Abstract
Neurogenesis takes place in the adult mammalian brain in three areas: Subgranular zone of the dentate gyrus (DG); subventricular zone of the lateral ventricle; olfactory bulb. Different molecular markers can be used to characterize the cells involved in adult neurogenesis. It has been recently suggested that a population of bone marrow (BM) progenitor cells may migrate to the brain and differentiate into neuronal lineage. To explore this hypothesis, we injected recombinant SV40-derived vectors into the BM and followed the potential migration of the transduced cells. Long-term BM-directed gene transfer using recombinant SV40-derived vectors leads to expression of the genes delivered to the BM firstly in circulating cells, then after several months in mature neurons and microglial cells, and thus without central nervous system (CNS) lesion. Most of transgene-expressing cells expressed NeuN, a marker of mature neurons. Thus, BM-derived cells may function as progenitors of CNS cells in adult animals. The mechanism by which the cells from the BM come to be neurons remains to be determined. Although the observed gradual increase in transgene-expressing neurons over 16 mo suggests that the pathway involved differentiation of BM-resident cells into neurons, cell fusion as the principal route cannot be totally ruled out. Additional studies using similar viral vectors showed that BM-derived progenitor cells migrating in the CNS express markers of neuronal precursors or immature neurons. Transgene-positive cells were found in the subgranular zone of the DG of the hippocampus 16 mo after intramarrow injection of the vector. In addition to cells expressing markers of mature neurons, transgene-positive cells were also positive for nestin and doublecortin, molecules expressed by developing neuronal cells. These cells were actively proliferating, as shown by short term BrdU incorporation studies. Inducing seizures by using kainic acid increased the number of BM progenitor cells transduced by SV40 vectors migrating to the hippocampus, and these cells were seen at earlier time points in the DG. We show that the cell membrane chemokine receptor, CCR5, and its ligands, enhance CNS inflammation and seizure activity in a model of neuronal excitotoxicity. SV40-based gene delivery of RNAi targeting CCR5 to the BM results in downregulating CCR5 in circulating cells, suggesting that CCR5 plays an important role in regulating traffic of BM-derived cells into the CNS, both in the basal state and in response to injury. Furthermore, reduction in CCR5 expression in circulating cells provides profound neuroprotection from excitotoxic neuronal injury, reduces neuroinflammation, and increases neuronal regeneration following this type of insult. These results suggest that BM-derived, transgene-expressing, cells can migrate to the brain and that they become neurons, at least in part, by differentiating into neuron precursors and subsequently developing into mature neurons.
Keywords: Stem cells, Bone marrow, Hippocampus, Cell therapy, SV40, Brain, Nestin, Doublecortin, Neurons, Development, Epilepsy, Seizures
Core tip: It was previously thought that the development of new neurons did not take place in the adult brain of higher vertebrates. There has been substantial progress in understanding neurogenesis in the adult brain during the last decade, showing that neural progenitor cells can induce neurogenesis, mainly in three areas: Subventricular zone, subgranular zone of the hippocampal dentate gyrus, and olfactory bulb. More recently, it has been shown that bone marrow progenitor cells can participate in neurogenesis in the adult brain. In this review, we discuss the mechanisms of the migration, differentiation, and maturation of bone marrow progenitor cells in the adult brain. We also consider the increase of adult neurogenesis following experimental seizures, provided that neuroinflammation is decreased by reducing the expression of chemokines, and consequently the related migration of inflammatory cells into the brain parenchyma.
ROLE OF BONE MARROW PROGENITOR CELLS IN ADULT NEUROGENESIS
Neurogenesis in the adult brain is a relatively new concept. There are three regions of the adult brain in Mammals where neurogenesis can take place in the adult mammal: Subgranular zone (SGZ) of the dentate gyrus (DG); subventricular zone (SVZ) of the lateral ventricle; and olfactory bulb (OB). The cells participating in neuronal development in adults have been characterized using molecular markers. It has been more recently suggested that a population of bone marrow (BM) progenitor cells could also contribute to adult neurogenesis. One way to verify if this hypothesis is correct would be to stain BM stem cells in situ and to track them in the body. In this review, we report that injecting the BM of rats and rabbits with SV40 vectors results in the transduction of BM precursor cells that are migrating, among other organs, to the brain where they differentiate in neurons and microglial cells. It has also been previously shown that neuroinflammation can hamper the process of neuroregeneration following insult in the DG. We show here that reducing the levels of certain chemokine receptors in circulating cells by gene transfer of siRNA against these receptors in a context of a rat model of neurotoxicity leads to a decrease in inflammation and an increase of BM-derived cells migrating to the brain.
GENE DELIVERY TO BONE MARROW PROGENITOR CELLS
Ex vivo gene delivery is the most utilized procedure for transducing hematopoietic stem cell (HSC). However, in order to replace the ex vivo approach of transduction and reimplantation HSC, direct delivery of viral vectors into the BM has been proposed[1-3]. This procedure has been suggested because ex vivo gene transduction and reimplantation may modify the homing properties and can change the functions of progenitor cells and HSC[4-6]. Furthermore, HSC transduced by ex vivo gene delivery procedures may become exposed to infectious agents[4]. We tried here to assess the efficiency of intramarrow injection in the femoral cavity of rats using rSV40 vectors. Levels of transgene expression were evaluated in peripheral blood population during several months[7]. Transgene expression was observed during several months in multiple BM and peripheral blood lineages by using this method[7]. Long term expression of transgene in platelets and the correction of haemophilia phenotype for at 5 mo were observed in other studies[8]. Sustained gene expression was also found present in neuronal cell after in vivo gene transfer[9]. The direct injection of viral gene delivery in the bone marrow can take full advantage of the stem cells that are present within the bone marrow including non hematopoietic cells[8,10]. The targeting of HSC within their niche may be advantageous in the treatment of Fanconi anemia (FA) by ensuring that they maintain their function and by enabling the correction of the remaining stem cells[6]. This approach also eliminates challenges, such as the requirement for preconditioning[8,10], thus making it very promising in the treatment of FA[6] and haemophilia[8]. Moreover, gene transfer based on direct intramarrow injection should prevent difficulties seen during ex vivo approach such as stimulation by cytokines, putative loss of engraftment, and keeping HSC properties overtime[8,10]. Gene transfer to the BM improves the efficiency of HSC viral vector transduction and strengthens the supportive microenvironment by opposition to intravenous inoculation that delivers vector to blood[8,10]. Consequently, this procedure leads to better preservation of stem cell viability and capacity[10]. We used the intrafemoral gene delivery approach to BM progenitor cells to study their fate in the body, and more particularly in the brain. We will first review the cells involved in adult neurogenesis, their molecular markers, the different areas involved in adult neurogenesis, and the factors influencing neuronal development. Then, by using gene delivery into the femoral cavity, we will show the migration and differentiation of transduced BM progenitors to the brain. Finally, we will demonstrate that reducing neuroinflammation in an animal model of experimental seizure leads to an increase of BM progenitor cells migrating towards the brain.
NEUROGENESIS IN THE ADULT BRAIN
The traditional concept was that the development and growth of new neurons from neuronal stem precursor cells does not take place in the adult brain of higher vertebrates. In 1962, Altman[11] demonstrated neurogenesis in the brain of an adult rat. There was no further report until Goldman and Nottebohm[12] reported evidence of neurogenesis in canaries. This was further substantiated in 1992 when studies reveal similar evidence of adult neurogenesis[13]. There has been substantial progress in understanding neurogenesis in the adult brain during the last decade. However, molecular events leading to the increase number, migration, and differentiation of progenitor cells in the brain need to be better characterized. The participation of BM progenitor cells to adult neurogenesis has also been suggested more recently.
Adult neurogenesis occurring in mammalian brain is now a well accepted idea, essentially in three regions: SGZ of the DG, SVZ of the lateral ventricle, and olfactory bulb[14]. Dividing cells see their number reduced after birth, except in the SGZ of the DG and the SVZ (Figure 1)[15].
Figure 1.
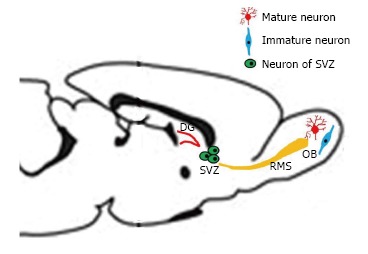
Different areas of neurogenesis in the adult rodent. The three areas of neurogenesis in the adult are the dentate gyrus of the hippocampus, the subventricular zone, and the olfactory bulb. Some progenitor cells are migrating from the SVZ to the OB, along a rostral migratory stream. DG: Dentate gyrus; SVZ: Subventricular zone; OB: Olfactory bulb; RMS: Rostral migratory stream.
Neuroblasts and progenitor cells from the SVZ also migrate through the rostral migratory stream (RMS) to the OB maturing into new neurons (Figure 1). The same process has been documented in primates, including humans[16-18]. Neurons, produced in the DG and the SVZ are primarily granular neurons[17] and to a lesser extent periglomerular neuronal cells for the OB[19].
The SVZ of the lateral ventricle is mainly located in the lateral wall of the lateral ventricle which is facing the striatum[20]. It contains various neural progenitor cells along its wall. Four different types of cells have been identified in the SVZ of the lateral ventricle of mice[20,21]. They are described as types A, B, C and E cells (Figure 2). These cells differ from each other based on the ultrastructure, morphology and molecular markers expressed by the cells. Type A cells, the most common ones, are darker than B cells in electron microscopy. Their cell bodies are elongated and contain up to two processes; they have abundant lax chromatin with two to four nucleoli; the cytoplasm is dark and contained free ribosomes. The rough endoplasmic reticulum has only a few short cisternae, the Golgi apparatus is small, and many microtubules are arranged along the long axis of the cell[20]. These type A cells were described as being connected to each other by junctional complexes[20]. Type A cells are neuroblasts expressing polysialated form of the neural cell adhesion molecule (PSA-NCAM). Type A cells are migratory in nature and course tangentially to the walls of the lateral ventricle. Type B cells are slow dividing astrocytes that enclose the migrating neuroblasts. These cells have different characteristics compared to type A cells[14]. Their nuclei is irregular and the cytoplasm is lighter stained. They are characterized by their abundant intermediate filaments and the dense bodies within their cytoplasm. A further subdivision of type B cells into type B1 and a type B2 has been reported[20]. Type B1 cells are larger, and contain more cytoplasm. It is believed that type B cells are the neural precursor cells and that they give rise to both type C and type A cells[22]. Type C cells are found only in the SVZ and they are rapidly dividing immature precursor cells arranged in clusters along the migrating chains. Type C cells are larger, more spherical, contain larger golgi apparatus than type B cells; however their size is similar to type B1 but they have fewer processes[20]. Type E cells belong to ependymocytes lineage (Figure 2).
Figure 2.
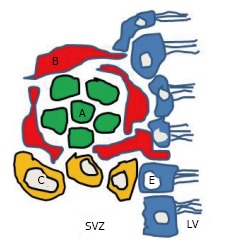
Four different types of progenitor cells in the subventricular zone. Type-A cells are migrating towards the olfactory bulb along the rostral migratory stream. Type B cells are believed to be the neural precursor cells and will give rise to both type C and type A cells. Type C cells are rapidly dividing immature precursor cells arranged in clusters along the migrating chains, and they are found only in the SVZ. E cells belong to ependymocytes lineage. LV: Lateral ventricle; SVZ: Subventricular zone.
Cells in the SVZ that are originating from the lateral wall of the lateral ventricle migrate along the RMS pathway to be incorporated into the OB[14,23]. The rostral migratory stream pathway is constituted mainly of type A and type B cells[20]. There has been no evidence so far of type C cells within the RMS and studies suggest that this region is devoid of these cells. Type A cells are arranged in chains surrounded by type B cells within the RMS, similarly as within the SVZ. These cells reach the olfactory bulb and become interneurons. In addition, microglial cells and endothelial cells are also present in the RMS[24]. The division and migration of the neuroblasts within and to the olfactory bulb is independent of the process that takes place within the olfactory bulb[25].
SGZ of the DG is situated between the hilus and the granular cell layer of the DG in the hippocampus. This is one of the main areas where neurogenesis takes place[17,26] with an estimated 9000 new cells generated each day[27]. Not all of the neurons generated in the hippocampus will survive and become incorporated into the neuronal circuit of the brain. Approximately 50% of the neurons born in the SGZ will die after birth without being incorporated in the neuronal circuit of the brain. Within the hippocampus, three types of cells are identified: (1) Type 1, radial cells, which give rise to type 2 cells; (2) Type 2, non-radial intermediate, neural progenitor cells; and (3) Neuroblasts, derived from type 2 cells[28]. Type 1 cells are found to be progenitor cells similar to type B cells found in SVZ and are slow dividing cells, whereas type 2 cells are rapidly dividing ones[29].
MOLECULAR MARKERS OF NEUROGENESIS
Adult neurogenesis involves a succession of events occurring in a specific order[30-32]. For example, hippocampal neurogenesis is thought to begin with stem cells located in the SGZ of the DG. There, these cells proliferate, differentiate and give rise to new neurons. Neurons at various stages of neurogenesis express different markers. Consequently, the fate, and differentiation, of cells during neurogenesis can be followed[33,34] (Figure 3 and Table 1).
Figure 3.
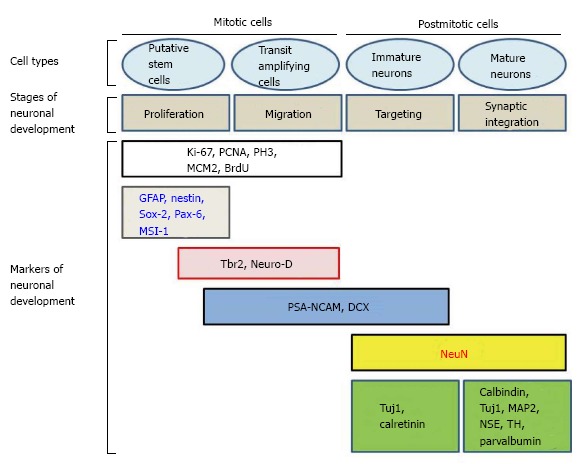
Different markers of neuronal development in mitotic and postmitotic cells (modified from Ref. [21,35]). PSA-NCAM: Polysialated form of the neural cell adhesion molecule; DCX: Doublecortin; GFAP: Glial fibrillary acidic protein; PCNA: Proliferating cell nuclear antigen; PH3: Phosphorylated form of histone 3; MCM2: Minichromosome maintenance protein 2; MSI-1: Musashi-1; MAP2: Microtubule-associated protein 2; NSE: Neuron specific enolase; TH: Tyrosine hydroxylase.
Table 1.
Markers of cells involved in neurogenesis with corresponding stages of neurogenesis
| Marker | Cells | Stage of neurogenesis |
| Nestin | Neuronal stem cells, radial glia cells, transit amplifying cells | Proliferation, differentiation |
| GFAP | Neuronal stem cell, mature astrocytes | Proliferation, differentiation |
| PSA-NCAM | Migrating neuroblast, immature neuron | Differentiation, migration, targeting |
| Tuj1 | Migrating neuroblast, immature neuron, mature neuron | Differentiation, migration, targeting |
| Doublecortin | Migrating neuroblast, immature neuron | Differentiation, migration, targeting |
| NeuN | Mature neuron | Targeting, synaptic integration |
GFAP: Glial fibrillary acidic protein; PSA-NCAM: Polysialated form of the neural cell adhesion molecule.
Different markers help to determine the cells in the stage of neurogenesis and their role during particular stages. Some of the most commonly used markers include glial fibrillary acidic protein (GFAP), nestin, Neuro D, Hu, TuJ1, doublecortin (DCX), PSA-NCAM, neuron specific enolase (NSE) and neuronal specific nuclear protein (NeuN). Markers are useful to distinguish cellular proliferation, early adult neurogenesis, later steps of adult neurogenesis, and mature stage.
Different markers of proliferation can be used. The most utilized markers are proliferating cell nuclear antigen, and Ki-67, respectfully characterized by a long half-life, and a short half-life of about one hour. Ki-67 is rarely used in this application. Other markers include minichromosome maintenance protein 2 and phosphorylated form of histone 3[35]. Bromodeoxyuridine (BrdU) incorporates into DNA during S phase and is also used as a marker of proliferating cells. To conclude that BrdU-positive cells are of neuronal lineage, and to characterize the cell lineages involved, BrdU-labeling needs to be combined with markers of neurons at different stages of neurogenesis[33,35].
