Abstract
Background and Purpose
Hydrogen sulfide (H2S) is a gasotransmitter produced from l‐cysteine through the enzymatic action of cystathionine‐γ‐lyase (CSE) and/or cystathionine‐β‐synthase. d‐Penicillamine is the d isomer of a dimethylated cysteine and has been used for the treatment of rheumatoid arthritis. As d‐penicillamine is structurally very similar to cysteine, we have investigated whether d‐penicillamine, as a cysteine analogue, has an effect on the H2S pathway.
Experimental Approach
We tested the effect of d‐penicillamine (0.01–1 mM) in mouse aortic rings mounted in isolated organ baths and determined whether it could affect H2S biosynthesis. In particular, we investigated any possible inhibitor or donor behaviour by using recombinant enzyme‐based assays and an in vivo approach.
Key Results
d‐Penicillamine, per se, showed little or no vasodilator effect, and it cannot be metabolized as a substrate in place of l‐cysteine. However, d‐penicillamine significantly reduced l‐cysteine‐induced vasodilatation in a concentration‐dependent manner through inhibition of H2S biosynthesis, and this effect occurred at concentrations 10 times lower than those needed to induce the release of H2S. In particular, d‐penicillamine selectively inhibited CSE in a pyridoxal‐5′‐phospate‐dependent manner.
Conclusions and Implications
Taken together, our results suggest that d‐penicillamine acts as a selective CSE inhibitor, leading to new perspectives in the design and use of specific pharmacological tools for H2S research. In addition, the inhibitory effect of d‐penicillamine on CSE could account for its beneficial action in rheumatoid arthritis patients, where H2S has been shown to have a detrimental effect.
Abbreviations
- MPST
3‐mercaptopyruvate sulfurtransferase
- CBS
cystathionine‐β‐synthase
- CSE
cystathionine‐γ‐lyase
- DPD
N,N‐dimethyl‐p‐phenylenediamine sulfate
- d‐pen
d‐penicillamine
- l‐pen
l‐penicillamine
- H2S
hydrogen sulfide
- IVM
intravital microscopy
- PAG
d,l‐propargylglycine
- PE
phenylephrine
- PLP
pyridoxal‐5′‐phosphate
- RA
rheumatoid arthritis
- TCA
trichloroacetic acid
- ZnAc
zinc acetate
Tables of Links
| TARGETS |
|---|
| 3‐mercaptopyruvate sulfurtransferase (MPST) |
| Cystathionine‐β‐synthase (CBS) |
| Cystathionine‐γ‐lyase (CSE) |
These Tables list key protein targets and ligands in this article which are hyperlinked to corresponding entries in http://www.guidetopharmacology.org, the common portal for data from the IUPHAR/BPS Guide to PHARMACOLOGY (Pawson et al., 2014) and are permanently archived in the Concise Guide to PHARMACOLOGY 2015/16 (Alexander et al., 2015).
Introduction
Hydrogen sulfide (H2S) is a gaseous molecule endogenously synthesized by cystathionine‐γ‐lyase (CSE), cystathionine‐β‐synthase (CBS) and 3‐mercaptopyruvate sulfurtransferase (MPST). These enzymes are differently distributed throughout the human body and within cell compartments. MPST is mainly expressed in mitochondria (Stipanuk, 2004; Shibuya et al., 2009), while CSE and CBS show a wide distribution in diverse cell types. CSE and CBS can both metabolize the substrate l‐cysteine to release H2S, and this reaction is strictly dependent upon the enzyme cofactor pyridoxal‐5′‐phosphate (PLP). The role of H2S has been widely investigated in several organs and tissues, and many studies have confirmed its crucial role in body physiology. For instance, CSE represents the prominent enzyme within the cardiovascular system, where H2S is a major player involved in regulating the function of heart and blood vessels (Predmore and Lefer, 2011; Wang, 2011). Yang and co‐workers (2008) have reported that a lack of functional CSE in mice leads to hypertension and reduces endothelial activation (Yang et al., 2008), while an increase in H2S levels leads to the attenuation of ischaemia‐induced heart failure in mice (Calvert et al., 2010). These observations highlight a key role for H2S in cardiovascular homeostasis. Moreover, recent reports have also revealed that H2S has a key role in the inflammatory process. Indeed, different studies have demonstrated that H2S is part of the pro‐resolutive system, stopping inflammation and promoting healing (Zanardo et al., 2006; Wallace, 2012). H2S has also been shown to counteract inflammation by triggering resolutive pathways through the engagement of annexin A1 (Brancaleone et al., 2014). Conversely, other reports have shown that H2S has a pro‐inflammatory role (Collin et al., 2005; Li et al., 2005; Zhang et al., 2006). Rheumatoid arthritis (RA) represents one of those particular conditions, where H2S has been described as a pro‐inflammatory mediator and arthritic patients have higher levels of H2S compared with healthy subjects (Whiteman et al., 2010; Kloesch et al., 2011; Muniraj et al., 2014). Interestingly, in the late 1970s RA patients were treated with d‐penicillamine (d‐pen), a degradation product derived from penicillin and used as a disease‐modifying anti‐rheumatic drug. d‐Pen was found to be effective at reducing the rheumatoid factor and ameliorating the symptoms of the disease (Jaffe, 1964; Dixon et al., 1975). From a purely chemical point of view, penicillamine is a β,β‐dimethyl‐cysteine (Figure 1). The structural analogy between cysteine and d‐pen recalled a similar analogy occurring between arginine and its methylated derivatives, l‐NΩ‐monomethyl‐arginine (l‐NMMA) and l‐asymmetric dimethylarginine (ADMA). l‐Arginine acts as the substrate for nitric oxide synthase (NOS) leading to NO production. However, l‐NMMA and ADMA are both inhibitors of NOS activity, and are obtained by inserting one or two methyl groups on the l‐arginine backbone. Although with penicillamine this occurs on the nitrogen atom rather than the α‐carbon, we hypothesized that the dimethylated cysteine, penicillamine, could inhibit H2S synthesizing enzymes. In the current literature, among the compounds used to efficiently block H2S biosynthesis, propargylglycine (PAG) is the most selective (Asimakopoulou et al., 2013). However, for PAG to be effective it has to be applied at concentrations within the millimolar range (up to 10 mM) (Whiteman et al., 2011). Such a high concentration can also lead to non‐selective effects and could interfer with the activity of other enzymes (Ressler et al., 1964; Rej, 1977; Burnett et al., 1980; Ochs and Harris, 1980). Therefore, there is a strong need for compounds that can be used at lower concentrations in order to minimize interactions with other enzymes unrelated to H2S. Here, we demonstrated that d‐pen is a selective CSE inhibitor, and is more potent at inhibiting the synthesis of H2S than PAG.
