Abstract
Objective. To compare the clinical outcome of patients presenting with symptoms of uncomplicated cystitis who were seen by a doctor, with patients who were given treatment following a diagnostic algorithm. Design. Randomized controlled trial. Setting. Out-of-hours service, Oslo, Norway. Intervention. Women with typical symptoms of uncomplicated cystitis were included in the trial in the time period September 2010–November 2011. They were randomized into two groups. One group received standard treatment according to the diagnostic algorithm, the other group received treatment after a regular consultation by a doctor. Subjects. Women (n = 441) aged 16–55 years. Mean age in both groups 27 years. Main outcome measures. Number of days until symptomatic resolution. Results. No significant differences were found between the groups in the basic patient demographics, severity of symptoms, or percentage of urine samples with single culture growth. A median of three days until symptomatic resolution was found in both groups. By day four 79% in the algorithm group and 72% in the regular consultation group were free of symptoms (p = 0.09). The number of patients who contacted a doctor again in the follow-up period and received alternative antibiotic treatment was insignificantly higher (p = 0.08) after regular consultation than after treatment according to the diagnostic algorithm. There were no cases of severe pyelonephritis or hospital admissions during the follow-up period. Conclusion. Using a diagnostic algorithm is a safe and efficient method for treating women with symptoms of uncomplicated cystitis at an out-of-hours service. This simplification of treatment strategy can lead to a more rational use of consultation time and a stricter adherence to National Antibiotic Guidelines for a common disorder.
Keywords: After-hours care, algorithms, amdinocillin (mecillinam), general practice, Norway, primary health care, urinary tract infection
Lower urinary tract infections in women are very common and account for a significant percentage of consultations in primary health care.
A search in relevant medical literature has not revealed any previous studies assessing the use of a diagnostic algorithm as part of the treatment scheme.
Our study demonstrated that the use of a diagnostic algorithm is a safe and efficient method for identifying women eligible for treatment without seeing a doctor.
This simplification of treatment strategy can contribute to shorter waiting time for the patient, better adherence to antibiotic guidelines, and a more rational use of consultation time in outpatient clinics.
Introduction
Uncomplicated cystitis accounts for approximately 95% of all consultations related to urinary tract infections (UTIs) in primary health care [1].
Several strategies have been explored to increase adherence to antibiotic guidelines for the treatment of acute urinary tract infection with variable results [2,3]. No antibiotic treatment, symptomatic treatment by the use of NSAIDs, cutting back on treatment length, and wait-and-see prescriptions are methods that aim to decrease the overall use of antibiotics. These strategies have a combined interest in preventing further development of antibiotic resistance [4–9]. However, refraining from antibiotic treatment increases the length of arduous symptoms [10,11].
Norwegian guidelines for the treatment of symptoms of acute cystitis in women have concluded that treatment can be given without a urine dipstick test. For repeated episodes of symptoms of cystitis, treatment can be given without seeing a physician, the suggested procedure being telephone contact with the medical secretary. Women suffering from recurring cystitis should see a doctor for examination and a urine culture [12].
The out-of-hours service in Oslo uses a questionnaire to identify patients who qualify for a simplified routine of diagnosing and treating uncomplicated cystitis, the objective being a safe, time-, and cost-saving procedure.
Mecillinam is the antibiotic of choice in treating cystitis according to the diagnostic algorithm. This is a Scandinavian agent. This choice is based on epidemiological findings of Escherichia coli (E.coli) as the predominating bacterial agent [13,14]. E.coli has a relatively low rate of resistance towards mecillinam [15–17]. It is also found to have a relatively low resistance driving effect, which has caused increased focus on the possibility of use in the context of the increasing load of multi-resistant bacteria [18,19].
Acute uncomplicated cystitis is a common disorder, but current diagnostic strategies in general practice show considerable variation. Population-based before-and-after studies have proved that use of a diagnostic algorithm significantly decreases the use of urine analysis, urine culture, and office visits. At the same time it also increases the number of patients receiving recommended antibiotics. Randomized controlled trials were sought to evaluate the use of a diagnostic algorithm in identifying women with uncomplicated cystitis [20–22].
The objectives of the study were:
to compare the clinical outcome of patients presenting with symptoms of uncomplicated cystitis who were treated after a regular consultation by a doctor with patients who were given treatment following a diagnostic algorithm;
to compare safety and rate of complications in the two groups.
