Abstract
Background
Serotonin (5-hydroxytryptamine, 5-HT) is modulated by sex steroid hormones and affects vascular function and mood. In the Kronos Early Estrogen Prevention Cognitive and Affective Ancillary Study (KEEPS-Cog), women randomized to oral conjugated equine estrogens (oCEE) showed greater benefit on affective mood states than women randomized to transdermal 17β-estradiol (tE2) or placebo (PL). This study examined the effect of these treatments on the platelet content of 5-HT as a surrogate measure of 5-HT synthesis and uptake in the brain.
Methods
The following were measured in a subset (n = 79) of women enrolled in KEEPS-Cog: 5-HT by ELISA, carotid intima-medial thickness (CIMT) by ultrasound, endothelial function by reactive hyperemia index (RHI), and self-reported symptoms of affective mood states by the Profile of Mood States (POMS) questionnaire.
Results
Mean platelet content of 5-HT increased by 107.0%, 84.5% and 39.8%, in tE2, oCEE and PL groups, respectively. Platelet 5-HT positively correlated with estrone in the oCEE group and with 17β- estradiol in the tE2 group. Platelet 5-HT showed a positive association with RHI, but not CIMT, in the PL and oCEE groups. Reduction in mood scores for depression-dejection and anger-hostility associated with elevations in platelet 5-HT only in the oCEE group (r = −0.5, p = 0.02).
Conclusions
Effects of oCEE compared to tE2 on RHI and mood may be related to mechanisms involving platelet, and perhaps neuronal, uptake and release of 5-HT and reflect conversion of estrone to bioavailable 17β- estradiol in platelets and the brain.
Keywords: conjugated equine estrogen, 17β-estradiol, 5-hydroxytryptamine, Kronos Early Estrogen Prevention Study, platelet
Introduction
Serotonin (5-hydroxytryptamine, 5-HT) is a monoamine that functions as a neurotransmitter in the brain and exerts complex effects on the cardiovascular system 1. Within the peripheral circulation, a portion of 5-HT is inactivated by the liver, but the majority is taken-up, transported and stored in dense granules of platelets. 5-HT released from activated platelets binds to serotonergic receptors on platelets initiating platelet aggregation, the vascular endothelium inducing the release of vasoactive substances and the vascular smooth muscle initiating either relaxation or contraction depending on the concentration and the anatomical origin of the blood vessel 2, 3.
In the brain, 5-HT is synthesized in serotonergic neurons from L-tryptophan by tryptophan hydroxylase. Most 5-HT in the brain is stored within synaptic vesicles of the raphe nuclei 4. 17β-estradiol increases mRNA of serotonin reuptake transporter (SERT) as well as the density of SERT-binding sites in the brains of experimental animals 5, 6. The 5-HT transporter on human platelets is identical to the brain 5-HT transporter 7; platelet 5-HT content has been used as a surrogate marker of its synthesis and uptake in the brain 8.
Evidence supports an association between depressive mood disturbances, low brain 5-HT, risk of cardiovascular disease and adverse cardiovascular events 9. Incidence of depression is greater in women compared to men, and following menopause, depressive symptoms and mood disturbances increase in some women 10, 11. Estrogens exert antidepressant effects, in part, by modulating synthesis and uptake of 5-HT 12. Estrogens affect vasomotor tone by increasing release of endothelium-derived vasodilatory substances and reducing platelet aggregation and secretion 13. Therefore, menopausal hormone therapy (MHT) may reduce depressive symptoms directly by modulating 5-HT levels in the brain and indirectly by modulating cerebral vascular reactivity 14.
Effects of MHT on mood and the relationship to 5-HT in the systemic circulation and cardiovascular risk remains unexplored in menopausal women. Several studies report a positive association between platelet 5-HT turnover and plasma estrogen levels in menopausal women 15-17, but none have related these to cardiovascular risk and only one compared two different formulations of estrogen. The objective of this study was to compare the association of two MHT formulations (oral conjugated equine estrogens and transdermal 17β-estradiol) with serum and platelet 5-HT levels in healthy, recently menopausal women and the relationship of these levels with progression of cardiovascular disease as defined by carotid intima-medial thickness (CIMT), and with endothelial function measured by pulse volume digital tonometry. In addition, using platelet 5-HT as a surrogate for 5-HT in the brain, the association between platelet 5-HT and mood also was examined.
Methods
Participants
Participants (n = 79) were a subset of Caucasian women enrolled in the Kronos Early Estrogen Prevention Cognitive and Affective Ancillary Study (KEEPS-Cog) at Mayo Clinic, Rochester, MN (Figure 1). The KEEPS-Cog, an ancillary study to the parent Kronos Early Estrogen Prevention Study (KEEPS), was designed to test the “critical period hypothesis” that MHT started early in menopause would improve mood and maintain cognition 18. The KEEPS was a multicenter, randomized, double-blinded, placebo-controlled clinical trial enrolling healthy women between July 2005-June 2008 without a prior history of cardiovascular or cerebrovascular disease (NCT00154180) 19. Briefly, inclusion criteria were: women between 42-58 years of age who experienced natural menopause and were within 6 months to 3 years of menopause. Menopause was defined by FSH level ≥ 35 ng/ml and estradiol levels <25 pg/ml. A normal mammogram within 1 year of randomization was required. Exclusion criteria were: coronary arterial calcification score of >50 Agatston Units, smoking over 10 cigarettes per day, BMI >35 kg/m2, history of cardiovascular disease, low density lipoprotein cholesterol >190 mg/dL, triglycerides >400 mg/dL, blood glucose >126 mg/dL, uncontrolled hypertension (systolic blood pressure >150 mmHg and/or diastolic blood pressure >95 mm Hg), current or recent (6 months) use of cholesterol lowering medications (statins, fibrate, or >500 mg/day niacin), follicle stimulating hormone (FSH) level ≤35 ng/mL, estrogen levels ≥ 25 pg/mL 19, and symptoms consistent with clinical depression (based on Beck’s Depression Inventory 2nd edition score of ≥ 28/63 or reporting suicidal ideation as assessed by the Beck’s Depression Inventory or scoring ≤ 22/30 on the Mini-Mental State Exam) 20, 21.
Figure 1.