Figure 1.
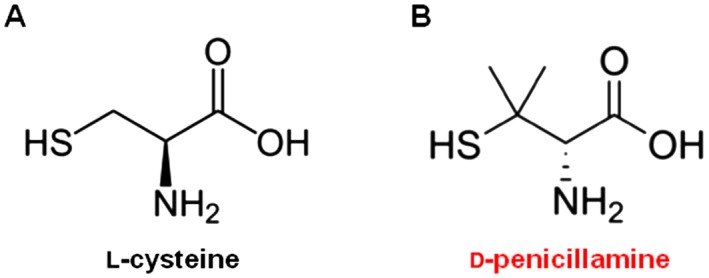
Chemical structures of (A) l‐cysteine and (B) d‐penicillamine.
Methods
Animals
CD‐1 male mice (8–12 weeks of age, 20–25 g of weight) were purchased from Charles River (Milano, Italy) and kept in animal care facility under controlled temperature, humidity and light/dark cycle and with food and water ad libitum. All animal procedures were performed according to the Declaration of Helsinki (European Union guidelines on use of animals in scientific experiments) and following ARRIVE guidelines (Kilkenny et al., 2010; McGrath & Lilley, 2015). All procedures were approved by the local animal care office (Centro Servizi Veterinari Università degli Studi di Napoli ‘Federico II’) and carried out following recommendations for experimental design and analysis in pharmacology as reported by Curtis et al., 2015. A total of 45 animals were used for the experiments described here.
Vascular tissue preparation
Thoracic aorta from CD‐1s male mice were used. Mice were anaesthetized with enflurane (5%) and then killed in CO2 chamber (70%); the aorta was rapidly harvested, and adherent connective and fat tissue were removed. Rings of 1–1.5 mm length were cut and placed in organ baths (3.0 mL) filled with oxygenated (95% O2–5% CO2) Krebs solution and kept at 37°C. The rings were connected to an isometric transducer (7006, Ugo Basile, Comerio, Italy) and changes in tension were continuously recorded with a computerized system (DataCapsule‐17400, UgoBasile, Comerio, Italy). The composition of the Krebs solution was as follows (mM): 118 NaCl, 4.7 KCl, 1.2 MgCl2, 1.2 KH2PO4, 2.5 CaCl2, 25 NaHCO3 and 10.1 glucose. The rings were initially stretched until a resting tension of 1.5 g was reached and then were allowed to equilibrate for at least 30 min; during this period the tension was adjusted, when necessary, to 1.5 g and the bath solution was periodically changed.
Isolated organ bath study
In each set of experiments, rings were firstly challenged with phenylephrine (PE, 1 μM) until the responses were reproducible. In order to verify the integrity of the endothelium, cumulative concentration‐response curves to ACh (10 nM–30 μM) were performed with PE pre‐contracted rings. Rings not reaching a relaxation response of at least 75% were discarded. Tissues were then washed and contracted with PE (1 μM) and, once the plateau was reached, cumulative concentration‐response curves to L‐cysteine (l‐cys, 100 nM–1 mM) were obtained. d‐Pen was first tested for its own vasoactive response (100 nM–1 mM). l‐Penicillamine (l‐pen, 100 nM–1 mM) was also tested as an l‐isomer control for d‐pen.
In a separate set of experiments the inhibitory effects of d‐pen on l‐cys‐induced relaxation were assessed; rings were pre‐incubated with d‐pen (0.01–0.1 mM) for 15 min and then an l‐cys cumulative concentration‐response curve was performed.
In a third set of experiments we applied the same protocols in the presence of an excess of PLP. Aortic rings were pre‐incubated with PLP (0.3 mM) for 5 min, then d‐pen (0.1 mM) was added. After 15 min, a cumulative concentration‐response curve to l‐cys was performed. The optimal concentration of PLP used in this setting was determined in preliminary experiments by assessing the effects of different concentration of PLP (0.1–0.3 mM). The % vasorelaxation was calculated as follows: [(maximal decrease in tension)/total amplitude of contraction] × 100. Krebs solution was used as the vehicle for all isolated organ bath experiments.
H2S assay
The concentrations of H2S in the thoracic aorta were determined by using a methylene blue‐based assay (Stipanuk and Beck, 1982). Briefly, thoracic aortas were dissected, placed in sterile PBS and any fat and connective tissue removed. Aortic rings were homogenized in a potassium phosphate lysis buffer, 100 mM pH 7.4, containing sodium orthovanadate (1 mM) and a protease inhibitor cocktail (1:1000 dilution), and the protein concentration was determined by using the Bradford assay (Bio‐Rad Laboratories, Milano, Italy). The lysates were added in a reaction mixture (total volume 500 μL) containing PLP (0.08 mM), l‐cys (0.4 mM) and saline (30 μL) in the presence of d‐pen (0.01–1 mM) or vehicle. d,l‐Propargylglycine (PAG, 1–10 mM) was used as positive control for inhibition of H2S biosynthesis. Inhibitors were added 10 min before the addition of l‐cys addition. In another set of experiments, the effect of d‐pen (0.1 mM) on H2S production was also tested in the presence of PLP (0.1–0.3 mM) in the reaction mixture. The reaction was performed in parafilm‐sealed eppendorf tubes and initiated by transferring tubes from ice to a 37°C water bath. After 40 min incubation, zinc acetate (ZnAc, 1%, 250 μL) was added to trap any H2S produced followed by trichloroacetic acid (TCA, 10%, 250 μL). Subsequently, N,N‐dimethyl‐p‐phenylenediamine sulfate (DPD, 20 μM, 133 μL) in 7.2 M HCl and FeCl3 (30 μM, 133 μL) in 1.2 M HCl were added. After 20 min, absorbance values were measured at a wavelength of 668 nm. All samples were assayed in duplicate, and the H2S concentration was calculated with reference to a calibration curve for NaHS (3.12–250 μM). Results are expressed as nmol mg‐1 of protein min‐1.
Recombinant CSE and CBS enzymes
The ability of d‐pen to inhibit CSE and/or CBS was determined by using GST‐CSE (rCSE) or GST‐CBS (rCBS) recombinant enzymes (5 μg of protein 100 μL‐1 of reaction mixture) as previously described (Asimakopoulou et al., 2013). All samples were assayed in duplicate, and H2S concentration was calculated with reference to a calibration curve for NaHS (3.12–250 μM). Results are expressed as nmol mg‐1 of protein min‐1.
