This study tested the hypothesis that an ex vivo T-cell suppression assay could estimate response to novel cell-based therapy for rheumatoid arthritis (RA). Results showed multipotent adult progenitor cell products suppressed RA effector T cells. The study demonstrated the feasibility of using suppressor assays to detect biological effects of cell-based therapy in RA and suggests these effects are dose-dependent.
Keywords: Arthritis, Rheumatoid, Autoimmune diseases, Biological markers, Mesenchymal stromal cells, T lymphocytes
Abstract
Cell-based therapy has potential therapeutic value in autoimmune diseases such as rheumatoid arthritis (RA). In RA, reduction of disease activity has been associated with improvement in the function of regulatory T cells (Treg) and attenuated responses of proinflammatory effector T cells (Teff). Mesenchymal stem cells (MSCs) and related multipotent adult progenitor cells (MAPC) have strong anti-inflammatory and immunomodulatory properties and may be able to “reset” the immune system to a pre-RA state. MAPC are MSC-like cells that are slightly earlier in lineage, have greater expansion capacity, and can be used as “off-the-shelf” therapy. Assessment of cell-based therapy to treat arthritis and related diseases is limited by the lack of available biological correlates that can be measured early on and indicate treatment response. We set out to develop a functional measure that could be used ex vivo as a biomarker of response. We were able to demonstrate that MAPC products could inhibit Teff responses from patients with active RA and that Treg from RA patients suppressed Teff. This assay used ex vivo can be used with MAPC or Treg alone or in combination and reflects the overall level of Teff suppression. Use of a novel functional biomarker as an exploratory endpoint in trials of cell-based therapy should be of value to detect biological outcomes at a point prior to the time that clinical response might be observed.
Significance
Therapy with mesenchymal stem cells and related multipotent adult progenitor cells is immune modifying in a variety of diseases. There is interest in using cell-based therapy in rheumatoid arthritis (RA) to induce tolerance and “reset” the immune system to its pre-RA state. In a clinical trial, it should be known as soon as possible if there is a chance of response. A biomarker has been developed that permits measurement of the effects of cell-based therapy on effector T cell function.
Introduction
Rheumatoid arthritis (RA) is a chronic autoimmune disease for which major advances in etiopathogenesis and therapy have occurred during the past two decades. The presence of disease-associated auto-antibodies (rheumatoid factor and anti-citrullinated peptide antibodies), referred to as seropositive RA, confers high risk for joint damage and work disability. In seropositive RA, breaks in B-cell tolerance also appear to be accompanied by breaks in T-cell tolerance; in systemic lupus erythematosus, B-cell depletion may improve T-regulatory cell (Treg) numbers and function [1]. Peripheral blood Treg function is attenuated in active RA and may be restored, at least in part, by effective therapy [2]. For patients with seropositive RA, therapy is generally lifelong and has side effects including risk for infection; disease remission is rarely sustained once disease-modifying antirheumatic drugs (DMARDs) or biological DMARDs are withdrawn. Most agents do not restore “immune tolerance,” although there is some evidence that therapy-induced clinical response is accompanied by increased Treg number and, in some cases, improvements in Treg function [2, 3].
Mesenchymal stem cells (MSCs), usually obtained from bone marrow of healthy adults, were originally described according to their adherence to plastic and capacity to differentiate into skeletal tissue (cartilage and bone), skin, and fat [4]. A major therapeutic effect of MSCs lies in their capacity to respond to immune derangement and restore immune homeostasis [5]. Use of adult adherent cell therapy with MSCs is an emerging therapeutic strategy promising for a number of human diseases and, unlike hematopoietic stem cell therapy, can be used without prior bone marrow ablation. Multipotent adult progenitor cells (MAPCs) appear to be an earlier lineage cell type than MSCs and possess greater proliferation potential than MSCs, although MAPCs appear to have the same or greater anti-inflammatory and immune modulatory capacity compared with MSCs. MAPCs have greater expansion capacity and can be used as “off-the-shelf” therapy. MAPCs have improved disease in several preclinical animal models, including models of ischemic stroke, traumatic brain injury, spinal cord injury, acute myocardial infarct, solid organ transplant, and graft-versus-host disease [6–10].
