad Software, Inc., La Jolla, CA, U.S.A.). In all the experiments, differences were considered significant, if the associated probability level (P) was lower than 0.05.
RESULTS
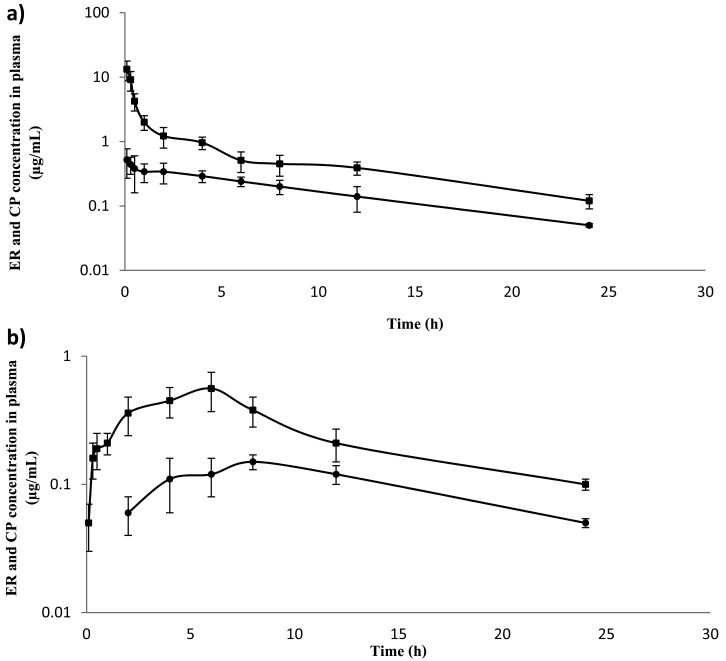
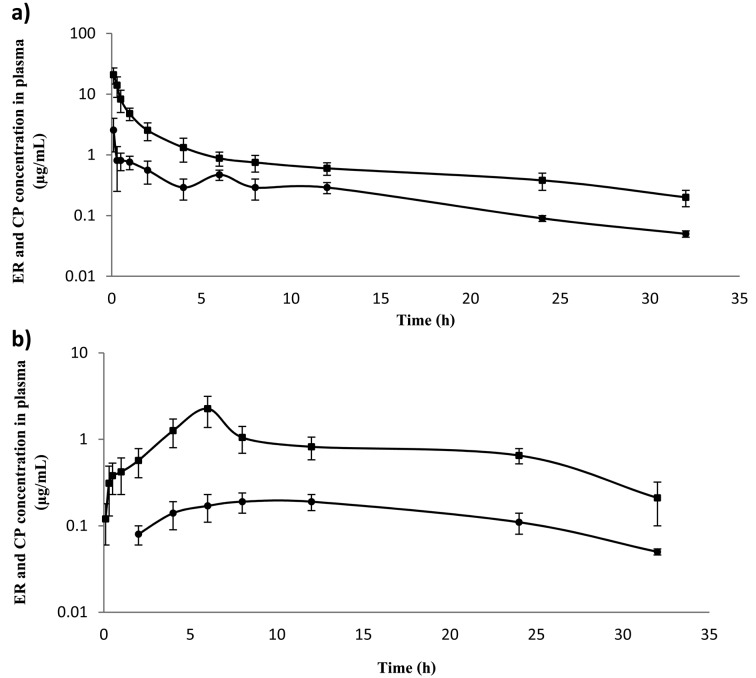
The determination of the ER and CP concentrations showed that ER and CP were detectable in the plasma of buffaloes following single i.v. and s.c. administrations. The semi-logarithmic plots of the mean plasma concentration-time curves of ER and CP at doses of 5.0 and 7.5 mg/kg BW are shown in Figs. 1 and 2. ER was measurable from 5 min, while CP was detectable from 30 min after s.c. administration. However, the drug ER and its metabolite CP were below the limit of detection of the method at 32 hr after i.v. and s.c. administrations, respectively, at doses of 5.0 and 7.5 mg/kg BW. Data for ER best fit a two-compartment model. The main pharmacokinetic parameters are reported in Table 1. Following i.v. administration, the AUC values increased with the dose, but not in a dose proportional fashion. In the s.c. treatment groups, Cmax and AUC values also increased, but not in a dose proportional fashion. Interestingly, Kel and T1/2β showed more rapid elimination of ER in the s.c. injection than in the i.v. injection. The absolute bioavailabilities of ER were 52.36 ± 4.24% and 72.26 ± 5.39% after s.c. administration at doses of 5.0 and 7.5 mg/kg BW, respectively (Table 1).