Material and methods
Since 2007 the out-of-hours service in Oslo has used a diagnostic algorithm to identify patients with an uncomplicated cystitis. Inclusion and exclusion criteria were chosen based on established symptoms and risk factors for complicated UTI. The study was preceded by a pilot investigation conducted for one week in June 2010, with the aim of identifying potential logistical challenges.
In the course of 14 months from September 2010–November 2011, 441 women in the age group 16–55 years were included. Patients eligible for inclusion were identified by use of a diagnostic algorithm as shown in Figure 1. Women presenting with dysuria and increased frequency of urination were included. Visible haematuria and increased urge for urination were also registered, but did not determine inclusion. Criteria for exclusion were relevant comorbidity (diabetes, kidney disease, and oesophageal passage problems), symptoms indicative of pyelonephritis or a complicated UTI, symptoms indicative of a sexually transmitted infection (STI), ongoing antibiotic/probenecid treatment, or a previous allergic reaction to penicillin. Temperature was measured and ongoing fever led to exclusion.
Figure 1.
Diagnostic algorithm.
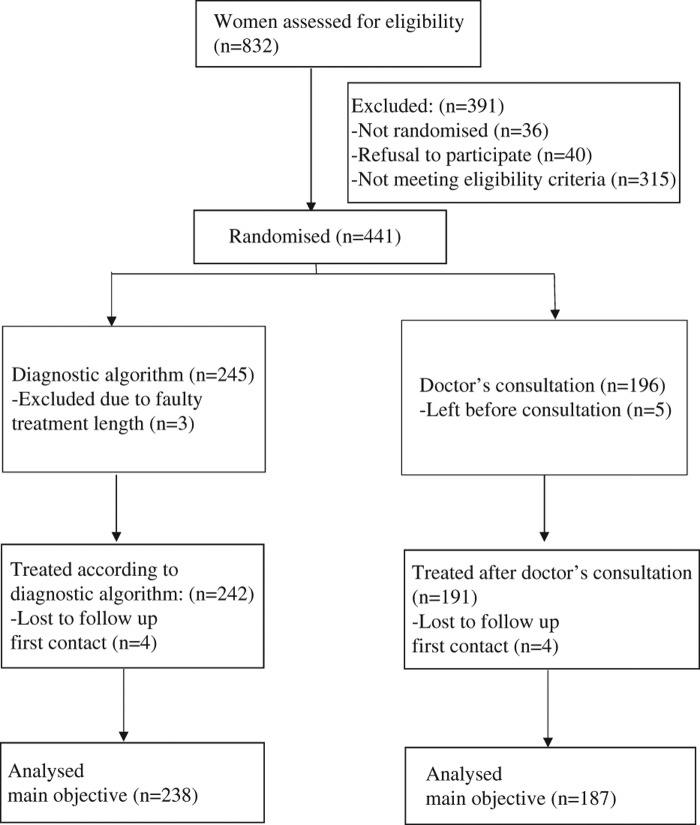
Included patients were randomized into two groups. The registering nurse completed the randomization process by drawing a number 1 or 2 from an envelope. The envelopes were generated by the study coordinator with an equal amount of numbers. We did not use block randomization. This may have contributed to the higher number of patients in one group; 242 patients were given diagnostic algorithm-based care versus 191 patients who were seen by a doctor (Figure 2).
Figure 2.
Trial flow chart: RCT of diagnostic algorithm for uncomplicated cystitis at an Out-of-hours service in Oslo, Norway.
The study group received pivmecillinam 200 mg × 3 for three days in accordance with the diagnostic algorithm. The control group was seen by a doctor who was not aware that the patient was included in the study.
Urine dipstick findings were evaluated in both groups along with a cultured urine sample taken on the day of presentation at the out-of-hours service.
In both groups the follow-up included a telephone call from the study coordinator one week after contact and two weeks after the treatment was ended. A total of 12 patients in the study group and 16 patients in the control group were lost during the follow-up. Four patients in each group were unreachable for the first phone call.
According to protocol, a urine sample was sent to the laboratory for evaluation one week after finishing treatment. The vast majority of samples were received during a time period of one to two weeks after treatment. All urine culture results were evaluated by both the study coordinator and the treating physician.
