Abstract
Background
Telomerase is a ribonucleoprotein enzyme that synthesises telomeres after cell division and maintains chromosomal length and stability thus leading to cellular immortalisation. The hTERT (human telomerase reverse transcriptase) subunit seems to be the rate-limiting determinant of telomerase and knowledge of factors controlling hTERT transcription may be useful in therapeutic strategies. The hTERT promoter contains binding sites for c-Myc and there is experimental and in vitro evidence that c-Myc may increase hTERT expression.
Materials and methods
RNA was extracted from 18 breast carcinomas and c-Myc mRNA expression was estimated by quantitative reverse transcriptase-PCR (RT-PCR) with Taqman methodology. These tumours had already been analysed for ER and PgR status using ligand-binding assays and had had their DNA ploidy and S-phase fractions measured by flow cytometry. Telomerase activity had already been determined by using a modified telomeric repeat and amplification protocol (TRAP) assay.
Results
Telomerase activity ranged from 0 to 246 units of Total Protein Generated (TPG), where one unit of TPG was equal to 600 molecules of telomerase substrate primers extended by at least three telomeric repeats. Median levels of TPG were 60 and mean levels 81. There was no significant correlation between levels of c-Myc mRNA expression, telomerase activity, S phase fraction or PgR. There was a significant negative correlation with ER status.
Conclusion
Although the hTERT promoter contains potential binding sites for c-Myc oncoprotein, we have found no correlation between c-Myc mRNA levels and telomerase activity.
Keywords: telomerase, c-Myc, breast, cancer
Introduction
Telomerase is a multi-component ribonucleoprotein located within the nucleus, the function of which is to synthesise the repetitive nucleotide sequence forming the telomeres at the end of chromosomes [1]. During cell division, DNA polymerase is unable to fully replicate the ends of linear DNA and genetic material is lost which eventually can result in chromosome instability and cellular senescence. Telomerase synthesises a new copy of the telomere repeat [1] so that cellular proliferation can continue leading to cellular immortality [2,3]. Telomerase is active in 70 – 90% of malignant tissues and many immortal cell lines, but most somatic cells have no detectable telomerase activity. Telomerase activity has been shown in some cancers to correlate with prognostic variables [4,5]. We detected telomerase activity in 74% of invasive breast cancers and in none of benign or normal breast tissue specimens. Furthermore, we observed a correlation between telomerase activity and tumour size, nodal status, lymphovascular invasion and Ki-67 expression [6,7]. The fundamental components of telomerase have been identified as the RNA template (hTR), the reverse transcriptase (hTERT) and telomerase associated proteins, including (TEP1) [1,8,9]. Of these hTR and TEP1 are expressed ubiquitously in both normal and cancerous tissue [8], whereas hTERT is detectable in tumour cells but not in normal somatic cells [9-11]. Telomerase enzyme activity can be reconstituted in fibroblasts by the ectopic expression of hTERT [12] and induction of hTERT expression has been shown to be essential for telomerase activation in cell-lines [9,10]. These observations suggest that hTERT is the rate-limiting determinant of telomerase enzyme activity. We have recently reported that hTERT mRNA is much higher in breast cancer specimens compared with adjacent non-cancerous breast tissue [13] and that telomerase activity is significantly correlated with hTERT mRNA expression [14]. Investigation of the mechanisms of hTERT control is important in elucidating the pathways that may be amenable to therapeutic manipulation and one such pathway involves the transcription factor Myc.
An increased level of c-Myc occurs frequently in a wide range of tumours [15,16] due to de-regulated expression of myc through gene amplification, retroviral insertion or chromosomal translocation. Sequence analysis of the hTERT gene promoter has shown the presence of at least 2 [17] and perhaps as many as 29 E boxes [18], potential binding sites for the Myc oncoprotein, and the possibility of a regulatory role for Myc has been explored in a number of studies. It has been found that purified Myc interacts with the E box sequences and that cotransfection of Myc induces activity in the isolated hTERT promoter [19]. It has been shown that retroviral expression of c-myc increases the amount of hTERT mRNA in human mammary epithelial cells and fibroblasts and telomerase activity could thereafter be detected [20]. It has also been reported that expression of c-Myc leads to increased hTERT expression and telomerase activity in human B cells [21]. Moreover, since this does not require protein synthesis this appears to be due to a direct effect of Myc on the hTERT promoter and not secondary to an increase in cellular proliferation by Myc [21]. In addition, the introduction of Myc anti-sense RNA has been shown to lead to a reduction in hTERT promoter activity [19].
