Abstract
Placental malaria caused by Plasmodium falciparum contributes to ~200,000 child deaths annually, mainly due to low birth weight (LBW). Parasitized erythrocyte sequestration and consequent inflammation in the placenta are common attributes of placental malaria. The precise molecular details of placental changes leading to LBW are still poorly understood. We hypothesized that placental malaria may disturb maternofetal exchange of vitamins, lipids, and hormones mediated by the multi-ligand (n ~ 50) scavenging/signaling receptor megalin, which is abundantly expressed in placenta but was not previously analyzed in pregnancy outcomes. We studied abundance of megalin and its intracellular adaptor protein Dab2 by immunofluorescence microscopy in placental biopsies from Ugandan women with (n = 8) and without (n = 20) active placental malaria. We found that: (a) abundances of both megalin (p = 0.01) and Dab2 (p = 0.006) were significantly reduced in brush border of syncytiotrophoblast of infected placentas; (b) amounts of megalin and Dab2 were strongly correlated (Spearman’s r = 0.53, p = 0.003); (c) abundances of megalin and Dab2 (p = 0.046) were reduced in infected placentas from women with LBW deliveries. This study provides first evidence that placental malaria infection is associated with reduced abundance of megalin transport/signaling system and indicate that these changes may contribute to the pathology of LBW.
During P. falciparum infection, pregnant women can suffer from placental malaria (PM), where parasitized erythrocytes (PE) sequester in the placenta1, which can lead to inflammatory response. PM contributes to about 200,000 neonatal and 10,000 maternal deaths annually in malaria endemic regions2. Infant mortality in PM is largely due to low birth weight (<2.5 kg), in addition to stillbirth, and abortion3. Sequestration of PE in the placenta occurs at the boundary surface of the syncytiotrophoblast4 – the interface between maternal blood and fetal vasculature where maternofetal exchange takes place5. Anatomically, fetal blood vessels branch in the placenta into villi that are covered by a single layer of multinucleated cell called the syncytiotrophoblast, which is created by fusion of underlying cytotrophoblast cells. The apical surface of the syncytiotrophoblast has microvilli (brush-border) expanding its surface to ~12.5 m2 for extensive molecular exchange5. Sequestration of PE on this surface can lead to macrophage infiltration into the intervillous space, local inflammation, and pathological changes in the syncytiotrophoblast4,6. These processes, in turn, may lead to reduced maternofetal exchange and reduced fetal growth during gestation, and finally to LBW and other poor outcomes including premature birth, pre-eclampsia, and small for gestational age babies7,8,9. Recent insights into mechanisms of the placental pathological processes during PM suggest involvement of various pathways, including angiogenesis10,11, insulin-like growth factor (IGF-1) axis12, and, potentially, mammalian target for rapamycin (mTOR) (all extensively reviewed in refs 6,13). Nevertheless, the molecular details of these processes, as well as the involvement and role of other proteins and pathways, are still poorly understood.
In this respect the glycoprotein megalin (also called gp330, gp600, and LRP2) is a particularly interesting candidate. It is a large (~600 kDa) multi-ligand single-spanning trans-membrane endocytic receptor with substantial physiological functions. It belongs to an ancient14 low density lipoprotein receptor family. The importance of this receptor has been demonstrated in a large number of experiments across multiple organs15, though its role in the placenta is not yet well characterized. It has been shown that megalin expresses in placenta and is localized on the surface of the syncytiotrophoblast16,17. Levels of megalin expression in the placenta are third highest following its expression by thyroid and kidney proximal tubular cells18. Many functions of megalin have been studied in kidney and in early embryonic development15,19. In mice, megalin expresses at very early stages of embryonic development20. Knockout of the megalin gene in mice leads to perinatal death (only 2% newborn mice survive) and severe pathologies in various organs, most notably in brain morphology, as well as in the kidney and lung20,21. Also, megalin-deficient mouse fetuses at mid-gestation were significantly smaller than wild-type20. It was hypothesized that developmental deficiency in these megalin knockout animals might be explained, at least in part, by a vitamin and lipoprotein deficiency due to defective/insufficient transport of these molecules (see below) to the fetus through megalin endocytosis in the yolk sac and placenta19,20. In humans, placental cytotrophoblast and syncytiotrophoblast megalin starts to express at least as early as 7–8 weeks gestation22 and expresses through term16,17,22,23. Substantial expression of megalin in the syncytiotrophoblast brush border16,17 indicates its importance for placenta function. In humans, mutation preventing megalin expression are extremely rare and only about a dozen surviving patients with Donnai-Barrow syndrome have been previously described24, with pathologies similar to those observed in megalin-knockout mice.
In epithelial cells, megalin is transferred with its cargo to the base of microvilli and is internalized through coated-pit endocytosis. We have demonstrated earlier that megalin interacts with a phosphotyrosine-interacting domain of the adaptor protein Dab225,26, which binds to the internalization motif27 in the megalin cytoplasmic domain. Dab2 is expressed in various tissues including syncytiotrophoblast and in trophoblast cells28,29. Its mRNA expression in placenta is highest among all tissues18. Based on previous studies of others on Dab2 functions28,30,31, we hypothesized that megalin and Dab2 might be involved in both signal transduction as well as in endocytosis through their interactions25. This hypothesis was experimentally confirmed in multiple studies15,19,32,33. As Dab2 is important for cell growth suppression28,30, it may also have an important role in placental growth and development.
Megalin interacts with an enormous suite of about 50 extracellular ligands which belong to several unrelated groups of proteins15. These ligands include nutrients, hormones and their carrier proteins, vitamin-binding and signaling molecules, morphogens, and extracellular matrix proteins including serum proteases and their inhibitors. Internalized molecules are either transcytosed or released in endosomes or lysosomes (vitamins, cholesterol, etc.) and then can be utilized within the cell or excreted on the cell surface by various transporters. Clearly, such diverse functions might have significant impact on the maternofetal exchange of nutrients and signals, as well as affect the homeostasis of various important molecules. This, in turn, may lead to placental pathology resulting in poor outcome for fetal development, including LBW, if the megalin system is somehow disturbed.
