Abstract
Electrophysiological and neurochemical studies implicate cholinergic signaling in the basolateral amygdala (BLA) in behaviors related to stress. Both animal studies and human clinical trials suggest that drugs that alter nicotinic acetylcholine receptor (nAChR) activity can affect behaviors related to mood and anxiety. Clinical studies also suggest that abnormalities in cholinergic signaling are associated with major depressive disorder, whereas pre-clinical studies have implicated both β2 subunit-containing (β2*) and α7 nAChRs in the effects of nicotine in models of anxiety- and depression-like behaviors. We therefore investigated whether nAChR signaling in the amygdala contributes to stress-mediated behaviors in mice. Local infusion of the non-competitive non-selective nAChR antagonist mecamylamine or viral-mediated downregulation of the β2 or α7 nAChR subunit in the amygdala all induced robust anxiolytic- and antidepressant-like effects in several mouse behavioral models. Further, whereas α7 nAChR subunit knockdown was somewhat more effective at decreasing anxiety-like behavior, only β2 subunit knockdown decreased resilience to social defeat stress and c-fos immunoreactivity in the BLA. In contrast, α7, but not β2, subunit knockdown effectively reversed the effect of increased ACh signaling in a mouse model of depression. These results suggest that signaling through β2* nAChRs is essential for baseline excitability of the BLA, and a decrease in signaling through β2 nAChRs alters anxiety- and depression-like behaviors even in unstressed animals. In contrast, stimulation of α7 nAChRs by acetylcholine may mediate the increased depression-like behaviors observed during the hypercholinergic state observed in depressed individuals.
INTRODUCTION
Major depressive disorder affects hundreds of millions of individuals worldwide, but its etiology is not fully elucidated because it is highly heterogeneous. Although there are available treatments, there is still a large proportion of patients that do not respond satisfactorily to traditional antidepressant therapies (Khan et al, 2012). Many etiological factors could underlie depressive disorders, including alterations in monoaminergic function, but several studies have implicated dysregulation of the cholinergic system in depression in human subjects (Janowsky et al, 1972; Risch et al, 1981). Recent human SPECT imaging studies show that depressed unipolar and bipolar subjects have increased levels of ACh throughout the brain (Saricicek et al, 2012; Hannestad et al, 2013). In humans, blocking the degradation of ACh can induce depressive symptoms even in individuals with no history of illness (Janowsky et al, 1972; Risch et al, 1981), whereas in mice, blocking ACh breakdown in the hippocampus alone is sufficient to induce depression- and anxiety-like behaviors (Mineur et al, 2013). Finally, activity of the medial habenula-IPN pathway (a region rich in α3β4 nAChRs; Gotti et al, 2006) is also critical for the control of anxiety induced by nicotine withdrawal (Zhao-Shea et al, 2015) and contributes to the aversive properties of self-administered nicotine through nAChRs containing the α5 subunit (Fowler et al, 2011).
The cholinergic system has been a recent focus for development of novel antidepressant medications (Drevets et al, 2013; Mineur and Picciotto, 2010; Picciotto et al, 2012; Saricicek et al, 2012). Acetylcholine signals through muscarinic and nicotinic acetylcholine receptors (mAChRs and nAChRs, respectively) and many studies of depression have focused on mAChR signaling (Cannon et al, 2011; Riemann et al, 1994); however, blockade of either mAChRs or nAChRs can reverse the depression-like effects of increased cholinergic signaling in mice (Mineur et al, 2013) and it is likely that multiple receptors can mediate the effects of cholinergic signaling on depression (Drevets et al, 2013). The recent failure of a large clinical trial (Vieta et al, 2013) suggests that global antagonism of nAChRs will not be a viable strategy for antidepressant development; however, small human (Philip et al, 2009), and a number of rodent studies (Mineur et al, 2009; Rollema et al, 2009) suggest that targeting specific nAChR subtypes may be a potential strategy for treating depression. Numerous studies suggest that nAChRs are important for cellular and behavioral responses to stress, and that modulation of corticolimbic circuits by nAChRs may be particularly critical (Mansvelder et al, 2009). Thus, studies identifying the locus for nAChR signaling could be important for unraveling the mechanisms underlying cholinergic control of stress-related behaviors.
Nicotinic antagonists at both β2* and α7 nAChRs can have antidepressant-like effects in mice (Andreasen et al, 2012, 2013; Mineur et al, 2009; Rollema et al, 2009). In addition, nAChR subtypes containing the α5, α2, α3, and β4 subunits have all been implicated in the aversive properties of nicotine, symptoms related to nicotine withdrawal and behaviors related to anxiety and depression (Fowler et al, 2011; Frahm et al, 2011; Salas et al, 2003; 2004; Upton and Lotfipour, 2015). The non-competitive, non-selective antagonist mecamylamine and the partial agonist cytisine are both effective in mouse models of antidepressant efficacy and converge to decrease neuronal activity in the basolateral amygdala (BLA) (Mineur et al, 2007), a brain region that is often hyperactivated in depressed patients (Drevets et al, 2008). In addition, nicotine can facilitate glutamatergic signaling in the amygdala through activity of α7 nAChRs (Jiang and Role, 2008) and these nAChRs are particularly critical for synaptic plasticity in this brain region (Jiang et al, 2013). Although many nAChR subtypes may contribute to anxiety- and depression-like behaviors, based on the critical role of the amygdala in emotional behaviors, the current study investigated whether altering cholinergic signaling through β2* or α7 nAChRs in the amygdala could affect neuronal activity and performance in behaviors related to stress and depression. We also investigated whether limiting the activity of these nAChRs in the amygdala was sufficient to reverse the effects of increasing cholinergic activity in a mouse model of depression to identify a causal link between amygdala cholinergic signaling and stress-induced behaviors.
MATERIALS AND METHODS
Animals
Male C57BL/6J mice (10–12-week-old upon arrival) were purchased from Jackson Laboratories (Bar Harbor, ME, USA) and were maintained in a temperature-controlled vivarium (21±2 °C) under a 12 h light–dark cycle with lights on at 7:00 AM. Unless separated for humane concerns or due to cannulation for local infusion, animals were housed five per cage. Food and water were available ad libitum. Ten to 12 animals were initially assigned to each treatment group. A summary of the different experimental groups and the experimental design is outlined in Table 1. All procedures were approved by the Yale University Animal Care and Use Committee.
