Abstract
Objective
To study the natural history, prognosticators, and outcomes in patients with RCC with extension of tumor beyond Gerota’s fascia and/or invading contiguously into the adrenal gland (pT4).
Patients and Methods
From 1992 to 2012, we identified 61 patients who underwent radical nephrectomy and were found to have pT4 disease. Clinicopathological variables were queried using univariate analysis to identify relevant prognostic variables. Cox proportional hazards model was used for multivariate analysis of predictors of cancer-specific survival. Survival plots were estimated using Kaplan-Meier method and survival analysis using log-rank test.
Results
Median age was 56 years (IQR 49-64) and 49 (81.7%) had ECOG PS 0-1. At diagnosis, 22 (36.1%) patients were M0 and 39 (63.9%) were M1. Forty-nine(80.3%) patients had clear cell RCC. Twenty-four(39.3%) had sarcomatoid features. Thirty-nine(69.6%) patients had Fuhrman grade 3-4. There were twenty-six(42.6%) pN0 patients, 16(26.2%) pN1, and 19(31.1%) pNx. Median CSS was 37 months for M0 and 8 months for M1 patients. On multivariate analysis, preoperative LDH and alkaline phosphatase, M stage, pN stage, and sarcomatoid dedifferentiation were significantly associated with survival.
Conclusions
Survival in patients with pT4 remains poor. pT4 is associated with a locally and regionally invasive biology that requires specific attention and warrants careful study. Understanding the drivers of this unique phenotype will generate therapeutic interventions that can change the behavior of these uniquely aggressive tumors.
Keywords: T4, renal cell carcinoma, predictors, nephrectomy, surgery
INTRODUCTION
In 2015, 61,560 patients are estimated to be diagnosed with tumors of the kidney and renal pelvis in the United States, with 14,080 expected deaths [1], with renal cell carcinoma (RCC) being the most common tumor. The stage distribution of RCC has migrated due to the widespread use of abdominal imaging and now over half are diagnosed as stage I. However, approximately 20% will be diagnosed with stage IV disease [2] and large retrospective studies estimate the incidence of pT4 disease between 5-15% [3, 4]. This subset of patients demonstrate poor outcomes with a 5-year survival rate of 20-30% and an estimated 10-year cancer specific survival rate (CSS) of 12% [5, 6]. Upfront aggressive surgery remains the standard of care for localized and locally advanced RCC and a cornerstone for the multimodal treatment of metastatic RCC [7, 8]. Nevertheless, tumors with adjacent organ involvement pose a challenging scenario even for experienced surgeons. These tumors are often associated with aggressive features such as Fuhrman grade 3 or 4, sarcomatoid dedifferentiation and lymphovascular invasion. Although several studies have shown promising results with neoadjuvant therapy in the setting of large surgical tumor burden [9-11], such pathologic features ultimately result in an intrinsically aggressive behavior with adjacent organ invasion, potentially leading to “unresectable” disease in spite of the best targeted therapies available.
To our knowledge, there are no studies specifically investigating pT4 RCC patients regardless of M stage. Moreover, there is paucity of data on the natural history of this high-risk population and the prognostic factors for survival. Our objective was therefore to study outcomes and prognostic variables (using preoperative and postoperative models) associated with survival in patients with pT4N0-1M0-1 RCC treated with radical nephrectomy.
PATIENTS AND METHODS
Study Population
This study was approved by the Institutional Review Board of The University of Texas MD Anderson Cancer Center. A retrospective chart review was performed from an institutional kidney cancer database of patients diagnosed with RCC who underwent radical nephrectomy at our institution from July 1992 to January 2012. We included patients with pT4 disease as defined by the 2010 TNM staging system [12]. As such, direct adrenal invasion formerly classified as pT3a was reassigned to pT4. Patients were included in this retrospective study if they underwent radical nephrectomy and were found to have pT4 on final pathology.