Fig. 1.
Mean values (± SD) of enrofloxacin (ER) (■) and ciprofloxacin (CP) (●) concentrations in plasma of Thai swamp buffaloes after intravenous (a) and subcutaneous (b) administrations at a dose of 5.0 mg⁄kg BW (n=5).
Fig. 2.
Mean values (± SD) of enrofloxacin (ER) (■) and ciprofloxacin (CP) (●) concentrations in plasma of Thai swamp buffaloes after intravenous (a) and subcutaneous (b) administrations at a dose of 7.5 mg⁄kg BW (n=5).
Table 1. Mean ± SD values of the pharmacokinetic parameters of enrofloxacin following intravenous and subcutaneous administrations at doses of 5.0 and 7.5 mg/kg BW in Thai swamp buffaloes (n=5 per group).
| Pharmacokinetic parameter (unit) | Intravenous | Subcutaneous | ||
|---|---|---|---|---|
| 5.0 mg/kg | 7.5 mg/kg | 5.0 mg/kg | 7.5 mg/kg | |
| Cp0 (µg/ml) | 16.32 ± 3.16 | 23.56 ± 5.23 | - | - |
| Kel (hr–1) | 0.1 ± 0.008 | 0.08 ± 0.005 | 0.20 ± 0.02* | 0.14 ± 0.01* |
| T1/2α(hr) | 3.35 ± 0.66 | 6.12 ± 0.86 | 2.21 ± 0.19* | 4.31 ± 0.61* |
| t1/2β (hr) | 7.05 ± 1.32 | 8.64 ± 1.15 | 3.43 ± 1.12* | 4.91 ± 0.91* |
| Cl§ (l/hr/kg) | 0.28 ± 0.04 | 0.21 ± 0.13 | 0.63 ± 0.11* | 0.28 ± 0.04* |
| Vd§(l/kg) | 2.82 ± 0.48 | 2.67 ± 0.33 | 3.12 ± 1.16 | 1.97 ± 0.32* |
| MRT (hr) | 15.0 ± 2.63 | 21.3 ± 3.12 | 8.70 ± 1.19* | 13.3 ± 2.65* |
| AUC0–24 (µg*hr/ml) | 16.93 ± 3.66 | 33.27 ± 4.63 | 8.12 ± 1.16* | 24.37 ± 3.28* |
| AUC0-∞ (µg*hr/ml) | 18.1 ± 3.29 | 35.0 ± 5.33 | 9.9 ± 2.23* | 27.0 ± 4.23* |
| Cmax (µg/ml) | - | - | 0.66 ± 0.15 | 2.26 ± 0.61 |
| Tmax (hr) | - | - | 6.0 ± 0.00 | 6.0 ± 0.00 |
| F (%) | - | - | 52.36 ± 4.24 | 72.26 ± 5.39 |
Cp0: plasma concentration at initial time; Kel: elimination rate constant of the beta phase; T1/2α: distribution half-life; T1/2β: elimination half-life; Cl: clearance; AUC0-∞: Area under the curve from time 0 to infinity; AUC0-24: Area under the curve from time 0 to 24 hr; Vd: Volume of distribution; MRT: Mean residence time; Cmax: Maximum concentration; Tmax: Time at maximum concentration. §This datum is normalized for bioavailability when calculated for the s.c. administration. *P<0.05 (statistically significant value if compared to the corresponding i.v. group).
The plasma concentrations of CP were lower than the respective concentrations of parental drug (Fig. 1). The curves were analyzed according to a non-compartmental method (Table 2). The average CP/ER AUCs ratio was 24.0 ± 2.1% and 18.3 ± 3.1% for the i.v. treatments (5.0 and 7.5 mg/kg, respectively) and 28.3 ± 3.97% and 26.6 ± 4.41% for s.c. treatments (5.0 and 7.5 mg/kg, respectively).