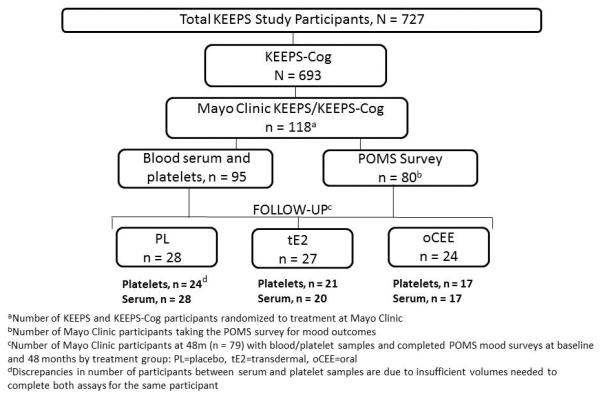
Flow chart defining the KEEPS-Cog cohort participating in this study. Total number of participants (n = 79) in whom sufficient serum and platelets were available for analysis is reported by treatment group in placebo (PL), transdermal (tE2) or oral (oCEE) estrogen preparations.
Study design
Women were randomized for 48 months to either: oral conjugated equine estrogens (o-CEE; Premarin, 0.45 mg/day); transdermal 17β-estradiol (t-E2; skin patch, Climera, 50 μg/day; or placebo pills and patch (PL). Women randomized to active treatments also received oral progesterone (Prometrium; micronized progesterone, 200 mg/day) for 12 days each month for the duration of the study to protect the uterus. Each participant underwent a medical examination, including body morphometrics and standard blood chemistries prior to randomization 19, 22.
Standard protocol approvals, registrations, and patient consents
This study was approved by the Mayo Clinic Institutional Review Board (IRB protocol #2241). All participants gave written informed consent.
Profile of Mood States (POMS) survey
POMS is a self-report survey composed of 65 psychological distress measurements, each ranked on a five-point Likert scale organized by mood inventories or subscales as depression-dejection, anger-hostility, tension-anxiety, confusion-bewilderment, fatigue-inertia and vigor-activity23. POMS was completed prior to randomization (baseline; BL) and after 48 months (48m) of treatment (Figure 1). In the KEEPS-Cog study, there were significant effects of MHT on depression-dejection, anger-hostility and tension-anxiety 21 and thus, these moods became the focus of the present evaluations with 5-HT.
Blood sampling and chemistries
Fasting venous blood was collected at BL and 48m after randomization into tubes containing hirudin plus soybean trypsin inhibitor or sodium citrate anticoagulants 24 and without anticoagulant for serum. Serum to be used for measurement of 5-HT was stored at −80°C until analysis. All samples were batched and assayed at the conclusion of the study. Serum 5-HT was quantified by ELISA (GenWay Biotech Inc., San Diego, CA). Inter- and intra-assay coefficients of variation were 7.6% (n=6) and 11% (n=5), respectively.
Serum total cholesterol, low-density lipoprotein cholesterol (LDL-C) and high-density lipoprotein cholesterol (HDL-C), triglycerides and fasting blood glucose (FBG) were measured by Kronos Science Laboratories (Phoenix, AZ). Estrone (E1), 17β-estradiol (E2), and testosterone (T) were measured using high performance liquid chromatography-tandem mass spectrometry (HPLC-MS/MS) and sex hormone binding globulin (SHBG) was measured by two-site chemiluminescent immunoassay, using the Immulite 2000 at the Mayo Clinic Department of Laboratory Medicine and Pathology (Rochester, MN).
Platelet lysates
Washed platelets were prepared as described previously 24. Absence of leukocytes contamination in the platelet preparation was verified by Coulter counter prior to storage of the platelets at −80°C 25. To prepare the lysate, the isolated stored platelets were thawed in a 37° water bath and placed in 200 μl of lysis buffer [(#0103004-L; RayBioTech, Norcross, GA) containing 5 mM TRIS, 0.5% TRITON X100, and protease inhibitor cocktail (#P8340, Sigma, St. Louis, MO], pH 7.4)]. This solution was passed through a 26-guage needle at least 3 times, sonicated and then centrifuged at 10,000g for 5min to remove lysed membrane particulates and organelles. The total protein concentration in the supernatant was measured by bicinchoninic acid assay (BCA kit #23225, Pierce, Rockford, IL) and 5-HT was analyzed by ELISA according to manufacturer’s instructions (GenWay Biotech, Inc., San Diego, CA). Inter- and intra-assay coefficients of variation were 4.5% (n = 11) at BL and 12.0% (n = 9) at 48m. Although serum and platelets were collected from all 79 participants, total volumes of materials in some cases were insufficient to perform all of the assays in all of the participants. The number of participants from whom platelet assays were performed is provided in Figure 1.
Carotid intima-medial thickness (CIMT)
CIMT, the primary outcome of KEEPS, was measured by B-mode ultrasound 22, 26.
Endothelial function
Endothelial function was measured using a peripheral tonometer to detect changes in pulse volume digital tonometry during reactive hyperemia (EndoPAT, model 2000; Itamar Medical, Ltd., Caesarea, Israel) 27, 28. The Reactive Hyperemia Index (RHI) was calculated by dedicated software in the system computer.
Statistical analysis
Baseline and follow-up participant data was evaluated for normality using histograms and box plots for graphical visualization of data points. Conventional cardiovascular risk factors (with the exception of participant count and percentage of smokers) met normality. Participant characteristics are shown as mean and standard deviation (mean ± SD) for continuous variables and as percentages for categorical variables. Hormones were treated as continuous variables. Mean change in mood scores were calculated as the difference in scores at 48m from BL. Sample size calculations were performed at 80% power (α=0.05). The effect size required to achieve statistical significance of MHT on platelet 5-HT levels at 48m is 8 participants. Linear regression was used to test the associations between serum and platelet lysate 5-HT concentrations and POMS outcomes. Correlation coefficients (r) and corresponding p-values were calculated for the simple linear regression model. One-way analysis of variance (ANOVA) was performed to compare the means of the three treatment groups, followed by Dunn’s and Holm-Sidak post-hoc tests to control Type I error rate. The familywise error rate (FWER) was set to 0.05.
Results
General characteristics
In this sub-set of KEEPS-Cog participants, the average age at enrollment was 52 years. Other demographics and clinical characteristics at BL and 48m are presented in Table 1. At BL, conventional cardiovascular risk factors were within normative ranges. At 48m, waist circumference was significantly larger in the PL group compared to tE2 group (p = 0.01) and triglyceride levels were significantly higher in the oCEE group compared to tE2 group (p = 0.02). The number of participants meeting criteria for metabolic syndrome 29 at 48m was similar among groups: PL: 1; tE2: 2; oCEE: 2.
Table 1.
