ABSTRACT
The understanding of bone repair phenomena is a fundamental part of dentistry and maxillofacial surgery.
Objective
The present study aimed to evaluate the influence of buried magnetic field stimulation on bone repair in rat calvaria after reconstruction with autogenous bone grafts, synthetic powdered hydroxyapatite, or allogeneic cartilage grafts, with or without exposure to magnetic stimulation.
Material and Methods
Ninety male Wistar rats were divided into 18 groups of five animals each. Critical bone defects were created in the rats’ calvaria and immediately reconstructed with autogenous bone, powdered synthetic hydroxyapatite or allogeneic cartilage. Magnetic implants were also placed in half the animals. Rats were euthanized for analysis at 15, 30, and 60 postoperative days. Histomorphometric analyses of the quantity of bone repair were performed at all times.
Results
These analyses showed significant group by postoperative time interactions (p=0.008). Among the rats subjected to autogenous bone reconstruction, those exposed to magnetic stimulation had higher bone fill percentages than those without magnetic implants. Results also showed that the quality of bone repair remained higher in the former group as compared to the latter at 60 postoperative days.
Conclusions
After 60 postoperative days, bone repair was greater in the group treated with autogenous bone grafts and exposed to a magnetic field, and bone repair was most pronounced in animals treated with autogenous bone grafts, followed by those treated with powdered synthetic hydroxyapatite and allogeneic cartilage grafts.
Keywords: Maxillofacial surgery, Magnetic field therapy, Bone substitutes
INTRODUCTION
The successful reconstruction of oral and maxillofacial bone defects has been recently enabled by advances in the understanding of bone physiology along with improvements in surgical techniques. Such improvements have allowed the development of techniques that promote biological repair, reestablishing the function of damaged tissues. Many biological, chemical, and physical stimuli have been found to have a positive influence on bone growth, repair, and remodeling. An example of such a stimulus is magnetic field stimulation, which has been found to have positive effects on tissue, cellular, and molecular processes 1 , 8 , 20 , 21 .
Several studies have been performed in order to assess the effects of static magnetic fields on different types of tissues 27 . Magnetic fields applied through the skin may activate iron atoms in hemoglobin, influencing oxygen transport, and stimulate osteogenesis by the activation of osteoblasts and by leading to an increase in blood flow to bone 6 , 20 , 21 . Additionally, results suggest that magnetic fields may increase the concentration of growth factors, accelerating the bone repair process 1 . Magnet therapy can be a treatment option, since static magnetic field have a positive influence on bone metabolism 4 .
The results of studies on the effects of magnetic stimulation on tissue repair are still controversial. There has been some favorable evidence of the effects of static magnetic fields generated by permanent magnets or by magnetized materials. Many studies have shown, for instance, that static magnetic fields can accelerate the repair of bones or tissues 6 , 20 , 21 .
The present study aimed to evaluate the influence of buried magnetic field stimulation on bone repair in rat calvaria after reconstruction with autogenous bone grafts, powdered synthetic hydroxyapatite or allogeneic cartilage, using bone histomorphometric analysis.
MATERIAL AND METHODS
The use of animals in the present study conformed to the State Code for Animal Protection and was in accordance with Normative Resolution 04/97 of the Research Ethics Committee of our institution (GPPG/HCPA), which reviewed and approved the present project (Project no. 10-0307). All experimental procedures were performed in the Animal Experimentation Unit of the Clinical Hospital of Porto Alegre (UEA-HCPA).
Sample size was estimated using G*Power software (version 3). A sample of 90 animals would have 80% power to detect a difference in bone fill percentage between two independent groups with a type I error rate of 5%.
Ninety male Wistar rats aged between seven and eight weeks with an average weight of 300 g were used in the present study. Block randomization was used to assign animals to one of 18 groups composed of five rats each, which were evaluated after 15, 30, and 60 postoperative days. The influence of buried magnetic fields in bone repair was evaluated by the implantation of two permanent magnets (Pan®, São Paulo, SP, Brazil) adjacent to a bone defect created in the calvarium. Each animal was individually subjected to a reconstruction procedure using an autogenous bone graft, a powdered synthetic hydroxyapatite implant (HAP 91®, JHS Biomateriais, Sabará, MG, Brazil) or an allogeneic cartilage graft.