A few interesting observations suggest that the megalin system might be affected in PM. When the megalin gene is knocked out, the number of microvilli and the amount of endocytosis are significantly reduced in kidney21. These changes are reminiscent of loss of syncytiotrophoblast microvilli reported during PM4 and strongly support the idea that megalin expression/distribution in PM might be disturbed. In addition, inflammatory processes, similar to that observed during Heymann nephritis, can lead to shedding of the megalin exodomain34.
To get insights into the megalin system during PM, we determined the abundance of megalin and its intracellular adaptor protein Dab2 in formalin-fixed paraffin-embedded placental samples obtained from women living in malaria endemic areas of Uganda, with and without PM, who had normal and low birth weights of newborn babies (Table 1 and Supplementary Table 1).
Table 1. Clinical characteristics of 28 Ugandan pregnant women at delivery.
| Mothers without placental infection (PE−) | Mothers with placental infection (PE + ) | P valuea | |
|---|---|---|---|
| Number of women | 20 | 8 | |
| Parity median [interquartile range]; n | 1 [1.25]; 16 | 1 [1]; 6 | 0.88 |
| Hemoglobin level [g/dL] mean (SD); n | 11.4 (2.3); 16 | 10.5 (2.1); 5 | 0.37 |
| Estimated gestational age [weeks] mean (SD); n | 39 (2); 15 | 38 (4); 5 | 0.88 |
| Birthweight [g] mean (SD); n | 2916 (513); 16 | 2942 (476); 5 | 0.89 |
| Low birth weight n (%) | 4/16 (25%) | 2/6 (33%) | |
| Stillbirth n (%)b | 0/16 (0%) | 1/6 (17%) |
aMann-Whitney test was used to calculate all p values.
bThis sample was included in categorical analysis as low birth weight.
Results
Patient characteristics
Samples were selected based on availability of samples from previous study35,36. Summary clinical information is presented in Table 1, and data from individual samples are presented in Supplementary Table S1. Groups of Ugandan mothers without (PE−) and with (PE + ) placental infection at delivery were similar in respect to parity, hemoglobin levels, estimated gestational age, and birthweight of newborns.
Expression of megalin and Dab2 in human syncytiotrophoblast in term placenta
Immunofluorescence studies confirmed substantial expression of megalin in the syncytiotrophoblast of human term placenta as previously reported17,22. However, this expression was not uniform throughout the syncytiotrophoblast; parts of the syncytiotrophoblast brush border may express very little of megalin (and Dab2), if any, as was noted previously for megalin in placenta22. Therefore, quantitative comparison of protein abundances in various samples is challenging. To overcome this difficulty the following steps were taken: (1) protein abundances were measured in the parts of syncytiotrophoblast brush border (BB) with the highest signal as reflecting the ability of the tissue to express these proteins and (2) repeated blind experiments were performed using different antibody preparations. Three non-overlapping areas encompassing the syncytiotrophoblast BB from each placental section were selected for the highest density of signal, measured, and averaged, as described in the Methods. Supplementary Figure S1 demonstrates that measurements obtained in independent blind experiments, using two independent preparations of anti-megalin C-terminal peptide (in cytoplasmic tail) antibodies and two preparations of anti-Dab2 antibodies raised against different regions, strongly correlate, suggesting that our measurements accurately reflect the levels of abundance of these proteins. As anti-megalin prep 2 (more recent preparation) antibody and anti-Dab2 commercial antibody had higher signal according to linear regression analysis (Supplementary Figure S1), they were used for all analyses described below.
Placental malaria is associated with reduced abundance of megalin and Dab2
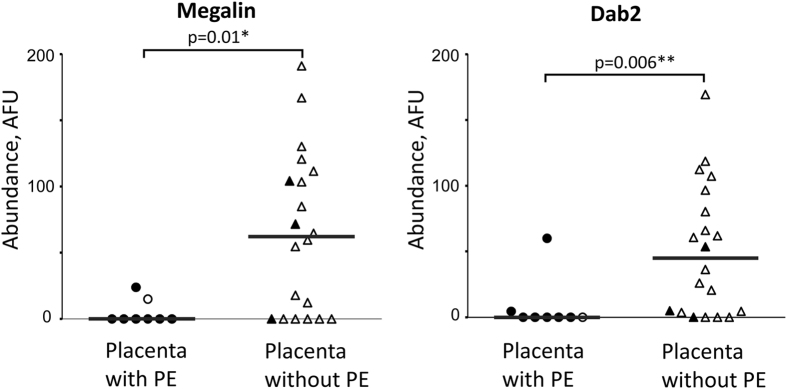
The abundance of both megalin and Dab2 was reduced in the syncytiotrophoblast brush border in malaria-infected placentas by indirect fluorescence microscopy, with statistical significance for both proteins (Fig. 1). Data on parity is available for 22 samples used in this analysis (Supplementary Table S1). Only including these 22 samples in the analysis shows that median parity is not different (p = 0.88) for groups stratified by placental infection, while abundance of both proteins is still reduced in the infected group (Supplementary Figure S2), demonstrating that parity is not a confounding factor. Samples with previous (resolved) malaria infection, as defined by the presence of extracellular hemozoin (Hz) in fibrin, are indicated in Fig. 1. If samples with active and past placental infection were combined, megalin and Dab2 continue to demonstrate a statistically significant reduction of abundance in these samples against normal controls (p = 0.04 and 0.005 for megalin and Dab2, respectively). Two data points for megalin and one for Dab2 with past (resolved) placental infections were above the median protein abundance value in the group of non-infected placenta samples (Fig. 1), which may point out the possibility for restoration of megalin and Dab2 expression in the placenta after placental infection is cleared. Similar results (as in Fig. 1) were obtained when protein abundance was measured in the entire syncytiotrophoblast when cytoplasmic staining was included (though fluorescence values were lower).
Figure 1. Megalin and Dab2 abundance is reduced in brush border area of syncytiotrophoblast in placentas with malarial infection at delivery.