Our aim in this study was to investigate whether the level of c-Myc mRNA expression correlates with telomerase activity in human breast carcinomas.
Materials and methods
Local ethical approval was obtained. Specimens of human breast carcinoma tissue (n = 18) were acquired from the MD Anderson centre (Texas, USA). These had been analysed already for quantitative telomerase activity (using a variation on the TRAP assay), ER (ER+ = ≥3 fmol/mg protein), PgR, (PgR+ = ≥5 fmol/mg protein), S-phase fraction (<6% = low, 6–10% = intermediate, >10% = high}DNA and ploidy status (diploid or aneuploid), by this unit as described previously [22].
RNA extraction
Total cellular RNA was extracted from 10–20 mg of these tumours using the RNeasy Mini isolation kit (Qiagen, Hilden, Germany) according to the manufacturer's protocol. Quantification of the RNA following treatment with DNase (Promega) was carried out in triplicate using the RiboGreen reagent (Molecular Probes Europe BV) according to the manufacturers protocol.
RT-PCR
TaqMan RT-PCR was performed in duplicate for each sample using the ABI PRISM 7700 Sequence Detector (Perkin-Elmer Applied Biosystems) and the TaqMan EZ RT-PCR Core Reagents Kit (Perkin-Elmer Applied Biosystems). The RT-PCR was performed for c-Myc using the forward and reverse oligonucleotide primers and TaqMan probes given in Table 1. These oligonucleotides were designed using Primer Express software (PE-Applied Biosystems, Warrington, UK) using gene sequences obtained from the GenBank database. Primers and probe were intron spanning to prevent amplification of genomic DNA and define a 71 base pair amplicon crossing from exons 2 to 3 in the c-myc gene (accession number V00568.1)
Table 1.
Sequences of TaqMan Primers and Probes for RT-PCR.
| Gene | Forward | Reverse | Probe |
| c-Myc | 5'TGA GGA GAC ACC GCCCAC3' | 5'CAA CAT CGA TTTCTT CCT CAT CTT C3' | 5'CCA GCA GCG ACT CTG AGG AGG AAC A3' |
Conditions for the reverse transcription step were 50°C for 2 minutes, 60°C for 10 minutes, and 92°C for 5 minutes. The polymerase chain reaction was carried out for 50 cycles of 20 seconds at 92°C and 25 seconds at 62°C. For the negative control RNase free water was used in the RT-PCR mix instead of RNA. For the positive control, the amplicon of C-Myc was used. A standard curve for c-Myc was constructed using serial dilutions of a single stranded sense oligonucleotide specifying the amplicon as previously described by Bustin et al [23] and this together with the known RNA concentration was used to quantify the mRNA copy number per microgram of RNA.
Results and statistical analysis
Telomerase activity varied between 0 and 246 units of Total Protein Generated (TPG), where one unit of TPG was equal to 600 molecules of telomerase substrate primers extended by at least three telomeric repeats [figure 1].
Figure 1.
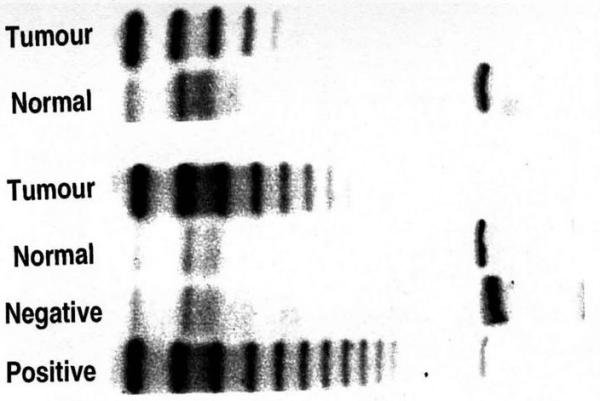
TRAP assay results showing telomerase positive and telomerase negative samples. Positivity is indicated by the presence of a DNA ladder after electrophoresis on a polyacrylamide gel.
