Abstract
Background:
Cyst and tumors arise from tissue remains of odontogenesis, these interactions have been considered to play an important role in the tumorigenesis of odontogenic lesions. The connective tissue stroma has an essential role in the preservation of epithelial tissues and minor alterations in the epithelium are followed by corresponding changes in the stroma, such as angiogenesis. Vascular endothelial growth factor (VEGF) is considered the first factor which maintains its position as the most critical driver of vascular formation and is required to initiate the formation of immature vessels, with this aim, present study was executed to evaluate VEGF expression in kertocystic odontogenic tumor, dentigerous cyst and radicular cyst (RC).
Materials and Methods:
A retrospective study was carried out comprising a total of 31 cases; 13 cases of keratocystic odontogenic tumor (KCOT), nine cases of dentigerous cyst (DC) and nine cases of RC. The sections were stained immunohistochemically with VEGF antibody and were evaluated for the presence and intensity of the immuno reactive cells. Statistical analysis was carried out using Chi-square test to inter-compare the VEGF expression between KCOT, DC, and RC.
Results:
VEGF expression in the epithelium and connective tissue was significantly higher in KCOT compared to dentigerous and RC. One case of KCOT with carcinomatous change also revealed positive results for the VEGF expression in the dysplastic epithelium, tumor islands, and connective tissue. The significant difference was observed on inter-comparison of the VEGF expression in the connective tissue of KCOT and DC, whereas no significant difference was observed in the VEGF expression in the connective tissue of KCOT and DC.
Conclusion:
The present study data supports the literature finding that angiogenesis can be important in the progression and enlargement of odontogenic cysts similarly to what occurs in neoplastic conditions and further it can be concluded that the higher positivity for VEGF of KCOT could help to explain in part the aggressive biological behavior of the lesion. The stroma of KCOT could be regarded not only as a structural support of the cyst wall but also as playing a part in the neoplastic behavior of cyst.
Keywords: Dentigerous cyst, keratocystic odontogenic tumor, odontogenic cyst, radicular cyst, vascular endothelial growth factor
INTRODUCTION
Odontogenesis is controlled by interactions between the epithelial and mesenchymal components of developing dental tissues. Regarding the fact that odontogenic.
Angiogenesis is the process of formation of new microvessels from the preexisting vasculature. It is thought to be initiated by an increase in the level of angiogenic stimuli and a concomitant decrease in the level of angiogenic inhibitors. These factors are produced by tumor cells, stromal cells, and inflammatory cells such as mast cells and macrophages.[1]
The angiogenic factors play a role as important mediators of tumor angiogenesis as in epithelial odontogenic tumors and the alterations might be associated with tumoriogenesis or malignant transformation of odontogenic tumors. The proangiogenic cytokine is capable of inducing micro vascular permeability leading to extravasation of plasma proteins, and a predictable sequence of proangiogenic stromal changes. Due to these functions, vascular endothelial growth factor (VEGF) has been implicated as an important factor in granulation tissue development and cyst enlargement. Furthermore, it is now assumed that the critical event in the regulation of angiogenesis is the signaling cascade involving vascular endothelium growth factor (VEGF).[2]
VEGFs comprise a family of multifunctional proteins mainly involved in normal and pathologic angiogenesis, defined as the formation of new vessels by sprouting of the preexisting endothelium.[3] The VEGF family includes VEGF-A or VEGF, also known as vascular permeability factor, VEGF-B, VEGF-C, VEGF-D, VEGF-E, and placental growth factors.[3,4,5]
Therefore, the present study attempted to assess the VEGF expression, in keratocystic odontogenic tumor (KCOT), dentigerous cyst (DC) and radicular cyst (RC) and correlate the role of VEGF and angiogenesis with cyst proliferation.
MATERIALS AND METHODS
The present study was carried out on 31 archival tissues that included13 cases of KCOT, nine cases of DC and nine cases of RC. Formalin fixed-paraffin embedded tissues were sectioned at 3–4 micron thickness and stained with hematoxylin and eosin stain for histopathological conformation.
VEGF expression was determined by immunohistochemistry using a rabbit polyclonal anti-VEGF antibody (BioGenex, USA).
The immunoexpression of VEGF in the epithelium was qualitatively evaluated at ×400 magnification as the criteria purposed by the by Leonardi, et al.,[6] the immunoexpression of the VEGF was classified according to the following scores: Score 0– no staining; Score 1– weak, staining in 11–25%of cells; Score 2– moderate, staining in 26–75% of cells; 3– strong, staining in more than 76% of cells.