An obstacle to evaluating the therapeutic utility of cell-based therapies in autoimmune disease(s) is the lack of identified biomarkers that precede and predict clinical response. We hypothesized that early in RA, a window of opportunity exists during which therapeutically delivered MSCs/MAPCs can induce “immune tolerance,” thus “resetting” immune homeostasis to a pre-RA state. This state is as close to cure as is currently possible. A specific goal of our studies was to develop a functional biomarker that would predict response to cell-based therapy in RA.
We aimed to (a) determine whether medium conditioned via culture on MAPCs (CCM) could inhibit effector T-cell (Teff) responses of patients with active RA, (b) determine whether Tregs from patients with RA suppressed Teffs, and (c) determine the potential for using this suppression assay as a functional biomarker in clinical trials of cell-based therapy.
Material and Methods
The Human Use Committee at MetroHealth Medical Center approved our studies. Seropositive patients with active RA (disease activity 28 scores: 4.5–5.8) starting a DMARD or biological DMARD were included. Control assays were performed using blood from healthy donors (HD). MAPCs were obtained from a bone marrow isolate (Lonza, Walkersville, MD, http://www.lonza.com) and then cultured as described [11] (supplemental online data). Peripheral blood T cells were isolated and separated into T-regulatory CD4+CD25hi cells and Effector CD4+CD25lo T cells by flow cytometry. Teffs were stimulated by cross-linking CD2/CD3/CD28 (Miltenyi Biotec, Auburn, CA, http://www.miltenyibiotec.com) in the presence of various amounts or combinations of CCM and Tregs. Tregs and Teffs were used from single donors or patients. Proliferation rates were measured using eFluor670 far-red vital dye (eBioscience, San Diego, CA, http://www.ebioscience.com), and suppression indices calculated [12].
Results and Discussion
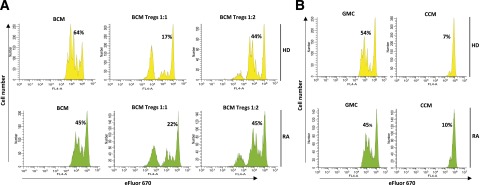
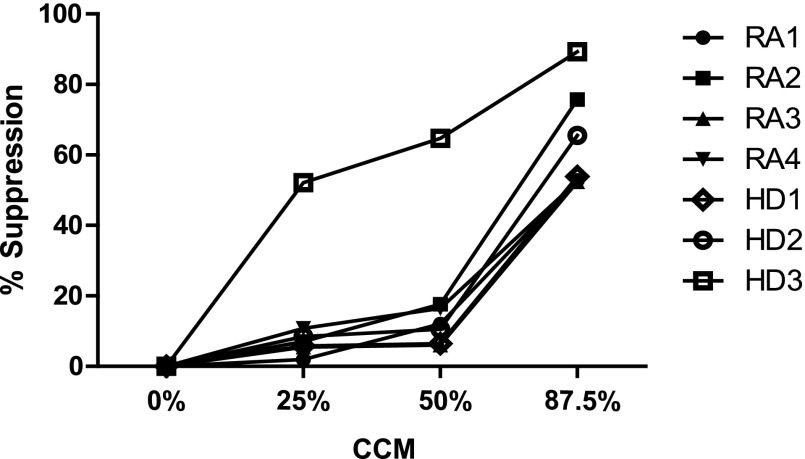
To characterize the effects of MAPC products on Teff proliferation or suppression, Teffs were stimulated using anti-CD2, -3, and -28 antibody beads. Teff from HD and RA proliferated and were suppressed by Tregs (Fig. 1A). HD and RA Teffs were both suppressed when cultured in CCM (Fig. 1B). Proliferation rates that dropped to 7% for HD and 10% for RA were observed in the presence of CCM. This correlated with 87% suppression of Teff proliferation in HD and 78% suppression in RA. Because might be anticipated for a drug, the effects of CCM on Teffs were dose dependent with concentrations from 12.5% to 87.5% of CCM tested. The greatest effects of CCM were observed at concentrations of CCM greater than 50% by volume (Fig. 2).
Figure 1.