H2S release in cell‐free assay
The ability of d‐pen to evoke the release of H2S was evaluated in an amperometric approach by using an Apollo‐4000 Free Radical Analyzer (WPI) detector and H2S‐selective minielectrodes, as previously described (Martelli et al., 2014). Briefly, the H2S‐selective minielectrode (polarizing voltage 150 mV) was equilibrated in 10 mL of the PBS solution at pH 7.4. Then, 100 μL of d‐pen solution were added to achieve a final concentration of 1 mM, and the generation of H2S was monitored for 15 min. The assay was performed in the presence of l‐cys, used as nucleophilic agent. l‐Cys per se does not cause the release of H2S (Martelli et al., 2014). The H2S concentration was determined by referring to a calibration curve plotted as amperometric currents (recorded in pA) against corresponding H2S standards obtained with NaHS (1–10 μM) at pH 4.0. In a separate set of experiments, we also evaluated the non‐enzymatic H2S release mediated by l‐pen. PBS was used as a vehicle control.
Intravital microscopy (IVM) in mouse mesenteric microcirculation
IVM was performed as previously reported (Gavins et al., 2003). Mice were treated with TNFα (500 ng per mouse i.p., 2 h) alone (vehicle, 0.2% carboxymethylcellulose, oral gavage 1 h before TNFα injection) or in combination with d‐pen (30 mg·kg−1, oral gavage, 1 h before TNFα injection). PAG (10 mg·kg‐1, i.p. 30 min before TNFα injection) was also used as control inhibitor of H2S biosynthesis. In all cases, CD‐1 mice were anaesthetized with ketamine (100 mg·kg−1) in combination with xylazine (10 mg·kg−1) and placed in the supine position on a heating pad (37°C). The level of anaesthesia was assessed by checking the hind limb pinch reflex. A cautery incision was made along the abdominal region, and the vascular bed was exposed and positioned under the microscope while superfused with warmed (37°C) bicarbonate‐buffered solution at a rate of 2 mL·min−1. Recording started after a 5 minute equilibration period and was made in one to three randomly selected postcapillary venules for each mouse (20–40 μm diameter; visible length > 100 μm). Thus, leukocyte adhesion reflected cells stationary for 30 s or longer, while leukocyte emigration was calculated as the number of cells in a 100 × 50 μm2 area, on both sides of the 100 μm vessel segment. At the end of the experiment, because this was a terminal procedure, all animals were killed by dislocation of the neck while still anaesthetized.
Materials
NaCl, KCl, MgCl2, KH2PO4, CaCl2, NaHCO3, glucose, PE, ACh, NaHS, l‐cys, PAG, PLP, d‐pen, l‐pen, K2HPO4, KH2PO4, PLP, ZnAc, DPD, FeCl3,TCA, ketamine, xylazine, sodium orthovanadate and protease inhibitors were all purchased from Sigma‐Aldrich (Milan, Italy). The TNFα was obtained from eBioscience (Hatfield, UK).
Statistical analysis
Statistical analysis was performed by using graphpad Prism 5.0 software (San Diego, CA, USA). All data are reported as mean ± SEM, and the number of replicates was at least n of 5 per group for each data set. Statistical analysis was performed by using one‐way ANOVA followed by Dunnett's post test when comparing more than two groups or two‐way analysis of variance (ANOVA) for multiple comparisons followed by Bonferroni's post test. Post hoc tests were performed when ANOVAs indicated that a significant difference existed between the groups. All statistical tests performed showed no significant variance in data set homogeneity. Data were considered statistically significant when a value of P < 0.05 was achieved. The data and statistical analysis comply with the recommendations on experimental design and analysis in pharmacology (Curtis et al., 2015).
Results
Effect of d‐pen on isolated aorta
In a preliminary set of experiments, we first evaluated whether d‐pen could induce a vasoactive effect by itself. Therefore, we performed a concentration‐response curve on aorta rings pre‐contracted with PE 1 μM. As reported in Figure 2, d‐pen did show a very weak vasodilator effect (~16%) when compared with that induced by l‐cys (Figure 2A). In addition, we also tested the l isomer of penicillamine (l‐pen) as a control. In contrast to d‐pen, l‐pen showed a consistent vasodilator effect, almost overlapping that induced by l‐cys (Supporting Information Fig. S1). Because penicillamine displays a free thiol group, we hypothesized that the ‐SH moiety might allow for non‐enzymatic release of H2S (Bucci et al., 2014). The amperometric measurement, a validated method for measuring H2S release (Supporting Information Fig. S2) (Martelli et al., 2014), revealed that d‐pen did not induce the release of appreciable amounts of H2S. However, in the presence of l‐cys, used as nucleophilic agent, a small effect was detectable. Conversely, when l‐pen was tested, the release of H2S was significantly higher than that detected for d‐pen (Supporting Information Fig. S3).
Figure 2.
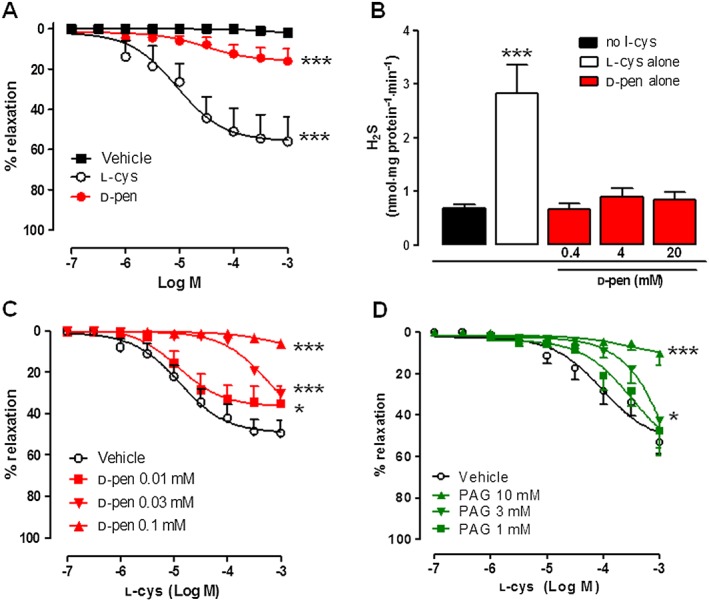
Vasoactive effect of d‐penicillamine (d‐pen). (A) Concentration‐response curves for d‐pen, l‐cysteine (l‐cys and the vehicle in aortic rings contracted with phenylephrine (1 uM, n = 6). * Indicates significant differences (P < 0.05) for comparisons with vehicle. (B) H2S production assay in homogenated whole aorta samples following l‐cys (0.4 mM) or d‐pen (0.4–20 mM) added as substrate for H2S biosynthesis (n = 6). * Indicates significant difference (P < 0.05) for comparison with no l‐cys. (C) Effect of d‐pen (0.01–0.1 mM, 15 min) on l‐cys induced vasodilatation in isolated aortic rings (n = 6). * Indicates significant difference (P < 0.05) for comparison with vehicle. (D) Effect of propargylglycine (PAG, 1–10 mM, 15 min, n = 6) on l‐cys‐induced vasodilatation in isolated aortic rings. * Indicates significant difference (P < 0.05) for comparison with vehicle.