All urine samples were cultured according to established procedures for identification and resistance patterns at the Department of Medical Microbiology, at Oslo University Hospital, Ullevål. The uropathogens were quantified in colony-forming units/mL. Significant bacteriuria was defined according to current European guidelines for patients with symptoms of UTI as ≥ 103 /mL for primary pathogens, ≥ 104/mL for secondary pathogens, and ≥ 105/mL for doubtful pathogens [23] (see Table II).
Table II.
Bacteriological findings in urine samples of female patients presenting with symptoms of urinary tract infection at an out-of-hours service in Oslo, Norway.
| Diagnostic algorithm | Doctor's consultation | p-value | |
|---|---|---|---|
| Urine samples taken at consultation, n | 234 | 172 | |
| Negative culture, n (%)1 | 102 (44) | 71 (41) | NS |
| Single culture isolates, n (%)2 | 132 (56) | 101 (59) | NS |
| Primary pathogens: | |||
| Escherichia coli, n (%) | 101 (77) | 79 (78) | NS |
| Staphylococcus saprophyticus, n (%) | 21 (16) | 17 (17) | NS |
| Secondary pathogens: | |||
| Klebsiella species, n (%) | 3 (2) | 2 (2) | NS |
| Proteus mirabilis, n (%) | 4 (3) | 0 | NS |
| Enterococcus fecalis, n (%) | 1 (1) | 1 (1) | NS |
| Enterobacter species, n (%) | 0 | 1 (1) | NS |
| Doubtful pathogens: | |||
| Streptococcus agalactiae, n (%) | 2 (2) | 0 | NS |
| Pseudomonas species, n (%) | 0 | 1 (1) | NS |
| Control urine sample, n (% of total) | 169 (78) | 130 (76) | |
| Negative culture, n (%)1 | 150 (89) | 117 (90) | 0.13 |
| Single culture isolates, n (%)2 | 19 (11) | 13 (10) | NS |
Notes: Crosstabs analysis. Pearson's chi-square p-value. NS = non-significant. p < 0.15 stated. Patients who did not deliver primary or control urine samples were excluded. 1Non-significant bacteriuria, see Material and methods. 2All species-specific percentage values are calculated for the total number of single-culture isolates.
The statistical power of the data was based on symptom relief in 85% of the control group versus 75% in the group treated according to the diagnostic algorithm by day four, and a given power of 80% and a p-value of < 5% (two-sided test). Sample size calculation indicated that 250 patients in each group were needed.
SPSS18 manufactured by IBM was used for statistical analysis. Descriptive analysis of data was done. For the evaluation of the primary outcome measurement a Kaplan–Meier plot was performed.
Results
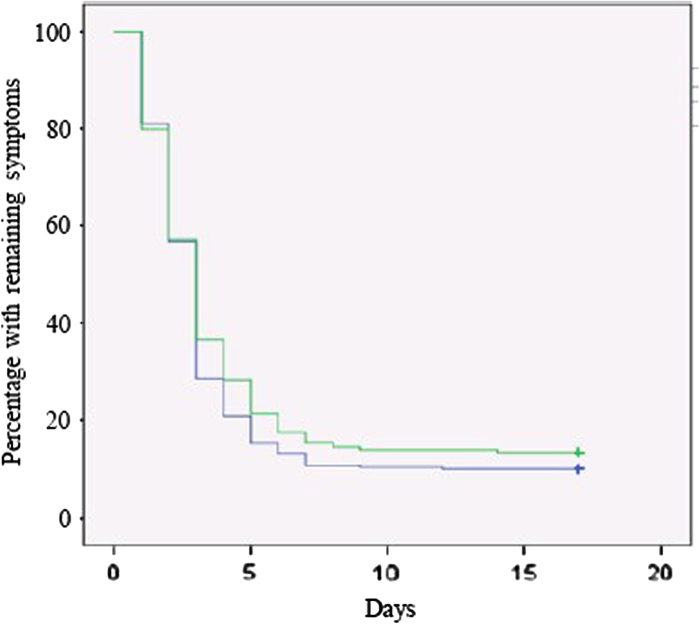
The main outcome measurement of the study was the number of days until symptomatic resolution in the two groups as shown in a Kaplan-Meier plot (Figure 3). By day four 188 of 238 (79%) in the group treated according to the diagnostic algorithm and 134 of 187 (72%) in the control group were free of symptoms (p = 0.09). A median of three days until symptom resolution was found in both groups, log rank test p = 0.3.