Table 2. Mean ± SD values of the pharmacokinetic parameters of ciprofloxacin following intravenous and subcutaneous administrations of enrofloxacin at doses of 5.0 and 7.5 mg/kg BW in Thai swamp buffaloes (n=5 per group).
| Pharmacokinetic parameters (unit) | Intravenous | Subcutaneous | ||
|---|---|---|---|---|
| 5.0 mg/kg | 7.5 mg/kg | 5.0 mg/kg | 7.5 mg/kg | |
| λz (hr−1) | 0.11 ± 0.025 | 0.11 ± 0.020 | 0.075 ± 0.013* | 0.11 ± 0.01 |
| t1/2λz (hr) | 6.44 ± 1.88 | 6.66 ± 1.35 | 9.43 ± 1.69* | 6.72 ± 1.28 |
| MRT (hr) | 9.63 ± 2.23 | 8.88 ± 1.15 | 16.44 ± 1.88* | 9.24 ± 1.04 |
| AUC0–24 (µg*hr/ml) | 4.13 ± 0.40 | 5.97 ± 1.68 | 2.25 ± 0.28* | 6.59 ± 1.29 |
| AUC0-∞ (µg*hr/ml) | 4.50 ± 0.39 | 6.42 ± 1.76 | 2.85 ± 0.34* | 7.11 ± 1.22 |
| Cmax (µg/ml) | - | - | 0.15 ± 0.022 | 2.29 ± 0.57 |
| Tmax (hr) | 0.14 ± 0.089 | 0.10 ± 0.00 | 6.0 ± 2.0* | 0.11 ± 0.00 |
λz: elimination constant; t1/2λz: Elimination half life; Cl: clearance; AUC0-∞: Area under the curve from time 0 to infinity; AUC0-24: Area under the curve from time 0 to 24 hr; MRT: Mean residence time; Cmax: Maximum concentration; Tmax: Time at maximum concentration. §This datum is normalized for bioavailability when calculated for the s.c. administration. *P<0.05 (statistically significant value if compared to the corresponding i.v. group).
The concentrations of ER and CP following i.v. and s.c. administrations at doses of 5.0 and 7.5 mg/kg BW in urine and feces, respectively, are shown in Tables 3 and 4. Both ER and CP were detectable in urine and feces up to 24 hr. Generally, ER and CP had higher concentrations in urine than in feces. The concentration of CP was higher than that of ER in urine.
Table 3. Mean ± SD values of enrofloxacin (ER) and ciprofloxacin (CP) in urine following intravenous and subcutaneous administrations at doses of 5.0 and 7.5 mg/kg BW in Thai swamp buffaloes (n=5 per group).
| Time (hr) | 5.0 mg/kg | 7.5 mg/kg | ||||||
|---|---|---|---|---|---|---|---|---|
| Intravenous | Subcutaneous | Intravenous | Subcutaneous | |||||
| ER (µg/ml) | CP (µg/ml) | ER (µg/ml) | CP (µg/ml) | ER (µg/ml) | CP (µg/ml) | ER (µg/ml) | CP (µg/ml) | |
| 0–4 | 2.34 ± 1.18 | 16.92 ± 4.93 | 0.50 ± 0.31* | 4.92 ± 3.83* | 5.14 ± 2.94 | 21.89 ± 7.49 | 0.35 ± 0.18* | 5.36 ± 2.13* |
| 4–12 | 0.42 ± 0.31 | 18.09 ± 3.57 | 0.70 ± 0.21 | 15.99 ± 4.70 | 0.54 ± 0.31 | 19.19 ± 1.48 | 0.56 ± 0.29 | 15.61 ± 4.91* |
| 12–24 | 0.12 ± 0.02 | 5.33 ± 3.86 | 0.36 ± 0.23 | 3.90 ± 1.51 | 0.16 ± 0.02 | 3.04 ± 1.32 | 0.31 ± 0.15 | 7.28 ± 2.66* |
*P<0.05 (statistically significant value if compared to the corresponding i.v. group).
