Abstract
Although radiofrequency (RF) ablation has been accepted as a curative treatment modality for solid organ tumors, intraductal RF ablation for malignant biliary obstruction has not been widely described.
The aim of this study was to evaluate the feasibility, safety, and efficacy (in terms of stent patency and survival) of intraductal RF ablation combined with biliary stent placement for nonresectable malignant biliary obstruction.
A search of the nonresectable malignant extrahepatic biliary obstruction database (179 patients) identified 18 consecutive patients who were treated with biliary intraluminal RF ablation during percutaneous transhepatic cholangiodrainage and inner stent placement (RF ablation group) and 18 patients who underwent inner stent placement without biliary intraluminal RF ablation (control group). The patients were matched for tumor type, location of obstruction, tumor stage, and Child–Pugh class status. Primary endpoints included safety, stent patency time, and survival rates. The secondary endpoint was effectiveness of the technique.
The RF ablation and control groups were closely matched in terms of age, diagnosis, presence of metastases, presence of locally advanced tumor, American Society of Anesthesiologists (ASA) grade, and chemotherapy regimen (all P > 0.05). The technical success rate for both groups was 100%. The median time of stent patency in the RF ablation and control groups were 5.8 (2.8–11.5) months and 4.5 (2.4–8.0) months, respectively (Kaplan–Meier analysis: P = 0.03). The median survival times in the RF ablation and control groups were 6.1 (4.8–15.2) months and 5.8 (4.2–16.5) months, with no significant difference according to Kaplan–Meier analysis (P = 0.45). In univariate and multivariate analyses, poorer overall survival was associated with advanced age and presence of metastases (P < 0.05).
Intraductal RF ablation combined with biliary stent placement for nonresectable malignant biliary obstruction is safe and feasible and effectively increases stent patency time. However, it does not improve patient survival.
INTRODUCTION
The vast majority of patients with extrahepatic cholangiocarcinoma present with malignant biliary obstruction. The diagnosis is usually made when the disease is already in an advanced stage, making it inoperable.1 In such cases, palliation with drainage of the biliary tree is of paramount importance to maintain the quality of life. Stent placement using endoscopic retrograde cholangiopancreatography (ERCP) or percutaneous transhepatic cholangiodrainage (PTCD) is the standard technique to ensure continued biliary drainage.2 Both the patient's quality of life and cost-effectiveness of the procedure depend on stent patency, making stent patency time a crucial factor. Unfortunately, the tumor may grow through the mesh of the stent, leading to in-stent occlusion.3
Radiofrequency (RF) ablation can lead to complete tumor necrosis and has been accepted as a curative treatment modality for hepatocellular carcinoma at an early stage owing to its safety and effectiveness.4 Recent preliminary reports have described intraductal RF ablation using the Habib EndoHBP catheter for malignant biliary obstruction.5–15 Intraductal RF ablation combined with biliary stent placement may potentially prolong stent patency.7–13 Moreover, sporadic reports show that RF ablation appears to improve survival in patients with malignant biliary strictures.14,15 In fact, intraductal RF ablation for nonresectable malignant biliary obstruction could reduce the tumor load and thus delay local tumor growth. However, radical tumor elimination is difficult to achieve with intraductal RF ablation because of the limited heat penetration depth.5,6 Therefore, intraductal RF ablation may eventually constitute a form of adjuvant therapy for unresectable extrahepatic cholangiocarcinoma.
The purpose of this study was to evaluate the feasibility, safety, and efficacy (stent patency and survival) of intraductal RF ablation combined with biliary stent placement for nonresectable malignant biliary obstruction. Moreover, we explored the feasibility of repeated RF ablation for in-stent restenosis.
PATIENTS AND METHODS
Study Design
The study was reviewed and approved by the ethics committees of Beijing Chao-yang Hospital Affiliated with Capital Medical University according to the standards of the Declaration of Helsinki. Written informed consent was waived because of the retrospective nature of this study. All the records of patients were anonymized and de-identified before analysis.