Markers of early stages of adult neurogenesis include GFAP, which is an intermediate filament protein expressed by mature astrocytes within the adult brain[33,36,37]. Its expression has been described in type B progenitor cells present in the SVZ and type 1 cells described in the SGZ[21,36-38]. Nestin is a structural analog to intermediate filament protein, found to be positive in type B neural progenitor cells and transit amplifying type C cells in the SZV[21], in type 1 (radial glial cells) and 2 cells in the SGZ[29,39]. Nestin is thought to be a marker of precursor cells. Consequently, nestin synthesis decreases, as differentiation of nervous tissue progresses. Neurofilament and glial (e.g., GFAP) proteins begin to be expressed during the differentiation of neurons and astrocytes respectively when nestin expression starts to be reduced. Nestin expression declines during the postnatal period. However, rare cells expressing nestin are present in the adult DG and the SVZ[40]. Expression of nestin is abruptly terminated. Other early markers of adult neurogenesis are Sox-2, a SRY-related HMG-box gene 2, paired box gene 6 (Pax-6), and musashi-1 (MSI-1), a RNA-binding protein preferentially expressed in the CNS. However, neuronal specificity of some of these markers is not absolute[35].
The markers of later stages of adult neurogenesis include DCX and PSA-NCAM, both markers of immature neurons. DCX is a protein positive in cells within the RMS and is present in migrating progenitor of neurons[41,42]. DCX induces polymerization of microtubules. DCX is a marker expressed by neuroblasts during their migration and immature neurons of the granular layer of the DG. It is also present in newly generated cells located at the border of the granular layer of the DG and in the SGZ. DCX is expressed in newly generated olfactory, hippocampal, and striatal neurons, but not in the cortex. DCX is a marker of late mitotic neuronal precursors and early postmitotic neurons. There is no overlap between the expression of DCX and nestin[33,34]. As such, DCX is a good marker for adult neurogenesis. Expression of the (PSA-NCAM) is observed at the same stage. Thus, both mitotic neuronal precursors and early postmitotic new neurons are positive for PSA-NCAM and DCX[35]. The basic helix-loop-helix protein NeuroD and T-box brain protein 2 (Tbr2) are also expressed during later steps of adult neurogenesis, and show some overlap with the expression of Pax-6. Neuro D is a protein inducing microtubule polymerization. NeuroD is expressed in proliferating neurons and in migrating neuroblasts[33]. Both Tbr2 and NeuroD are coexpressed with DCX and PSA-NCAM. Moreover, Tbr2 is downregulated when cells are committed to neuronal lineage[35].
Finally, NeuN is a soluble protein that is found in the nucleus and cytoplasm of postmitotic neurons. NeuN is expressed by mature neurons[33]. In the hippocampus (HC), postmitotic cells are immunopositive for NeuN. NeuN is a marker of both newly generated postmitotic neurons and “normal” postmitotic neurons. There is a correlation between the decreased expression of DCX and the beginning of NeuN expression[33]. Additional markers can be used to characterize mature neurons, and some of them can be specific of certain areas of the brain. Hu is a RNA binding protein from the elav family expressed from the early stages of neurogenesis to the end[43]. Tyrosine hydroxylase is the enzyme that catalyzes the formation of L-DOPA, precursor of dopamine. Several calcium-binding proteins can be expressed in mature neurons: Calbindin-D28k is particularly abundant in the cerebellum; calretinin is a 29 k protein with 58% homologies to calbindin-D28k; parvalbumin is a small calcium-binding albumin protein involved in calcium signaling. Microtubule-associated protein 2 induces assembly of microtubules, an essential stage in neuritogenesis. NSE is often used as a marker of mature neurons. Neuron-specific class III β-tubulin (Tuj1) contributes to axonal transport and provides stability to microtubules in axons and somas. However, the list of the markers for mature neurons is not exhaustive (Figure 3).
Markers expressed in the SVZ and the subgranular zone of the DG during adult neurogenesis are presented in Tables 2 and 3 respectively.
Table 2.
Markers expressed during neurogenesis originating in the subventricular zone
| Neuronal lineage | Type B cells | Type C cells | Type A cells | Neuronal cells |
| Cell identity | SVZ Astrocytes | Transit amplifying progenitor cell | Migrating neuroblast | Mature and immature neuron |
| Putative stem cells | ||||
| Identifying marker | GFAP | Nestin | PSA-NCAM | DCX |
| Nestin | Tuj1 | NeuN | ||
| Hu |
SVZ: Subventricular zone; GFAP: Glial fibrillary acidic protein; PSA-NCAM: Polysialated form of the neural cell adhesion molecule; DCX: Doublecortin; NeuN: Neuronal specific nuclear protein.
Table 3.
Markers expressed during neurogenesis in the subgranular zone of the dentate gyrus
| Mitotic cells | Post mitotic cells | |||
| Stages of neuronal development | Putative stem cell | Transit amplifying progenitors | Immature neurons | Mature neurons |
| Markers for neuronal development | GFAP Nestin | Nestin Doublecortin | Doublecortin Tuj1 | Tuj1 NeuN NSE |
GFAP: Glial fibrillary acidic protein; NeuN: Neuronal specific nuclear protein; NSE: Neuron specific enolase.
MIGRATION OF BONE MARROW PROGENITORS TO THE BRAIN AND THEIR DIFFERENTIATION INTO CELLS OF DIFFERENT CNS LINEAGES
rSV40-transduced bone marrow progenitor cells migrate to the brain
Gene delivery to the brain has focused mainly on transducing neurons directly. However, an alternative approach may be to consider those areas of the brain where neurogenesis continues well into adult life: The DG of the HC, and the SVZ. New neurons are generate throughout life in the DG. These new neurons are involved in the repair of brain insults. Targeting endogenous brain cell progenitors in situ might be attempted in order to genetically engineer them. For example, proliferation of such engineered cells after injury or during disorder of the brain would lead to functional brain cells expressing a transgene of potential interest. However, this approach is limited by the low number of endogenous progenitors in the adult brain. Furthermore, the life span of endogenous stem cells might be not as long compared to pluripotent stem cells with different properties as well.
Migration of BM stem cells to the brain and their differentiation into different types of brain cells has been reported in rodents[44,45] as well as in humans[46]. When they are injected intravascularly or intraperitoneally in rodents, adult BM progenitor cells are able to migrate to the adult CNS where they differentiate into neuronal[45,47], or non-neuronal cells[48]. In patients with transplantation of BM, the autopsy of brains demonstrated that human HSC can trans-differentiate into neurons, astrocytes, and microglia following long term marrow engraftment. These results were observed without fusion between cells. They suggest that human HSC coming from BM-transplantation could be used as a potential therapeutic source not only for long term regenerative neuropoiesis[46], but also for gene delivery in the brain.
The potential of direct transduction of HSC in the BM has been raised. However, in situ in vivo gene delivery to HSC/progenitors by direct injection of viral vectors in the BM has been rarely described, despite the putative interest of this method. The fate of cells positive for the transgene in the body has never been studied in this context. Our group previously reported that the inoculation of a Tag-deleted recombinant SV40 vector carrying a marker gene (FLAG epitope appended to HIV-1 Nef as a carrier protein) in the femoral BM of rats caused positive results[7]. Expression of the transgene in the blood lasted throughout the 16 mo of the study. Twenty-five percent of femoral marrow cells and between 4%-12% (average, 5%) of blood nucleated cells of all lineages were positive for the transgene FLAG throughout the whole study. However, it remained to be determined if HSC could migrate to the brain in this experimental paradigm. The aim of the study was to determine the localization and the type of transgene-positive cells in the DG and in the SVZ. DG is composed of the hilus area surrounded by the granule cell layer (GCL), formed by an inner (upper) and outer (lower) blades. We therefore investigated by immunohistochemistry[49,50] if some BM progenitor cells, transduced in vivo by intramarrow injection of a rSV40 vector, could migrate to the adult CNS and differentiate into different brain cell lineages[51,52].
Transgene expression was seen in cells with shape of neurons in the DG 16 mo after intramarrow injection of the vector. Transgene expression was not seen in the DG of control animals whose BM was either inoculated with a control vector [SB(BUGT)], or saline. Transgene-positive cells were mostly observed in the DG and SVZ (Figure 4A). Transgene-positive cells were not seen before 1 mo and few of them were detected at 4 mo. Numerous transgene-positive cells were observed in the DG and SVZ 16 mo after intramarrow injection of the vector. Some of the transgene-expressing cells were also positive for NeuN, a marker of mature neurons, not only in the GCL but also in the hilus. Transgene-positive cells expressing NeuN had the shape of neurons of the DG. There were also some transgene-expressing cells that were not NeuN positive in the same areas, and some of these cells had the morphology of microglial cells and were immunopositive for CD11b-C3bi, and CD68, both markers of microglial cells. Very rare transgene-positive were astrocytes. Sixteen months after intramarrow injection of the vector, approximately 5% of DG cells were positive for the transgene. Forty-eight point six percent, 49.7% and 1.6% of these transgene-positive cells were respectively expressing markers of neurons, microglia, and astrocytes, as assessed by double immunocytochemistry for the transgene and lineage markers. We also determined transgene expression in the SVZ. The pattern of FLAG expression in the SVZ was similar as in the DG. One and 4 mo after intramarrow injection of the vector, no or few cells were expressing FLAG; numerous transgene-positive cells were observed in the SVZ 16 mo after intramarrow injection of SV(Nef-FLAG). About 50% of transgene-positive cells were of neuronal lineage because stained by neurotrace (NT), a neuronal marker. This percentage is close to the one observed in the DG. Thus, transgene-positive cells can be observed in areas of the CNS other than the DG. Our results show that cells present in the adult BM can migrate to the CNS where they differentiate into cells of CNS lineages.
Figure 4.
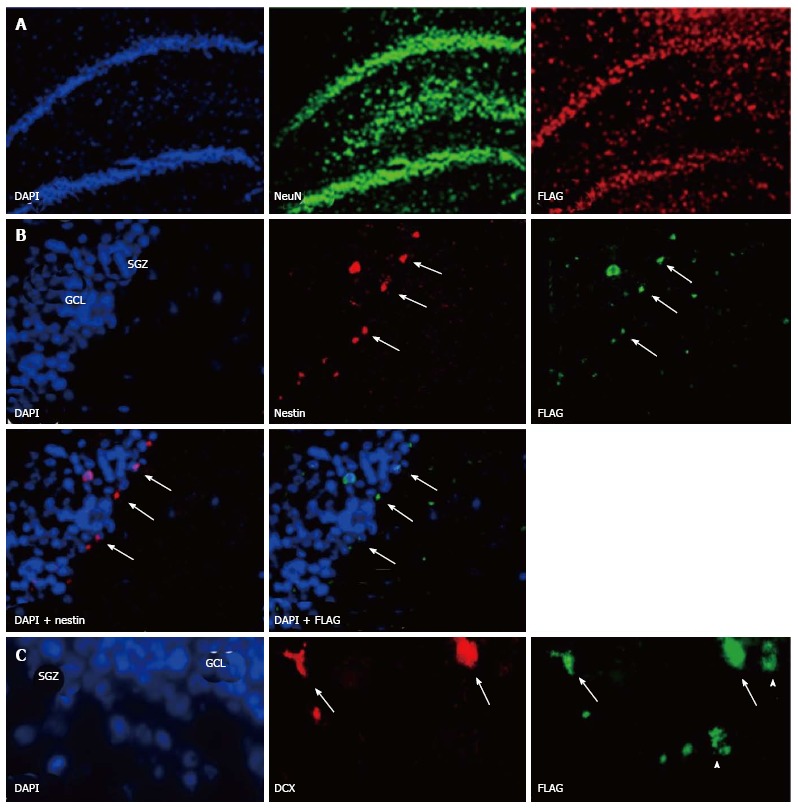
Bone marrow derived cells can migrate to the rat normal hippocampus. Sixteen months after injection of SV (Nef-FLAG) into the rat bone marrow (BM), transgene expressing cells were detected in the dentate gyrus (DG). A: FLAG-positive cells colocalized with NeuN, a marker of mature neurons. FLAG+/NeuN+ cells were located in the granular cell layer (GCL), as well as in the subgranular zone (SGZ) and the hilus. No FLAG-expressing cells were detected in the brain after injection of SV (BUGT), a control vector, in the BM; B: Nestin+/FLAG+ cells were detected mainly at the border SGZ/GCL, and more rarely in the GCL (arrows); C: DCX+ positive cells expressing FLAG were seen at the border SGZ/GCL and more rarely in the GCL (arrows). Note that some FLAG+ cells were DCX-, and were probably mature neurons (arrowheads). In all experiments, nuclei were stained in blue by Vectashield containing DAPI.
Consequently, it appears, that gene delivery to the BM induces transgene expression in brain cells of different lineages, in the DG and the SVZ. These results show that adult neurogenesis continues in the DG and SVZ during adult life, and that at least some of the cells generated that way derive from one or more populations of resident BM cells at the time of the administration.
Other hypotheses can explain these observations. rSV40 vectors can transduce CNS cells when directly administered into the brain[53]. However, DG cells would unlikely be transduced at the time of the injection of the vector in the BM. Only 16 mo after the injection were transgene-positive cells seen in the DG. Transgene-positive cells were absent the first weeks after injection of the vector and were rare 4 mo after intramarrow injection of the vector. Moreover, transgene-positive cells were rarely detected in epithelial cells of other organs usually transduced by intravenous rSV40 administration (e.g., liver, kidney), at all times after injection, suggesting that the vector did not diffuse significantly from the site of injection[54,55].
Others also suggested that BM progenitor cells might differentiate into mature brain cells. It has been shown that endogenous neural stem cells are present in the DG and SVZ, and can generate different types of brain cells[56]. These endogenous progenitor cells may be responsible for the constant remodeling taking place in the HC and OB[15,16]. The apparent maturation and differentiation of BM progenitors seen in the present study is likely a physiological process because no insult was caused to the brain. Various types of injury can lead the adult rodent brain to repair itself by neurogenesis[57-59]. Neurogenesis taking place in the HC and SVZ following such insults is based on the generation of new neurons that are able to migrate a considerable distance from their origin[57]. Moreover, these regenerated neurons can replace dying cortical ones[60,61].
It is still unclear if such process takes place in humans during physiologic or pathologic conditions. However, our results were observed without injury given to the CNS. Both experimental and human data showed that transplantation of male HSC coming from the BM into female recipients resulted in CNS neurons that were bearing Y chromosome[45,46]. Cogle et al[46] detected neurons in the HC of women recipients coming from male BM transplants. However, this process took several months to occur. These data are coherent with our results[46,62,63]. Similarly, BM progenitor cells from male mice, transplanted into immunocompromised female recipients, generated cells positive for Y-chromosome, that were detected in the brain and were positive for neuronal markers[45,46].
Studies using gene-marked BM cells have shown that BM-derived cells may transdifferentiate into CNS cells of one or more lineages[47]. Cell-cell fusion might support some of the reports[62-64]. However, when they are exposed to certain growth factors (i.e., brain-derived neurotrophic factor, epidermal growth factor), or when they are grown together with fetal mesencephalic or striatal cells, cultured BM stromal cells can express some glial and neuronal antigens[65]. Similarly, when they are administered into brain ventricles of neonates, BM stromal cells can differentiate into cells immunopositive for astrocytic and neuronal markers[66]. It has been suggested that BM progenitor cells can trans-differentiate into brain cells of different lineages[45]. However, other reasons can explain these observations as well, and the mechanism(s) of such trans-differentiation are not clear as yet.