Clinical characteristics of the subset of KEEPS-Cog participants at baseline (BL) and 48 months (48m) following randomization to treatment.
| Placebo (PL) | Transdermal (tE2) | Oral (oCEE) | ||||
|---|---|---|---|---|---|---|
| BL | 48m | BL | 48m | BL | 48m | |
| Number of participants | 28 | 27 | 24 | |||
| Age (years) | 52.7±0.3 | 57.2±2.0 | 52.8±0.6 | 57.3±2.5 | 52.2±0.4 | 57.0±2.3 |
| Time past menopause (months) |
16.0±1.7 | 64.0±8.5 | 17.1±1.9 | 69.0±8.9 | 19.3±2.2 | 67.0±10.3 |
| Mean systolic blood pressure (mmHg) |
120.9±13.3 | 121.0±14.7 | 118.6±15.8 | 119.0±16.1 | 123.6±12.3 | 121.0±12.4 |
| Mean diastolic blood pressure (mmHg) |
74.2±7.6 | 77.0±8.9 | 72.5±8.4 | 73.0±9.8 | 78.2±7.0 | 76.0±8.8 |
| Body mass index (kg/m2) | 27.7±3.6 | 29.0±3.9 | 26.0±4.4 | 26.0±4.3 | 27.7±4.4 | 28.0±5.1 |
| Waist circumference (cm) | 86.2±2.1 | 93.0±10.0 | 83.0±2.5 | 83.0±11.7* | 86.2±3.8 | 89.0±12.6 |
| Low-density lipoprotein (LDL, mg/dL) |
136.9±7.0 | 117.0±29.1 | 125.2±6.4 | 111.0±33.1 | 137.3±6.1 | 101.0±39.5 |
| High-density lipoprotein (HDL, mg/dL) |
60.7±3.0 | 72.0±13.5 | 59.9±3.1 | 74.0±12.1 | 60.1±2.8 | 77.0±17.1 |
| Triglycerides (mg/dL) |
91.1±9.9 | 92.0±55.7 | 94.0±7.6 | 81.0±36.3 | 95.5±10.2 | 111.0±40.4** |
| Fasting blood glucose (mg/dL) |
93.6±1.6 | 81.0±6.3 | 95.0±1.3 | 79.0±7.1 | 91.1±2.2 | 81.0±10.4 |
| Carotid intima medial thickness (CIMT;mm) |
0.67±0.07 | 0.70±0.09 | 0.69±0.09 | 0.73±0.11 | 0.67±0.07 | 0.69±0.08 |
| Reactive Hyperemia Index | 2.4±0.9 | 2.6±0.7 | 2.4±0.7 | 2.2±0.7 | 2.5±0.7 | 2.5±0.6 |
| Platelet count, citrate (103/μL) |
240.7±37.1 | 243.2±59.0 | 237.9±53.4 | 262.4±61.8 | 226.7±52.8 | 243.7±56.7 |
| Smoking status | ||||||
| Never | 21 | 17 | 15 | |||
| Past | 7 | 6 | 9 | |||
| Current | 4 | |||||
Conventional cardiovascular (CV) risk factors measured at baseline and 48 months are reported as mean ± SD; the number of participants is reported as counts and smoking status is presented as percentage of counts (%). Statistical significance denoted as:
PL vs. tE2 at 48 months, p=0.01;
oCEE vs. tE2 at 48 months, p=0.02.
At 48m, compared to PL, serum concentrations of E1 were significantly higher in the oCEE group (p = 0.001); E2 levels were significantly higher in tE2 group (p < 0.0001). Serum concentrations of SHBG were significantly higher in oCEE group as compared to tE2 and PL groups (p = 0.001; Table 2). Serum total testosterone did not differ among groups.
Table 2.
Hormone concentrations in serum of KEEPS-Cog participants at 48 months.
| Hormone |
PL
(n = 23) |
tE2
(n = 25) |
oCEE
(n = 20) |
|---|---|---|---|
| Estrone (E1) (pg/mL) | 20±6.3 | 34±13.2 | 67±36.8††† |
| Estradiol (E2) (pg/mL) | 6.1±2.4 | 29±27.8† | 14±6.9 |
| Testosterone (T) (ng/dL) | 23±16.0 | 24.2±11.6 | 27.7±11.7 |
| Sex Hormone Binding Globulin (SHBG) (nmol/L) |
50.3±27.4 | 58.6±24.5 | 106.6±47.6†† |
compared to PL, p=0.001;
compared to PL, p<0.0001;
compared to PL and tE2, p=0.001.
Effects of MHT on serum and platelet 5-HT
At BL, serum 5-HT levels showed large variability and did not differ among treatment assignments (p = 0.98) averaging 77.0 ± 8.7 ng/mL (mean ± SD) for all participants. Serum levels of 5-HT did not change significantly from BL nor differ significantly among groups after 48m of treatment: 75.1 ± 7.0, 78.4 ± 8.4, 79.5 ± 9.2 ng/mL for PL, tE2 and oCEE, respectively (p = 0.16; Figure 2).
Figure 2.
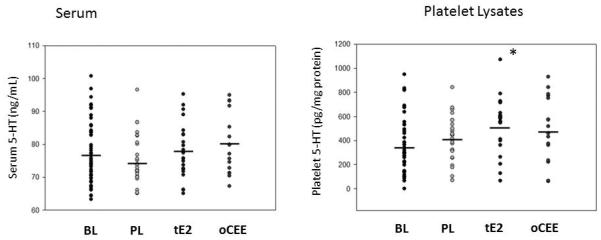
Levels of 5-HT in serum (left panel) and platelet lysates (right panel) at baseline (BL) and after 48 months (48m) of treatment with placebo (PL), transdermal 17β estradiol (tE2) or oral conjugated equine estrogens(oCEE). Asterisk denotes statistical significant difference from BL (p = 0.002*).
Platelet count did not vary significantly over the course of the study or among groups at follow-up (Table 1). At BL, platelet content of 5-HT did not differ among groups (p = 0.80): 294.5 ± 218.3 (n=24), 355.6 ± 283.1 (n=21) and 252.2 ±157.0 (n=17) pg/mg of protein in the PL, tE2 and oCEE groups, respectively. After 48m of treatment, mean platelet 5-HT content increased by 39.8% (to 411.2 ± 204.5 pg/mg protein) in the PL group and by 107% (to 529.8 ± 236.7 pg/mg protein) and 84.5% (to 465.4 ± 278.9 pg/mg protein) in the tE2 and oCEE groups, respectively (Figure 2). The change in mean platelet content between the tE2 and BL group was statistically significant (p = 0.02). There was a positive association between serum and platelet 5-HT only in the tE2 group (p = 0.04; data not shown).