Between May, 2011, and August, 2014, 18 patients diagnosed with unresectable malignant extrahepatic bile duct obstruction were treated with endobiliary RF ablation via a percutaneous transhepatic cholangial approach, which was followed by a placement of metallic stent. Malignant extrahepatic biliary obstruction was caused by cholangiocarcinoma, pancreatic cancer, or gallbladder cancer. Only patients who had the life expectancy longer than 3 months were included in this study.12 Exclusion criteria were (1) tumor load of >70%; (2) partial or complete main portal vein tumor thrombosis; (3) Child–Pugh class C, serum creatinine level > 2 × baseline level, or American Society of Anesthesiologists (ASA) grade ≥ 4; (4) intractable severe blood coagulation dysfunction; (5) massive refractory ascites; or (6) refusal of biliary intraluminal RF ablation treatment.
A cohort of 179 patients who were diagnosed with unresectable malignant extrahepatic biliary obstruction and underwent biliary stent placement alone were used as the candidates of subjects in the control group. To obtain a robust comparison of outcomes, we selected a subgroup of patients from the 179 patients who well matched the patients treated with endobiliary RF ablation plus biliary stenting in the context of their tumor type, location of obstruction, relative tumor stage, ASA category, and the presence or absence of metastases at the time of treatment.
Each case was discussed in a multidisciplinary setting. Locally advanced biliary tract cancer was defined as a cancer for which surgical resection was impossible even via an aggressive surgical procedure, including combined vascular resection. In practice, this referred to (1) local invasion of the hepatic artery precluding reconstruction; (2) local invasion of the portal vein precluding reconstruction; (3) local invasion of the hepatic vein precluding reconstruction; (4) extensive infiltration of the bile duct precluding curative resection; and (5) extensive hepatic invasion precluding excision owing to insufficient remnant liver volume even after portal vein embolization.16 Locally advanced pancreatic cancer was defined as a cancer of stage III (T4, any N, M0), or a cancer with encasement of >180° of the circumference of the superior mesenteric artery or the coeliac arteries, or a cancer with any unreconstructable venous involvement.17
RF Ablation Combined With Biliary Stent Placement
Intraductal RF ablation combined with biliary stent placement was performed by 2 physicians with 10 and 20 years of experience. The patient was placed in a supine position on the table. An access to the biliary system was established through the procedure of percutaneous transhepatic cholangial drainage under general anesthesia. After the location and extent of the malignant stricture was identified, under fluoroscopic guidance, a Habib EndoHPB bipolar RF ablation catheter (EMcision, Hitchin Herts, UK) was inserted in the extrahepatic bile duct and the electrode was positioned exactly at the site of stenotic bile duct. The ablation procedure was started at the distal portion of the stricture. The output power was 10 W, and the ablation time was 1.5 to 2 minutes. For the stricture which was longer than 2.5 cm, sequential applications were applied to ensure RF ablation treatment throughout the length of the stricture. A dilation balloon was inserted over a 0.035 inch guide wire to dilate the stenosis in case it was not able to be reopened by RF ablation. Uncovered self-expanding metal stents (SEMSs) were deployed across the stenosis after RF ablation. A following cholangiography was performed to confirm the opening and location of the SEMS. After RF ablation and stenting, a 10 French drainage catheter was kept in the bile duct for bile drainage. After 5 to 7 days of the RF ablation, a cholangiography confirmed that biliary patency and then the drainage catheter were removed.
In the control group, patients underwent stent insertion alone and the stent type and technique were the same as those in the RF ablation group.
Postoperative Evaluation
After the procedure, the patients were administered prophylactic antibiotics and received symptom-based treatments. At 1 week after the procedure, they were examined for remission of jaundice and short-term complications, and the serum bilirubin concentration was recorded.
After discharge, stent patency and patient survival were recorded based on follow-up outpatient visits or telephone interviews. A stent was considered patent if jaundice was not observed, direct bilirubin level was normal, and bile duct expansion was not displayed on contrast-enhanced computed tomography (CT) or ultrasonography during postoperative follow-up. If jaundice was observed and liver function tests showed an increase in the total or direct bilirubin level, or if jaundice was not observed but the total or direct bilirubin level was found to have increased during follow-up, contrast-enhanced CT was performed to confirm stent obstruction. On imaging, the bile duct above the stent appeared obviously expanded if stent blockage had occurred, indicating the loss of patency. We repeated RF ablation and/or inserted a new stent in such cases in the RF ablation group and the control group. If the new stent could not be fully expanded, it was dilated with a balloon. All patients were followed up once per month until death.