The first step in the migration of BM progenitor cells to the brain would be the homing of the BM-derived population(s) to their target. The mobilization pathway of HSC towards the brain involves a complex and intimate collaboration between adhesion molecules, cytokines, proteolytic enzymes, stromal cells, and HSC. This process can explain the regulation of HSC release, migration, and homing from the BM to the brain[67]. Vascular and extracellular matrix molecules have an important role as well[22,45,68-70]. Numerous reports involve mechanisms and factors inducing or promoting mobilization of HSC from BM into peripheral blood (PB). Several cell membrane proteins, including CXCR4 and its ligand, α-chemokine stromal-derived growth factor-1 (SDF-1), are involved in the mobilization of HSC to the PB[71-74]. Different factors can increase the number of HSC migrating to the PB. Among these factors, some are related to tissue or organ injury, strenuous exercise and stress, local or systemic inflammation, and finally pharmacological agents such as CXCR4 small-molecule antagonist AMD3100 and granulocyte colony-stimulating factor (G-CSF)[74-77]. Among these factors affecting HSC mobilization, G-CSF and SDF-1 are the best known. It must be noted that the activation of the complement cascade is activated by all these processes[74,76].
BM niches are retaining HSC through the interaction between the chemokine CXCR4 receptor and α4β1 integrin. The respective ligands of CXCR4 and α4β1 integrin are α-chemokine SDF-1 and vascular adhesion molecule-1 (VCAM-1, also known as CD106), and they are present on cells in the BM niches (e.g., fibroblasts and osteoblasts)[74-77]. One of the main factors affecting HSC mobilization, G-CSF, operates in two ways: First, it disrupts the anchoring relationship by decreasing the expression of SDF-1, thus reducing the binding of SDF-1 to CXCR4. Secondly, G-CSF enhances serum levels of other cytokines and growth factors[67,78,79].
SDF-1 (chemokine CXCL12) is highly expressed in the BM where it is generated by osteoblasts in the endosteal region, as well as by endothelial cells and reticular cells located in the BM stroma. SDF-1 is a potent chemoattractant for HSC and it controls cell adhesion and survival as well. Synthesis of SDF-1 obeys to a circadian rhythm regulated by the sympathetic nervous system. Noradrenaline operates via β2-adrenoreceptors present on osteoblasts and via β3 adrenoreceptors expressed in nestin-positive stem cells in order to decrease their production of SDF-1[80,81].
CXCR4 and CXCR7 have been described as two chemokine receptors for SDF-1. The relationship between SDF-1 and CXCR4 in HSC is believed a key factor in the control of the traffic of HSC in the BM[82,83]. AMD3100 is a powerful bicyclam CXCR4 antagonist that acts synergistically with G-CSF in humans. AMD3100 enhances mobilization of HSC in the BM[84]. Once mobilized, HSC express decreased levels of CXCR4. CXCR7 is a second high-affinity receptor for SDF-1. However, CXCR7 is not linked to signaling pathways for migration of HSC. SDF-1 is internalized then degraded once bound to CXCR7; consequently, CXCR7 appears to act as a SDF-1 sink[67,78]. CXCR4 and CXCR7 interact, but CXCR4 inhibition does not seem to modify the role of CXCR7[67].
There are several factors influencing the interactions SDF-1-CXCR4 and α4β1 integrin-VCAM-1. For example, the sensitivity to SDF-1 depends on the incorporation of the CXCR4 receptor into membrane lipid rafts[67,85,86]. As a consequence, the migration of HSC is influenced by gradients of the bioactive lipids sphingosine-1 phosphate (S1P) and ceramide-1 phosphate (CP1). S1P and CP1 result from membrane lipid metabolism and are involved in stem cell trafficking.
Other important molecules involved in HSC mobilization are proteolytic enzymes released from activated granulocytes and monocytes that are present in the BM. These enzymes operate by attenuating SDF-1-CXCR4 and α4β1 integrin-VCAM-1 interactions in the BM microenvironment[74-77]. Another example of molecular intervention is the role of innate immunity in this process. One of the molecules involved in innate immunity, β2-defensin (β2-D), influences the sensitivity of HSC to SDF-1[76]. Different proteolytic cascades such as the complement cascade, coagulation cascade, and fibrinolytic cascade, as well as several other proteolytic enzymes secreted by cells present in the BM might have a role as well[87]. Different stress situations, local or systemic inflammation, and administration of pharmacological mobilizing agents (e.g., G-CSF and AMD3100) can influence these proteolytic cascades[76].
Activated complement stimulates oxidative stress, activation of platelets, and injury of erythrocyte membranes, interacts with different proteolytic cascades in the BM, and consequently, triggers mobilization of HSC[67]. The third (C3a) and fifth protein components of complement (C5) have also an important role in the mobilization of HSC[88]. The first cells migrating from the BM are neutrophils[88]. C5b-C9 complex (or membrane attack complex) induces release of S1P, from red blood cells and platelets[89]. Thus, inflammatory process and innate immunity will induce the migration of HSC from BM into PB[76].
In another cascade involved in the mobilization of HSC, the fibrinolytic cascade, plasminogen binds to the BM extracellular matrix (ECM). Secondly, various proteins components of the ECM, including fibrin, laminin, are damaged by plasminogen after it converted into plasmin. Other proteases, such as metalloproteinases MMP-3, MMP-9, MMP-12 and MMP-13, are also activated by plasminogen in order to reduce the levels of other components of the ECM, such as collagen[90].
Heme oxygenase 1 (HO-1) is also involved in the mobilization process of the HSC[91]. Among other roles, HO-1 mitigates the inflammation linked to the complement by increasing the expression of complement inhibitors CD55 and CD59 on endothelial cells[92]. HO-1 participates also in the regulation of the expression of SDF-1[93], a major factor in the retention of HSC in BM niches[74-77]. It has been shown recently that HO-1 influences actually negatively the adhesion and migration of neutrophils during acute inflammation[94]. It has been suggested that negative regulators of the mobilization of HSC from BM exist. But few results have been reported so far. Serine protease inhibitors (serpins)[95] and tissue inhibitors of metalloproteinases (TIMPs)[96] seem to have such inhibitory effects on mobilization of HSC. HO-1 seem to influence negatively the migration of HSC[74].
Concerning homing of progenitor cells to organs, and not PB, other factors can be involved. For example, resveratrol enhances migration of mesenchymal stem cells to injured liver[97].
After mobilization from the BM, the second step would be the differentiation into CNS cells of different lineages. However, mechanisms of differentiation are not totally clear yet[98]. Neural cell adhesion molecules (i.e., N-CAM), proteins regulating cell cycle transit[99] and transcription factors[100] have been implicated in this process, as well as molecules of the ECM[68,69]. Ectopic expression of a specific set of transcription factors (c-Myc, Sox2, Oct4 and Klf4) can reprogram mouse embryonic and adult fibroblasts into embryonic stem-like cells[101]. These cells were called induced pluripotent stem (iPS) cells. It has been reported that stem cells can be reprogrammed into iPS cells more effectively than into mature cells. Other types of cells can demonstrate multipotency. Among these cells, multilineage-differentiating stress-enduring (MUSE) cells[102] and mesenchymal stem cells (MSC) have been described[103,104]. MUSE cells are adult stem cells. They are characterized by differentiation into different lineage (mesodermal-, ectodermal- and endodermal) cells from a single cell. MUSE cells are tolerant to stress. They express markers of pluripotency, and are able of self renewal[102]. They can be obtained from fibroblasts. Properties of MUSE cells resemble those of iPS cells, but they are devoid of tumorigenicity[102]. MSC are a population of multipotent, self-renewing cells, mostly located in a bony niche, that regulate skeletal tissue and repair[105].
High levels of sonic hedgehog and induction of Wnt signaling induce derivation of floor plate from pluripotent stem cells, increasing the quantity and quality of dopaminergic (DA) neurons consequently produced[106,107]. These results led to the development of protocols for differentiation of mouse iPSCs into DA neurons[108]. These newly generated cells expressed markers of DA neurons, such as the enzyme tyrosine hydroxylase, as well as the transcription factors Nurr1, and Pitx3. Once transplanted in a rat model of Parkinson’s disease, these DA cells could repair lesions seen in the animal model[108].
Transdifferentiation of fibroblasts to neurons has been reported. The introduction of three specific factors of neurodevelopment (Brn2, Ascl1 and Myth1l, or BAM) in mouse fibroblasts, directly generated neuronal cells, called induced neurons, or iNs[109]. Similar approach was used with the same results by using human fibroblasts[110]. Mouse and human iNs were characterized by immunopositivity for neuronal markers such as Tuj1, Map2, Tau and synapsin. Moreover, the combination of the iN factors with Lmx1a and FoxA2[111] or a combination of Lmx1a and Nurr1[112] in fibroblasts can directly generate cells with DA neuronal characteristics, named iDA neurons. The combination of the three neurodevelopmental factors (Brn2, Ascl1 and Myth1l, or BAM) with other factors implicated in the embryonic development of DA neurons enhances the transdifferentiation towards cells presenting characteristics of DA neurons[113-118].
It is also possible to induce the transformation of acinar exocrine cells from pancreas of cadavers into pancreatic β-cells. Cells generated that way can produce insulin, and are glucose-regulated. Once transplanted into immunocompromised diabetic mice, these cells can normalize glycemia[119].
In our experiments, we examined the brain and other organs[120] to assess the transgene expression outside of the BM in animals whose BM was injected with SV40-derived vector. We found that CNS cells of different lineages expressed the transgene several months after intramarrow administration of the vector, suggesting that transgene-positive cells were likely to have migrated from the BM. About 5% of DG cells were transgene-positive. BM progenitor cells were also migrating towards other organs (i.e., spleen, lungs, liver). In the lungs, FLAG-expressing cells were mainly seen in the alveoli and were coexpressing markers of progenitor cells (i.e., TTF-1) and of macrophages. Our studies do not determine the nature of the BM progenitors migrating to the CNS. These cells may be of hematopoietic, stromal or other origin. The hypothesis that BM cells can transdifferentiate into CNS cells of different lineages has been the topics of several conflicting reports. However, various animal models, different experimental paradigms and diverse methods have been used and could explain the discrepancies observed in these reports. In most of the studies against the hypothesis of BM to CNS transdifferentiation, the markers used are protein products of transgenes[121-123]. Transgene delivered by numerous vectors, integrating or not, may disappear with time, particularly if they are of protein origin. By contrast, rSV40 vectors lead to long-term transgene expression[54,124]. In other studies, DNA markers have been used to assess the engraftment of stem cells and the differentiation of donor cells in to host cells[125,126]. DNA probes, such as the ones in FISH assays, were used in most studies sustaining the hypothesis of BM to CNS transdifferentiation. One of the important factors explaining the conflicting data appears to be the time between the transplantation of progenitor cells in the brain or in the periphery and their differentiation. Human cells transplanted into the mouse lateral ventricle differentiate into neurons in the OB only after several months[61]. The number of transgene-positive neurons enumerated in the brain after injection of GFP-expressing HSC increases with age[62,63]. The brief time intervals between the injection of the animals with progenitor cells and the harvest of the tissues might explain some negative results[121]. However, these negative results might be due to the full maturity of the neurons, to the molecular marker used, and to the type of neurons as well. Our work suggest that BM-derived cells found in the DG are mature neurons, because expressing NeuN. They also have the shape of mature neurons in the considered area. However, we do not know at this point what is their physiological function and what type of synapses are established.
The settings of the experimental system might also explain the results observed. For example, some experiments include treating HSC in vitro with growth factors that might modify their properties before their administration. Such treatment could potentially influence the homing of transplanted cells towards the brain or their differentiation into cells of CNS lineages. These experimental settings do not reproduce physiological conditions. By avoiding some of these treatments, we were able to transduce the BM directly without perturbing the different BM populations of cells. Thus, we can suggest that BM resident cells can transdifferentiate into cells of CNS lineages. However, additional studies would be useful to explore the functions of these cells.
Bone marrow progenitor cells express different markers of neuronal differentiation in the brain
We reported above that permanent BM-directed gene transfer using recombinant SV40-derived vectors led to expression of the transgene in mature neurons, and thus without CNS lesion indicating that BM progenitor cells can differentiate into cells of different CNS lineages. Most of transgene-expresssing cells also expressed NeuN, a marker of mature neurons. However, it remained to be determined by what mechanism the cells from the BM come to be neurons. Although the observed gradual increase in transgene-expressing neurons over 16 mo suggested that the pathway involved differentiation of BM-resident cells into neurons, we could not rule out cell fusion as the principal route. Therefore, we tested here whether BM-derived progenitor cells migrating in the CNS could express markers of neuronal precursors or immature neurons. We injected SV40-derived vectors, carrying marker epitopes (FLAG or AU1), into the femoral cavities of rats or rabbits. Control animals received a control vector, SV (BUGT), injected into the BM as well. AU1- or FLAG-positive cells were seen in the DG 16 mo after injection of respectively SV (RevM10.AU1) or SV (Nef-FLAG) in the BM. In addition to cells expressing markers of mature neurons, transgene-positive cells were also positive for nestin and doublecortin, molecules expressed by developing neuronal cells. These cells were actively proliferating, as shown by short term BrdU incorporation studies (Figures 4B, 4C and Figure 5). These results confirm that BM progenitors migrate to the CNS where they become neurons, by differentiating into neuron precursors and subsequently developing into mature neurons. Similar results were seen in the rat (Figures 4B, 4C and 5) and the rabbit (Figure 6).
Figure 5.
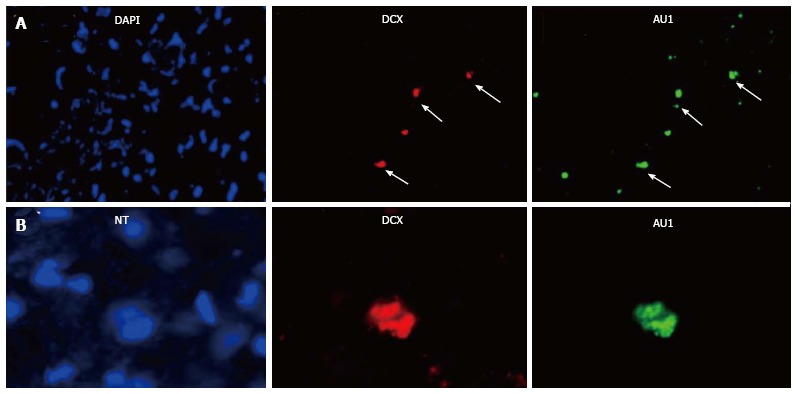
Transgene (AU1) expression in the hippocampus of rats whose bone marrow has been injected with SV (RevM10.AU1). A: Almost all DCX-positive cells were expressing AU1 (arrows); B: DCX+/AU1+ cells were of neuronal lineage, because they were stained by NT (arrows). No AU1 expression was seen after bone marrow injection of SV (BUGT), a control vector. DCX: Doublecortin; NT: Neurotrace.
Figure 6.
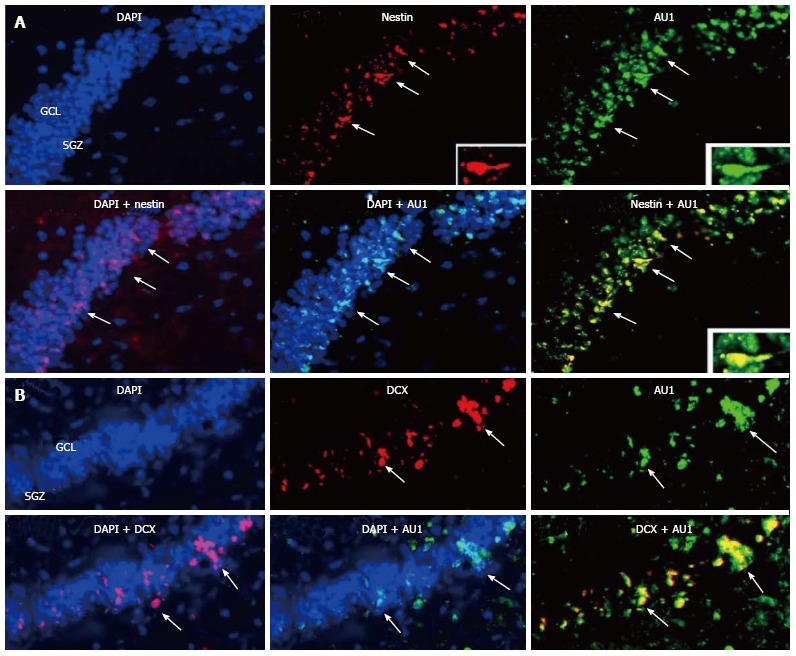
Migration of bone marrow derived cells to the rabbit hippocampus. Sixteen months after injection of SV (RevM10.AU1) into the rabbit bone marrow (BM), transgene-positive cells were seen in the dentate gyrus. Numerous AU1-positive cells colocalized with NeuN, a marker of postmitotic neurons (not shown). A: Nestin+/AU1+ cells were seen mainly at the border SGZ/GCL, and more rarely in the GCL (arrows); B: DCX+ positive cells expressing AU1 were detected at the border SGZ/GCL and more rarely in the GCL (arrows). SGZ: Subgranular zone; GCL: Granular cell layer; DCX: Doublecortin.