In the autogenous bone group, the bone graft collected during the creation of the cranial defect was repositioned for reconstruction. The powdered synthetic hydroxyapatite was absorbable and porous, and was prepared using a sieve with a mesh size of 2 mm. It was directly implanted on the receiver, according to manufacturer’s instructions. Allogeneic cartilage grafts were obtained as described by Vieira, et al. 25 (1993). A piece of cartilage without perichondrium was first harvested, then washed with sterile saline solution for 15 minutes and preserved in a 70% ethanol solution under refrigeration (2° to 8°C) for 20 days. Particulate cartilage grafts were implanted in the bone defect according to the protocol described by Bercini & Puricelli 3 (1992).
The magnetic field was generated by two neodymium iron boron magnets implanted adjacent to the bone defect. All magnets used in the study were measured by a gauss meter (Magnet-Physik FH 35, Magnet-Physik Steingroever, Köln, Nordrhein-Westfalen, Germany), and the average intensity of the magnetic field in the central region of the bone defect was 84.3G. Commercially pure titanium discs (Promm® Surgical Materials Industry, Porto Alegre, RS, Brazil) were used in the calvaria of control group rats in order to simulate the presence of magnets.
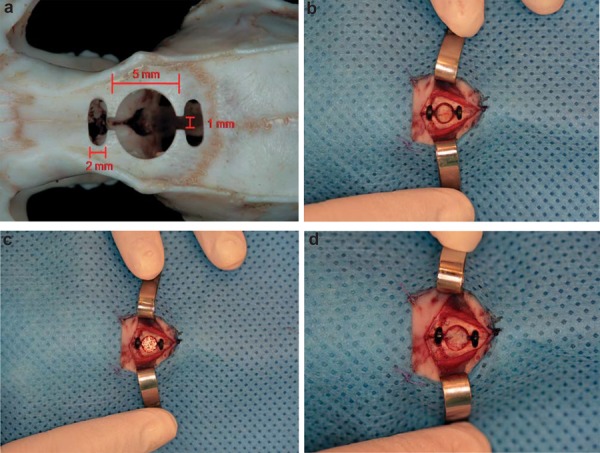
Strict asepsis was observed during the procedures. The rats were anesthetized by intraperitoneal ketamine hydrochloride (100 mg/kg) and xylazine hydrochloride (10 mg/kg), as well as local bupivacaine (2 mg/kg). A trephine drill (Neodent®, Curitiba, PR, Brazil) was used to create a bicortical defect in the frontal bone, measuring 5 mm in diameter and 1 mm in depth. Two 2 mm osteotomies with 1 mm gaps were created anterior and posterior to the bone defect for the placement of the two magnets. This method was used to facilitate the penetration of the magnetic field in the bone defect. The defect was filled with each of the different materials (Figure 1). During the postoperative period, the rats received food and water, and 5 mg/kg Tramadol for pain relief. Animals were euthanized by decapitation after the previously described postoperative periods.
Figure 1. Reconstruction of critical bone defects in rat calvaria using different materials. a) Dimensions of critical bone defects; b) Reconstruction with autogenous bone graft; c) Reconstruction with powdered synthetic hydroxyapatite implant; d) Reconstruction with allogeneic cartilage graft.
Histological preparation
The material was fixed in 10% neutral buffered formalin for 24 hours, after which the pieces were decalcified in a 50% formic acid and 20% sodium citrate solution (1:1). All metallic devices were carefully removed during decalcification. Once the process was complete, a median longitudinal section of the calvarium was taken. The pieces were embedded in paraffin, and 4 µ-histological sections were taken from the central area of the bone defect. Slides were then stained with hematoxylin and eosin (HE) and coded to allow for a blind evaluation.
To ensure correct calibration, ten slides were evaluated twice with an interval of seven days. The values obtained for the two calibration measurements were analyzed using R software (version 2.9.0, R Development Core Team 2010, Auckland, Auckland, Nova Zelândia). The intraclass correlation coefficient (ICC) for the two calibration measurements was 0.98, with a confidence interval (CI) of 0.61 to 0.99.
The histological field examined included the entire length of the bone defect, and 100X-magnified images of the histological slides were captured by an Olympus® video camera (Model 5, Qcolor Cooler, RTV) coupled to a binocular microscope (Olympus Optical Co.®, CX41RF) and a Dell computer (Dimension 5150) running Qcapture® software (version 2.81; Quantitative Imaging Corporation, Inc.; 2005). Photomicrographs were grouped side by side in a mosaic-like arrangement to allow for the visualization and measurement of the total area of the bone defect. Subsequently, the external images of the bone defect were deleted, and only the images of the central portion of the defect were used for analysis (Figure 2).