Multipotent adult progenitor cells (MAPCs) suppressed active RA and HD effector T-cell (Teff) response. CD4+ cells were isolated from peripheral blood mononuclear cells from HD and patients with RA. Cells were stained and then sorted into Teff and Treg populations based on CD4 and CD25 expression. Teffs were stimulated with anti-CD2/CD3/CD28-coated beads and then loaded with eFluor670 vital dye. Teff proliferation was measured using flow cytometry. (A): T-cell suppression assays were performed in the presence of BCM with or without Tregs at ratios of 1:1 and 1:2. (B): T-cell suppression assays were performed with CCM or GMC. Abbreviations: BCM, basal medium; CCM, multipotent adult progenitor cell-conditioned medium; GMC, control growth medium; HD, blood from healthy donors; RA, rheumatoid arthritis patient; Treg, T-regulatory cell.
Figure 2.
Effector T cells are suppressed by multipotent adult progenitor cell (MAPC)-conditioned medium in a dose-dependent manner in RA and HD. MAPCs were grown in serum-free MAPC medium that contains tumor necrosis factor-α (10 ng/ml), interleukin-1β (10 ng/ml), and an equivalent amount of interferon (IFN) γ. After 3 days IFNγ was immunodepleted. Control growth medium was used as a control for CCM; it is the initial medium but has not been exposed to MAPCs. CCM concentrations between 12.5% and 87.5% of the total medium were tested. Suppression was observed at CCM concentrations of greater than 50% by volume. Abbreviations: CCM, multipotent adult progenitor cell-conditioned medium; HD1–HD3, blood from healthy donors specific to this experiment; RA1–RA4, distinct rheumatoid arthritis patients specific to this experiment.
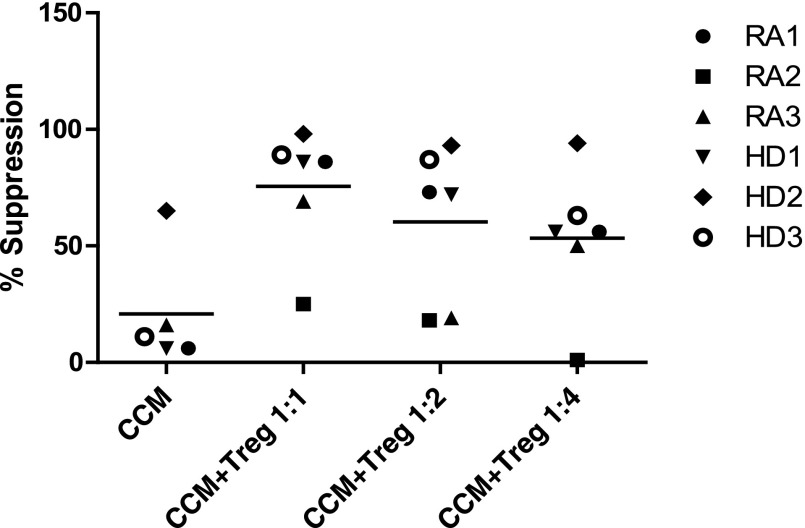
We further hypothesized that in vivo, MAPCs and MSCs might act directly and indirectly on Teffs by also improving Treg function. To determine whether CCM improved Treg function, we performed suppression assays using 50% CCM, a concentration that suboptimally suppressed Teffs (Fig. 2). Using CCM alone, suppression of Teffs (mean ± SEM) was 59% ± 6% (Fig. 3). Addition of Tregs increased suppression to 90% ± 3% (1:1 Tregs to Teffs), 84% ± 3% (1:2 Tregs to Teffs), and 71% ± 6% (1:4 Tregs to Teffs).
Figure 3.
CCM improved Treg function. Suppression assays of effector T cells (Teffs) using combination of 50% by volume CCM and Tregs at ratios of Teff to Tregs varying from 1:1 to 1:4. Bars represent the mean values. Abbreviations: CCM, multipotent adult progenitor cell-conditioned medium; HD1–HD3, healthy donors specific to each experiment; RA1–RA4, distinct rheumatoid arthritis patients specific to this experiment; Treg, T-regulatory cell.