Study Endpoints
The primary endpoints of the study were safety, stent patency time, and survival rates. The secondary endpoint was the effectiveness of the primary technique.
Statistical Analysis
All statistical analyses were performed using the SPSS 15.0 statistical software (SPSS Inc, Chicago, IL). Continuous data were expressed as mean ± SD. Continuous variables of the 2 groups were compared using Student's t test. Categorical variables were analyzed using the χ2 test or Fisher's exact test. Stent patency time and survival rates were calculated using the Kaplan–Meier method and compared using the log-rank test. Multivariate analysis was performed using Cox proportional hazards modeling. All reported P-values are 2-sided. A P-value < 0.05 was considered statistically significant.
RESULTS
Baseline Characteristics and Technique Effectiveness
Of the 36 patients, 26 (72.2%) were men and 10 (27.8%) were women. The RF ablation and control groups were closely matched in terms of age, diagnosis, presence of metastases, presence of locally advanced tumor, ASA grade, and chemotherapy regimen (P > 0.05). The patients’ demographic characteristics are shown in Table 1.
TABLE 1.
Demographic Characteristics of Patients in the Study

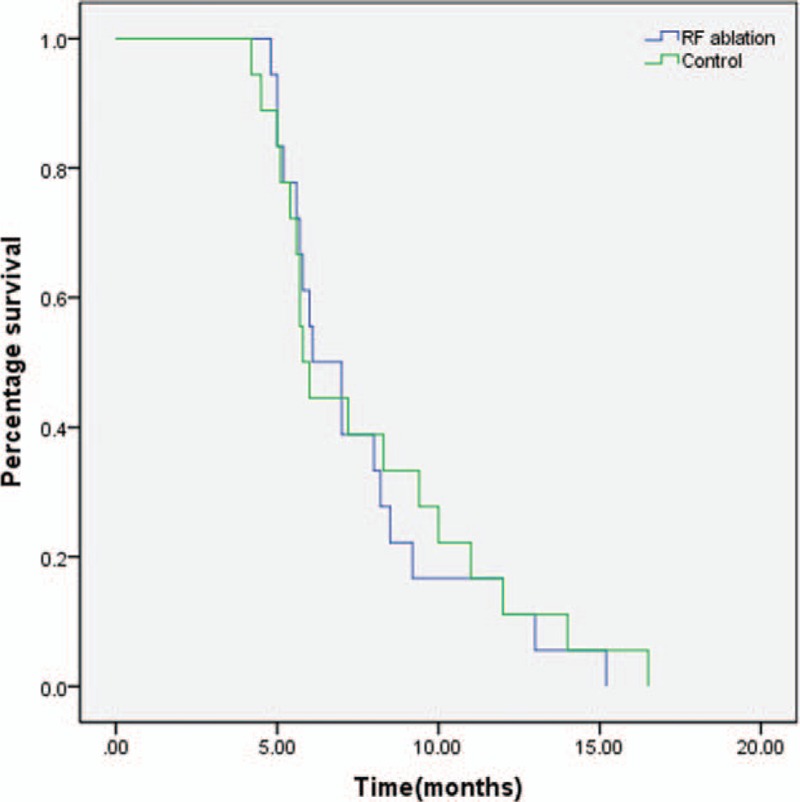
Both procedures were performed successfully in all the patients (Figure 1). Each patient received 1 SEMS. The mean stricture length was 38.4 ± 10.6 mm in the RF ablation group versus 40.5 ± 9.5 mm in the control group (P = 0.65; Table 2). There was no difference in pre- and post-stent stricture diameter between the 2 groups (P > 0.05; Table 2). However, there was a significant improvement in stricture diameter in both the RF ablation (4.2 ± 1.2 vs. 1.0 ± 0.7 mm, P = 0.01) and control groups (3.4 ± 1.4 versus 1.0 ± 0.9 mm, P < 0.01) after stent placement.
FIGURE 1.