In order to further clarify whether d‐pen might act as a substrate for H2S production, we performed H2S biosynthesis assay in homogenated aorta samples. Whole aorta lysate was used as an enzyme source and d‐pen was added to the sample instead of l‐cys to induce H2S production. As shown in Figure 2B, in contrast to l‐cys, the addition of d‐pen up to a concentration of 20 mM did not cause any significant increase in H2S production (Figure 2B). Therefore, in order to test its putative inhibitory profile, aortic rings were pre‐incubated with d‐pen (0.01–0.1 mM) and were challenged with l‐cys (0.1 μM–1 mM). On the basis of a preliminary set of experiments, the optimal time of pretreatment was determined as 15 min (Supporting Information Fig. S4). We observed that d‐pen significantly inhibited l‐cys‐induced relaxation in a concentration‐dependent fashion (Figure 2C). PAG, tested as a comparative control, produced a similar inhibitory effect, although at concentrations higher than those tested for d‐pen (1–10 mM) (Figure 2D).
Effect of d‐pen on H2S biosynthesis and on rCSE or rCBS
Because d‐pen inhibited l‐cys‐induced relaxation, we investigated whether this effect was related to suppression of H2S biosynthesis. Therefore, we performed an in vitro assay by using homogenated aorta samples as an enzyme source. The assay was run in the presence of different concentration of d‐pen (0.01–1 mM), and H2S production was assessed. As shown in Figure 3, following incubation with the substrate l‐cys, H2S biosynthesis was significantly reduced by d‐pen in a concentration‐dependent fashion (Figure 3A). In a parallel experiment, PAG, a selective CSE inhibitor, was used as a positive control (Figure 3A). The inhibition observed with d‐pen was achieved at concentrations lower than those of PAG (IC50 0.044 mM and 1.7 mM, for d‐pen and PAG, respectively). Because the inhibitory effect of d‐pen was achieved in homogenated samples of whole aortas, we could not distinguish whether d‐pen blocked CSE, CBS or both enzymes. In order to further elucidate this point, we used recombinant CSE and CBS (rCSE and rCBS) (Asimakopoulou et al., 2013), and we tested the effect of d‐pen on H2S biosynthesis in this cell‐free system. d‐Pen significantly inhibited both enzymes in a concentration‐dependent manner (Figure 3B). However, more interestingly, the concentration of d‐pen needed to inhibit CSE (range of 0.01–1 mM) was lower than that required to block CBS (range of 1–10 mM). Therefore, because the IC50 of d‐pen against CSE is 0.27 mM, and the IC50 for CBS is 8.5 mM, d‐pen is about 30 times more selective for CSE.
Figure 3.
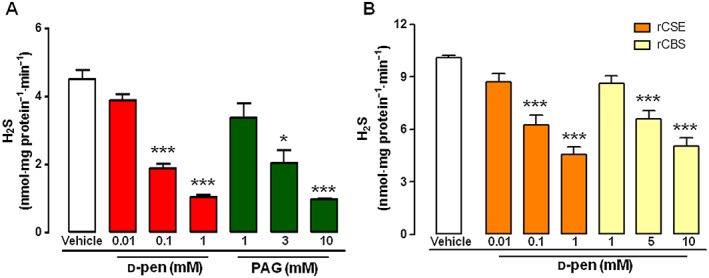
Effect of d‐pen on enzymatic H2S biosynthesis in homogenated mouse aorta samples and recombinant (r) CSE and CBS. (A) H2S production in homogenated aorta samples following l‐cys (1 mM) stimulus in the presence of increasing concentration of d‐pen (0.01–1 mM) or vehicle. Propargylglycine (PAG, 1–10 mM) has been used as a control for inhibition of H2S biosynthesis (n = 6). (B) H2S production in rCSE and rCBS following l‐cys stimulus in the presence of increasing concentrations of d‐pen (0.01–1 mM for rCSE, 1–10 mM for rCBS) or vehicle (n = 6). * Indicates significant difference (P < 0.05) for comparison with vehicle.
Inhibitory effect of d‐pen is PLP‐dependent
Because d‐pen inhibited l‐cys‐induced vasodilatation and H2S biosynthesis, we investigated the mechanism by which interfers with the H2S pathway. Based on the fact that CBS and CSE enzymatic activity is PLP‐dependent and several inhibitors inactivate this cofactor, we investigated the involvement of PLP in the inhibitory effect of d‐pen. In order to verify this hypothesis, we added PLP to the organ bath and performed concentration‐response curves to l‐cys in the presence of d‐pen (0.1 mM). As shown in Figure 4, the vasodilatation induced by l‐cys and blocked by the addition of d‐pen was significantly restored by the addition of PLP (0.3 mM) (Figure 4A). In order to further confirm this finding, we applied the PLP supplementation approach to the H2S biosynthesis experiment. In this setting, homogenated samples of aorta were used as an enzyme source and PLP was added in the presence of d‐pen. Increasing the concentration of PLP significantly blunted the d‐pen effect in a concentration‐dependent manner (Figure 4B).
Figure 4.
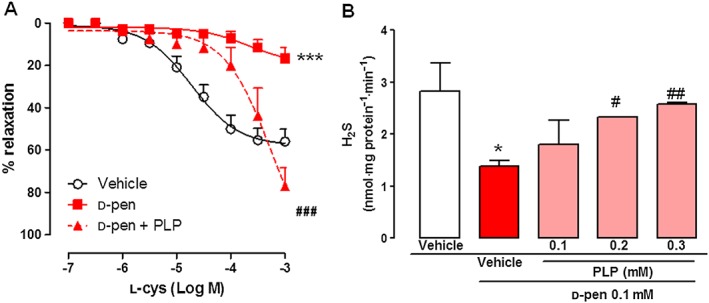
Effect of the addition of pyridoxal‐5′‐phosphate (PLP) on the inhibitory effect of d‐pen in mouse aorta. (A) l‐Cys concentration‐response curve in isolated aortic rings in the presence of d‐pen alone (0.1 mM, 15 min) or d‐pen in combination with PLP (0.3 mM, 15 min, n = 6). * and # indicate significant differences (P < 0.05) for comparisons with vehicle and d‐pen, respectively. (B) H2S production following stimulation with l‐cys in homogenated aorta samples in the presence of d‐pen alone (0.1 mM) or d‐pen in combination with increasing PLP concentrations (0.1–0.3 mM) or vehicle (n = 6). * and # indicate significant difference (P < 0.05) for comparisons with vehicle and d‐pen, respectively.