Abundance of megalin and Dab2 was assessed in brush border of placentas with PE (n = 8) and without PE (n = 20) using immunofluorescence assay as described in Methods. Medians of arbitrary fluorescence units (AFU) are reported (gray bars). Filled symbols represent samples with extracellular hemozoin in fibrin indicative of previous (resolved) placental infections, clear symbols represent samples without hemozoin. Protein abundance between 2 groups was compared using Mann-Whitney test. PE – parasitized erythrocytes in the placenta at the time of delivery.
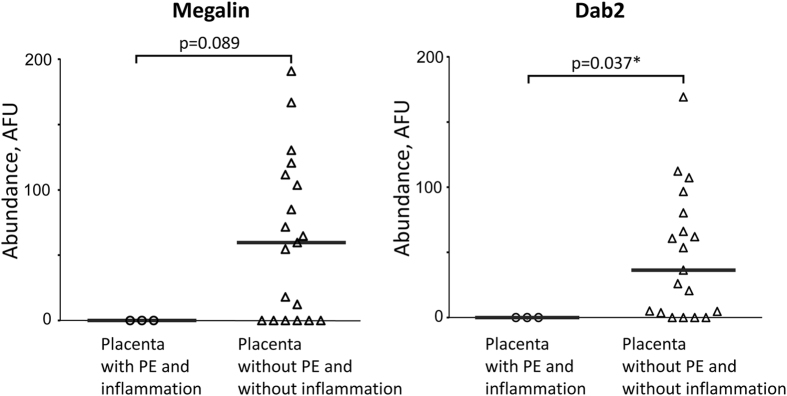
When data were stratified by the presence of placental inflammation in malaria-infected samples (n = 3) against samples without infection and inflammation, a similar decrease in megalin and Dab2 abundance was observed (Fig. 2), with statistical significance for Dab2.
Figure 2. Megalin and Dab2 abundance is reduced in brush border area of syncytiotrophoblast in placentas with malarial intervillous inflammatory infiltrates.
Megalin and Dab 2 abundance in placentas with PE and malaria-relevant inflammation (n = 3) and without PE and inflammation (n = 19) was measured using immunofluorescence assay. Medians of arbitrary fluorescence units (AFU) are reported (gray bars). Protein abundance between 2 groups was compared using Mann-Whitney test. PE – parasitized erythrocytes in the placenta at the time of delivery.
Also, irrespective of PM status of samples, statistically significant positive correlation of megalin and Dab2 expression in the brush border region was observed (Fig. 3). A similar correlation was observed when the entire syncytiotrophoblast was analyzed (data not shown).
Figure 3. Correlation of megalin and Dab2 abundance in brush border area of syncytiotrophoblast.
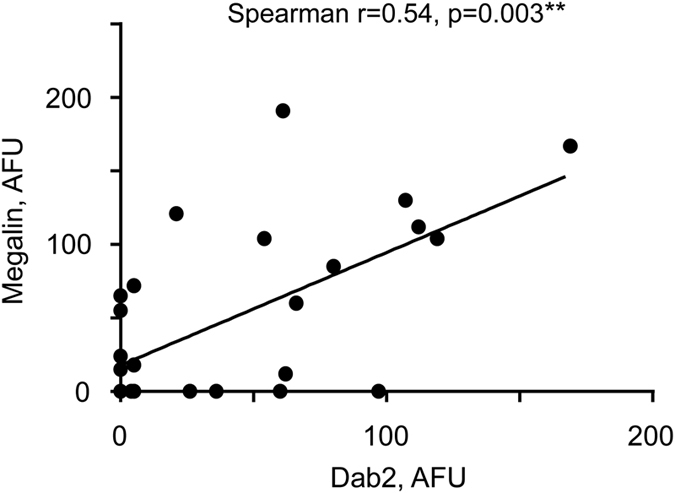
Correlation between amounts of Megalin and Dab2 was assessed using Spearman’s rank correlation coefficient (r). Megalin and Dab 2 abundance in all placentas (n = 28) was measured in arbitrary fluorescence units (AFU) using immunofluorescence assay as described in Methods. Six study participants (4 with PE and hemozoin, 1 with hemozoin only, and 1 without PE or hemozoin) did not have detectable megalin and Dab2. Line indicates linear regression, slope = 0.77 + 0.19, p = 0.0003. PE – parasitized erythrocytes in the placenta at the time of delivery. Hemozoin in the placenta at the time of delivery is indicative of past infection.
Expression of megalin and Dab2 is reduced in placentas with malaria parasites and low birth weight
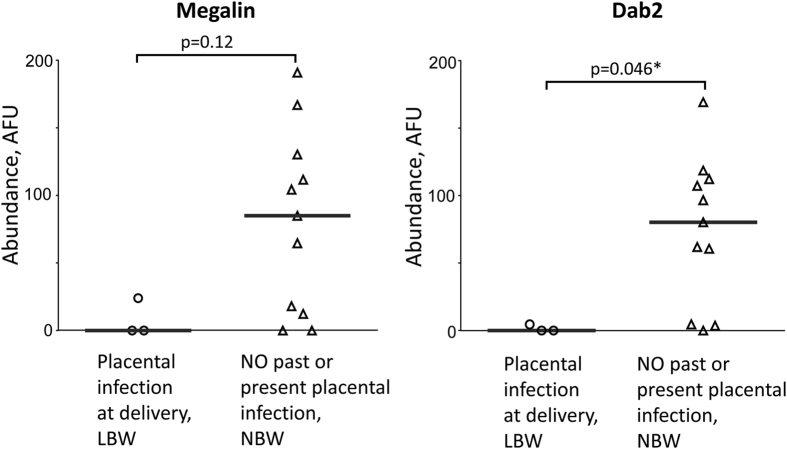
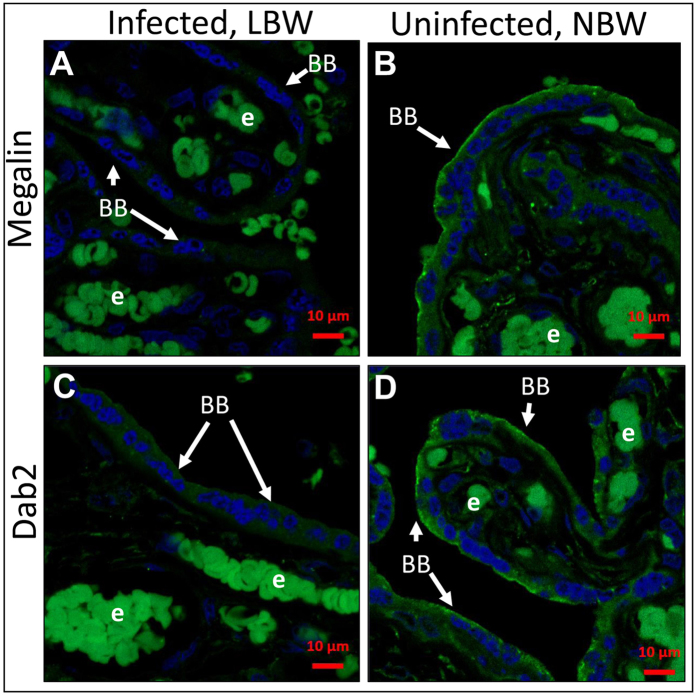
Moderate correlation of birth weight with abundance of megalin and Dab2 in the brush border of the syncytiotrophoblast was identified, though below the level of statistical significance (Spearman correlation coefficients, Megalin: r = 0.35 [CI = −0.09981 to 0.6782], p = 0.11; Dab2: r = 0.23 [CI = −0.2242 to 0.6030, p = 0.3). When Dab2 expression levels were stratified by median Dab2 abundance, birth weight was 350 g greater in those with high (above median) versus low (below median) placental expression, although the difference was not statistically significant (p = 0.095) (a woman with stillbirth and no detectable Dab2 expression was excluded). Abundance of both megalin and Dab2 is substantially reduced in the brush border of malaria-infected placentas obtained from LBW deliveries (Fig. 4), with statistical significance for Dab2 (p = 0.046). Figure 5 illustrates comparative differences observed between two samples from infected placenta and LBW versus uninfected placenta and normal birth weight (NBW). Specifically, the brush border of syncytiotrophoblast demonstrates the low or absent levels of megalin and Dab2 in the LBW samples with active malaria infection.
Figure 4. Megalin and Dab2 abundance is reduced in brush border area of syncytiotrophoblast in placentas with malarial infection and low birth weight.
Megalin and Dab 2 abundance in placentas with PE and low birth weight (LBW) (n = 3) and without PE or past infection and normal birth weight (NBW) (n = 11) was measured using immunofluorescence assay. Medians of arbitrary fluorescence units (AFU) are reported (gray bars). Protein abundance between 2 groups was compared using Mann-Whitney test. PE – parasitized erythrocytes in the placenta at the time of delivery.