This progression recapitulates the sequential stages of neurogenesis. Transgene-expressing cells positive for nestin and DCX were mainly localized at the border between SGZ and GCL, and less frequently in the GCL. This location is the one usually reported for immature neurons in the hippocampus. Cells in the SGZ have been shown to migrate into the GCL[30].
In assessing the studies that report support for, or evidence against, BM cell transdifferentiation into other lineages[121,122], the time between the administration of stem cells and harvest of the tissue is a critical factor[61-63,66]. Thus, a brief time interval between injecting animals and collecting tissue may not identify markers belonging to the BM cells in the target organ studied. We have found that increases in neuronal and microglial cell populations expressing transgenes delivered to BM were gradual and protracted. No transgene-expressing neurons were seen 1 mo following BM injection, a few at 4 mo and many were at 16 mo[51]. BM cells can undergo spontaneous fusion with other cell types, a process that is probably unlikely. Higher levels of transdifferentiation have been reported in some studies[127]. The results observed in physiological conditions, as here, should also be compared to the ones seen during brain damage[128-132] in order to determine the putative factors influencing the migration and differentiation of BM-derived cells in adult CNS.
FACTORS INFLUENCING NEUROGENESIS IN ADULT BRAIN AND ROLE OF SEIZURES
Factors influencing neurogenesis
Various physiological and pathological factors may increase neurogenesis in different areas of the brain. Neurogenesis is found to be increased in acute neurodegenerative disease[28,133]. Hippocampal neurogenesis is enhanced during Alzheimer’s disease[134]. Brain injury such as stroke also leads to an increase in neurogenesis in humans[135]. Neurogenesis may be increased following damage to the adult brain[57,59]. Different classes of molecules have affected the rate of neurogenesis, including growth factor, hormones[136] and neurotransmitters. Testosterone can increase generation of new neurons in the DG of adult male rodents[136]. Blockade of neuroinflammation can restore adult neurogenesis in the hippocampus[137].
Neuronal loss, inflammation, regeneration and gliosis following kainic acid-induced seizures
It has been reported that seizures can induce neuronal regeneration in the DG, but the extent of the process is hampered by the inflammation following the seizure. We first identified neuronal regeneration post kainic acid (KA)-induced seizure. For this purpose, we injected intraperitoneally 10 mg/kg KA in rats. KA causes tonic-clonic seizures. After the animals recovered from these seizures, they were rested for 7 d and their brains were examined for neuron loss in the HC, using either NeuN or NT as neuronal markers. We observed that neuron loss was most prominent in the CA1 and CA2/CA3 regions of the hippocampus, but loss of neurons was statistically significant, compared to control rats receiving vehicle only, in the hilus, SGZ/GCL and in CA1, CA2 and CA3.
At the same time, we demonstrated an inflammatory infiltration in the HC by enumerating macrophage/microglial cells using antibodies against Iba-1 (quiescent and activated cells) and CD68 (activated cells), and astrocytes using antibody against GFAP. Numbers of GFAP- and Iba-1-positive cells were increased in the HCs of KA recipients, compared to controls, indicating that brain injury related to KA administration led to infiltration of the affected area by macrophages and microglia, and to an astrocytic response. Neuroinflammation was associated with an increased expression of cytokines and chemokines, particularly regulated on activation, normal T cell expressed and secreted (RANTES) and macrophage inflammatory protein 1 α (MIP-1α), as well as C-C chemokine receptor type 5 (CCR5) itself. RANTES and MIP-1α are both ligands of CCR5 receptors. KA-induced increases in production of CCR5 ligands and ICAM-1 within blood vessels suggests that CCR5+ cells may be increased in the hippocampi of KA-treated rats, compared to control animals. We therefore enumerated CCR5+ cells in several areas of the HCs of rats treated with KA or saline. No CCR5+ cells were detected in control rat HCs. In contrast, CCR5+ cells were significantly more abundant throughout the hippocampi in KA recipients. In HCs of rats injected with KA, CCR5 was expressed mainly by lymphocytes, monocytes/macrophages, microglial cells, to a lesser extent by neurons, and rarely with astrocytes. This expression was seen not only in inflammatory cells in brain parenchyma, but in vessel walls as well.
KA-induced injury also elicits a regenerative response: Increased new neurons are formed following the insult. We assessed the level of cell proliferation and the populations of cells involved by labeling with BrdU. BrdU-expressing cells were seen in the GCL and SGZ of HCs from rats injected with KA. Very rare cells positive for BrdU were seen in HCs of controls. In KA recipients, 80.2% of these cells were neurons. In addition, proliferation of both immature neurons and neural stem cells was involved, as evidenced by BrdU positivity in cells that also expressed doublecortin and nestin, respectively (Figure 7).
Figure 7.
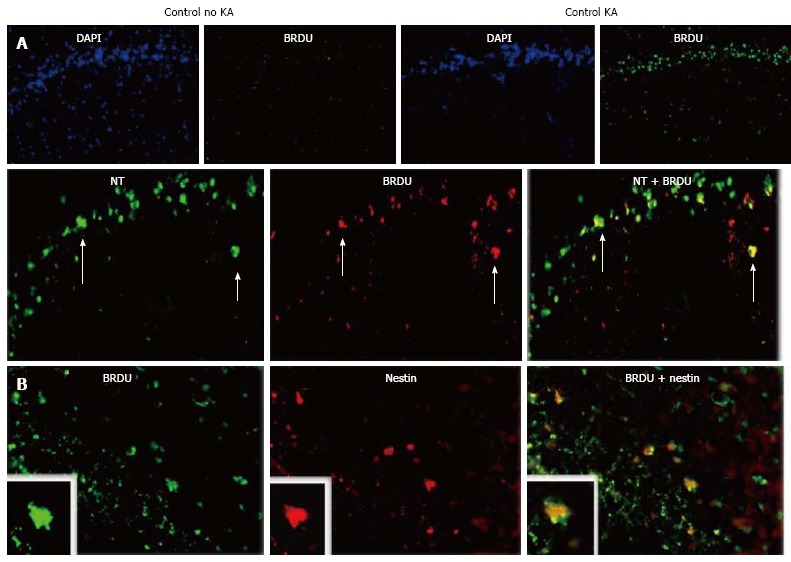
Kainic acid-induced regeneration in the hippocampus. Rats were injected with kainic acid (KA) intraperitoneally (i.p.), 10 mg/kg, and their hippocampi (HCs) analyzed by immunomicroscopy 7 d thereafter. A: Neurogenesis following KA treatment. Rats given KA or saline were injected with BrdU. Then, at 7 d post-KA, their DGs were immunostained for BrdU to visualize proliferating cells. DAPI counterstain is shown to facilitate interpretation. Arrows show neurons positive for BrDU. Double staining with Neurotrace (NT) for neurons is shown below; B: Double staining for nestin, a marker of proliferating and migrating neural cells (modified from Ref. [132]).
CCR5 regulates migration of bone marrow progenitors to the DG and limits neuroregeneration in response to injury
We reported above that KA elicits injury, inflammation and neuron regeneration in the HC. In the next step, we showed that decreasing CCR5 on bone marrow-derived cells reduces the number of KA-induced seizures, and related injury and inflammation. We used for that a bifunctional vector composed of a vector targeting CCR5 (RNAiR5) and a vector carrying a protein tag, used to evaluate transgene expression. The effect of RNAiR5 gene (targeting CCR5) delivery to the BM on the migration of BM progenitors to the DG and their differentiation into neurons was assessed, with and without KA treatment.
Intraperitoneal injection of 10 mg/kg KA causes grade 5 tonic-clonic seizures (range of seizure duration, 30-80 s) in normal rats beginning an average of 32.5 ± 4.7 min post-injection (range, 20-45 min). The average number of such seizures within a 4 h period after KA administration was 23.6 ± 3.4 (range, 21-28). Rats were given SV (RNAiR5-RevM10.AU1), the monofunctional vectors SV (RevM10.AU1) or SV (RNAiR5) intrafemorally. SV (BUGT) was used as control vector. Animals were challenged 4 mo later by i.p. injection of KA. Rats given RNAiR5-containing vectors were significantly protected from KA-induced seizure activity compared to rats receiving either SV (BUGT) or SV (RevM10.AU1), by all criteria applied: Time of seizure onset, number and severity of seizures and recovery time. The two RNAiR5-carrying vectors were comparable in protecting from KA-induced seizure activity, and were highly significantly different from the two control vectors [SV (RevM10.AU1), SV (BUGT)].
Four months after injection of the SV40-derived vector that does not target CCR5, and without KA administration, very few BM progenitors were seen in the DG. This is consistent with the observations reported about concerning the migration of BM-derived cells to the CNS. By contrast, in rats receiving a vector carrying the RNAi that targets CCR5, there was a significant increase in the numbers of bone marrow-derived cells expressing neuronal markers. Thus, decreasing CCR5 led to increased bone marrow cell migration to the brain and increased DG neurons derived from those cells. However, in the absence of KA treatment, the number of cells originating from the BM was low. After KA treatment, and notwithstanding the ability of RNAiR5 to mitigate KA-induced injury, numbers of DG cells derived from the bone marrow was far greater in recipients of the vector targeting CCR5 than in recipients of an unrelated vector.
We examined, further, the influence of bone marrow-directed gene delivery of RNAiR5 on DG neuroregeneration. Proliferating cells were visualized using BrdU, and immature neurons were identified using doublecortin and nestin as markers. Again, despite the mitigating effect of bone marrow-directed RNAiR5 on DG injury and neuroinflammation, proliferation of neuronal precursors in response to KA treatment was approximately 3-fold that seen in recipients of the control vector only. Moreover, 72.2% ± 7.4% of BrdU-positive cells were also transgene-positive, and 79.8% ± 6.9% of transgene-positive cells were positive for BrdU. Most of these cells were of neuronal lineage, as demonstrated by immunopositivity for nestin and/or doublecortin or NeuN (Figure 8).
Figure 8.
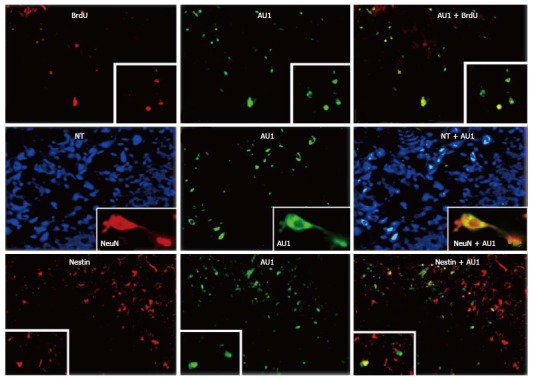
RNAiR5 gene delivery to the bone marrow increases numbers of bone marrow-derived cells in the hippocampus and neuroproliferative activity in the hippocampus, in the resting state and in response to kainic acid treatment. Bone marrow-derived cells in the hippocampus (HC) were all neurons, and most proliferating cells in the HC were bone marrow-derived. Double immunostaining for AU1 plus BrdU (upper row) and nestin (lower row), and neuron identification using Neurotrace (NT) and NeuN (middle row) was performed. Shown are HC sections from kainic acid-treated rats, transduced with SV (RNAiR5-RevM10.AU1). Most of the AU1+ cells were also positive for BrdU (79.8%, ± 6.9%) as a marker of cell proliferation, and most BrdU+ cells were also AU1+ (72.2%, ± 7.4%). Numerous AU1+ cells were also positive for nestin, a marker of proliferating and migrating neurons, although there were as well many nestin-positive cells that did not express AU1. The equivalence of NT identification of neurons and NeuN immunostaining of neurons is illustrated in the insets in the middle row. Data are representative of 3 independent experiments (modified from Ref. [132]).
We reported that bone marrow-directed gene delivery of RNAi targeting CCR5 using a recombinant SV40-derived vector results in high levels of gene modification of bone marrow-derived cells, particularly Sca-1+ cells. As a consequence of the effectiveness of this approach to downregulating CCR5, our data suggest that the cell membrane chemokine receptor, CCR5, influences greatly the regulation of the traffic of BM progenitors towards the CNS, both in the basal state and in response to injury. Furthermore, reduction in CCR5 expression in circulating cells provides profound neuroprotection from, in this case, excitotoxic neuronal injury. CCR5 and its ligands enhance CNS inflammation and seizure activity, and may result in increased CNS injury as a result.
Inflammatory cell infiltration of the CNS entails adhesion of lymphocytes and monocytes in the blood to cerebrovascular endothelium, mediated by endothelial cell production of chemokines, clustering of integrins and migration of peripheral blood mononuclear cells (PBMC) through the vascular endothelium into the brain. CCR5 and its ligands, RANTES and MIP-1α, are known to be involved in this process[138-144]. Thus, such transmigration is stimulated by RANTES but decreased by anti-CCR5 antibodies[142,143]. Our data are consistent with these findings. Expression of MIP-1α and RANTES on brain microvessels and endothelial cells is greatly increased after KA administration, and we showed that reducing cell membrane CCR5 probably decreases PBMC adhesion to CCR5 ligands[145]. Lower levels of CCR5 on PBMC membranes following BM-directed gene delivery also decreases production of those chemokines by brain vascular endothelium. Migration of CCR5+ microglia and monocyte-derived macrophages is stimulated by CCR5 ligands and these cells, in turn, stimulate both endothelial activation and production of proinflammatory cytokines[141]. Our results show also that the initial production of CCR5 ligands after KA administration is unaltered in rats injected with both control and rSV40s vectors targeting CCR5, but this changed over time between the recipients of control vector and rSV40 vectors reducing CCR5. Thus, CCR5 is involved in a multiplicative effect of chemotaxis, stimulation of chemotaxis and then more chemotaxis. Altering this cycle by reductions in PBMC CCR5 may thus have neuroprotective and antiinflammatory effects that are disproportionate to the magnitude of the decrease in CCR5.
These results showing the interaction between CCR5 on PBMC and its ligands at the vascular level emphasize the role of vascular inflammation in KA-induced seizures. It has been reported that vascular inflammation and leukocyte-endothelial adhesion can participate in the development of seizures. α4 integrin and VCAM-1 antibodies can mitigate leukocyte-vascular interaction and prevent pilocarpine-induced seizures[146]. It has been recently suggested that blood-brain barrier (BBB) breakdown can induce epileptiform activity[147,148]. BBB disruption has been described after KA administration[149-151]. During KA-induced seizures, disruption of the BBB is characterized by disappearance of tight junction ZO-1 and occludin, recruitment of neutrophils[149], increase in the production of tissue plasminogen activator and NO[150]. Activation of astrocytes, for example by glutamate agonists, can influence vessel permeability[149]. Another chemokine, MCP-1 (CCL2), can induce BBB opening and KA-induced upregulation of MCP-1 mediates recruitment of macrophages and granulocytes[151]. Increase of MCP-1 in blood vessels of HCs after seizures might lead to modifications of permeability of the BBB[152]. Enhanced permeability of BBB can increase the access of KA to the parenchyma[146]. In the pilocarpine model, it has been suggested that the effect of the drug can be to allow focal BBB leakage, which then synergizes with the CNS effects of pilocarpine to induce seizures. Leukocyte adhesion blockade prevented BBB opening in the pilocarpine model[146]. In the present results, much less leakage of vascular contents from blood vessels was seen in HCs of rats given rSV40s vectors targeting CCR5, suggesting that the experimental reduction of the interaction between CCR5 on PBMC and CCR5 ligands on vessels limited BBB leakage.