Figure 3.
Symptom resolution: Percentage of patients with remaining symptoms versus number of days since the start of treatment, Kaplan–Meier plot. Blue line: Treated according to the diagnostic algorithm. Green line: Treatment chosen after a doctor's consultation. P-value (log rank): 0.3.
Patient characteristics
Table I gives basic demographic data, including age and the country of origin. No significant differences between the two groups were found. Increased frequency of urination and pain on urination along with female sex and age were valued as absolute criteria of inclusion. The symptom scores did not reveal significant differences between the two groups.
Table I.
Baseline demographic and clinical characteristics of consecutive female patients presenting with symptoms of urinary tract infection at an out-of-hours service in Oslo, Norway.
| Diagnostic algorithm (n = 242) | Doctor's consult (n = 191) | p-value | |
|---|---|---|---|
| Age, mean (SD), y | 27 (8) | 27 (8) | NS |
| Country of origin | |||
| Norwegian, n, (%) | 206 (85) | 159 (83) | NS |
| Other European, n, (%) | 25 (10) | 27 (14) | NS |
| Outside Europe, n, (%) | 9 (4) | 4 (2) | NS |
| Unknown, n, (%) | 2 (1) | 1 (1) | NS |
| Patient-reported symptoms | |||
| Painful urination | |||
| Strong, n (%) | 85 (35) | 48 (25) | NS |
| Moderate, n (%) | 126 (52) | 111 (58) | NS |
| Mild, n (%) | 31 (13) | 32 (17) | NS |
| Increased frequency of urination | |||
| Strong, n (%) | 89 (37) | 64 (34) | NS |
| Moderate, n (%) | 132 (54) | 110 (58) | NS |
| Mild, n (%) | 21 (9) | 17 (9) | NS |
| Increased need to urinate, n (%) | 237 (98) | 190 (99) | 0.07 |
| Macroscopic haematuria, n (%) | 92 (38) | 78 (41) | NS |
| Urinary dipstick findings | n = 160 | n = 187 | |
| Nitrate positive, n (%) | 29 (18) | 29(16) | NS |
| Leukocyte esterase positive n (%) | 141(88) | 164(88) | NS |
Notes: Crosstabs analysis. Pearson's chi-square p-value. NS = non-significant. p < 0.15 stated.
Urine dipstick findings were evaluated; there were no significant differences in the number of patients who presented with a positive nitrate or leukocyte esterase test (see Table I). Bacteriological findings showed an insignificantly higher rate of growth of single-culture bacteria in the group seen by a doctor (Table II).
The value of the control urine samples was compromised by a group of patients who failed to deliver the samples. However, there was an insignificantly higher (p = 0.13) number of negative control urine samples in the group who had seen a doctor.
A majority of the patients included in the group seen by a doctor were diagnosed with uncomplicated cystitis and treated with mecillinam, which is the standard procedure for treatment according to the diagnostic algorithm. Of 191 patients, 186 were diagnosed with cystitis, two were diagnosed with pyelonephritis, one was diagnosed with bacterial vaginosis, two were diagnosed with cystitis and other illness (bacterial vaginosis and muscular pain), and in two patients no illness was found.
In the group seen by a doctor 80% were treated with mecillinam. Other antibiotics used were trimethoprim (13%) and nitrofurantoin (6%). Two patients (1%) were not given any antibiotic treatment.
During the follow-up period 12% of the patients treated according to the diagnostic algorithm contacted a doctor versus 18% in the group seen by a doctor (p 0.08). An alternative antibiotic regime was given to 11% in the group treated according to the diagnostic algorithm versus 16% in the group seen by a doctor.
For those who made follow-up contact significant bacteriuria was found in 19% of urine specimens in the diagnostic algorithm group and 26% in the control group. For those who did not contact a doctor again 10% significant bacteriuria was found in both groups. There were no cases of severe pyelonephritis or hospital admissions during the follow-up period.
The trial ended when the time frame for inclusion as agreed with the out-of-hours service in Oslo was reached. We managed to reach the needed sample in the algorithm group, but not in the control group. This was due to practical problems with recruitment of patients and the time frame given.