Median levels of TPG were 60 and mean levels 81. C-Myc mRNA levels were between 1.14 × 106 and 2.14 × 108 (mean 6.92 × 107) copy numbers per μg of RNA. Statistical analysis was performed using SAS (Version 6.11, SAS Institute, Cary, North Carolina). The C-Myc expression, ER, PgR, S-phase fraction and telomerase activity were considered as continuous variables and the relationship between them was investigated using Spearman's nonparametric correlation coefficient. No significant relationship was found between C-Myc expression and telomerase activity (rs = 0.05691, p = 0.8225), [figure 2]. C-Myc mRNA expression was however, found to be inversely correlated with ER status (rs = -0.52583, p = 0.0250) but not to correlate with PgR status (rs = -0.149, p = 0.55) or S phase fraction (rs= -0.334, p = 0.1708).
Figure 2.
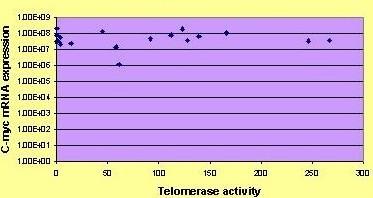
Graphic presentation of c-Myc mRNA expression and telomerase activity.
We also examined whether there are any differences in c-Myc mRNA expression by ER status (ER+ = ≥3 fmol/mgprotein), PgRstatus (PgR+ = ≥5 fmol/mg protein), DNA ploidy status (diploid or aneuploid), S-phase fraction (<6% = low, 6–10% = intermediate, >10% = high) and observed a strong relationship was observed between c-Myc mRNA expression and diploidy (p = 0.03) [Tables 2, 3].
Table 2.
The relationships between c-Myc mRNA expression and, ER, PgR, ploidy and SPF using Kruskal-Wallis p-values.
| factor | N | Mean | SD | Median | p-value |
| cmyc | |||||
| ER- | 17 | 69580146 | 61984126 | 40202108 | 0.63 |
| ER+ | 1 | 63344107 | . | . | |
| PgR- | 10 | 59102005 | 59941659 | 38086495 | 0.21 |
| PgR+ | 8 | 81898317 | 61947631 | 74842241 | |
| Diploid | 9 | 1.0277E8 | 68918774 | 86487036 | 0.038 |
| Aneuploid | 9 | 35698918 | 20212590 | 39212925 | |
| LowSPF | 9 | 85567170 | 55632400 | 86340375 | 0.34 |
| IntermediateSPF | 3 | 79641669 | 117177069 | 23457155 | |
| HighSPF | 6 | 39529509 | 15928240 | 39707516 |
Table 3.
Spearman correlation coefficients (SCCs) demonstrating no significant correlation between c-Myc mRNA expression and TPG by telomerase. However there is a significant correlation between hTERT mRNA expression and TPG and between c-Myc and absence of ER. Each cell of the table shows SCC, p-value and number of observation.
| htert | cmyc | |
| ER | -0.36467 | -0.52583 |
| 0.1368 | 0.0250 | |
| 18 | 18 | |
| PgR | 0.07671 | -0.14910 |
| 0.7622 | 0.5549 | |
| 18 | 18 | |
| S-phase | -0.21981 | -0.33746 |
| 0.3808 | 0.1708 | |
| 18 | 18 | |
| TPG | 0.51112 | 0.05691 |
| 0.0302 | 0.8225 | |
| 18 |
Conclusion
Although the hTERT promoter contains E-boxes, we have found no correlation between c-Myc mRNA levels and telomerase activity.
The control of hTERT is undoubtedly a complex one and it is likely that a number of other transcription factors influence its expression than c-Myc. These might act together with c-Myc, as has been shown for Sp1 [24] or independently. In this respect, it has been shown that transfer of a normal chromosome 3 into human breast carcinoma cells results in abolition of hTERT transcripts without any change in c-Myc levels [25]. Furthermore, it is known that another member of the Myc family, Mad1 forms a complex with Max and acts as a transcriptional repressor at the same binding sites as Myc-Max [16]. It has been shown that, the proportion of Mad1 binding to the hTERT promoter rises and that of Myc falls, during the differentiation of HL60 cells [26]. This is associated with reduced acetylation of the hTERT promoter and measurement of the Mad/Myc ratio is likely to be important in establishing the overall level of transcriptional activation of hTERT.