Table 1. Experimental Groups and Design.
| Figure | Manipulation | Treatment | Regimen | Assays performed | Other assays |
|---|---|---|---|---|---|
| 1 | Amygdala infusion | Mecamylamine (1 ng @ 2 μg/ml) | Acute (30 min before test) | Tail suspension | |
| 2, 3 | β2 amygdala knockdown | Chronic (~5 weeks before test) | Light/dark, tail suspension, forced swim, social defeat | c-fos, locomotion | |
| α7 amygdala knockdown | |||||
| 4 | Amygdala knockdown (β2 or α7) | Mecamylamine (1 mg/kg) | Acute (30 min before each test) | Tail suspension, social defeat | Locomotion |
| 5 | Amygdala knockdown (β2 or α7) | Physostigmine (0.15 mg/kg) | Acute (60 min before test) | Tail suspension, forced swim | Locomotion |
Drugs
All drugs were purchased from Sigma-Aldrich. For i.p. injections, mecamylamine was dissolved in phosphate-buffered saline (saline; 1 mM KH2PO4, 155 mM NaCl, 3 mM Na2HPO4, pH=7.4) at 0.1 mg/ml and physostigmine was dissolved at 0.15 mg/ml. Solutions were injected i.p. at 10 ml/kg of body weight unless otherwise described.
Cannulation and Local Infusion
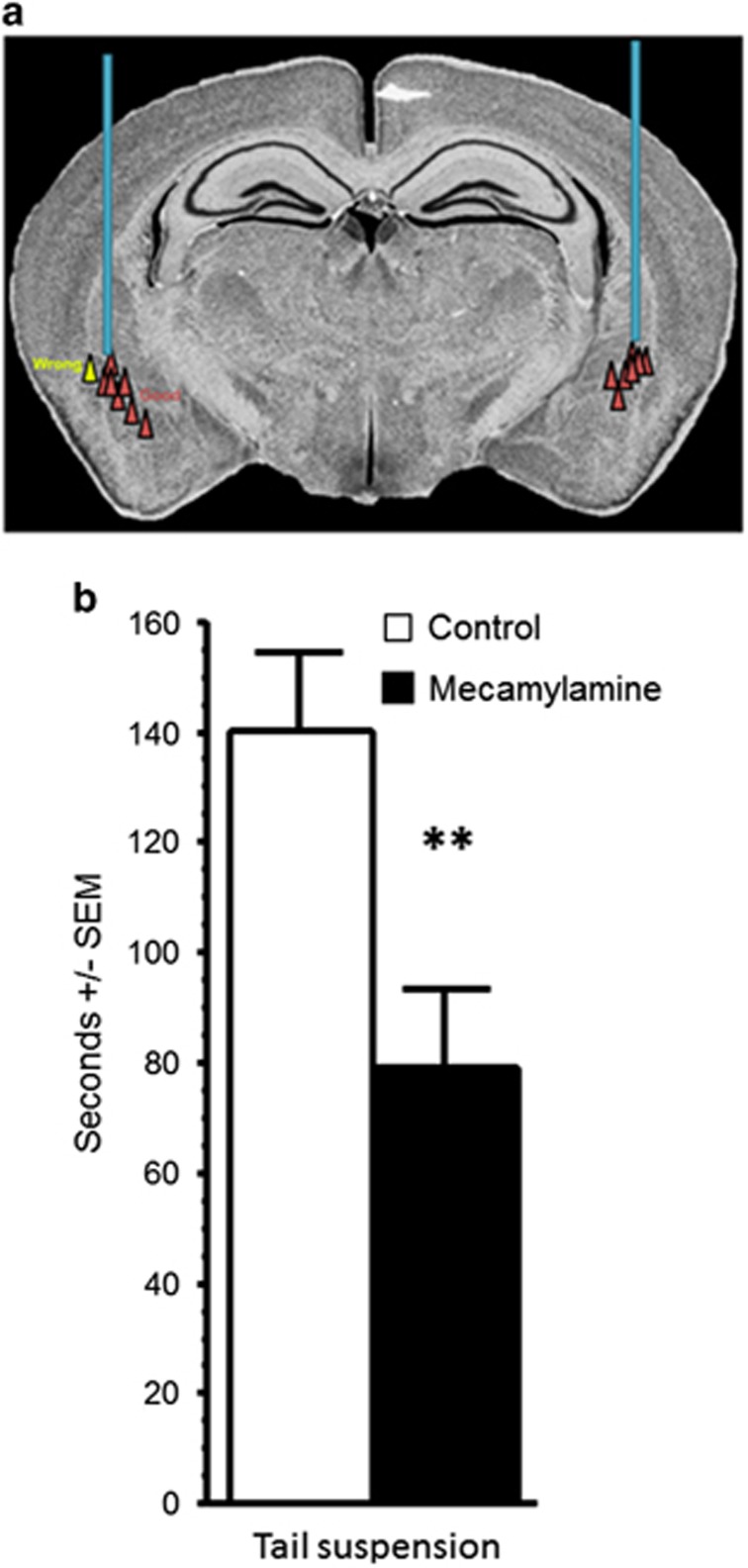
Mecamylamine was infused into the amygdala at a concentration of 2 μg/ml, though bi-lateral cannulae. Placement was checked by injecting blue dye through the cannula (Figure 1a) and subjects with off-target placements were not analyzed further. Knockdown was verified in vivo by equilibrium binding (see Supplementary Materials and Supplementary Figure S1).
Figure 1.
Mecamylamine infusion into the amygdala has an antidepressant-like effect. (a) Placement of all on-target infusions into the amygdala. The yellow triangle represents an example of an off-target infusion. (b) Time spent immobile in the tail suspension test following acute infusion of mecamylamine. Data are expressed as means ±SEM. **p<0.01.
AAV-shRNA Infusions into the Amygdala
Details of design, purification, and validation of viral vectors can be found in the Supplementary Materials. Infusion of viral vectors into the amygdala was performed with a stereotaxic frame mounted with 5 μl Hamilton syringes under 2% Isoflurane anesthesia. In total, 0.5 μl per side of a mix of the different AAV-shRNA for each gene was infused into the amygdala with the following coordinates (from Bregma): anterior/posterior=–1.8 mm; lateral=±3.4 mm; dorsal/ventral=−5.2 mm. The infusion rate was~0.1 μl/min and the needle was then left in place for an additional 5 min to allow for diffusion. Control animals received a scrambled shRNA (Scr), ie, a sequence that does not have a target in the mouse genome/transcriptome. Infusion sites were assessed by detection of GFP in brain slices from paraformaldehyde (PFA)-perfused animals (see immunohistochemistry for details). Subjects with off-target cannula placement (0–3 per group) were removed from further analyses.