Inhibitory effect of d‐pen in an in vivo model of inflammation
So far, all these findings indicated that d‐pen acts as an inhibitor of H2S biosynthesis. H2S has been shown to have anti‐inflammatory effects that have been demonstrated in different settings (Zanardo et al., 2006; Brancaleone et al., 2014). Thus, inhibiting the synthesis of H2S should have a detrimental effect on an inflammatory response. In order to further confirm that the inhibitory effect d‐pen on H2S biosynthesis also occurred in vivo, we tested its effect on a mouse model of inflammation, TNFα‐induced mesenteric inflammation followed by IVM analysis. Results from the IVM analysis demonstrated that administration of d‐pen significantly exacerbated the TNFα‐induced vascular inflammation. In particular, a significant increase in the number of adherent leukocytes was observed (Figure 5A). Similarly, IVM analysis also showed that d‐pen significantly augmented the transmigration of leukocytes (Figure 5B); this effect was also observed following the administration of PAG (Figure 5).
Figure 5.

Effect of d‐pen on TNFα (500 ng per mouse, i.p., 2 h)‐induced mouse mesenteric inflammation. (A) d‐Pen (30 mg·kg−1, oral gavage 1 h before TNFα injection) negatively modulated levels of adherent leukocytes (n = 5). * Indicates significant difference (P < 0.05) for comparison with vehicle. (B) d‐Pen (30 mg·kg−1, oral gavage 1 h before TNFα injection) exacerbated vascular inflammation measured as number of emigrated leukocytes (n = 5). * Indicates significant difference (P < 0.05) for comparison with vehicle. PAG (10 mg·kg−1, i.p. 30 min before TNFα injection) was used as a control.
Discussion and conclusions
In this work, we have investigated whether d‐pen, used as an anti‐rheumatic drug in the late 1970's and still present in British National Formulary, can interfere with the H2S biosynthetic machinery. We formulated this hypothesis based on the structural similarity that penicillamine shares with cysteine, the physiological precursor for the generation of H2S. The key role of H2S plays in physiopathology has been widely established; however, there is a need for novel inhibitors as the present compounds, such as PAG, have been found to interfer with other unrelated pathways (Rej, 1977; Burnett et al., 1980; Papapetropoulos et al., 2015).
Firstly, we tested the ability of d‐pen to induce vasodilatation, and found it had almost no relaxant effect in vitro (~16%). d‐Pen, used in place of l‐cys in the biosynthesis assay, up to a concentration of 20 mM, did not trigger any H2S production. These findings clearly demonstrated that d‐pen is unable to metabolize and produce H2S in vitro. Indeed, this finding led us to check whether the free ‐SH moiety could somehow release H2S in a non‐enzymatic manner, as described for other molecules with free thiol groups (Bucci et al., 2014). The amperometric analysis, which evaluates the spontaneous generation of H2S, demonstrated that d‐pen did not induce the release of appreciable amounts of H2S and the weak release observed, in the presence of the nucleophilic agent, could be ascribed to contaminant traces of polysulfides, thus being irrelevant to our study. Conversely, d‐pen at 0.1 mM completely blocked l‐cys‐induced vasodilatation. Therefore, d‐pen acts as an inhibitor at a concentration 10 times lower than that needed to directly cause the weak relaxing effect observed on aortic rings. In order to further characterize the d‐pen inhibitory profile, we tested its potential as an inhibitor in an H2S biosynthesis assay by using homogenated aorta samples as an enzyme source. d‐Pen significantly inhibited l‐cys‐stimulated H2S production, thus indicating that its inhibitory effect observed in vitro in isolated aortas involves the modulation of H2S‐synthesizing enzymes. In order to further determine whether this inhibitory effect of d‐pen was associated with the inhibition of CSE and/or CBS, we tested its effect in a cell‐free assay by using recombinant enzymes and measuring H2S production in this setting. We found that d‐pen, also in this case, reduced H2S biosynthesis in a concentration‐dependent manner. More interestingly, we also observed that d‐pen was about 30 times more selective for CSE than for CBS. Indeed, the IC50 of d‐pen for rCSE was calculated to be 0.271 mM.
One of the mechanisms involved in CSE and CBS inhibition mediated by commonly used inhibitors involves binding to PLP, which is an essential cofactor for CBS and CSE. Indeed, PAG, the most selective and widely used CSE inhibitor, forms a bridge with PLP and binds to Tyr114, thereby blocking the catalytic activity of CSE (Sun et al., 2009). Therefore, we investigated whether the inhibitory effect of d‐pen might depend on its ability to interfer with the function of PLP, rather than on a direct interaction with the enzyme. To this purpose, we performed experiments aimed at evaluating the effect of d‐pen in the presence of PLP, and we found that the inhibitory effect of d‐pen was reversed by the addition of an excess of PLP. It should be noted that the addition of PLP normally only has an effect if its original concentration is lower than its physiological level (Elsey et al., 2010; Mikami et al., 2013). The additional PLP used in our study was kept within 0.1–0.3 mM, a concentration range shown to have no non‐specific effects (Stipanuk and Beck, 1982; Li et al., 2005; Asimakopoulou et al., 2013; DeRatt et al., 2014). Taken together, the results from the tissue and biochemical experiments indicate that d‐pen exerts its effect on the activity of CSE and synthesis of H2S by interfering with PLP.