Demographics including age, gender, BMI, ECOG PS, smoking history, and relevant symptoms were collected. Local symptoms were defined as flank pain or hematuria and systemic symptoms were defined as fever, night sweats, weight loss >10 pounds or malaise. Palpable abdominal mass reported during preoperative physical examination in the ipsilateral flank region consistent with a renal mass was recorded. All patients underwent a metastatic evaluation including one chest computed tomography (CT) scan or chest X-ray (CXR), cross sectional abdominal imaging, and if indicated bone scan or brain imaging. Clinical staging was based on the 2010 AJCC TNM staging system [12]. A dedicated genitourinary pathologist reviewed all available pathology slides. Tumor grade was determined using the Fuhrman grading system and tumor histology was reported according to the 2004 WHO criteria [13]. Preoperative laboratory values such as lactate dehydrogenase (LDH), alkaline phosphatase, hemoglobin and eGFR were registered and characterized either normal or abnormal based on standard institutional laboratory values. Surgical characteristics for all patients such as estimated blood loss (EBL) and surgery duration were recorded. Systemic therapy administration and timing were at the discretion of the treating physician and were not standardized.
Study Design
The primary objective of the study was to assess the outcomes of patients with RCC and adjacent organ invasion treated with radical nephrectomy. Our secondary objective was to establish predictors of cancer-specific survival in this cohort of patients.
Data Analysis
Cancer-specific survival (CSS) was measured in months and defined as the time from date of nephrectomy to death of disease or last follow up. The distribution of each categorical variable was summarized in terms of frequencies and percentages, and continuous nonparametric variables were summarized by median and interquartile range (IQR). Univariate (UVA) Cox proportional hazard regression model was performed to determine clinical and pathological features associated with disease-specific death. From the existing covariates with statistical significance (P<0.05) on UVA, we further developed two multivariate models to predict CSS after RN based on widely available preoperative and postoperative variables. Using the stepwise model selection, which requires two significance levels: one for adding variables and one for removing variables, we systematically examined all significant univariate variables and identified based on multivariate analysis (MVA), two separate models of preoperative and postoperative predictors of survival in pT4 RCC patients. Survival curves were estimated according to Kaplan-Meier methods and were compared using the log rank test. All analyses were carried out in SAS 9.3 (SAS Institute Inc, Cary, NC).
RESULTS
Clinical and pathological characteristics
This study included 61 patients diagnosed with pT4 renal cell carcinoma treated with radical nephrectomy at our institution. Out of the 61 patients, 51 presented with single-organ invasion and 10 with multiple-organ invasion. Patients were classified as pT4 due to invasion of adrenal gland in 29, liver in 11, pancreas in 10, colon in 8, small bowel in 4, spleen in 3, mesentery in 3, and muscle in 2. Table 1 shows the baseline patient demographic and clinical characteristics. Median age was 56 years (IQR 49-64), with 42 males (68.9%) and 49 (81.7%) with ECOG PS 0-1 at presentation. Thirty-six (59%) patients presented with at least one relevant local symptom. Forty-two (68.9%) presented with systemic symptoms. On preoperative assessment, 42 patients (68.9%) were categorized as clinical T4, 32 (52.5%) as clinical N1 (median number of lymph nodes removed was 6.5, IQR 2-11), and 39 (63.9%) as clinical M1 at the time of diagnosis. Fourteen (23%) patients received neoadjuvant systemic therapy (at the discretion of the treating physicians): sunitinib (n=7), IL-2/interferon/5-FU (n=3), capecitabine/gemcitabine (n=2), bevacizumab (n=1), and 5-FU/adriamycin/cisplatin (n=1). 42(75%) patients received salvage systemic therapy.
Table 1.
Demographics and clinical characteristics in 61 patients with pathological T4 RCC treated with RN
| N (%) or Median (IQR) | |
|---|---|
| All patients | 61 (100) |
| Age, years | 56 (49-64) |
|
Gender
Female Male |
19 (31.1) 42 (68.9) |
|
Race
White African American Hispanic |
49 (80.3) 6 (9.8) 6 (9.8) |
| BMI, kg/m2 | 28 (22- 34) |
|
ECOG performance status
0 1 2 3 |
13 (21.6) 36 (60) 10 (16.7) 1 (1.7) |
|
Local symptoms
Yes No |
36 (59) 25 (41) |
|
Systemic symptoms
Yes No |
42 (68.9) 19 (31.1) |
|
Clinical (Preoperative) T stage
<T4 T4 |
19 (31.1) 42 (68.9) |
|
Clinical (Preoperative) N stage
N0 N1 |
29 (47.5) 32 (52.5) |
|
Clinical (Preoperative) M stage
M0 M1 |
22 (36.1) 39 (63.9) |
|
Hemoglobin
Normal Abnormal |
8 (13.1) 53 (86.9) |
|
LDH
Normal Abnormal |
40 (71.4) 16 (28.6) |
|
Alkaline phosphatase
Normal Abnormal |
35 (61.4) 22 (38.6) |
|
Estimated Glomerular Filtration Rate
>60 <60 |
35 (64.8) 19 (35.2) |
|
Neoadjuvant Systemic Therapy
Yes No |
14 (23) 47 (77) |
* Numbers do not always add to 61 due to missing values
Median OR time was 224.5 minutes (IQR 137 – 354.5), and median EBL was 1450 ml (IQR 800 – 2200). Table 2 shows surgical and pathological characteristics. All patients underwent open radical surgery. Median tumor size was 13cm (IQR 10-16). Forty-seven patients (77.0%) had clear cell RCC, 39 (69.6%) had Fuhrman grade 4, and sarcomatoid dedifferentiation were present in 24 (39.3%). Twenty-six (42.6%), 16 (26.2%), and 19 (31.1%) patients were pN0, pN1, and pNx, respectively. Five patients had Clavien grade 3 and higher complications (3 grade 3a, 1 grade 3b, 1 grade 5).