Behavioral Testing
Behavioral tests were conducted in sequence, with 24–48 h between each assay, as previously described (Mineur et al, 2013) and based on published recommendations. For local infusion experiments, only the tail suspension test was performed as the protruding cannula was incompatible with other behavioral tests. In knockdown experiments combining shRNA infusion with injections of mecamylamine or physostigmine, mice were tested in the tail suspension test and the social defeat paradigm only. This choice was made to avoid repeated exposure to these pharmacological agents that could lead to adaptation. Animals that only received AAV-shRNA infusion were tested in sequence in the light–dark exploration box, the tail suspension test, the forced swim test, the social defeat paradigm, and a final evaluation of locomotor activity. Each group of nAChR knockdown animals (KD) was tested side-by-side with an equivalent control group (Scr) that underwent surgery at the same time to avoid any between-experiment differences that might bias outcomes. Data are expressed as percent control within each group to normalize the data across experiments.
Light–dark exploration box
A rectangular box was split evenly into two compartments: one covered and the other open and illuminated by a 60 W light. Mice were placed into the farthest point of the light side and the time taken to cross to the dark compartment was recorded. After the first crossing, time in the dark side and the number of crossings was recorded over a 6 min period.
Tail suspension test
Mice were suspended gently by the tail for a period of 6 min, behavior was videotaped, and the number of bouts and time spent immobile were scored manually.
Forced swim test
Each mouse was placed in a 4-l beaker of room temperature water for 15 min. For experiments with physostigmine, a second forced swim test was performed 24 h later for 5 min to limit confounds due to hypothermia. Behavior was videotaped and time spent immobile was scored manually.
Social defeat paradigm
CD1 mice were selected for aggressive behavior as described previously (Mineur et al, 2013, 2015). On the initial defeat day, each C57BL/6J mouse was placed into a CD1 aggressor mouse home cage. The time taken until the first fight was recorded and the two mice were then separated using a metal mesh, with the CD1 mouse receiving two thirds of the home cage. After 10 min, the C57BL/6J mouse was returned to its home cage and the metal mesh was removed. This process occurred twice a day for 3 days, with the C57BL/6J mouse exposed to a different CD1 aggressor mouse each day. On the fourth day, social interaction was assessed. Initially, each tested mouse was placed in the open field and allowed to roam freely. The time spent in the area surrounding the holding square was recorded for the first 2.5 min, after which a CD1 mouse was placed inside this square. The time spent in the area around the holding square was then recorded for another 2.5 min. Data are expressed a ratio of time spent in the area around the holding square with or without the CD1 mouse.
Locomotor activity
Locomotor activity was measured once the battery of tests was completed in a 48 cm × 22 cm × 18 cm plastic cage for 30 min. None of the treatments evaluated induced a significant change in locomotion compared with animals from the comparable control group.
C-fos Immunohistochemistry
Following a 15-min forced swim stress, mice were irreversibly anesthetized with pentobarbital (50 mg/kg) and perfused intracardially with 4% PFA in PBS. Brains were post-fixed in PFA overnight, cryoprotected in 30% PBS/sucrose solution and sliced at 40 μm with a sliding microtome. Immunostaining was conducted on one in six amygdala slices (4–6 slices per animal) for 6–8 animals per treatment group. Tissue was pre-incubated with 0.3% Triton/1% normal goat serum/PBS (pH=7.4) for 30 min and was then incubated with the same solution combined to a pre-conjugated anti-c-fos antibody (1 : 1000; Santa Cruz SC-52) for 48 h at 4 °C. Positively identified cells were counted within a defined area (expressed in pixels) of the BLA, and number of counted cell was then normalized to area. For each brain, both the left and right amygdala was sampled and an average per amygdala was taken of these two values for statistical analysis.
Statistical Analysis
Results of behavioral and c-fos assays were analyzed by ANOVA with ‘drug' and/or ‘virus' when relevant, as between-factors. Post hoc analyses were then performed by t-test with Bonferroni corrections. Significance was set at p<0.05.
RESULTS
Amygdala Infusion of the Non-Competitive, Non-Selective Antagonist Mecamylamine has an Antidepressant-Like Effect in the Tail Suspension Test
An increase in cholinergic tone can be observed in depressed patients (Hannestad et al, 2013; Saricicek et al, 2012). In contrast, treatment with the non-competitive non-selective antagonist mecamylamine can have an antidepressant-like effect in mice, which is correlated with a decrease in amygdala activity (Mineur et al, 2007). Thus, we first determined whether decreasing cholinergic signaling through nAChRs in the amygdala with mecamylamine could recapitulate the antidepressant-like effect observed when this drug is administered systemically. Mice infused with mecamylamine showed a significant decrease in immobility in the tail suspension test, suggesting that limiting nicotinic transmission in the amygdala is sufficient to induce an antidepressant-like effect (Figure 1b; F(1,17)=7.97, p=0.01). Because mecamylamine is a non-competitive non-selective antagonist, we then investigated whether specific nAChR subtypes contribute to this change in behavior.
