Abstract
A phosphate tether-mediated ring-closing metathesis study to the synthesis of Z-configured, P-stereogenic bicyclo[7.3.1]- and bicyclo[8.3.1]phosphates is reported. Investigations suggest that C3-substitution, olefin substitution, and proximity of the forming olefin to the bridgehead carbon of the bicyclic affect the efficiency and stereochemical outcome of the RCM event. This study demonstrates the utility of phosphate tether-mediated desymmetrization of C2-symmetric, 1,3-anti-diol-containing dienes in the generation of macrocyclic phosphates with potential synthetic and biological utility.
Keywords: phosphate tether, tripodal coupling, macrocyclic ring-closing metathesis, bicyclic phosphate triester, diastereotopic group differentiation, desymmetrization of C2-symmetric polyols, Z-selective ring-closing metathesis
Graphical Abstract

Introduction
The development of methods that allow for the mild, high-yielding, and predictable coupling of simple chemical fragments to provide complex intermediates from core motifs common to a variety of natural products stands at the forefront of modern-day organic synthesis. In particular, methods that employ temporary tether strategies have emerged as intriguing tools to control substrate reactivity, site specificity and stereochemical outcome of tether-mediated transformations.1 While the use and study of temporary silicon-based tether systems in RCM is well represented in the literature,2 on-going efforts in our group have sought to exploit the inherent properties of phosphate triesters to develop tripodal P-tether systems3 for the generation of complex polyol-containing intermediates en route to the total synthesis of natural products. This work has led to the completion of a number of total and formal syntheses, including dolabelide C,4 salicylihalimide A (formal synthesis),5 (−)-tetrahydrolipstatin,6 (+)-strictifolione,7 and lyngbouilloside (core macrolactone).8 In an effort to expand the scope of the method, in 2013, we completed a detailed study on the effects of ring size and stereochemical complexity on the successful formation of bicyclo[4.3.1]-,9 bicyclo[5.3.1]-,10 and bicyclo[7.3.1]phosphates11,12 via diastereoselective RCM (Figure 1).13 Earlier this year, we published a continuation of these efforts to provide a number of stereochemically complex bicyclo[6.3.1]phosphates,14 along with a few simple examples of bicyclo[7.3.1]- and bicyclo[8.3.1]phosphates (Figure 1).15,16 However, the complexity of large ring dynamics—combined with the small sample size of bicyclo[7.3.1]- and bicyclo[8.3.1]phosphates in both studies—can complicate the generalization of those findings, and since the previous work represented some of the first reports of RCM to afford phosphates of this ring size, much remains to be discovered in this particular area of macrocyclic phosphate synthesis. The work reported herein expands upon previous efforts by focusing on the formation of bicyclo[7.3.1]- and bicyclo[8.3.1]phosphates (10- and 11-membered RCM), with a special emphasis on the effect of olefin- and C3-substition (Figure 1), as well as proximity of the forming olefin to the bridgehead carbon of the resultant bicyclic phosphate, on the success and stereochemical outcome of the macrocyclic, ring-closing metathesis event. In addition, the bicyclic phosphates formed after RCM-mediated macrocyclization represent a unique class of phosphates with potential synthetic, as well as biological, utility.
Figure 1.
Ring-closing metathesis to medium- and large-ring containing bicyclo[n.3.1]phosphates
Results and Discussion
Synthesis of Bicyclo[7.3.1]phosphates
Prior to this report, the few examples of RCM to afford bicyclo[7.3.1]phosphates were limited to those resulting from the formation of a C7–C8 double bond within the bicyclic framework, and surprisingly, these reactions—when successful—provided the corresponding product as the E-isomer diastereoselectively (in diastereomeric ratios of > 20:1, Figure 1).12,16 However, as the RCM of analogous monocyclic Si-tethered systems proceeds with predominantly Z-configured olefin formation—even at larger ring sizes,17 there was some interest in attempting to perturb the stereoselectivity of the phosphate tether-mediated RCM to provide 10- and 11-membered rings to broaden the scope of the molecules accessible by this method. Thus, trienes 3 and 4 were chosen as model substrates to assess the effect of olefin substitution on the phosphate tether-mediated RCM to generate bicyclo[7.3.1]phosphates (Scheme 1).18
Scheme 1.
Ring-closing metathesis to bicyclo[7.3.1]phosphates via formation of C7–C8-trisubstituted olefins
Enantiomeric monochlorophosphates (R,R)-1 and (S,S)-13 were added to the lithium alkoxides of 5-hexyn-1-ol and 5-methyl-5-hexen-1-ol, respectively, to afford alkynyl diene 2 and triene 3 in 65% and 68% yield, respectively. Subsequent hydrosilylation of 2, using conditions developed by Trost and Ball,19 provided vinyl silyloxy-containing triene 4 in moderate yield. Unfortunately, though a variety of catalysts and reaction conditions were attempted, the formation of bicyclo[7.3.1]phosphates 5 and 6 could not be achieved by RCM. With these results in hand, combined with those obtained in the previous study,16 we hypothesized that the proximity of the forming olefin (C7–C8) to the bridgehead carbon (C9) of the resultant bicyclo[7.3.1]phosphate, in combination with C3-substitution, was a significant contributor to the success and stereoselectivity of the phosphate tether-mediated RCM. Moreover, it was conjectured that homologation of the C2-symmetric dienediol used to generate the corresponding phosphate trienes could have important stereochemical and conformational effects on the synthesis of bicyclo[7.3.1]phosphates by virtue of increasing the distance between the forming olefin (now, C6–C7) and the bridgehead carbon (C9).
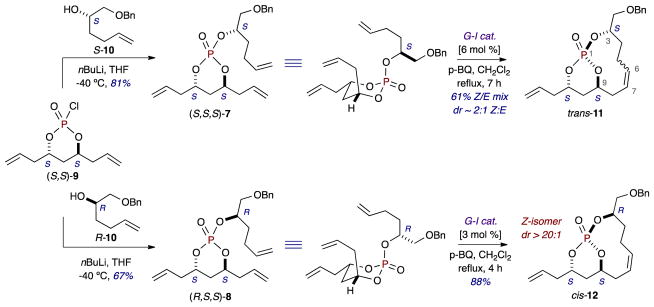
To assess this hypothesis, diastereomeric phosphate trienes 7 and 8 were synthesized from the coupling of monochlorophosphate (S,S)-912,16 and the corresponding chiral, non-racemic olefinic alcohols (S)-10 and (R)-10, respectively (Scheme 2). Treatment of triene 7 with Grubbs first generation catalyst (G-I), added in 1 mol % portions over 7 hours (6 mol %, total), provided the corresponding bicyclo[7.3.1]phosphates trans-1120 in 61% combined yield of a separable 2:1 mixture of Z/E-stereoisomers (with Z-trans-11 as the major stereoisomer).21 Notably, the formation of the analogous equivalent of this trans-bicyclo[7.3.1]phosphate via C7–C8 olefin formation was unsuccessful under a variety of RCM conditions,16 suggesting that the extension of the forming olefin from C7–C8 to C6–7 could provide for conformational flexibility that allows for the reaction of the latter to proceed (albeit with lower stereoselectivity). Conversely, RCM of triene 8 with G-I (3 mol %) provided Z-olefin-containing cis-12 in 88% as a single stereoisomer (dr > 20:1).
Scheme 2.
Synthesis of bicyclo[7.3.1]phosphates via formation of C6–C7-disubstituted olefins
In addition to C3-substitution, olefin substitution was introduced to assess the effect—if any—on the success and stereochemical outcome of RCM to generate bicyclo[7.3.1]phosphates. To separate the effect of olefin substitution from C3-substitution, we first synthesized simple triene 13 via the coupling of monochlorophosphate (R,R)-1412,16 with 4-penten-1-ol (Scheme 3). Exposure of 13 to Grubbs second-generation catalyst (G-II) provided the corresponding bicyclic phosphate 15 as the Z-isomer in 87% yield. Olefin configuration was confirmed by X-ray crystallographic analysis, as shown in Scheme 3.22 Interestingly, C3-substitution had little effect on the outcome of RCM for this set of bicyclo[7.3.1]phosphates, as the treatment of diastereomeric trienes 16 and 17 with G-II catalyst provided the expected bicyclic phosphates cis-18 and trans-19 in 81% and 87% yields, respectively, with excellent Z:E-diastereomeric ratios in each case. However, this substitution had a significant effect on the 1H and 13C NMR of trans-19, as high temperatures (95°C, DMSO-d6) and extended experiment times were required to partially resolve proton signals and properly identify short, broad carbon signals (See Supporting Information).
Scheme 3.
Synthesis of bicyclo[7.3.1]phosphates via formation of C6–C7-trisubstituted olefins
Finally, monochlorophosphate (S,S)-9 was coupled with 4-methyl-4-penten-1-ol to provide triene 20, armed with olefin substitution on the alcohol cross-partner, in 90% yield (Scheme 4). Triene 20 was treated with o-tolyl-Grubbs second-generation catalyst-variant Ru-21,23 in toluene at 35°C, to generate bicyclic phosphate 22 in 26% yield as the Z-isomer—in addition with a significant amount of oligomerization by-product. It should be noted that a number of reaction conditions were attempted to decrease the amount of oligomerization of 20 that occurs upon treatment with metathesis catalyst. However, extended reaction times and portion-wise addition of catalyst only led to an increase in oligomerization and a decrease in yield, presumably due to the type I nature of the mono-substituted olefins in pseudo-C2-symmetric triene 20 and the resultant exocyclic olefin in bicyclic phosphate 22.24
Scheme 4.
Synthesis of bicyclo[7.3.1]phosphates via formation of C6–C7-trisubstituted olefins
Synthesis of Bicyclo[8.3.1]phosphates
The method was extended to include the synthesis of bicyclo[8.3.1]phosphates via the formation of a C7–C8 olefin in order to determine the effects of C3-stereochemistry, olefin substitution, and proximity of the forming olefin to the bridgehead carbon (C10) on the success and stereochemical outcome of the RCM event. Studies commenced with the synthesis of diastereomeric trienes 23 and 24 from monochlorophosphate 9 and the corresponding chiral, non-racemic olefin-containing alcohols (S)-25 and (R)-25 (Scheme 5). Triene 23 was treated with G-I catalyst, in refluxing methylene chloride, to provide trans-26 in 63% as the Z-isomer (dr > 20:1). Conversely, the exposure of triene 24 to RCM conditions afforded bicyclic phosphates cis-27 as an inseparable mixture of Z/E-isomers (dr = 2:1 Z:E).25 These results were somewhat surprising, as the stereoselectivity of RCM to afford these C3-substituted bicyclo[8.3.1]phosphates was opposite the selectivity observed for the analogous bicyclo[7.3.1]phosphate systems (trans-11 and cis-12, Scheme 2, vide supra).
Scheme 5.
Synthesis of bicyclo[8.3.1]phosphates via formation of C7–C8-disubstituted olefins
The effect of olefin substitution on the success and stereochemical outcome of RCM to bicyclo[8.3.1]phosphates was next examined. In this regard, simple triene 28 was synthesized, in excellent yield, from the coupling of (R,R)-14 and 5-hexen-1-ol (Scheme 6). Exposure of 28 to G-II catalyst (7 mol % over 9 hours) provided the corresponding bicyclo[7.3.1]phosphate 29 with exclusive Z-selectivity (dr > 20:1, as observed by NMR). To examine the effect of C3-substitution in these systems, we next synthesized diastereomeric phosphate trienes 30 and 31 from the coupling of monochlorophosphate 14 with chiral, non-racemic alcohols (S)-25 and (R)-25, respectively. Treatment of phosphate trienes 30 and 31 with G-II catalyst provided the expected products (cis-32 and trans-33) in good yield and selectivity.
Scheme 6.
Synthesis of bicyclo[8.3.1]phosphates via formation of C7–C8-disubstituted olefins
X-ray crystallographic analysis of trans-33 confirmed the stereochemical result determined by NMR analysis (Figure 2). Interestingly, while the X-ray structures of every bicyclic phosphate obtained to date reveal an expected chair-conformation of the 6-membered ring of the bicyclic phosphate, the X-ray crystallographic analysis of trans-33 show a distorted boat geometry of the 6-membered ring, compensated for by a distorted tetrahedral geometry of the phosphate itself. In addition, the C3-substituent is directed away from the concave interior of the bicyclic phosphate. Taken collectively, this X-ray, in combination with experimental observation (gathered for both 10- and 11-membered ring formation, vide supra), suggest that C3-substitution could have a significant effect on large ring dynamics that influences the stereoselectivity of the RCM event.
Figure 2.
X-ray confirmation of olefin stereochemistry and revelation of distorted boat geometry in trans-33
Conclusion
In summary, phosphate tether-mediated RCM to provide a variety of P-stereogenic bicyclo[7.3.1]- and bicyclo[8.3.1]phosphates is reported. Unlike previous reports,12,16 the examples presented in this manuscript afford the corresponding bicyclic phosphates with predominantly Z-selectivity, though C3-substitution can affect the level of selectivity for disubstituted olefin formation. Olefin substitution (methyl) was well-tolerated, though higher catalyst loadings and extended reaction times were sometimes required for full conversion (6.2). Taken collectively, these results, in combination with those obtained in previous reports, suggest that C3-substitution, olefin substitution, the proximity of the forming olefin to the bridgehead carbon of resultant the bicyclic phosphate are all important factors in the success and stereochemical outcome of the RCM event. In addition, the bicyclic phosphates generated in this report represent an interesting class of macrocyclic phosphates with potential synthetic and biological utility as small molecule probes.
Experimental Section
All reactions were carried out in oven- or flame-dried glassware, under argon atmosphere, using standard gas-tight syringes, cannulae, and septa. Stirring was achieved with oven-dried magnetic stir bars. Toluene, THF, and CH2Cl2 were purified by passage through a purification system employing activated Al2O3.26 Et3N was purified by passage over basic alumina and stored over KOH. Butyllithium was purchased from Aldrich and titrated prior to use. All olefin metathesis catalysts were acquired from Materia and used without further purification. Flash column chromatography was performed with silica gel (40–63 μm), and thin layer chromatography was performed on silica gel 60F254 plates. 1H, 13C, DEPT, COSY, HSQC, HMBC and NOESY NMR spectra were recorded in CDCl3 (unless otherwise mentioned) at 500 MHz, and 125 MHz, respectively, calibrated to the solvent peak, and utilized to unambiguously assign proton and carbon signals. Correspondingly, 31P NMR spectra were recorded in CDCl3 at 162 MHz. High-resolution mass spectrometry (HRMS) was recorded using ESI (MeOH), and exact masses were observed for (2M+Na)+ ions.
General Procedure for Phosphate Triester Triene Generation (General Procedure 1)
To a solution of alcohol (1.1 equiv) in THF (0.2 M), at -40 °C under argon, was added n-BuLi (2.5 M in hexanes, 1 equiv), dropwise. The mixture was allowed to stir for 5 minutes, at which point a solution of phosphate monochloride (1.2 equiv) in THF (1 mL) was slowly added to the reaction vessel via cannulation. The mixture stirred at –40 °C for 2 hours (monitored by TLC) and was quenched with aqueous NH4Cl (sat.). The biphasic solution was separated, and the aqueous layer was extracted EtOAc (3 × 5 mL). The combined organic layers were washed with brine, dried (Na2SO4), and concentrated under reduced pressure. Purification via flash chromatography (silica, 0% to 50% EtOAc in Hexanes) provided triene-containing monocyclic phosphate triester product.
General procedure for RCM to provide bicyclo[7.3.1]- and bicyclo[8.3.1]phosphates (General Procedure 2)
To a flask containing monocyclic phosphate triester (1 mmol) in CH2Cl2 (dry, degassed, 0.001 M), equipped with an argon inlet and reflux condenser, was added p-benzoquinone (10 mol %). Then, G-I or G-II catalyst [see reaction schemes, vide supra] was added to the reaction [portion-wise in 1 mol % quantities over the allotted reaction time], and the reaction mixture was heated to reflux. Upon completion (monitored by TLC), the reaction was cooled to room temperature, where 5 drops of DMSO were added and the reaction was concentrated under reduced pressure. Purification via flash chromatography (silica, 0% to 60% or 75% EtOAc in Hexanes) provided the corresponding bicyclic phosphate.