Figure 2. Mosaic-like arrangement of the entire length of the bone defect and central portion of the defect. Superior image: BDL indicates the bone defect limit, DC indicates position of the metal device used (magnet or titanium disk), BD indicates the bone defect area; Inferior image: it shows the image obtained for histomorphometric analysis after exclusion of areas outside the critical bone defect created; the black line delimits the autogenous bone graft and the green line delimits the new bone.
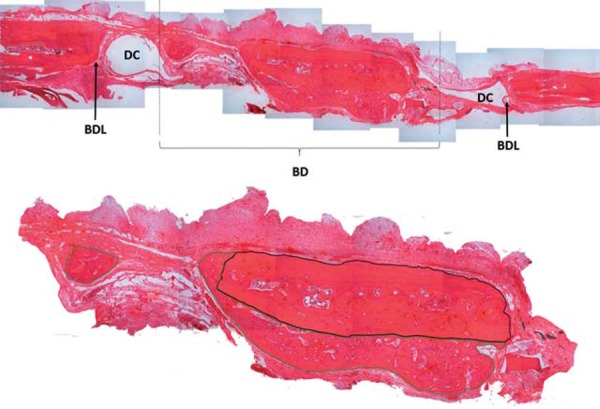
Histomorphometric analysis consisted of the demarcation of the total area of the bone defect and of regenerated bone areas within the defect. These measurements were obtained in Pixel 2 units using Axiovision® software, version 4.6.3, (Carl Zeiss Imaging) and used to calculate the percentage area of the defect containing regenerated bone (Figure 2).
For statistical analysis of the bone fill percentage, we used the mixed models test with the covariance structure chosen by the smallest criterion using Akaike’s information, which, in this case, was the diagonal. The post hoc test used was Bonferroni’s test.
RESULTS
The sample (N=90) did not show any postoperative infectious complications. However, one rat was excluded from the sample because of magnet displacement and another because the quality of the material obtained was unsuitable for histological assessment.
Histomorphometric analysis
The bone fill percentage was calculated based on the total area of the bone defect and the size of the areas containing newly-formed bone in its interior. This variable showed a normal distribution. The model showed a time interaction of p=0.008. Mean values, standard errors, and confidence intervals for the bone fill percentages identified in each group are shown in Table 1.
Table 1. Bone fill percentages in all experimental groups after 15, 30, and 60 postoperative days.
| Group | Time (days) | Average | Standard error | Confidence interval of 95% | |
|---|---|---|---|---|---|
| Lower limit | Upper limit | ||||
| CT | 15 | 1.631 | 1.643 | -1.776 | 5.039 |
| 30 | 8.335 | 3.413 | 1.29 | 15.38 | |
| 60 | 14.055 | 3.964 | 5.875 | 22.236 | |
| CT + M | 15 | 3.989 | 1.47 | 0.941 | 7.037 |
| 30 | 17.018 | 3.413 | 9.973 | 24.063 | |
| 60 | 19.12 | 3.964 | 10.940 | 27.300 | |
| HA | 15 | 6.258 | 1.47 | 3.21 | 9.306 |
| 30 | 19.372 | 3.413 | 12.327 | 26.417 | |
| 60 | 25.5 | 3.964 | 17.320 | 33.680 | |
| HA + M | 15 | 3.811 | 1.47 | 0.764 | 6.859 |
| 30 | 13.617 | 3.413 | 6.572 | 20.661 | |
| 60 | 31.199 | 3.964 | 23.019 | 39.380 | |
| AB | 15 | 3.069 | 1.643 | -0.339 | 6.476 |
| 30 | 16.384 | 3.413 | 9.339 | 23.429 | |
| 60 | 15.544 | 3.964 | 7.364 | 23.724 | |
| AB + M | 15 | 8.644 | 1.47 | 5.596 | 11.692 |
