Abstract
Gastrinoma is a gastrin-secreting tumor that is associated with Zollinger-Ellison syndrome. The majority of cases occur in the pancreas, followed by the duodenum. Early diagnosis is difficult due to the relative rarity of the tumor and the lack of specific symptoms. In the current study, a 68-year-old female patient presented at the Second Affiliated Hospital, Zhejiang University (Hangzhou, China) due to intermittent abdominal pain and watery diarrhea. The patient was treated by surgical resection and was pathologically diagnosed with a well-differentiated pancreatic neuroendocrine tumor (gastrinoma; grade 1). No evidence of recurrence was observed during 1 year of follow-up. Furthermore, a review of the Chinese literature was performed, which analyzed an additional 17 published cases of gastrinoma. The tumor size ranged between 0.5×0.5 cm and 7.5×6.3×5.1 cm. The pancreas was the most common site of occurrence, accounting for 72% (13/18) of cases, followed by the duodenum (28%; 5/18). The most common initial symptom was abdominal pain (89%; 16/18), followed by diarrhea (56%; 10/18). In 18 cases, including the present case and 17 previous cases, the level of gastrin ranged between 137 and 1,550 pg/ml (normal range, 5–100 pg/ml). Of the 17 previous cases, 11 patients underwent surgery and 6 patients received conservative therapy due to metastasis or patient choice. Overall, gastrinoma remains a rare disease. Complete removal of the lesion is the standard curative treatment and conservative treatment is only recommended for patients unsuitable for surgery or for those with widespread metastasis.
Keywords: gastrinoma, Zollinger-Ellison syndrome, neuroendocrine tumor, annular pancreas
Introduction
Gastrinoma is a rare neuroendocrine tumor (NET), with an incidence of 0.1–15 cases per million individuals worldwide (1). A total of 0.1% of peptic ulcers and 2–5% of recurrent ulcers are caused by gastrinoma (2). Gastrinoma may also be referred to as Zollinger-Ellison syndrome, which presents as gastric hypersecretion, hyperacidity and atypical peptic ulceration, and was first reported by Zollinger and Ellison in 1955 (3). Gastrinoma is often misdiagnosed due to its relative rarity and lack of specific symptoms. Typical symptoms include abdominal pain, secretory diarrhea, esophagitis and hypercalcemia (4). Serum gastrin level is of great significance, while final diagnosis depends on pathological and immunohistochemical analysis. Surgical management remains the only curative treatment for gastrinoma (5). The current study reports the case of a 68-year-old female patient diagnosed with gastrinoma, who was treated successfully by surgical resection. Written informed consent was obtained from the patient for publication of this study. In addition, 17 cases of gastrinoma published in the Chinese literature are reviewed. The epidemiology, histopathology, imaging characteristics and clinical management of the disease are reported.
Case report
In November 2013, a 68-year-old female was admitted to the Department of Surgery at The Second Affiliated Hospital, Zhejiang University (Hangzhou, China), with intermittent abdominal pain and watery diarrhea that had been present for 1 year. The symptoms could be temporarily relieved by symptomatic treatment. No nausea, vomiting, melena or tenesmus was reported, and the patient's medical and family histories were unremarkable. Physical examination revealed mild tenderness in the epigastrium, without rebound tenderness. No evident abnormalities in thyroid function or tumor markers were identified. The patient's gastrin level was elevated (864.19 pg/ml; normal range, 25–100 pg/ml). Parathyroid hormone (PTH) and blood calcium levels were normal. Gastroscopy revealed an ulcer in the descending duodenum and biopsy identified an NET. Computed tomography (CT; LightSpeed VTC; GE Healthcare Life Sciences, Chalfont, UK) and magnetic resonance imaging (MRI; Signa HDxt 3.0T; GE Healthcare, Life Sciences) revealed no abnormalities.