VEGF expression in the connective tissue was semi-quantitatively evaluated, calculating the percentage of VEGF-positive vessels, fibroblasts, and inflammatory cells in 10 representative fields by using a binuclear microscope at ×400 magnification. The scoring were divided in the following categories: Score 0 (≤10% positivecells); Score 1 (>10% and <50% positivecells); Score 2 (≥50% positivecells).[7]
The Chi-square test was applied to inter-compare the VEGF expression between KCOT, DC and RC. P < 0.05 was considered to be statistically significant.
RESULTS
On inter-comparison of vascular endothelial growth factor expression in the epithelium between keratocystic odontogenic tumor, dentigerous cyst and radicular cyst
Positive immune reactivity was observed in all cases of KCOT, [Figure 1] DC [Figure 2] where as in 66.66% cases of RC. All the epithelial layers, with the exception of the parakeratin layer in two cases of KCOT, were stained. In KCOT strong expression was observed in six cases (46.15%) moderate expression in six cases (46.15%), weak expression in one case (7.69%). One case of KCOT (out of 13) with carcinomatous change revealed the moderate intensity of VEGF both in the epithelium, tumor islands, and connective tissue stroma.
Figure 1.
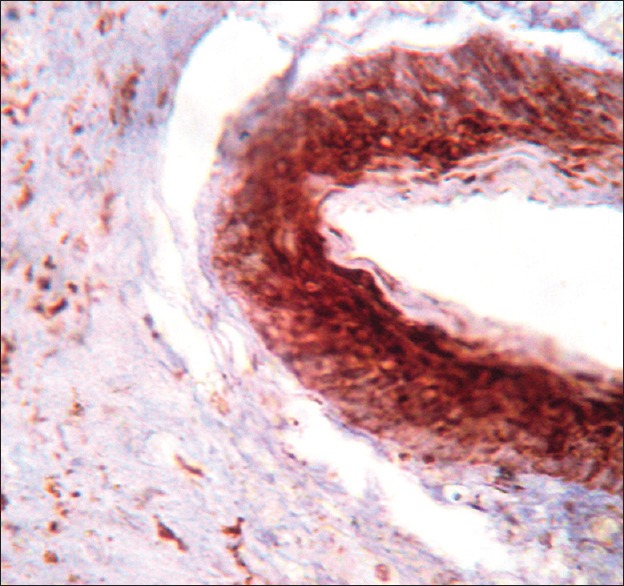
Vascular endothelial growth factor expression in keratocystic odontogenic tumor
Figure 2.
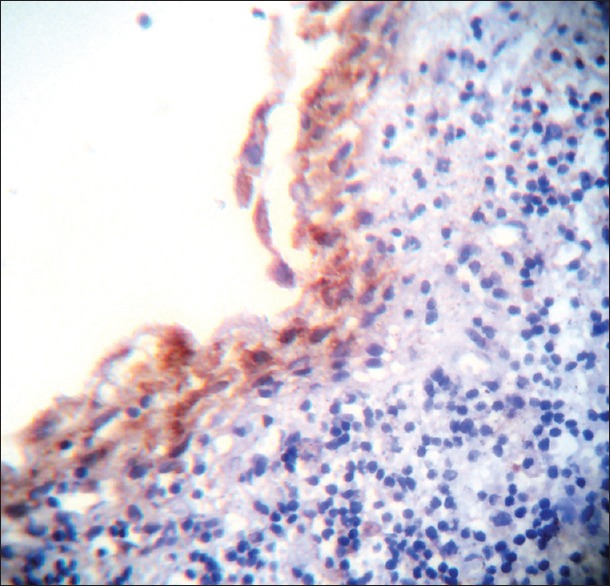
Vascular endothelial growth factor expression in dentigerous cyst
In DC strong expression was observed in four cases (44.44%), moderate expression in two cases (22.22%), and weak expression in three cases (33.33%). In RC strong expression was observed in [Figure 3] one case (11.11%), moderate expression in one case (11.11%), weak expression in four cases (44.44%) and three cases (33.33%) revealed negative staining. A significant difference was observed with the P = 0.018 on inter-comparison of VEGF expression in the epithelium between KCOT, DC, and RC.
Figure 3.