Figure 5. Expression of megalin and Dab2 in placentas from mothers with low birth weight babies is reduced.
Megalin (A,B) and Dab2 (C,D) expression in syncytiotrophoblast of placentas with malaria infection and low birth weight (A,C) and without malaria infection and normal weight (B,D) was measured using immunofluorescence assay. Protein expression is shown in green, DAPI nuclear stain in blue. Pairs of pictures (A–D) were taken and processed under identical conditions. Bright cells inside of fetal vessels are auto-fluorescing erythrocytes (e). Brush border (BB) is indicated by arrows. LBW – low birth weight. NBW – normal birth weight.
Discussion
PM-related changes in the maternofetal interface of the placenta are associated with LBW6. Several mechanisms contributing to LBW have been suggested, including dysregulated angiogenesis9,10,37, impaired growth hormone production12,38, and decreased nutrient transport6. In this study a novel role for megalin system in PM pathologies was explored, including PM-associated LBW. Results demonstrate that PM is associated with reduced syncytiotrophoblast abundance of megalin and Dab2 proteins, known to provide endocytosis and signaling pathways, which, in turn, may play a role in fetal growth restriction and low birth weight pathology.
Specifically, the study provides evidence that megalin and Dab 2 abundance is reduced in placentas with active infections (Fig. 1). Similar reduction (with statistical significance for Dab2) was also observed in infected placentas with inflammation (Fig. 2). As PM is often characterized by infiltration of immune cells, especially monocytes and macrophages, into intervillous space39, these results suggest that inflammation may affect megalin system expression in syncytiotrophoblast. The small number of samples with malaria-related inflammation (n = 3) limits statistical power in our analysis.
Further, amounts of megalin and Dab2 are positively correlated in the placental brush border (Fig. 3). It has been shown earlier that Dab2, as an intracellular ligand of megalin25, co-localizes with megalin in renal proximal tubules40. In addition, expression levels of both proteins are mutually dependent, where knockout of one of them reduces expression of the other one40. The results presented here expand this observation to placental tissue and indicate inter-relevance of megalin and Dab2 expression. Moreover, this underscores the importance of significant associations identified in this work, even found for one of these proteins, as reduction of its abundance may affect the function of the entire system.
While all samples with placental infection and LBW had low or undetectable megalin and Dab2 (3/3), there were 4/11 and 3/11 normal controls with NBW that also had low or undetectable levels of megalin and Dab2, respectively (Fig. 4). In addition, the reduction of megalin system proteins in the placenta correlated with the reduction in birth weight irrespective of PM status, but these data were not statistically significant. Though small sample size definitely limited the power of our analysis, it allows to speculate that such correlation or association might be the case not only during PM, but, potentially, in other diseases of pregnancy. Low levels of these proteins in a proportion of malaria-negative placentas may indicate that other factors might affect their abundance. For example, renal inflammatory processes such as those that occur in Heymann nephritis can lead to shedding of the megalin exodomain34; both megalin and its co-receptor cubilin were downregulated in gallbladder epithelium in gallstone patients at the mRNA and protein levels41; mice fed a cholesterol rich lithogenic diet showed a reduction in megalin mRNA expression in these cells15. Megalin expression might be responsive to treatment, for example, rosiglitazone significantly increased megalin mRNA expression in the gallbladder15. Similarly, multiple factors may contribute to the expression of megalin system proteins in uninfected placentas. Also, in spite of megalin importance, even megalin knock-out newborn mice can survive, though in low numbers (2%), indicating that some redundant mechanism(s) may compensate for megalin system insufficiency during fetal development which allows for survival20,21.