Contributor Information
Katharine L Kirkpatrick, Email: kaffdabe@aol.com.
Robert F Newbold, Email: robert.newbold@brunel.ac.uk.
Kefah Mokbel, Email: kefahmokbel@hotmail.com.
References
- Mokbel K. The role of telomerase in breast cancer. [Review] [68 refs] Eur J Surg Oncol. 2000;26:509–514. doi: 10.1053/ejso.1999.0932. [DOI] [PubMed] [Google Scholar]
- Blackburn E. Switching and Signaling at the Telomere. Cell. 2001;106:661–673. doi: 10.1016/S0092-8674(01)00492-5. [DOI] [PubMed] [Google Scholar]
- Zhu J, Wang H, Bishop J, Blackburn E. Telomerase Extends the Lifespan of Birus-Transformed Human Cells Without Net Telomere Lengthening. Proc Natl Acad Sci USA. 1999;96:3723–3728. doi: 10.1073/pnas.96.7.3723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim N, Piatyszek MA, Prowse K. Specific association of human telomerase activity with immortal cells and cancer. Science. 1994;266:2011–2015. doi: 10.1126/science.7605428. [DOI] [PubMed] [Google Scholar]
- Shay JW, Bacchetti S. A survey of telomerase activity in human cancer. Eur J Cancer. 1997;33:787–791. doi: 10.1016/S0959-8049(97)00062-2. [DOI] [PubMed] [Google Scholar]
- Mokbel K, Parris CN, Ghilchik M, Williams G, Newbold RF. The association between telomerase, histopathological parameters, and KI-67 expression in breast cancer. Am J Surg. 1999;178:69–72. doi: 10.1016/S0002-9610(99)00128-2. [DOI] [PubMed] [Google Scholar]
- Mokbel KM, Parris CN, Ghilchik M, Amerasinghe CN, Newbold RF. Telomerase activity and lymphovascular invasion in breast cancer. Eur J Surg Oncol. 2000;26:30–33. doi: 10.1053/ejso.1999.0736. [DOI] [PubMed] [Google Scholar]
- Wu A, Ichihashi M, Ueda M. Correlation of the Expression of Human Telomerase Subunits With telomerase activity in Normal Skin and Skin Tumors. Cancer. 1999;86:2038–2044. doi: 10.1002/(SICI)1097-0142(19991115)86:10<2038::AID-CNCR22>3.0.CO;2-A. [DOI] [PubMed] [Google Scholar]
- Kirkpatrick KL, Mokbel K. The significance of human telomerase reverse transcriptase in human cancer. Eur J Surg Oncol. 2001;27:754–760. doi: 10.1053/ejso.2001.1151. [DOI] [PubMed] [Google Scholar]
- Meyerson M, Counter CM, Eaton EN, Ellisen LW, Steiner P, Caddle SD, Ziaugra L, Beijersbergen RL, Davidoff MJ, Liu Q, Bacchetti S, Haber DA, Weinberg RA. hEST2, the putative human telomerase catalytic subunit gene, is up-regulated in tumor cells and during immortalization. Cell. 1997;90:785–795. doi: 10.1016/S0092-8674(00)80538-3. [DOI] [PubMed] [Google Scholar]
- Nakamura TM, Cooper JP, Cech TR. Two modes of survival of fission yeast without telomerase. Science. 1998;282:493–496. doi: 10.1126/science.282.5388.493. [DOI] [PubMed] [Google Scholar]
- Weinrich SL, Pruzan R, Ma L, Ouellette M, Tesmer VM, Holt SE, Bodnar AG, Lichtsteiner S, Kim MW, Trager JB, Taylor RD, Carlos R, Andrews WH, Wright JE, Shay JW, Harley CB, Morin GB. Reconstitution of human telomerase with the template RNA component hTR and the catalytic protein subunit hTRT. Nat Genet. 1997;17:498–502. doi: 10.1038/ng1297-498. [DOI] [PubMed] [Google Scholar]
- Kirkpatrick KL, Ogunkolade W, Elkak AE, Bustin SA, Jenkins P, Ghilchik M, Newbold RF, Mokbel K. hTERT Expression in human breast cancer and non-cancerous breast tissue; correlation with tumour stage and c-Myc expression. Breast Cancer Res Treat. 2003;77:277–284. doi: 10.1023/A:1021849217054. [DOI] [PubMed] [Google Scholar]