Downregulation of α7 and β2* nAChRs in the Amygdala has Anxiolytic- and Antidepressant-Like Effects
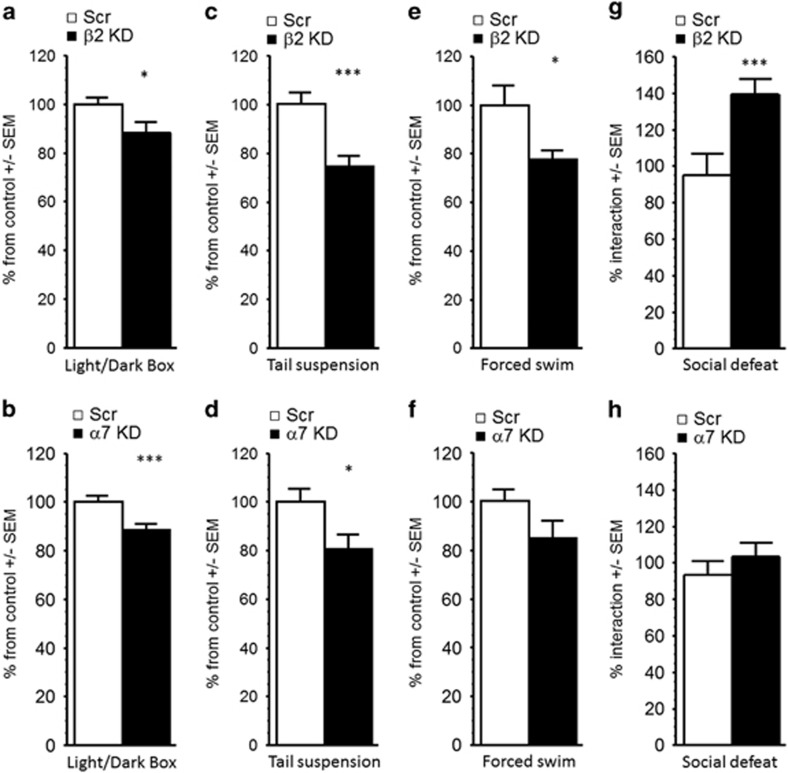
To determine the consequences of altering nicotinic signaling through specific nAChR subtypes in tests of antidepressant and anxiolytic efficacy and in response to social stress, AAV-shRNAs targeting either the α7 or the β2 nAChR subunit were infused into the amygdala of C57BL6/J mice. In the light–dark box, knockdown of either the α7 or the β2 subunit decreased the time spent in the dark compartment, suggesting an anxiolytic-like effect mediated by the blockade of either nAChR subtype, although the effect of α7 knockdown was somewhat more pronounced (F(1, 19)=4.26, p=0.04 and F(1, 18)=10.51, p=0.004, respectively; Figure 2a and b). None of the treatments had a significant effect on the overall number of transitions between chambers (Fs<1); Supplementary Figure S2A and B). In the tail suspension test, shRNAs targeting either the α7 or the β2 nAChR subunit decreased the time spent immobile (F(1, 18)=6.71, p=0.02 and F(1, 19)=16.03, p=0.001; Figure 2c and d), suggesting an antidepressant-like effect; a similar pattern was also observed in the forced swim test in mice following β2 subunit knockdown in the amygdala (F(1, 18)=5.59, p=0.03; Figure 2e), but there was only a non-significant trend following α7 knockdown in the amygdala (F<1; Figure 2f). In the social defeat paradigm, β2 subunit knockdown resulted in a significant increase in social interaction compared with control mice (F(1, 21)=10.33, p=0.004 Figure 2g), whereas α7 knockdown did not alter the interaction score (F<1; Figure 2h). These results suggest that both α7 and β2* nAChRs in the amygdala contribute to behavior in tests of anxiolytic and antidepressant efficacy in the absence of stress, but only blockade of β2 signaling in the amygdala increases resilience to social stress.
Figure 2.
Effects of β2 and α7 nAChR subunit knockdown in the amygdala in tests of anxiety- and depression-like phenotypes. Time spent in the dark side of the light/dark box following (a) β2 or (b) α7 nAChR subunit knockdown in the amygdala. Time spent immobile in the tail suspension test following (c) β2 or (d) α7 nAChR subunit knockdown in the amygdala. Time spent immobile in the forced swim test following (e) β2 or (f) α7 nAChR subunit knockdown in the amygdala. Time spent interacting with a CD1 mouse after 4 days of social defeat following (g) β2 or (h) α7 nAChR subunit knockdown in the amygdala. Data are expressed as % means ±SEM normalized to each control group. *p<0.05; ***p<0.001.
Downregulation of β2*, but not α7, nAChRs in the Amygdala, Significantly Decreases C-Fos Expression
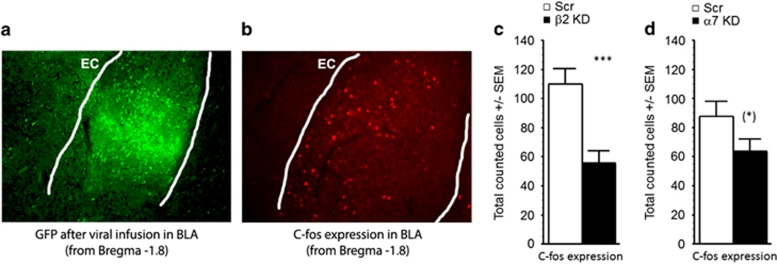
Mice were challenged with 15 min of forced swim and were killed 90 min later. After checking for GFP expression to ensure proper targeting of the BLA (Figure 3a), GFP-positive brain slices were processed for c-fos immunoreactivity, a marker of neuronal activation (Figure 3b). Following β2 subunit knockdown in the amygdala, there was a highly significant ~50% decrease in c-fos expression (F(1, 30)=13.92, p=0.0008); Figure 3c), similar to what had been described previously following systemic injection of mecamylamine (Mineur et al, 2007). Conversely, knocking down the α7 subunit had little effect on c-fos immunoreactivity (F<1; Figure 3d), suggesting that blockade of β2* nAChRs in the amygdala was critical for regulation of overall activity of BLA neurons.
Figure 3.
Immunohistochemistry for GFP and c-fos in the amygdala after β2 or α7 knockdown. (a) Representative photomicrograph of GFP expression in the BLA following viral infusion. (b) Representative photomicrograph of c-fos immunohistochemistry in the amygdala following viral infusion. EC, external capsule. (c, d) Total number of c-fos immunoreactive cells in the amygdala following (c) β2 or (d) α7 nAChR subunit knockdown. Data are expressed as means ±SEM. *p<0.06; ***p<0.001.