| 30 | 29.041 | 3.413 | 21.996 | 36.086 | |
| 60 | 34.749 | 3.964 | 26.568 | 42.929 | |
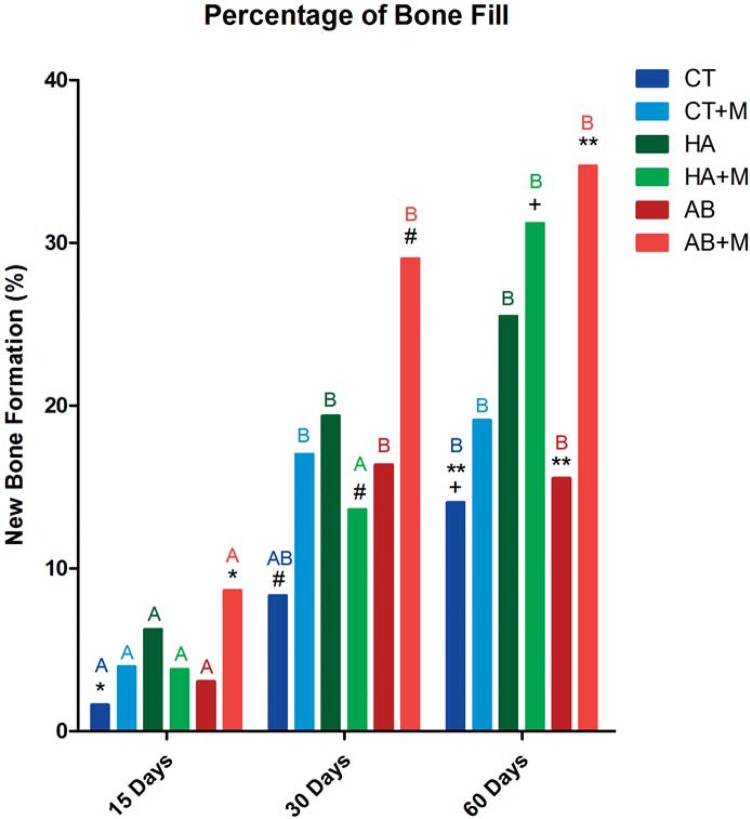
Between-group comparisons of bone fill percentage at 15 postoperative days showed that the autogenous bone graft with magnetic field group (AB+M) had a higher bone fill percentage than the allogeneic cartilage graft without magnetic field group (CT) – indicated in the graph for *. At 30 postoperative days, the bone fill percentage of AB+M rats was still higher than observed in the CT group, and was also significantly greater than that seen in the hydroxyapatite implant with magnetic field group (HA+M) – indicated in the graph for #. After 60 days, the HA+M group showed a higher bone fill percentage than the CT group – indicated in the graph for +, and the AB+M group had higher bone fill than the CT and autogenous bone graft without magnetic field (AB) groups – indicated in the graph for **. These results showed that 60 days after autogenous bone reconstruction, rats exposed to magnetic fields had a higher bone fill percentage than those without stimulation (Figure 3).
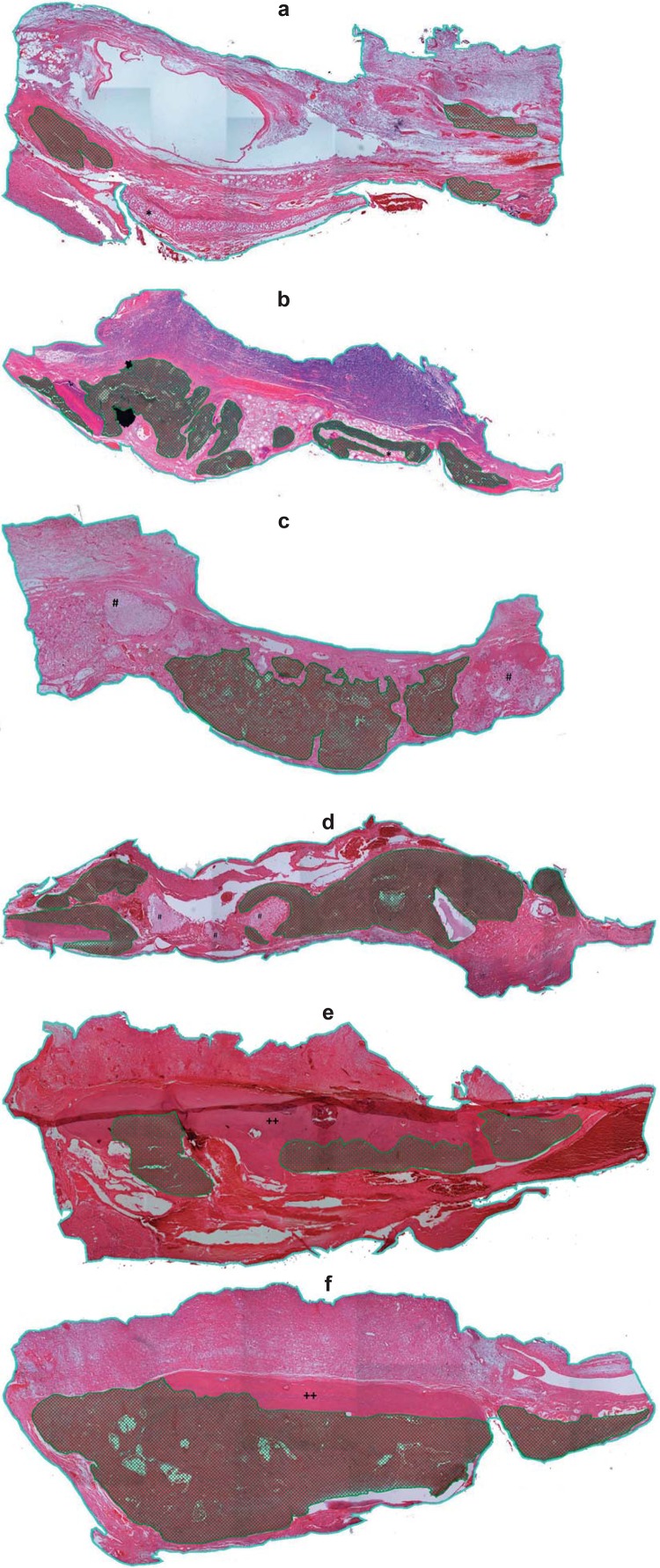
Figure 3. Histomorphometric analysis after reconstruction with autogenous bone graft, powdered synthetic hydroxyapatite implant, andallogeneic cartilage graft, with or without exposure to buried magnetic field, at 60 postoperative days (Blue lines define the total area of the bone defects, and cross hatched areas represent areas of new bone formation within the critical defect). d) Powdered synthetic hydroxyapatite group exposed to buried magnetic stimulation; e) Autogenous bone graft group not exposed to buried magnetic stimulation; f) Autogenous bone graft group exposed to buried magnetic stimulation. * indicates cartilage graft; # indicates hydroxyapatite implant; ++ indicates bone graft.
Longitudinal analyses were also performed to evaluate the development of each group over time. In the allogeneic cartilage graft with magnetic field (CT+M), hydroxyapatite implant without magnetic field (HA), AB, and AB+M groups, bone fill percentage after 15 days was lower than that found after 30 and 60 days. In HA+M rats, differences were only observed after 60 days, at which point bone fill percentages were significantly higher than those observed after 15 and 30 days. In the CT group, although differences in bone fill were observed between 15 and 60 days, bone fill at 30 postoperative days did not differ from that found after 15 and 60 days - represented in the graph by different capital letters. The graphical representation of the percentage of bone fill shows the between-group and longitudinal comparisons (Figure 4).