Table 2.
Surgical and pathological characteristics in 61 patients with pathological T4 RCC treated with RN
| N (%) or Median (IQR) | |
|---|---|
| All patients | 61 (100) |
| Tumor diameter at nephrectomy, cm | 13 (10 - 16) |
|
Pathological N Stage
N0 N1 Nx |
26 (42.6) 16 (26.2) 19 (31.1) |
|
Histology
Clear Cell Non-Clear Cell |
47 (77.0) 14 (23.0) |
|
Fuhrman grade
1 2 3 4 |
0 (0) 3 (5.4) 14 (25) 39 (69.6) |
|
Sarcomatoid dedifferentiation
Yes No |
24 (39.3) 37 (60.7) |
|
Necrosis
Yes No |
45 (78.9) 12 (21.1) |
|
Lymphovascular Invasion
Yes No |
16 (26.7) 44 (73.3) |
|
IVC thrombectomy
Yes No |
11 (18) 50 (82) |
|
Direct adrenal extension
Yes No |
29 (50.9) 28 (49.1) |
|
Positive surgical margin
Yes No |
10 (16.3) 51 (83.7) |
* Numbers do not always add to 61 due to missing values
Survival
Seventeen patients (27.8%) were NED after surgery. Of these, 11 (64.7%) later recurred: seven patients had distant recurrence, two had only local recurrence and two presented with both. The median cancer specific survival (CSS) for the entire cohort was 10 months (95% CI: 8.7-17.2). At a median follow-up of 12 months (IQR 5.25 – 41.5 months) from time of diagnosis we censored 45 (72.6%) cancer-related deaths. Of the patients who were alive at last follow-up, 5 were NED and 11 were with disease.
UVA Cox proportional hazards regression analyses revealed that systemic symptoms, preoperative LDH and alkaline phosphatase, M stage at diagnosis, Fuhrman grade, histologic subtype, sarcomatoid dedifferentiation, and pN stage were associated with CSS (Table 3).
Table 3.
Univariate Cox Regression Analysis of Clinical and Pathological Features for the prediction of Cancer Specific Survival in 61 patients with pT4 RCC treated with RN
| Univariate | ||
|---|---|---|
| HR (95% CI) | P Value | |
| Variables | ||
| ECOG Performance Status (PS 2-3 versus 0-1) | 1.70 (0.85-3.38) | 0.13 |
| Systemic Symptoms (Yes versus No) | 2.78 (1.30-5.94) | 0.01 |
| M stage (M1 versus M0) | 2.48 (1.26-4.89) | 0.01 |
| Histology (Clear Cell versus Non-Clear Cell) | 2.41 (1.24-4.66) | 0.01 |
| Sarcomatoid (Yes versus No) | 2.67 (1.42-5.02) | <0.01 |
| Fuhrman grade (Grade 3-4 versus 1-2) | 2.59 (1.21-5.57) | 0.01 |
| Positive surgical margin (Yes versus No) | 1.54 (0.73-3.26) | 0.26 |
| Abnormal Hemoglobin (Yes versus No) | 1.71 (0.61-4.78) | 0.31 |
| Abnormal Corrected Calcium (Yes versus No) | 1.49 (0.77-2.88) | 0.24 |
| Abnormal LDH (Yes versus No) | 2.72 (1.39-5.33) | <0.01 |
| Abnormal Alkaline Phosphatase (Yes versus No) | 3.26 (1.72-6.18) | <0.01 |
| Pathological Nodal status N1 versus N0 Nx versus N0 |
5.38 (2.36-12.26) 2.52 (1.19-5.35) |
<0.01 <0.01 0.02 |
We then developed a preoperative model for individualized prediction of CSS based on significant UVA covariates. MVA revealed that LDH and alkaline phosphatase remained significant predictors of survival. A postoperative model was also developed, and LDH, alkaline phosphatase, as well as M stage at diagnosis, pN stage and sarcomatoid dedifferentiation were significant predictors of survival (Tables 4 and 5).