Knockdown of the α7 or β2 nAChR Subunit in the Amygdala Occludes the Effect of Mecamylamine in the Tail Suspension Test
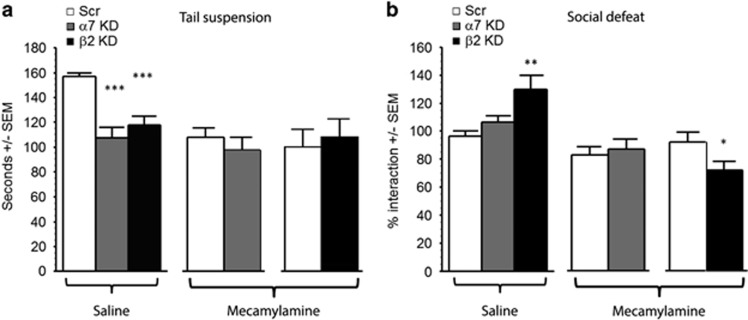
To determine whether the effects of mecamylamine were mediated through activity at α7 or β2* nAChRs in the amygdala, and to eliminate the possibility of off-target effects (including, among others, at NMDA receptors (Gonzales et al, 1993; O'Dell and Christensen, 1988)), we infused shRNAs targeting each subunit into the amygdala and challenged the animals with mecamylamine or vehicle. Following knockdown of either the α7 or the β2 subunit in the amygdala, we observed the expected antidepressant-like effect at baseline (ANOVA: F(2, 26)=17.91, p<0.0001; post hoc: Scr vs α7 KD: p<0.001; Scr vsβ2 KD, p=0.0001; Figure 4a, left). Following mecamylamine injection, control mice displayed an antidepressant-like response in the tail suspension test, and this effect was occluded in mice that received shRNAs targeting the α7 (Figure 4a, middle) or the β2 subunit (Figure 4a, right; all Fs<1) in the amygdala. Mice infused with shRNAs targeting the α7 subunit showed no change in social behavior, whereas β2 subunit knockdown in the amydala significantly increased interaction after social defeat (ANOVA: F(2, 26)=6.58, p=0.004; post hoc: Scr vs α7 KD: p=0.29; Scr vsβ2 KD, p=0.001; Figure 4b, left). Unexpectedly, the administration of mecamylamine modestly, but significantly, decreased social interaction in mice following knockdown of the β2 nAChR subunit in the amygdala (F(1, 25)=4.77, p=0.04; Figure 4b, middle), whereas there was no effect of mecamylamine following infusion of the Scr or knockdown of the α7 subunit in the amygdala (F<1); Figure 4b, right). Thus, a depressant-like effect of mecamylamine in the social defeat paradigm is unmasked following knockdown of the β2 subunit in the amygdala. These data suggest that mecamylamine can promote stress-induced social avoidance through a mechanism independent of β2* or α7 nAChR signaling in the amygdala.
Figure 4.
α7 and β2* nAChRs in the amygdala mediate the antidepressant-like effects of a nicotinic antagonist. Effect of acute mecamylamine injection on (a) the time spent immobile in the tail suspension test and (b) time spent interacting with a CD1 mouse in the social defeat test following α7 or β2 nAChR subunit knockdown in the amygdala. Data are expressed as means ±SEM. *p<0.05; **p<0.01; ***p<0.001.
Systemic, but not Local, Blockade of Acetylcholine Esterase Increases Depression-Like Behavior and this is Partially Reversed by α7 or β2 nAChR Knockdown in the Amygdala
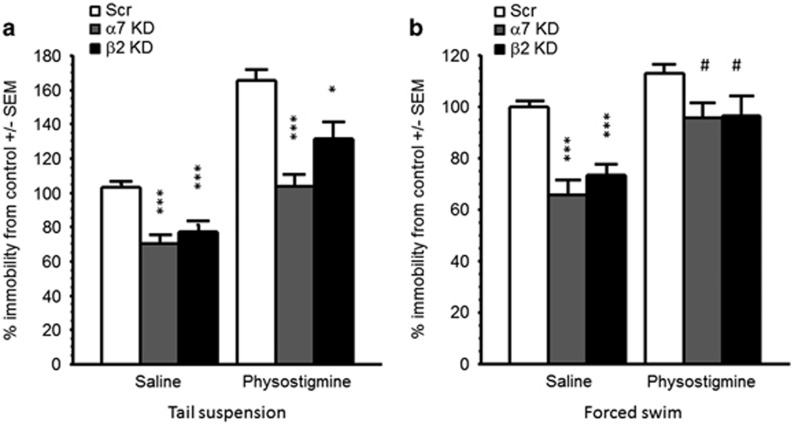
Blocking or knocking down nAChRs in the amygdala has antidepressant-like effects, so we then determined whether increasing ACh levels by infusing shRNAs targeting acetylcholinesterase (AChE: the main enzyme degrading ACh) into the amygdala could have the opposite effect in tests of antidepressant efficacy. Contrary to what was observed when AChE was knocked down in the hippocampus (Mineur et al, 2013), knockdown in the amygdala had no effect on behavior in the tail suspension test (control vs AChE knockdown: 175 ±24.75 vs 195 ±25.24 s, F<1), nor did it affect c-fos expression (control vs AChE knockdown: 110 ±34.4 vs 87 ±37.4 counted cells per section, F<1). We then determined whether knockdown of the α7 or β2 nAChR subunit in the amygdala could reverse the effect of systemic antagonism of AChE activity by physostigmine. ANOVAs reveal an overall physostigmine and knockdown effect (F(1, 63)=66.38, p<0.0001 and F(2, 63)=22.46, p<0.0001, respectively), but no interaction (F<1), suggesting that the nAChR knockdown had a similar effect on immobility in the tail suspension test regardless of the degree of AChE activity (Figure 5a). Post hoc analyses revealed that infusion of shRNAs targeting the α7 or β2 nAChR subunit into the amygdala significantly reduced time spent immobile in the tail suspension test compared ;with saline treated, scrambled shRNA-infused controls ;(all p<0.001). A similar effect was observed in the forced swim test (Figure 5b) with an overall physostigmine (F(1, 61)=29.65, p<0.0001) and knockdown (F(1, 61)=17.59, p<0.0001) effect, but no interaction (F <1). Post hoc analyses showed that β2 and α7 subunit knockdown in the amygdala decreased immobility at baseline in the forced swim test (all p<0.0001) but only marginally decreased immobility following physostigmine administration (p=0.022). It is possible that confounding factors could have affected these results as cholinesterase inhibitors can significantly alter body temperature. Alternatively, post hoc corrections for multiple comparisons (where the p cutoff value is 0.017 in this design) could underestimate the effect. These experiments suggest that decreasing signaling through α7 or β2 nAChRs in the amygdala results in an antidepressant-like effect that can partially reverse the consequences of increased ACh levels systemically.
Figure 5.
Knockdown of α7 or β2* nAChRs in the amygdala can oppose the effects of systemic physostigmine injection. (a) Time spent immobile in the tail suspension test. (b) Time spent interacting with a CD1 mouse in the forced swim test. Data are expressed as means ±SEM. *p<0.05; ***p<0.001; #p<0.007.