Figure 4. Graphical representation of the percentage of bone formation over time for all groups at 15 postoperative days: AB+M > CT (*); At 30 postoperative days: AB+M > CT (#); At 60 postoperative days: HA+M > CT (+); At 60 postoperative days: AB+M > CT e AB (**). Different capital letters show longitudinal differences.
DISCUSSION
The present study was based on the current understanding of the physiology of bone formation, and aimed to contribute to existing knowledge of the mechanisms involved in bone repair. The latter process is a key part of the response of the organism to bone tissue damage 13 , 23 .
In order to understand the influence of magnetic fields on bone repair, several studies have focused on histological parameters 20 , 21 , assessing the concentration of growth factors 1 , 11 , the deposition of calcium ions during ossification 26 , and even the influence of magnetism on cell plasma membranes 11 . The ability of different interventions to accelerate the bone repair process and contribute to the restoration of bone form and function has also been widely studied, often by the use of critical bone defects in rats 9 , 22 . Lastly, the quantification and comparison of tissues present in histological sections are generally performed by histomorphometric analysis 8 , 9 , 16 , 19 .
The present study evaluated the effects of a buried magnetic field on bone repair through the placement of magnets adjacent to a bone defect created according to the method outlined by Puricelli, et al. 21 (2006). Our results revealed that, after 60 postoperative days, rats treated with autogenous bone reconstruction and exposed to magnetic fields showed significantly higher bone fill than those who received a similar treatment but no magnetic stimulation. In the early postoperative period, although the average fill percentage was higher in groups which received magnetic stimulation, these differences were not statistically significant.
Autogenous grafts are considered the gold-standard of bone-grafting, and in the present study, rats that received such a treatment in addition to magnetic stimulation had the highest percentage of bone fill. The osteoinductive, osteoconductive and osteogenetic properties of autogenous grafts are known to have a positive influence on bone repair. Our results also point to a sustained positive effect of magnetic field exposure on bone repair.
Although the present study has produced promising evidence of the influence of magnetic fields on bone repair, there is still a need to identify the best method to evaluate this process, and to clarify its underlying biological mechanisms. Studies on tissue engineering using magnetized scaffolds have shown promising results 4 , as well as the influence of the static magnetic field in cell culture 28 .