The major findings of our investigation are that (a) products secreted into culture supernatant from MAPCs (i.e., CCM) suppressed Teff responses from active RA and HD, (b) CCM suppressed Teffs in a dose-dependent fashion, and (c) there was a trend toward combinations of CCM (50% by volume) and RA Tregs suppressing RA Teffs beyond that of CCM alone. The main focus of our work was to test the hypothesis that an ex vivo T-cell suppression assay could estimate response to novel cell-based therapy. Suppression of Teffs by soluble MAPC products has previously pointed toward indoleamine 2,3-dioxygenase being the major factor in contact-dependent suppression of Teffs [11]. To our knowledge, our assays are the first to demonstrate that MAPC products suppress Teffs in a system that lacks professional antigen-presenting cells and thereby lacks replenishment of cytokines and related secreted products. This is also the first study, to our knowledge, that demonstrates suppression specifically of RA Teffs by MAPC products. Importantly, this study demonstrated the feasibility of using suppressor assays to detect biological effects of cell-based therapy in RA and our ex vivo data suggest that these effects are dose dependent. Our hypothesis is that tolerance, an active state in which Teff have attenuated responses to specific antigen but are not globally suppressed, may be induced by MAPCs delivered therapeutically.
In active RA, derangement in Treg function has been reported, but controversy exists because there are also reports of normal Treg function in active RA. MSCs from aging and diseased individuals are also prematurely senescent [13], and RA MSCs display defects in their capacity to inhibit Th17-cell polarization [14]. Methotrexate, a conventional DMARD, has recently been demonstrated to improve Treg function through demethylation of the FoxP3 promoter [15, 16]. These data, combined with our ability to optimize measurement of a functional biomarker across applications and diseases, makes our study of the effects of allogeneic MAPCs relevant to future therapeutic applications. Our data are compatible with our hypothesis that MAPC-dependent effects on Teffs and MAPC-dependent effects on regulatory cell populations may be synergistic.
We propose that our approach of using a functional suppressor ex vivo in clinical trials is vital to understanding early in a trial if cell-based therapy is having a biological effect and the nature of that effect on the effector and regulatory compartments. This system can also be modified to measure other critical Teff function(s), such as cytokine production, that are also known to be regulated by MAPCs or MSCs.
Conclusion
Taken together, our data suggest that early studies of cell-based therapy in RA can and should include functional measures of immune status. Longitudinal use of this assay, proposed as a “functional biomarker” before and after cell-based therapy, should give us a more complete understanding of when and how cell-based therapy can be used to redirect the immune system to its pre-RA status.
Supplementary Material
Acknowledgments
This study was supported in part by R01AR056273 and the CWRU Clinical and Translational Science Award Grant No. UL1TR000439, a component of the National Institutes of Health (NIH) and NIH roadmap for Medical Research. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIH.
Author Contributions
G.S.-M.: collection and/or assembly of data, data analysis and interpretation; D.C.S.: collection and/or assembly of data; A.E.T. and D.D.A. conception and design, data analysis and interpretation, final approval of manuscript; N.A.L.: collection and/or assembly of data; E.B.: conception and design, collection and/or assembly of data, final approval of manuscript; T.S.M.: conception and design; H.M.L. and A.I.C.: conception and design, final approval of manuscript; M.B.: data analysis and interpretation, manuscript writing, final approval of manuscript; N.G.S.: conception and design, provision of study material or patients, data analysis and interpretation, manuscript writing, final approval of manuscript.
Disclosure of Potential Conflicts of Interest
A.E.T. and N.A.L. are compensated employees of Athersys, Inc. A.I.C. has compensated intellectual property rights through Case Western Reserve University and is an uncompensated consultant. The other authors indicated no potential conflicts of interest.