Table 4. Mean ± SD values of enrofloxacin (ER) and ciprofloxacin (CP) in feces following intravenous and subcutaneous administrations at doses of 5.0 and 7.5 mg/kg BW in Thai swamp buffaloes (n=5 per group).
| Time (hr) | 5.0 mg/kg | 7.5 mg/kg | ||||||
|---|---|---|---|---|---|---|---|---|
| Intravenous | Subcutaneous | Intravenous | Subcutaneous | |||||
| ER (µg/ml) | CP (µg/ml) | ER (µg/ml) | CP (µg/ml) | ER (µg/ml) | CP (µg/ml) | ER (µg/ml) | CP (µg/ml) | |
| 0–4 | NF | NF | 0.03 ± 0.003* | 0.05 ± 0.04* | 0.24 ± 0.05 | ND | NF | NF |
| 4–12 | 3.63 ± 1.12 | 3.46 ± 0.72 | 0.57 ± 0.33* | 0.20 ± 0.17* | 2.06 ± 0.13 | 0.51 ± 0.45 | 0.33 ± 0.06* | 0.14 ± 0.06 |
| 12–24 | 0.10 ± 0.33 | 0.32 ± 0.03 | 0.62 ± 0.14* | 0.28 ± 0.08 | 0.24 ± 0.10 | 0.20 ± 0.08 | 0.86 ± 0.53* | 0.53 ± 0.21* |
NF: no fecal collection; ND: not detected. *P<0.05 (statistically significant value if compared to the corresponding i.v. group).
DISCUSSION
Our study was designed to characterize the disposition of ER following i.v. and s.c. administrations at doses of 5.0 and 7.5 mg/kg BW in Thai swamp buffaloes. This animal is important for the rural economy of Thailand, but is often affected by bacterial infections that can reduce its food-production ability (http://www.dld.go.th/th/index.php/2010-02-16-08-16) [8]. Several pharmacokinetic differences have been previously reported for ER within various animal species. For this reason, it is important to have a clear pharmacokinetic profile of this drug in each target species to formulate a sound therapeutic treatment plan.
The elimination half-life range of ER after i.v. administration was 7.05–8.64. This is slightly lower than the value reported for donkeys (9.54 hr) [35], but higher than those reported in buffalo calves (2.92 hr) [20], sheep (3.25 hr) [10], goats (2.73 hr) [10], lactating dairy cows (3.69 hr) [16] and beef steers (5.15 hr) [16]. Compared to other ruminant species, the plasma clearance in Thai swamp buffaloes was lower than those reported in buffalo calves (1.94 l/hr/kg) [20], sheep (0.53 l/hr/kg) [10], goats (0.70 l/hr/kg) [10], lactating dairy cows (1.45 l/hr/kg) [16] and beef steers (0.7 l/hr/kg) [16]. Data from the present study indicated a slower rate of elimination of ER in Thai swamp buffaloes compared to the aforementioned ruminant species. Parameters related to elimination (such as Kel and T1/2β) showed rapid elimination of s.c. injection than i.v. injection. It might be due to the saturated elimination of ER at highest concentration of drug. In fish species, ER has been reported to inhibit cytochrome P450 [37]. On the contrary, Cl values (5.0 vs. 7.5 mg/kg i.v. treatments) appeared to be unchanged, despite a large variability has been found in the 7.5 mg/kg group. However, further studies (with a larger animal sample size) are warranted to support this hypothesis. On the other hand, the Vd of ER found in the present study was large after both i.v. and s.c. administrations. These data are in agreement with the results from previous investigations in other ruminant species [14, 18, 20, 25, 32], which indicated that ER was able to penetrate well into various tissues.
Surprisingly, the AUC values increased irrespective of the dose (2 and 3 times after i.v. and s.c. administrations, respectively). This is in disagreement with other animal species where ER showed dose-dependent pharmacokinetic parameters [7]. The reason for this phenomenon is not clear, and a study with larger animal sample size should be used to verify this datum.
Following s.c. administration, ER given at a dose of 7.5 mg/kg was absorbed more efficiently than when given at a dose of 5.0 mg/kg as indicated by its higher bioavailability (72.26%). Some studies involving ER injected via extravascular injective routes, reported that local irritation and soft tissue necrosis can occur in the injection area [28]. Although this possibility was not checked at the time of experiment, the higher dose of the drug might have evoked a stronger inflammation response, increasing the portion of drug absorbed by dilated blood vessels. It might be a concern in clinical practice, however, some recent studies reported in turtles have shown that an intramuscular injection of a diluted concentration of ER (10 mg/ml) [10, 34] did not produce irritative effects as the more concentrate drug marketed solution (25 mg/ml) for pets [6, 31]. However, the nonlinear AUC values increase on increasing dose might suggest elimination saturation and consequently a non-linear pharmacokinetic. Under this condition, the F% data should have a negligible value and should be used with caution. Further studies are necessary to clarify this issue.