Discussion
Main findings
Our study demonstrated that an algorithm-based diagnosis of acute uncomplicated cystitis correlated well with the diagnosis given after a blinded doctor's consultation. There was no significant difference between the two groups in the number of days until clinical cure or in the number of patients who again contacted a doctor during the follow-up period. Although we did not manage to include a sufficient number of patients in the comparison group to meet our power calculation, we conclude that algorithm-based handling of patients was equivalent to an ordinary consultation. This conclusion is strengthened by the fact that we found an insignificant tendency for shorter duration of symptoms in the algorithm group.
A strict adherence to the algorithm to identify inclusion and exclusion criteria is necessary in order to ensure safe use of the algorithm. No cases of serious pyelonephritis or hospital admission were reported in the two groups.
Strengths and weaknesses
The study has been carried out in a setting with a high number of unselected patients with uncomplicated cystitis. During the follow-up, nearly all patients were successfully contacted regarding clinical cure, but a follow-up urine specimen was more often not received. All follow-up contacts were made by the same clinician.
With a mean age of 27 years in our patient population, the study is less representative of the elderly segment of included patients. In a young study population STIs are more common and it has been noted that adolescent females with symptoms of UTI could benefit from testing for both STI and UTI [24]. A Swedish study conducted in general practice found a prevalence of 4% of Chlamydia trachomatis among young women with symptoms of UTI; this is comparable to the prevalence of 3% in the general population of women in the same age group [25].
Symptoms of STIs determine exclusion in the diagnostic algorithm; however, it is possible that some patients had a co-infection or a primary STI. There were no patients in the control group diagnosed with an STI. In addition, equal symptomatic and bacteriological outcomes in the two arms make a significant number of STIs less probable.
It is noteworthy that the doctor who treated the control patients was able to consider the urine dipstick findings when choosing antibiotic treatment. One could speculate this this would result in the doctor choosing an alternative regimen to ensure an effect against non-nitrate-producing organisms. Our findings did not show such a change in treatment choice. It is possible that the treating physician did not consider the nitrate status of the urine dipstick findings.
Urine samples were spontaneous samples taken at the time of consultation, thus a number of women had not kept the urine in the bladder for at least four hours, which is regarded as necessary to demonstrate significant bacteriuria. This may explain why the studied population showed a lower percentage of significant bacteriuria than previous studies [15,26]. There was no significant difference between the groups as to proportion of significant bacteriuria in the primary urine samples.
Whether it is feasible from a resistance point of view to keep mecillinam as the treatment of choice according to the diagnostic algorithm, or whether variation among the recommended treatment regimens would be more beneficial should be considered. In Norway recommended treatment regimens include mecillinam, nitrofurantoin, and trimethoprim. Ciprofloxacin is not a recommended agent for the treatment of uncomplicated cystitis in Norway [16].
Comparison with other studies
To the best of our knowledge this is the first randomized controlled study to assess whether an algorithm-based care is equivalent to doctor-based care in treating uncomplicated cystitis.
An article referring to a telephone-based nurse evaluation and treatment algorithm concluded that algorithm-based care could allow for successful management of uncomplicated cystitis. [27]
Previous studies have been performed to evaluate symptoms or a group of symptoms as a prognostic indicator of the presence of a UTI with variable findings.
A review article evaluated which data from the history and clinical findings gave significantly increased diagnostic precision with regard to acute uncomplicated cystitis. It was found that a combination of dysuria and increased frequency of urination without increased vaginal secretions gave a 96% chance of a UTI [28]. Another study found the prognostic value of symptoms and clinical findings to be low in regard to how well they predicted a UTI [29].
Recently the results of a self-reporting questionnaire to assess UTIs and differential diagnosis was published and found to be applicable for clinical studies and practice for diagnosis of uncomplicated UTI. The study was, however, not a randomized controlled trial and could not provide a comparison with a control group for symptomatic and bacteriological outcome [30].
Implications/conclusion
This simplification of treatment strategy can give a shorter waiting period for the patient, better adherence to antibiotic guidelines, and more rational use of consultation time in outpatient clinics.
We conclude that our algorithm is a feasible and safe way to handle young women with symptoms of an acute cystitis, and that the strategy can be transferred to other out-of-hours services.