In addition to the potential positive effect of magnetic fields on bone graft healing, other factors may also have a direct influence on the incorporation of bone grafts. For instance, one factor is the type of graft used. The present study involved the use of block cortical grafts harvested from the cranial bone. This type of graft has shown slower revascularization than bone marrow grafts; consequently, the incorporation of the former is always slower than that of the latter 13 . Another factor which may influence graft healing is the size of the graft particles used. In a study conducted in rabbits, the early stages of bone repair were found to be influenced by the size of the autogenous bone particles in the grafts used 17 . In the present work, the use of block grafts probably had a negative influence on bone repair after 15 and 30 postoperative days. According to Shapoff, et al. 24 (1980), the total volume of newly-formed bone in defects filled with small particles may be higher than that found in defects filled with larger particles after similar postoperative periods. Additionally, as Nagata, et al. 13 (2009) have also pointed out, there is a need to establish a lower limit for particle size, since bone particles smaller than 125 µm are susceptible to removal by macrophages.
Despite being considered the gold standard for bone reconstruction, autogenous bone grafting is associated with several limitations, the most important of which are surgical morbidity at the donor site, the limited supply of graft quantity, and the irregular resorption of the graft 2 . Therefore, the present study sought to evaluate alternative materials that could effectively replace autogenous bone. The main requirements for the success of a bone substitute are biocompatibility, bioactivity, and adequate mechanical properties. Hydroxyapatite has been extensively studied as a bone substitute, and has been widely used for the treatment of bone defects. Its chemical formula is similar to that of inorganic bone tissue, which may explain its intense affinity to bone 7 . Although the quality of hydroxyapatite may vary between manufacturers, this has not had a significant impact on the results of studies involving the use of this material. However, the size of hydroxyapatite pores has been found to influence its filling by osteoblasts, and materials with a pore size of 150 to 500 µm are considered ideal for grafting 15 .
Our histological assessments allowed for the confirmation of the biocompatibility of the materials used, since graft rejection was not observed in any of the animals used. In the vast majority of cases, hydroxyapatite granules were surrounded by granulation tissue, suggesting possible future bone neoformation. The osteoconductivity of hydroxyapatite was also demonstrated in the present study, corroborating the results found by other authors 7 , 18 . Bone formation was observed on the surface of hydroxyapatite at all operative times.
The materials involved in the present study did not adapt easily to the contours of the bone defects, possibly because of the shallowness of the bone cavities themselves. This relative instability of the material can lead to variations in the amount of newly-formed bone within the same experimental group 7 . Some authors have used bone substitutes supported by membranes 9 , 10 , 12 or to secure graft stability. The use of this method has led to promising results, especially when used in conjunction with growth factors and osteoinductive substances 7 , 14 .
The role of cartilage in bone formation and repair was also examined in the present study, since bone defects in one of the experimental groups were treated with allogeneic cartilage grafts. Cartilage grafts have been used by several authors in oral and maxillofacial reconstruction, and has been found to have several advantages associated with its long-term integrity and survival 5 . In the present study, the animals treated with allogeneic cartilage grafts showed the lowest percentage of bone fill, possibly because the resorption of this material was slower than that of the other bone substitutes used. In a study on rats performed by Vieira, et al. 25 (1993), in which different means of cartilage graft preservation were compared, the authors found that the cartilage only began to be replaced by bone after 120 postoperative days.
The results obtained in the present study emphasize the importance of removing the bone segments between the magnets and the central region of the bone defect to allow for more intense magnetic fields and produce more favorable effects on bone repair.
CONCLUSION
The present findings led to the following conclusions:
a) After 60 postoperative days, bone repair, as indicated by bone fill percentages, was exposed to a magnetic field and it was greater in the group treated with autogenous bone grafts than in the group treated with autogenous bone grafts and not exposed to magnetic fields;
b) Bone repair was most pronounced in animals treated with autogenous bone grafts, followed by those treated with powdered synthetic hydroxyapatite and allogeneic cartilage grafts.
The present research has contributed to the understanding of the influence of buried magnetic fields on bone repair. It is suggested that future studies invest in new methods that allow them to complement the present results and strengthen this line of research.
ACKNOWLEDGEMENTS
This study was undertaken by Surg. Maíra Cavallet de Abreu as a requirement for her Master’s degree in oral and maxillofacial surgery, under the supervision of Professor Dr. Edela Puricelli (School of Dentistry/Federal University of Rio Grande do Sul). The authors declare no conflict of interest. This study received financial support from the Research Incentive Fund of Hospital de Clínicas de Porto Alegre (FIPE/HCPA). Financial support for the purchase of histopathology equipment was obtained from Rio Grande do Sul Research Foundation (FAPERGS, PROAP04/2005, Grant number 0410882).