Table 4.
Multivariate Cox regression analysis of Clinical Features for Preoperative Prediction of Cancer Specific Survival in 61 patients with pT4 RCC treated with RN
| Multivariate | ||
|---|---|---|
| HR (95% CI) | P Value | |
| Variables | ||
| Clinical (Preoperative) N stage at diagnosis (N1 versus N0) |
0.63 (0.33 – 1.22) | 0.17 |
| M stage at diagnosis (M1 versus M0) | 1.77 (0.81 – 3.85) | 0.15 |
| Abnormal LDH (Yes versus No) | 3.97 (1.87 – 8.43) | <0.01 |
| Abnormal Alkaline Phosphatase (Yes versus No) | 2.92 (1.47 – 5.80) | <0.01 |
Table 5.
Multivariate Cox regression analysis of Clinical and Pathological Features for the Postoperative Prediction of Cancer Specific Survival in 61 patients with pT4 RCC treated with RN
| Multivariate | ||
|---|---|---|
| HR (95% CI) | P Value | |
| Variables | ||
| Sarcomatoid dedifferentiation (Yes versus No) | 3.11 (1.51- 6.42) | <0.01 |
| Pathological N Stage pN1 versus pN0 pNx versus pN0 |
5.16 (1.94 - 13.69) 2.58 (1.15 – 5.79) |
<0.01 0.02 |
| M stage (M1 versus M0) | 2.24 (1.01 – 4.98) | 0.05 |
| Abnormal LDH (Yes versus No) | 2.32 (1.04 – 5.17) | 0.04 |
| Abnormal Alkaline Phosphatase (Yes versus No) | 4.29 (2.04 – 9.00) | <0.01 |
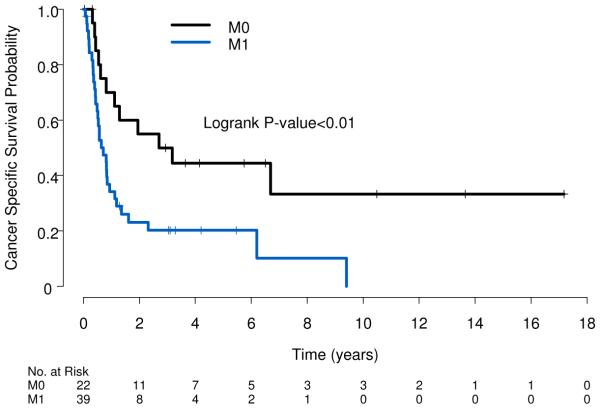
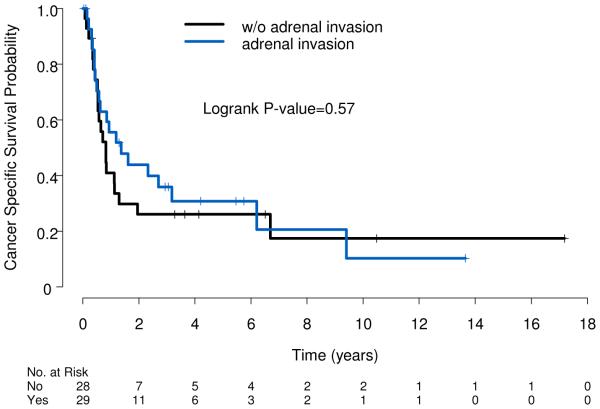
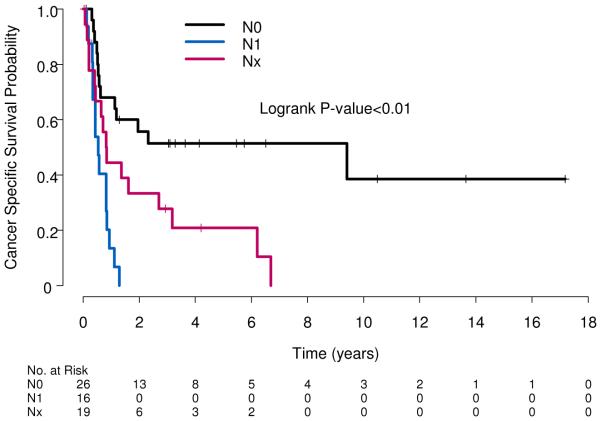
Figures 1-5 display KM curves of variables of interest. As expected, patients with M1 disease had a significantly worse median CSS of 8 months, versus 37 months in patients with M0 disease (Figure 1, p<0.01). Patients with direct adrenal involvement faired as poorly as the rest of the pT4 group, supporting the inclusion of direct adrenal invasion in the pT4 group in the AJCC staging (Figure 4).