References
- 1.Roccatello D, Sciascia S, Baldovino S, et al. A 4-year observation in lupus nephritis patients treated with an intensified B-lymphocyte depletion without immunosuppressive maintenance treatment-Clinical response compared to literature and immunological re-assessment. Autoimmun Rev. 2015;14:1123–1130. doi: 10.1016/j.autrev.2015.07.017. [DOI] [PubMed] [Google Scholar]
- 2.Valencia X, Stephens G, Goldbach-Mansky R, et al. TNF downmodulates the function of human CD4+CD25hi T-regulatory cells. Blood. 2006;108:253–261. doi: 10.1182/blood-2005-11-4567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Emery P, Burmester GR, Bykerk VP, et al. Evaluating drug-free remission with abatacept in early rheumatoid arthritis: results from the phase 3b, multicentre, randomised, active-controlled AVERT study of 24 months, with a 12-month, double-blind treatment period. Ann Rheum Dis. 2015;74:19–26. doi: 10.1136/annrheumdis-2014-206106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Pittenger MF, Mackay AM, Beck SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–147. doi: 10.1126/science.284.5411.143. [DOI] [PubMed] [Google Scholar]
- 5.Prockop DJ, Oh JY. Mesenchymal stem/stromal cells (MSCs): Role as guardians of inflammation. Mol Ther. 2012;20:14–20. doi: 10.1038/mt.2011.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pelacho B, Nakamura Y, Zhang J, et al. Multipotent adult progenitor cell transplantation increases vascularity and improves left ventricular function after myocardial infarction. J Tissue Eng Regen Med. 2007;1:51–59. doi: 10.1002/term.7. [DOI] [PubMed] [Google Scholar]
- 7.Franquesa M, Hoogduijn MJ, Reinders ME, et al. Mesenchymal Stem Cells in Solid Organ Transplantation (MiSOT) Fourth Meeting: Lessons learned from first clinical trials [published correction appears in Transplantation 2013;96:e49] Transplantation. 2013;96:234–238. doi: 10.1097/TP.0b013e318298f9fa. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Vaes B, Van’t Hof W, Deans R, et al. Application of MultiStem(®) allogeneic cells for immunomodulatory therapy: clinical progress and pre-clinical challenges in prophylaxis for graft versus host disease. Front Immunol. 2012;3:345. doi: 10.3389/fimmu.2012.00345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Walker PA, Bedi SS, Shah SK, et al. Intravenous multipotent adult progenitor cell therapy after traumatic brain injury: Modulation of the resident microglia population. J Neuroinflammation. 2012;9:228. doi: 10.1186/1742-2094-9-228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hess DC, Sila CA, Furlan AJ, et al. A double-blind placebo-controlled clinical evaluation of MultiStem for the treatment of ischemic stroke. Int J Stroke. 2014;9:381–386. doi: 10.1111/ijs.12065. [DOI] [PubMed] [Google Scholar]
- 11.Reading JL, Yang JH, Sabbah S, et al. Clinical-grade multipotent adult progenitor cells durably control pathogenic T cell responses in human models of transplantation and autoimmunity. J Immunol. 2013;190:4542–4552. doi: 10.4049/jimmunol.1202710. [DOI] [PubMed] [Google Scholar]
- 12.McMurchy AN, Levings MK. Suppression assays with human T regulatory cells: A technical guide. Eur J Immunol. 2012;42:27–34. doi: 10.1002/eji.201141651. [DOI] [PubMed] [Google Scholar]
- 13.Lepperdinger G, Brunauer R, Gassner R, et al. Changes of the functional capacity of mesenchymal stem cells due to aging or age-associated disease - implications for clinical applications and donor recruitment. Transfus Med Hemother. 2008;35:299–305. doi: 10.1159/000142373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sun Y, Deng W, Geng L, et al. Mesenchymal stem cells from patients with rheumatoid arthritis display impaired function in inhibiting Th17 cells. J Immunol Res. 2015;2015:284215. doi: 10.1155/2015/284215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cribbs AP, Kennedy A, Penn H, et al. Treg cell function in rheumatoid arthritis is compromised by ctla-4 promoter methylation resulting in a failure to activate the indoleamine 2,3-dioxygenase pathway. Arthritis Rheumatol. 2014;66:2344–2354. doi: 10.1002/art.38715. [DOI] [PubMed] [Google Scholar]
- 16.Bending D, Pesenacker AM, Ursu S, et al. Hypomethylation at the regulatory T cell-specific demethylated region in CD25hi T cells is decoupled from FOXP3 expression at the inflamed site in childhood arthritis. J Immunol. 2014;193:2699–2708. doi: 10.4049/jimmunol.1400599. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.