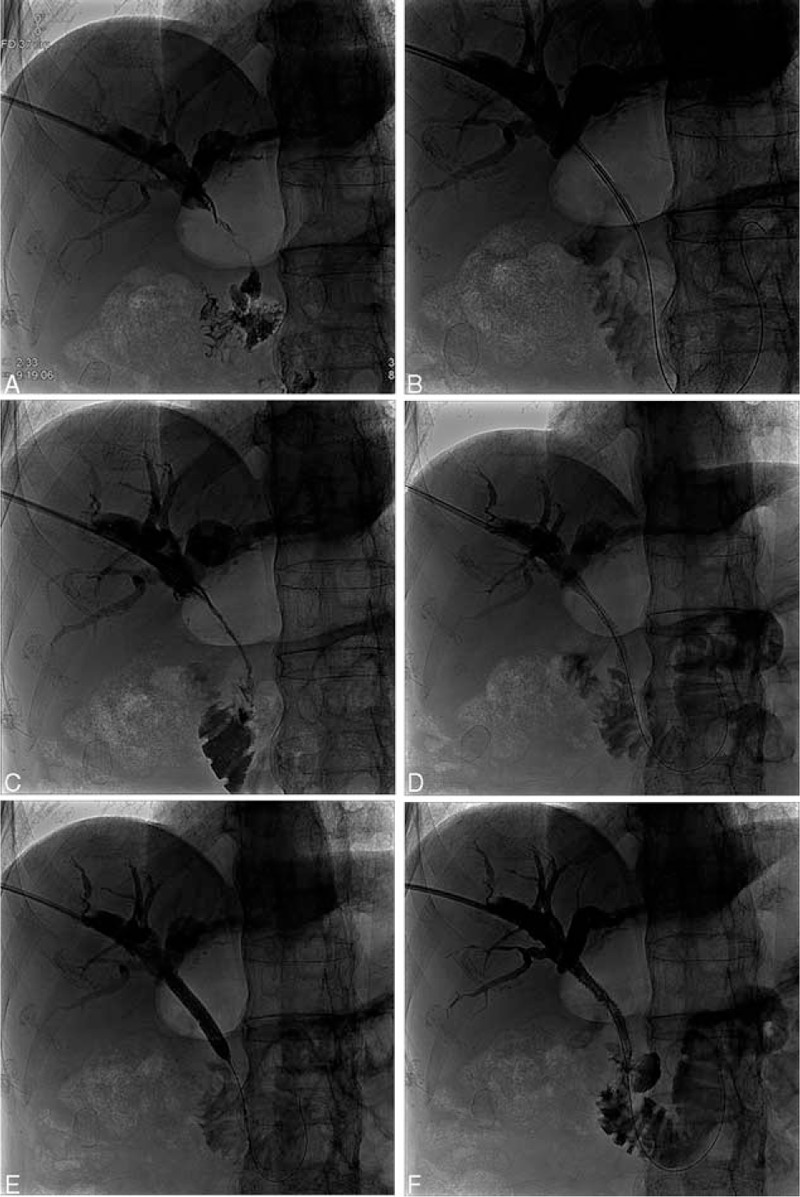
The site of biliary obstruction due to the tumor in a 63-year-old man with a highly differentiated adenocarcinoma of the proximal third of the bile duct confirmed by pathologic diagnostic forceps biopsy. (A) Cholangiography before ablation showed dilated intrahepatic biliary ducts and obstruction at the upper bile duct. (B) Intraductal RF ablation was performed with a percutaneous RF catheter using a 0.035-inch guide wire. (C) The obstruction was relieved slightly by the ablation procedure. (D) A balloon catheter was inserted into the bile duct. (E) Balloon dilating after the intraductal RF ablation. (F) A self-expanding metallic stent was inserted after the ablation. Cholangiography showed that the obstruction was relieved. RF = radiofrequency.
TABLE 2.
Stricture and Procedure Characteristics
Complications
In both groups, no electrocardiographic abnormalities were observed during the procedure. There were also no instances of bile duct perforation, peribiliary sepsis, or hemorrhage. After the operation, 3 patients in the RF ablation group had symptoms of cholangitis (1, 3, and 8 h after the procedure) such as abdominal pain, chills, and fever, which were resolved with antibiotic therapy and other conservative treatments. Subsequent repeat liver function and routine blood tests showed that the laboratory values returned to baseline levels. There were no cases of hemobilia, perforation, bile leak, or pancreatitis.