Figure 1.
Cancer-specific survival stratified by M stage. (Log rank p<0.01).
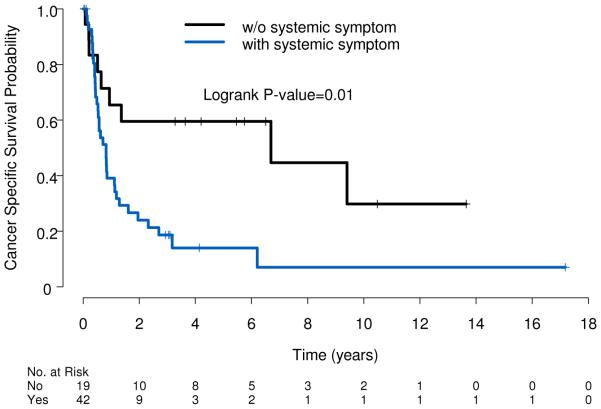
Figure 5.
Cancer-specific survival stratified by presence of systemic symptoms at time of diagnosis. (Log rank p= 0.01).
Figure 4.
Cancer-specific survival stratified by adrenal involvement. (Log rank p=0.57).
DISCUSSION
In our cohort of patients with pT4 RCC who underwent radical nephrectomy, we found that abnormal preoperative LDH and alkaline phosphatase, presence of metastases at time of diagnosis, pathologically confirmed lymph node involvement, and sarcomatoid dedifferentiation were independent predictors of survival in a postoperative model. Given the advanced disease state, most patients in this study were symptomatic, presented with large masses and had aggressive tumor features on final pathology. In addition, approximately two thirds of patients with pT4 had distant metastasis at time of diagnosis, consistent with prior smaller studies [14]. As reflected in the 2010 AJCC TNM staging, patients with tumor extension beyond Gerota’s fascia are classified as stage IV, in the same category as patients with distant metastatic disease, consistent with generally poor outcome in these patients. Moreover, while targeted therapy has improved outcomes in patients with metastatic RCC, surgery remains the cornerstone for treatment of advanced RCC, and patients with suspected T4 RCC should not be routinely excluded from surgery [7]. We and others have shown in the past that in spite of a surgically challenging scenario, resection of the primary tumor in patients whose RCC shows adjacent organ invasion is feasible without significantly increased morbidity, and could provide improved survival in patients with no distant metastasis. Using variables available preoperatively, we found that routine laboratory values (LDH and alkaline phosphatase) were independent predictors of CSS in this cohort. If externally validated, these factors can be potentially used to select patients who should undergo upfront surgical treatment and those who could benefit from presurgical targeted therapy.
At present, accurate preoperative identification of patients with pT4 disease remains difficult. In a previous study, we investigated 30 patients who were thought to have clinical T4 RCC and underwent nephrectomy. Final pathology revealed that only 12 of these patients actually (40%) had pT4. On the other hand, in the current study, of the 61 pT4 patients, only 68.9% were predicted to have clinical T4 on preoperative imaging. These findings reveal the unmet need of better preoperative identification of patients with pT4 disease.
