Abstract
Background
RET rearrangement, a hallmark of radiation-induced thyroid cancer, has been reported to occur in 1% of lung adenocarcinoma patients. Patients with this rearrangement tend to be younger and never smokers, raising a possibility of other causes, such as radiation. We hypothesized that RET chromosomal rearrangement may represent a genetic mechanism of radiation-induced lung cancer.
Methods
Two hundred forty-five consecutive primary lung adenocarcinomas without history of radiation and 38 lung adenocarcinoma patients with a history of therapeutic radiation for breast carcinoma or mediastinal Hodkgin lymphoma were tested for RET rearrangement by fluorescence in situ hybridization. Human lung adenocarcinoma cells (201T) were subjected to γ radiation and tested for RET gene fusions by reverse transcriptase-polymerase chain reaction and Southern blot hybridization.
Results
We identified one case with RET rearrangement in the group without history of radiation (1 of 240; 0.4%) and two cases in the group with history of radiation (2 of 37; 5.4%; P=0.0436). Both these patients were women, who were former smokers with a history of breast carcinoma treated with surgery and radiation. Furthermore, we found that RET fusions could be directly induced in 201T human lung cells by exposure to 1 Gy of γ radiation. All fusions identified were between RET and KIF5B genes, and no RET fusions to CCDC6 or NCOA4 genes, characteristic for thyroid cancer, were identified in the irradiated lung cells.
Conclusion
RET fusions may represent a genetic mechanism of radiation-induced lung adenocarcinoma.
Keywords: Lung adenocarcinoma, RET proto oncogene, Radiation
Approximately 1% of lung adenocarcinomas were reported to harbor a novel gene fusion involving the RET tyrosine-kinase gene partnered with either kinesin family member 5B (KIF5B) or coiled-coil domain containing 6 (CCDC6).1–5 RET is a receptor tyrosine-kinase involved in cell proliferation, migration, cell differentiation, and neuronal navigation. Germline and somatic mutations in RET are associated with multiple endocrine neoplasia type 2 and sporadic medullary carcinoma of thyroid. It is interesting that RET rearrangements, mostly CCDC6-RET (papillary thyroid carcinoma [PTC1]) and nuclear receptor coactivator 4 (NCOA4) nuclear receptor coactivator 4 (NCOA4)-RET (PTC3), develop with a large prevalence in radiation-induced thyroid cancers.6 It is intriguing that patients with lung adenocarcinomas with RET fusion gene have a tendency to be younger and never-smokers, raising a possibility of other causes, such as radiation. This is particularly likely because the RET gene rearrangements represent a hallmark of radiation-induced cancer in the thyroid gland.7 Furthermore, exposure to radon is believed to be the second most common cause of lung cancer in the United States, and both medical therapeutic and accidental environmental irradiation are associated with the increased risk of lung cancer. The genetic mechanisms of radiation-induced lung cancer are largely unknown. This led us to the hypothesis that RET chromosomal rearrangements represent a genetic mechanism of radiation-induced lung cancer.
PATIENTS AND METHODS
Patient Selection and Biomarker Testing
Two groups of patients were selected for the study. The first group included 245 consecutive newly diagnosed primary lung adenocarcinomas (stages I–IV). This group of patients had no documented history of medical, accidental, or professional irradiation. The second group included 38 lung cancer patients (stages I–IV) with the history of therapeutic radiation for breast carcinomas or mediastinal Hodkgin’s lymphomas at least 3 years before the diagnosis of lung cancer. The study was approved by the University of Pittsburgh Institutional Review Board (IRB #PRO12070216).
RET Fluorescence In Situ Hybridization
Fluorescence in situ hybridization (FISH) analysis was performed by using standard method with the fusion probe for RET (Abnova, Taipei City, Taiwan) and paraffin pretreatment reagent kit (Vysis, Inc., Downers Grove, IL). At least 60 cells were scored for each case and control. Tumors with 12.8% or more of cells displaying RET rearrangement were classified as FISH positive. Each FISH assay included normal lung tissue sections as a negative control, and sections of lung non–small-cell carcinoma previously identified with KIF5B-RET translocation as a positive control.