Nevertheless, categorical analysis of protein abundance data stratified by LBW and placental infection revealed statistically significant reduction for Dab2 abundance (and very similar trend for megalin) compared to non-infected placental samples with NBW (Fig. 4). This result suggests that reduction in abundance of megalin transport/signaling system proteins in PM may contribute to the pathology of LBW. Below is a discussion of the potential mechanisms of pathological changes in pregnancy in relevance to the disturbance of the megalin system in placenta, which was observed in this work during PM.
Maternal cholesterol is essential for fetal growth42. It has been shown that fetal growth restriction is associated with alterations in placental lipoprotein receptors (LDL, LRP1) and maternal lipoprotein composition43. Megalin is involved in transport of the same lipoparticles, plus it interacts and transports additional types of Apo-lipoproteins and might substantially contribute to cholesterol transport, necessary for fetal growth.
Megalin also plays an important role in vitamin homeostasis by re-absorbing various vitamins in complexes with their carrier proteins in kidneys19,44. Because vitamins for the developing fetus can only be supplied from maternal blood, megalin may play a similar function in placenta supplying retinol22, cobalamins45, folate, and vitamin B1245, as well as vitamin D, which is important for regulation of calcium homeostasis46. Moreover, deficiency of vitamin D in pregnancy may increase the risk of preterm delivery and fetal growth restriction47.
Further, megalin co-expresses on the syncytiotrophoblast surface with several receptors specialized for interactions with hormones/growth factors (that are also megalin ligands) including insulin-like growth factor-1 (IGF-1), parathyroid hormone and epidermal growth factor5, which play important roles in the regulation of fetal growth throughout pregnancy48. Thyroid hormones (TH) are involved in placenta villous development and free TH (T4) levels are associated with birth weight49. In maternal blood, T4 is almost completely bound to transthyretin, a ligand for megalin50 that is secreted to maternal circulation and re-absorbed by trophoblasts51, and therefore may maintain T4 homeostasis. As mentioned above, PM was associated with disturbance of the IGF axis, with significant changes in IGF-1 but not IGF-2, and no detectable changes in mRNA levels for IGF-1 receptor12. Since IGF-1 is also a ligand for megalin and modulation of the megalin system may affect levels of IGF-152, megalin reduction may contribute to the effects of PM on IGF-1 level changes12.
How does PM affect the megalin system? One potential mechanism might be that sequestration of PE through surface-expressed PfEMP1 parasite adhesins may cross-link the corresponding host adhesion receptor, chondroitin sulfate A1, on the syncytiotrophoblast surface. This clustering of the surface receptors may directly affect multiple membrane processes including megalin-mediated endocytic and/or signaling pathways, as was the case in other studies on the effects of cross-linking of other cell surface receptors53. Infiltrating macrophages during PM may directly damage the syncytiotrophoblast surface, linking placental inflammation and megalin system abundance (Fig. 2) and/or function.
TGFβ negatively regulates endocytosis of albumin through megalin and cubilin54. During placental malaria TGF-beta concentration in placenta is significantly increased55. Hence, systemic regulation of placental megalin expression in malaria is possible and may lead to reduced endocytosis in infected placentas.
This study is limited by the number of samples investigated. A logical continuation would be a similar study with larger sample size and samples obtained from various geographical areas, taking into account a larger set of variables, including timing of malaria infection during pregnancy and nutritional status. Further, effects of PE adhesion to cytotrophoblast primary cells or cell lines, like BeWo cells, on megalin system functioning in the absence and presence of monocytes/macrophages may test our hypothesis in vitro. These studies may contribute to development of therapy alleviating pathological effects of megalin system dysfunction, for example by affecting transcription of relevant genes, and/or by supplementation of nutrients and ligands.
In summary, it is likely megalin is involved in fetal growth via a complex network of regulatory activities participating in endocrine, paracrine, and autocrine signaling by interacting with and internalizing various ligands of maternal and fetal origin. We propose that dysregulation of these networks in the placenta affects development of the fetus. To our knowledge, this is the first report linking the abundance of placental megalin system proteins with the birth weight of newborn babies and associating PM infection with changes in abundance of megalin system proteins.
Methods
Ethics Statement
Written informed consent was provided and specimens were collected under approval by the Uganda National Committee for Science and Technology (#HS207). The use of coded specimens was approved and deemed not human subjects research by the University of Washington Human Subjects Division (#35425) and by the Florida Atlantic University Institutional Review Board (#801504). All experiments were performed in accordance with the relevant guidelines and regulations.
Placental samples
Specimens were from a cohort of a nested randomized control trial of artemether-lumefantrine versus quinine to treat malaria in pregnancy conducted in 2006–2009 in Mbarara, Uganda, as previously described35. Briefly, the randomized controlled trial inclusion criteria included viable pregnancy at gestational age of 13 weeks or later and positive malaria blood smear by microscopy; exclusion criteria included P. falciparum parasitemia greater than 250 × 103/μL, severe anemia (hemoglobin < 7 g/dL), or severe or complicated malaria needing parenteral treatment. As previously described56, placental biopsies were fixed and stored in neutral buffered formalin, routinely processed and scored for the presence or absence of parasitized erythrocytes (PE + or PE−) and hemozoin in fibrin57, which were semi-quantitatively scored together with the presence of intervillous inflammatory infiltrates58. Placentas positive for parasitized erythrocytes (PE) by histology were referred as malaria-infected. To select samples for this study (n = 28) all available samples that had parasites identified by histology (n = 8) were matched to sequential controls that had no identifiable placental malaria-related changes on histopathology (n = 17), maintaining similarities in parity (Supplementary Figure S2). These control samples included available placental specimens from the larger cohort of women, who were screened negative for malaria by blood smear at weekly visits and not included in the randomized controlled trial36 (samples 23–28 in Supplementary Table S1). In addition, a subset of women with a relatively large amount of hemozoin in fibrin but no parasites (heavy past infections) were also available and were included (n = 3).
For the immunofluorescence assays, sections of 6 μm were prepared from paraffin embedded blocks of formalin-fixed placentas obtained from these women. Relevant clinical parameters of placenta samples are described in Supplementary Table S1.