(4R,6R)-2-(hex-5-yn-1-yloxy)-4,6-divinyl-1,3,2-dioxaphosphinane 2-oxide (2)
Following General Procedure 1, monochlorophosphate (R,R)-1 (0.4210 g, 2.018 mmol) and 5-hexyn-1-ol (0.1816 g, 1.850 mmol) were converted to phosphate triene 2 (0.3562 g, 1.318 mmol, 65%) which was isolated as a colorless oil. FTIR (neat): 3296, 3234, 2955, 2939, 2870, 1431, 1281, 1119, 1020, 993, 932, 874, 847, 725, 644, 548 cm−1; [α]D = –49.1 (c 1.96, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 6.00 (ddd, J = 16.9, 10.6, 6.0 Hz, 1H, H2C=CH-CHO(P)CH2), 5.89 (dddd, J = 17.3, 10.6, 5.2, 1.6 Hz, 1H, H2C=CH-CHO(P)CH2), 5.44 (ddd, J = 17.0, 1.2, 1.2 Hz, 1H, H-HC=CH-CHO(P)CH2), 5.36 (ddd, J = 17.2, 1.2, 1.2 Hz, 1H, H-HC=CH-CHO(P)CH2), 5.31–5.26 (m, 2H, H-HC=CH-CHO(P)CH2, H-HC=CH-CHO(P)CH2), 5.06–4.93 (m, 2H, H2C=CH-CHO(P)CH2, H2C=CH-CHO(P)CH2), 4.13–4.06 (m, 2H, (P)OCH2CH2CH2CH2CCH), 2.21 (td, J = 7.0, 2.7 Hz, 2H, (P)OCH2CH2CH2CH2CCH), 2.15 (dddd, J = 14.7, 8.2, 4.9, 1.6 Hz, 1H, H2C=CH-CHO(P)CHaHbCHO[P]), 2.03 (dddd, J = 14.8, 5.5, 3.6, 1.9 Hz, 1H, H2C=CH-CHO(P)CHaHbCHO[P]), 1.94 (t, J = 2.6 Hz, 1H, (P)OCH2CH2CH2CH2CCH), 1.79 (dt, J = 12.5, 6.4 Hz, 2H, (P)OCH2CH2CH2CH2CCH), 1.61 (p, J = 7.2 Hz, 2H, (P)OCH2CH2CH2CH2CCH); 13C NMR (126 MHz, CDCl3) δ ppm 134.86 (d, JCP = 10.9 Hz, CH), 134.85 (CH), 118.1 (CH2), 117.4 (CH2), 83.6 (C), 77.5 (d, JCP = 6.7 Hz, CH), 76.1 (d, JCP = 6.2 Hz, CH), 68.7 (CH), 67.2 (d, JCP = 5.9 Hz, CH2), 35.0 (d, JCP = 7.7 Hz, CH2), 29.1 (d, JCP = 6.9 Hz, CH2), 24.4 (CH2), 17.8 (CH2); 31P NMR (162 MHz, CDCl3) δ ppm –7.66 (dq, JPH = 13.9, 6.9 Hz); HRMS (TOF MS ES+) calcd for (C13H19O4P)2Na (2M+Na)+ 563.1940; found 563.1929.
(4S,6S)-2-((5-methylhex-5-en-1-yl)oxy)-4,6-divinyl-1,3,2-dioxaphosphinane 2-oxide (3)
Following General Procedure 1, monochlorophosphate (S,S)-1 (0.3380 g, 1.620 mmol) and 5-methyl-5-penten-1-ol (0.1700 g, 1.485 mmol) were converted to phosphate triene 3 (0.2634 g, 0.9200 mmol, 68%) which was isolated as a colorless oil. FTIR (neat): 2937, 2868, 1429, 1412, 1283, 1119, 1018, 926, 876, 849, 725, 646, 546 cm−1; [α]D = +51.7 (c 1.27, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 6.03 (ddd, J = 16.9, 10.6, 6.0 Hz, 1H, H2C=CH-CHO(P)CH2), 5.91 (ddddd, J = 17.3, 10.7, 5.3, 1.5, 1.5 Hz, 1H, H2C=CH-CHO(P)CH2), 5.46 (dd, J = 17.2, 1.3 Hz, 1H, H-HC=CH-CHO(P)CH2), 5.37 (dd, J = 17.2, 1.3 Hz, 1H, H-HC=CH-CHO(P)CH2), 5.31 (dd, J = 4.3, 1.2 Hz, 1H, H-HC=CH-CHO(P)CH2), 5.29 (dd, J = 4.3, 1.2 Hz, 1H, H-HC=CH-CHO(P)CH2), 5.07–5.01 (m, 1H, H2C=CH-CHO(P)CH2), 5.00–4.94 (m, 1H, H2C=CH-CHO(P)CH2), 4.71 (d, J = 2.1 Hz, 1H, (P)OCH2CH2CH2CH2C(CH3)=CHaHb), 4.67–4.66 (m, 1H, (P)OCH2CH2CH2CH2C(CH3)=CHaHb), 4.13–4.08 (m, 2H, (P)OCH2CH2CH2CH2C(CH3)=CH2), 2.21–2.13 (m, 1H, H2C=CH-CHO(P)CHaHbCHO[P]),), 2.07–2.01 (m, 3H, H2C=CH-CHO(P)CHaHbCHO[P], (P)OCH2CH2CH2CH2C(CH3)=CH2), 1.71–1.66 (m, 5H, (P)OCH2CH2CH2CH2C(CH3)=CH2, (P)OCH2CH2CH2CH2C(CH3)=CH2), 1.53 (dtd, J = 8.8, 7.3, 5.5 Hz, 2H, (P)OCH2CH2CH2CH2C(CH3)=CH2); 13C NMR (126 MHz, CDCl3) δ ppm 145.2 (C), 135.0 (d, JCP = 4.7 Hz, CH), 134.96 (CH), 118.0 (CH2), 117.4 (CH2), 110.2 (CH2), 77.5 (d, JCP = 6.8 Hz, CH), 76.1 (d, JCP = 6.0 Hz, CH), 67.8 (d, JCP = 6.0 Hz, CH2), 37.1 (CH2), 35.1 (d, JCP = 7.6 Hz, CH2), 29.8 (d, JCP = 6.8 Hz, CH2), 23.3 (CH2), 22.2 (CH3); 31P NMR (162 MHz, CDCl3) δ ppm –7.55 (dd, JPH = 14.2, 7.1 Hz); HRMS (TOF MS ES+) calcd for (C14H23O4P)2Na (2M+Na)+ 595.2566; found 595.2557.
(4R,6R)-2-((5-(triethoxysilyl)hex-5-en-1-yl)oxy)-4,6-divinyl-1,3,2-dioxaphosphinane 2-oxide (4)
To a clean, dry round bottom flask, equipped with a magnetic stirbar, was added 2 (0.0720 g, 0.2664 mmol) and CH2Cl2 (dry, degassed, 3 mL) under Ar. The flask was cooled to 0 °C, where (Cp*Ru[MeCN]3)PF6 (6.7 mg, 0.01332 mmol, 5 mol %) was added in two portions. The flask was removed from the cooling bath and warmed to room temperature. The reaction stirred at room temperature for 1 hour (monitored by TLC) and, upon completion, concentrated under reduced pressure. The crude mixture was separated via flash chromatography (silica, 0% to 50% EtOAc in Hexanes) to afford 4 (0.0791 g, 0.1820 mmol, 68%) as a colorless oil. 4 was stored in dry, degassed toluene under Ar in a flammable freezer for further use, as storage neat under Ar at lower temperatures resulted in decomposition after 2 weeks. FTIR (neat): 2974, 2925, 2895, 1285, 1101, 1078, 1020, 991, 960, 781, 752, 550 cm−1; [α]D = –28.6 (c 0.37, benzene); 1H NMR (500 MHz, CDCl3) δ ppm 6.01 (ddd, JPH = 16.7, 10.4, 5.9 Hz, 1H, H2C=CH-CHO(P)CH2), 5.89 (dddd, J = 17.2, 10.6, 5.2, 2.0 Hz, 1H, H2C=CH-CHO(P)CH2), 5.70 (dd, J = 3.6, 1.8 Hz, 1H, –C[Si(OEt)3]=CHaHb), 5.62 (d, J = 3.2 Hz, 1H–C[Si(OEt)3]=CHaHb), 5.45 (dd, J = 16.9, 1.9 Hz, 1H, H-HC=CH-CHO(P)CH2), 5.36 (dd, J = 17.2, 0.7 Hz, 1H, H-HC=CH-CHO(P)CH2), 5.31–5.26 (m, 2H, H-HC=CH-CHO(P)CH2, H-HC=CH-CHO(P)CH2), 5.05–4.99 (m, 1H, H2C=CH-CHO(P)CH2), 4.98–4.93 (m, 1H, H2C=CH-CHO(P)CH2), 4.14–4.04 (m, 2H, (P)OCH2CH2CH2CH2C[Si(OEt)3]=CH2), 3.80 (dtt, J = 9.3, 4.5, 2.2 Hz, 6H, –Si(OCH2CH3)3), 2.21–2.11 (m, 3H, H2C=CH-CHO(P)CHaHbCHO[P]), (P)OCH2CH2CH2CH2C[Si(OEt)3]=CH2), 2.06–2.00 (m, 1H, H2C=CH-CHO(P)CHaHbCHO[P]), 1.68 (q, J = 7.0, 6.4 Hz, 2H, (P)OCH2CH2CH2CH2C[Si(OEt)3]=CH2), 1.54 (tt, J = 9.6, 6.3 Hz, 2H, (P)OCH2CH2CH2CH2C[Si(OEt)3]=CH2), 1.21 (td, J = 7.0, 2.0 Hz, 9H, –Si(OCH2CH3)3).; 13C NMR (126 MHz, CDCl3) δ ppm 143.0 (C), 134.98 (d, JCP = 9.6 Hz, CH), 134.96 (CH), 129.4 (CH2), 118.0 (CH2), 117.4 (CH2), 77.5 (d, JCP = 6.8 Hz, CH), 76.0 (d, JCP = 6.0 Hz, CH), 67.8 (d, JCP = 6.0 Hz, CH2), 58.4 (3 x CH2), 35.3 (CH2), 35.1 (d, JCP = 7.7 Hz, CH2), 29.9 (d, JCP = 6.8 Hz, CH2), 24.5 (CH2), 18.1 (3 x CH3); 31P NMR (162 MHz, CDCl3) δ ppm –7.58 (dd, JPH = 13.9, 7.0 Hz); HRMS (TOF MS ES+) calcd for (C19H35O7PSi)2Na (2M+Na)+ 891.3677; found 891.3659.
(4S,6S)-4,6-diallyl-2-(((S)-1-(benzyloxy)hex-5-en-2-yl)oxy)-1,3,2-dioxaphosphinane 2-oxide (7)
Following General Procedure 1, monochlorophosphate (S,S)-9 (0.182 g, 0.769 mmol) and alcohol (S)-10 (0.145 g, 0.705 mmol) were converted into 7 (0.211 g, 0.519 mmol, 81%) which was isolated as a pale yellow oil. FTIR (neat): 2976, 2930, 2860, 1452, 1362, 1286, 1097, 997, 918, 739, 698, 629, 505 cm−1; [α]D = –34.4 (c 1.85, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 7.37–7.26 (m, 5H, Aromatic C-H), 5.86–5.69 (m, 3H, H2C=CH-CH2-CH2-CHO(P)CH2OBn, H2C=CH-CH2-CHO(P)CH2, H2C=CH-CH2-CHO(P)CH2), 5.16 (dd, J = 8.7, 2.1 Hz, 1H, HaHbC=CH-CH2CHO(P)CH2), 5.14–5.11 (m, 2H, HaHbC=CH-CH2CHO(P)CH2, HaHbC=CH-CH2CHO(P)CH2), 5.10 (dd, J = 3.2, 1.6 Hz, 1H, HaHbC=CH-CH2CHO(P)CH2), 5.05 (dq, J = 17.2, 1.5 Hz, 1H, HaHbC=CH-CH2-CH2-CHO(P)CH2OBn), 4.99 (dt, J = 10.2, 1.5 Hz, 1H, HaHbC=CH-CH2-CH2-CHO(P)CH2OBn), 4.66–4.48 (m, 5H, H2C=CHCH2-CHO(P)CH2, H2C=CHCH2-CHO(P)CH2, CHO(P)CH2OBn, OCH2Ph), 3.63–3.60 (m, 2H, CHO(P)CH2OBn), 2.66–2.52 (m, 2H, H2C=CH-CH2CHO(P)CH2, H2C=CH-CH2CHO(P)CH2), 2.39 (dt, J = 14.6, 7.4 Hz, 2H, H2C=CH-CH2CHO(P)CH2, H2C=CH-CH2CHO(P)CH2), 2.23–2.09 (m, 2H, H2C=CH-CH2-CH2-CHO(P)CH2OBn), 2.03–1.95 (m, 1H, H2C=CHCH2-CHO(P)CHaHbCHO[P]), 1.89 (dqd, J = 14.5, 3.8, 1.8 Hz, 1H, H2C=CHCH2-CHO(P)CHaHbCHO[P]), 1.85–1.79 (m, 2H, H2C=CH-CH2-CH2-CHO(P)CH2OBn); 13C NMR (126 MHz, CDCl3) δ ppm 138.0 (C), 137.4 (CH), 132.6 (CH), 132.3 (CH), 128.3 (2 x CH), 127.61 (2 x CH), 127.59 (CH), 118.9 (CH2), 118.7 (CH2), 115.2 (CH2), 77.8 (d, JCP = 6.3 Hz, CH), 76.8 (d, JCP = 7.9 Hz, CH), 75.5 (d, JCP = 6.6 Hz, CH), 73.2 (CH2), 71.6 (d, JCP = 4.0 Hz, CH2), 39.9 (d, JCP = 7.3 Hz, CH2), 39.1 (d, JCP = 3.9 Hz, CH2), 33.1 (d, JCP = 6.7 Hz, CH2), 31.4 (d, JCP = 5.0 Hz, CH2), 29.2 (CH2); 31P NMR (162 MHz, CDCl3) δ ppm –7.04 (dt, JPH = 13.7, 6.9 Hz); HRMS (TOF MS ES+) calcd for (C22H31O5P)2Na (2M+Na)+ 835.3716; found 835.3748.