CP is the main active metabolite of ER, possessing a similar spectrum of antibacterial activity. Its concentration has been reported as always being lower than that of parental molecule in mammals, birds and reptiles. CP is known to account for about 25–35% of the amount of the parental drug in mammals [33]. These earlier data are in line with the results of the CP/ER AUC ratio reported in the present study (range 18.3–28.3%).
ER and CP were detectable in urine and feces up to 24 hr after i.v. and s.c. administrations, in the present study. However, the concentration of CP in urine was significantly higher than that detected in feces. This indicated that the urine was a major route of excretion of ER metabolite. This is in line with a previous study on the ER excretion in pigs [40].
It has been reported that ER can kill bacteria in a concentration-dependent manner [1, 9]. The minimum inhibitory concentration (MIC) values of ER for most veterinary pathogens range from 0.001 to 0.1 µg/ml [29]; however, bovine pathogens are usually susceptible to ER at MIC ≤ 0.06 µg/ml (Baytril-100® package insert). In addition, ER was proven to be one of the most active antimicrobial agents against Pasteurella multicida with an MIC of 90% of the isolates (MIC90) of 0.05 µg/ml [39]. This organism causes a disease called hemorrhagic septicemia (HS) or bovine respiratory disease (BRD), which is considered to be a livestock disease of major economic importance in Southeast Asia [30]. The maximum serum concentration/minimum inhibitory concentration (Cmax/MIC) ratio and the area under the serum concentration-time curve (AUC0–24/MIC) ratio are considered as the major pharmacokinetic-pharmacodynamic (PK-PD) indices for antimicrobial activity and optimal dose determination. The criteria of Cmax/MIC ratios of 8–10 or AUC/MIC ratios >100–125 have been suggested for maximum therapeutic efficacy and clinical effectiveness [15, 22, 23, 38]. Using an MIC breakpoint of 0.06 µg/ml, the values of the Cmax/MIC ratio and the AUC0–24/MIC ratio obtained from s.c. administration were 11.0 and 135.3 hr, respectively, whereas they were 37.67 and 406.2 hr at a dose of 5.0 and 7.5 mg/kg BW, respectively. All of these ratio values were above the suggested values. Hence, s.c. administration of ER at a dose of 5.0 and 7.5 mg/kg BW might provide effective plasma concentration for the treatment of susceptible bacterial diseases (MIC ≤ 0.06 µg/ml) in Thai swamp buffaloes. However, the Cmax/MIC (6.6) and AUC0–24/MIC (81.2 hr) values were below the recommended ratios for an MIC value of 0.1 µg/ml after s.c. administration of ER at 5.0 mg/kg BW. It is noteworthy that in this theoretical calculation, the efficacy due to CP is not included. CP is reported to have similar efficacy to its parental drug, and an increase of around 20% (equivalent to the fraction of metabolite formed) should be expected in the in vivo activity of the drug. Regarding our study, the drug plasma level was maintained at a higher level than the MIC (0.06 µg/ml) for 12 and 24 hr after ER s.c. administration at doses of 5.0 and 7.5 mg/kg BW. The side effects of ER, such as vomiting, anorexia, diarrhea and nervous signs, were not observed after s.c. drug administration with either dose.
In conclusion, the plasma concentrations of ER and its metabolite CP were measured in Thai swamp buffaloes, B. bubalis, after s.c. administration at doses of 5.0 and 7.5 mg/kg BW. The metabolism of ER to CP was identified also in this species, as occurs in other ruminants. ER and CP were excreted via the urine. Based on the pharmacokinetic data derived from our study and PK-PD indices, s.c. administration of ER at a dose of 5.0 and 7.5 mg/kg BW would be appropriate for the treatment of susceptible bacterial diseases in Thai swamp buffaloes. However, the dose rate of 7.5 mg/kg is recommended to treat infectious diseases caused by other veterinary pathogens having MIC ≤ 0.1 µg/ml.
Acknowledgments
This work was financially supported by the Faculty of Veterinary Medicine, Kasetsart University, Bangkok, Thailand. Authors acknowledge the English editing of the manuscript to Dr. H. Owen (University of Queensland, Australia).