BBB leakage is partly mediated by leukocytes through different mechanisms: Generation of oxygen free radicals, enhanced production of cytokines and chemokines, vascular alterations, release of cytotoxic enzymes. Adhesion of leukocytes to endothelium produces changes in small GTPases involved in cytoskeletal organization, and in calcium signaling, as well as in activation of kinases[146,153]. Our demonstration that inhibition of interactions between CCR5 on PBMC and CCR5 ligands in vessels prevents BBB disruption after KA is consistent with prior observations about the effects of leukocyte adhesion on vascular permeability[146]. Thus, our results show that CCR5 influences the synergistic interactions between leukocyte adhesion, endothelial activation, BBB leakage and seizure activity.
CCL3 (MIP-1α)[154,155] and CCL5 (RANTES)[156,157] can be expressed by brain endothelial cells. Different mechanisms can be responsible for the induction of CCL5 and CCL3 in the endothelium. Several studies have demonstrated the involvement of TNF-α and IL1-β in seizure activity[129,158-160]. TNF-α and IL1-β can be rapidly produced by microglial cells[161]. TNF-α and IL1-β stimulate expression of CCL5 in endothelial cells[156,157,162]. TNF-α can also activate NF kappaB, the role of which has been underscored in seizure activity, including KA-induced neurotoxicity[163]. TNF-α-induced CCL5 transcription involves cis-regulatory promoter elements, i.e., NF-κB, C/EBPb, NF-IL-6, NF-AT and the cAMP response element, CRE[164]. TNF-α can induce NF-κB, but apparently not AP-1, activity in endothelial cells[165]. Other mechanisms upregulating CCL5 in endothelial cells involve HIF-1α, JNK-2 and AP-1 (JunD). Lysophosphatidylcholine can induce rapid expression of CCL5 in endothelial cells by activating of multiple kinases[166]. Increase of PLA2 activity has been reported in different models of epilepsy[167] including kainic acid treatment[168-170].
The type of benefit from decreasing CCR5 described in the present study has been seen in studies in which TAK-779, an inhibitor of CCR5, mitigates CNS damage due to ischemia[171]. Targeting MIP-1α may also protect from experimental autoimmune encephalomyelitis[172] and other types of CNS injury[173-176]. Another important consequence of BM-delivered, RNAi-mediated, decrease in CCR5 expression in blood cells is increased migration of BM progenitors to the DG, where they become neurons. These new neurons are generated in absence of lesions, but also can assist in repairing insults to the CNS[177-179]. BM progenitors can migrate to the brain and become neurons by transdifferentiation or other processes[44-48]. Inflammation may limit the ability of the brain to repair itself by neurogenesis, as the generation and survival of new neurons is inversely proportional to the magnitude of the inflammation[128,180], and is facilitated by administration of inflammation inhibitors such as indomethacin[137].
Our results underscore the relationship between ongoing neurogenesis and BM-derived cells. In those studies, we found almost no such cells in the DG at 4 mo after gene delivery to the BM, but numerous cells 15 mo after intramarrow inoculation of the vector. Our current data show that decreasing CCR5 expression on bone marrow-derived cells both increased the basal level of neurogenesis from BM-derived progenitors and greatly amplified the regenerative response to excitotoxic injury. This observation is even more striking in light of the fact that recipients of the vectors targeting CCR5 had much less severe neuron loss than did controls. Furthermore, the level of neuron proliferation, as measured by BrdU incorporation, was also several-fold higher in recipients of vectors that contained RNAiR5 than in controls. It is likely, therefore, that targeting CCR5 by gene transfer or pharmacologic means may promote ongoing neurogenesis and neuroregenerative responses to injury. It might be due to an inhibition of inflammation, known for limiting neurogenesis[128,137,180]. However, a direct role of CCR5 in regulating neuron regeneration cannot be excluded. It is not known whether the cell population(s) in the bone marrow that provide these neuron precursors are of hematopoietic or other origin.
Thus, the consequences of acquired deficiency in CCR5 highlights the role of CCR5 in neuroinflammation, excitotoxic injury, chemotaxis and astrocyte proliferation, and decreases in CCR5 may provide pronounced neuroprotection from such injury. In the present study, cells in the treated animals expressed less CCR5 (molecule per cell) than those from control animals. Even if the cells have not become negative for CCR5, the decreased expression of this receptor did affect their function. The distinction between a complete shutdown of CCR5 and reduced expression due to the RNAi is important, because if marginal reductions in a target can result in a disproportionate loss of function (due for example to reduced total avidity at the cell surface) there is greater hope for the use of such vectors in other in vivo applications.
In conclusion, our data demonstrate the centrality of CCR5 and its ligands in mediating injury-induced inflammation, and suggest that decreasing levels of CCR5 may have as its consequences neuroprotection and enhanced neuroregeneration. We confirm here that BM progenitor cells participate in neurogenesis in the adult brain, and migrate towards the DG and SVZ (Figure 9). SV40-based gene delivery of RNAi targeting CCR5 to the BM results in downregulating CCR5 in circulating cells. Consequently, the inhibition of interactions between CCR5 on peripheral blood mononuclear cells and CCR5 ligands in vessels prevents BBB disruption after KA treatment. The decrease of leukocyte-vascular interaction affects vascular permeability, thus, infiltration of parenchyma by inflammatory cells, and reduces neuroinflammation. Subsequently, our results imply that CCR5 influences the interactions between leukocyte adhesion, endothelial activation, BBB leakage and seizure activity. However, given the redundancy of cytokines and chemokines, CCR5 might be just one of the components implicated in the interaction between leukocytes and vessels, and other chemokines, or other molecules, might be involved as well. For example, receptors for IL-1 and TNF-α are upregulated rapidly during seizures[160]. The magnitude of seizure activity impacts on the inflammatory responses that follow seizures[181-183]. Microglial activation, and production of IL-1β, IL6, TNF-α and free radical species, directly affect the process of post-seizure neurogenesis[181-184] and the survival of the neurons that are produced as a result[159,182,185,186].
Figure 9.
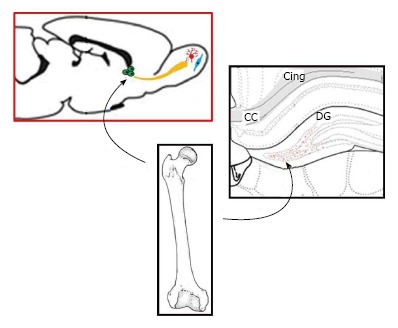
Figure suggesting the participation of bone marrow progenitor cells to neurogenesis in adults. DG: Dentate gyrus.
Footnotes
Conflict-of-interest statement: There is no conflict of interest.
Open-Access: This article is an open-access article which was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
Peer-review started: June 7, 2015
First decision: July 6, 2015
Article in press: February 16, 2016
P- Reviewer: Barnes DW, Dunbar GL, Li SC, Saeki K S- Editor: Gong XM L- Editor: A E- Editor: Lu YJ
References
- 1.Nelson DM, Metzger ME, Donahue RE, Morgan RA. In vivo retrovirus-mediated gene transfer into multiple hematopoietic lineages in rabbits without preconditioning. Hum Gene Ther. 1997;8:747–754. doi: 10.1089/hum.1997.8.6-747. [DOI] [PubMed] [Google Scholar]
- 2.Porada CD, Tran ND, Zhao Y, Anderson WF, Zanjani ED. Neonatal gene therapy. transfer and expression of exogenous genes in neonatal sheep following direct injection of retroviral vectors into the bone marrow space. Exp Hematol. 2000;28:642–650. doi: 10.1016/s0301-472x(00)00158-2. [DOI] [PubMed] [Google Scholar]
- 3.McCauslin CS, Wine J, Cheng L, Klarmann KD, Candotti F, Clausen PA, Spence SE, Keller JR. In vivo retroviral gene transfer by direct intrafemoral injection results in correction of the SCID phenotype in Jak3 knock-out animals. Blood. 2003;102:843–848. doi: 10.1182/blood-2002-12-3859. [DOI] [PubMed] [Google Scholar]
- 4.Mátrai J, Chuah MK, VandenDriessche T. Recent advances in lentiviral vector development and applications. Mol Ther. 2010;18:477–490. doi: 10.1038/mt.2009.319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Frecha C, Costa C, Nègre D, Amirache F, Trono D, Rio P, Bueren J, Cosset FL, Verhoeyen E. A novel lentiviral vector targets gene transfer into human hematopoietic stem cells in marrow from patients with bone marrow failure syndrome and in vivo in humanized mice. Blood. 2012;119:1139–1150. doi: 10.1182/blood-2011-04-346619. [DOI] [PubMed] [Google Scholar]
- 6.Habi O, Girard J, Bourdages V, Delisle MC, Carreau M. Correction of Fanconi Anemia Group C Hematopoietic Stem Cells Following Intrafemoral Gene Transfer. Anemia. 2010;2010:pii: 947816. doi: 10.1155/2010/947816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Liu B, Daviau J, Nichols CN, Strayer DS. In vivo gene transfer into rat bone marrow progenitor cells using rSV40 viral vectors. Blood. 2005;106:2655–2662. doi: 10.1182/blood-2005-01-0028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wang X, Shin SC, Chiang AF, Khan I, Pan D, Rawlings DJ, Miao CH. Intraosseous delivery of lentiviral vectors targeting factor VIII expression in platelets corrects murine hemophilia A. Mol Ther. 2015;23:617–626. doi: 10.1038/mt.2015.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Worsham DN, Schuesler T, von Kalle C, Pan D. In vivo gene transfer into adult stem cells in unconditioned mice by in situ delivery of a lentiviral vector. Mol Ther. 2006;14:514–524. doi: 10.1016/j.ymthe.2006.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pan D. In situ (in vivo) gene transfer into murine bone marrow stem cells. Methods Mol Biol. 2009;506:159–169. doi: 10.1007/978-1-59745-409-4_12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Altman J. Are new neurons formed in the brains of adult mammals? Science. 1962;135:1127–1128. doi: 10.1126/science.135.3509.1127. [DOI] [PubMed] [Google Scholar]
- 12.Goldman SA, Nottebohm F. Neuronal production, migration, and differentiation in a vocal control nucleus of the adult female canary brain. Proc Natl Acad Sci USA. 1983;80:2390–2394. doi: 10.1073/pnas.80.8.2390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Reynolds BA, Weiss S. Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science. 1992;255:1707–1710. doi: 10.1126/science.1553558. [DOI] [PubMed] [Google Scholar]
- 14.Sohur US, Emsley JG, Mitchell BD, Macklis JD. Adult neurogenesis and cellular brain repair with neural progenitors, precursors and stem cells. Philos Trans R Soc Lond B Biol Sci. 2006;361:1477–1497. doi: 10.1098/rstb.2006.1887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Thomas LB, Gates MA, Steindler DA. Young neurons from the adult subependymal zone proliferate and migrate along an astrocyte, extracellular matrix-rich pathway. Glia. 1996;17:1–14. doi: 10.1002/(SICI)1098-1136(199605)17:1<1::AID-GLIA1>3.0.CO;2-7. [DOI] [PubMed] [Google Scholar]
- 16.Eriksson PS, Perfilieva E, Björk-Eriksson T, Alborn AM, Nordborg C, Peterson DA, Gage FH. Neurogenesis in the adult human hippocampus. Nat Med. 1998;4:1313–1317. doi: 10.1038/3305. [DOI] [PubMed] [Google Scholar]
- 17.Gould E, Reeves AJ, Fallah M, Tanapat P, Gross CG, Fuchs E. Hippocampal neurogenesis in adult Old World primates. Proc Natl Acad Sci USA. 1999;96:5263–5267. doi: 10.1073/pnas.96.9.5263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kornack DR, Rakic P. The generation, migration, and differentiation of olfactory neurons in the adult primate brain. Proc Natl Acad Sci USA. 2001;98:4752–4757. doi: 10.1073/pnas.081074998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lois C, Alvarez-Buylla A. Long-distance neuronal migration in the adult mammalian brain. Science. 1994;264:1145–1148. doi: 10.1126/science.8178174. [DOI] [PubMed] [Google Scholar]
- 20.Doetsch F, García-Verdugo JM, Alvarez-Buylla A. Cellular composition and three-dimensional organization of the subventricular germinal zone in the adult mammalian brain. J Neurosci. 1997;17:5046–5061. doi: 10.1523/JNEUROSCI.17-13-05046.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Abrous DN, Koehl M, Le Moal M. Adult neurogenesis: from precursors to network and physiology. Physiol Rev. 2005;85:523–569. doi: 10.1152/physrev.00055.2003. [DOI] [PubMed] [Google Scholar]
- 22.Doetsch F, Caillé I, Lim DA, García-Verdugo JM, Alvarez-Buylla A. Subventricular zone astrocytes are neural stem cells in the adult mammalian brain. Cell. 1999;97:703–716. doi: 10.1016/s0092-8674(00)80783-7. [DOI] [PubMed] [Google Scholar]
- 23.Altman J. Autoradiographic and histological studies of postnatal neurogenesis. IV. Cell proliferation and migration in the anterior forebrain, with special reference to persisting neurogenesis in the olfactory bulb. J Comp Neurol. 1969;137:433–457. doi: 10.1002/cne.901370404. [DOI] [PubMed] [Google Scholar]
- 24.Lois C, García-Verdugo JM, Alvarez-Buylla A. Chain migration of neuronal precursors. Science. 1996;271:978–981. doi: 10.1126/science.271.5251.978. [DOI] [PubMed] [Google Scholar]
- 25.Rochefort C, Gheusi G, Vincent JD, Lledo PM. Enriched odor exposure increases the number of newborn neurons in the adult olfactory bulb and improves odor memory. J Neurosci. 2002;22:2679–2689. doi: 10.1523/JNEUROSCI.22-07-02679.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Altman J, Das GD. Post-natal origin of microneurones in the rat brain. Nature. 1965;207:953–956. doi: 10.1038/207953a0. [DOI] [PubMed] [Google Scholar]
- 27.Cameron HA, McKay RD. Adult neurogenesis produces a large pool of new granule cells in the dentate gyrus. J Comp Neurol. 2001;435:406–417. doi: 10.1002/cne.1040. [DOI] [PubMed] [Google Scholar]
- 28.Mu Y, Gage FH. Adult hippocampal neurogenesis and its role in Alzheimer’s disease. Mol Neurodegener. 2011;6:85. doi: 10.1186/1750-1326-6-85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Miller JA, Nathanson J, Franjic D, Shim S, Dalley RA, Shapouri S, Smith KA, Sunkin SM, Bernard A, Bennett JL, et al. Conserved molecular signatures of neurogenesis in the hippocampal subgranular zone of rodents and primates. Development. 2013;140:4633–4644. doi: 10.1242/dev.097212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Song HJ, Stevens CF, Gage FH. Neural stem cells from adult hippocampus develop essential properties of functional CNS neurons. Nat Neurosci. 2002;5:438–445. doi: 10.1038/nn844. [DOI] [PubMed] [Google Scholar]