(1R,3S,9S,11S,Z)-11-allyl-3-((benzyloxy)methyl)-2,12,13-trioxa-1-phosphabicyclo[7.3.1]tridec-6-ene 1-oxide (trans-11, Z-product [major])
Following General Procedure 2, triene 7 (40.0 mg, 0.0984 mmol) was exposed to G-I catalyst (6 mol %, over 7 hours) in refluxing CH2Cl2 to provide the corresponding bicyclic phosphates trans-11 (22.8 mg, 0.0603 mmol, 61% combined yield) as a 2:1 mixture of Z:E-stereoisomers. Purification by flash chromatography provided clean Z-product (16.0 mg, 0.0422 mmol, 43%) as a colorless oil. FTIR (neat): 2924, 1452, 1277, 1105, 1022, 976, 930, 737, 698 cm−1; [α]D = –40.5 (c 0.83, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 7.34 (d, J = 4.5 Hz, 4H, Aromatic C-H), 7.32–7.28 (m, 1H, Aromatic C-H), 5.80 (dddd, J = 17.2, 10.2, 7.9, 6.0 Hz, 1H, CH2(P)OCHCH2CH=CH2), 5.63–5.53 (m, 2H, CH2(P)OCHCH2CH=CHCH2), 5.22–5.14 (m, 2H, CH2(P)OCHCH2CH=CH2), 4.92–4.86 (m, 1H, CH2(P)OCHCH2CH=CHCH2), 4.66 (dtdd, J = 14.5, 7.2, 6.1, 3.7 Hz, 1H, CH2(P)OCHCH2CH=CH2), 4.58 (d, J = 11.9 Hz, 1H, –OCHaHbPh), 4.55 (d, J = 11.9 Hz, 1H, –OCHaHbPh), 4.24 (tdd, J = 9.1, 7.5, 4.5 Hz, 1H, -HC=CHCH2CH2CHO(P)CH2OBn), 3.66 (dd, J = 10.1, 5.7 Hz, 1H, -CHO(P)CHaHbOBn), 3.50 (dd, J = 10.1, 6.4 Hz, 1H, -CHO(P)CHaHbOBn), 2.80–2.70 (m, 3H, CH2(P)OCHCHaHbCH=CH2, CH2(P)OCHCHaHbCH=CHCHaHb-), 2.49 (dt, J = 14.8, 7.7 Hz, 1H, CH2(P)OCHCHaHbCH=CH2), 2.28 (d, J = 18.2 Hz, 1H, CH2(P)OCHCHaHbCH=CHCH2-), 2.16–2.05 (m, 2H, CH2(P)OCHCH2CH=CHCHaHbCH2, H2C=CHCH2-CHO(P)CHaHbCHO[P]), 1.96–1.87 (m, 1H, H2C=CHCH2-CHO(P)CHaHbCHO[P]), 1.87–1.80 (m, 2H, CH2(P)OCHCH2CH=CHCH2CH2CHO[P]); 13C NMR (126 MHz, CDCl3) δ ppm 138.0 (C), 135.7 (CH), 132.3 (CH), 128.3 (2 x CH), 127.66 (2 x CH), 127.62 (CH), 123.8 (CH), 119.0 (CH2), 78.0 (d, JCP = 6.3 Hz, CH), 76.5 (d, JCP = 8.3 Hz, 2 x CH), 73.5 (CH2), 72.7 (d, JCP = 5.9 Hz, CH2), 39.0 (d, JCP = 3.5 Hz, CH2), 32.1 (d, JCP = 4.5 Hz, CH2), 31.3 (d, JCP = 3.0 Hz, CH2), 31.1 (CH2), 23.8 (CH2); 31P NMR (162 MHz, CDCl3) δ ppm –14.14 (dt, JPH = 13.9, 7.0 Hz); HRMS (TOF MS ES+) calcd for (C20H27O5P)2Na (2M+Na)+ 779.3090; found 779.3094.
(1S,3S,9R,11R,E)-11-allyl-3-((benzyloxy)methyl)-2,12,13-trioxa-1-phosphabicyclo[7.3.1]tridec-6-ene 1-oxide (trans-11, E-product [minor])
Following General Procedure 2, triene 7 (40.0 mg, 0.0984 mmol) was exposed to G-I catalyst (6 mol %, over 7 hours) in refluxing CH2Cl2 to provide the corresponding bicyclic phosphates trans-11 (22.8 mg, 0.0603 mmol, 61% combined yield) as a 2:1 mixture of Z:E-stereoisomers. Purification by flash chromatography provided clean E-product (6.8 mg, 0.0180 mmol, 18%) as a colorless oil. FTIR (neat): 2924, 2853, 1452, 1364, 1273, 1097, 1024, 982, 932, 698 cm−1; [α]D = –77.6 (c 0.21, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 7.35 (d, J = 4.4 Hz, 4H, Aromatic C-H), 7.32–7.28 (m, 1H, Aromatic C-H), 5.87–5.71 (m, 2H, CH2(P)OCHCH2CH=CH2, CH2(P)OCHCH2CH=CHCH2), 5.50 (dddd, J = 15.3, 10.1, 5.5, 1.7 Hz, 1H, CH2(P)OCHCH2CH=CHCH2), 5.24–5.15 (m, 2H, CH2(P)OCHCH2CH=CH2), 4.94–4.84 (m, 1H, CH2(P)OCHCH2CH=CHCH2), 4.69–4.51 (m, 3H, CH2(P)OCHCH2CH=CH2, -OCH2Ph), 4.14 (dddd, J = 13.8, 9.8, 6.5, 1.1 Hz, 1H, -HC=CHCH2CH2CHO(P)CH2OBn), 3.66 (dd, J = 9.7, 5.3 Hz, 1H, -HC=CHCH2CH2CHO(P)CHaHbOBn), 3.46 (dd, J = 9.7, 7.4 Hz, 1H, HC=CHCH2CH2CHO(P)CHaHbOBn), 2.72 (dddt, J = 14.5, 7.4, 6.1, 1.5 Hz, 1H, CH2(P)OCHCHaHbCH=CH2), 2.63 (ddd, J = 14.5, 10.1, 4.9 Hz, 1H, CH2(P)OCHCHaHbCH=CHCH2), 2.52–2.43 (m, 2H, CH2(P)OCHCHaHbCH=CHCH2, CH2(P)OCHCH2CH=CHCHaHbCH2), 2.17–2.11 (m, 1H, CH2(P)OCHCHaHbCH=CHCH2), 2.10–1.92 (m, 4H, CH2(P)OCHCH2CH=CHCHaHbCHaHbCHO[P]CH2OBn, H2C=CHCH2-CHO(P)CH2CHO[P]), 1.75 (dddd, J = 15.1, 13.0, 8.5, 4.5 Hz, 1H, CH2(P)OCHCH2CH=CHCH2CHaHbCHO[P]CH2OBn); 13C NMR (126 MHz, CDCl3) δ ppm 138.8 (CH), 138.0 (C), 132.2 (CH), 128.4 (2 x CH), 127.8 (2 x CH), 127.7 (CH), 122.1 (CH), 119.1 (CH2), 78.8 (d, JCP = 9.5 Hz, CH), 78.0 (d, JCP = 6.4 Hz, CH), 77.8 (d, JCP = 9.5 Hz, CH), 73.6 (CH2), 72.6 (d, JCP = 1.7 Hz, CH2), 39.3 (d, JCP = 3.7 Hz, CH2), 36.8 (CH2), 32.0 (CH2), 31.9 (d, JCP = 9.5 Hz, CH2), 31.0 (d, JCP = 7.5 Hz, CH2); 31P NMR (162 MHz, CDCl3) δ ppm –15.94 (d, JPH = 12.8 Hz); HRMS (TOF MS ES+) calcd for (C20H27O5P)2Na (2M+Na)+ 779.3090; found 779.3069.
(4S,6S)-4,6-diallyl-2-(((R)-1-(benzyloxy)hex-5-en-2-yl)oxy)-1,3,2-dioxaphosphinane 2-oxide (8)
Following General Procedure 1, monochlorophosphate (S,S)-9 (0.182 g, 0.769 mmol) and alcohol (R)-10 (0.145 g, 0.705 mmol) were converted into 8 (0.175 g, 0.430 mmol, 67%) which was isolated as a colorless oil. FTIR (neat): 2930, 2862, 1452, 1366, 1286, 1097, 1078, 997, 976, 916, 816, 739, 629, 550 cm−1; [α]D = –38.2 (c 0.60, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 7.38–7.28 (m, 5H, Aromatic C-H), 5.86–5.63 (m, 3H, H2C=CH-CH2-CH2-CHO(P)CH2OBn, H2C=CH-CH2-CH2-CHO(P)CH2OBn), 5.15–5.10 (m, 3H, H2C=CH-CH2CHO(P)CH2, HaHbC=CH-CH2CHO(P)CH2), 5.08 (d, J = 1.4 Hz, 1H, HaHbC=CH-CH2CHO(P)CH2), 5.04 (dq, J = 17.1, 1.7 Hz, 1H, HaHbC=CH-CH2-CH2-CHO(P)CH2OBn), 4.98 (dq, J = 10.2, 1.4 Hz, 1H, HaHbC=CH-CH2-CH2-CHO(P)CH2OBn), 4.66–4.47 (m, 5H, H2C=CHCH2-CHO(P)CH2, H2C=CHCH2-CHO(P)CH2, CHO(P)CH2OBn, OCH2Ph), 3.66–3.53 (m, 2H, CHO(P)CH2OBn), 2.68 (dddt, J = 13.5, 7.5, 6.3, 1.5 Hz, 1H, H2C=CH-CHaHbCHO(P)CH2), 2.49–2.43 (m, 1H, H2C=CH-CHaHbCHO(P)CH2), 2.43–2.35 (m, 1H, H2C=CH-CHaHbCHO(P)CH2), 2.34–2.27 (m, 1H, H2C=CH-CHaHbCHO(P)CH2), 2.17 (tddd, J = 7.8, 6.5, 5.1, 1.4 Hz, 2H, H2C=CH-CH2-CH2-CHO(P)CH2OBn), 1.97 (dddd, J = 14.8, 8.6, 5.0, 1.4 Hz, 1H, H2C=CHCH2-CHO(P)CHaHbCHO[P]), 1.91–1.76 (m, 3H, H2C=CHCH2-CHO(P)CHaHbCHO[P], H2C=CH-CH2-CH2-CHO(P)CH2OBn); 13C NMR (126 MHz, CDCl3) δ ppm 137.9 (C), 137.5 (CH), 132.8 (CH), 132.3 (CH), 128.4 (2 x CH), 127.7 (CH), 127.6 (2 x CH), 118.7 (CH2), 118.6 (CH2), 115.2 (CH2), 77.4 (d, JCP = 2.4 Hz, CH), 77.4 (d, JCP = 3.3 Hz, CH), 75.0 (d, JCP = 6.7 Hz, CH), 73.2 (CH2), 71.8 (d, JCP = 4.2 Hz, CH2), 40.0 (d, JCP = 7.8 Hz, CH2), 38.9 (d, JCP = 3.2 Hz, CH2), 33.0 (d, JCP = 6.8 Hz, CH2), 31.5 (d, JCP = 4.9 Hz, CH2), 29.2 (CH2); 31P NMR (162 MHz, CDCl3) δ ppm –7.18 (s); HRMS (TOF MS ES+) calcd for (C22H31O5P)2Na (2M+Na)+ 835.3716; found 835.3719.
(1R,3R,9S,11S,Z)-11-allyl-3-((benzyloxy)methyl)-2,12,13-trioxa-1-phosphabicyclo[7.3.1]tridec-6-ene 1-oxide (cis-12)
Following General Procedure 2, triene 8 (30.0 mg, 0.0738 mmol) was exposed to G-I catalyst (3 mol %, over 4 hours) in refluxing CH2Cl2 to provide the corresponding bicyclic phosphate cis-12 (24.6 mg, 0.0650 mmol, 88%) as a colorless oil. FTIR (neat): 2924, 2858, 1454, 1364, 1285, 1096, 978, 735, 698, 559 cm−1; [α]D = –14.9 (c 1.01, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 7.33 (d, J = 4.4 Hz, 4H, Aromatic C-H), 7.30–7.25 (m, 1H, Aromatic C-H), 5.83 (ddt, J = 16.6, 10.8, 7.1 Hz, 1H, CH2(P)OCHCH2CH=CH2), 5.63–5.53 (m, 2H, CH2(P)OCHCH2CH=CHCH2), 5.18 (dd, J = 6.8, 1.5 Hz, 1H, CH2(P)OCHCH2CH=CHaHb), 5.15 (d, J = 1.4 Hz, 1H, CH2(P)OCHCH2CH=CHaHb), 4.79 (dtd, J = 12.1, 6.2, 1.9 Hz, 1H, CH2(P)OCHCH2CH=CHCH2), 4.72–4.60 (m, 2H, CH2(P)OCHCH2CH=CH2, –OCHaHbPh), 4.52 (d, J = 12.0 Hz, 1H, –OCHaHbPh), 4.37 (dtt, J = 12.0, 5.9, 2.9 Hz, 1H, -HC=CHCH2CH2CHO(P)CH2OBn), 3.94 (dd, J = 10.2, 5.7 Hz, 1H, -CHO(P)CHaHbOBn), 3.79 (dd, J = 10.1, 2.7 Hz, 1H, -CHO(P)CHaHbOBn), 3.23 (q, J = 12.5, 11.7 Hz, 1H, CH2(P)OCHCHaHbCH=CHCH2), 2.62 (tq, J = 13.1, 10.8, 4.9 Hz, 1H, CH2(P)OCHCH2CH=CHCHaHbCH2), 2.53 (dtd, J = 14.2, 6.4, 1.5 Hz, 1H, CH2(P)OCHCHaHbCH=CH2), 2.45–2.35 (m, 1H, CH2(P)OCHCHaHbCH=CH2), 2.25 (ddd, J = 14.6, 11.9, 5.6 Hz, 1H, H2C=CHCH2-CHO(P)CHaHbCHO[P]), 2.09 (dddd, J = 14.6, 11.8, 4.8, 2.8 Hz, 1H, CH2(P)OCHCH2CH=CHCH2CHaHbCHO[P]), 2.01 (d, J = 13.1 Hz, 1H, CH2(P)OCHCH2CH=CHCHaHbCH2), 1.94 (d, J = 14.0 Hz, 1H, CH2(P)OCHCHaHbCH=CHCH2), 1.80–1.74 (m, 1H, CH2(P)OCHCH2CH=CHCH2CHaHbCHO[P]), 1.70 (dq, J = 14.7, 1.9 Hz, 1H, H2C=CHCH2-CHO(P)CHaHbCHO[P]); 13C NMR (126 MHz, CDCl3) δ ppm 138.3 (C), 131.9 (CH), 130.2 (CH), 128.3 (2 x CH), 127.7 (2 x CH), 127.5 (CH), 126.0 (CH), 119.1 (CH2), 76.9 (d, JCP = 7.7 Hz, CH), 75.7 (d, JCP = 7.3 Hz, CH), 74.7 (d, JCP = 2.7 Hz, CH), 73.5 (CH2), 71.9 (CH2), 40.6 (d, JCP = 9.0 Hz, CH2), 34.3 (d, JCP = 6.9 Hz, CH2), 31.5 (CH2), 29.8 (d, JCP = 9.6 Hz, CH2), 21.9 (CH2); 31P NMR (162 MHz, CDCl3) δ ppm –9.76 (d, JPH = 23.5 Hz); HRMS (TOF MS ES+) calcd for (C20H27O5P)2Na (2M+Na)+ 779.3090; found 779.3095.