Acknowledgements
The authors would like to thank the nursing staff, doctors, administration, and patients at the out- of-hours service in Oslo.
The full study protocol can be obtained from Marianne Bollestad, email: marianne.bollestad@medisin.uio.no
Source of funding
National Centre for Emergency Primary Health Care, Norway. The Norwegian Committee on Research in General Practice. The Antibiotic Centre for Primary Care, Norway.
Ethical approval
The study is registered in Clinical trials (reference number: NCT01132131) and has been approved by the Regional Ethical committee (reference number: 2010/486).
Declaration of interest
There are no conflicts of interest in connection with the paper. The authors alone are responsible for the content and writing of the paper.
References
- Baerheim A. Empirical treatment of uncomplicated cystitis. Scand J Prim Health Care 2012;30:1–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arnold S, Straus S. Interventions to improve antibiotic prescribing practices in ambulatory care. Cochrane Database Syst Rev 2005;19:CD003539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willems L, Denckens P, Philips H, Henriquez R, Remmen R. Can we improve adherence to guidelines for the treatment of lower urinary tract infection? A simple, multifaceted intervention in out-of-hours services. J Antimicrob Chemother 2012;67:2997–3000. [DOI] [PubMed] [Google Scholar]
- Christiaens TC, De Meyere M, Verschraegen G, Peersman W, Heytens S, De Maeseneer JM. Randomised controlled trial of nitrofurantoin versus placebo in the treatment of uncomplicated urinary tract infection in adult women. Br J Gen Pract 2002;52:729–34. [PMC free article] [PubMed] [Google Scholar]
- Høye S, Frich JS, Lindbæk M. Use and feasibility of delayed prescribing for respiratory tract infections: A questionnaire survey. BMC Fam Pract 2011;18:12–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bleidorn J, Gagyor I, Kochen MM, Wegscheider K, Hummers-Pradier E. Symptomatic treatment (ibuprofen) or antibiotics (ciprofloxacin) for urinary tract infections? Results from a randomized controlled pilot trial. BMC Med 2010;8:30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Little P, Moore MV, Turner S, Rumsby K, Warner G, Lowes JA et al. Effectiveness of five different approaches in management of urinary tract infection: Randomised controlled trial. BMJ 2010;340:c199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katchman EA, Milo G, Paul M, Christiaens T, Baerheim A, Leibovici L. Three days vs longer duration of antibiotic treatment in cystitis in women: Systematic review and meta-analysis. Am J Med 2005;118:1196–207. [DOI] [PubMed] [Google Scholar]
- Skudal HK, Grude N, Kristiansen BK. Økende forekomst av antibiotikaresistens ved urinveisinfeksjoner [Increasing antibiotic resistance in urinary tract infections]. Tidsskr Nor Laegeforen 2006;126:1058–60. [PubMed] [Google Scholar]
- Little P, Merriman R, Turner S, Rumsby K, Warner G, Lowes JA, et al. Presentation, pattern and natural course of severe symptoms, and role of antibiotics and antibiotic resistance among patients presenting with suspected uncomplicated urinary tract infection in primary care: Observational study. BMJ 2010;340:b5633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferry S, Holm SE, Stenlund H, Lundholm R, Monsen TJ. Clinical and bacteriological outcome of different doses and duration of pivmecillinam compared with placebo therapy of uncomplicated lower urinary tract infection in women: The LUTIW project. Scand J Prim Health Care 2007;25:49–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flottorp S, Oxman AD, Cooper JG, Hjortdahl P, Sandberg S, Vorland LH. Retningslinjer for diagnostikk og behandling av akutte vannlatingsplager hos kvinner [Guidelines for diagnosis and treatment of acute urinary tract problems in women]. Tidsskrift Nor Laegeforen 2000;120:1748–53. [PubMed] [Google Scholar]
- Grude N, Tveten Y, Kristiansen BE. Urinary tract infections in Norway: Bacterial etiology and susceptibility. A retrospective study of clinical isolates. Clin Microbio Infect 2001;7:543–7. [PubMed] [Google Scholar]
- Jureen R, Digranes A, Bærheim A. Urinveispatogene bakterier ved ukompliserte nedre urinveisinfeksjoner hos kvinner [Urinary tract pathogens in uncomplicated lower urinary tract infections in women in Norway]. Tidsskr Nor Laegeforen 2003;123:2021–2. [PubMed] [Google Scholar]
- Grude N, Tveten Y, Jenkins A, Kristiansen BE. Uncomplicated urinary tract infections: Bacterial findings and efficacy of empirical antibacterial treatment. Scand J Prim Health Care 2005;23:115–19. [DOI] [PubMed] [Google Scholar]
- Lindbæk M, Berild D, Eliassen KE, Fetveit A, Grude N, Hjortdahl P. Nasjonale faglige retningslinjer for antibiotikabruk i primærhelsetjenesten [National guidelines for use of antibiotics in primary health care]. Helsedirektoratet. 2013. Available at http://www.antibiotikasenteret.no/index.php/retningslinjer-for-antibiotikabruk (accessed October 2, 2014).