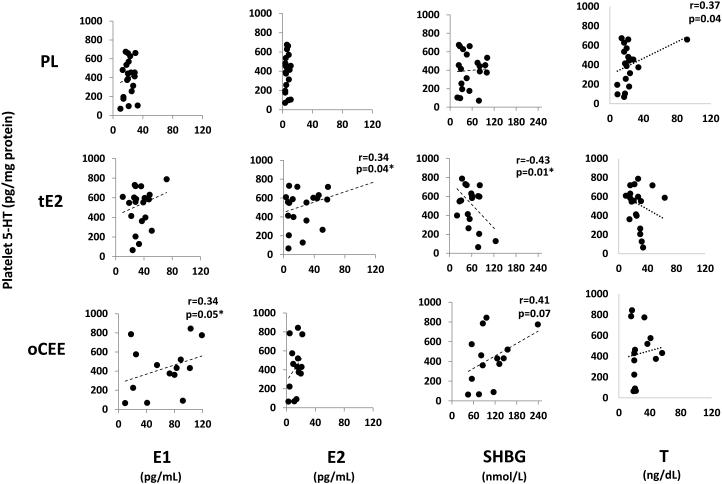
There were nominal positive associations between E2 and E1 and platelet levels of 5-HT in the tE2 (r = 0.34, p = 0.04) and oCEE (r = 0.34, p = 0.05; Figure 3) groups, respectively and between testosterone and platelet 5-HT levels in the PL group at 48m (R=0.37, p=0.04). Associations of SHBG with platelet 5-HT concentrations were negative and statistically significant for tE2 (r = −0.43, p = 0.01) but showed a positive trend for oCEE (r = 0.41, p = 0.07; Figure 3). There was a nominally significant positive association between E2/E1 ratio with platelet 5-HT (r = 0.38, p = 0.04) in the tE2 group. The range in E2 concentrations in the oCEE group was not broad enough to establish meaningful evaluation of these ratios in this group.
Figure 3.
Correlations of serum estrone (E1), estradiol (E2), sex hormone binding globulin (SHBG) and testosterone (T) with platelet lysate levels of 5-HT in women receiving placebo (PL) and transdermal (tE2, 17β-estradiol; Climera, 50 μg/day) or oral (oCEE; Premarin, 0.45 mg/day) hormone preparations. Each dot represents an individual. .
Platelet 5-HT and vascular assessments
There were no significant associations between either serum or platelet 5-HT and changes in CIMT (data not shown). However, platelet 5-HT content was positively and significantly associated with higher RHI in women of the PL and oCEE groups at 48m (r = 0.42, p = 0.03; r = 0.57, p = 0.02, respectively; Figure 4).
Figure 4.
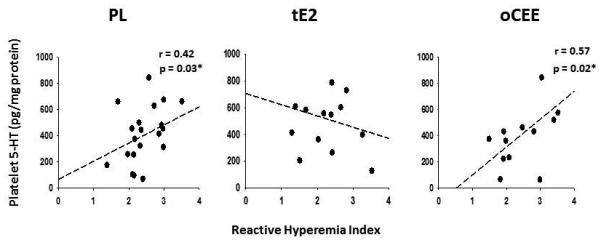
Relationship between platelet 5-HT and endothelial function as measured by flow-mediated reactive hyperemia index (RHI) via pulse volume digital tonometry at 48m. Correlations are presented by treatment in placebo (PL, n = 21), transdermal (tE2, n = 14) and oral (oCEE, n = 13) groups. Each symbol represents data from an individual.
Platelet 5-HT and relationship to mood
There were no statistically significant differences in overall POMS scores or individual components of POMS among group assignments at baseline. Compared to BL, at 48 months, overall negative mood states as measured by the POMS scale increased (worsened) in the PL group by 10.5% but significantly decreased (improved) by 4.7% and 7% in the tE2 and oCEE groups, respectively (Table 3). Changes in scores for individual components of mood followed this general pattern with reduction of symptoms for depression-dejection, anger-hostility and tension-anxiety being greater in both the oCEE and tE2 groups compared to PL (p = 0.001). Reduction in scores in the oCEE group were greater for anger-hostility and tension-anxiety than in the tE2 group (p = 0.001). Lower scores for depression-dejection and anger-hostility significantly correlated with increases in platelet content of 5-HT only in women of the oCEE group (Figure 5). Serum testosterone levels did not correlated with anger-hostility scores in any group (data not shown).
Table 3.
Baseline and mean changes in mood parameters of KEEPS-Cog participants following menopausal hormone treatment.
| Mood Parameters | PL | tE2 | oCEE | |||
|---|---|---|---|---|---|---|
|
Mean (SD)
BL |
Mean
Change |
Mean (SD)
BL |
Mean
Change |
Mean (SD)
BL |
Mean
Change |
|
| Profile of Mood States (POMS) |
37.7±14.4 | 4.1±5.9 | 42.8±12.2 | −1.7±3.2* | 42.9±13.1 | −2.5±1.6* |
| Depression-Dejection | 3.0±3.7 | 1.8±0.6 | 4.4±3.8 | −0.2±1.0* | 4.5±4.6 | 0.6±2.0*† |
| Anger-Hostility | 2.9±3.3 | 0.9±4.2 | 3.8±3.6 | −1.0±0.6** | 5.3±5.3 | −1.3±1.7** |
| Tension-Anxiety | 4.4±4.1 | −0.5±0.7 | 6.0±3.8 | −1.9±0.1* | 5.4±3.5 | −1.3±0.2*‡ |
Self-reported Profile of Mood States (POMS) parameters of KEEPS participants by treatment group, presented as mean ± SD; mean change was calculated as the difference in scores at 48 months for PL (n=28), tE2 (n=27) and oCEE (n=21) groups from baseline. Statistical significance reported for mean change:
tE2 and oCEE vs. PL, p = 0.001;
tE2 and oCEE vs. PL, p = 0.02;
oCEE vs. tE2, p = 0.03;
oCEE vs. tE2, p = 0.001.
Figure 5.
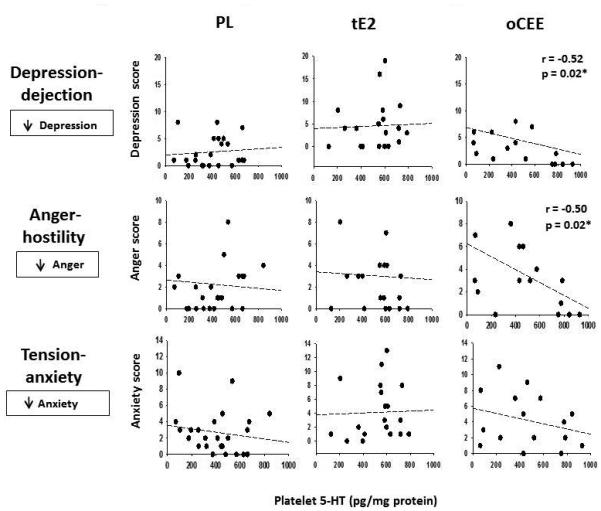
Associations of peripheral platelet 5-HT levels (pg/mg protein) with components of mood by treatment group at 48m. A lower self-reported mood score represents improved mood. Consistent significant associations were found between increases in platelet 5-HT and decreases in scores for depression-dejection and anger-hostility (less depression-dejection and anger-hostility) in the oCEE group at follow up (r = −0.52, p = 0.02).