- Kirkpatrick K, Clark G, Ghilchick M, Newbold RF, Mokbel K. hTERT mRNA expression correlates with telomerase activity in human breast cancer. Eur J Surg Oncol. 2003;29:321–326. doi: 10.1053/ejso.2002.1374. [DOI] [PubMed] [Google Scholar]
- Liao D, Dickson R. c-Myc in breast cancer. Endocr Relat Cancer. 2000;7:143–164. doi: 10.1677/erc.0.0070143. [DOI] [PubMed] [Google Scholar]
- Grandori C, Cowley S, James L, Eisenman R. The myc/max/mad network and the transcriptional control of cell behaviour. Annu Rev Cell Dev Biol. 2000;16:653–699. doi: 10.1146/annurev.cellbio.16.1.653. [DOI] [PubMed] [Google Scholar]
- Cong YS, Wen J, Bacchetti S. The human telomerase catalytic subunit hTERT: organization of the gene and characterization of the promoter. Hum Mol Genet. 1999;8:137–142. doi: 10.1093/hmg/8.1.137. [DOI] [PubMed] [Google Scholar]
- Cerni C. Telomeres, telomerase, and myc. An update. [Review] [139 refs] Mutat Res. 2000;462:31–47. doi: 10.1016/S1383-5742(99)00091-5. [DOI] [PubMed] [Google Scholar]
- Oh S, Song YH, Kim UJ, Yim J, Kim TK. In vivo and in vitro analyses of Myc for differential promoter activities of the human telomerase (hTERT) gene in normal and tumor cells. Biochem Biophys Res Commun. 1999;263:361–365. doi: 10.1006/bbrc.1999.1366. [DOI] [PubMed] [Google Scholar]
- Wang J, Xie LY, Allan S, Beach D, Hannon GJ. Myc activates telomerase. Genes Dev. 1998;12:1769–1774. doi: 10.1101/gad.12.12.1769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu KJ, Grandori C, Amacker M, Simon-Vermot N, Polack A, Lingner J, Dalla-Favera R. Direct activation of TERT transcription by c-MYC. Nat Genet. 1999;21:220–224. doi: 10.1038/6010. [DOI] [PubMed] [Google Scholar]
- Clark G, Osborne C, Levitt D, Wu F, Kim N. Telomerase Activity and Survival of Patients with Node-Positive Breast Cancer. J Natl Cancer Inst. 1997;89:1875–1881. doi: 10.1093/jnci/89.24.1874. [DOI] [PubMed] [Google Scholar]
- Bustin SA, Gyselman VG, Williams NS, Dorudi S. Detection of cytokeratins 19/20 and guanylyl cyclase C in peripheral blood of colorectal cancer patients. Br J Cancer. 1999;79:1813–1820. doi: 10.1038/sj.bjc.6690289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kyo S, Takakura M, Taira T, Kanaya T, Itoh H, Yutsudo M, Ariga H, Inoue M. Sp1 cooperates with c-Myc to activate transcription of the human telomerase reverse transcriptase gene (hTERT) Nucleic Acids Res. 2000;28:669–677. doi: 10.1093/nar/28.3.669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ducrest AL, Amacker M, Mathieu Y, Cuthbert AP, Trott DA, Newbold RF, Nabholz M, Lingner J. Regulation of Human Telomerase Activity. Cancer Res. 2001;61:7594–7602. [PubMed] [Google Scholar]
- Xu D, Popov N, Hou M, Wang Q, Bjorkholm M, Gruber A, Menkel AR, Henriksson M. Switch from Myc/Max to Mad1/Max binding and decrease in histone acetylation at the telomerase reverse transcriptase promoter during differentiation of HL60 cells. Proc Natl Acad Sci USA. 2001;98:3826–3831. doi: 10.1073/pnas.071043198. [DOI] [PMC free article] [PubMed] [Google Scholar]