REFERENCES
- 1.Aliabadi F. S., Lees P.1997. Pharmacodynamic and pharmacokinetic inter-relationship of antibacterial drugs. J. Vet. Pharmacol. Ther. 20: 14–17. [Google Scholar]
- 2.Anadón A., Matinez Larranaga M. R., Diaz M. J., Bringas P., Martinez M. A., Fernandez Cruz M. L., Fernandez M. C., Fernandez R.1995. Pharmacokinetics and residues of enrofloxacin in chickens. Am. J. Vet. Res. 56: 501–506. [PubMed] [Google Scholar]
- 3.Anadón A., Matinez Larranaga M. R., Diaz M. J., Fernandez Cruz M. L., Martinez M. A., Frejo M. T., Martinez M., Iturbe J., Tafur M.1999. Pharmacokinetic variables and tissue residues of enrofloxacin and ciprofloxacin in healthy pigs. Am. J. Vet. Res. 60: 1377–1382. [PubMed] [Google Scholar]
- 4.Araneda C., Villar P., Cuadros C., Valle M., Nunes P., Santelices M.2013. Single and multiple pharmacokinetics of enrofloxacin and ciprofloxacin in pigs. J. Bioequiv. Availab. 5: 41–46. doi: 10.4172/jbb.1000132 [DOI] [Google Scholar]
- 5.Aramayona J. J., Mora L., Fraile J., Garcia M. A., Abadia A. R., Bregante M. A.1996. Penetration of enrofloxacin and ciprofloxacin into breast milk, and pharmacokinetics of the drugs in lactating rabbits and neonatal offspring. Am. J. Vet. Res. 57: 547–553. [PubMed] [Google Scholar]
- 6.Casares M., Enders F.1996. Enrofloxacin side-effects in a Galapagos tortoise (Geochelone elephantopus nigra). Proc. Am. Assoc. Zoo. Vet. 96: 446–447. [Google Scholar]
- 7.De Lucas J. J., Solano J., Gonzalez F., Ballesteros C., San Andres M. I., Martin Von Kauffmann C., Rodriguez C.2013. Pharmacokinetics of enrofloxacin after multiple subcutaneous and intramuscular administrations in adult ostriches. Br. Poult. Sci. 54: 391–397. [DOI] [PubMed] [Google Scholar]
- 8.Department of Livestock Development, Ministry of Agriculture and Cooperatives, Thailand. Infectious diseases in cattle, buffalo, pig, goat and sheep in raining season of Thailand. http://www.dld.go.th/th/index.php/2010-02-16–08-16; 6 July, 2010.
- 9.Drusano G. L., Johnson D. E., Rosen M., Standiford H. C.1993. Pharmacodynamics of fluoroquinolone antimicrobial agent in a neutropenic rat model of Pseudomonas sepsis. Antimicrob. Agents Chemother. 37: 483–490. doi: 10.1128/AAC.37.3.483 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Elsheikh H. A., Taha A. A. W., Khalafallah A. I., Osman I. A. M.2002. Disposition kinetics of enrofloxacin (Baytril 5%) in sheep and goats following intravenous and intramuscular injection using a microbiological assay. Res. Vet. Sci. 73: 125–129. doi: 10.1016/S0034-5288(02)00020-6 [DOI] [PubMed] [Google Scholar]
- 11.Giguère S., Sweeney R. W., Belanger M.1996. Pharmacokinetics of enrofloxacin in adult horses and concentration of the drug in serum, body fluids and endometrial tissues after repeated intragastrically administered doses. Am. J. Vet. Res. 57: 1025–1030. [PubMed] [Google Scholar]
- 12.Giorgi M., Rota S., Giorgi T., Capasso M., Briganti A.2013. Blood concentrations of enrofloxacin and the metabolite ciprofloxacin in yellow-bellied slider turtles (trachemys scripta scripta) after a single intracoelomic injection of enrofloxacin. J. Exot. Pet Med. 22: 192–199. doi: 10.1053/j.jepm.2013.05.009 [DOI] [Google Scholar]
- 13.Haines G. R., Brown M. P., Gronwell R. R., Merritt K. A.2000. Serum concentrations and pharmacokinetics of enrofloxacin after intravenous and intragastric administration to mares. Can. J. Vet. Res. 64: 171–177. [PMC free article] [PubMed] [Google Scholar]