However, our in vitro results do not necessarily reflect what would be the outcome in vivo following the administration of d‐pen. Indeed, this non‐conventional aminoacid has been reported to reach only low micromolar concentrations in plasma despite the high doses administered to humans (Muijsers et al., 1984; Joyce and Day, 1990). This discrepancy might be explained by the fact that d‐pen quickly forms disulfides, and can be stored as a different species. For instance, the formation of d‐pen disulfide conjugated with plasma albumin or cysteine has been well documented and may be responsible for the drastic reduction in the bioavailability of d‐pen (Nakaike et al., 1983). This aspect needs to be considered with respect to the clinical relevance of d‐pen treatment, as its blood concentration may fluctuate depending on several kinetic factors and, thus, affect the clinical outcome. However, it is noteworthy to underline that the high potency of d‐pen in inhibiting CSE demonstrated here might overcome this issue. Indeed, the plasma concentration of free d‐pen could be enough to achieve a reliable inhibitory effect on CSE and, in turn, a relevant clinical effect. Furthermore, the formation of d‐pen disulfides, although reducing free compound levels, could enable d‐pen to be available for a longer time within the body. Based on these observations, we decided to test whether d‐pen was also effective as an inhibitor of H2S biosynthesis in vivo. In the current literature it has been reported that H2S has an anti‐inflammatory role in diverse preclinical experimental settings (Brancaleone et al., 2014) (reviewed in (Wallace et al., 2015)). To this purpose, we tested the effect of d‐pen on mesenteric inflammation triggered by TNFα by using IVM analysis (Zanardo et al., 2006; Brancaleone et al., 2014). In contrast to the effects described for H2S donors (Sidhapuriwala et al., 2007; Jain et al., 2010; Ekundi‐Valentim et al., 2013; Wallace et al., 2015), the oral treatment with d‐pen increased the trafficking of leukocytes triggered by TNFα, exacerbating the inflammatory response and, thus, confirming it also has an inhibitory action on H2S biosynthesis in vivo. It is important to note that the role of H2S in inflammation is still questionable. In particular, despite its well known anti‐inflammatory properties, H2S clearly has a detrimental effect in RA patients, where its levels have been found to be higher than those detected in healthy volunteers (Whiteman et al., 2010; Kloesch et al., 2011; Muniraj et al., 2014). Therefore, if we consider this clinical setting and we bear in mind the inhibitory effect of d‐pen on H2S production, it is not difficult to speculate that such a mechanism could account for the beneficial effects observed following its administration in arthritic patients.
In conclusion, we demonstrated that the d isomer of penicillamine is a selective inhibitor of CSE, as it reduced H2S biosynthesis in a PLP‐sensitive fashion both in vitro and in vivo (Figure 6). In all the in vitro assays performed d‐pen was about 30 times more potent than PAG, the most widely used CSE inhibitor. Finally, d‐pen could represent a useful scaffold for the development and design of novel and more selective inhibitors of H2S based on the cysteine structure.
Figure 6.
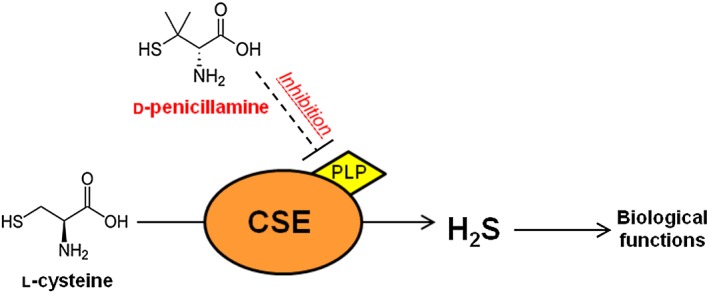
Schematic summary of the effects of d‐pen on the H2S pathway. The plain arrow (→) indicates physiological pathway for H2S biosynthesis from l‐cysteine. The dashed line (‐‐‐‐) indicates inhibitory effect of d‐pen on CSE, occurring in a PLP‐dependent manner.
Author contributions
V.B. designed and performed the in vivo experiments, analysed the data and wrote the manuscript; I.E. performed the biochemical assays and in vitro experiments; A.G performed the in vitro animal experiments; V.V. contributed to the in vitro experiments; A.A. performed the recombinant enzyme experiments; V.C. did the in vitro analytical measurements; V.C. analysed the in vitro measurements and provided intellectual contributions to the manuscript; T.G. performed the in vivo animal experiments; M.P. helped to prepare the manuscript; A.P. analysed the recombinant enzyme data and provided intellectual support in preparing the manuscript; M.B. provided intellectual contributions and helped to prepare the manuscript; G.C. supervised all the experiments, revised, critically the intellectual contributions to the manuscript and gave final approval to the publication.
Conflict of interest
The authors declare no conflicts of interest.
Declaration of transparency and scientific rigour
This Declaration acknowledges that this paper adheres to the principles for transparent reporting and scientific rigour of preclinical research recommended by funding agencies, publishers and other organizations engaged with supporting research.
Supporting information
Figure S1 Concentration‐response curves for l‐pen, l‐cysteine (l‐cys) and the vehicle in aorta rings contracted with phenylephrine (1uM, n = 6). * indicate significant differences (P < 0.05) for comparisons with vehicle.
Figure S2 (a) Calibration curve for H2S non‐enzymatic release measured as known concentration of H2S vs current measured in pAmps (pH 4) (r 2 = 0.966). Data are expressed as mean±SEM. (b) Representative graph showing increase in current (pAmps) induced by H2S release vs time (pH 4). In addition, the effect of zinc acetate [1% w/v Zn(AcO)2] addition on H2S non‐enzymatic release is also displayed. The graph shows that H2S‐induced current decreased as soon as Zn(AcO)2 was added to the reaction mixture.
Figure S3 Amperometric measurement vs time of cell free H2S release by d‐pen (1 mM) or l‐pen (1 mM) in aqueous buffer vehicle (n = 6). The assay has been run in presence of l‐cysteine, used as nucleophilic agent. ° indicates significant difference (P < 0.05) for comparisons with l‐pen. * indicates significant difference (P < 0.05) for comparisons with assay run in absence of l‐cysteine.
Figure S4 Effect of D‐pen 100 μM on l‐cys induced vasodilation in isolated aorta rings at different time of incubation (5, 15, 30 min). D‐pen was already effective after 5 min pre‐incubation, however maximum effect was achieved after 15 min. Pre‐incubation for 30 min did not result in any significant effect on l‐cys‐induced vasodilation. * indicates significant difference (P < 0.05) for comparison with vehicle.
Supporting info item
Acknowledgements
This work was supported by Italian Ministry of University and Research (MIUR, PRIN‐2012), by the European Union (European Social Fund – ESF), by Greek national funds through the Operational Program ‘Education and Lifelong Learning’ of the National Strategic Reference Framework (NSRF)‐Research Funding Program: Aristeia 2011 (1436) to AP, and by the COST Action BM1005 (ENOG: European Network on Gasotransmitters). This research was also funded by Regione Campania under POR Campania FESR 2007‐2013‐O.O.2.1 (FarmaBioNet).
Brancaleone, V. , Esposito, I. , Gargiulo, A. , Vellecco, V. , Asimakopoulou, A. , Citi, V. , Calderone, V. , Gobbetti, T. , Perretti, M. , Papapetropoulos, A. , Bucci, M. , and Cirino, G. (2016) d‐Penicillamine modulates hydrogen sulfide (H2S) pathway through selective inhibition of cystathionine‐γ‐lyase. British Journal of Pharmacology, 173: 1556–1565. doi: 10.1111/bph.13459.