- 31.Kempermann G, Gast D, Kronenberg G, Yamaguchi M, Gage FH. Early determination and long-term persistence of adult-generated new neurons in the hippocampus of mice. Development. 2003;130:391–399. doi: 10.1242/dev.00203. [DOI] [PubMed] [Google Scholar]
- 32.Kempermann G, Jessberger S, Steiner B, Kronenberg G. Milestones of neuronal development in the adult hippocampus. Trends Neurosci. 2004;27:447–452. doi: 10.1016/j.tins.2004.05.013. [DOI] [PubMed] [Google Scholar]
- 33.von Bohlen Und Halbach O. Immunohistological markers for staging neurogenesis in adult hippocampus. Cell Tissue Res. 2007;329:409–420. doi: 10.1007/s00441-007-0432-4. [DOI] [PubMed] [Google Scholar]
- 34.Magavi SS, Macklis JD. Immunocytochemical analysis of neuronal differentiation. Methods Mol Biol. 2008;438:345–352. doi: 10.1007/978-1-59745-133-8_26. [DOI] [PubMed] [Google Scholar]
- 35.von Bohlen Und Halbach O. Immunocytochemical identification of Mammalian differentiating neuronsin the process of adult neurogenesis in the hippocampus dentate gyrus. In: Merighi A, Lossi L, editors. Immunocytochemistry and related techniques. New York, NY: Humana Press, Springer Science Business Media; 2015. pp. 109–122. [Google Scholar]
- 36.Alvarez-Buylla A, Seri B, Doetsch F. Identification of neural stem cells in the adult vertebrate brain. Brain Res Bull. 2002;57:751–758. doi: 10.1016/s0361-9230(01)00770-5. [DOI] [PubMed] [Google Scholar]
- 37.Alvarez-Buylla A, Garcia-Verdugo JM. Neurogenesis in adult subventricular zone. J Neurosci. 2002;22:629–634. doi: 10.1523/JNEUROSCI.22-03-00629.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Urbán N, Guillemot F. Neurogenesis in the embryonic and adult brain: same regulators, different roles. Front Cell Neurosci. 2014;8:396. doi: 10.3389/fncel.2014.00396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Götz M, Huttner WB. The cell biology of neurogenesis. Nat Rev Mol Cell Biol. 2005;6:777–788. doi: 10.1038/nrm1739. [DOI] [PubMed] [Google Scholar]
- 40.Gilyarov AV. Nestin in central nervous system cells. Neurosci Behav Physiol. 2008;38:165–169. doi: 10.1007/s11055-008-0025-z. [DOI] [PubMed] [Google Scholar]
- 41.Francis F, Koulakoff A, Boucher D, Chafey P, Schaar B, Vinet MC, Friocourt G, McDonnell N, Reiner O, Kahn A, et al. Doublecortin is a developmentally regulated, microtubule-associated protein expressed in migrating and differentiating neurons. Neuron. 1999;23:247–256. doi: 10.1016/s0896-6273(00)80777-1. [DOI] [PubMed] [Google Scholar]
- 42.Brown JP, Couillard-Després S, Cooper-Kuhn CM, Winkler J, Aigner L, Kuhn HG. Transient expression of doublecortin during adult neurogenesis. J Comp Neurol. 2003;467:1–10. doi: 10.1002/cne.10874. [DOI] [PubMed] [Google Scholar]
- 43.Ball NS, King PH. Neuron-specific hel-N1 and HuD as novel molecular markers of neuroblastoma: a correlation of HuD messenger RNA levels with favorable prognostic features. Clin Cancer Res. 1997;3:1859–1865. [PubMed] [Google Scholar]
- 44.Theele DP, Streit WJ. A chronicle of microglial ontogeny. Glia. 1993;7:5–8. doi: 10.1002/glia.440070104. [DOI] [PubMed] [Google Scholar]
- 45.Mezey E, Chandross KJ, Harta G, Maki RA, McKercher SR. Turning blood into brain: cells bearing neuronal antigens generated in vivo from bone marrow. Science. 2000;290:1779–1782. doi: 10.1126/science.290.5497.1779. [DOI] [PubMed] [Google Scholar]
- 46.Cogle CR, Yachnis AT, Laywell ED, Zander DS, Wingard JR, Steindler DA, Scott EW. Bone marrow transdifferentiation in brain after transplantation: a retrospective study. Lancet. 2004;363:1432–1437. doi: 10.1016/S0140-6736(04)16102-3. [DOI] [PubMed] [Google Scholar]
- 47.Brazelton TR, Rossi FM, Keshet GI, Blau HM. From marrow to brain: expression of neuronal phenotypes in adult mice. Science. 2000;290:1775–1779. doi: 10.1126/science.290.5497.1775. [DOI] [PubMed] [Google Scholar]
- 48.Eglitis MA, Mezey E. Hematopoietic cells differentiate into both microglia and macroglia in the brains of adult mice. Proc Natl Acad Sci USA. 1997;94:4080–4085. doi: 10.1073/pnas.94.8.4080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Louboutin JP, Chekmasova AA, Marusich E, Chowdhury JR, Strayer DS. Efficient CNS gene delivery by intravenous injection. Nat Methods. 2010;7:905–907. doi: 10.1038/nmeth.1518. [DOI] [PubMed] [Google Scholar]
- 50.Louboutin JP. Immunocytochemical assessement of blood-brain barrier: structure, function, and damage. In: Merighi A, Lossi L, editors. Immunocytochemistry and related techniques. New York, NY: Humana Press, Springer Science Business Media; 2015. pp. 225–253. [Google Scholar]
- 51.Louboutin JP, Liu B, Reyes BA, Van Bockstaele EJ, Strayer DS. Rat bone marrow progenitor cells transduced in situ by rSV40 vectors differentiate into multiple central nervous system cell lineages. Stem Cells. 2006;24:2801–2809. doi: 10.1634/stemcells.2006-0124. [DOI] [PubMed] [Google Scholar]
- 52.Louboutin JP, Chekmasova AA, Liu B, Strayer DS. Efficient in vivo gene transfer into rat bone marrow progenitor cells using SV40-derived viral vectors: migration and differentiation of transduced bone marrow resident cells to other organs. In: Greene AL, editor. Encyclopedia of stem cell research. Hauppauge, NY: Nova Science Publishers; 2012. [Google Scholar]
- 53.Cordelier P, Van Bockstaele E, Calarota SA, Strayer DS. Inhibiting AIDS in the central nervous system: gene delivery to protect neurons from HIV. Mol Ther. 2003;7:801–810. doi: 10.1016/s1525-0016(03)00093-5. [DOI] [PubMed] [Google Scholar]
- 54.Sauter BV, Parashar B, Chowdhury NR, Kadakol A, Ilan Y, Singh H, Milano J, Strayer DS, Chowdhury JR. A replication-deficient rSV40 mediates liver-directed gene transfer and a long-term amelioration of jaundice in gunn rats. Gastroenterology. 2000;119:1348–1357. doi: 10.1053/gast.2000.19577. [DOI] [PubMed] [Google Scholar]
- 55.Strayer DS, Milano J. SV40 mediates stable gene transfer in vivo. Gene Ther. 1996;3:581–587. [PubMed] [Google Scholar]
- 56.Scheffler B, Horn M, Blumcke I, Laywell ED, Coomes D, Kukekov VG, Steindler DA. Marrow-mindedness: a perspective on neuropoiesis. Trends Neurosci. 1999;22:348–357. doi: 10.1016/s0166-2236(99)01416-2. [DOI] [PubMed] [Google Scholar]
- 57.Scharfman HE, Goodman JH, Sollas AL. Granule-like neurons at the hilar/CA3 border after status epilepticus and their synchrony with area CA3 pyramidal cells: functional implications of seizure-induced neurogenesis. J Neurosci. 2000;20:6144–6158. doi: 10.1523/JNEUROSCI.20-16-06144.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Jin K, Minami M, Lan JQ, Mao XO, Batteur S, Simon RP, Greenberg DA. Neurogenesis in dentate subgranular zone and rostral subventricular zone after focal cerebral ischemia in the rat. Proc Natl Acad Sci USA. 2001;98:4710–4715. doi: 10.1073/pnas.081011098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.van Praag H, Schinder AF, Christie BR, Toni N, Palmer TD, Gage FH. Functional neurogenesis in the adult hippocampus. Nature. 2002;415:1030–1034. doi: 10.1038/4151030a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Magavi SS, Leavitt BR, Macklis JD. Induction of neurogenesis in the neocortex of adult mice. Nature. 2000;405:951–955. doi: 10.1038/35016083. [DOI] [PubMed] [Google Scholar]
- 61.Tamaki S, Eckert K, He D, Sutton R, Doshe M, Jain G, Tushinski R, Reitsma M, Harris B, Tsukamoto A, et al. Engraftment of sorted/expanded human central nervous system stem cells from fetal brain. J Neurosci Res. 2002;69:976–986. doi: 10.1002/jnr.10412. [DOI] [PubMed] [Google Scholar]
- 62.Weimann JM, Johansson CB, Trejo A, Blau HM. Stable reprogrammed heterokaryons form spontaneously in Purkinje neurons after bone marrow transplant. Nat Cell Biol. 2003;5:959–966. doi: 10.1038/ncb1053. [DOI] [PubMed] [Google Scholar]
- 63.Weimann JM, Charlton CA, Brazelton TR, Hackman RC, Blau HM. Contribution of transplanted bone marrow cells to Purkinje neurons in human adult brains. Proc Natl Acad Sci USA. 2003;100:2088–2093. doi: 10.1073/pnas.0337659100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Alvarez-Dolado M, Pardal R, Garcia-Verdugo JM, Fike JR, Lee HO, Pfeffer K, Lois C, Morrison SJ, Alvarez-Buylla A. Fusion of bone-marrow-derived cells with Purkinje neurons, cardiomyocytes and hepatocytes. Nature. 2003;425:968–973. doi: 10.1038/nature02069. [DOI] [PubMed] [Google Scholar]
- 65.Sanchez-Ramos J, Song S, Cardozo-Pelaez F, Hazzi C, Stedeford T, Willing A, Freeman TB, Saporta S, Janssen W, Patel N, et al. Adult bone marrow stromal cells differentiate into neural cells in vitro. Exp Neurol. 2000;164:247–256. doi: 10.1006/exnr.2000.7389. [DOI] [PubMed] [Google Scholar]
- 66.Kopen GC, Prockop DJ, Phinney DG. Marrow stromal cells migrate throughout forebrain and cerebellum, and they differentiate into astrocytes after injection into neonatal mouse brains. Proc Natl Acad Sci USA. 1999;96:10711–10716. doi: 10.1073/pnas.96.19.10711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Alvarez P, Carrillo E, Vélez C, Hita-Contreras F, Martínez-Amat A, Rodríguez-Serrano F, Boulaiz H, Ortiz R, Melguizo C, Prados J, et al. Regulatory systems in bone marrow for hematopoietic stem/progenitor cells mobilization and homing. Biomed Res Int. 2013;2013:312656. doi: 10.1155/2013/312656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Prockop DJ. Marrow stromal cells as stem cells for nonhematopoietic tissues. Science. 1997;276:71–74. doi: 10.1126/science.276.5309.71. [DOI] [PubMed] [Google Scholar]
- 69.Palmer TD, Willhoite AR, Gage FH. Vascular niche for adult hippocampal neurogenesis. J Comp Neurol. 2000;425:479–494. doi: 10.1002/1096-9861(20001002)425:4<479::aid-cne2>3.0.co;2-3. [DOI] [PubMed] [Google Scholar]
- 70.Shors TJ, Miesegaes G, Beylin A, Zhao M, Rydel T, Gould E. Neurogenesis in the adult is involved in the formation of trace memories. Nature. 2001;410:372–376. doi: 10.1038/35066584. [DOI] [PubMed] [Google Scholar]
- 71.Ma Q, Jones D, Borghesani PR, Segal RA, Nagasawa T, Kishimoto T, Bronson RT, Springer TA. Impaired B-lymphopoiesis, myelopoiesis, and derailed cerebellar neuron migration in CXCR4- and SDF-1-deficient mice. Proc Natl Acad Sci USA. 1998;95:9448–9453. doi: 10.1073/pnas.95.16.9448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Klein RS, Rubin JB, Gibson HD, DeHaan EN, Alvarez-Hernandez X, Segal RA, Luster AD. SDF-1 alpha induces chemotaxis and enhances Sonic hedgehog-induced proliferation of cerebellar granule cells. Development. 2001;128:1971–1981. doi: 10.1242/dev.128.11.1971. [DOI] [PubMed] [Google Scholar]
- 73.Hatch HM, Zheng D, Jorgensen ML, Petersen BE. SDF-1alpha/CXCR4: a mechanism for hepatic oval cell activation and bone marrow stem cell recruitment to the injured liver of rats. Cloning Stem Cells. 2002;4:339–351. doi: 10.1089/153623002321025014. [DOI] [PubMed] [Google Scholar]
- 74.Wysoczynski M, Ratajczak J, Pedziwiatr D, Rokosh G, Bolli R, Ratajczak MZ. Identification of heme oxygenase 1 (HO-1) as a novel negative regulator of mobilization of hematopoietic stem/progenitor cells. Stem Cell Rev. 2015;11:110–118. doi: 10.1007/s12015-014-9547-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Lévesque JP, Helwani FM, Winkler IG. The endosteal ‘osteoblastic’ niche and its role in hematopoietic stem cell homing and mobilization. Leukemia. 2010;24:1979–1992. doi: 10.1038/leu.2010.214. [DOI] [PubMed] [Google Scholar]
- 76.Ratajczak MZ, Kim CH, Wojakowski W, Janowska-Wieczorek A, Kucia M, Ratajczak J. Innate immunity as orchestrator of stem cell mobilization. Leukemia. 2010;24:1667–1675. doi: 10.1038/leu.2010.162. [DOI] [PubMed] [Google Scholar]
- 77.Bonig H, Papayannopoulou T. Hematopoietic stem cell mobilization: updated conceptual renditions. Leukemia. 2013;27:24–31. doi: 10.1038/leu.2012.254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Kränkel N, Spinetti G, Amadesi S, Madeddu P. Targeting stem cell niches and trafficking for cardiovascular therapy. Pharmacol Ther. 2011;129:62–81. doi: 10.1016/j.pharmthera.2010.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Campbell TB, Hangoc G, Liu Y, Pollok K, Broxmeyer HE. Inhibition of CD26 in human cord blood CD34+ cells enhances their engraftment of nonobese diabetic/severe combined immunodeficiency mice. Stem Cells Dev. 2007;16:347–354. doi: 10.1089/scd.2007.9995. [DOI] [PubMed] [Google Scholar]
- 80.Sugiyama T, Kohara H, Noda M, Nagasawa T. Maintenance of the hematopoietic stem cell pool by CXCL12-CXCR4 chemokine signaling in bone marrow stromal cell niches. Immunity. 2006;25:977–988. doi: 10.1016/j.immuni.2006.10.016. [DOI] [PubMed] [Google Scholar]
- 81.Méndez-Ferrer S, Lucas D, Battista M, Frenette PS. Haematopoietic stem cell release is regulated by circadian oscillations. Nature. 2008;452:442–447. doi: 10.1038/nature06685. [DOI] [PubMed] [Google Scholar]
- 82.Balabanian K, Lagane B, Infantino S, Chow KY, Harriague J, Moepps B, Arenzana-Seisdedos F, Thelen M, Bachelerie F. The chemokine SDF-1/CXCL12 binds to and signals through the orphan receptor RDC1 in T lymphocytes. J Biol Chem. 2005;280:35760–35766. doi: 10.1074/jbc.M508234200. [DOI] [PubMed] [Google Scholar]
- 83.Ma Q, Jones D, Springer TA. The chemokine receptor CXCR4 is required for the retention of B lineage and granulocytic precursors within the bone marrow microenvironment. Immunity. 1999;10:463–471. doi: 10.1016/s1074-7613(00)80046-1. [DOI] [PubMed] [Google Scholar]
- 84.Flomenberg N, Devine SM, Dipersio JF, Liesveld JL, McCarty JM, Rowley SD, Vesole DH, Badel K, Calandra G. The use of AMD3100 plus G-CSF for autologous hematopoietic progenitor cell mobilization is superior to G-CSF alone. Blood. 2005;106:1867–1874. doi: 10.1182/blood-2005-02-0468. [DOI] [PubMed] [Google Scholar]
- 85.Walter DH, Rochwalsky U, Reinhold J, Seeger F, Aicher A, Urbich C, Spyridopoulos I, Chun J, Brinkmann V, Keul P, et al. Sphingosine-1-phosphate stimulates the functional capacity of progenitor cells by activation of the CXCR4-dependent signaling pathway via the S1P3 receptor. Arterioscler Thromb Vasc Biol. 2007;27:275–282. doi: 10.1161/01.ATV.0000254669.12675.70. [DOI] [PubMed] [Google Scholar]