Antibodies
Primary antibodies used were monospecific purified rabbit antibodies, raised against megalin C-terminal 17-mer peptide (2 independently purified preparations) and Dab2 phosphotyrosine interaction domain, which were described and characterized previously25, and commercial anti-Dab2 antibody H-110 (# sc-13982, Santa Cruz Biotechnology, CA). Secondary antibodies used were Alexa Fluor 594-labeled polyclonal Donkey anti-Rabbit IgG (#711-585-152, Jackson Immunoresearch, West Grove, PA).
Measurement of megalin and Dab2 abundance by indirect fluorescence microscopy
The paraffin embedded tissue samples were rehydrated by a series (3 min each) of graded xylene/ethanol washes (2 times with 100% xylene, once with 1:1 xylene/ethanol, once sequentially with 100%, 95%, 70% and 50% ethanol in deionized water), followed by a final wash in phosphate buffered saline (PBS) for 5 min. The rehydrated slides were then heated in Tris-EDTA buffer (10 mM Tris Base, 1 mM EDTA solution, 0.05% Tween-20, pH 9.0) and microwaved to boiling at full power and then maintained at 50% power for 8 minutes and cooled in PBS59. Sections were then pre-incubated with PBS, 1% BSA and 0.25% Triton-X for 30 minutes, incubated with primary antibody (Dab2 commercial 1:100; Dab2-PID 1:100; Megalin C-terminal 17-mer peptide 1:50) in the presence of 2.5% non-immune donkey serum (#017-000-121, Jackson Immunoresearch, West Grove, PA, USA), either for one hour at room temperature or overnight at 4 °C in a humidified chamber and washed with PBS for 5 minutes 3 times. Slides were then incubated with secondary antibody in the presence of non-immune donkey IgG (# 017-000-002, 1:100, Jackson Immunoresearch, West Grove, PA, USA) for one hour at room temperature and counterstained with DAPI (5 μg/mL, Sigma) for 3 minutes. After three washes with PBS for 5 minutes, slides were processed with mounting medium (Vectashield H-1000, Vector Laboratories, Burlingame, CA, USA) and coverslip applied. Negative controls were processed without primary antibody or with antibodies purified from the pre-immunized animal serum on the same column as monospecific antibodies. Both methods produced similar background levels (data not shown). For the staining, to provide maximally identical conditions, each placental section was divided in sub-sections processed with all primary antibodies, and then incubated with secondary antibody simultaneously. As a positive control for the procedures used, we stained fresh-frozen mouse kidney 6 μm sections as described above and obtained strong staining of the proximal tubule cells with anti-megalin and anti-Dab 2 antibodies, as described previously40,60 (data not shown).
Slides were imaged using a confocal microscope (Zeiss LSM 700 equipped with Plan-Apochromat 63x/1.40 Oil DIC M27 objective) and processed using Zen 2012 Blue software (Zeiss, Jena, Germany). Images were captured using identical conditions to allow for quantification of signal strength. Collected images of syncytiotrophoblast were selected for the brush border only (~1 μm from the surface to inside of the cell) or the entire syncytiotrophoblast (up to ~10 μm in depth and excluding nuclei). The process of selection is illustrated in Supplementary Figure S3: selected region A indicates brush border area, and region A+B indicates entire syncytiotrophoblast. In each placental section at least 3 non-overlapping areas encompassing syncytiotrophoblast 50 μm–100 μm along the surface were selected for the highest density of signal (in arbitrary fluorescence units, AFU) using visual appearance and density-measuring feature of the Zen 2012 Blue software, measured as described above, and averaged for each placental sample. If staining was similar throughout the section, 3 random areas were selected. Negative control AFU were then subtracted from the experimental AFU to obtain final AFU for each sample used in the analysis. All slides were processed by the same individual, blindly.
Statistical analyses
Statistical analyses were performed using GraphPad Prism 6 (La Jolla, California, US) statistical software using non-parametric statistics: Mann-Whitney tests (two-tail) for group differences and Spearman tests for analyses of correlations. P value of <0.05 was considered significant. Qualitative level of significance indicated by stars: *p < 0.05, **p < 0.01, ***p < 0.001.
Additional Information
How to cite this article: Lybbert, J. et al. Abundance of megalin and Dab2 is reduced in syncytiotrophoblast during placental malaria, which may contribute to low birth weight. Sci. Rep. 6, 24508; doi: 10.1038/srep24508 (2016).
Supplementary Material
Acknowledgments
This work was supported by the National Institutes of Health (grants 5R01HD058005 and 1R21AI105506 to A.V.O.), by the Florida Atlantic University Graduate Research & Inquiry Program (GRIP grant to J.L.), and by the Florida Atlantic University (Start-up fund to A.V.O.). The original study which produced the initial placental sample collection was supported by Médecins Sans Frontières and the European Commission. We are grateful to the entire research team at the Epicentre, Mbarara, Uganda and all the study participants and their families who made this study possible.
Footnotes
Author Contributions A.V.O. conceived and designed the experiments. J.L., J.G., O.C. and A.M. performed the experiments. J.L., J.G., and A.V.O. analyzed the data. E.T., M.D., P.J.G., P.P. and A.M. contributed reagents/materials. A.M. and A.V.O. wrote the manuscript.