Discussion
Results of this study provide evidence that oral conjugated equine estrogens and transdermal 17β-estradiol differentially affect content of serotonin in platelets and components of mood in healthy menopausal women. At menopause, risk increases for developing mood disturbances and cardiovascular disease 10, 11. Serotonin may contribute to both processes as a neurotransmitter in the brain and as a vasoactive amine that promotes endothelium-dependent relaxation but causes contraction of vascular smooth muscle and promotes platelet aggregation 1, 14.
Evidence suggests a relationship between depression and cardiovascular disease. However, the relationship of MHT and 5-HT to both processes is controversial, in part, due to regulation and expression of 5-HT receptor subtypes, differences in measurement of 5-HT and differences in dose and type of estrogen treatments used in various studies 3, 30, 31. The present study compared two MHT products which are used clinically for relief of menopausal symptoms and represent doses consistent with clinical guidelines 32.
Results of the present study are consistent with other observations that MHT correlates with circulating (platelet) 5-HT levels and turnover 15, 17 and improves mood and reduces depressive symptoms 33. The progesterone component of MHT may be involved in these effects, as progesterone to upregulates the 5-HT(2A) receptor binding potential 34 and serum levels of progesterone correlated with increased metabolism of 5-HT in the brain 35. In addition, our results provide insight into differences in effects of oral CEE, which contain multiple metabolites of 17β-estradiol, the primary of which is estrone 36 as compared to transdermal 17β-estradiol. In addition, variation in serum concentrations of SHBG between treatment groups indicates direct effects of the oral product on the liver similar to what is reported for effects on pro-inflammatory cytokines such as C-reactive protein 37.
Serum concentrations of 5-HT reflect, in part, amine released from activated platelets in coagulated blood. Levels of serum 5-HT measured in the present study are consistent with those measured by others in postmenopausal women with and without MHT 38, 39. However, serum levels do not account for incomplete activation or release of 5-HT from the platelets as indicated by the lack of positive correlation between serum and platelet content of 5-HT in the PL and oCEE groups. Thus, serum levels of 5-HT could underestimate effects of the treatment on synthesis and uptake of the amine.
Differential effects of MHT on platelet 5-HT and vascular function
The platelet count did not differ significantly among the groups after 48 months of treatment. Both hormone treatments tended to increase 5-HT content of platelets with the major estrogenic compound in each treatment having a positive association with platelet content of 5-HT. The E2/E1 ratio may reflect total biologically available estrogens in menopausal women 40, 41. The platelet content of 5-HT was positively associated with the E2/E1 ratio only in the tE2 group, suggesting that the relative concentration of estrogen metabolites may affect synthesis, uptake, release and degradation of the amine 12, 42.
Increases in SHBG may partially explain the differences in platelet content of 5-HT between oCEE and tE2 groups. Binding affinity of sex steroids to SHBG is T>E2>E1 thus reducing bio-available E2. Conversion of E1 to E2 at the cellular level would increase site-specific of E2 43. Results of the present study are consistent with those of others showing that oCEE treatment increases serum SHBG 44 and extend observations to indicate a negative association between serum SHBG and platelet 5-HT levels in the tE2 group but a positive correlation in the oCEE group.
Uptake of 5-HT from the blood by platelets, endothelium and nerve terminals may be cardioprotective, but with onset of cardiovascular disease, the sensitivity of platelets to 5-HT uptake and clearance by the endothelium may diminish 1, 45. RHI, as a measure of vasodilatation, increased with increasing platelet content of 5-HT in the oCEE group. 5-HT released from platelets can augment release of endothelium-derived relaxing factors though activation of receptors on the endothelium 46. The absence of an association with tE2 may reflect the relationship between platelet and serum levels 5-HT in this group and not the others. Furthermore, 5-HT is one of many vasoactive and mitogenic products released from activated platelets 47 and the measure of individual components may not represent the collective effect on vascular remodeling.
CIMT increases with age and the increases in CIMT were comparable among treatment groups 48 which reflects, in part, the low cardiovascular risk status of women at the time of enrollment, the collective effect of other vasoactive and mitogenic products that are released by activated platelets 47, the genomic interactions with treatment 49, as well as the short duration of the study. It remains to be seen if changes in CIMT would be different among treatment assignments after women completed study drugs. Additional, follow-up studies of KEEPS participants would provide insight into temporal relationships of MHT use and vascular remodeling.
Differential effects of MHT on platelet 5-HT and mood
Results of the present study are consistent with other reports of MHT improving mood and a sense of well-being in menopausal women with absent or mild depressive symptoms 50. The changes in mood scores are consistent with clinical meaningful improvement in mood based on the standard established for clinical trials examining pain control 51. An expert panel recommended that a change of one standard error of the mean or half of a standard deviation should be considered clinically significant. According to these guidelines, the o-CEE group demonstrated clinically meaningful change at 48m on the depression-dejection and tension-anxiety subscales (Table 3).
Results of the present study also are consistent with other reports of positive correlations between plasma estrone and 17β-estradiol on platelet 5-HT levels in non-depressed postmenopausal women 15. However, only in the oCEE group were scores for depression-dejection and anger-hostility related to platelet content of 5-HT. This difference between oCEE and tE2 treatments most likely reflects different pharmacokinetics and physiological mechanisms of action of estrogens metabolites associated with the two formulations. The oral preparation of conjugated equine estrogens (CEE) contains E1, as well as estrone sulfate and 17β-estradiol sulfate, which have lower binding affinity for the estrogen receptor than 17β-estradiol. Therefore, ligand-bound estrogen receptors will affect modulation of the serotonergic system differently at multiple control points, including increasing enzymatic expression of tryptophan hydroxylase-2, the rate limiting enzyme for 5-HT synthesis, and release and uptake through increased 5-HT receptor and transporter expression 5, 52, 53. E1 can be converted to 17β-estradiol and as such may serve as a circulating storage form of the hormone 40. Therefore, circulating levels of E2 may not be reflective of conversion of E1 to E2 at the tissue level in the oCEE group.