REFERENCES
- 1.Aaron RK, Boyan BD, Ciombor DM, Schwartz Z, Simon BJ. Stimulation of growth factor synthesis by electric and electromagnetic fields. Clin Orthop Relat Res. 2005;(419):30–37. doi: 10.1097/00003086-200402000-00006. [DOI] [PubMed] [Google Scholar]
- 2.Albrektsson T, Johansson C. Osteoinduction, osteoconduction and osseointegration. Eur Spine J. 2001;10(2):S96–101. doi: 10.1007/s005860100282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bercini F, Puricelli E. Intra-osseous and subperiosteal grafts of cartilage, removing the perichondrium. Rev Odonto Ciênc. 1992;7(14):9–24. [Google Scholar]
- 4.Bock N, Riminucci A, Dionigi C, Russo A, Tampieri A, Landi E, et al. A novel route in bone tissue engineering: magnetic biomimetic scaffolds. Acta Biometer. 2010;6:786–796. doi: 10.1016/j.actbio.2009.09.017. [DOI] [PubMed] [Google Scholar]
- 5.Cavaliere M, Mottola G, Rondinelli M, Iemma M. Tragal cartilage in tympanoplasty: anatomic and functional results in 306 cases. Acta Otorhinolaryngol Ital. 2009;29(1):27–32. [PMC free article] [PubMed] [Google Scholar]
- 6.Costantino C, Pogliacomi F, Passera F, Concari G. Treatment of wrist and hand fractures with natural magnets: preliminary report. Acta Biomed. 2007;78(3):198–203. [PubMed] [Google Scholar]
- 7.Dinarvand P, Seyedjafari E, Shafiee A, Jandaghi AB, Doostmohammadi A, Fathi MH, et al. New approach to bone tissue engineering: simultaneous application of hydroxyapatite and bioactive glass coated on a poly(L-lactic acid) scaffold. ACS Appl Mater Interfaces. 2011;3(11):4518–4524. doi: 10.1021/am201212u. [DOI] [PubMed] [Google Scholar]
- 8.Fini M, Cadossi R, Canè V, Cavani F, Giavaresi G, Krajewski A, et al. The effect of pulsed electromagnetic fields on the osteointegration of hydroxyapatite implants in cancellous bone: a morphologic and microstructural in vivo study. J Orthop Res. 2002;20(4):756–763. doi: 10.1016/S0736-0266(01)00158-9. [DOI] [PubMed] [Google Scholar]
- 9.Furlaneto FA, Nagata MJ, Fucini SE, Deliberador TM, Okamoto T, Messora MR. Bone healing in critical-size defects treated with bioactive glass/calcium sulfate: a histologic and histometric study in rat calvaria. 311Clin Oral Implants Res. 2007;18(3) doi: 10.1111/j.1600-0501.2006.01331.x. [DOI] [PubMed] [Google Scholar]
- 10.Haddad AJ, Peel SA, Clokie CM, Sándor GK. Closure of rabbit calvarial critical-sized defects using protective composite allogeneic and alloplastic bone substitutes. J Craniofac Surg. 2006;17(5):926–934. doi: 10.1097/01.scs.0000230615.49270.d1. [DOI] [PubMed] [Google Scholar]
- 11.Icaro Cornaglia A, Casasco M, Riva F, Farina A, Fassina L, Visai L, et al. Stimulation of osteoblast growth by an electromagnetic field in a model of bone-like construct. Eur J Histochem. 2006;50(3):199–204. [PubMed] [Google Scholar]
- 12.Marzouk KM, Gamal AY, Al-Awady AA, Sharawy MM. Osteoconductive effects of vinyl styrene microbeads in rat calvarial defects. J Oral Maxillofac Surg. 2007;65(8):1508–1516. doi: 10.1016/j.joms.2006.10.039. [DOI] [PubMed] [Google Scholar]
- 13.Nagata M, Messora M, Okamoto R, Campos N, Pola N, Esper L, et al. Influence of the proportion of particulate autogenous bone graft/platelet-rich plasma on bone healing in critical-size defects: an immunohistochemical analysis in rat calvaria. Bone. 2009;45(2):339–345. doi: 10.1016/j.bone.2009.04.246. [DOI] [PubMed] [Google Scholar]
- 14.Neovius E, Engstrand T. Craniofacial reconstruction with bone and biomaterials: review over the last 11 years. J Plast Reconstr Aesthet Surg. 2010;63(10):1615–1623. doi: 10.1016/j.bjps.2009.06.003. [DOI] [PubMed] [Google Scholar]