We previously showed that aggressive surgical resection in patients with pT4M1 RCC can achieve palliation from local and systemic symptoms [15]. However, patient survival in this small series was poor at a median of 6.8 months, and the benefit of surgical intervention remains unclear for asymptomatic patients with pT4M1 [15]. Karellas and colleagues [7] studied 38 patients with locally advanced RCC (T3 and T4) who had complex adjacent organ resection and showed that prognosis is generally poor, with outcomes comparable to patients with metastatic disease. Moreover, 90% of the patients in their series died from disease, with a median CSS of 11.7 months. Surgical margin status was the only significant predictor of recurrence and survival. Takagi and colleagues [16] reported on 39 patients with locally advanced RCC with distant metastasis, 14 (36%) of which were pT4, and showed a median CSS of 4.5 months in this subgroup. The authors found that lymph node metastasis, higher pT stage, and non-clear or sarcomatoid histology were predictors of CSS on multivariate analysis. In the current study, although patients with pT4M1 disease had a median CSS of 8 months, those with pT4M0 had significantly longer survival, with median CSS of 37 months, indicating a benefit to surgical resection in patients with non-metastatic disease. As such, patients suspected to have T4 RCC should not be necessarily excluded from surgical resection simply because of their presumed advanced local stage.
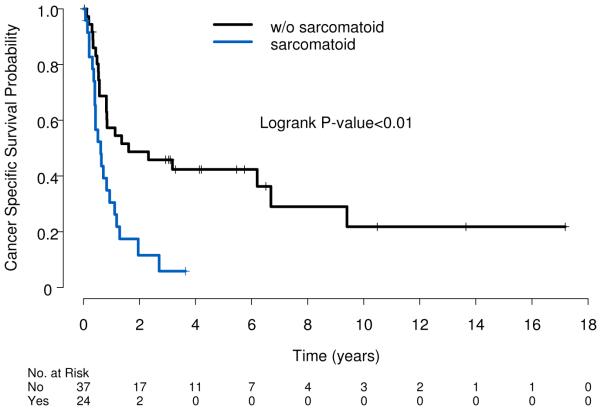
In our series, almost 40% of the patients had sarcomatoid dedifferentiation, illustrating a substantially greater proportion than the approximate incidence in RCC patients[17]. Sarcomatoid dedifferentiation is an independent risk factor for poor prognosis, with worse survival throughout the spectrum of the disease, from clinically localized to metastatic [18, 19]. Patients with sarcomatoid dedifferentiation undergoing resection have poor survival and often may not be able to proceed to postoperative systemic therapy [20]. In fact, only 2 of our 24 patients with pT4 and sarcomatoid dedifferentiation were still alive at 2 years after surgery, underscoring the importance of preoperative identification of these factors for patient counseling.
Although controversy exists on whether lymph node dissection (LND) should be routinely offered, studies suggest that pT4 patients are at the highest risk of lymph node metastasis with a rate up to 35% [21]. We previously showed that nodal stage, along with pT stage, represents a significant independent predictor of disease recurrence and survival in this group [22]. Our current results show that pathologic lymph node involvement influence CSS with pN0/Nx patients having a survival of 2.7 years versus 0.5 years in pN1 patients. Capitanio and colleagues [23] proposed that in pT4 patients, LND may add a survival benefit beyond just staging purposes. In a retrospectively analysis of 44 pT4 patients, when stratified for nodal status, CSS rates at 1, 2 and 3 years were 65.0, 36.1, and 9.0% for pN0 versus 13.3, 13.0, and 6.7% for pN1, respectively (p=0.004). Moreover the number of positive lymph nodes and the number of nodes removed achieved independent predictors status on MVA for CSS (HR 1.25, p=0.001) and as protective effect on survival (HR 0.84 p=0.007), respectively. At present, in our institution, patients with suspected pT4 disease undergo a thorough lymph node dissection for staging, and potentially therapeutic purposes.
Our study has several limitations. This was a retrospective single-center study that spanned a 20-year period. Patients received a variety of systemic therapies in different treatment eras at the discretion of the treating physician, which cannot be accounted for in the analysis given the relatively small number of patients. This study was based on a highly selected cohort of patients who underwent radical nephrectomy. As a result, we were not able to compare non-surgical treatments in patients with pT4 disease. However, to our knowledge, this is the largest study specifically reporting on surgical outcomes from a pathologically confirmed T4 patient cohort based on the 2010 AJCC TNM system, and describing two simple, yet potentially clinically useful models for preoperative and postoperative prognostication in this rare group of patients.
CONCLUSIONS
Survival in patients with pT4 RCC remains poor, with an estimated median survival of 10 months. Preoperative identification of patients with pT4 is in need of refinement, and could help improve patient counseling, management and selection for inclusion in neoadjuvant/presurgical trials. pT4 is associated with a locally and regionally invasive biology (especially sarcomatoid dedifferentiation) that requires specific attention and warrants careful study. Understanding the drivers of this unique phenotype will generate therapeutic interventions that can change the behavior of these aggressive tumors.