Limitations
This study has several limitations. First, the analysis utilized only change from baseline at one time point (48 months) and does not take into account longitudinal variability 54. Second, concentrations of 5-HT in platelet lysate may not reflect that released following agonist activation of platelets as might occur during reactive hyperemia 55. Third, 5-HT measured in platelets is only an estimate of synthesis and transporter activity in the brain. Fourth, effects of MHT on mood reporting may be biased as POMS is a profile of immediate mood state and may be influenced by personal transient circumstances. Fifth, 5-HT is a monoamine derived from the amino acid tryptophan, the levels of which are affected by food intake. Oral intake of 5-HT cannot pass into the serotonergic pathway of CNS, unless it is metabolized to 5-hydroxytryptophan (5-HTP), although this factor may be limited as samples were collected after an overnight fast. In addition, the active smokers were in the tE2 group and it is unclear how this cardiovascular risk factor would affect 5-HT metabolism. Finally, KEEPS-Cog participants were a homogenous population of healthy Caucasian women and therefore, results may not generalize to a population of women with various cardiovascular risk factors.
Conclusion
The platelet content of 5-HT and its associations to E2/E1 ratio differ between oCEE and transdermal E2, suggesting that local concentrations of estrogen metabolites affect 5-HT uptake and synthesis. Although both oCEE and transdermal tE2 improved mood, mechanisms involving synthesis and activation of 5-HT transporters with neuronal activation may differ as higher platelet content of 5-HT, as a surrogate for synthesis and uptake, was associated with improved mood only in the oCEE group. It was not possible to identify a relationship between platelet 5-HT and vascular remodeling reflected by changes in CIMT. However, analysis of platelet 5-HT with a more dynamic and transient measure of cardiovascular function, for example, endothelial reactive hyperemia, provides insight into interactions among MHT, 5-HT, endothelial and platelet physiology. Additional studies are needed to determine how various formulations of MHT affect mood, 5-HT and cardiovascular risk relative to variants in genes encoding enzymes which metabolize E1 to E2 and their sulfonation and genes encoding estrogen receptors.
Acknowledgments
Study Funding: The Aurora Foundation to the Kronos Longevity Research Institute, NIH HL90639, NS066147, HD65987, AG029624, P50 AG033514, UL1 RR0241501 and the Mayo Foundation. Study medications for KEEPS were supplied in part by Bayer Health Care and by Abbott Pharmaceuticals.
Additional Contributions: We gratefully acknowledge the dedicated efforts of all the investigators and staff at the KEEPS clinical centers, the KEEPS Data Coordinating Center at KLRI, study coordinator, Teresa G. Zais (deceased), and Robert D. Litwiller for technical assistance collecting the blood. Above all, we recognize and thank the KEEPS participants for their dedication and commitment to the programs of the Mayo Clinic Women’s Health Research Center.
Footnotes
Disclosures
Author Contributions:
Dr. Raz: conceptualization of the study, analysis and interpretation of the data, drafting the manuscript.
Larry Hunter: data collection, analysis and revising the manuscript
Dr. Dowling: data collection, interpretation of data and revising the manuscript
Dr. Wharton: data collection, interpretation of data and revising the manuscript
Dr. Gleason: data collection, interpretation of data and revising the manuscript
Dr. Jayachandran: analysis and interpretation of the data, revising the manuscript
Layne Anderson: data collection
Dr. Asthana: design or conceptualization of the study; interpretation of data and revising the manuscript
Dr. Miller: design or conceptualization of the study, interpretation of data and revising the manuscript.
From the National Center for Research Resources (NCRR), a component of the National Institutes of Health (NIH), and the NIH Roadmap for Medical Research. Contents of this paper are solely the responsibility of the authors and do not necessarily represent the official view of NCRR or NIH.
Author Disclosures: None
References
- 1.Fidalgo S, Ivanov DK, Wood SH. Serotonin: from top to bottom. Biogerontology. 2013;14:21–45. doi: 10.1007/s10522-012-9406-3. [DOI] [PubMed] [Google Scholar]
- 2.Riad M, Tong XK, el Mestikawy S, Hamon M, Hamel E, Descarries L. Endothelial expression of the 5-hydroxytryptamine1B antimigraine drug receptor in rat and human brain microvessels. Neuroscience. 1998;86:1031–5. doi: 10.1016/s0306-4522(98)00196-1. [DOI] [PubMed] [Google Scholar]
- 3.Vanhoutte PM, Luscher TF. Serotonin and the blood vessel wall. J Hyperten. 1986;4:S29–S35. [PubMed] [Google Scholar]
- 4.Gaspar P, Lillesaar C. Probing the diversity of serotonin neurons. Philos Trans R Soc Lond B Biol Sci. 2012;367:2382–94. doi: 10.1098/rstb.2011.0378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.McQueen JK, Wilson H, Fink G. Estradiol-17 beta increases serotonin transporter (SERT) mRNA levels and the density of SERT-binding sites in female rat brain. Brain Res Mol Brain Res. 1997;45:13–23. doi: 10.1016/s0169-328x(96)00233-1. [DOI] [PubMed] [Google Scholar]
- 6.Sanchez MG, Morissette M, Di Paolo T. Oestradiol modulation of serotonin reuptake transporter and serotonin metabolism in the brain of monkeys. J Neuroendocrinol. 2013;25:560–9. doi: 10.1111/jne.12034. [DOI] [PubMed] [Google Scholar]
- 7.Lesch KP, Wolozin BL, Murphy DL, Reiderer P. Primary structure of the human platelet serotonin uptake site: identity with the brain serotonin transporter. J Neurochem. 1993;60:2319–22. doi: 10.1111/j.1471-4159.1993.tb03522.x. [DOI] [PubMed] [Google Scholar]