In Vitro Exposure to External γ Radiation and RET Rearrangements in Human Lung Cells
Human adenocarcinoma cells 201T (wild type for EGFR, KRAS, and met proto oncogene [MET]) were subjected to γ radiation, a type of low-linear energy transfer (LET) radiation which is similar to radiographs used for radiotherapy.8 Cells (1 × 106) growing in T75 flasks were exposed to 1 Gy of γ radiation from Cs-137 delivered as a single dose by using the Gamma Cell 40 (Nordion, Kanta, Ontario, Canada) irradiator split, into 30 flasks, grown for 8 days (~4 population doublings), and harvested.9 Cell pellets were used to isolate RNA, purify mRNA, and test for RET gene fusions by reverse transcriptase polymerase chain reaction followed by Southern blot hybridization. Detection of RET/PTC1 and RET/PTC3 rearrangements was achieved using polymerase chain reaction primers reported before.10 This highly sensitive assay detects the formation of gene fusions with the rate as low as one in 106 cells. All experiments were performed in duplicate. Induction of DNA double-strand breaks (DSBs) was monitored by formation of γhistone H2AX foci. Cell survival was assessed using by counting floating cells and tryptan blue staining.
Statistical Analysis
Fisher’s exact probability test was used to compare the rates of positives between the groups. p Values less than 0.05 were considered statistically significant.
RESULTS
Detection of RET Rearrangement in Patient Samples
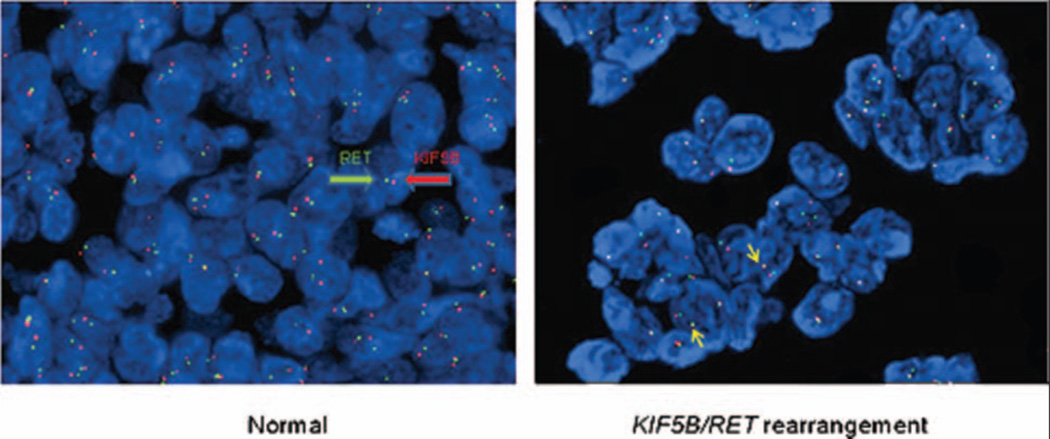
FISH was successful in 240 cases of unselected, consecutively diagnosed lung adenocarcinomas. Technically suboptimal hybridization occurred in five samples. One of 240 cases (0.4%) was classified as positive for RET rearrangement, with 15% of tumor cells showing the fused 3'5' RET and the split/single 3' RET and the split/single 5' RET signal (Fig. 1).
Figure 1.
KIF5B/RET rearrangement in the adenocarcinoma cells from a patient with a history of radiation therapy for breast cancer. Normal cells without rearrangement show two red and two green signals separated, whereas cells with rearrangement show one red and one green signal separated and one pair of red and green signals in juxtaposition.
Of 38 cases with a history of therapeutic radiation (dosage range, 40–60 Gy) there were 30 women with a history of breast cancer and eight men with a history of mediastinal Hodgkin’s lymphoma. All patients developed a primary lung adenocarcinoma. FISH assay was successful in 37 cases. Two of 37 cases (5.4%) were positive for RET rearrangement, with 20.3% and 19.2% of the tumor cells showing FISH rearrangement pattern. Both patients were women, who were former smokers with a history of breast carcinoma. The first patient received the 45 Gy dose followed by the 14 Gy boost to the tumor 5 years before the development of lung cancer. The second patient received the 50 Gy dose followed by the 10 Gy boost to the tumor bed 7 years before the development of lung cancer. The difference in frequency of RET rearrangement between the two study groups was statistically significant (p=0.0436).