References
- Alexander SPH, Fabbro D, Kelly E, Marrion N, Peters JA, Benson HE, et al. (2015). The Concise Guide to PHARMACOLOGY 2015/16: Enzymes. Br J Pharmacol 172: 6024–6109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asimakopoulou A, Panopoulos P, Chasapis CT, Coletta C, Zhou Z, Cirino G, et al. (2013). Selectivity of commonly used pharmacological inhibitors for cystathionine beta synthase (CBS) and cystathionine gamma lyase (CSE). Br J Pharmacol 169: 922–932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brancaleone V, Mitidieri E, Flower RJ, Cirino G, Perretti M (2014). Annexin A1 mediates hydrogen sulfide properties in the control of inflammation. J Pharmacol Exp Ther 351: 96–104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bucci M, Vellecco V, Cantalupo A, Brancaleone V, Zhou Z, Evangelista S, et al. (2014). Hydrogen sulfide accounts for the peripheral vascular effects of zofenopril independently of ACE inhibition. Cardiovasc Res 102: 138–147. [DOI] [PubMed] [Google Scholar]
- Burnett G, Marcotte P, Walsh C (1980). Mechanism‐based inactivation of pig heart l‐alanine transaminase by l‐propargylglycine. Half‐site reactivity. J Biol Chem 255: 3487–3491. [PubMed] [Google Scholar]
- Calvert JW, Elston M, Nicholson CK, Gundewar S, Jha S, Elrod JW, et al. (2010). Genetic and pharmacologic hydrogen sulfide therapy attenuates ischemia‐induced heart failure in mice. Circulation 122: 11–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collin M, Anuar FB, Murch O, Bhatia M, Moore PK, Thiemermann C (2005). Inhibition of endogenous hydrogen sulfide formation reduces the organ injury caused by endotoxemia. Br J Pharmacol 146: 498–505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curtis MJ, Bond RA, Spina D, Ahluwalia A, Alexander SP, Giembycz MA, et al. (2015). Experimental design and analysis and their reporting: new guidance for publication in BJP. Br J Pharmacol 172: 3461–3471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeRatt BN, Ralat MA, Kabil O, Chi YY, Banerjee R, Gregory JF 3rd (2014). Vitamin B‐6 restriction reduces the production of hydrogen sulfide and its biomarkers by the transsulfuration pathway in cultured human hepatoma cells. J Nutr 144: 1501–1508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dixon AJ, Davies J, Dormandy TL, Hamilton EB, Holt PJ, Mason RM, et al. (1975). Synthetic d(‐)penicillamine in rheumatoid arthritis. Double‐blind controlled study of a high and low dosage regimen. Ann Rheum Dis 34: 416–421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ekundi‐Valentim E, Mesquita FP, Santos KT, de Paula MA, Florenzano J, Zanoni CI, et al. (2013). A comparative study on the anti‐inflammatory effects of single oral doses of naproxen and its hydrogen sulfide (H2S)‐releasing derivative ATB‐346 in rats with carrageenan‐induced synovitis. Med Gas Res 3: 24–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elsey DJ, Fowkes RC, Baxter GF (2010). l‐cysteine stimulates hydrogen sulfide synthesis in myocardium associated with attenuation of ischemia‐reperfusion injury. J Cardiovasc Pharmacol Ther 15: 53–59. [DOI] [PubMed] [Google Scholar]
- Gavins FN, Yona S, Kamal AM, Flower RJ, Perretti M (2003). Leukocyte antiadhesive actions of annexin 1: ALXR‐ and FPR‐related anti‐inflammatory mechanisms. Blood 101: 4140–4147. [DOI] [PubMed] [Google Scholar]
- Jaffe IA (1964). Rheumatoid arthritis with arteritis; report of a case treated with penicillamine. Ann Intern Med 61: 556–563. [DOI] [PubMed] [Google Scholar]
- Jain SK, Bull R, Rains JL, Bass PF, Levine SN, Reddy S, et al. (2010). Low levels of hydrogen sulfide in the blood of diabetes patients and streptozotocin‐treated rats causes vascular inflammation? Antioxid Redox Signal 12: 1333–1337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joyce DA, Day RO (1990). d‐penicillamine and d‐penicillamine‐protein disulphide in plasma and synovial fluid of patients with rheumatoid arthritis. Br J Clin Pharmacol 30: 511–517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kilkenny C, Browne W, Cuthill IC, Emerson M, Altman DG (2010). Animal research: reporting in vivo experiments: the ARRIVE guidelines. Br J Pharmacol 160: 1577–1579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kloesch B, Liszt M, Krehan D, Broell J, Kiener H, Steiner G (2011). High concentrations of hydrogen sulphide elevate the expression of a series of pro‐inflammatory genes in fibroblast‐like synoviocytes derived from rheumatoid and osteoarthritis patients. Immunol Lett 141: 197–203. [DOI] [PubMed] [Google Scholar]
- Li L, Bhatia M, Zhu YZ, Zhu YC, Ramnath RD, Wang ZJ, et al. (2005). Hydrogen sulfide is a novel mediator of lipopolysaccharide‐induced inflammation in the mouse. FASEB J 19: 1196–1198. [DOI] [PubMed] [Google Scholar]
- Martelli A, Testai L, Citi V, Marino A, Bellagambi FG, Ghimenti S, et al. (2014). Pharmacological characterization of the vascular effects of aryl isothiocyanates: is hydrogen sulfide the real player? Vascul Pharmacol 60: 32–41. [DOI] [PubMed] [Google Scholar]
- McGrath JC, Lilley E (2015). Implementing guidelines on reporting research using animals (ARRIVE etc.): new requirements for publication in BJP. Br J Pharmacol 172: 3189–3193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mikami Y, Shibuya N, Ogasawara Y, Kimura H (2013). Hydrogen sulfide is produced by cystathionine gamma‐lyase at the steady‐state low intracellular Ca(2+) concentrations. Biochem Biophys Res Commun 431: 131–135. [DOI] [PubMed] [Google Scholar]
- Muijsers AO, van de Stadt RJ, Henrichs AM, Ament HJ, van der Korst JK (1984). d‐penicillamine in patients with rheumatoid arthritis. Serum levels, pharmacokinetic aspects, and correlation with clinical course and side effects. Arthritis Rheum 27: 1362–1369. [DOI] [PubMed] [Google Scholar]