- 86.Kim CH, Wu W, Wysoczynski M, Abdel-Latif A, Sunkara M, Morris A, Kucia M, Ratajczak J, Ratajczak MZ. Conditioning for hematopoietic transplantation activates the complement cascade and induces a proteolytic environment in bone marrow: a novel role for bioactive lipids and soluble C5b-C9 as homing factors. Leukemia. 2012;26:106–116. doi: 10.1038/leu.2011.185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Borkowska S, Suszynska M, Mierzejewska K, Ismail A, Budkowska M, Salata D, Dolegowska B, Kucia M, Ratajczak J, Ratajczak MZ. Novel evidence that crosstalk between the complement, coagulation and fibrinolysis proteolytic cascades is involved in mobilization of hematopoietic stem/progenitor cells (HSPCs) Leukemia. 2014;28:2148–2154. doi: 10.1038/leu.2014.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Lee HM, Wu W, Wysoczynski M, Liu R, Zuba-Surma EK, Kucia M, Ratajczak J, Ratajczak MZ. Impaired mobilization of hematopoietic stem/progenitor cells in C5-deficient mice supports the pivotal involvement of innate immunity in this process and reveals novel promobilization effects of granulocytes. Leukemia. 2009;23:2052–2062. doi: 10.1038/leu.2009.158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Ratajczak MZ, Lee H, Wysoczynski M, Wan W, Marlicz W, Laughlin MJ, Kucia M, Janowska-Wieczorek A, Ratajczak J. Novel insight into stem cell mobilization-plasma sphingosine-1-phosphate is a major chemoattractant that directs the egress of hematopoietic stem progenitor cells from the bone marrow and its level in peripheral blood increases during mobilization due to activation of complement cascade/membrane attack complex. Leukemia. 2010;24:976–985. doi: 10.1038/leu.2010.53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Gong Y, Hoover-Plow J. The plasminogen system in regulating stem cell mobilization. J Biomed Biotechnol. 2012;2012:437920. doi: 10.1155/2012/437920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Paine A, Eiz-Vesper B, Blasczyk R, Immenschuh S. Signaling to heme oxygenase-1 and its anti-inflammatory therapeutic potential. Biochem Pharmacol. 2010;80:1895–1903. doi: 10.1016/j.bcp.2010.07.014. [DOI] [PubMed] [Google Scholar]
- 92.Kinderlerer AR, Pombo Gregoire I, Hamdulay SS, Ali F, Steinberg R, Silva G, Ali N, Wang B, Haskard DO, Soares MP, et al. Heme oxygenase-1 expression enhances vascular endothelial resistance to complement-mediated injury through induction of decay-accelerating factor: a role for increased bilirubin and ferritin. Blood. 2009;113:1598–1607. doi: 10.1182/blood-2008-04-152934. [DOI] [PubMed] [Google Scholar]
- 93.Lin HH, Chen YH, Chang PF, Lee YT, Yet SF, Chau LY. Heme oxygenase-1 promotes neovascularization in ischemic heart by coinduction of VEGF and SDF-1. J Mol Cell Cardiol. 2008;45:44–55. doi: 10.1016/j.yjmcc.2008.04.011. [DOI] [PubMed] [Google Scholar]
- 94.Freitas A, Alves-Filho JC, Secco DD, Neto AF, Ferreira SH, Barja-Fidalgo C, Cunha FQ. Heme oxygenase/carbon monoxide-biliverdin pathway down regulates neutrophil rolling, adhesion and migration in acute inflammation. Br J Pharmacol. 2006;149:345–354. doi: 10.1038/sj.bjp.0706882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Winkler IG, Hendy J, Coughlin P, Horvath A, Lévesque JP. Serine protease inhibitors serpina1 and serpina3 are down-regulated in bone marrow during hematopoietic progenitor mobilization. J Exp Med. 2005;201:1077–1088. doi: 10.1084/jem.20042299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Janowska-Wieczorek A, Marquez LA, Dobrowsky A, Ratajczak MZ, Cabuhat ML. Differential MMP and TIMP production by human marrow and peripheral blood CD34(+) cells in response to chemokines. Exp Hematol. 2000;28:1274–1285. doi: 10.1016/s0301-472x(00)00532-4. [DOI] [PubMed] [Google Scholar]
- 97.Okay E, Simsek T, Subasi C, Gunes A, Duruksu G, Gurbuz Y, Gacar G, Karaoz E. Cross effects of resveratrol and mesenchymal stem cells on liver regeneration and homing in partially hepatectomized rats. Stem Cell Rev. 2015;11:322–331. doi: 10.1007/s12015-014-9572-6. [DOI] [PubMed] [Google Scholar]
- 98.Steindler DA, Pincus DW. Stem cells and neuropoiesis in the adult human brain. Lancet. 2002;359:1047–1054. doi: 10.1016/S0140-6736(02)08096-0. [DOI] [PubMed] [Google Scholar]
- 99.Zindy F, Cunningham JJ, Sherr CJ, Jogal S, Smeyne RJ, Roussel MF. Postnatal neuronal proliferation in mice lacking Ink4d and Kip1 inhibitors of cyclin-dependent kinases. Proc Natl Acad Sci USA. 1999;96:13462–13467. doi: 10.1073/pnas.96.23.13462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Yang XW, Zhong R, Heintz N. Granule cell specification in the developing mouse brain as defined by expression of the zinc finger transcription factor RU49. Development. 1996;122:555–566. doi: 10.1242/dev.122.2.555. [DOI] [PubMed] [Google Scholar]
- 101.Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663–676. doi: 10.1016/j.cell.2006.07.024. [DOI] [PubMed] [Google Scholar]
- 102.Wakao S, Kitada M, Kuroda Y, Shigemoto T, Matsuse D, Akashi H, Tanimura Y, Tsuchiyama K, Kikuchi T, Goda M, et al. Multilineage-differentiating stress-enduring (Muse) cells are a primary source of induced pluripotent stem cells in human fibroblasts. Proc Natl Acad Sci USA. 2011;108:9875–9880. doi: 10.1073/pnas.1100816108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Houlihan DD, Mabuchi Y, Morikawa S, Niibe K, Araki D, Suzuki S, Okano H, Matsuzaki Y. Isolation of mouse mesenchymal stem cells on the basis of expression of Sca-1 and PDGFR-α. Nat Protoc. 2012;7:2103–2111. doi: 10.1038/nprot.2012.125. [DOI] [PubMed] [Google Scholar]
- 104.Niibe K, Kawamura Y, Araki D, Morikawa S, Miura K, Suzuki S, Shimmura S, Sunabori T, Mabuchi Y, Nagai Y, et al. Purified mesenchymal stem cells are an efficient source for iPS cell induction. PLoS One. 2011;6:e17610. doi: 10.1371/journal.pone.0017610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Caplan AI. Mesenchymal stem cells. J Orthop Res. 1991;9:641–650. doi: 10.1002/jor.1100090504. [DOI] [PubMed] [Google Scholar]
- 106.Fasano CA, Chambers SM, Lee G, Tomishima MJ, Studer L. Efficient derivation of functional floor plate tissue from human embryonic stem cells. Cell Stem Cell. 2010;6:336–347. doi: 10.1016/j.stem.2010.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 107.Kriks S, Shim JW, Piao J, Ganat YM, Wakeman DR, Xie Z, Carrillo-Reid L, Auyeung G, Antonacci C, Buch A, et al. Dopamine neurons derived from human ES cells efficiently engraft in animal models of Parkinson’s disease. Nature. 2011;480:547–551. doi: 10.1038/nature10648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Wernig M, Zhao JP, Pruszak J, Hedlund E, Fu D, Soldner F, Broccoli V, Constantine-Paton M, Isacson O, Jaenisch R. Neurons derived from reprogrammed fibroblasts functionally integrate into the fetal brain and improve symptoms of rats with Parkinson’s disease. Proc Natl Acad Sci USA. 2008;105:5856–5861. doi: 10.1073/pnas.0801677105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Vierbuchen T, Ostermeier A, Pang ZP, Kokubu Y, Südhof TC, Wernig M. Direct conversion of fibroblasts to functional neurons by defined factors. Nature. 2010;463:1035–1041. doi: 10.1038/nature08797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Pang ZP, Yang N, Vierbuchen T, Ostermeier A, Fuentes DR, Yang TQ, Citri A, Sebastiano V, Marro S, Südhof TC, et al. Induction of human neuronal cells by defined transcription factors. Nature. 2011;476:220–223. doi: 10.1038/nature10202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Pfisterer U, Kirkeby A, Torper O, Wood J, Nelander J, Dufour A, Björklund A, Lindvall O, Jakobsson J, Parmar M. Direct conversion of human fibroblasts to dopaminergic neurons. Proc Natl Acad Sci USA. 2011;108:10343–10348. doi: 10.1073/pnas.1105135108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Caiazzo M, Dell’Anno MT, Dvoretskova E, Lazarevic D, Taverna S, Leo D, Sotnikova TD, Menegon A, Roncaglia P, Colciago G, et al. Direct generation of functional dopaminergic neurons from mouse and human fibroblasts. Nature. 2011;476:224–227. doi: 10.1038/nature10284. [DOI] [PubMed] [Google Scholar]
- 113.Lee HS, Bae EJ, Yi SH, Shim JW, Jo AY, Kang JS, Yoon EH, Rhee YH, Park CH, Koh HC, et al. Foxa2 and Nurr1 synergistically yield A9 nigral dopamine neurons exhibiting improved differentiation, function, and cell survival. Stem Cells. 2010;28:501–512. doi: 10.1002/stem.294. [DOI] [PubMed] [Google Scholar]
- 114.Nakatani T, Kumai M, Mizuhara E, Minaki Y, Ono Y. Lmx1a and Lmx1b cooperate with Foxa2 to coordinate the specification of dopaminergic neurons and control of floor plate cell differentiation in the developing mesencephalon. Dev Biol. 2010;339:101–113. doi: 10.1016/j.ydbio.2009.12.017. [DOI] [PubMed] [Google Scholar]
- 115.Chung S, Leung A, Han BS, Chang MY, Moon JI, Kim CH, Hong S, Pruszak J, Isacson O, Kim KS. Wnt1-lmx1a forms a novel autoregulatory loop and controls midbrain dopaminergic differentiation synergistically with the SHH-FoxA2 pathway. Cell Stem Cell. 2009;5:646–658. doi: 10.1016/j.stem.2009.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Andersson E, Tryggvason U, Deng Q, Friling S, Alekseenko Z, Robert B, Perlmann T, Ericson J. Identification of intrinsic determinants of midbrain dopamine neurons. Cell. 2006;124:393–405. doi: 10.1016/j.cell.2005.10.037. [DOI] [PubMed] [Google Scholar]
- 117.Cai J, Donaldson A, Yang M, German MS, Enikolopov G, Iacovitti L. The role of Lmx1a in the differentiation of human embryonic stem cells into midbrain dopamine neurons in culture and after transplantation into a Parkinson’s disease model. Stem Cells. 2009;27:220–229. doi: 10.1634/stemcells.2008-0734. [DOI] [PubMed] [Google Scholar]
- 118.Roessler R, Boddeke E, Copray S. Induced pluripotent stem cell technology and direct conversion: new possibilities to study and treat Parkinson’s disease. Stem Cell Rev. 2013;9:505–513. doi: 10.1007/s12015-012-9369-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Baeyens L, Bouwens L. Can beta-cells be derived from exocrine pancreas? Diabetes Obes Metab. 2008;10 Suppl 4:170–178. doi: 10.1111/j.1463-1326.2008.00949.x. [DOI] [PubMed] [Google Scholar]
- 120.Louboutin JP, Liu B, Chekmasova AA, Reyes BA, van Bockstaele EJ, Strayer DS. Delivering genes to the organ-localized immune system: long-term results of direct intramarrow transduction. J Gene Med. 2007;9:843–851. doi: 10.1002/jgm.1084. [DOI] [PubMed] [Google Scholar]
- 121.Castro RF, Jackson KA, Goodell MA, Robertson CS, Liu H, Shine HD. Failure of bone marrow cells to transdifferentiate into neural cells in vivo. Science. 2002;297:1299. doi: 10.1126/science.297.5585.1299. [DOI] [PubMed] [Google Scholar]
- 122.Wagers AJ, Sherwood RI, Christensen JL, Weissman IL. Little evidence for developmental plasticity of adult hematopoietic stem cells. Science. 2002;297:2256–2259. doi: 10.1126/science.1074807. [DOI] [PubMed] [Google Scholar]
- 123.Yagi T, McMahon EJ, Takikita S, Mohri I, Matsushima GK, Suzuki K. Fate of donor hematopoietic cells in demyelinating mutant mouse, twitcher, following transplantation of GFP+ bone marrow cells. Neurobiol Dis. 2004;16:98–109. doi: 10.1016/j.nbd.2004.01.002. [DOI] [PubMed] [Google Scholar]
- 124.Duan YY, Wu J, Zhu JL, Liu SL, Ozaki I, Strayer DS, Zern MA. Gene therapy for human alpha1-antitrypsin deficiency in an animal model using SV40-derived vectors. Gastroenterology. 2004;127:1222–1232. doi: 10.1053/j.gastro.2004.07.058. [DOI] [PubMed] [Google Scholar]
- 125.Mezey E, Key S, Vogelsang G, Szalayova I, Lange GD, Crain B. Transplanted bone marrow generates new neurons in human brains. Proc Natl Acad Sci USA. 2003;100:1364–1369. doi: 10.1073/pnas.0336479100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Mezey E, Nagy A, Szalayova I, Key S, Bratincsak A, Baffi J, Shahar T. Comment on “Failure of bone marrow cells to transdifferentiate into neural cells in vivo”. Science. 2003;299:1184; author reply 1184. doi: 10.1126/science.1079318. [DOI] [PubMed] [Google Scholar]
- 127.Lagasse E, Connors H, Al-Dhalimy M, Reitsma M, Dohse M, Osborne L, Wang X, Finegold M, Weissman IL, Grompe M. Purified hematopoietic stem cells can differentiate into hepatocytes in vivo. Nat Med. 2000;6:1229–1234. doi: 10.1038/81326. [DOI] [PubMed] [Google Scholar]
- 128.Mohapel P, Ekdahl CT, Lindvall O. Status epilepticus severity influences the long-term outcome of neurogenesis in the adult dentate gyrus. Neurobiol Dis. 2004;15:196–205. doi: 10.1016/j.nbd.2003.11.010. [DOI] [PubMed] [Google Scholar]
- 129.Vezzani A, Granata T. Brain inflammation in epilepsy: experimental and clinical evidence. Epilepsia. 2005;46:1724–1743. doi: 10.1111/j.1528-1167.2005.00298.x. [DOI] [PubMed] [Google Scholar]
- 130.Johnson EA, Kan RK. The acute phase response and soman-induced status epilepticus: temporal, regional and cellular changes in rat brain cytokine concentrations. J Neuroinflammation. 2010;7:40. doi: 10.1186/1742-2094-7-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Costa-Ferro ZS, Vitola AS, Pedroso MF, Cunha FB, Xavier LL, Machado DC, Soares MB, Ribeiro-dos-Santos R, DaCosta JC. Prevention of seizures and reorganization of hippocampal functions by transplantation of bone marrow cells in the acute phase of experimental epilepsy. Seizure. 2010;19:84–92. doi: 10.1016/j.seizure.2009.12.003. [DOI] [PubMed] [Google Scholar]
- 132.Louboutin JP, Chekmasova A, Marusich E, Agrawal L, Strayer DS. Role of CCR5 and its ligands in the control of vascular inflammation and leukocyte recruitment required for acute excitotoxic seizure induction and neural damage. FASEB J. 2011;25:737–753. doi: 10.1096/fj.10-161851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Jin K, Sun Y, Xie L, Peel A, Mao XO, Batteur S, Greenberg DA. Directed migration of neuronal precursors into the ischemic cerebral cortex and striatum. Mol Cell Neurosci. 2003;24:171–189. doi: 10.1016/s1044-7431(03)00159-3. [DOI] [PubMed] [Google Scholar]