- Kahlmeter G, Poulson HO. Antimicrobial susceptibility of Escherichia coli from community-acquired urinary tract infections in Europe: The ECO-SENS study revisited. Int J Antimicrob Agents 2012;9:45–51. [DOI] [PubMed] [Google Scholar]
- Monsen TJ, Holm S, Ferry BM, Ferry S. Mecillinam resistance and outcome of pivmecillinam treatment in uncomplicated urinary tract infection in women. APMIS 2014; 122:317–23. [DOI] [PubMed] [Google Scholar]
- Dewar S. Emerging clinical role of pivmecillinam in the treatment of urinary tract infection in the context of multidrug-resistant bacteria. J Antimicrob Chemother 2014;69:303–8. [DOI] [PubMed] [Google Scholar]
- Bent S, Saint S. The optimal use of diagnostic testing in women with acute uncomplicated cystitis. Am J Med 2002;113:20–8. [DOI] [PubMed] [Google Scholar]
- Flottorp S, Oxman AD, Håvelsrud K, Treweek S, Herrin J. Cluster randomised controlled trial of tailored interventions to improve the management of urinary tract infections in women and sore throat. BMJ.2002;325:367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grigoryan L, Trautner BW, Gupta K. Diagnosis and management of urinary tract infections in the outpatient setting: A review. JAMA 2014;312:1677–84. [DOI] [PubMed] [Google Scholar]
- Aspevall O, Hallander H, Gant V, Kouri T. European guidelines for urinalysis: A collaborative document produced by European clinical microbiologists and clinical chemists under ECLM in collaboration with ESCMID. Clin Microbiol Infect 2001;7:173–8. [DOI] [PubMed] [Google Scholar]
- Huppert JS, Biro F, Lan D, Mortensen JE, Reed J, Slap GB. Urinary symptoms in adolescent females: STI or UTI? J Adolesc Health 2007;40:418–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Österberg E, Aspevall O, Grillner L, Persson E. Young women with symptoms of urinary tract infection: Prevalence and diagnosis of chlamydial infection and evaluation of rapid screening of bacteriuria. Scand J Prim Health Care 1996;14:43–9. [DOI] [PubMed] [Google Scholar]
- Kahlmeter G. An international survey of the antimicrobial susceptibility of pathogens from uncomplicated urinary tract infections: The ECO.SENS Project. Clin Microbiol Infect 2003;51:67–76. [DOI] [PubMed] [Google Scholar]
- Schauberger CW, Merkitch KW, Prell AM. Acute cystitis in women: Experience with a telephone-based algorithm. WMJ 2007;106:326–9. [PubMed] [Google Scholar]
- Stephen B, Nallamothu BK, Simel DL, Fihn SD, Saint S. Does this woman have an acute uncomplicated urinary tract infection? JAMA 2002;287:2701–10. [DOI] [PubMed] [Google Scholar]
- Medina-Bombardo D, Jover-Palmer A. Does clinical examination aid in the diagnosis of urinary tract infections in women? A systematic review and meta-analysis. BMC Fam Pract 2011;12:111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alidjanov JF, Abdufattaev UA, Makhsudov SA, Pilatz A, Akilov FA, Naber KG, et al. New self-reporting questionnaire to assess urinary tract infections and differential diagnosis: Acute cystitis symptoms score. Urol Int 2014;92:230–6. [DOI] [PubMed] [Google Scholar]