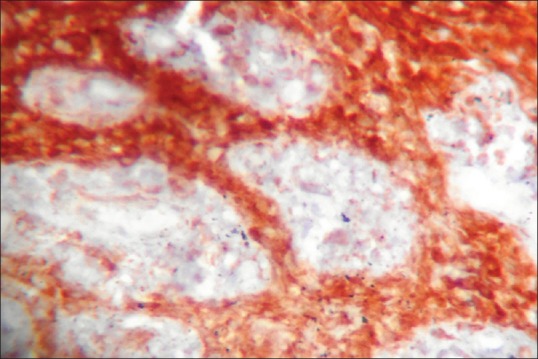
Vascular endothelial growth factor expression in radicular cyst
On inter-comparison of VEGF expression in the connective tissue between KCOT, DC and RC. In KCOT more number of cases of KCOT six cases (46.15%), revealed strong expression as compared to dentigerous three cases (33.33%) and RC four cases (44.44%).
Inter-comparison of VEGF expression in the epithelium between KCOT and DC revealed statistically highly significant results with the P = 0.0032 [Table 1].
Table 1.
Inter-comparison of VEGF expression in the epithelium

Inter-comparison of VEGF expression in the connective tissue between KCOT and DC revealed a significant difference with the P = 0.002 [Table 2].
Table 2.
Inter-comparison of VEGF expression in the connective tissue

Inter-comparison of VEGF expression in the epithelium between KCOT and RC revealed statistically highly significant results with the P = 0.00156 [Table 1].
Inter-comparison of VEGF expression in the connective tissue between KCOT and RC revealed Anon significant difference with the P = 0.1281 [Table 2].
Inter-comparison of VEGF expression in the epithelium between DC and RC revealed statistically significant results with the P = 0.0321 [Table 1].
Inter-comparison of VEGF expression in the connective tissue between DC and RC revealed a nonsignificant difference with the P = 0.145 [Table 2].
DISCUSSION
The present study assessed the expression of VEGF in 13 cases of KCOT and nine cases each of DC and RC. Statistically significant differences in VEGF expression in the epithelium (0.018) and connective tissue (0.0155) were observed between KCOT, DC and RC. All epithelial cell layers, with the exception of the parakeratin layer in 12 out of 13 cases of KCOT revealed positive immunoreactivity for VEGF [Figure 1]. These results might suggest that the critical event in the regulation of angiogenesis is the signaling cascade involving vascular endothelium growth factor (VEGF). This conclusion is based first of all on the biological properties of this growth factor.
On literature review, only one study was found comparing VEGF expression in KCOT, DC and RC by Mitrou et al.[8] who observed immune reactivity for VEGF in 35 cases out of 37 odontogenic keratocysts (OKCs) and all DCs and RCs, as well as adjacent endothelial cells, fibroblasts, and inflammatory cells. Our results were consistent with the findings of Mitrou et al. as we also observed positive expression of VEGF in the epithelium of all the cases of KCOT 31(out of 31) DC 9(out of 9), RC 9(out of 9) with all cases of KCOT, DC, while only 66.66% cases of RC 6 (out of 9) revealing positive VEGF expression in the connective tissue.
Mitrou et al.[8] concluded that VEGF expression in the lining epithelium of KCOT, DC, and RC was not associated with the presence of inflammation. Although it can be assumed that preexisting inflammation may upregulate VEGF expression, particularly in RCs, their findings are suggestive of a role for VEGF regardless of inflammation.
The epithelial lining of odontogenic cysts can undergo simple cystic expansion, keratinization, or dysplastic transformation. However, it is well known that the lining of an odontogenic cyst may transform into a mucoepidermoid carcinoma or a squamous cell carcinoma (SCC)[9] with carcinomatous change occurring in <1% of the odontogenic cysts.[10]
In the present study, one case of KCOT with carcinomatous changes revealed the moderate intensity of VEGF both in the epithelium, tumor islands, and connective tissue stroma. On review of literature no study was found evaluating the VEGF expression in the case of KCOT transforming into SCC, moreover, only 15 reports of SCC developing from KCOT have been reported.
VEGF has been implicated in the pathogenesis of odontogenic tumors and cysts, while acquisition of anangiogenic phenotype appears to be a crucial aspect in the transition from hyperplasia to neoplasia and VEGF has also been found to be produced by tumor cells. High expression by tumor cells is strongly correlated with high expression of VEGF receptors (VEGFR)-2 and VEGFR-3 by the same cells, suggesting the presence of an autocrine loop that induces epithelial proliferation.