- 14.Haritova A., Lashev L., Pashov D.2003. Pharmacokinetics of enrofloxacin in lactating sheep. Res. Vet. Sci. 74: 241–245. doi: 10.1016/S0034-5288(03)00003-1 [DOI] [PubMed] [Google Scholar]
- 15.Hyatt J. M., McKinnon P. S., Zimmer G. S., Schentag J. J.1995. The importance of pharmacokinetic/pharmacodynamics surrogate markers to outcome: focus on antibacterial agents. Clin. Pharmacokinet. 28: 143–160. doi: 10.2165/00003088-199528020-00005 [DOI] [PubMed] [Google Scholar]
- 16.Idowu O. R., Peggins J. O., Cullison R., Bredow J.2010. Comparative pharmacokinetics of enrofloxacin and ciprofloxacin in lactating dairy cows and beef steers following intravenous administration of enrofloxacin. Res. Vet. Sci. 89: 230–235. doi: 10.1016/j.rvsc.2009.12.019 [DOI] [PubMed] [Google Scholar]
- 17.Kaartinen L., Salonen M., Älli L., Pyörälä S.1995. Pharmacokinetics of enrofloxacin after single intravenous, intramuscular and subcutaneous injections in lactating cows. J. Vet. Pharmacol. Ther. 18: 357–362. doi: 10.1111/j.1365-2885.1995.tb00604.x [DOI] [PubMed] [Google Scholar]
- 18.Kaartinen L., Pyörälä S., Moilanen M., Raisanen S.1997. Pharmacokinetics of newborn and one-week-old calves. J. Vet. Pharmacol. Ther. 20: 479–482. doi: 10.1046/j.1365-2885.1997.00100.x [DOI] [PubMed] [Google Scholar]
- 19.Kamberi M., Tsutsumi K., Kotegawa T., Nakamura K., Nakano S.1998. Determination of ciprofloxacin in plasma and urine by HPLC with ultraviolet detection. Clin. Chem. 44: 1251–1255. [PubMed] [Google Scholar]
- 20.Kumar N., Jayachandran C.2008. Pharmacokinetics of enrofloxacin and its metabolite ciprofloxacin after single dose intravenous administration of enrofloxacin in buffalo calves. J. Vet. Pharmacol. Toxicol. 7: 42–47. [DOI] [PubMed] [Google Scholar]
- 21.Küng K., Riond J. L., Wolffram S., Wanner M.1993. Comparison of an HPLC and bioassay method to determine antimicrobial concentrations after intravenous and oral administration of enrofloxacin in four dogs. Res. Vet. Sci. 54: 247–248. doi: 10.1016/0034-5288(93)90065-N [DOI] [PubMed] [Google Scholar]
- 22.Lode H., Borner K., Koeppe P.1998. Pharmacodynamics of fluoroquinolones. Clin. Infect. Dis. 27: 33–39. doi: 10.1086/514623 [DOI] [PubMed] [Google Scholar]
- 23.McKellar Q. A., Sanchez Bruni S. F., Jones D. G.2004. Pharmacokinetic/pharmacodynamic relationships of antimicrobial drugs used in veterinary medicine. J. Vet. Pharmacol. Ther. 27: 503–514. doi: 10.1111/j.1365-2885.2004.00603.x [DOI] [PubMed] [Google Scholar]
- 24.Messenger K. M., Papich M. G., Blikslager A. T.2012. Distribution of enrofloxacin and its active metabolite, using an in vivo ultrafiltration sampling technique after the injection of enrofloxacin to pigs. J. Vet. Pharmacol. Ther. 35: 452–459. doi: 10.1111/j.1365-2885.2011.01338.x [DOI] [PubMed] [Google Scholar]
- 25.Mengozzi G., Intorre L., Bertini S., Soldani G.1996. Pharmacokinetics of enrofloxacin and its metabolite ciprofloxacin after intravenous and intramuscular administration in sheep. Am. J. Vet. Res. 57: 1040–1043. [PubMed] [Google Scholar]
- 26.Nielsen P., Gyrd-Harren N.1997. Bioavailability of enrofloxacin after oral administration to fed and fasted pigs. Pharmacol. Toxicol. 80: 246–250. doi: 10.1111/j.1600-0773.1997.tb01967.x [DOI] [PubMed] [Google Scholar]