- 8.Yubero-Lahoz S, Robledo P, Farre M, de laTorre R. Platelet SERT as a peripheral biomarker of serotonergic neurotransmission in the central nervous system. Curr Med Chem. 2013;20:1382–96. doi: 10.2174/0929867311320110003. [DOI] [PubMed] [Google Scholar]
- 9.Janssen I, Powell LH, Matthews KA, et al. Depressive symptoms are related to progression of coronary calcium in midlife women: the Study of Women's Health Across the Nation (SWAN) Heart Study. Am Heart J. 2011;161:1186–91 e1. doi: 10.1016/j.ahj.2011.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bromberger JT, Schott LL, Kravitz HM, et al. Longitudinal change in reproductive hormones and depressive symptoms across the menopausal transition: results from the Study of Women's Health Across the Nation (SWAN) Arch Gen Psychiatry. 2010;67:598–607. doi: 10.1001/archgenpsychiatry.2010.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Deecher D, Andree TH, Sloan D, Schechter LE. From menarche to menopause: Exploring the underying biology of depression in women experiencing hormonal changes. Psychoneuroendocrinology. 2008;33:3–17. doi: 10.1016/j.psyneuen.2007.10.006. [DOI] [PubMed] [Google Scholar]
- 12.Osterlund MK. Underlying mechanisms mediating the antidepressant effects of estrogens. Biochim Biophys Acta. 2010;1800:1136–44. doi: 10.1016/j.bbagen.2009.11.001. [DOI] [PubMed] [Google Scholar]
- 13.Miller VM, Duckles SP. Vascular actions of estrogens: functional implications. Pharmacol Rev. 2008;60:210–41. doi: 10.1124/pr.107.08002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Park KE, Pepine CJ. Pathophysiologic mechanisms linking impaired cardiovascular health and neurologic dysfunction: the year in review. Cleve Clin J Med. 2010;77(Suppl 3):S40–5. doi: 10.3949/ccjm.77.s3.07. [DOI] [PubMed] [Google Scholar]
- 15.Guicheney P, Leger D, Barrat J, et al. Platelet serotonin content and plasma tryptophan in peri- and postmenopausal women: variations with plasma oestrogen levels and depressive symptoms. Eur J Clin Invest. 1988;18:297–304. doi: 10.1111/j.1365-2362.1988.tb01262.x. [DOI] [PubMed] [Google Scholar]
- 16.Flaumenhaft R, Nachtigall M, Lowenstein J, Nachtigall L, Nachtigall R. Association of oral but not transdermal estrogen therapy with enhanced platelet reactivity in a subset of postmenopausal women. Menopause. 2009;16:407–12. doi: 10.1097/gme.0b013e3181833886. [DOI] [PubMed] [Google Scholar]
- 17.Lippert TH, Filshie M, Muck AO, Seeger H, Zwirner M. Serotonin metabolite excretion after postmenopausal estradiol therapy. Maturitas. 1996;24:37–41. doi: 10.1016/0378-5122(95)00998-1. [DOI] [PubMed] [Google Scholar]
- 18.Wharton W, Gleason CE, Miller VM, Asthana S. Rationale and design of the Kronos Early Estrogen Prevention Study (KEEPS) and the KEEPS cognitive and affective sub study (KEEPS Cog) Brain Res. 2013;1514:12–7. doi: 10.1016/j.brainres.2013.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Harman SM, Brinton EA, Cedars M, et al. KEEPS: The Kronos Early Estrogen Prevention Study. Climacteric. 2005;8:3–12. doi: 10.1080/13697130500042417. [DOI] [PubMed] [Google Scholar]
- 20.Wharton W, Gleason CE, Dowling NM, et al. The KEEPS-Cognitive and Affective Study: Baseline Associations between Vascular Risk Factors and Cognition. J Alzheimers Dis. 2014;40:331–41. doi: 10.3233/JAD-130245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gleason CE, Dowling NM, Wharton W, et al. Effects of Hormone Therapy on Cognition and Mood in Recently Postmenopausal Women: Findings from the Randomized, Controlled KEEPS-Cognitive and Affective Study. PLoS medicine. 2015;12:e1001833. doi: 10.1371/journal.pmed.1001833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Miller VM, Hunter LW, Chu K, et al. Biologic nanoparticles and platelet reactivity. Nanomedicine. 2009;4:725–33. doi: 10.2217/nnm.09.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Curran SL, Andrykowski MA, Stutds JL. Short Form of the Profile of Mood States (POMS-SF): Psychometric information. Psychological Assessment. 7(1):80–3. [Google Scholar]
- 24.Jayachandran M, Litwiller RD, Lahr BD, et al. Alterations in Platelet Function and Cell-Derived Microvesicles in Recently Menopausal Women: Relationship to Metabolic Syndrome and Atherogenic Risk. J Cardiovasc Transl Res. 2011;4:811–22. doi: 10.1007/s12265-011-9296-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jayachandran M, Miller VM. Ovariectomy upregulates expression of estrogen receptors, NOS, and HSPs in porcine platelets. Am J Physiol:Heart Circ Physiol. 2002;283:H220–H6. doi: 10.1152/ajpheart.00950.2001. [DOI] [PubMed] [Google Scholar]
- 26.Hodis H, Mack W, Lobo R, et al. Estrogen in the Prevention of Atherosclerosis. a randomized, double-blind, placebo-controlled trial. Ann Intern Med. 2001;135:939–53. doi: 10.7326/0003-4819-135-11-200112040-00005. [DOI] [PubMed] [Google Scholar]
- 27.Mulvagh SL, Behrenbeck T, Lahr BA, et al. Endothelial function and cardiovascular risk stratification in menopausal women. Climacteric. 2010;13:45–54. doi: 10.3109/13697130902943287. [DOI] [PubMed] [Google Scholar]
- 28.Kling JM, Lahr B, Bailey K, Harman SM, Miller V, Mulvagh SL. Endothelial function in women of the Kronos Early Estrogen Prevention Study. Climacteric. 2015;18:1–11. doi: 10.3109/13697137.2014.986719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Alberti KG, Zimmet P, Shaw J. Metabolic syndrome--a new world-wide definition. A Consensus Statement from the International Diabetes Federation. Diabet Med. 2006;23:469–80. doi: 10.1111/j.1464-5491.2006.01858.x. [DOI] [PubMed] [Google Scholar]
- 30.Cao C, Han JH, Kim SZ, Cho KW, Kim SH. Diverse regulation of atrial natriuretic peptide secretion by serotonin receptor subtypes. Cardiovasc Res. 2003;59:360–8. doi: 10.1016/s0008-6363(03)00394-8. [DOI] [PubMed] [Google Scholar]
- 31.Greendale GA, Derby CA, Maki PM. Perimenopause and cognition. Obstet Gynecol Clin North Am. 2011;38:519–35. doi: 10.1016/j.ogc.2011.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.de Villiers TJ, Gass ML, Haines CJ, et al. Global Consensus Statement on menopausal hormone therapy. Maturitas. 2013;74:391–2. doi: 10.1016/j.maturitas.2013.02.001. [DOI] [PubMed] [Google Scholar]