RET Rearrangements in Human Lung Cells Exposed to γ Radiation
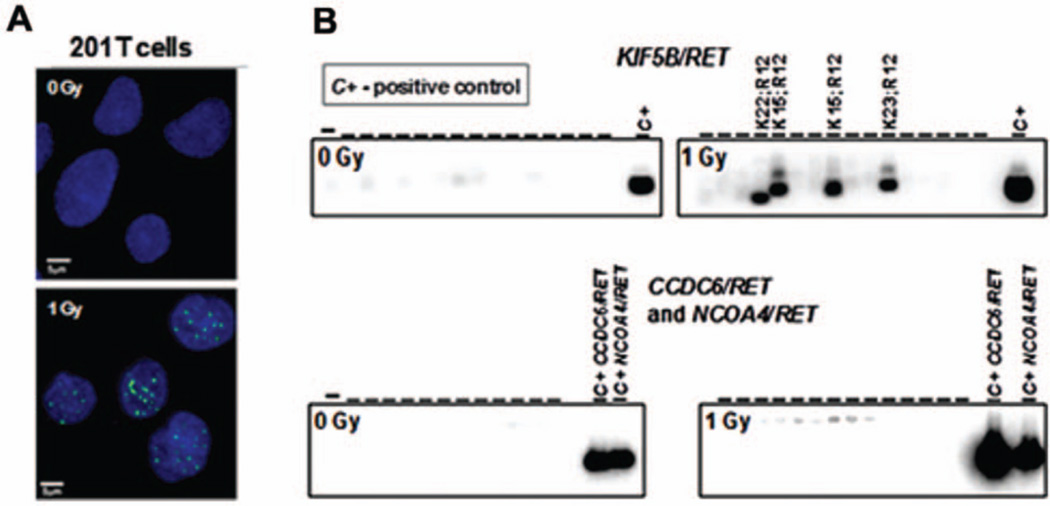
We found that RET fusions could be directly induced in 201T human lung cells by 1 Gy of γ radiation. The induction of DSBs was confirmed by γH2AX nuclear foci formation (Fig. 2A). In both experimental replicates, we observed no formation of RET fusions in cells not exposed to radiation, whereas exposure to 1 Gy of radiation resulted in the formation of two KIF5B-RET fusions in each experiment involving 1 × 106 cells (Fig. 2B). Overall, five KIF5B/RET fusions were detected, with the following cDNA isoforms: exon 15 of KIF5B fused to exon 12 of RET (×3), exon 22 (×1), and exon 23 (×1) (Table 1). No fusions was found between RET and either CCDC6 (RET/PTC1) or NCOA4 (RET/PTC3) genes, typical of radiation-associated thyroid cancer.10
Figure 2.
A, Generation of DSBs was monitored by formation of γH2AX nuclear foci. B, Induction of KIF5B/RET, but not CCDC6/RET or NCOA4/RET rearrangements in 201T lung cells by γ radiation.
TABLE 1.
Carcinogenic Rearrangements Detected in 201T Lung Cells
| Sample ID | Number of Cells Positive for gH2AX Foci |
Number of Clones |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Detected by
Hybridization |
Mutation Rate Per 106
Cells |
||||||||||
|
KIF5B/RET |
RET/PTC |
KIF5B/RET |
RET/PTC |
||||||||
| K15/R12 | K23/R12 | K22/R12 | PTC1 | PTC3 | K15/R12 | K23/R12 | K22/R12 | PTC1 | PTC3 | ||
| 0 Gy | 5% | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 0 Gy | 5% | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 1 Gy | 100% | 2 | 0 | 1 | 0 | 0 | 2 × 10−6 | 0 | 1 × 10−6 | 0 | 0 |
| 1 Gy | 100% | 1 | 1 | 0 | 0 | 0 | 1 × 10−6 | 1 × 10−6 | 0 | 0 | 0 |
DISCUSSION
The frequency of RET rearrangements in our unselected group of patients was lower than reported in literature. This could be explained by the fact that most reported studies are from Asia, and frequency of RET rearrangements in other racial and ethnical groups is still under investigation. Furthermore, our patient population tends to be older and having a history of smoking.11 Our preliminary data described above provide evidence that RET rearrangements occur with a significant frequency in lung adenocarcinomas from patients exposed to therapeutic radiation. Furthermore, we were able to induce RET fusions in human lung cells by γ radiation in vitro. This allowed us to propose the hypothesis that RET chromosomal rearrangement represents a genetic mechanism of radiation-induced lung cancer. Matsubara et al.12 recently reported the presence of CCDC6-RET fusion in a lung carcinoma cell line. In contrast, our in vitro experiments showed only KIF5B-RET fusions, and no fusions between RET and either CCDC6 or NCOA4 genes, which are typical of radiation-associated thyroid cancer. Our observations suggest that patients with radiation-induced lung cancer likely represent a subset of lung carcinoma patients who might benefit from clinically available tyrosine-kinase inhibitors that target RET kinase activity.