DISCUSSION
Depression is a complex, multidimensional disorder, and it is likely that multiple neurotransmitter systems and brain regions contribute to its pathophysiology. This may explain why a significant proportion of depressed individuals do not show complete remission in response to existing antidepressant treatments. Thus, it remains critical to identify the mechanisms underlying the development of mood disorders.
Recent studies have suggested that individuals with uni- or bipolar illness have increased ACh levels throughout the brain when they are acutely depressed (Saricicek et al, 2012; Hannestad et al, 2013). A number of studies have also addressed the possibility that nAChRs could be involved in depression, because depressed individuals are twice as likely to smoke, have a harder time quitting, and can develop symptoms of depression during withdrawal (for reviews, see Moylan et al, 2012; Picciotto et al, 2012). Despite controversy about the connection that may exist between nAChRs and mood regulation, many studies have shown that decreasing ACh signaling through nAChRs can have antidepressant-like properties in mice (Mineur et al, 2007, 2009) and can improve symptoms of depression in some clinical studies (George et al, 2008). Although both the nicotinic antagonist mecamylamine and the low efficacy partial agonist cytisine can decrease c-fos activity in the amygdala (Mineur et al, 2007), it is not known where in the brain nAChRs are required to alter behaviors relevant to stress and depression. Many brain areas that receive significant ACh innervation, including hippocampus, prefrontal cortex, habenuala, IPN, ventral tegmental area, and nucleus accumbens/striatum (Leao et al, 2015; Mineur et al, 2013, 2015; Picciotto and Mineur, 2014; Polter and Kauer, 2014; Zhao-Shea et al, 2013, 2015) could be involved in nAChR-related mood regulation. We demonstrate here that the non-selective nAChR antagonist mecamylamine can induce antidepressant-like effects when infused locally into the amygdala, suggesting that nAChR signaling is critical for baseline neuronal activity in this brain region, and that decreasing nAChR activity in amygdala is sufficient for an antidepressant-like response.
A combination of pharmacological and molecular genetic experiments has suggested that β2* nAChRs contribute to stress-induced behaviors and may contribute to the effects of antidepressant medications effective in human patients (Caldarone et al, 2004; Harrist et al, 2004). The effect of α7 nAChRs on behaviors related to depression is more complex, however. Although the antidepressant-like effects of the non-competitive, non-selective antagonist mecamylamine are not observed in knockout mice lacking the α7 subunit (Rabenstein et al, 2006), α7 nAChR agonists can potentiate the antidepressant-like effects of a monoamine reuptake inhibitor, with no effects on their own (Andreasen et al, 2013). The current studies demonstrate that decreasing expression of either the α7 or β2 nAChR subunit in the amygdala mimics the effect of classical antidepressants in behavioral assays in mice, and that the effect of either knockdown is similar to local infusion of mecamylamine into the amygdala. It also suggests that there may be distinct effects of nAChR signaling in the amygdala that could account for specific endophenotypes related to depression as compared other with stress-induced behaviors, as social defeat was only different following β2 knockdown. Further, α7 and β2* nAChRs may be expressed in distinct subcellular compartments, cell types or subregions of the amygdala, resulting in differing transduction of ACh signaling in this brain region. This idea is supported by studies of c-fos immunoreactivity performed here, showing that knockdown of the β2 nAChR subunit significantly decreased neuronal activity in the BLA, whereas α7 subunit knockdown had much less effect on c-fos expression.
The results presented here are consistent with the role of β2* nAChRs described in the VTA, in which blockade decreases the reactivity of dopaminergic neurons to a broad range of stimulation (Koranda et al, 2014). Conversely, these studies suggest that the role of α7 nAChR signaling in stress-related behaviors is specific to the endophenotype assessed. α7 nAChRs are highly expressed in BLA and have a critical role in neuronal excitability (Almeida-Suhett et al, 2014; Jiang and Role, 2008; Klein and Yakel, 2006). Because the α7 subunit is expressed in both glutamatergic and GABAergic neurons in the amygdala and can modulate both excitatory and inhibitory neurons (Pidoplichko et al, 2013), decreasing the activity of this nAChR subtype could have opposite behavioral consequences depending on whether the nucleus is highly active or quiescent (Pandya and Yakel, 2013). It has been suggested that the primary role of α7 nAChRs in BLA is to enhance inhibitory synaptic transmission (Pidoplichko et al, 2013), but several studies have demonstrated that signaling through α7 nAChRs can increase excitatory transmission (Barazangi and Role, 2001; Jiang and Role, 2008; Klein and Yakel, 2006). Along these lines, regulation of excitability of the BLA by α7 nAChRs has been associated with increased anxiety-like behavior in a model of traumatic brain injury (Almeida-Suhett et al, 2014).
These observations suggest that the net effect of altering α7 signaling is highly dependent on the physiological status of the amygdala but do not explain why knockdown of AChE in the amygdala has no effect on behavior in the tail suspension test or on c-fos expression. Thus, fluctuations in levels of ACh in amygdala may not be the primary mediator of stress-related changes in behavior. Rather, ACh acting through nAChRs at baseline could be required for normal amygdala function, and decreasing cholinergic activity in this structure could be protective against changes in neurotransmitter systems at other brain sites in response to stress, including changes in α7-regulated glutamatergic projections (Yang et al, 2013). This would be consistent with electrophysiological studies demonstrating that cholinergic neurons of the nucleus basalis, a major source of ACh to the amygdala, have a high firing rate when animals are awake, leading to a high level of baseline cholinergic signaling in the amygdala even in the absence of a stressor (Whalen et al, 1994). This is also consistent with the observation that inescapable stress increases extracellular ACh levels in the hippocampus as measured by microdialysis, but has little effect on ACh levels in the amygdala (Mark et al, 1996).
Signaling through the α7 and β2* nAChR subtypes in the amygdala is critical for stress-related behaviors, but there are likely differences in the neurobiological substrates underlying the effects of each nAChR subtype. In a model of depression induced by systemic physostigmine administration, knockdown of the β2 subunit was not as effective as α7 subunit knockdown, suggesting that α7 has a permissive role in the amygdala that may amplify the effects of stress-induced glutamate release, similar to what has been reported in other brain regions. In conclusion, dysregulation of nAChR activity in amygdala may alter susceptibility to stress and contribute to maladaptive behaviors.
Funding and disclosure
The authors declare no conflict of interest.
Acknowledgments
This work was supported by NIH grants DA033945 (ORWH, NIDA, FDA), MH077681, and MH105824 from the National Institute on Drug Abuse (NIDA), the Food and Drug Administration (FDA), and the Office of Research on Women's Health (ORWH).