Further, in the present study with the P = 0.0032 and 0.002 significant differences were observed in the epithelium and connective tissue when VEGF expression was inter-compared between KCOT and DC. Moderate to strong expression was observed in the majority of cases of KCOT (92.3%), on the other hand, 66.7% cases of DC revealed moderate to strong expression. These findings are suggestive of the fact that KCOT has increased epithelial proliferation rate, and increased level of angiogenesis may be related to the increase in tissue metabolism and nutrition requirement of the proliferating epithelium. These findings also help to explain the biological behavior of KCOT that should be considered to be a benign odontogenic tumor rather than merely cyst.
On literature review only two studies by Mitrou et al.[8] and Rubini et al.[7] were found comparing VEGF expression in KCOT and DC. Our results were consistent with the findings of these studies. Mitrou et al.[8] observed very strong expression in 72.97% of KCOT, while weak expression in the majority of DCs (38.89%). Rubini et al.[7] observed a significant different expression of VEGF in all cell components in OKCs compared to follicular cysts (FCs). Indeed, the majority of parakeratotic OKCs (80%) and OOKCs (68%) showed more than 50% VEGF-positive epithelial cells, whereas the majority of FCs (71%) were either negative in the epithelium or showed <10% positive cells (χ2, P < 0.0005). This higher positivity for VEGF of KCOT could help to explain in part the infiltrative characteristic of this lesion. They also observed a correlation between VEGF expression in epithelial cells and capsular fibroblasts and vessels, suggesting that these cell types might form a cellular network sharing regulation by the stimulatory signals promoting angiogenesis.
el-Labban and Aghabeigi[11] morphometrically and ultrastructurally evaluated blood vessels in KCOTs and DCs. Their ultrastructural examination revealed the presence of thrombosis in KCOTs, but rarely in DCs. The thrombosed vessels in KCOT probably contain a large number of platelets, which can produce angiogenic and proangiogenic factors such as platelet-derived growth factor, VEGF, basic fibroblast growth factor and transforming growth factor-beta1 (Brill et al. and Anitua et al).[12,13] These proteins, especially VEGF, can induce endothelial cell proliferation, migration and vascular permeability at different levels (Maharaj and D’Amore, 2007)[14] leading to increased angiogenesis and microvessel formation.
In this study, we observed a highly significant difference on inter-comparison of the VEGF expression in the epithelium between KCOT and RC (0.00156) where as a nonsignificant difference was observed on inter- comparison of the VEGF expression in the connective tissue between KCOT and RC (0.1281). Although it can be assumed that preexisting inflammation may upregulate VEGF expression, particularly in RCs, but the present study findings are suggestive of a role for VEGF regardless of inflammation. These results were in accordance with the study by Mitrou et al.[8] who also observed no statistically significant difference between KCOT and RC (0.228).
A significant difference in the epithelium (0.0321) and the nonsignificant difference in the connective tissue (0.145) were observed on inter-comparison of the VEGF expression between DC and RC. These results were in accordance with that of Mitrou et al.[8] who also observed no statistical significant difference between DC and RC (P = 0.26).de Moraes et al.[15] compared the VEGF and angiogenic index in RC and DC. They observed higher expression of VEGF in the epithelium and capsule of Rc as compared to DC and suggested that the receptor activator of nuclear factor kappa-B (NF-κB) and receptor activator of NF-κB ligand play an important role in bone resorption in DC and the hemorrhagic areas in the capsule of DC could be explained by increased vessel's number and the higher VEGF expression in RC might be related to nature of these lesions, where the inflammatory process contributes significantly to these findings.
The epithelium status of the RCs has been suggested as a reliable histological parameter of biological activity and/or inactivity of cystic growth. The epithelial expression of VEGF in DCs might constitute an additional mechanism for the enlargement of these lesions, maintaining the stimulus for angiogenesis and vascular permeability.
In conclusion, the results of the present study suggest that higher positivity of VEGF expression by the epithelial cells in KCOT may promote growth via an autocrine proliferative effect on the cystic epithelium, while paracrine stimulation of the vascular net work may maintain survival and growth, and promote enlargement and expansion through protein accumulation in the cystic cavity and bone resorption.