(4R,6R)-4,6-bis(2-methylallyl)-2-(pent-4-en-1-yloxy)-1,3,2-dioxaphosphinane 2-oxide (13)
Following General Procedure 1, (R,R)-monochlorophosphate 14 (0.145 g, 0.548 mmol) and 4-penten-1-ol (43.2 mg, 0.502 mmol) were converted to phosphate triene 13 (0.115 g, 0.366 mmol, 80%) which was isolated as a colorless oil. FTIR (neat): 2966, 2939, 1445, 1377, 1288.4, 1011, 970, 893, 814, 799, 542 cm−1; [α]D = +44.1 (c 0.88, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 5.80 (ddt, J = 16.9, 10.2, 6.6 Hz, 1H, (P)OCH2CH2CH2CH=CH2), 5.05 (dq, J = 17.2, 1.7 Hz, 1H, (P)OCH2CH2CH2CH=CHaHb), 5.00 (dq, J = 10.2, 1.5 Hz, 1H, (P)OCH2CH2CH2CH=CHaHb), 4.87 (q, J = 1.5 Hz, 2H, HaHbC=C(CH3)-CH2CHO(P)CH2, HaHbC=C(CH3)-CH2CHO(P)CH2), 4.78 (d, J = 5.94 Hz, 2H, HaHbC=C(CH3)-CH2CHO(P)CH2, HaHbC=C(CH3)-CH2CHO(P)CH2), 4.77–4.71 (m, 1H, H2C=C(CH3)-CH2CHO(P)CH2), 4.63 (dtdd, J = 8.7, 6.8, 5.2, 3.6 Hz, 1H, H2C=C(CH3)-CH2CHO(P)CH2), 4.10 (q, J = 6.7 Hz, 2H, (P)OCH2CH2CH2CH=CH2), 2.62 (dd, J = 14.2, 7.0 Hz, 1H, H2C=C(CH3)-CHaHbCHO(P)CH2), 2.56 (dd, J = 14.3, 6.9 Hz, 1H, H2C=C(CH3)-CHaHbCHO(P)CH2), 2.41–2.30 (m, 2H, H2C=C(CH3)-CHaHbCHO(P)CH2, H2C=C(CH3)-CHaHbCHO(P)CH2), 2.16 (dt, J = 8.0, 6.7 Hz, 2H, (P)OCH2CH2CH2CH=CH2), 2.01 (dddd, J = 14.8, 8.5, 5.0, 1.4 Hz, 1H, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P]), 1.87 (dddd, J = 14.7, 5.1, 3.5, 1.8 Hz, 1H, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P]), 1.83–1.78 (m, 2H, (P)OCH2CH2CH2CH=CH2), 1.76 (dd, J = 5.3, 1.1 Hz, 6H, H2C=C(CH3)-CH2CHO(P)CH2); 13C NMR (126 MHz, CDCl3) δ ppm 140.3 (C), 140.2 (C), 137.2 (CH), 115.4 (CH2), 114.2 (2 x CH2), 75.8 (d, JCP = 6.8 Hz, CH), 74.5 (d, JCP = 6.5 Hz, CH), 67.2 (d, JCP = 5.9 Hz, CH2), 43.7 (d, JCP = 7.2 Hz, CH2), 42.7 (d, JCP = 3.2 Hz, CH2), 33.4 (d, JCP = 6.9 Hz, CH2), 29.6 (CH2), 29.5 (d, JCP = 6.7 Hz, CH2), 22.6 (CH3), 22.3 (CH3); 31P NMR (162 MHz, CDCl3) δ ppm –6.71 (d, JPH = 7.2 Hz); HRMS (TOF MS ES+) calcd for (C16H27O4P)2Na (2M+Na)+ 651.3192; found 651.3194.
(1S,9R,11R,Z)-7-methyl-11-(2-methylallyl)-2,12,13-trioxa-1-phosphabicyclo[7.3.1]tridec-6-ene 1-oxide (15)
Following General Procedure 2, triene 13 (20.0 mg, 0.0636 mmol) was exposed to G-II catalyst (2 mol %, over 2 hours) in refluxing CH2Cl2 to provide the corresponding bicyclic phosphate 15 (15.8 mg, 0.0552 mmol, 87%) as a white, crystalline solid, which was recrystallized via vapor-diffusion method (EtOAc:Hexanes) to obtain X-ray quality crystals for X-ray crystallographic analysis (which confirmed stereochemical olefin assignment as Z). FTIR (neat): 2964, 2926, 2860, 1435, 1379, 1279, 1221, 1101, 1086, 1069, 1024, 984, 953, 914, 843, 791, 764, 546 cm−1; [α]D = –18.7 (c 0.70, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 5.25 (ddt, J = 10.6, 3.4, 1.6 Hz, 1H, CH2(P)OCHCH2C(CH3)=CHCH2), 4.91–4.79 (m, 4H, H2C=C(CH3)CH2-CHO(P)CH2, H2C=C(CH3)CH2-CHO(P)CH2CHO[P]), 4.04 (dt, J = 7.9, 1.8 Hz, 2H, (P)OCH2CH2CH2CH=C(CH3)CH2), 3.35 (t, J = 13.5 Hz, 1H, CH2(P)OCHCHaHbC(CH3)=CHCH2), 2.57–2.44 (m, 2H, H2C=C(CH3)CHaHb-CHO(P)CH2, CH2(P)OCHCH2C(CH3)=CHCHaHbCH2), 2.30 (ddd, J = 14.3, 6.4, 2.8 Hz, 1H, H2C=C(CH3)CHaHb-CHO(P)CH2), 2.22 (ddd, J = 14.6, 11.8, 5.6 Hz, 1H, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P]), 1.99 (ddt, J = 14.2, 9.8, 4.3 Hz, 2H, CH2(P)OCHCH2C(CH3)=CHCHaHbCH2, (P)OCH2CHaHbCH2CH=C(CH3)CH2), 1.80 (s, 3H, CH2(P)OCHCH2C(CH3)=CHCH2), 1.79 (dd, J = 1.1, 1.1 Hz, 3H, H2C=C(CH3)CH2-CHO(P)CH2), 1.75 (dd, J = 14.1, 2.3 Hz, 1H, CH2(P)OCHCHaHbC(CH3)=CHCH2), 1.70 (d, J = 14.7 Hz, 1H, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P]), 1.51 (dddd, J = 17.2, 10.4, 5.4, 3.2 Hz, 1H, (P)OCH2CHaHbCH2CH=C(CH3)CH2); 13C NMR (126 MHz, CDCl3) δ ppm 140.0 (C), 132.8 (C), 125.0 (CH), 114.3 (CH2), 75.2 (d, JCP = 7.5 Hz, CH), 74.8 (d, JCP = 7.0 Hz, CH), 64.2 (d, JCP = 5.0 Hz, CH2), 44.4 (d, JCP = 9.1 Hz, CH2), 34.9 (CH2), 34.7 (d, JCP = 6.7 Hz, CH2), 26.8 (d, JCP = 10.1 Hz, CH2), 22.9 (CH3), 22.5 (CH2), 22.0 (CH3); 31P NMR (162 MHz, CDCl3) δ ppm –8.16 (d, JPH = 23.5 Hz); HRMS (TOF MS ES+) calcd for (C14H23O4P)2Na (2M+Na)+ 595.2566; found 595.2538.
(4R,6R)-2-(((S)-1-(benzyloxy)hex-5-en-2-yl)oxy)-4,6-bis(2-methylallyl)-1,3,2-dioxaphosphinane 2-oxide (16)
Following General Procedure 1, monochlorophosphate (R,R)-14 (0.180 g, 0.680 mmol) and alcohol (S)-10 (0.129 g, 0.623 mmol) were converted into phosphate triene 16 (0.194 g, 0.447 mmol, 79%) which was isolated as a colorless oil. FTIR (neat): 2964, 2936, 2860, 1454, 1377, 1367, 1288, 1101, 1072, 990, 895, 814, 737, 698, 552 cm−1; [α]D = +36.5 (c 1.97, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 7.36–7.27 (m, 5H, Aromatic C-H), 5.80 (ddt, J = 16.9, 10.2, 6.6 Hz, 1H, H2C=CH-CH2-CH2-CHO(P)CH2OBn), 5.03 (dd, J = 17.2, 1.7 Hz, 1H, HaHbC=CH-CH2-CH2-CHO(P)CH2OBn), 4.97 (dd, J = 10.2, 1.7 Hz, 1H, HaHbC=CH-CH2-CH2-CHO(P)CH2OBn), 4.85 (d, J = 1.7 Hz, 1H, HaHbC=C(CH3)-CH2CHO(P)CH2), 4.82 (dd, J = 1.7, 1.7 Hz, 1H, HaHbC=C(CH3)-CH2CHO(P)CH2), 4.76–4.74 (m, 2H, HaHbC=C(CH3)-CH2CHO(P)CH2, HaHbC=C(CH3)-CH2CHO(P)CH2), 4.73–4.65 (m, 2H, H2C=C(CH3)-CH2CHO(P)CH2, H2C=C(CH3)-CH2CHO(P)CH2), 4.63–4.51 (m, 3H, H2C=CH-CH2-CH2-CHO(P)CH2OBn, OCH2Ph), 3.62–3.55 (m, 2H, H2C=CH-CH2-CH2-CHO(P)CH2OBn), 2.67–2.61 (m, 1H, H2C=C(CH3)-CHaHbCHO(P)CH2), 2.49 (dd, J = 14.1, 6.5 Hz, 1H, H2C=C(CH3)-CHaHbCHO(P)CH2), 2.36 (dd, J = 14.2, 7.4 Hz, 1H, H2C=C(CH3)-CHaHbCHO(P)CH2), 2.29 (ddt, J = 14.1, 7.3, 1.2 Hz, 1H, H2C=C(CH3)-CHaHbCHO(P)CH2), 2.16 (dddd, J = 15.6, 9.1, 7.1, 4.0 Hz, 2H, H2C=CH-CH2-CH2-CHO(P)CH2OBn), 1.96 (dddd, J = 14.5, 8.2, 4.9, 1.4 Hz, 1H, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P]), 1.90–1.76 (m, 3H, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P], H2C=CH-CH2-CH2-CHO(P)CH2OBn), 1.74 (t, J = 1.0 Hz, 3H, H2C=C(CH3)-CH2CHO(P)CH2), 1.69 (t, J = 1.1 Hz, 3H, H2C=C(CH3)-CH2CHO(P)CH2); 13C NMR (126 MHz, CDCl3) δ ppm 140.4 (C), 140.3 (C), 137.8(C), 137.5 (CH), 128.3 (2 x CH), 127.7 (CH), 127.6 (2 x CH), 115.1 (CH2), 114.1 (2 x CH2), 77.4 (d, JCP = 6.2 Hz, CH), 76.0 (d, JCP = 6.8 Hz, CH), 74.3 (d, JCP = 6.6 Hz, CH), 73.1 (CH2), 71.7 (d, JCP = 4.2 Hz, CH2), 43.7 (d, JCP = 7.4 Hz, CH2), 42.8 (d, JCP = 3.5 Hz, CH2), 33.3 (d, JCP = 6.9 Hz, CH2), 31.4 (d, JCP = 4.7 Hz, CH2), 29.1 (CH2), 22.6 (CH3), 22.3 (CH3); 31P NMR (162 MHz, CDCl3) δ ppm –7.13 (m);27 HRMS (TOF MS ES+) calcd for (C24H35O5P)2Na (2M+Na)+ 891.4342; found 891.4326.
(1S,3S,9R,11R,Z)-3-((benzyloxy)methyl)-7-methyl-11-(2-methylallyl)-2,12,13-trioxa-1-phosphabicyclo[7.3.1]tridec-6-ene 1-oxide (cis-18)
Following General Procedure 2, triene 16 (30.0 mg, 0.0690 mmol) was exposed to G-II catalyst (2 mol %, over 2 hours) in refluxing CH2Cl2 to provide the corresponding bicyclic phosphate cis-18 (22.6 mg, 0.0556 mmol, 81%) as a colorless oil. FTIR (neat): 2966, 2920, 1454, 1377, 1285, 1099, 1072, 980, 916, 793, 698, 546 cm−1; [α]D = –4.6 (c 0.67, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 7.34 (d, J = 4.5 Hz, 4H, Aromatic C-H), 7.30–7.25 (m, 1H, Aromatic C-H), 5.28 (ddt, J = 10.6, 3.2, 1.5 Hz, 1H, CH2(P)OCHCH2C(CH3)=CHCH2), 4.93–4.78 (m, 4H, H2C=C(CH3)CH2-CHO(P)CH2, H2C=C(CH3)CH2-CHO(P)CH2CHO[P]), 4.62 (d, J = 12.0 Hz, 1H, -OCHaHbPh), 4.53 (d, J = 12.0 Hz, 1H, -OCHaHbPh), 4.36 (dtt, J = 12.0, 5.9, 2.8 Hz, 1H, CH2(CH3)C=CHCH2CH2CHO(P)CH2OBn), 3.92 (dd, J = 10.2, 5.8 Hz, 1H, CH2CHO(P)CHaHbOBn), 3.80 (dd, J = 10.1, 2.7 Hz, 1H, CH2CHO(P)CHaHbOBn), 3.32 (t, J = 13.5 Hz, 1H, CH2(P)OCHCHaHbC(CH3)=CHCH2), 2.52 (tdd, J = 16.9, 9.3, 4.4 Hz, 2H, CH2(CH3)C=CHCHaHbCH2CHO(P)CH2OBn, H2C=C(CH3)CHaHb-CHO(P)CH2), 2.32 (ddd, J = 14.3, 7.1, 2.2 Hz, 1H, H2C=C(CH3)CHaHb-CHO(P)CH2), 2.23 (ddd, J = 14.5, 11.8, 5.8 Hz, 1H, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P]), 2.09–1.97 (m, 2H, CH2(CH3)C=CHCHaHbCHaHbCHO(P)CH2OBn), 1.79 (d, J = 1.4 Hz, 6H), 1.72 (ddt, J = 16.9, 14.7, 2.4 Hz, 3H, CH2(P)OCHCHaHbC(CH3)=CHCH2, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P], CH2(CH3)C=CHCH2CHaHbCHO(P)CH2OBn); 13C NMR (126 MHz, CDCl3) δ ppm 140.0 (C), 138.3 (C), 131.9 (C), 128.3 (2 x CH), 127.7 (2 x CH), 127.5 (CH), 125.9 (CH), 114.4 (CH2), 75.0 (d, J = 7.8 Hz, CH), 74.6 (d, J = 5.7 Hz, CH), 74.6 (CH), 73.5 (CH2), 72.0 (CH2), 44.3 (d, J = 9.1 Hz, CH2), 35.1 (CH2), 34.5 (d, J = 6.8 Hz, CH2), 30.2 (d, J = 9.7 Hz, CH2), 22.9 (CH3), 22.7 (CH2), 22.0 (CH3); 31P NMR (162 MHz, CDCl3) δ ppm –9.52 (d, JPH = 23.3 Hz); HRMS (TOF MS ES+) calcd for (C22H31O5P)2Na (2M+Na)+ 835.3716; found 835.3714.
(4R,6R)-2-(((R)-1-(benzyloxy)hex-5-en-2-yl)oxy)-4,6-bis(2-methylallyl)-1,3,2-dioxaphosphinane 2-oxide (17)
Following General Procedure 1, monochlorophosphate (R,R)-14 (0.180 g, 0.680 mmol) and alcohol (R)-10 (0.129 g, 0.623 mmol) were converted into phosphate triene 17 (0.201 g, 0.463 mmol, 82%) which was isolated as a pale yellow oil. FTIR (neat): 2964, 2936, 2860, 1452, 1375, 1366, 1285, 1099, 1070, 999, 899, 810, 737, 698, 548 cm−1; [α]D = +31.7 (c 1.29, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 7.34 (d, J = 4.4 Hz, 4H, aromatic C-H), 7.29 (dd, J = 4.9, 3.8 Hz, 1H, aromatic C-H), 5.81 (ddt, J = 16.9, 10.2, 6.5 Hz, 1H, H2C=CH-CH2-CH2-CHO(P)CH2OBn), 5.04 (dq, J = 17.1, 1.7 Hz, 1H, HaHbC=CH-CH2-CH2-CHO(P)CH2OBn), 4.99 (dd, J = 10.2, 1.6 Hz, 1H, HaHbC=CH-CH2-CH2-CHO(P)CH2OBn), 4.85 (dt, J = 13.5, 1.7 Hz, 2H, H2C=C(CH3)-CH2CHO(P)CH2), 4.79–4.74 (m, 2H, H2C=C(CH3)-CH2CHO(P)CH2), 4.74–4.61 (m, 3H, H2C=C(CH3)-CH2CHO(P)CH2, H2C=C(CH3)-CH2CHO(P)CH2, H2C=CH-CH2-CH2-CHO(P)CH2OBn), 4.59 (d, J = 12.0 Hz, 1H, OCHaHbPh), 4.53 (d, J = 12.0 Hz, 1H, OCHaHbPh), 3.61 (dd, J = 4.8, 2.6 Hz, 2H, H2C=CH-CH2-CH2-CHO(P)CH2OBn), 2.57 (td, J = 14.5, 6.8 Hz, 2H, H2C=C(CH3)-CHaHbCHO(P)CH2, H2C=C(CH3)-CHaHbCHO(P)CH2), 2.40–2.30 (m, 2H, H2C=C(CH3)-CHaHbCHO(P)CH2, H2C=C(CH3)-CHaHbCHO(P)CH2), 2.24–2.08 (m, 2H, H2C=CH-CH2-CH2-CHO(P)CH2OBn), 1.98 (dddd, J = 14.3, 8.0, 4.8, 1.4 Hz, 1H, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P],), 1.89 (dddd, J = 14.7, 5.7, 4.0, 1.7 Hz, 1H, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P],), 1.82 (dddd, J = 8.8, 7.4, 6.2, 1.6 Hz, 2H, H2C=CH-CH2-CH2-CHO(P)CH2OBn), 1.75 (d, J = 1.1 Hz, 3H, H2C=C(CH3)-CH2CHO(P)CH2), 1.72 (d, J = 1.1 Hz, 3H, H2C=C(CH3)-CH2CHO(P)CH2); 13C NMR (126 MHz, CDCl3) δ ppm 140.4 (C), 140.2 (C), 138.0 (C), 137.4 (CH), 128.3 (2 x CH), 127.59 (2 x CH), 127.57 (CH), 115.2 (CH2), 114.24 (CH2), 114.15 (CH2), 77.7 (d, JCP = 6.3 Hz, CH), 75.6 (d, JCP = 6.7 Hz, CH), 74.6 (d, JCP = 6.5 Hz, CH), 73.2 (CH2), 71.6 (d, JCP = 3.9 Hz, CH2), 43.7 (d, JCP = 7.1 Hz, CH2), 43.0 (d, JCP = 4.1 Hz, CH2), 33.3 (d, JCP = 6.7 Hz, CH2), 31.4 (d, JCP = 5.1 Hz, CH2), 29.2 (CH2), 22.6 (CH3), 22.3 (CH3); 31P NMR (162 MHz, CDCl3) δ ppm –6.95 (m); HRMS (TOF MS ES+) calcd for (C24H35O5P)2Na (2M+Na)+ 891.4342; found 891.4314.