Footnotes
Supplementary Information accompanies the paper on the Neuropsychopharmacology website (http://www.nature.com/npp)
Supplementary Material
References
- Almeida-Suhett CP, Prager EM, Pidoplichko V, Figueiredo TH, Marini AM, Li Z et al (2014). Reduced GABAergic inhibition in the basolateral amygdala and the development of anxiety-like behaviors after mild traumatic brain injury. PLoS One 9: e102627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andreasen JT, Redrobe JP, Nielsen EO (2012). Combined alpha7 nicotinic acetylcholine receptor agonism and partial serotonin transporter inhibition produce antidepressant-like effects in the mouse forced swim and tail suspension tests: a comparison of SSR180711 and PNU-282987. Pharmacol Biochem Behav 100: 624–629. [DOI] [PubMed] [Google Scholar]
- Andreasen JT, Redrobe JP, Nielsen EO, Christensen JK, Olsen GM, Peters D (2013). A combined alpha7 nicotinic acetylcholine receptor agonist and monoamine reuptake inhibitor, NS9775, represents a novel profile with potential benefits in emotional and cognitive disturbances. Neuropharmacology 73: 183–191. [DOI] [PubMed] [Google Scholar]
- Barazangi N, Role LW (2001). Nicotine-induced enhancement of glutamatergic and GABAergic synaptic transmission in the mouse amygdala. J Neurophysiol 86: 463–474. [DOI] [PubMed] [Google Scholar]
- Caldarone BJ, Harrist A, Cleary MA, Beech RD, King SL, Picciotto MR (2004). High-affinity nicotinic acetylcholine receptors are required for antidepressant effects of amitriptyline on behavior and hippocampal cell proliferation. Biol Psychiatry 56: 657–664. [DOI] [PubMed] [Google Scholar]
- Cannon DM, Klaver JK, Gandhi SK, Solorio G, Peck SA, Erickson K et al (2011). Genetic variation in cholinergic muscarinic-2 receptor gene modulates M2 receptor binding in vivo and accounts for reduced binding in bipolar disorder. Mol Psychiatry 16: 407–418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drevets WC, Price JL, Furey ML (2008). Brain structural and functional abnormalities in mood disorders: implications for neurocircuitry models of depression. Brain Struct Funct 213: 93–118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drevets WC, Zarate CA Jr, Furey ML (2013). Antidepressant effects of the muscarinic cholinergic receptor antagonist scopolamine: a review. Biol Psychiatry 73: 1156–1163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fowler CD, Lu Q, Johnson PM, Marks MJ, Kenny PJ (2011). Habenular alpha5 nicotinic receptor subunit signalling controls nicotine intake. Nature 471: 597–601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frahm S, Slimak MA, Ferrarese L, Santos-Torres J, Antolin-Fontes B, Auer S et al (2011). Aversion to nicotine is regulated by the balanced activity of beta4 and alpha5 nicotinic receptor subunits in the medial habenula. Neuron 70: 522–535. [DOI] [PubMed] [Google Scholar]
- George TP, Sacco KA, Vessicchio JC, Weinberger AH, Shytle RD (2008). nicotinic antagonist augmentation of selective serotonin reuptake inhibitor-refractory major depressive disorder: a preliminary study. J Clin Psychopharmacol 28: 340–344. [DOI] [PubMed] [Google Scholar]
- Gonzales RA, Roper LC, Westbrook SL (1993). Cholinergic modulation of N-methyl-D-aspartate-evoked [3H]norepinephrine release from rat cortical slices. J Pharmacol Exp Ther 264: 282–288. [PubMed] [Google Scholar]
- Gotti C, Zoli M, Clementi F (2006). Brain nicotinic acetylcholine receptors: native subtypes and their relevance. Trends Pharmacol Sci 27: 482–491. [DOI] [PubMed] [Google Scholar]
- Hannestad JO, Cosgrove KP, DellaGioia NF, Perkins E, Bois F, Bhagwagar Z et al (2013). Changes in the cholinergic system between bipolar depression and euthymia as measured with [123I]5IA single photon emission computed tomography. Biol Psychiatry 74: 768–776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrist A, Beech RD, King SL, Zanardi A, Cleary MA, Caldarone BJ et al (2004). Alteration of hippocampal cell proliferation in mice lacking the beta 2 subunit of the neuronal nicotinic acetylcholine receptor. Synapse 54: 200–206. [DOI] [PubMed] [Google Scholar]
- Janowsky DS, el-Yousef MK, Davis JM, Sekerke HJ (1972). A cholinergic-adrenergic hypothesis of mania and depression. Lancet 2: 632–635. [DOI] [PubMed] [Google Scholar]
- Jiang L, Emmetsberger J, Talmage DA, Role LW (2013). Type III neuregulin 1 is required for multiple forms of excitatory synaptic plasticity of mouse cortico-amygdala circuits. J Neurosci 33: 9655–9666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang L, Role LW (2008). Facilitation of cortico-amygdala synapses by nicotine: activity-dependent modulation of glutamatergic transmission. J Neurophysiol 99: 1988–1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan A, Faucett J, Lichtenberg P, Kirsch I, Brown WA (2012). A systematic review of comparative efficacy of treatments and controls for depression. PLoS One 7: e41778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klein RC, Yakel JL (2006). Functional somato-dendritic alpha7-containing nicotinic acetylcholine receptors in the rat basolateral amygdala complex. J Physiol 576(Pt 3): 865–872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koranda JL, Cone JJ, McGehee DS, Roitman MF, Beeler JA, Zhuang X (2014). Nicotinic receptors regulate the dynamic range of dopamine release in vivo. J Neurophysiol 111: 103–111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leao RM, Cruz FC, Vendruscolo LF, de Guglielmo G, Logrip ML, Planeta CS et al (2015). Chronic nicotine activates stress/reward-related brain regions and facilitates the transition to compulsive alcohol drinking. J Neurosci 35: 6241–6253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mansvelder HD, Mertz M, Role LW (2009). Nicotinic modulation of synaptic transmission and plasticity in cortico-limbic circuits. Semin Cell Dev B iol 20: 432–440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mark GP, Rada PV, Shors TJ (1996). Inescapable stress enhances extracellular acetylcholine in the rat hippocampus and prefrontal cortex but not the nucleus accumbens or amygdala. Neuroscience 74: 767–774. [DOI] [PubMed] [Google Scholar]