Stent Patency Time
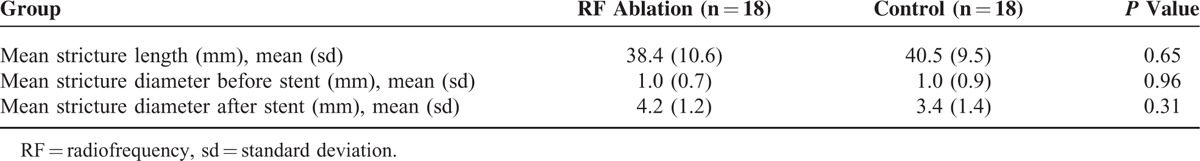
Stent patency time was counted from the day of stent insertion to the day of proven stent occlusion or patient's death. The median stent patency time in the RF ablation and control groups were 5.8 (2.8–11.5) months and 4.5 (2.4–8.0) months, respectively, with a significant difference revealed with Kaplan–Meier analysis (P = 0.03) (Figure 2).
FIGURE 2.
A Kaplan–Meier curve showing a significant difference in overall stent patency time between the RF ablation group and the control group (P = 0.03). The median stent patency times in the RF ablation and control groups were 5.8 (2.8–11.5) months and 4.5 (2.4–8.0) months, respectively. RF = radiofrequency.
In the RF ablation group, 3 patients underwent repeat PTCD to confirm stent occlusion and received new stents after RF ablation of the obstructed stent. After confirming stent occlusion with percutaneous transhepatic cholangiography, a guide wire was inserted through the obstructed stent. An ablation catheter was introduced, and the obstruction was ablated. After ablation, a half-inflated balloon was inserted through the guide wire and moved back and forth through the stent into the duodenum to remove ablated tissue and debris from the metal stent. A new SEMS was inserted. Cholangiography after stent placement showed that the obstruction was relieved.
In the control group, 10 patients had confirmed stent occlusion and received new stents. Moreover, 11 patients in the 2 groups received palliative PTCD after stent occlusion because of life expectancy being <3 months and/or poor performance status.
Survival Rates
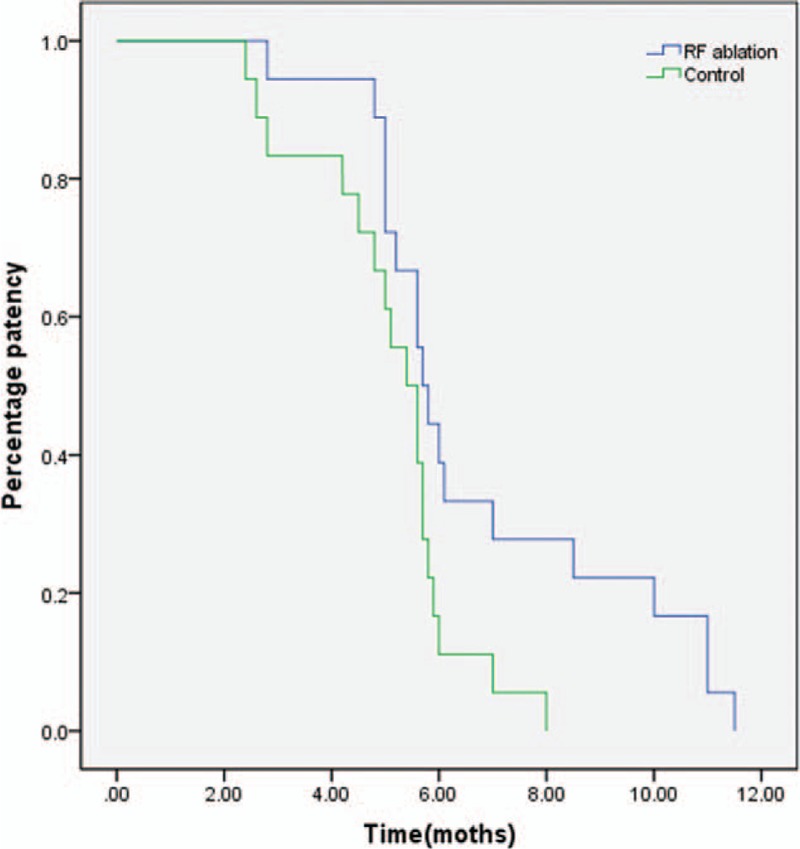
The median survival time in the RF ablation and control groups was 6.1 (4.8–15.2) months and 5.8 (4.2–16.5) months, respectively, with no difference revealed by Kaplan–Meier analysis (P = 0.45) (Figure 3).
FIGURE 3.