- 15.Notodihardjo FZ, Kakudo N, Kushida S, Suzuki K, Kusumoto K. Bone regeneration with BMP-2 and hydroxyapatite in critical-size calvarial defects in rats. J Craniomaxillofac Surg. 2012;40(3):287–291. doi: 10.1016/j.jcms.2011.04.008. [DOI] [PubMed] [Google Scholar]
- 16.Oliveira RC, Oliveira FH, Cestari TM, Taga R, Granjeiro JM. Morphometric evaluation of the repair of critical-size defects using demineralized bovine bone and autogenous bone grafts in rat calvaria. Clin Oral Implants Res. 2008;19(8):749–754. doi: 10.1111/j.1600-0501.2008.01521.x. [DOI] [PubMed] [Google Scholar]
- 17.Pallesen L, Schou S, Aaboe M, Hjørting-Hansen E, Nattestad A, Melsen F. Influence of particle size of autogenous bone grafts on the early stages of bone regeneration: a histologic and stereologic study in rabbit calvarium. Int J Oral Maxillofac Implants. 2002;17(4):498–506. [PubMed] [Google Scholar]
- 18.Paris MF, Oliveira MG, Puricelli E, Ramalho LP. Reconstruction of alveolar bone in dentolous area, with use of the hidroxiapatite, histological analysis: Experimental study. Rev Odonto Ciênc. 2003;18(39):89–98. [Google Scholar]
- 19.Pryor ME, Polimeni G, Koo KT, Hartman MJ, Gross H, April M, et al. Analysis of rat calvaria defects implanted with a platelet-rich plasma preparation: histologic and histometric observations. J Clin Periodontol. 2005;32(9):966–972. doi: 10.1111/j.1600-051X.2005.00772.x. [DOI] [PubMed] [Google Scholar]
- 20.Puricelli E, Dutra NB, Ponzoni D. Histological evaluation of the influence of magnetic field application in autogenous bone grafts in rats. 1Head Face Med. 2009;5 doi: 10.1186/1746-160X-5-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Puricelli E, Ulbrich LM, Ponzoni D, Filho JJ. Histological analysis of the effects of a static magnetic field on bone healing process in rat femurs. 43Head Face Med. 2006;2 doi: 10.1186/1746-160X-2-43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schmitz JP, Hollinger JO. The critical size defect as an experimental model for craniomandibulofacial nonunions. Clin Orthop Relat Res. 1986;(205):299–308. [PubMed] [Google Scholar]
- 23.Shapiro F. Bone development and its relation to fracture repair. The role of mesenchymal osteoblasts and surface osteoblasts. Eur Cell Mater. 2008;15:53–76. doi: 10.22203/ecm.v015a05. [DOI] [PubMed] [Google Scholar]
- 24.Shapoff CA, Bowers GM, Levy B, Mellonig JT, Yukna RA. The effect of particle size on the osteogenic activity of composite grafts of allogeneic freeze-dried bone and autogenous marrow. J Periodontol. 1980;51(11):625–630. doi: 10.1902/jop.1980.51.11.625. [DOI] [PubMed] [Google Scholar]
- 25.Vieira EH, Gabrielli MA, Okamoto T, Gabrielli MF, Scarso J, Filho, Ramalho LO, et al. Allogeneic transplants of rib cartilage preserved in 98% glycerol or 70% alcohol into the malar process of rats: a comparative histological study. J Nihon Univ Sch Dent. 1993;35(2):96–103. doi: 10.2334/josnusd1959.35.96. [DOI] [PubMed] [Google Scholar]
- 26.Yan QC, Tomita N, Ikada Y. Effects of static magnetic field on bone formation of rat femurs. Med Eng Phys. 1998;20(6):397–402. doi: 10.1016/s1350-4533(98)00051-4. [DOI] [PubMed] [Google Scholar]
- 27.Zhang J, Ding C, Ren L, Zhou Y, Shang P. The effects of static magnetic fields on bone. Prog Biophys Mol Biol. 2014;114(3):146–152. doi: 10.1016/j.pbiomolbio.2014.02.001. [DOI] [PubMed] [Google Scholar]
- 28.Zhang J, Ding C, Shang P. Alterations of mineral elements in osteoblast during differentiation under hypo, moderate and high static magnetic fields. Biol Trace Elem Res. 2014;162(1-3):153–157. doi: 10.1007/s12011-014-0157-7. [DOI] [PubMed] [Google Scholar]