Footnotes
Disclosure: The authors declare no conflict of interest.
REFERENCES
- 1.Kohno T, Ichikawa H, Totoki Y, et al. KIF5B-RET fusions in lung adenocarcinoma. Nat Med. 2012;18:375–377. doi: 10.1038/nm.2644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Takeuchi K, Soda M, Togashi Y, et al. RET, ROS1 and ALK fusions in lung cancer. Nat Med. 2012;18:378–381. doi: 10.1038/nm.2658. [DOI] [PubMed] [Google Scholar]
- 3.Wang R, Hu H, Pan Y, et al. RET fusions define a unique molecular and clinicopathologic subtype of non-small-cell lung cancer. J Clin Oncol. 2012;30:4352–4359. doi: 10.1200/JCO.2012.44.1477. [DOI] [PubMed] [Google Scholar]
- 4.Suehara Y, Arcila M, Wang L, et al. Identification of KIF5B-RET and GOPCROS1 fusions in lung adenocarcinomas through a comprehensive mRNA-based screen for tyrosine kinase fusions. Clin Cancer Res. 2012;18:6599–6608. doi: 10.1158/1078-0432.CCR-12-0838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ju YS, Lee WC, Shin JY, et al. A transforming KIF5B and RET gene fusion in lung adenocarcinoma revealed from whole-genome and transcriptome sequencing. Genome Res. 2012;22:436–445. doi: 10.1101/gr.133645.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hamatani K, Eguchi H, Ito R, et al. RET/PTC rearrangements preferentially occurred in papillary thyroid cancer among atomic bomb survivors exposed to high radiation dose. Cancer Res. 2008;68:7176–7182. doi: 10.1158/0008-5472.CAN-08-0293. [DOI] [PubMed] [Google Scholar]
- 7.Bounacer A, Wicker R, Caillou B, et al. High prevalence of activating ret proto-oncogene rearrangements, in thyroid tumors from patients who had received external radiation. Oncogene. 1997;15:1263–1273. doi: 10.1038/sj.onc.1200206. [DOI] [PubMed] [Google Scholar]
- 8.Siegfried JM, Krishnamachary N, Gaither Davis A, Gubish C, Hunt JD, Shriver SP. Evidence for autocrine actions of neuromedin B and gastrin-releasing peptide in non-small cell lung cancer. Pulm Pharmacol Ther. 1999;12:291–302. doi: 10.1006/pupt.1999.0210. [DOI] [PubMed] [Google Scholar]
- 9.Evdokimova V, Gandhi M, Rayapureddi J, Stringer JR, Nikiforov YE. Formation of carcinogenic chromosomal rearrangements in human thyroid cells after induction of double-strand DNA breaks by restriction endonucleases. Endocr Relat Cancer. 2012;19:271–281. doi: 10.1530/ERC-11-0314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Caudill CM, Zhu Z, Ciampi R, Stringer JR, Nikiforov YE. Dose-dependent generation of RET/PTC in human thyroid cells after in vitro exposure to gamma-radiation: a model of carcinogenic chromosomal rearrangement induced by ionizing radiation. J Clin Endocrinol Metab. 2005;90:2364–2369. doi: 10.1210/jc.2004-1811. [DOI] [PubMed] [Google Scholar]
- 11.Dacic S, Shuai Y, Yousem S, Ohori P, Nikiforova M. Clinicopathological predictors of EGFR/KRAS mutational status in primary lung adenocarcinomas. Mod Pathol. 2010;23:159–168. doi: 10.1038/modpathol.2009.154. [DOI] [PubMed] [Google Scholar]
- 12.Matsubara D, Kanai Y, Ishikawa S, et al. Identification of CCDC6-RET fusion in the human lung adenocarcinoma cell line, LC-2/ad. J Thorac Oncol. 2012;7:1872–1876. doi: 10.1097/JTO.0b013e3182721ed1. [DOI] [PubMed] [Google Scholar]