- Mineur YS, Eibl C, Young G, Kochevar C, Papke RL, Gundisch D et al (2009). Cytisine-based nicotinic partial agonists as novel antidepressant compounds. J Pharmacol Exp Ther 329: 377–386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mineur YS, Einstein EB, Bentham MP, Wigestrand MB, Blakeman S, Newbold SA et al (2015). Expression of the 5-HT1A serotonin receptor in the hippocampus is required for social stress resilience and the antidepressant-like effects induced by the nicotinic partial agonist cytisine. Neuropsychopharmacology 40: 938–946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mineur YS, Obayemi A, Wigestrand MB, Fote GM, Calarco CA, Li AM et al (2013). Cholinergic signaling in the hippocampus regulates social stress resilience and anxiety- and depression-like behavior. Proc Natl Acad Sci USA 110: 3573–3578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mineur YS, Picciotto MR (2010). Nicotine receptors and depression: revisiting and revising the cholinergic hypothesis. Trends Pharmacol Sci 31: 580–586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mineur YS, Somenzi O, Picciotto MR (2007). Cytisine, a partial agonist of high-affinity nicotinic acetylcholine receptors, has antidepressant-like properties in male C57BL/6J mice. Neuropharmacology 52: 1256–1262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moylan S, Jacka FN, Pasco JA, Berk M (2012). Cigarette smoking, nicotine dependence and anxiety disorders: a systematic review of population-based, epidemiological studies. BMC Med 10: 123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Dell TJ, Christensen BN (1988). Mecamylamine is a selective non-competitive antagonist of N-methyl-D-aspartate- and aspartate-induced currents in horizontal cells dissociated from the catfish retina. Neurosci Lett 94: 93–98. [DOI] [PubMed] [Google Scholar]
- Pandya AA, Yakel JL (2013). Effects of neuronal nicotinic acetylcholine receptor allosteric modulators in animal behavior studies. Biochem Pharmacol 86: 1054–1062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Philip NS, Carpenter LL, Tyrka AR, Whiteley LB, Price LH (2009). Varenicline augmentation in depressed smokers: an 8-week, open-label study. J Clin Psychiatry 70: 1026–1031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Picciotto MR, Higley MJ, Mineur YS (2012). Acetylcholine as a neuromodulator: cholinergic signaling shapes nervous system function and behavior. Neuron 76: 116–129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Picciotto MR, Mineur YS (2014). Molecules and circuits involved in nicotine addiction: the many faces of smoking. Neuropharmacology 76: 545–553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pidoplichko VI, Prager EM, Aroniadou-Anderjaska V, Braga MF (2013). alpha7-containing nicotinic acetylcholine receptors on interneurons of the basolateral amygdala and their role in the regulation of the network excitability. J Neurophysiol 110: 2358–2369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polter AM, Kauer JA (2014). Stress and VTA synapses: implications for addiction and depression. Eur J Neurosci 39: 1179–1188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rabenstein RL, Caldarone BJ, Picciotto MR (2006). The nicotinic antagonist mecamylamine has antidepressant-like effects in wild-type but not beta2- or alpha7-nicotinic acetylcholine receptor subunit knockout mice. Psychopharmacology 189: 395–401. [DOI] [PubMed] [Google Scholar]
- Riemann D, Hohagen F, Krieger S, Gann H, Muller WE, Olbrich R et al (1994). Cholinergic REM induction test: muscarinic supersensitivity underlies polysomnographic findings in both depression and schizophrenia. J Psychiatr Res 28: 195–210. [DOI] [PubMed] [Google Scholar]
- Risch SC, Cohen RM, Janowsky DS, Kalin NH, Sitaram N, Gillin JC et al (1981). Physostigmine induction of depressive symptomatology in normal human subjects. Psychiatry Res 4: 89–94. [DOI] [PubMed] [Google Scholar]
- Rollema H, Guanowsky V, Mineur YS, Shrikhande A, Coe JW, Seymour PA et al (2009). Varenicline has antidepressant-like activity in the forced swim test and augments sertraline's effect. Eur J Pharmacol 605: 114–116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salas R, Pieri F, De Biasi M (2004). Decreased signs of nicotine withdrawal in mice null for the beta4 nicotinic acetylcholine receptor subunit. J Neurosci 24: 10035–10039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salas R, Pieri F, Fung B, Dani JA, De Biasi M (2003). Altered anxiety-related responses in mutant mice lacking the beta4 subunit of the nicotinic receptor. J Neurosci 23: 6255–6263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saricicek A, Esterlis I, Maloney KH, Mineur YS, Ruf BM, Muralidharan A et al (2012). Persistent beta2*-nicotinic acetylcholinergic receptor dysfunction in major depressive disorder. Am J Psychiatry 169: 851–859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Upton M, Lotfipour S (2015). alpha2-Null mutant mice have altered levels of neuronal activity in restricted midbrain and limbic brain regions during nicotine withdrawal as demonstrated by cfos expression. Biochem Pharmacol 97: 558–565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vieta E, Thase ME, Naber D, D'Souza B, Rancans E, Lepola U et al (2013). Efficacy and tolerability of flexibly-dosed adjunct TC-5214 (dexmecamylamine) in patients with major depressive disorder and inadequate response to prior antidepressant. Eur Neuropsychopharmacol 24: 564–574. [DOI] [PubMed] [Google Scholar]
- Whalen PJ, Kapp BS, Pascoe JP (1994). Neuronal activity within the nucleus basalis and conditioned neocortical electroencephalographic activation. J Neurosci 14(3 Pt 2): 1623–1633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Y, Paspalas CD, Jin LE, Picciotto MR, Arnsten AF, Wang M (2013). Nicotinic alpha7 receptors enhance NMDA cognitive circuits in dorsolateral prefrontal cortex. Proc Natl Acad Sci USA 110: 12078–12083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao-Shea R, DeGroot SR, Liu L, Vallaster M, Pang X, Su Q et al (2015). Increased CRF signalling in a ventral tegmental area-interpeduncular nucleus-medial habenula circuit induces anxiety during nicotine withdrawal. Nat Commun 6: 6770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao-Shea R, Liu L, Pang X, Gardner PD, Tapper AR (2013). Activation of GABAergic neurons in the interpeduncular nucleus triggers physical nicotine withdrawal symptoms. Curr Biol 23: 2327–2335. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.