- Muniraj N, Stamp LK, Badiei A, Hegde A, Cameron V, Bhatia M (2014). Hydrogen sulfide acts as a pro‐inflammatory mediator in rheumatic disease. Int J Rheum Dis. doi: 10.1111/1756-185X.12472 [DOI] [PubMed] [Google Scholar]
- Nakaike S, Tanaka C, Tomita N, Umemoto M, Otomo S, Aihara H (1983). Immunopharmacologic studies of d‐penicillamine‐l‐cysteine disulfide. Jpn J Pharmacol 33: 983–990. [DOI] [PubMed] [Google Scholar]
- Ochs RS, Harris RA (1980). Aminooxyacetate inhibits gluconeogenesis by isolated chicken hepatocytes. Biochim Biophys Acta 632: 260–269. [DOI] [PubMed] [Google Scholar]
- Papapetropoulos A, Whiteman M, Cirino G (2015). Pharmacological tools for hydrogen sulphide research: a brief, introductory guide for beginners. Br J Pharmacol 172: 1633–1637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pawson AJ, Sharman JL, Benson HE, Faccenda E, Alexander SP, Buneman OP, et al. (2014). The IUPHAR/BPS guide to PHARMACOLOGY: an expert‐driven knowledge base of drug targets and their ligands. Nucleic Acids Res 42: D1098–D1106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Predmore BL, Lefer DJ (2011). Hydrogen sulfide‐mediated myocardial pre‐ and post‐conditioning. Expert Rev Clin Pharmacol 4: 83–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rej R (1977). Aminooxyacetate is not an adequate differential inhibitor of aspartate aminotransferase isoenzymes. Clin Chem 23: 1508–1509. [PubMed] [Google Scholar]
- Ressler C, Nelson J, Pfeffer M (1964). A pyridoxal‐beta‐cyanoalanine relation in the rat. Nature 203: 1286–1287. [DOI] [PubMed] [Google Scholar]
- Shibuya N, Mikami Y, Kimura Y, Nagahara N, Kimura H (2009). Vascular endothelium expresses 3‐mercaptopyruvate sulfurtransferase and produces hydrogen sulfide. J Biochem 146: 623–626. [DOI] [PubMed] [Google Scholar]
- Sidhapuriwala J, Li L, Sparatore A, Bhatia M, Moore PK (2007). Effect of S‐diclofenac, a novel hydrogen sulfide releasing derivative, on carrageenan‐induced hindpaw oedema formation in the rat. Eur J Pharmacol 569: 149–154. [DOI] [PubMed] [Google Scholar]
- Stipanuk MH (2004). Sulfur amino acid metabolism: pathways for production and removal of homocysteine and cysteine. Annu Rev Nutr 24: 539–577. [DOI] [PubMed] [Google Scholar]
- Stipanuk MH, Beck PW (1982). Characterization of the enzymic capacity for cysteine desulphhydration in liver and kidney of the rat. Biochem J 206: 267–277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun Q, Collins R, Huang S, Holmberg‐Schiavone L, Anand GS, Tan CH, et al. (2009). Structural basis for the inhibition mechanism of human cystathionine gamma‐lyase, an enzyme responsible for the production of H(2)S. J Biol Chem 284: 3076–3085. [DOI] [PubMed] [Google Scholar]
- Wallace JL (2012). Hydrogen sulfide: a rescue molecule for mucosal defence and repair. Dig Dis Sci 57: 1432–1434. [DOI] [PubMed] [Google Scholar]
- Wallace JL, Ianaro A, Flannigan KL, Cirino G (2015). Gaseous mediators in resolution of inflammation. Semin Immunol 27: 227–233. [DOI] [PubMed] [Google Scholar]
- Wang R (2011). Signaling pathways for the vascular effects of hydrogen sulfide. Curr Opin Nephrol Hypertens 20: 107–112. [DOI] [PubMed] [Google Scholar]
- Whiteman M, Haigh R, Tarr JM, Gooding KM, Shore AC, Winyard PG (2010). Detection of hydrogen sulfide in plasma and knee‐joint synovial fluid from rheumatoid arthritis patients: relation to clinical and laboratory measures of inflammation. Ann N Y Acad Sci 1203: 146–150. [DOI] [PubMed] [Google Scholar]
- Whiteman M, Le Trionnaire S, Chopra M, Fox B, Whatmore J (2011). Emerging role of hydrogen sulfide in health and disease: critical appraisal of biomarkers and pharmacological tools. Clin Sci (Lond) 121: 459–488. [DOI] [PubMed] [Google Scholar]
- Yang G, Wu L, Jiang B, Yang W, Qi J, Cao K, et al (2008). H2S as a physiologic vasorelaxant: hypertension in mice with deletion of cystathionine gamma‐lyase. Science 322: 587–590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zanardo RC, Brancaleone V, Distrutti E, Fiorucci S, Cirino G, Wallace JL (2006). Hydrogen sulfide is an endogenous modulator of leukocyte‐mediated inflammation. FASEB J 20: 2118–2120. [DOI] [PubMed] [Google Scholar]
- Zhang HL, Zhi L, Moore PK, Bhatia M (2006). Role of hydrogen sulfide in cecal ligation and puncture‐induced sepsis in the mouse. Am J Physiol Lung Cell Mol Physiol 290: L1193–L1201. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1 Concentration‐response curves for l‐pen, l‐cysteine (l‐cys) and the vehicle in aorta rings contracted with phenylephrine (1uM, n = 6). * indicate significant differences (P < 0.05) for comparisons with vehicle.
Figure S2 (a) Calibration curve for H2S non‐enzymatic release measured as known concentration of H2S vs current measured in pAmps (pH 4) (r 2 = 0.966). Data are expressed as mean±SEM. (b) Representative graph showing increase in current (pAmps) induced by H2S release vs time (pH 4). In addition, the effect of zinc acetate [1% w/v Zn(AcO)2] addition on H2S non‐enzymatic release is also displayed. The graph shows that H2S‐induced current decreased as soon as Zn(AcO)2 was added to the reaction mixture.
Figure S3 Amperometric measurement vs time of cell free H2S release by d‐pen (1 mM) or l‐pen (1 mM) in aqueous buffer vehicle (n = 6). The assay has been run in presence of l‐cysteine, used as nucleophilic agent. ° indicates significant difference (P < 0.05) for comparisons with l‐pen. * indicates significant difference (P < 0.05) for comparisons with assay run in absence of l‐cysteine.
Figure S4 Effect of D‐pen 100 μM on l‐cys induced vasodilation in isolated aorta rings at different time of incubation (5, 15, 30 min). D‐pen was already effective after 5 min pre‐incubation, however maximum effect was achieved after 15 min. Pre‐incubation for 30 min did not result in any significant effect on l‐cys‐induced vasodilation. * indicates significant difference (P < 0.05) for comparison with vehicle.
Supporting info item