(1S,3R,9R,11R,Z)-3-((benzyloxy)methyl)-7-methyl-11-(2-methylallyl)-2,12,13-trioxa-1-phosphabicyclo[7.3.1]tridec-6-ene 1-oxide (trans-19)
Following General Procedure 2, triene 17 (30.0 mg, 0.0690 mmol) was exposed to G-II catalyst (2 mol %, over 2 hours) in refluxing CH2Cl2 to provide the corresponding bicyclic phosphate trans-19 (24.5 mg, 0.0603 mmol, 87%) as a pale yellow oil. Full characterization in CDCl3 and DMSO-d6 at ambient temperature—even after extended scan times—provided incomplete information for 13C and carbon-related 2D NMR. As such, full characterization of trans-19 required elevated temperatures (95 °C) to fully identify short, broad carbon signals and correlate these 13C signals to 1H signals (HSQC, HMBC). FTIR (neat): 2928, 2855, 1450, 1377, 1281, 1103, 1020, 972, 957, 907, 791, 745, 698 cm−1; [α]D = +57.5 (c 0.36, CHCl3); 1H NMR (500 MHz, 95 °C, DMSO-d6) δ ppm 7.37–7.26 (m, 5H, aromatic C-H), 5.33 (t, J = 8.2 Hz, 1H, CH2(P)OCHCH2C(CH3)=CHCH2), 4.96–4.78 (m, 4H, H2C=C(CH3)CH2-CHO(P)CH2, H2C=C(CH3)CH2-CHO(P)CH2CHO[P]), 4.51 (s, 2H, -OCH2Ph), 4.24 (q, J = 8.8, 5.9 Hz, 1H, CH2(CH3)C=CHCH2CH2CHO(P)CH2OBn), 3.61 (dd, J = 10.3, 5.8 Hz, 1H, CH2CHO(P)CHaHbOBn), 3.48 (dd, J = 10.4, 5.7 Hz, 1H, CH2CHO(P)CHaHbOBn), 2.60–2.52 (s, 1H, CH2(P)OCHCHaHbC(CH3)=CHCH2), 2.49–2.34 (m, 4H, CH2(P)OCHCHaHbC(CH3)=CHCH2, CH2(CH3)C=CHCHaHbCH2CHO(P)CH2OBn, H2C=C(CH3)CH2-CHO(P)CH2), 2.18 (s, 1H, CH2(CH3)C=CHCHaHbCH2CHO(P)CH2OBn), 1.99 (q, J = 5.9, 5.1 Hz, 2H, H2C=C(CH3)CH2-CHO(P)CH2CHO[P]), 1.75 (m, J = 6.8 Hz, 7H, CH2(CH3)C=CHCH2CHaHbCHO(P)CH2OBn, CH2(P)OCHCH2C(CH3)=CHCH2, H2C=C(CH3)CH2-CHO(P)CH2), 1.63 (tt, J = 10.2, 5.1 Hz, 1H, CH2(CH3)C=CHCH2CHaHbCHO(P)CH2OBn); 13C NMR (126 MHz, 95 °C, DMSO-d6) δ ppm 140.3 (C), 138.0 (C), 130.7 (C), 128.3 (CH), 127.8 (2 x CH), 127.03 (2 x CH), 126.95 (CH), 113.2 (CH2), 76.6 (s, br, CH), 76.0 (d, JCP = 6.7 Hz, CH), 75.5 (d, JCP = 8.3 Hz, CH), 72.01 (CH2), 71.98 (CH2), 42.3 (d, JCP = 4.5 Hz, CH2), 35.8 (CH2), 32.0 (s, br, CH2), 30.7 (d, JCP = 3.7 Hz, CH2), 24.9 (s, br, CH3), 23.9 (CH2), 21.9 (CH3); 31P NMR (162 MHz, 95 °C, DMSO-d6) δ ppm –14.7 (s); HRMS (TOF MS ES+) calcd for (C22H31O5P)2Na (2M+Na)+ 835.3716; found 835.3685.
(4S,6S)-4,6-diallyl-2-((4-methylpent-4-en-1-yl)oxy)-1,3,2-dioxaphosphinane 2-oxide (20)
Following General Procedure 1, monochlorophosphate (S,S)-9 (0.2593 g, 1.096 mmol) and 4-methyl-4-penten-1-ol (0.1006 g, 1.005 mmol) were converted into 20 (0.2456 g, 0.8178 mmol, 90%) which was isolated as a colorless oil. FTIR (neat): 2959, 2937, 1443, 1373, 1286, 1096, 1078, 1015, 974, 918, 898, 814, 631, 509 cm−1; [α]D = –35.2 (c 0.84, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 5.84–5.72 (m, 2H, H2C=CH-CH2CHO(P)CH2, H2C=CH-CH2CHO(P)CH2), 5.18–5.12 (m, 4H, H2C=CH-CH2CHO(P)CH2, H2C=CH-CH2CHO(P)CH2), 4.76–4.73 (m, 1H, HaHbC=C(CH3)-CH2-CH2-CH2O(P)), 4.69 (dt, J = 2.3, 1.1 Hz, 1H, HaHbC=C(CH3)-CH2-CH2-CH2O(P)), 4.61 (dddd, J = 14.4, 12.0, 7.0, 5.0 Hz, 1H, H2C=CHCH2-CHO(P)CH2), 4.54–4.46 (m, 1H, H2C=CHCH2-CHO(P)CH2), 4.09 (dt, J = 8.1, 6.6 Hz, 2H, H2C=C(CH3)-CH2-CH2-CH2O(P)), 2.66 (ddd, J = 7.0, 13.9, 7.0 Hz, 1H, H2C=CHCHaHb-CHO(P)CH2), 2.56 (ddd, J = 7.0, 13.9, 7.0 Hz, 1H, H2C=CHCHaHb-CHO(P)CH2), 2.39 (ddd, J = 7.0, 14.1, 7.0 Hz 2H, H2C=CHCHaHb-CHO(P)CH2, H2C=CHCHaHb-CHO(P)CH2), 2.09 (dd, J = 7.33, 7.33 Hz, 2H, H2C=C(CH3)-CH2-CH2-CH2O(P)), 2.02 (dddd, J = 14.9, 8.6, 5.0, 1.4 Hz, 1H, H2C=CHCH2-CHO(P)CHaHbCHO[P]), 1.90–1.80 (m, 3H, H2C=CHCH2-CHO(P)CHaHbCHO[P], H2C=C(CH3)-CH2-CH2-CH2O(P)), 1.74–1.69 (m, 3H, H2C=C(CH3)-CH2-CH2-CH2O(P)); 13C NMR (126 MHz, CDCl3) δ ppm 144.4 (C), 132.6 (CH), 132.2 (CH), 119.0 (CH2), 118.8 (CH2), 110.6 (CH2), 77.0 (d, JCP = 6.8 Hz, CH), 75.5 (d, JCP = 6.6 Hz, CH), 67.5 (d, JCP = 5.9 Hz, CH2), 40.0 (d, JCP = 7.4 Hz, CH2), 38.9 (d, JCP = 3.2 Hz, CH2), 33.6 (CH2), 33.1 (d, JCP = 6.8 Hz, CH2), 28.3 (d, JCP = 6.6 Hz, CH2), 22.4 (CH3); 31P NMR (162 MHz, CDCl3) δ ppm –6.77 (dd, JPH = 14.3, 7.0 Hz); HRMS (TOF MS ES+) calcd for (C15H25O4P)2Na (2M+Na)+ 623.2879; found 623.2886.
(1R,9S,11S,Z)-11-allyl-6-methyl-2,12,13-trioxa-1-phosphabicyclo[7.3.1]tridec-6-ene 1-oxide (22)
To a clean, dry, round bottom flask, equipped with a stirbar, was added triene 20 (25.0 mg, 0.0832 mmol), toluene (dry, degassed, 84 mL), and Ru-21 catalyst (3.3 mg, 0.00416 mmol, 5 mol %). High vacuum was applied to the flask (1 min), and the flask was charged with Ar (repeated 5 times). The flask was equipped with an Ar inlet and heated to 35 °C. The reaction stirred at 35 °C for 2.5 hours, where another 5 mol % of Ru-21 (3.3 mg, 0.00416 mmol) was added. The reaction continued to stir for 1 hour (monitored by TLC), at which point 5 drops of DMSO were added, and the solvent was removed under reduced pressure. The crude mixture was separated by flash chromatography (silica, 0%-50% EtOAc in hexanes) to provide 22 (5.8 mg, 0.0213 mmol, 26%) as a colorless oil. FTIR (neat): 2961, 2928, 1464, 1433, 1281, 1223, 1105, 1072, 1013, 978, 912, 866, 804, 552 cm−1; [α]D = –33.5 (c 0.19, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 5.89–5.78 (m, 1H, CH2(P)OCHCH2CH=CH2), 5.37 (dd, J = 11.5, 2.4 Hz, 1H, CH2(P)OCHCH2CH=C(CH3)CH2), 5.20–5.17 (m, 1H, CH2(P)OCHCH2CH=CHaHb), 5.16 (t, J = 1.3 Hz, 1H, CH2(P)OCHCH2CH=CHaHb), 4.78 (dtd, J = 12.2, 6.2, 2.0 Hz, 1H, CH2(P)OCHCH2CH=CH2), 4.70–4.59 (m, 1H, CH2(P)OCHCH2CH=C(CH3)CH2), 4.06 (ddd, J = 9.9, 4.6, 1.6 Hz, 1H, (P)OCHaHbCH2CH2C(CH3)=CHCH2), 3.92 (dddd, J = 12.8, 9.9, 5.9, 2.6 Hz, 1H, (P)OCHaHbCH2CH2C(CH3)=CHCH2), 3.16 (dt, J = 14.3, 12.5 Hz, 1H, CH2(P)OCHCHaHbCH=C(CH3)CH2), 2.72 (td, J = 13.1, 5.3 Hz, 1H, (P)OCH2CH2CHaHbC(CH3)=CHCH2), 2.57–2.46 (m, 1H, CH2(P)OCHCHaHbCH=CH2), 2.44–2.33 (m, 1H, CH2(P)OCHCHaHbCH=CH2), 2.22 (dddd, J = 14.6, 12.0, 5.5, 0.8 Hz, 1H, H2C=CHCH2-CHO(P)CHaHbCHO[P]), 1.95 (dddt, J = 15.0, 10.1, 5.1, 2.7 Hz, 2H, CH2(P)OCHCHaHbCH=C(CH3)CH2, (P)OCH2CHaHbCH2C(CH3)=CHCH2), 1.89–1.81 (m, 1H, (P)OCH2CH2CHaHbC(CH3)=CHCH2), 1.78–1.72 (m, 1H, (P)OCH2CHaHbCH2C(CH3)=CHCH2), 1.71 (dd, J = 1.7, 1.7 Hz, 3H, CH2(P)OCHCH2CH=C(CH3)CH2), 1.70–1.66 (m, 1H, H2C=CHCH2-CHO(P)CHaHbCHO[P]); 13C NMR (126 MHz, CDCl3) δ ppm 135.2 (C), 132.0 (CH), 122.2 (CH2), 119.0 (CH2), 77.5 (d, JCP = 7.3 Hz, CH), 75.9 (d, JCP = 7.3 Hz, CH), 63.9 (d, JCP = 4.9 Hz, CH2), 40.6 (d, JCP = 9.0 Hz, CH2), 34.5 (d, JCP = 6.7 Hz, CH2), 32.6 (CH2), 26.1 (CH2), 24.9 (d, JCP = 10.0 Hz, CH2), 22.5 (CH3); 31P NMR (162 MHz, CDCl3) δ ppm –8.22 (d, JPH = 24.1 Hz); HRMS (TOF MS ES+) calcd for (C13H21O4P)2Na (2M+Na)+ 567.2253; found 567.2241.
(4S,6S)-4,6-diallyl-2-(((S)-1-(benzyloxy)hept-6-en-2-yl)oxy)-1,3,2-dioxaphosphinane 2-oxide (23)
Following General Procedure 1, monochlorophosphate (S,S)-9 (0.117 g, 0.494 mmol) and alcohol (S)-25 (0.0990 g, 0.450 mmol) were converted into 23 (0.109 g, 0.259 mmol, 58%) which was isolated as a pale yellow oil. FTIR (neat): 2932, 2862, 1454, 1435, 1362, 1286, 1097, 1007, 976, 916, 797, 739, 698, 629, 513 cm−1; [α]D = –33.2 (c 1.13, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 7.34 (d, J = 4.4 Hz, 4H, aromatic C-H), 7.32–7.27 (m, 1H, aromatic C-H), 5.85–5.68 (m, 3H, H2C=CH-CH2CHO(P)CH2, H2C=CH-CH2CHO(P)CH2, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 5.16 (dt, J = 9.0, 1.5 Hz, 1H, HaHbC=CH-CH2CHO(P)CH2), 5.13 (dt, J = 3.6, 1.3 Hz, 2H, HaHbC=CH-CH2CHO(P)CH2, HaHbC=CH-CH2CHO(P)CH2), 5.10 (q, J = 1.6 Hz, 1H, HaHbC=CH-CH2CHO(P)CH2), 5.04–4.99 (m, 1H, HaHbC=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 4.97 (ddd, J = 10.2, 2.2, 1.1 Hz, 1H, HaHbC=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 4.65–4.48 (m, 5H, H2C=CHCH2-CHO(P)CH2, H2C=CHCH2-CHO(P)CH2, CHO(P)CH2OBn, OCH2Ph), 3.61 (dd, J = 4.9, 3.5 Hz, 2H, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 2.63 (ddd, J = 6.7, 13.9, 6.7 Hz, 1H, H2C=CH-CHaHbCHO(P)CH2), 2.56 (m, ddd, J = 6.3, 13.5, 6.3 Hz, 1H, H2C=CH-CHaHbCHO(P)CH2), 2.43–2.33 (m, 2H, H2C=CH-CHaHbCHO(P)CH2, H2C=CH-CHaHbCHO(P)CH2), 2.09 (dtdd, J = 7.9, 6.6, 4.1, 1.3 Hz, 2H, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 2.00 (dddd, J = 14.6, 8.4, 5.0, 1.3 Hz, 1H, H2C=CHCH2-CHO(P)CHaHbCHO[P]), 1.89 (dddd, J = 14.6, 5.7, 4.0, 1.7 Hz, 1H, H2C=CHCH2-CHO(P)CHaHbCHO[P]), 1.73 (dt, J = 8.6, 6.8 Hz, 2H, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 1.57–1.42 (m, 2H, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn); 13C NMR (126 MHz, CDCl3) δ ppm 138.2 (CH), 138.0 (C), 132.6 (CH), 132.3 (CH), 128.3 (CH), 127.62 (2 x CH), 127.59 (2 x CH), 118.8 (CH2), 118.7 (CH2), 114.9 (CH2), 78.2 (d, JCP = 6.3 Hz, CH), 76.8 (d, JCP = 6.9 Hz, CH), 75.4 (d, JCP = 6.8 Hz, CH), 73.2 (CH2), 71.7 (d, JCP = 4.1 Hz, CH2), 40.0 (d, JCP = 7.3 Hz, CH2), 39.1 (d, JCP = 4.0 Hz, CH2), 33.4 (CH2), 33.1 (d, JCP = 6.7 Hz, CH2), 31.6 (d, JCP = 4.9 Hz, CH2), 24.3 (CH2); 31P NMR (162 MHz, CDCl3) δ ppm –6.96 (dt, JPH = 13.6, 7.0 Hz); HRMS (TOF MS ES+) calcd for (C23H33O5P)2Na (2M+Na)+ 863.4029; found 863.4056.