Figure 2.
Cancer-specific survival stratified by presence of sarcomatoid features. (Log rank p<0.01).
Figure 3.
Cancer-specific survival stratified by pathological nodal status. (Log rank p<0.01).
Highlights.
In patients with pT4 RCC who underwent RN, we found that abnormal preoperative LDH and ALP, metastases at diagnosis, pN1, and sarcomatoid dedifferentiation were independent predictors of survival.
Accurate preoperative identification of patients with pT4 disease remains difficult. In our study, only 68.9% were predicted to have clinical T4 on preoperative imaging.
Although patients with pT4M1 disease had a median CSS of 8 months, those with pT4M0 had with median CSS of 37 months, indicating a benefit to surgical resection in patients with non-metastatic disease.
Acknowledgements
The Biostatistics Resource Group is supported by the National Institutes for Health/National Cancer Institute under award number P30CA016672.
Financial Disclosures: Jose A. Karam has served as a one-time consultant to Pfizer in 2013. Christopher G. Wood has received research funding from Pfizer and served as a consultant and on its advisory board. None of these disclosures by both authors are related to the current manuscript.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- [1].Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015;65:5–29. doi: 10.3322/caac.21254. [DOI] [PubMed] [Google Scholar]
- [2].Kane CJ, Mallin K, Ritchey J, Cooperberg MR, Carroll PR. Renal cell cancer stage migration: analysis of the National Cancer Data Base. Cancer. 2008;113:78–83. doi: 10.1002/cncr.23518. [DOI] [PubMed] [Google Scholar]
- [3].Lam JS, Belldegrun AS, Pantuck AJ. Long-term outcomes of the surgical management of renal cell carcinoma. World journal of urology. 2006;24:255–66. doi: 10.1007/s00345-006-0055-5. [DOI] [PubMed] [Google Scholar]
- [4].Frank I, Blute ML, Cheville JC, Lohse CM, Weaver AL, Zincke H. An outcome prediction model for patients with clear cell renal cell carcinoma treated with radical nephrectomy based on tumor stage, size, grade and necrosis: the SSIGN score. The Journal of urology. 2002;168:2395–400. doi: 10.1016/S0022-5347(05)64153-5. [DOI] [PubMed] [Google Scholar]
- [5].Kim SP, Alt AL, Weight CJ, Costello BA, Cheville JC, Lohse C, et al. Independent validation of the 2010 American Joint Committee on Cancer TNM classification for renal cell carcinoma: results from a large, single institution cohort. The Journal of urology. 2011;185:2035–9. doi: 10.1016/j.juro.2011.02.059. [DOI] [PubMed] [Google Scholar]
- [6].Stewart GD, Ang WJ, Laird A, Tolley DA, Riddick AC, McNeill SA. The operative safety and oncological outcomes of laparoscopic nephrectomy for T3 renal cell cancer. BJU international. 2012;110:884–90. doi: 10.1111/j.1464-410X.2011.10850.x. [DOI] [PubMed] [Google Scholar]
- [7].Karellas ME, Jang TL, Kagiwada MA, Kinnaman MD, Jarnagin WR, Russo P. Advanced-stage renal cell carcinoma treated by radical nephrectomy and adjacent organ or structure resection. BJU international. 2009;103:160–4. doi: 10.1111/j.1464-410X.2008.08025.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Escudier B, Porta C, Schmidinger M, Algaba F, Patard JJ, Khoo V, et al. Renal cell carcinoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of oncology : official journal of the European Society for Medical Oncology / ESMO. >2014;25(Suppl 3):iii49–56. doi: 10.1093/annonc/mdu259. [DOI] [PubMed] [Google Scholar]
- [9].Ueda K, Noguchi M, Suekane S, Moriya F, Nagayoshi R, Ejima K, et al. Successful treatment of T4 renal cell carcinoma after a neoadjuvant targeted therapy using sunitinib: report of a case. The Kurume medical journal. 2011;58:95–8. doi: 10.2739/kurumemedj.58.95. [DOI] [PubMed] [Google Scholar]