Kaplan–Meier analysis showed that there was no difference in survival between the RF ablation group and the control group (P = 0.45). The median survival times in the RF ablation and control groups were 6.1 (4.8–15.2) months and 5.8 (4.2–16.5) months, respectively. RF = radiofrequency.
Table 3 lists the results of univariate and multivariate analyses using Cox regression tests to identify independent prognostic factors for poor survival. Factors that significantly predicted poor survival in the univariate analysis were advanced age and presence of metastases. The multivariate analysis confirmed that advanced age (odds ratio [OR] = 1.076; 95% confidence interval [CI]: 0.745–1.380; P = 0.04) and presence of metastases (OR = 1.905 95%CI: 1.237–2.984; P = 0.03; Table 3) were associated with poor survival. All the 36 patients died of progressive tumor (n = 29) or systemic failure (n = 7) rather than from cholangitis or biliary obstruction.
TABLE 3.
Significant Variables for Overall Survival in the Univariate and Multivariate Analysis (n = 36)

DISCUSSION
The main finding of our study is that intraductal RF ablation combined with biliary stent placement is a safe and effective therapeutic alternative for nonresectable malignant biliary obstruction. The RF ablation and stenting procedure was successful in all the 18 patients. Moreover, there were no complications such as biliary tract bleeding, perforation, or bile leakage in the RF ablation group. The stent patency time was much higher in the RF ablation group than in the control group, indicating that local tumor growth was indeed inhibited. In addition, repeated RF ablation combined with stent placement was feasible in cases with recurrence of biliary obstruction.
Over the last 2 decades, endobiliary stenting via a PTCD or ERCP approach has been accepted as the preferred option for resolving the malignant stenosis in patients who are not candidates of surgical removal of the tumors. However, it is a challenging task to achieve a long-term biliary patency using a palliative treatment of endobiliary stent placement for patients with unresectable malignant biliary obstruction.2,3 To improve the dismal short-time patency of plastic stents, metallic stents including bare metal, covered-metal or recently used drug-eluting stents have been used to obtain a much longer time of patency. However, recurrence of bile duct occlusion usually occurs within 6 to 8 months of stent placement due to tumor ingrowth or overgrowth, sludge accumulation, or biofilm formation.14 The placement of covered SEMSs and photodynamic therapy have been reported as alternative approaches to prolong the time of stent patency but each has its own inherent shortfall. Covered SEMSs are associated with an increased risk of pancreatitis and cholecystitis and the application of photodynamic therapy is prone to inducing cholangitis and photosensitivity.14 At our institution, all patients with nonresectable malignant biliary obstruction receive uncovered SEMSs.
RF ablation has been widely used clinically for the treatment of solid organ tumors, especially hepatic malignancies.4 Because of its presumably local effect on tumor tissue, it has recently been tested for ability to improve stent patency.5–13 The procedure requires a disposable, bipolar, over-the-wire RF catheter suitable for endoluminal delivery of RF into the biliary tree (Habib EndoHPB, EMcision Ltd, London, UK). It is an 8-F (2.6-mm) catheter with a 180-cm working length that can be deployed through endoscope working channels with a diameter of at least 3.2 mm.
In the first clinical study to assess the safety and efficacy of RF ablation in management of malignant biliary obstruction by Steel et al,7 the ablation was performed at a power of 7 to 10 W for 2 minutes to achieve the balance between efficient tumor necrosis and limited thermal injury to adjacent tissues. Several studies8–15 mostly conducted in patients with cholangiocarcinoma (>75%) have subsequently demonstrated that intraductal RF ablation combined with biliary stent placement for malignant biliary obstruction is safe and feasible and effectively increases stent patency time. Furthermore, intraductal RF ablation has also been used in the treatment of occluded SEMSs secondary to tumor ingrowth or overgrowth, a frequent complication of SEMS placement.13 Our study confirms that RF ablation in combination with stent placement is a safe and effective technique for biliary decompression in patients with either nonresectable malignant biliary obstruction or occluded SEMSs.