(1R,3S,10S,12S,Z)-12-allyl-3-((benzyloxy)methyl)-2,13,14-trioxa-1-phosphabicyclo[8.3.1]tetradec-7-ene 1-oxide (trans-26)
Following General Procedure 2, triene 5.2 (20.0 mg, 0.0476 mmol) was exposed to G-I catalyst (2 mol %, over 3 hours) in refluxing CH2Cl2 to provide the corresponding bicyclic phosphate trans-5.4 (11.8 mg, 0.0301 mmol, 63%) as a colorless oil. FTIR (neat): 2922, 2860, 1452, 1364, 1273, 1097, 1012, 972, 953, 920, 791, 735, 698, 554 cm−1; [α]D = –63.9 (c 0.44, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 7.39–7.32 (m, 4H, Aromatic C-H), 7.32–7.27 (m, 1H, Aromatic C-H), 5.80 (dddd, J = 16.9, 10.2, 7.5, 6.5 Hz, 1H, CH2(P)OCHCH2CH=CH2), 5.58–5.47 (m, 2H, CH2(P)OCHCH2CH=CHCH2), 5.16 (dq, J = 17.2, 1.6 Hz, 1H, CH2(P)OCHCH2CH=CHaHb), 5.12 (ddt, J = 10.2, 2.1, 1.2 Hz, 1H, CH2(P)OCHCH2CH=CHaHb), 4.72–4.61 (m, 3H, CH2(P)OCHCH2CH=CHCH2, CH2(P)OCHCH2CH=CH2, –OCHaHbPh), 4.57 (d, J = 11.8 Hz, 1H, –OCHaHbPh), 4.38–4.27 (m, 1H, -CH2HC=CHCH2CH2CH2CHO(P)CH2OBn), 3.72–3.55 (m, 2H, CH2CHO(P)CH2OBn), 2.98–2.89 (m, 1H, CH2(P)OCHCHaHbCH=CHCH2), 2.62–2.45 (m, 2H, CH2(P)OCHCHaHbCH=CH2, H2C=CHCH2-CHO(P)CHaHbCHO[P]), 2.45–2.34 (m, 2H, CH2(P)OCHCHaHbCH=CH2, CH2HC=CHCHaHbCH2CH2CHO(P)), 2.00–1.86 (m, 3H, CH2(P)OCHCHaHbCH=CHCH2, CH2HC=CHCH2CHaHbCH2CHO(P), CH2HC=CHCHaHbCH2CH2CHO(P)), 1.83 (dtd, J = 15.1, 2.8, 1.5 Hz, 1H, H2C=CHCH2-CHO(P)CHaHbCHO[P]), 1.62–1.57 (m, 1H, CH2HC=CHCH2CHaHbCH2CHO(P)), 1.53 (dqd, J = 10.7, 5.3, 3.6 Hz, 2H, CH2HC=CHCH2CH2CH2CHO(P)); 13C NMR (126 MHz, CDCl3) δ ppm 138.1 (C), 132.8 (CH), 132.1 (CH), 128.3 (2 x CH), 127.7 (2 x CH), 127.6 (CH), 125.1 (CH), 118.9 (CH2), 77.5 (d, JCP = 8.4 Hz, CH), 76.9 (d, JCP = 6.5 Hz, CH), 76.4 (d, JCP = 6.1 Hz, CH), 73.6 (CH2), 72.3 (d, JCP = 6.8 Hz, CH2), 40.9 (d, JCP = 4.6 Hz, CH2), 34.5 (d, JCP = 12.0 Hz, CH2), 33.1 (d, JCP = 3.1 Hz, CH2), 28.9 (CH2), 24.1 (CH2), 23.8 (CH2); 31P NMR (162 MHz, CDCl3) δ ppm –10.13 (m); HRMS (TOF MS ES+) calcd for (C21H29O5P)2Na (2M+Na)+ 807.3403; found 807.3404.
(4S,6S)-4,6-diallyl-2-(((R)-1-(benzyloxy)hept-6-en-2-yl)oxy)-1,3,2-dioxaphosphinane 2-oxide (24)
Following General Procedure 1, monochlorophosphate (S,S)-9 (0.117 g, 0.494 mmol) and alcohol (R)-25 (0.0990 g, 0.450 mmol) were converted into 24 (0.120 g, 0.259 mmol, 63%) which was isolated as a colorless oil. FTIR (neat): 2930, 2862, 1454, 1435, 1362, 1288, 1099, 1078, 999, 974, 916, 800, 739, 698, 629, 548 cm−1; [α]D = –39.7 (c 1.08, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 7.36–7.28 (m, 5H, aromatic C-H), 5.83–5.73 (m, 2H, H2C=CH-CH2CHO(P)CH2, H2C=CH-CH2CHO(P)CH2), 5.73–5.63 (m, 1H, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 5.14–5.10 (m, 2H, HaHbC=CH-CH2CHO(P)CH2, HaHbC=CH-CH2CHO(P)CH2), 5.10–5.07 (m, 2H, HaHbC=CH-CH2CHO(P)CH2, HaHbC=CH-CH2CHO(P)CH2), 5.01 (dq, J = 17.1, 1.7 Hz, 1H, HaHbC=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 4.95 (ddt, J = 10.2, 2.3, 1.2 Hz, 1H, HaHbC=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 4.61–4.50 (m, 5H, H2C=CHCH2-CHO(P)CH2, H2C=CHCH2-CHO(P)CH2, CHO(P)CH2OBn, OCH2Ph), 3.62–3.54 (m, 2H, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 2.72–2.64 (m, 1H, H2C=CH-CHaHbCHO(P)CH2), 2.48–2.41 (m, 1H, H2C=CH-CHaHbCHO(P)CH2), 2.41–2.34 (m, 1H, H2C=CH-CHaHbCHO(P)CH2), 2.34–2.25 (m, 1H, H2C=CH-CHaHbCHO(P)CH2), 2.12–2.04 (m, 2H, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 1.96 (dddd, J = 14.8, 8.7, 5.0, 1.4 Hz, 1H, H2C=CHCH2-CHO(P)CHaHbCHO[P]), 1.82 (dddd, J = 14.6, 5.1, 3.7, 1.8 Hz, 1H, H2C=CHCH2-CHO(P)CHaHbCHO[P]), 1.78–1.66 (m, 2H, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 1.58–1.41 (m, 2H, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn); 13C NMR (126 MHz, CDCl3) δ ppm 138.3 (CH), 137.9 (C), 132.8 (CH), 132.3 (CH), 128.3 (2 x CH), 127.7 (CH), 127.6 (2 x CH), 118.7 (CH2), 118.6 (CH2), 114.8 (CH2), 77.8 (d, JCP = 6.2 Hz, CH), 77.4 (d, JCP = 6.9 Hz, CH), 74.9 (d, JCP = 6.5 Hz, CH), 73.2 (CH2), 71.9 (d, JCP = 4.5 Hz, CH2), 40.0 (d, JCP = 7.8 Hz, CH2), 38.8 (d, JCP = 3.2 Hz, CH2), 33.4 (CH2), 33.0 (d, JCP = 6.7 Hz, CH2), 31.7 (d, JCP = 4.5 Hz, CH2), 24.2 (CH2); 31P NMR (162 MHz, CDCl3) δ ppm –7.15 (dt, JPH = 13.0, 5.4 Hz); HRMS (TOF MS ES+) calcd for (C23H33O5P)2Na (2M+Na)+ 863.4029; found 863.4053.
(1R,3R,10S,12S,Z)-12-allyl-3-((benzyloxy)methyl)-2,13,14-trioxa-1-phosphabicyclo[8.3.1]tetradec-7-ene 1-oxide (cis-27, Z-product [major])
Following General Procedure 2, triene 24 (25.0 mg, 0.0595 mmol) was exposed to G-I catalyst (3 mol %, over 4 hours) in refluxing CH2Cl2 to provide the corresponding bicyclic phosphates cis-27 (13.8 mg, 0.0352 mmol, 59% combined yield, colorless oil) as 2:1 mixture of Z/E-isomers, which was further purified to a 10:1 mixture of Z/E-isomers for full characterization of the major product. FTIR (neat): 2930, 2862, 1452, 1362, 1283, 1101, 1078, 1042, 997, 974, 918, 826, 795, 739, 698, 557 cm−1; [α]D = –41.0 (c 0.20, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 7.38–7.33 (m, 4H, Aromatic C-H), 7.32–7.28 (m, 1H, Aromatic C-H), 5.79 (dddd, J = 17.0, 10.2, 7.5, 6.5 Hz, 1H, CH2(P)OCHCH2CH=CH2), 5.49 (dddd, J = 10.8, 10.8, 3.3, 2.0 Hz, 1H, CH2(P)OCHCH2CH=CHCH2), 5.47 (dddd, J = 11.0, 11.0, 3.2, 1.9 Hz, 1H, CH2(P)OCHCH2CH=CHCH2), 5.14 (dq, J = 17.1, 1.6 Hz, 1H, CH2(P)OCHCH2CH=CHaHb), 5.10 (ddd, J = 10.2, 2.0, 1.0 Hz, 1H, CH2(P)OCHCH2CH=CHaHb), 5.06 (dtd, J = 9.9, 4.8, 2.5 Hz, 1H, CH2CHO(P)CH2OBn), 4.72–4.65 (m, 1H, CH2(P)OCHCH2CH=CH2), 4.61 (m, 2H, CH2(P)OCHCH2CH=CHCH2, -OCHaHbPh), 4.56 (d, J = 12.3 Hz, 1H, -OCHaHbPh), 3.70–3.65 (m, 1H, CH2CHO(P)CHaHbOBn), 3.58 (ddd, J = 10.5, 5.3, 2.0 Hz, 1H, CH2CHO(P)CHaHbOBn), 3.19 (ddd, J = 14.5, 12.6, 10.5 Hz, 1H, CH2(P)OCHCHaHbCH=CHCH2), 2.63–2.53 (m, 1H, -CH2HC=CHCHaHbCH2CH2CHO(P)), 2.48 (dddd, J = 12.8, 7.8, 4.9, 1.4 Hz, 1H, CH2(P)OCHCHaHbCH=CH2), 2.40–2.32 (m, 1H, CH2(P)OCHCHaHbCH=CH2), 2.25–2.16 (m, 1H, H2C=CHCH2-CHO(P)CHaHbCHO[P]), 1.95 (ddt, J = 15.6, 12.9, 2.9 Hz, 2H, -CH2HC=CHCHaHbCH2CHaHbCHO(P)), 1.87 (dt, J = 14.8, 2.7 Hz, 1H, CH2(P)OCHCHaHbCH=CHCH2), 1.78 (tdd, J = 16.0, 6.1, 3.5 Hz, 1H, -CH2HC=CHCH2CHaHbCH2CHO(P)), 1.73–1.65 (m, 2H, CH2HC=CHCH2CH2CHaHbCHO(P), H2C=CHCH2-CHO(P)CHaHbCHO[P]), 1.45 (ddtd, J = 14.6, 12.2, 4.8, 2.8 Hz, 1H, -CH2HC=CHCH2CHaHbCH2CHO(P)); 13C NMR (126 MHz, CDCl3) δ ppm 138.1 (C), 132.5 (CH), 132.1 (CH), 128.3 (2 x CH), 127.6 (CH), 127.5 (2 x CH), 125.3 (CH), 118.8 (CH2), 78.1 (d, JCP = 7.8 Hz, CH), 77.9 (d, JCP = 6.8 Hz, CH), 75.0 (d, JCP = 7.2 Hz, CH), 72.9 (CH2), 70.3 (d, JCP = 6.6 Hz, CH2), 40.6 (d, JCP = 9.0 Hz, CH2), 34.4 (d, JCP = 6.7 Hz, CH2), 32.4 (CH2), 28.4 (d, JCP = 2.2 Hz, CH2), 25.8 (CH2), 22.9 (CH2); 31P NMR (162 MHz, CDCl3) δ ppm –8.85 (dd, JPH = 23.8, 9.5 Hz); HRMS (TOF MS ES+) calcd for (C21H29O5P)2Na (2M+Na)+ 807.3403; found 807.3422.
(4R,6R)-2-(hex-5-en-1-yloxy)-4,6-bis(2-methylallyl)-1,3,2-dioxaphosphinane 2-oxide (28)
Following General Procedure 1, (R,R)-monochlorophosphate 14 (0.215 g, 0.812 mmol) and 5-hexen-1-ol (74.6 mg, 0.745 mmol) were converted to phosphate triene 28 (0.193 g, 0.5877 mmol, 87%) which was isolated as a colorless oil. FTIR (neat): 2964, 2936, 2858, 1443, 1377, 1288, 1026, 1007, 970, 893, 799, 546 cm−1; [α]D = +44.3 (c 0.14, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 5.79 (ddt, J = 16.9, 10.2, 6.7 Hz, 1H, (P)OCH2CH2CH2CH2CH=CH2), 5.02 (dd, J = 17.2, 1.9 Hz, 1H, (P)OCH2CH2CH2CH=CHaHb), 4.97 (dd, J = 10.3, 1.7 Hz, 1H, (P)OCH2CH2CH2CH=CHaHb), 4.87 (q, J = 1.7 Hz, 2H, (P)OCH2CH2CH2C(CH3)=CHaHb, (P)OCH2CH2CH2C(CH3)=CHaHb), 4.79–4.77 (m, 2H, (P)OCH2CH2CH2C(CH3)=CHaHb, (P)OCH2CH2CH2C(CH3)=CHaHb), 4.78 (d, J = 7.0 Hz, 1H, H2C=C(CH3)-CH2CHO(P)CH2), 4.63 (dtdd, J = 8.7, 6.9, 5.1, 3.6 Hz, 1H, H2C=C(CH3)-CH2CHO(P)CH2), 4.08 (q, J = 6.8 Hz, 2H, (P)OCH2CH2CH2CH2CH=CH2), 2.62 (dd, J = 14.2, 7.0 Hz, 1H, H2C=C(CH3)-CHaHbCHO(P)CH2), 2.55 (dd, J = 14.2, 6.9 Hz, 1H, H2C=C(CH3)-CHaHbCHO(P)CH2), 2.40–2.30 (m, 2H, H2C=C(CH3)-CHaHbCHO(P)CH2, H2C=C(CH3)-CHaHbCHO(P)CH2), 2.09 (q, J = 7.1 Hz, 2H, (P)OCH2CH2CH2CH=CH2), 2.01 (dddd, J = 13.5, 8.6, 5.0, 1.3 Hz, 1H, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P]), 1.86 (dddd, J = 14.7, 5.2, 3.5, 1.8 Hz, 1H, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P]), 1.76 (d, J = 5.9 Hz, 6H, (P)OCH2CH2CH2C(CH3)=CH2, (P)OCH2CH2CH2C(CH3)=CH2), 1.74–1.67 (m, 2H, H2C=CH-CH2-CH2-CH2-CHO(P)), 1.49 (p, J = 7.5 Hz, 2H, H2C=CH-CH2-CH2-CH2-CHO(P)); 13C NMR (126 MHz, CDCl3) δ ppm 140.3 (C), 140.2 (C), 138.2 (CH), 114.9 (CH2), 114.18 (CH2), 114.17 (CH2), 75.8 (d, JCP = 6.8 Hz, CH), 74.5 (d, JCP = 6.5 Hz, CH), 67.7 (d, JCP = 5.9 Hz, CH2), 43.7 (d, JCP = 7.3 Hz, CH2), 42.7 (d, JCP = 3.2 Hz, CH2), 33.4 (d, JCP = 6.8 Hz, CH2), 33.1 (CH2), 29.7 (d, JCP = 6.7 Hz, CH2), 24.8 (CH2), 22.6 (CH3), 22.3 (CH3); 31P NMR (162 MHz, CDCl3) δ ppm –6.67 (s); HRMS (TOF MS ES+) calcd for (C17H29O4P)2Na (2M+Na)+ 679.3505; found 679.3520.