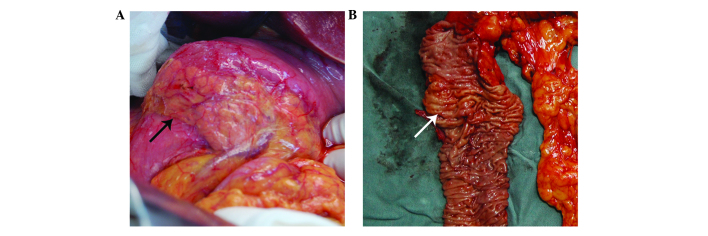
During surgery, a ring of pancreatic tissue encircling the descending portion of the duodenum was identified, a condition termed an ‘annular pancreas’ (Fig. 1A). Following the resection, a 0.7×0.7-cm thick lesion was identified proximal to the duodenal papilla (Fig. 1B). In addition, an enlarged lymph node, 1.5 cm in diameter, was identified behind the head of pancreas. Subsequently, the patient underwent a duodenopancreatectomy.
Figure 1.
Macroscopic images of the sample. (A) An annular pancreas was observed, where a ring of pancreatic tissue encircled the duodenum (black arrow). (B) A 0.7×0.7-cm thick lesion was identified proximal to the duodenal papilla (white arrow).
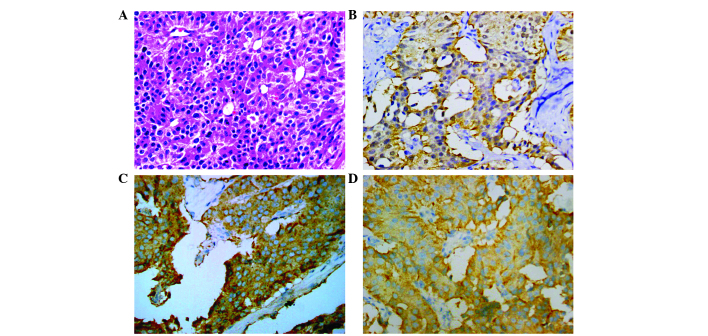
The tumor was pathologically diagnosed, using hematoxylin and esoin stainning, as a well-differentiated pancreatic NET using the World Health Organization 2010 classification system (grade 1) (6), due to the presence of round/oval nuclei and that the tumor had infiltrated the layer. Immunohistochemical analysis was performed at the Department of Pathology of The Second Affiliated Hospital of Zhejiang University, and revealed that the tumor was strongly positive for synaptophysin (Syn; +++), chromogranin A (CgA; +++), cytokeratin (CK)7 (+++), CK19 (++) and gastrin (++) (Fig. 2); positive for somatostatin (+), SRY-box 10 (+), neuron-specific enolase (+), p53 (+) and Ki-67 (+; 2%); and negative for insulin, progesterone receptor, CK20, CK34BE12 and glucagon. + indicates <20% of cells stained, ++ indicates 20–50% of cells stained and +++ indicates >50% of cells stained.
Figure 2.
Microscopic images of the tumor (magnification, ×400). (A) Hematoxylin and eosin staining revealing a trabecular pattern. Immunohistochemical staining revealing positivity for (B) gastrin (++), (C) chromogranin A (+++) and (D) synaptophysin (+++).
Following surgery, the patient recovered well and the gastrin level decreased to 32.6 pg/ml. The patient underwent follow-up for 1 year, and no evidence of recurrence was observed during this period. At present, the patient remains alive after 2 years of follow-up.
Literature review
The Chinese Biology and Medicine Database (www.sinomed.ac.cn), the Chinese periodical Database of Science and Technology (www.cqvip.com), and the China Hospital Knowledge Database (www.kns.chkd.cnki.net.) were searched for gastrinoma cases published in the previous 10 years using the search terms ‘gastrinoma’ and ‘case report’. Studies were included based on the following criteria: i) The study was a case report; ii) the final diagnosis of each case was confirmed using pathological and immunohistochemical results; and iii) the manuscript was written in the Chinese language. Case reports that did not provide sufficient data were excluded. Duplicate reports were excluded and 17 published cases, in addition to the present case, were identified (Table I) (7–21).
Table I.