- 27.Otero J. L., Mestorino N., Errecalde J. O.2009. Pharmacokinetics of enrofloxacin after single intravenous administration in sheep. Rev. Sci. Tech. OIE 28: 1129–1142. [DOI] [PubMed] [Google Scholar]
- 28.Plumb D. C.2011. Enrofloxacin. pp. 504–509. In: Veterinary Drug Handbook, 7th ed., Wiley Blackwell, Ames. [Google Scholar]
- 29.Prescott J. F., Yielding K. M.1990. In vitro susceptibility of selected veterinary bacterial pathogens to ciprofloxacin, enrofloxacin and norfloxacin. Can. J. Vet. Res. 54: 195–197. [PMC free article] [PubMed] [Google Scholar]
- 30.Ramdani, Dawkins H. J. S., Johnson R. B., Spencer T. L., Adler B.1990. Pasteurella multocida infections in mice with reference to haemorrhagic septicaemia in cattle and buffalo. Immunol. Cell Biol. 68: 57–61. doi: 10.1038/icb.1990.8 [DOI] [PubMed] [Google Scholar]
- 31.Raphael B. L., Papich M., Cook R. A.1994. Pharmcokinetics of enrofloxacin after a single intramuscular injection in Indian star tortoises (Geochelone elegans). J. Zoo Wildl. Med. 25: 88–94. [Google Scholar]
- 32.Rao G. S., Ramesh S., Ahmad A. H., Tripathi H. C., Sharma L. D., Malik J. K.2002. Pharmacokinetics of enrofloxacin and its metabolite ciprofloxacin in goats given enrofloxacin alone and in combination with probenecid. Vet. J. 163: 85–93. doi: 10.1053/tvjl.2001.0594 [DOI] [PubMed] [Google Scholar]
- 33.Rao G. S., Ramesh S., Ahmad A. H., Tripathi H. C., Sharma L. D., Malik J. K.2001. Pharmacokinetics of Enrofloxacin and its metabolite Ciprofloxacin after intramuscular administration of Enrofloxacin in goats. Vet. Res. Commun. 25: 197–204. doi: 10.1023/A:1006481625615 [DOI] [PubMed] [Google Scholar]
- 34.Salvadori M., De Vito V., Owen H., Giorgi M.2015. Pharmacokinetics of enrofloxacin and its metabolite ciprofloxacin after intracoelomic administration in Tortoises (Testudo hermanni). Isr. J. Vet. Med. 70: 45–48. [Google Scholar]
- 35.Sekkin S., Gokbulut C., Kum C., Karademir U.2012. Plasma disposition of enrofloxacin following intravenous and intramuscular administration in donkeys. Vet. Rec. 171: 447–451. doi: 10.1136/vr.100653 [DOI] [PubMed] [Google Scholar]
- 36.Sunderland J., Lovering A. M., Tobin C. M., MacGowan A. P., Roe J. M., Delsol A. A.2004. A reverse-phase HPLC assay for the simultaneous determination of enrofloxacin and ciprofloxacin in pig faeces. Int. J. Antimicrob. Agents 23: 390–393. doi: 10.1016/j.ijantimicag.2003.07.014 [DOI] [PubMed] [Google Scholar]
- 37.Vaccaro E., Giorgi M., Longo V., Mengozzi G., Gervasi P. G.2003. Inhibition of cytochrome p450 enzymes by enrofloxacin in the sea bass (Dicentrarchus labrax). Aquat. Toxicol. 62: 27–33. doi: 10.1016/S0166-445X(02)00064-4 [DOI] [PubMed] [Google Scholar]
- 38.Wright D. H., Brown G. H., Peterson M. L., Rotschafer J. C.2000. Application of fluoroquinolone pharmacodynamics. J. Antimicrob. Chemother. 46: 669–683. doi: 10.1093/jac/46.5.669 [DOI] [PubMed] [Google Scholar]
- 39.Yoshimura H., Ishimaru M., Endoh Y. S., Kojima A.2001. Antimicrobial susceptibility of Pasteurellamultocida isolated from cattle and pigs. J. Vet. Med. B Infect. Dis. Vet. Public Health 48: 555–560. doi: 10.1046/j.1439-0450.2001.00468.x [DOI] [PubMed] [Google Scholar]
- 40.Zhou X., Chen C., Yue L., Sun Y., Ding H., Liu Y.2008. Excretion of enrofloxacin in pigs and its effect on ecological environment. Environ. Toxicol. Pharmacol. 26: 272–277. doi: 10.1016/j.etap.2008.04.004 [DOI] [PubMed] [Google Scholar]