- [10].Rini BI, Garcia J, Elson P, Wood L, Shah S, Stephenson A, et al. The effect of sunitinib on primary renal cell carcinoma and facilitation of subsequent surgery. The Journal of urology. 2012;187:1548–54. doi: 10.1016/j.juro.2011.12.075. [DOI] [PubMed] [Google Scholar]
- [11].Karam JA, Devine CE, Urbauer DL, Lozano M, Maity T, Ahrar K, et al. Phase 2 Trial of Neoadjuvant Axitinib in Patients with Locally Advanced Nonmetastatic Clear Cell Renal Cell Carcinoma. European urology. 2014 doi: 10.1016/j.eururo.2014.01.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Annals of surgical oncology. 2010;17:1471–4. doi: 10.1245/s10434-010-0985-4. [DOI] [PubMed] [Google Scholar]
- [13].Eble John N., Epstein Jonathan I. Pathology and Genetics of Tumours of the Urinary System and Male Genital Organs. IARCPress; Lyon: 2004. GS. [Google Scholar]
- [14].Lam JS, Klatte T, Patard JJ, Goel RH, Guille F, Lobel B, et al. Prognostic relevance of tumour size in T3a renal cell carcinoma: a multicentre experience. European urology. 2007;52:155–62. doi: 10.1016/j.eururo.2007.01.106. [DOI] [PubMed] [Google Scholar]
- [15].Kassouf W, Sanchez-Ortiz R, Tamboli P, Jonasch E, Merchant MM, Spiess PE, et al. Cytoreductive nephrectomy for T4NxM1 renal cell carcinoma: the M.D. Anderson Cancer Center experience. Urology. 2007;69:835–8. doi: 10.1016/j.urology.2007.01.034. [DOI] [PubMed] [Google Scholar]
- [16].Takagi T, Kondo T, Iizuka J, Kobayashi H, Hashimoto Y, Tanabe K. Cytoreductive nephrectomy for pT3b-T4 M1 renal cell carcinoma in 39 patients: single center analysis. International journal of urology : official journal of the Japanese Urological Association. 2012;19:875–9. doi: 10.1111/j.1442-2042.2012.03038.x. [DOI] [PubMed] [Google Scholar]
- [17].Shuch B, Bratslavsky G, Linehan WM, Srinivasan R. Sarcomatoid renal cell carcinoma: a comprehensive review of the biology and current treatment strategies. The oncologist. 2012;17:46–54. doi: 10.1634/theoncologist.2011-0227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Merrill MM, Wood CG, Tannir NM, Slack RS, Babaian KN, Jonasch E, et al. Clinically nonmetastatic renal cell carcinoma with sarcomatoid dedifferentiation: Natural history and outcomes after surgical resection with curative intent. Urologic oncology. 2015 doi: 10.1016/j.urolonc.2014.11.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Kyriakopoulos CE, Chittoria N, Choueiri TK, Kroeger N, Lee JL, Srinivas S, et al. Outcome of Patients With Metastatic Sarcomatoid Renal Cell Carcinoma: Results From the International Metastatic Renal Cell Carcinoma Database Consortium. Clinical genitourinary cancer. 2014 doi: 10.1016/j.clgc.2014.08.011. [DOI] [PubMed] [Google Scholar]
- [20].Shuch B, Said J, La Rochelle JC, Zhou Y, Li G, Klatte T, et al. Cytoreductive nephrectomy for kidney cancer with sarcomatoid histology--is up-front resection indicated and, if not, is it avoidable? The Journal of urology. 2009;182:2164–71. doi: 10.1016/j.juro.2009.07.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Sun M, Bianchi M, Hansen J, Abdollah F, Trinh QD, Lughezzani G, et al. Nodal involvement at nephrectomy is associated with worse survival: a stage-for-stage and grade-for-grade analysis. International journal of urology : official journal of the Japanese Urological Association. 2013;20:372–80. doi: 10.1111/j.1442-2042.2012.03170.x. [DOI] [PubMed] [Google Scholar]
- [22].Margulis V, Sanchez-Ortiz RF, Tamboli P, Cohen DD, Swanson DA, Wood CG. Renal cell carcinoma clinically involving adjacent organs: experience with aggressive surgical management. Cancer. 2007;109:2025–30. doi: 10.1002/cncr.22629. [DOI] [PubMed] [Google Scholar]
- [23].Capitanio U, Matloob R, Suardi N, Abdollah F, Castiglione F, Di Trapani D, et al. The extent of lymphadenectomy does affect cancer specific survival in pathologically confirmed T4 renal cell carcinoma. Urologia. 2012;79:109–15. doi: 10.5301/RU.2012.9255. [DOI] [PubMed] [Google Scholar]