- 33.Shors TJ, Leuner B. Estrogen-mediated effects on depression and memory formation in females. J Affect Disord. 2003;74:85–96. doi: 10.1016/s0165-0327(02)00428-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Moses-Kolko EL, Berga SL, Greer PJ, Smith G, Cidis Meltzer C, Drevets WC. Widespread increases of cortical serotonin type 2A receptor availability after hormone therapy in euthymic postmenopausal women. Fertil Steril. 2003;80:554–9. doi: 10.1016/s0015-0282(03)00973-7. [DOI] [PubMed] [Google Scholar]
- 35.Ladisich W. Influence of progesterone on serotonin metabolism: a possible causal factor for mood changes. Psychoneuroendocrinology. 1977;2:257–66. doi: 10.1016/0306-4530(77)90042-7. [DOI] [PubMed] [Google Scholar]
- 36.Miller VM. Estrogen metabolomics: A physiologists perspective. Hypertension. 2010;56:816–8. doi: 10.1161/HYPERTENSIONAHA.110.154385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mueck A, Gennazzani A, Samsioe G, Vukovic-Sysocki I, Seeger H. Low-dose continuous combinations of hormone therapy and biochemical surrogate markers for vascular tone and inflammation: transdermal versus oral application. Menopause. 2007;14:1–7. doi: 10.1097/gme.0b013e318054e2e7. [DOI] [PubMed] [Google Scholar]
- 38.Modder UI, Achenbach SJ, Amin S, Riggs BL, Melton LJ, 3rd, Khosla S. Relation of serum serotonin levels to bone density and structural parameters in women. J Bone Miner Res. 2010;25:415–22. doi: 10.1359/jbmr.090721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Blum I, Vered Y, Lifshitz A, et al. The effect of estrogen replacement therapy on plasma serotonin and catecholamines of postmenopausal women. Isr J Med Sci. 1996;32:1158–62. [PubMed] [Google Scholar]
- 40.Aedo AR, Landgren BM, Diczfalusy E. Pharmacokinetics and biotransformation of orally administered oestrone sulphate and oestradiol valerate in post-menopausal women. Maturitas. 1990;12:333–43. doi: 10.1016/0378-5122(90)90013-v. [DOI] [PubMed] [Google Scholar]
- 41.De Lignieres B, Basdevant A, Thomas G, et al. Biological effects of estradiol-17 beta in postmenopausal women: oral versus percutaneous administration. J Clin Endocrinol Metab. 1986;62:536–41. doi: 10.1210/jcem-62-3-536. [DOI] [PubMed] [Google Scholar]
- 42.Steiner M, Dunn E, Born L. Hormones and mood: from menarche to menopause and beyond. J Affect Disord. 2003;74:67–83. doi: 10.1016/s0165-0327(02)00432-9. [DOI] [PubMed] [Google Scholar]
- 43.Kopper NW, Gudeman J, Thompson DJ. Transdermal hormone therapy in postmenopausal women: a review of metabolic effects and drug delivery technologies. Drug Des Devel Ther. 2009;2:193–202. doi: 10.2147/dddt.s4146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Campagnoli C, Biglia N, Altare F, et al. Differential effects of oral conjugated estrogens and transdermal estradiol on insulin-like growth factor 1, growth hormone and sex hormone binding globulin serum levels. Gynecol Endocrinol. 1993;7:251–8. doi: 10.3109/09513599309152509. [DOI] [PubMed] [Google Scholar]
- 45.Nobler MS, Mann JJ, Sackeim HA. Serotonin, cerebral blood flow, and cerebral metabolic rate in geriatric major depression and normal aging. Brain Res Brain Res Rev. 1999;30:250–63. doi: 10.1016/s0165-0173(99)00019-3. [DOI] [PubMed] [Google Scholar]
- 46.Houston DS, Shepherd JT, Vanhoutte PM. Adenine nucleotides, serotonin, and endothelium-dependent relaxations to platelets. Am J Physiol. 1985;248:H389–H95. doi: 10.1152/ajpheart.1985.248.3.H389. [DOI] [PubMed] [Google Scholar]
- 47.Miller VM, Lahr BD, Bailey KR, Heit JA, Harman SM, Jayachandran M. Longitudinal effects of menopausal hormone treatments on platelet characteristics and cell-derived microvesicles. Platelets. 2015:1–11. doi: 10.3109/09537104.2015.1023273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Harman SM, Black DM, Naftolin F, et al. Arterial imaging outcomes and cardiovascular risk factors in recently menopausal women: A randomized trial. Ann Intern Med. 2014;161:249–60. doi: 10.7326/M14-0353. [DOI] [PubMed] [Google Scholar]
- 49.Miller V, Jenkins G, Biernacka J, et al. Pharmacogenomics of estrogens on changes in carotid artery intima-medial thickness and coronary arterial calcification Kronos Early Estrogen Prevention Study. Physiol Genomics. 2015:25. doi: 10.1152/physiolgenomics.00029.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Schmidt PJ, Nieman L, Danaceau MA, et al. Estrogen replacement in perimenopause-related depression: a preliminary report. Am J Obstet Gynecol. 2000;183:414–20. doi: 10.1067/mob.2000.106004. [DOI] [PubMed] [Google Scholar]
- 51.Dworkin RH, Turk DC, Wyrwich KW, et al. Interpreting the clinical importance of treatment outcomes in chronic pain clinical trials: IMMPACT recommendations. J Pain. 2008;9:105–21. doi: 10.1016/j.jpain.2007.09.005. [DOI] [PubMed] [Google Scholar]
- 52.Sumner BE, Grant KE, Rosie R, Hegele-Hartung C, Fritzemeier KH, Fink G. Effects of tamoxifen on serotonin transporter and 5-hydroxytryptamine(2A) receptor binding sites and mRNA levels in the brain of ovariectomized rats with or without acute estradiol replacement. Brain Res Mol Brain Res. 1999;73:119–28. doi: 10.1016/s0169-328x(99)00243-0. [DOI] [PubMed] [Google Scholar]
- 53.Rubinow DR, Schmidt PJ, Roca CA. Estrogen-serotonin interactions: implications for affective regulation. Biol Psychiatry. 1998;44:839–50. doi: 10.1016/s0006-3223(98)00162-0. [DOI] [PubMed] [Google Scholar]
- 54.Senn S. Change from baseline and analysis of covariance revisited. Stat Med. 2006;25:4334–44. doi: 10.1002/sim.2682. [DOI] [PubMed] [Google Scholar]
- 55.Miller VM, Lewis DA, Barber DA. Gender differences and endothelium- and platelet-derived factors in the coronary circulation. Clin Exp Pharmacol Physiol. 1999;26:132–6. doi: 10.1046/j.1440-1681.1999.02997.x. [DOI] [PubMed] [Google Scholar]