References
- Fried M. & Duffy P. E. Adherence of Plasmodium falciparum to chondroitin sulfate A in the human placenta. Science 272, 1502–1504. (1996). [DOI] [PubMed] [Google Scholar]
- Andrew E. V. W. et al. Knowledge, Attitudes, and Practices Concerning Malaria in Pregnancy: Results from a Qualitative Study in Madang, Papua New Guinea. PLoS One 10, 0119077 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGregor I. A. Thoughts on malaria in pregnancy with consideration of some factors which influence remedial strategies. Parassitologia 29, 153–163 (1987). [PubMed] [Google Scholar]
- Walter P. R., Garin Y. & Blot P. Placental pathologic changes in malaria. A histologic and ultrastructural study. Am J Pathol 109, 330–342 (1982). [PMC free article] [PubMed] [Google Scholar]
- Castelucci M. & Kaufman P. In Pathology of the Human Placenta (eds Benirschke K., Kaufmann P., & Baergen R. N.) Ch. 6, 50–120 (Springer-Verlag, 2006). [Google Scholar]
- Kidima W. B. Syncytiotrophoblast Functions and Fetal Growth Restriction during Placental Malaria: Updates and Implication for Future Interventions. Biomed Res Int 2015, 451735 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chua C. L., Brown G., Hamilton J. A., Rogerson S. & Boeuf P. Monocytes and macrophages in malaria: protection or pathology? Trends Parasitol 29, 26–34 (2013). [DOI] [PubMed] [Google Scholar]
- Menendez C. et al. The impact of placental malaria on gestational age and birth weight. J Infect Dis 181, 1740–1745 (2000). [DOI] [PubMed] [Google Scholar]
- Fried M. & Duffy P. E. Maternal malaria and parasite adhesion. J Mol Med (Berl) 76, 162–171 (1998). [DOI] [PubMed] [Google Scholar]
- Silver K. L., Zhong K., Leke R. G., Taylor D. W. & Kain K. C. Dysregulation of angiopoietins is associated with placental malaria and low birth weight. PLoS One 5, e9481 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ataide R. et al. Malaria in Pregnancy Interacts with and Alters the Angiogenic Profiles of the Placenta. PLoS Negl Trop Dis 9, e0003824 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Umbers A. J. et al. Placental malaria-associated inflammation disturbs the insulin-like growth factor axis of fetal growth regulation. J Infect Dis 203, 561–569 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Umbers A. J., Aitken E. H. & Rogerson S. J. Malaria in pregnancy: small babies, big problem. Trends Parasitol 27, 168–175 (2011). [DOI] [PubMed] [Google Scholar]
- Yochem J. & Greenwald I. A gene for a low density lipoprotein receptor-related protein in the nematode Caenorhabditis elegans. Proc Natl Acad Sci USA 90, 4572–4576, (1993). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marzolo M. P. & Farfan P. New insights into the roles of megalin/LRP2 and the regulation of its functional expression. Biol Res 44, 89–105 (2011). [DOI] [PubMed] [Google Scholar]
- Lundgren S. et al. Tissue distribution of human gp330/megalin, a putative Ca(2 + )-sensitive protein. J Histochem Cytochem 45, 383–392 (1997). [DOI] [PubMed] [Google Scholar]
- Lambot N. et al. Evidence for a clathrin-mediated recycling of albumin in human term placenta. Biol Reprod 75, 90–97 (2006). [DOI] [PubMed] [Google Scholar]
- Su A. I. et al. A gene atlas of the mouse and human protein-encoding transcriptomes. Proc Natl Acad Sci USA 101, 6062–6067 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christensen E. I. & Birn H. Megalin and cubilin: multifunctional endocytic receptors. Nat Rev Mol Cell Biol 3, 256–266 (2002). [DOI] [PubMed] [Google Scholar]
- Willnow T. E. et al. Defective forebrain development in mice lacking gp330/megalin. Proc Natl Acad Sci USA 93, 8460–8464 (1996). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leheste J. R. et al. Megalin knockout mice as an animal model of low molecular weight proteinuria. Am J Pathol 155, 1361–1370 (1999). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burke K. A., Jauniaux E., Burton G. J. & Cindrova-Davies T. Expression and immunolocalisation of the endocytic receptors megalin and cubilin in the human yolk sac and placenta across gestation. Placenta 34, 1105–1109 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akour A. A., Gerk P. & Kennedy M. J. Megalin expression in human term and preterm placental villous tissues: effect of gestational age and sample processing and storage time. J Pharmacol Toxicol Methods 71, 147–154 (2015). [DOI] [PubMed] [Google Scholar]
- Kantarci S. et al. Mutations in LRP2, which encodes the multiligand receptor megalin, cause Donnai-Barrow and facio-oculo-acoustico-renal syndromes. Nat Genet 39, 957–959 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oleinikov A. V., Zhao J. & Makker S. P. Cytosolic adaptor protein Dab2 is an intracellular ligand of endocytic receptor gp600/megalin. Biochemical J. 347, 613–621, (2000). [PMC free article] [PubMed] [Google Scholar]
- Gallagher H., Oleinikov A. V., Fenske C. & Newman D. J. The adaptor disabled-2 binds to the third Psi xNPxY sequence on the cytoplasmic tail of megalin. Biochimie 86, 179–182 (2004). [DOI] [PubMed] [Google Scholar]
- Chen W. J., Goldstein J. L. & Brown M. S. NPXY, a sequence often found in cytoplasmic tails, is required for coated pit-mediated internalization of the low density lipoprotein receptor. J Biol Chem 265, 3116–3123 (1990). [PubMed] [Google Scholar]
- Mok S. C. et al. DOC-2, a candidate tumor suppressor gene in human epithelial ovarian cancer. Oncogene 16, 2381–2387 (1998). [DOI] [PubMed] [Google Scholar]
- Fulop V. et al. DOC-2/hDab2, a candidate tumor suppressor gene involved in the development of gestational trophoblastic diseases. Oncogene 17, 419–424 (1998). [DOI] [PubMed] [Google Scholar]
- Tseng C. P. et al. The role of DOC-2/DAB2 protein phosphorylation in the inhibition of AP-1 activity. An underlying mechanism of its tumor-suppressive function in prostate cancer. J Biol Chem 274, 31981–31986 (1999). [DOI] [PubMed] [Google Scholar]
- Xu X. X., Yi T., Tang B. & Lambeth J. D. Disabled-2 (Dab2) is an SH3 domain-binding partner of Grb2. Oncogene 16, 1561–1569, (1998). [DOI] [PubMed] [Google Scholar]