CONCLUSION
Findings of the present study further stress on the hypo thesis that the stroma of KCOT could be regarded not only as a structural support of the cyst wall but also as playing a part in the neoplastic behavior of cyst.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- 1.Polverini PJ. The pathophysiology of angiogenesis. Crit Rev Oral Biol Med. 1995;6:230–47. doi: 10.1177/10454411950060030501. [DOI] [PubMed] [Google Scholar]
- 2.Karamysheva AF. Mechanisms of angiogenesis. Biochemistry (Mosc) 2008;73:751–62. doi: 10.1134/s0006297908070031. [DOI] [PubMed] [Google Scholar]
- 3.Carmeliet P, Collen D. Molecular analysis of blood vessel formation and disease. Am J Physiol. 1997;273(5 Pt 2):H2091–104. doi: 10.1152/ajpheart.1997.273.5.H2091. [DOI] [PubMed] [Google Scholar]
- 4.Ferrara N, Davis-Smyth T. The biology of vascular endothelial growth factor. Endocr Rev. 1997;18:4–25. doi: 10.1210/edrv.18.1.0287. [DOI] [PubMed] [Google Scholar]
- 5.Olofsson B, Jeltsch M, Eriksson U, Alitalo K. Current biology of VEGF-B and VEGF-C. Curr Opin Biotechnol. 1999;10:528–35. doi: 10.1016/s0958-1669(99)00024-5. [DOI] [PubMed] [Google Scholar]
- 6.Leonardi R, Caltabiano M, Pagano M, Pezzuto V, Loreto C, Palestro G. Detection of vascular endothelial growth factor/vascular permeability factor in periapical lesions. J Endod. 2003;29:180–3. doi: 10.1097/00004770-200303000-00004. [DOI] [PubMed] [Google Scholar]
- 7.Rubini C, Artese L, Zizzi A, Fioroni M, Ascani G, Goteri G, et al. Immunohistochemical expression of vascular endothelial growth factor (VEGF) in different types of odontogenic cysts. Clin Oral Investig. 2011;15:757–61. doi: 10.1007/s00784-010-0433-7. [DOI] [PubMed] [Google Scholar]
- 8.Mitrou GK, Tosios KI, Kyroudi A, Sklavounou A. Odontogenic keratocyst expresses vascular endothelial growth factor: An immunohistochemical study. J Oral Pathol Med. 2009;38:470–5. doi: 10.1111/j.1600-0714.2009.00755.x. [DOI] [PubMed] [Google Scholar]
- 9.Moosvi Z, Tayaar SA, Kumar GS. Neoplastic potential of odontogenic cysts. Contemp Clin Dent. 2011;2:106–9. doi: 10.4103/0976-237X.83073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Stoelinga PJ, Bronkhorst FB. The incidence, multiple presentation and recurrence of aggressive cysts of the jaws. J Craniomaxillofac Surg. 1988;16:184–95. doi: 10.1016/s1010-5182(88)80044-1. [DOI] [PubMed] [Google Scholar]
- 11.el-Labban NG, Aghabeigi B. A comparative stereologic and ultrastructural study of blood vessels in odontogenic keratocysts and dentigerous cysts. J Oral Pathol Med. 1990;19:442–6. doi: 10.1111/j.1600-0714.1990.tb00784.x. [DOI] [PubMed] [Google Scholar]
- 12.Brill A, Elinav H, Varon D. Differential role of platelet granular mediators in angiogenesis. Cardiovasc Res. 2004;63:226–35. doi: 10.1016/j.cardiores.2004.04.012. [DOI] [PubMed] [Google Scholar]
- 13.Anitua E, Sanchez M, Nurden AT, Zalduendo M, de la Fuente M, Azofra J, et al. Reciprocal actions of platelet-secreted TGF-beta1on the production of VEGF and HGF by human tendon cells. Plast Reconstr Surg. 2007;119:950–9. doi: 10.1097/01.prs.0000255543.43695.1d. [DOI] [PubMed] [Google Scholar]
- 14.Maharaj AS, D’Amore PA. Roles for VEGF in the adult. Microvasc Res. 2007;74:100–13. doi: 10.1016/j.mvr.2007.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.de Moraes M, de Matos FR, de Souza LB, de Almeida Freitas R, de Lisboa Lopes Costa A. Immuno expression of RANK, RANKL, OPG, VEGF, and vWF in radicular and dentigerous cysts. J Oral Pathol Med. 2013;42:468–73. doi: 10.1111/jop.12036. [DOI] [PubMed] [Google Scholar]