A retrospective study comparing RF ablation combined with SEMS placement with SEMS placement alone showed that RF ablation appears to improve survival in patients with end-stage cholangiocarcinoma and pancreatic cancer.14 In this published study, although there was no difference in survival between the 2 groups, RF ablation was an independent predictor of survival along with age and use of chemotherapy. Recently, Kallis et al15 carried out a retrospective analysis of 23 patients with nonresectable pancreatic carcinoma and malignant biliary obstruction who underwent endoscopic RF ablation and SEMSs insertion and of 46 controls who were treated with SEMS insertion alone. According to their report, RF ablation did not appear to alter absolute stent patency time. Although Kaplan–Meier analysis revealed no obvious differences in survival between the 2 groups (P = 0.09, hazard ratio 0.660, 95% CI 0.410–1.063), RF ablation was independently predictive of survival at 90 days (OR 21.07, 95 % CI 1.45–306.64, P = 0.03) and 180 days (OR 4.48, 95% CI 1.04–19.30, P = 0.04) in multivariate analysis. The authors suggested that RF ablation may have indirect antitumor effects that improve the survival of the patients.14,15
In contrast, our data show that RF ablation did not offer a survival benefit in patients with nonresectable malignant biliary obstruction. There are at least 2 explanations for this result. First, as Kallis et al have stated,15 the vast majority of patients with nonresectable malignant biliary obstruction die of progressive malignant disease or carcinomatosis rather than from cholangitis or stent obstruction. Second, we used RF ablation as a clinical adjuvant therapy to eliminate the nonresectable malignant mass causing biliary obstruction rather than as a radical treatment. Recurrence of the residual tumor after such incomplete RF ablation is likely inevitable and eventually influences survival. Furthermore, the phenomenon of rapid tumor progression after RF ablation observed in many clinical centers is a poor prognostic indicator for malignant tumors and may indicate uncontrollable tumor progression and metastasis.18–22 We did not observe this phenomenon in the present study, which is indirectly reflected by the prolonged stent patency. However, our results also showed that incomplete RF ablation does not increase the survival rate in patients with nonresectable malignant biliary obstruction.
This study is limited by its retrospective nature and small patient population. Furthermore, although we attempted to identify the best matches among the control patients, selection bias is not excluded. Moreover, as the main aim was to control tumor growth, we used RF ablation in all the patients from the study group without considering its potential effect on some tumors, such as extrahepatic cholangiocarcinoma and gallbladder adenocarcinoma. Prospective, randomized, single-disease, controlled studies with a large number of patients are underway to determine the efficacy of intraductal RF ablation for nonresectable malignant biliary obstruction and its effects on long-term biliary stent patency and survival rates.
In conclusion, intraductal RF ablation combined with biliary stent placement for nonresectable malignant biliary obstruction is safe and feasible and effectively increases stent patency time. However, it does not improve patient survival.
Footnotes
Abbreviations: CI = confidence interval, CT = computed tomography, ERCP = endoscopic retrograde cholangiopancreatography, OR = odds ratio, PTCD = percutaneous transhepatic cholangiodrainage, RF = radiofrequency, SEMS = self-expanding metal stent.
JW and LZ contributed equally to this study.
The authors have no funding and conflicts of interest to disclose.
REFERENCES
- 1.Singh A, Siddiqui UD. The role of endoscopy in the diagnosis and management of cholangiocarcinoma. J Clin Gastroenterol 2015; 49:725–737. [DOI] [PubMed] [Google Scholar]
- 2.Krokidis M, Hatzidakis A. Percutaneous minimally invasive treatment of malignant biliary strictures: current status. Cardiovasc Intervent Radiol 2014; 37:316–323. [DOI] [PubMed] [Google Scholar]
- 3.Saleem A, Leggett CL, Murad MH, et al. Meta-analysis of randomized trials comparing the patency of covered and uncovered self-expandable metal stents for palliation of distal malignant bile duct obstruction. Gastrointest Endoscop 2011; 74:321–327.e1-e3. [DOI] [PubMed] [Google Scholar]