(1S,10R,12R,Z)-8-methyl-12-(2-methylallyl)-2,13,14-trioxa-1-phosphabicyclo[8.3.1]tetradec-7-ene 1-oxide (29)
Following General Procedure 2, triene 28 (20.0 mg, 0.0609 mmol) was exposed to G-II catalyst (7 mol %, over 9 hours) in refluxing CH2Cl2 to provide the corresponding bicyclic phosphate 6.2 (14.8 mg, 0.0493 mmol, 81%) as a colorless oil. FTIR (neat): 2963, 2916, 1855, 1443, 1377, 1103, 1072, 1053, 1020, 1005, 978, 943, 924, 897, 793, 762, 544 cm−1; [α]D = +37.3 (c 0.15, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 5.18 (ddd, J = 12.0, 3.4, 1.7 Hz, 1H, (P)OCHCH2C(CH3)=CHCH2), 4.93–4.83 (m, 2H, (P)OCHCH2C(CH3)=CHaHb), 4.83–4.76 (m, 2H, (P)OCHCH2C(CH3)=CHaHb, (P)OCHCH2C(CH3)=CHCH2), 4.74–4.68 (m, 1H, (P)OCHaHbCH2CH2CH2CH=C(CH3)CH2), 3.89 (dddd, J = 19.0, 11.7, 10.6, 1.2 Hz, 1H, (P)OCHaHbCH2CH2CH2CH=C(CH3)CH2), 3.31 (dd, J = 14.1, 13.0 Hz, 1H, (P)OCHCHaHbC(CH3)=CHCH2), 2.60 (dddd, J = 14.2, 11.9, 9.4, 6.3 Hz, 1H, (P)OCH2CH2CH2CHaHbCH=C(CH3)CH2), 2.51 (dd, J = 14.2, 6.7 Hz, 1H, (P)OCHCHaHbC(CH3)=CH2), 2.29 (dddd, J = 14.3, 6.5, 2.8, 1.0 Hz, 1H, (P)OCHCHaHbC(CH3)=CH2), 2.25–2.16 (m, 1H, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P]), 2.03–1.96 (m, 1H, (P)OCH2CH2CH2CHaHbCH=C(CH3)CH2), 1.90–1.81 (m, 1H, (P)OCH2CHaHbCH2CH2CH=C(CH3)CH2), 1.79 (dt, J = 3.9, 1.1 Hz, 6H, (P)OCHCH2C(CH3)=CHCH2, (P)OCHCH2C(CH3)=CH2), 1.72–1.62 (m, 4H, (P)OCHCHaHbC(CH3)=CHCH2, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P], (P)OCH2CH2CH2CH2CH=C(CH3)CH2), 1.59–1.53 (m, 1H, (P)OCH2CHaHbCH2CH2CH=C(CH3)CH2); 13C NMR (126 MHz, CDCl3) δ ppm 140.1 (C), 130.2 (C), 128.8 (CH), 114.2 (CH2), 75.8 (d, JCP = 8.0 Hz, CH), 74.0 (d, JCP = 6.9 Hz, CH), 71.6 (d, JCP = 6.6 Hz, CH2), 44.4 (d, JCP = 8.8 Hz, CH2), 36.0 (CH2), 34.7 (d, JCP = 7.0 Hz, CH2), 28.6 (CH2), 27.4 (CH2), 26.0 (CH2), 22.9 (CH3), 22.2 (CH3); 31P NMR (162 MHz, CDCl3) δ ppm –7.95 (td, JPH = 20.5, 9.7 Hz); HRMS (TOF MS ES+) calcd for (C15H25O4P)2Na (2M+Na)+ 623.2879; found 623.2889.
(4R,6R)-2-(((S)-1-(benzyloxy)hept-6-en-2-yl)oxy)-4,6-bis(2-methylallyl)-1,3,2-dioxaphosphinane 2-oxide (30)
Following General Procedure 1, monochlorophosphate (R,R)-14 (0.180 g, 0.679 mmol) and alcohol (S)-25 (0.137 g, 0.622 mmol) were converted into 30 (0.158 g, 0.352 mmol, 62%) which was isolated as a colorless oil. FTIR (neat): 2936, 2862, 1452, 1375, 1366, 1286, 1207, 1003, 1101, 1070, 1003, 970, 8945, 812, 739, 698, 553 cm−1; [α]D = +34.9 (c = 0.93, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 7.34 (d, J = 3.7 Hz, 4H, aromatic C-H), 7.32–7.28 (m, 1H, aromatic C-H), 5.78 (ddt, J = 16.9, 10.2, 6.7 Hz, 1H, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 5.00 (dq, J = 17.1, 1.7 Hz, 1H, HaHbC=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 4.95 (ddt, J = 10.2, 2.2, 1.2 Hz, 1H, HaHbC=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 4.84 (dt, J = 14.7, 1.6 Hz, 2H, H2C=C(CH3)-CH2CHO(P)CH2), 4.77–4.66 (m, 4H, H2C=C(CH3)-CH2CHO(P)CH2, H2C=C(CH3)-CH2CHO(P)CH2, H2C=C(CH3)-CH2CHO(P)CH2), 4.62–4.51 (m, 3H, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn, OCH2Ph), 3.58 (dd, J = 4.8, 1.2 Hz, 2H, -CH2OBn), 2.63 (ddd, J = 14.2, 7.1, 1.2 Hz, 1H, H2C=C(CH3)-CHaHbCHO(P)CH2), 2.49 (dd, J = 14.2, 6.4 Hz, 1H, H2C=C(CH3)-CHaHbCHO(P)CH2), 2.36 (dd, J = 14.1, 7.4 Hz, 1H, H2C=C(CH3)-CHaHbCHO(P)CH2), 2.29 (ddt, J = 14.2, 7.4, 1.3 Hz, 1H, H2C=C(CH3)-CHaHbCHO(P)CH2), 2.07 (qdd, J = 6.4, 2.9, 1.4 Hz, 2H, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 1.96 (dddd, J = 14.5, 8.1, 4.9, 1.4 Hz, 1H, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P]), 1.86 (dddd, J = 14.6, 5.4, 3.8, 1.7 Hz, 1H, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P]), 1.75 (s, 3H, H2C=C(CH3)CH2-CHO(P)), 1.74–1.70 (m, 2H, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 1.69 (t, J = 1.2 Hz, 3H, H2C=C(CH3)CH2-CHO(P)), 1.57–1.41 (m, 2H, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn); 13C NMR (126 MHz, CDCl3) δ ppm 140.5 (C), 140.3 (C), 138.3 (CH), 137.9 (C), 128.4 (2 x CH), 127.7 (CH), 127.6 (2 x CH), 114.8 (CH2), 114.12 (CH2), 114.11 (CH2), 77.9 (d, J = 6.2 Hz, CH), 76.0 (d, J = 6.8 Hz, CH), 74.3 (d, J = 6.5 Hz, CH), 73.2 (CH2), 71.9 (d, J = 4.5 Hz, CH2), 43.8 (d, J = 7.5 Hz, CH2), 42.8 (d, J = 3.6 Hz, CH2), 33.5 (CH2), 33.3 (d, J = 6.8 Hz, CH2), 31.7 (d, J = 4.5 Hz, CH2), 24.2 (CH2), 22.6 (CH3), 22.4 (CH3); 31P NMR (162 MHz, CDCl3) δ ppm –7.07 (dt, JPH = 13.1, 6.0 Hz); HRMS (TOF MS ES+) calcd for (C25H37O5P)2Na (2M+Na)+ 919.4655; found 919.4643.
(1S,3S,10R,12R,Z)-3-((benzyloxy)methyl)-8-methyl-12-(2-methylallyl)-2,13,14-trioxa-1-phosphabicyclo[8.3.1]tetradec-7-ene 1-oxide (cis-32)
Following General Procedure 2, triene 30 (30.0 mg, 0.0669 mmol) was exposed to G-II catalyst (4 mol %, over 5 hours) in refluxing CH2Cl2 to provide the corresponding bicyclic phosphate cis-32 (19.2 mg, 0.0457 mmol, 68%) as a colorless oil. FTIR (neat): 2963, 2936, 2920, 2862, 1452, 1377, 1283, 1103, 1074, 1003, 972, 918, 797, 698, 550 cm−1; [α]D = +32.4 (c 0.17, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 7.37–7.33 (m, 4H, Aromatic C-H), 7.32–7.28 (m, 1H, Aromatic C-H), 5.20 (ddt, J = 11.8, 3.2, 1.5 Hz, 1H, (P)OCHCH2C(CH3)=CHCH2), 5.05 (ddtd, J = 10.0, 7.7, 4.9, 3.0 Hz, 1H, (P)OCHCH2C(CH3)=CHCH2), 4.88 (ddt, J = 15.8, 9.5, 4.4 Hz, 1H, (P)OCHCH2C(CH3)=CH2), 4.83–4.76 (m, 3H, (P)OCHCH2C(CH3)=CH2, CH2(CH3)C=CHCH2CH2CH2CHO(P)CH2OBn), 4.59 (d, J = 12.0 Hz, 1H, -OCHaHbPh), 4.56 (d, J = 12.0 Hz, 1H, -OCHaHbPh), 3.66 (dd, J = 10.4, 7.3 Hz, 1H, CH2CHO(P)CHaHbOBn), 3.56 (ddd, J = 10.4, 5.2, 1.9 Hz, 1H, CH2CHO(P)CHaHbOBn), 3.29 (t, J = 13.6 Hz, 1H, (P)OCHCHaHbC(CH3)=CHCH2), 2.58–2.50 (m, 1H, CH2(CH3)C=CHCHaHbCH2CH2CHO(P)), 2.47 (dd, J = 14.4, 7.0 Hz, 1H, (P)OCHCHaHbC(CH3)=CH2), 2.28 (dddd, J = 14.5, 6.2, 3.0, 1.0 Hz, 1H, (P)OCHCHaHbC(CH3)=CH2), 2.24–2.16 (m, 1H, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P]), 1.94 (ddt, J = 15.5, 5.7, 2.9 Hz, 2H, CH2(CH3)C=CHCHaHbCH2CH2CHO(P), CH2(CH3)C=CHCH2CH2CHaHbCHO(P)CH2OBn), 1.80–1.72 (m, 7H, CH2(CH3)C=CHCH2CHaHbCH2CHO(P)CH2OBn, (P)OCHCH2C(CH3)=CHCH2, (P)OCHCH2C(CH3)=CH2), 1.72–1.65 (m, 3H, (P)OCHCHaHbC(CH3)=CHCH2, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P], CH2(CH3)C=CHCH2CH2CHaHbCHO(P)CH2OBn), 1.42 (ddtd, J = 14.6, 12.5, 5.1, 2.7 Hz, 1H, CH2(CH3)C=CHCH2CHaHbCH2CHO(P)CH2OBn); 13C NMR (126 MHz, CDCl3) δ ppm 140.2 (C), 138.1 (C), 130.3 (C), 128.6 (CH), 128.3 (2 x CH), 127.53 (CH), 127.52 (2 x CH), 114.0 (CH2), 78.1 (d, JCP = 6.9 Hz, CH), 75.7 (d, JCP = 8.1 Hz, CH), 73.9 (d, JCP = 6.9 Hz, CH), 72.9 (CH2), 70.5 (d, JCP = 6.3 Hz, CH2), 44.3 (d, JCP = 8.9 Hz, CH2), 36.0 (CH2), 34.7 (d, JCP = 6.7 Hz, CH2), 28.3 (d, JCP = 2.3 Hz, CH2), 26.5 (CH2), 23.3 (CH2), 22.8 (CH3), 22.2 (CH3); 31P NMR (162 MHz, CDCl3) δ ppm –8.71 (dd, JPH = 23.2, 9.4 Hz); HRMS (TOF MS ES+) calcd for (C23H33O5P)2Na (2M+Na)+ 863.4029; found 863.4021.
(4R,6R)-2-(((R)-1-(benzyloxy)hept-6-en-2-yl)oxy)-4,6-bis(2-methylallyl)-1,3,2-dioxaphosphinane 2-oxide (31)
Following General Procedure 1, monochlorophosphate (R,R)-14 (0.180 g, 0.679 mmol) and alcohol (R)-25 (0.137 g, 0.622 mmol) were converted into 31 (0.118 g, 0.263 mmol, 47%) which was isolated as a colorless oil. FTIR (neat): 2936, 2860, 1452, 1377, 1366, 1285, 1101, 1069, 1003, 897, 847, 808, 739, 698, 550 cm−1; [α]D = +30.5 (c 1.00, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 7.34 (d, J = 4.4 Hz, 4H, aromatic C-H), 7.31–7.27 (m, 1H, aromatic C-H), 5.79 (ddt, J = 16.9, 10.1, 6.7 Hz, 1H, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 5.02 (dq, J = 17.1, 1.6 Hz, 1H, HaHbC=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 4.96 (ddt, J = 10.2, 2.2, 1.2 Hz, 1H, HaHbC=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 4.87 (t, J = 1.7 Hz, 1H, HaHbC=C(CH3)-CH2CHO(P)CH2), 4.85–4.83 (m, 1H, HaHbC=C(CH3)-CH2CHO(P)CH2), 4.79 (dq, J = 1.9, 0.9 Hz, 1H, HaHbC=C(CH3)-CH2CHO(P)CH2), 4.76–4.70 (m, 2H, HaHbC=C(CH3)-CH2CHO(P)CH2, H2C=C(CH3)-CH2CHO(P)CH2), 4.67–4.57 (m, 3H, H2C=C(CH3)-CH2CHO(P)CH2, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn, OCHaHbPh), 4.53 (d, J = 12.0 Hz, 1H, OCHaHbPh), 3.61 (t, J = 4.9 Hz, 2H, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 2.64–2.51 (m, 2H, H2C=C(CH3)-CH2CHO(P)CH2), 2.40–2.31 (m, 2H, H2C=C(CH3)-CH2CHO(P)CH2), 2.09 (dtdt, J = 7.9, 6.5, 3.6, 1.3 Hz, 2H, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 1.98 (dddd, J = 14.4, 8.0, 4.9, 1.5 Hz, 1H, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P]), 1.89 (dddd, J = 14.6, 5.6, 3.9, 1.7 Hz, 1H, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P]), 1.78–1.69 (m, 8H, H2C=C(CH3)-CH2CHO(P)CH2, H2C=C(CH3)-CH2CHO(P)CH2, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn), 1.57–1.41 (m, 2H, H2C=CH-CH2-CH2-CH2-CHO(P)CH2OBn); 13C NMR (126 MHz, CDCl3) δ ppm 140.4 (C), 140.3 (C), 138.2 (CH), 138.1 (C), 128.3 (2 x CH), 127.60 (2 x CH), 127.57 (CH), 114.9 (CH2), 114.20 (CH2), 114.15 (CH2), 78.2 (d, J = 6.3 Hz, CH), 75.6 (d, J = 6.6 Hz, CH), 74.6 (d, J = 6.7 Hz, CH), 73.2 (CH2), 71.7 (d, J = 4.1 Hz, CH2), 43.7 (d, J = 7.1 Hz, CH2), 43.0 (d, J = 4.1 Hz, CH2), 33.4 (CH2), 33.3 (d, J = 6.7 Hz, CH2), 31.6 (d, J = 4.9 Hz, CH2), 24.3 (CH2), 22.6 (CH3), 22.3 (CH3); 31P NMR (162 MHz, CDCl3) δ ppm –6.90 (dt, JPH = 13.5, 6.8 Hz); HRMS (TOF MS ES+) calcd for (C25H37O5P)2Na (2M+Na)+ 919.4655; found 919.4681.