- 134.Jin K, Peel AL, Mao XO, Xie L, Cottrell BA, Henshall DC, Greenberg DA. Increased hippocampal neurogenesis in Alzheimer’s disease. Proc Natl Acad Sci USA. 2004;101:343–347. doi: 10.1073/pnas.2634794100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Jin K, Wang X, Xie L, Mao XO, Zhu W, Wang Y, Shen J, Mao Y, Banwait S, Greenberg DA. Evidence for stroke-induced neurogenesis in the human brain. Proc Natl Acad Sci USA. 2006;103:13198–13202. doi: 10.1073/pnas.0603512103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Spritzer MD, Ibler E, Inglis W, Curtis MG. Testosterone and social isolation influence adult neurogenesis in the dentate gyrus of male rats. Neuroscience. 2011;195:180–190. doi: 10.1016/j.neuroscience.2011.08.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Monje ML, Toda H, Palmer TD. Inflammatory blockade restores adult hippocampal neurogenesis. Science. 2003;302:1760–1765. doi: 10.1126/science.1088417. [DOI] [PubMed] [Google Scholar]
- 138.Andjelkovic AV, Spencer DD, Pachter JS. Visualization of chemokine binding sites on human brain microvessels. J Cell Biol. 1999;145:403–412. doi: 10.1083/jcb.145.2.403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Andjelkovic AV, Pachter JS. Characterization of binding sites for chemokines MCP-1 and MIP-1alpha on human brain microvessels. J Neurochem. 2000;75:1898–1906. doi: 10.1046/j.1471-4159.2000.0751898.x. [DOI] [PubMed] [Google Scholar]
- 140.Man S, Ubogu EE, Ransohoff RM. Inflammatory cell migration into the central nervous system: a few new twists on an old tale. Brain Pathol. 2007;17:243–250. doi: 10.1111/j.1750-3639.2007.00067.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Ubogu EE, Callahan MK, Tucky BH, Ransohoff RM. Determinants of CCL5-driven mononuclear cell migration across the blood-brain barrier. Implications for therapeutically modulating neuroinflammation. J Neuroimmunol. 2006;179:132–144. doi: 10.1016/j.jneuroim.2006.06.004. [DOI] [PubMed] [Google Scholar]
- 142.Ubogu EE, Callahan MK, Tucky BH, Ransohoff RM. CCR5 expression on monocytes and T cells: modulation by transmigration across the blood-brain barrier in vitro. Cell Immunol. 2006;243:19–29. doi: 10.1016/j.cellimm.2006.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Zozulya AL, Reinke E, Baiu DC, Karman J, Sandor M, Fabry Z. Dendritic cell transmigration through brain microvessel endothelium is regulated by MIP-1alpha chemokine and matrix metalloproteinases. J Immunol. 2007;178:520–529. doi: 10.4049/jimmunol.178.1.520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Schreiber TH, Shinder V, Cain DW, Alon R, Sackstein R. Shear flow-dependent integration of apical and subendothelial chemokines in T-cell transmigration: implications for locomotion and the multistep paradigm. Blood. 2007;109:1381–1386. doi: 10.1182/blood-2006-07-032995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Marusich E, Louboutin JP, Chekmasova AA, Strayer DS. Lymphocyte adhesion to CCR5 ligands is reduced by anti-CCR5 gene delivery. J Neurol Sci. 2011;308:25–27. doi: 10.1016/j.jns.2011.06.039. [DOI] [PubMed] [Google Scholar]
- 146.Fabene PF, Navarro Mora G, Martinello M, Rossi B, Merigo F, Ottoboni L, Bach S, Angiari S, Benati D, Chakir A, et al. A role for leukocyte-endothelial adhesion mechanisms in epilepsy. Nat Med. 2008;14:1377–1383. doi: 10.1038/nm.1878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Marchi N, Angelov L, Masaryk T, Fazio V, Granata T, Hernandez N, Hallene K, Diglaw T, Franic L, Najm I, et al. Seizure-promoting effect of blood-brain barrier disruption. Epilepsia. 2007;48:732–742. doi: 10.1111/j.1528-1167.2007.00988.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.van Vliet EA, da Costa Araújo S, Redeker S, van Schaik R, Aronica E, Gorter JA. Blood-brain barrier leakage may lead to progression of temporal lobe epilepsy. Brain. 2007;130:521–534. doi: 10.1093/brain/awl318. [DOI] [PubMed] [Google Scholar]
- 149.Bolton SJ, Perry VH. Differential blood-brain barrier breakdown and leucocyte recruitment following excitotoxic lesions in juvenile and adult rats. Exp Neurol. 1998;154:231–240. doi: 10.1006/exnr.1998.6927. [DOI] [PubMed] [Google Scholar]
- 150.Parathath SR, Parathath S, Tsirka SE. Nitric oxide mediates neurodegeneration and breakdown of the blood-brain barrier in tPA-dependent excitotoxic injury in mice. J Cell Sci. 2006;119:339–349. doi: 10.1242/jcs.02734. [DOI] [PubMed] [Google Scholar]
- 151.Manley NC, Bertrand AA, Kinney KS, Hing TC, Sapolsky RM. Characterization of monocyte chemoattractant protein-1 expression following a kainate model of status epilepticus. Brain Res. 2007;1182:138–143. doi: 10.1016/j.brainres.2007.08.092. [DOI] [PubMed] [Google Scholar]
- 152.Xu JH, Long L, Tang YC, Zhang JT, Hut HT, Tang FR. CCR3, CCR2A and macrophage inflammatory protein (MIP)-1a, monocyte chemotactic protein-1 (MCP-1) in the mouse hippocampus during and after pilocarpine-induced status epilepticus (PISE) Neuropathol Appl Neurobiol. 2009;35:496–514. doi: 10.1111/j.1365-2990.2009.01022.x. [DOI] [PubMed] [Google Scholar]
- 153.Greenwood J, Etienne-Manneville S, Adamson P, Couraud PO. Lymphocyte migration into the central nervous system: implication of ICAM-1 signalling at the blood-brain barrier. Vascul Pharmacol. 2002;38:315–322. doi: 10.1016/s1537-1891(02)00199-4. [DOI] [PubMed] [Google Scholar]
- 154.Tripathy D, Thirumangalakudi L, Grammas P. Expression of macrophage inflammatory protein 1-alpha is elevated in Alzheimer’s vessels and is regulated by oxidative stress. J Alzheimers Dis. 2007;11:447–455. doi: 10.3233/jad-2007-11405. [DOI] [PubMed] [Google Scholar]
- 155.Chui R, Dorovini-Zis K. Regulation of CCL2 and CCL3 expression in human brain endothelial cells by cytokines and lipopolysaccharide. J Neuroinflammation. 2010;7:1. doi: 10.1186/1742-2094-7-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Shukaliak JA, Dorovini-Zis K. Expression of the beta-chemokines RANTES and MIP-1 beta by human brain microvessel endothelial cells in primary culture. J Neuropathol Exp Neurol. 2000;59:339–352. doi: 10.1093/jnen/59.5.339. [DOI] [PubMed] [Google Scholar]
- 157.Subileau EA, Rezaie P, Davies HA, Colyer FM, Greenwood J, Male DK, Romero IA. Expression of chemokines and their receptors by human brain endothelium: implications for multiple sclerosis. J Neuropathol Exp Neurol. 2009;68:227–240. doi: 10.1097/NEN.0b013e318197eca7. [DOI] [PubMed] [Google Scholar]
- 158.Yabuuchi K, Minami M, Katsumata S, Satoh M. In situ hybridization study of interleukin-1 beta mRNA induced by kainic acid in the rat brain. Brain Res Mol Brain Res. 1993;20:153–161. doi: 10.1016/0169-328x(93)90121-5. [DOI] [PubMed] [Google Scholar]
- 159.De Simoni MG, Perego C, Ravizza T, Moneta D, Conti M, Marchesi F, De Luigi A, Garattini S, Vezzani A. Inflammatory cytokines and related genes are induced in the rat hippocampus by limbic status epilepticus. Eur J Neurosci. 2000;12:2623–2633. doi: 10.1046/j.1460-9568.2000.00140.x. [DOI] [PubMed] [Google Scholar]
- 160.Gorter JA, van Vliet EA, Aronica E, Breit T, Rauwerda H, Lopes da Silva FH, Wadman WJ. Potential new antiepileptogenic targets indicated by microarray analysis in a rat model for temporal lobe epilepsy. J Neurosci. 2006;26:11083–11110. doi: 10.1523/JNEUROSCI.2766-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Vezzani A, Conti M, De Luigi A, Ravizza T, Moneta D, Marchesi F, De Simoni MG. Interleukin-1beta immunoreactivity and microglia are enhanced in the rat hippocampus by focal kainate application: functional evidence for enhancement of electrographic seizures. J Neurosci. 1999;19:5054–5065. doi: 10.1523/JNEUROSCI.19-12-05054.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Briones MA, Phillips DJ, Renshaw MA, Hooper WC. Expression of chemokine by human coronary-artery and umbilical-vein endothelial cells and its regulation by inflammatory cytokines. Coron Artery Dis. 2001;12:179–186. doi: 10.1097/00019501-200105000-00004. [DOI] [PubMed] [Google Scholar]
- 163.Won SJ, Ko HW, Kim EY, Park EC, Huh K, Jung NP, Choi I, Oh YK, Shin HC, Gwag BJ. Nuclear factor kappa B-mediated kainate neurotoxicity in the rat and hamster hippocampus. Neuroscience. 1999;94:83–91. doi: 10.1016/s0306-4522(99)00196-7. [DOI] [PubMed] [Google Scholar]
- 164.Yeligar SM, Machida K, Tsukamoto H, Kalra VK. Ethanol augments RANTES/CCL5 expression in rat liver sinusoidal endothelial cells and human endothelial cells via activation of NF-kappa B, HIF-1 alpha, and AP-1. J Immunol. 2009;183:5964–5976. doi: 10.4049/jimmunol.0901564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Roebuck KA, Carpenter LR, Lakshminarayanan V, Page SM, Moy JN, Thomas LL. Stimulus-specific regulation of chemokine expression involves differential activation of the redox-responsive transcription factors AP-1 and NF-kappaB. J Leukoc Biol. 1999;65:291–298. doi: 10.1002/jlb.65.3.291. [DOI] [PubMed] [Google Scholar]
- 166.Murugesan G, Sandhya Rani MR, Gerber CE, Mukhopadhyay C, Ransohoff RM, Chisolm GM, Kottke-Marchant K. Lysophosphatidylcholine regulates human microvascular endothelial cell expression of chemokines. J Mol Cell Cardiol. 2003;35:1375–1384. doi: 10.1016/j.yjmcc.2003.08.004. [DOI] [PubMed] [Google Scholar]
- 167.Yegin A, Akbas SH, Ozben T, Korgun DK. Secretory phospholipase A2 and phospholipids in neural membranes in an experimental epilepsy model. Acta Neurol Scand. 2002;106:258–262. doi: 10.1034/j.1600-0404.2002.01238.x. [DOI] [PubMed] [Google Scholar]
- 168.Sandhya TL, Ong WY, Horrocks LA, Farooqui AA. A light and electron microscopic study of cytoplasmic phospholipase A2 and cyclooxygenase-2 in the hippocampus after kainate lesions. Brain Res. 1998;788:223–231. doi: 10.1016/s0006-8993(97)01552-7. [DOI] [PubMed] [Google Scholar]
- 169.Thwin MM, Ong WY, Fong CW, Sato K, Kodama K, Farooqui AA, Gopalakrishnakone P. Secretory phospholipase A2 activity in the normal and kainate injected rat brain, and inhibition by a peptide derived from python serum. Exp Brain Res. 2003;150:427–433. doi: 10.1007/s00221-003-1476-7. [DOI] [PubMed] [Google Scholar]
- 170.Farooqui AA. Studies on plasmalogen-selective phospholipase A2 in brain. Mol Neurobiol. 2010;41:267–273. doi: 10.1007/s12035-009-8091-y. [DOI] [PubMed] [Google Scholar]
- 171.Takami S, Minami M, Katayama T, Nagata I, Namura S, Satoh M. TAK-779, a nonpeptide CC chemokine receptor antagonist, protects the brain against focal cerebral ischemia in mice. J Cereb Blood Flow Metab. 2002;22:780–784. doi: 10.1097/00004647-200207000-00003. [DOI] [PubMed] [Google Scholar]
- 172.Youssef S, Wildbaum G, Karin N. Prevention of experimental autoimmune encephalomyelitis by MIP-1alpha and MCP-1 naked DNA vaccines. J Autoimmun. 1999;13:21–29. doi: 10.1006/jaut.1999.0306. [DOI] [PubMed] [Google Scholar]
- 173.Xia MQ, Qin SX, Wu LJ, Mackay CR, Hyman BT. Immunohistochemical study of the beta-chemokine receptors CCR3 and CCR5 and their ligands in normal and Alzheimer’s disease brains. Am J Pathol. 1998;153:31–37. doi: 10.1016/s0002-9440(10)65542-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Balistreri CR, Grimaldi MP, Vasto S, Listi F, Chiappelli M, Licastro F, Lio D, Caruso C, Candore G. Association between the polymorphism of CCR5 and Alzheimer’s disease: results of a study performed on male and female patients from Northern Italy. Ann N Y Acad Sci. 2006;1089:454–461. doi: 10.1196/annals.1386.012. [DOI] [PubMed] [Google Scholar]
- 175.Combarros O, Infante J, Llorca J, Peña N, Fernández-Viadero C, Berciano J. The chemokine receptor CCR5-Delta32 gene mutation is not protective against Alzheimer’s disease. Neurosci Lett. 2004;366:312–314. doi: 10.1016/j.neulet.2004.05.058. [DOI] [PubMed] [Google Scholar]
- 176.Galimberti D, Fenoglio C, Lovati C, Gatti A, Guidi I, Venturelli E, Cutter GR, Mariani C, Forloni G, Pettenati C, et al. CCR2-64I polymorphism and CCR5Delta32 deletion in patients with Alzheimer’s disease. J Neurol Sci. 2004;225:79–83. doi: 10.1016/j.jns.2004.07.005. [DOI] [PubMed] [Google Scholar]
- 177.Liu J, Solway K, Messing RO, Sharp FR. Increased neurogenesis in the dentate gyrus after transient global ischemia in gerbils. J Neurosci. 1998;18:7768–7778. doi: 10.1523/JNEUROSCI.18-19-07768.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Arvidsson A, Collin T, Kirik D, Kokaia Z, Lindvall O. Neuronal replacement from endogenous precursors in the adult brain after stroke. Nat Med. 2002;8:963–970. doi: 10.1038/nm747. [DOI] [PubMed] [Google Scholar]
- 179.Bengzon J, Kokaia Z, Elmér E, Nanobashvili A, Kokaia M, Lindvall O. Apoptosis and proliferation of dentate gyrus neurons after single and intermittent limbic seizures. Proc Natl Acad Sci USA. 1997;94:10432–10437. doi: 10.1073/pnas.94.19.10432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Ekdahl CT, Mohapel P, Elmér E, Lindvall O. Caspase inhibitors increase short-term survival of progenitor-cell progeny in the adult rat dentate gyrus following status epilepticus. Eur J Neurosci. 2001;14:937–945. doi: 10.1046/j.0953-816x.2001.01713.x. [DOI] [PubMed] [Google Scholar]
- 181.Ekdahl CT, Claasen JH, Bonde S, Kokaia Z, Lindvall O. Inflammation is detrimental for neurogenesis in adult brain. Proc Natl Acad Sci USA. 2003;100:13632–13637. doi: 10.1073/pnas.2234031100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Bonde S, Ekdahl CT, Lindvall O. Long-term neuronal replacement in adult rat hippocampus after status epilepticus despite chronic inflammation. Eur J Neurosci. 2006;23:965–974. doi: 10.1111/j.1460-9568.2006.04635.x. [DOI] [PubMed] [Google Scholar]
- 183.Hattiangady B, Rao MS, Shetty AK. Chronic temporal lobe epilepsy is associated with severely declined dentate neurogenesis in the adult hippocampus. Neurobiol Dis. 2004;17:473–490. doi: 10.1016/j.nbd.2004.08.008. [DOI] [PubMed] [Google Scholar]
- 184.Pocock JM, Liddle AC. Microglial signalling cascades in neurodegenerative disease. Prog Brain Res. 2001;132:555–565. doi: 10.1016/S0079-6123(01)32103-9. [DOI] [PubMed] [Google Scholar]
- 185.Rizzi M, Perego C, Aliprandi M, Richichi C, Ravizza T, Colella D, Velískŏvá J, Moshé SL, De Simoni MG, Vezzani A. Glia activation and cytokine increase in rat hippocampus by kainic acid-induced status epilepticus during postnatal development. Neurobiol Dis. 2003;14:494–503. doi: 10.1016/j.nbd.2003.08.001. [DOI] [PubMed] [Google Scholar]
- 186.Rosell DR, Nacher J, Akama KT, McEwen BS. Spatiotemporal distribution of gp130 cytokines and their receptors after status epilepticus: comparison with neuronal degeneration and microglial activation. Neuroscience. 2003;122:329–348. doi: 10.1016/s0306-4522(03)00593-1. [DOI] [PubMed] [Google Scholar]