- 4.Gao J, Wang SH, Ding XM, et al. Radiofrequency ablation for single hepatocellular carcinoma 3 cm or less as first-line treatment. World J Gastroenterol 2015; 21:5287–5294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Itoi T, Isayama H, Sofuni A, et al. Evaluation of effects of a novel endoscopically applied radiofrequency ablation biliary catheter using an ex-vivo pig liver. J Hepatobiliary Pancreat Sci 2012; 19:543–547. [DOI] [PubMed] [Google Scholar]
- 6.Zacharoulis D, Lazoura O, Sioka E, et al. Habib EndoHPB: a novel endobiliary radiofrequency ablation device. An experimental study. J Invest Surg 2013; 26:6–10. [DOI] [PubMed] [Google Scholar]
- 7.Steel AW, Postgate AJ, Khorsandi S, et al. Endoscopically applied radiofrequency ablation appears to be safe in the treatment of malignant biliary obstruction. Gastrointest Endoscop 2011; 73:149–153. [DOI] [PubMed] [Google Scholar]
- 8.Alis H, Sengoz C, Gonenc M, et al. Endobiliary radiofrequency ablation for malignant biliary obstruction. Hepatobiliary Pancreat Dis Int 2013; 12:423–427. [DOI] [PubMed] [Google Scholar]
- 9.Figueroa-Barojas P, Bakhru MR, Habib NA, et al. Safety and efficacy of radiofrequency ablation in the management of unresectable bile duct and pancreatic cancer: a novel palliation technique. J Oncol 2013; 2013:910897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mao EJ, Watson JB, Soares G, et al. Successful percutaneous endobiliary radiofrequency ablation for unresectable malignant biliary obstruction: a case report and review of the literature. J Gastrointest Cancer 2014; 45 Suppl 1:55–57. [DOI] [PubMed] [Google Scholar]
- 11.Rustagi T, Jamidar PA. Intraductal radiofrequency ablation for management of malignant biliary obstruction. Dig Dis Sci 2014; 59:2635–2641. [DOI] [PubMed] [Google Scholar]
- 12.Wu TT, Li HC, Li WM, et al. Percutaneous intraluminal radiofrequency ablation for malignant extrahepatic biliary obstruction: a safe and feasible method. Dig Dis Sci 2015; 60:2158–2163. [DOI] [PubMed] [Google Scholar]
- 13.Li TF, Huang GH, Li Z, et al. Percutaneous transhepatic cholangiography and intraductal radiofrequency ablation combined with biliary stent placement for malignant biliary obstruction. J Vasc Intervent Radiol 2015; 26:715–721. [DOI] [PubMed] [Google Scholar]
- 14.Sharaiha RZ, Natov N, Glockenberg KS, et al. Comparison of metal stenting with radiofrequency ablation versus stenting alone for treating malignant biliary strictures: is there an added benefit? Dig Dis Sci 2014; 59:3099–3102. [DOI] [PubMed] [Google Scholar]
- 15.Kallis Y, Phillips N, Steel A, et al. Analysis of endoscopic radiofrequency ablation of biliary malignant strictures in pancreatic cancer suggests potential survival benefit. Dig Dis Sci 2015; 60:3449–3455. [DOI] [PubMed] [Google Scholar]
- 16.Kato A, Shimizu H, Ohtsuka M, et al. Surgical resection after downsizing chemotherapy for initially unresectable locally advanced biliary tract cancer: a retrospective single-center study. Ann Surg Oncol 2013; 20:318–324. [DOI] [PubMed] [Google Scholar]
- 17.Vincent A, Herman J, Schulick R, et al. Pancreatic cancer. Lancet 2011; 378:607–620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Seki T, Tamai T, Ikeda K, et al. Rapid progression of hepatocellular carcinoma after transcatheter arterial chemoembolization and percutaneous radiofrequency ablation in the primary tumour region. Eur J Gastroenterol Hepatol 2001; 13:291–294. [DOI] [PubMed] [Google Scholar]
- 19.Imamura Y, Kohara K, Shibatou T, et al. Two cases of hepatocellular carcinoma showing rapid progression after radio-frequency ablation therapy. Nihon Shokakibyo Gakkai Zasshi 2002; 99:40–44.[in Japanese]. [PubMed] [Google Scholar]
- 20.Ruzzenente A, Manzoni GD, Molfetta M, et al. Rapid progression of hepatocellular carcinoma after radiofrequency ablation. World J Gastroenterol 2004; 10:1137–1140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Saboo SS, Krajewski KM, Jagannathan JP, et al. Rapid progression of combined hepatocellular carcinoma and cholangiocarcinoma. Cancer Imaging 2011; 11:37–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yamamoto K, Hiura M, Tabaru A, et al. Rapid progression of hepatocellular carcinoma in a patient with autoimmune hepatitis. Inter Med 2011; 50:1409–1413. [DOI] [PubMed] [Google Scholar]