(1S,3R,10R,12R,Z)-3-((benzyloxy)methyl)-8-methyl-12-(2-methylallyl)-2,13,14-trioxa-1-phosphabicyclo[8.3.1]tetradec-7-ene 1-oxide (trans-33)
Following General Procedure 2, triene 31 (20.0 mg, 0.0446 mmol) was exposed to G-II catalyst (2 mol %, over 3 hours) in refluxing CH2Cl2 to provide the corresponding bicyclic phosphate trans-33 (13.5 mg, 0.0321 mmol, 72%) as a white, crystalline solid, which was recrystallized via vapor-diffusion method (EtOAc:Hexanes) to afford X-ray quality crystals for X-ray crystallographic analysis (which confirmed stereochemical olefin assignment as Z). FTIR (neat): 2961, 2924, 2858, 1452, 1378, 1273, 1105, 1094, 1076, 1020, 974, 951, 908, 783, 739, 698, 542 cm−1; [α]D = +54.4 (c 0.86, CHCl3); 1H NMR (500 MHz, CDCl3) δ ppm 7.38–7.32 (m, 4H, Aromatic C-H), 7.29 (dt, J = 6.8, 3.0 Hz, 1H, Aromatic C-H), 5.29 (dd, J = 12.3, 4.0 Hz, 1H, CH2(P)OCHCH2C(CH3)=CHCH2), 4.89–4.82 (m, 2H, CH2(P)OCHCH2C(CH3)=CHCH2, CH2(P)OCHCH2C(CH3)=CHaHb), 4.80 (dd, J = 2.1, 1.0 Hz, 1H, CH2(P)OCHCH2C(CH3)=CHaHb), 4.76 (ddd, J = 11.3, 6.5, 2.5 Hz, 1H, CH2(P)OCHCH2C(CH3)=CH2), 4.61 (d, J = 11.9 Hz, 1H, -OCHaHbPh), 4.56 (d, J = 11.9 Hz, 1H, -OCHaHbPh), 4.31 (dddd, J = 14.0, 11.5, 6.8, 4.6 Hz, 1H, CH2(CH3)C=CHCH2CH2CH2CHO(P)CH2OBn), 3.63 (dd, J = 10.8, 6.6 Hz, 1H, CH2CHO(P)CHaHbOBn), 3.58 (ddd, J = 10.7, 3.9, 2.4 Hz, 1H, CH2CHO(P)CHaHbOBn), 3.12–3.05 (m, 1H, CH2(P)OCHCHaHbC(CH3)=CHCH2), 2.57 (dd, J = 14.2, 6.6 Hz, 1H, CH2(P)OCHCHaHbC(CH3)=CH2), 2.50 (ddd, J = 15.1, 11.3, 6.2 Hz, 1H, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P]), 2.35 (ddt, J = 14.3, 6.8, 1.3 Hz, 1H, CH2(P)OCHCHaHbC(CH3)=CH2), 2.32–2.25 (m, 1H, CH2(CH3)C=CHCHaHbCH2CH2CHO(P)CH2OBn), 1.93–1.80 (m, 3H, H2C=C(CH3)CH2-CHO(P)CHaHbCHO[P], CH2(CH3)C=CHCHaHbCH2CHaHbCHO(P)CH2OBn), 1.76 (dt, J = 10.3, 1.4 Hz, 7H, CH2(P)OCHCHaHbC(CH3)=CHCH2, CH2(P)OCHCH2C(CH3)=CHCH2, CH2(P)OCHCH2C(CH3)=CH2), 1.53 (dddd, J = 25.2, 12.3, 6.8, 3.4 Hz, 3H, CH2(CH3)C=CHCH2CH2CHaHbCHO(P)CH2OBn); 13C NMR (126 MHz, CDCl3) δ ppm 140.3 (C), 138.1 (C), 130.1 (C), 128.6 (CH), 128.3 (2 x CH), 127.7 (2 x CH), 127.6 (CH), 114.1 (CH2), 77.1 (d, JCP = 8.6 Hz, CH), 75.8 (d, JCP = 6.1 Hz, CH), 74.3 (d, JCP = 6.4 Hz, CH), 73.6 (CH2), 72.4 (d, JCP = 7.0 Hz, CH2), 44.5 (d, JCP = 4.9 Hz, CH2), 37.4 (d, JCP = 4.2 Hz, CH2), 34.8 (d, JCP = 11.8 Hz, CH2), 29.1 (d, JCP = 1.9 Hz, CH2), 25.0 (CH2), 23.9 (CH2), 23.0 (CH3), 22.8 (CH3); 31P NMR (162 MHz, CDCl3) δ ppm –9.90 (d, JPH = 9.8 Hz); HRMS (TOF MS ES+) calcd for (C23H33O5P)2Na (2M+Na)+ 863.4029; found 863.4023.
Supplementary Material
Acknowledgments
This investigation was generously supported by funds provided by the National Institute of General Medical Sciences (NIH R01 GM077309-7). The authors thank Justin Douglas and Sarah Neuenswander in the University of Kansas NMR Laboratory and Dr. Todd Williams for HRMS analysis. Support for the NMR instrumentation was provided by NSF Grant #9512331, #9977422, #0320648 and NIH Center Grant #P20 GM103418, #S10RR024664 and #S10 OD016360. We kindly acknowledge Dr. Victor Day of the Molecular Structure Group (MSG) at the University of Kansas for X-ray analysis (NSF-MRI grant CHE-0923449). The authors thank the University of Kansas and the State of Kansas for support of our program. The authors also thank Materia, Inc. for supplying metathesis catalysts.
Footnotes
For NMR spectra of all new compounds and X-ray crystallographic data (including CIF files, thermal ellipsoid plots, and select tabular X-ray crystallographic data) of 15 and trans-33, see the provided Supporting Information files.
References
- 1.For an excellent review on reversible, covalent bonding and induced intramolecularity as it relates to catalysis, see: Tan KL. ACS Catal. 2011;1:877–886.
- 2.(a) Evans PA. Temporary Silicon-Tethered Ring-Closing Metathesis Reactions in Natural Product Synthesis. In: Cossy J, Areniyadis S, Meyer C, editors. Metathesis in Natural Product Synthesis. Wiley-VCH; Weinheim, Germany: 2010. pp. 225–259. [Google Scholar]; (b) Cusak A. Chem Eur J. 2012;18:5800–5824. doi: 10.1002/chem.201103186. [DOI] [PubMed] [Google Scholar]; (c) Evans PA, Cui J, Buffone GP. Angew Chem, Int Ed. 2003;42:1734–1737. doi: 10.1002/anie.200250486. [DOI] [PMC free article] [PubMed] [Google Scholar]; (d) Matsui R, Seto K, Fujita K, Suzuki T, Nakazaki A, Kobayashi S. Angew Chem, Int Ed. 2010;49:10068–10073. doi: 10.1002/anie.201004746. [DOI] [PubMed] [Google Scholar]
- 3.Whitehead A, McReynolds MD, Moore JD, Hanson PR. Org Lett. 2005;7:3375–3378. doi: 10.1021/ol0512886.Whitehead A, McParland JP, Hanson PR. Org Lett. 2006;8:5025–5028. doi: 10.1021/ol061756r.Thomas CD, McParland JP, Hanson PR. Eur J Org Chem. 2009:5487–5500. doi: 10.1002/ejoc.200900560.For recent examples of RCM to afford other P-heterocyclic systems, see: Schmidt B, Kunz O. Org Lett. 2013;15:4470–4473. doi: 10.1021/ol4020078.Fourgeaud P, Midrier C, Vors JP, Volle JN, Pirat JL, Virieux D. Tetrahedron. 2010;66:758–764.Kumar D, Singh N, Keshav K, Elias AJ. Inorg Chem. 2011;50:250–260. doi: 10.1021/ic101884s.Wu X, O’Brien P, Ellwood S, Secci F, Kelly B. Org Lett. 2013;15:192–195. doi: 10.1021/ol303253h.Hetherington L, Greedy B, Gouverneur V. Tetrahedron. 2000;56:2053–2060.For a recent review on general RCM that contains a section on RCM to P-heterocycles, see: Maitra S, Chegondi R, Markley JL, Hanson PR. General Ring Closing Metathesis. In: Grubbs RH, O’Leary DJ, editors. Handbook of metathesis: Application in Organic Synthesis. 2. Vol. 2. Wiley-VCH; Weinheim, Germany: 2015. pp. 1–170.
- 4.Hanson PR, Chegondi R, Nguyen J, Thomas CD, Waetzig J, Whitehead A. J Org Chem. 2011;76:4358–4370. doi: 10.1021/jo2003506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chegondi R, Tan MML, Hanson PR. J Org Chem. 2011;76:3909–3916. doi: 10.1021/jo200337v. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Venukadasula PKM, Chegondi R, Maitra S, Hanson PR. Org Lett. 2010;12:1556–1559. doi: 10.1021/ol1002913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jayasinghe S, Venukadasula PKM, Hanson PR. Org Lett. 2014;16:122–125. doi: 10.1021/ol403110p. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chegondi R, Hanson PR. Tetrahedron Lett. 2015;56:3330–3333. doi: 10.1016/j.tetlet.2015.01.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.In a manner similar to the naming system used in previous reports, we have generated a simplified set of identifiers that is a short-hand description of the IUPAC-designated names for each class of synthesized phosphate in order to emphasize the type of bicyclic structure formed after successful RCM. The proper names of each bicyclic phosphate are reported in the experimental section and corresponding supporting information. In addition, each proper name will be denoted as references throughout the manuscript. In this case, bicyclo[4.3.1]phosphates refers to “2,9,10-trioxa-1-phosphabicyclo(4.3.1)dec-4-ene 1-oxides.”
- 10.“Bicyclo[5.3.1]phosphates” refers to “2,10,11-trioxa-1-phosphabicyclo(5.3.1) undec-5-ene 1-oxides” and “2,10,11-trioxa-1-phosphabicyclo(5.3.1)undec-4-ene 1-oxides.”
- 11.“Bicyclo[7.3.1]phosphates” refers to “2,12,13-trioxa-1-phosphabicyclo(7.3.1)tridec-7-ene 1-oxides” and “2,12,13-trioxa-1-phosphabicyclo(7.3.1)tridec-6-ene 1-oxides.”
- 12.Chegondi R, Maitra S, Markley JL, Hanson PR. Chem Eur J. 2013;19:8088–8093. doi: 10.1002/chem.201300913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.The term “diastereoselective,” while seemingly unintuitive, refers to the newly formed stereogenic center at phosphorus, which forms stereoselectively based upon the inherent stereochemistry of chiral, non-racemic, C2-symmetric, 1,3-anti-diene diol utilized in the reaction; for a detailed discussion on this selectivity, see references 3a–c. Essentially, this term describes the diastereoselective RCM that results in the formation of a stereogenic phosphorus atom from RCM precursor trienes that are pseudo-C2-symmetric.
- 14.“Bicyclo[6.3.1]phosphates” refers to “2,11,12-trioxa-1-phosphabicyclo(6.3.1)dodec-5-ene 1 oxides” and “2,11,12-trioxa-1-phosphabicyclo(6.3.1)dodec-6-ene 1-oxides.”
- 15.“Bicyclo[8.3.1]phosphates” refers to “2,13,14-trioxa-1-phosphabicyclo(8.3.1)tetra-dec-8-ene 1 oxides” and “2,13,14-trioxa-1-phosphabicyclo(8.3.1)tetra-dec-7-ene 1 oxides.”
- 16.Maitra S, Markley JL, Chegondi R, Hanson PR. Tetrahedron. 2015;71:5734–5740. doi: 10.1016/j.tet.2015.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hoye TR, Promo MA. Tetrahedron Lett. 1999;40:1429–1432.Scalzullo SM, Islam RU, Morgans GL, Michael JP, van Otterlo WAL. Tetrahedron Lett. 2008;49:7403–7405.Urosa A, Marcos IS, Díez D, Padrón JM, Basabe P. J Org Chem. 2015;80:6447–6455. doi: 10.1021/acs.joc.5b00876.En route to the total synthesis of (+)-gigantecin, Hoye and coworkers observed an unexpected 11-membered RCM of a silicon-tethered intermediate to provide the corresponding monocycle with predominantly Z-olefin selectivity, see: Hoye TR, Eklov BM, Jeon J, Khoroosi M. Org Lett. 2006;8:3383–3386. doi: 10.1021/ol061383u.
- 18.The use of a removable silyl group to control the stereoselectivity of macrocyclic RCM was first published by Schreiber and Young in the following report: Wang Y, Jimenez M, Hansen AS, Raiber E-A, Schreiber S, Young DW. J Am Chem Soc. 2011;133:9196–9199. doi: 10.1021/ja202012s.
- 19.Trost BM, Ball ZT. J Am Chem Soc. 2005;127:17644–17655. doi: 10.1021/ja0528580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.The cis-/trans- descriptors in cis-12 and trans-11 refer to the relative stereochemistry between the substituents at C3 and C9 in the bicyclo[7.3.1]phosphates.
- 21.Olefin stereochemistry (Z- versus E-configuration) was determined by detailed NMR analysis of both coupling constants of olefinic protons, proton shift, and NOESY correlation of olefinic protons and/or vinylic methylene (CH2) signals. Unambiguous definition of each proton signal was necessary to arrive at meaningful conclusions, and as such, it is included in the Experimental Section of this manuscript.
- 22.All X-ray crystallographic data has been submitted to the Cambridge Crystallographic Data Centre, and the structures were assigned the following deposition numbers: 15 (1431827) and trans-33 (1431826).
- 23.Stewart IC, Ung T, Pletnev AA, Berlin JM, Grubbs RH, Schrodi Y. Org Lett. 2007;9:1589–1592. doi: 10.1021/ol0705144. [DOI] [PubMed] [Google Scholar]
- 24.Chatterjee AK, Choi TL, Sanders DP, Grubbs RH. J Am Chem Soc. 2003;125:11360–11370. doi: 10.1021/ja0214882. [DOI] [PubMed] [Google Scholar]
- 25.Diastereoselectivity was determined by NMR analysis (1H and proton-decoupled 13P NMR). The 2:1 mixture of Z- and E-isomers of cis-27 was purified to a 10:1 mixture of the major stereoisomer (Z-configured) to the minor stereoisomer (E-configured, confirmed by LC-MS) for full characterization and confirmation of the olefin configuration.
- 26.Pangborn AB, Giardello MA, Grubbs RH, Rosen RK, Timmers FJ. Organometallics. 1996;15:1518–1520. [Google Scholar]
- 27.Due to complex PH coupling, some 31P NMR for new compounds appear as a single peak with complex splitting; this multiplet collapses to a singlet in proton-decoupled 31P {1H} NMR (See Supporting Information).
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.