- Maurer M. E. & Cooper J. A. Endocytosis of megalin by visceral endoderm cells requires the Dab2 adaptor protein. J Cell Sci 118, 5345–5355 (2005). [DOI] [PubMed] [Google Scholar]
- Saito A., Sato H., Iino N. & Takeda T. Molecular mechanisms of receptor-mediated endocytosis in the renal proximal tubular epithelium. J Biomed Biotechnol 2010, 403272, (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raychowdhury R., Zheng G., Brown D. & McCluskey R. T. Induction of Heymann nephritis with a gp330/megalin fusion protein. Am J Pathol 148, 1613–1623 (1996). [PMC free article] [PubMed] [Google Scholar]
- Piola P. et al. Efficacy and safety of artemether-lumefantrine compared with quinine in pregnant women with uncomplicated Plasmodium falciparum malaria: an open-label, randomised, non-inferiority trial. Lancet Infect Dis 10, 762–769 (2010). [DOI] [PubMed] [Google Scholar]
- De Beaudrap P. et al. Impact of malaria during pregnancy on pregnancy outcomes in a Ugandan prospective cohort with intensive malaria screening and prompt treatment. Malar J 12, 139 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conroy A. L. et al. Complement activation and the resulting placental vascular insufficiency drives fetal growth restriction associated with placental malaria. Cell Host Microbe 13, 215–226 (2013). [DOI] [PubMed] [Google Scholar]
- Muehlenbachs A., Mutabingwa T. K., Edmonds S., Fried M. & Duffy P. E. Hypertension and maternal-fetal conflict during placental malaria. PLoS Med 3, e446 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ordi J. et al. Massive chronic intervillositis of the placenta associated with malaria infection. Am J Surg Pathol 22, 1006–1011 (1998). [DOI] [PubMed] [Google Scholar]
- Nagai J. et al. Mutually dependent localization of megalin and Dab2 in the renal proximal tubule. Am J Physiol Renal Physiol 289, F569–576 (2005). [DOI] [PubMed] [Google Scholar]
- Tsaroucha A. K. et al. Megalin and cubilin in the human gallbladder epithelium. Clin Exp Med 8, 165–170, (2008). [DOI] [PubMed] [Google Scholar]
- Woollett L. A. Review: Transport of maternal cholesterol to the fetal circulation. Placenta 32 Suppl 2, S218–221 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wadsack C. et al. Intrauterine growth restriction is associated with alterations in placental lipoprotein receptors and maternal lipoprotein composition. Am J Physiol Endocrinol Metab 292, E476–484 (2007). [DOI] [PubMed] [Google Scholar]
- Marino M., Andrews D., Brown D. & McCluskey R. T. Transcytosis of Retinol-Binding Protein across Renal Proximal Tubule Cells after Megalin (gp 330)-Mediated Endocytosis. J Am Soc Nephrol 12, 637–648 (2001). [DOI] [PubMed] [Google Scholar]
- Birn H. et al. Megalin binds and mediates cellular internalization of folate binding protein. Febs J 272, 4423–4430 (2005). [DOI] [PubMed] [Google Scholar]
- Nykjaer A. et al. An endocytic pathway essential for renal uptake and activation of the steroid 25-(OH) vitamin D3. Cell 96, 507–515 (1999). [DOI] [PubMed] [Google Scholar]
- Ray J. G. & Laskin C. A. Folic acid and homocyst(e)ine metabolic defects and the risk of placental abruption, pre-eclampsia and spontaneous pregnancy loss: A systematic review. Placenta 20, 519–529 (1999). [DOI] [PubMed] [Google Scholar]
- Murphy V. E., Smith R., Giles W. B. & Clifton V. L. Endocrine regulation of human fetal growth: the role of the mother, placenta, and fetus. Endocr Rev 27, 141–169 (2006). [DOI] [PubMed] [Google Scholar]
- Shields B. M., Knight B. A., Hill A., Hattersley A. T. & Vaidya B. Fetal thyroid hormone level at birth is associated with fetal growth. J Clin Endocrinol Metab 96, E934–938 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sousa M. M. et al. Evidence for the role of megalin in renal uptake of transthyretin. J Biol Chem 275, 38176–38181 (2000). [DOI] [PubMed] [Google Scholar]
- Patel J., Landers K., Li H., Mortimer R. H. & Richard K. Delivery of maternal thyroid hormones to the fetus. Trends Endocrinol Metab 22, 164–170 (2011). [DOI] [PubMed] [Google Scholar]
- Bolos M., Fernandez S. & Torres-Aleman I. Oral administration of a GSK3 inhibitor increases brain insulin-like growth factor I levels. J Biol Chem 285, 17693–17700 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huet C., Ash J. F. & Singer S. J. The antibody-induced clustering and endocytosis of HLA antigens on cultured human fibroblasts. Cell 21, 429–438 (1980). [DOI] [PubMed] [Google Scholar]
- Gekle M. et al. Transforming growth factor-beta1 reduces megalin- and cubilin-mediated endocytosis of albumin in proximal-tubule-derived opossum kidney cells. J Physiol 552, 471–481 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fried M., Muga R. O., Misore A. O. & Duffy P. E. Malaria elicits type 1 cytokines in the human placenta: IFN-gamma and TNF-alpha associated with pregnancy outcomes. J Immunol 160, 2523–2530 (1998). [PubMed] [Google Scholar]
- Muehlenbachs A. et al. Artemether-lumefantrine to treat malaria in pregnancy is associated with reduced placental haemozoin deposition compared to quinine in a randomized controlled trial. Malar J 11, 150 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bulmer J. N., Rasheed F. N., Francis N., Morrison L. & Greenwood B. M. Placental malaria. I. Pathological classification. Histopathology 22, 211–218 (1993). [DOI] [PubMed] [Google Scholar]
- Muehlenbachs A. et al. A novel histological grading scheme for placental malaria applied in areas of high and low malaria transmission. J Infect Dis 202, 1608–1616 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shahed A. & Young K. A. Anti-Mullerian hormone (AMH), inhibin-alpha, growth differentiation factor 9 (GDF9), and bone morphogenic protein-15 (BMP15) mRNA and protein are influenced by photoperiod-induced ovarian regression and recrudescence in Siberian hamster ovaries. Mol Reprod Dev 80, 895–907 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oleinikov A. V. & Makker S. P. Increased expression of cytoplasmic tail-containing form of gp600/megalin in active Heymann nephritis. J Pathol 192, 251–256 (2000). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.