Clinical data of the 18 gastrinoma cases (including the present case) reported in the Chinese literature.
| Patient no. | Author, year | Gender | Age, years | Tumor size, cm | Localization | Symptoms | Gastrin, pg/ml | Treatment | (Ref.) |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Wang et al, 2008 | M | 70 | 7.5×6.3×5.1 | Duodenum | Abdominal pain | NA | Surgery | (7) |
| 2 | Wang et al, 2010 | F | 61 | 1.8×2.1 | Pancreas | Abdominal pain and diarrhea | 1250 | Conservative therapy | (8) |
| 3 | Wang et al, 2010 | M | 38 | 1.5×2.0 | Cauda pancreatis | Abdominal pain | NA | Surgery | (8) |
| 4 | Li et al, 2006 | M | 45 | 2.0×2.0 | Cauda pancreatis | Abdominal pain and melena | 1550 | Surgery | (9) |
| 5 | Li et al, 2006 | M | 37 | 2.0×2.0 | Pancreas | Abdominal pain | NA | Surgery | (9) |
| 6 | Hou et al, 2009 | M | 61 | 6.0×5.6 | Caput pancreatis and duodenum | Abdominal pain and diarrhea | 406 | Conservative therapy | (10) |
| 7 | Zhan et al, 2012 | F | 71 | 5.0×4.0 | Caput pancreatis and duodenum | Abdominal pain and diarrhea | NA | Conservative therapy | (11) |
| 8 | Yang and Wang, 2008 | F | 56 | NA | Liver | Abdominal pain and diarrhea | 615 | Conservative therapy | (12) |
| 9 | Hou et al, 2005 | M | 25 | NA | Stomach | Abdominal pain and diarrhea | 960 | Surgery | (13) |
| 10 | Dai et al, 2010 | M | 40 | 3.1×2.0 | Pancreas | Vomit and diarrhea | 675 | Surgery | (14) |
| 11 | Zhang and Liu, 2011 | F | 48 | NA | Duodenum | Abdominal pain | 138 | Conservative therapy | (15) |
| 12 | Rajiv et al, 2009 | M | 64 | 4.0×3.0 | Cauda pancreatis | Abdominal pain and diarrhea | 1250 | Conservative therapy | (16) |
| 13 | Wang, 2008 | F | 69 | 3.5×2.8 | Cauda pancreatis | Abdominal pain and diarrhea | NA | Surgery | (17) |
| 14 | Chen and Wang, 2003 | F | 41 | 1.0×1.5 | Pancreas | Abdominal pain and diarrhea | 137 | Surgery | (18) |
| 15 | Wu and Shi, 2003 | F | 58 | 0.5×0.5 | Caput pancreatis | Diarrhea | 217 | Surgery | (19) |
| 16 | Wang, 2003 | M | 58 | 2.0×2.0 | Cauda pancreatis | Abdominal pain | NA | Surgery | (20) |
| 17 | Shen et al, 2007 | M | 64 | 5.2×4.0×3.8 | Caput pancreatis | Abdominal pain | 200 | NA | (21) |
| Present case | F | 68 | 2.0×1.0 | Duodenum | Abdominal pain and diarrhea | 864 | Surgery | – |
M, male; F, female; NA, not available.
The mean age of the 18 patients at presentation was 54.1 years (range, 25–71 years), and the male to female ratio was 5:4. The tumor sizes ranged between 0.5×0.5 cm and 7.5×6.3×5.1 cm. The pancreas was the most common site of gastrinoma occurrence, accounting for 72% (13/18) of cases, followed by the duodenum (28%; 5/18). A single case of stomach gastrinoma and a single case of liver gastrinoma were also observed. The most common initial symptom was abdominal pain (89%; 16/18), followed by diarrhea (56%; 10/18). Vomiting and melena were also reported in one case. The level of gastrin ranged between 137 and 1,550 pg/ml (normal range, 5–100 pg/ml). In the 17 previously published cases, 11 patients underwent surgery and 6 patients received conservative therapy due to the presence metastasis or patient choice.
Discussion
Gastrinoma is a relatively rare NET with an incidence of 0.1–15 cases per million individuals worldwide (1). Gastrinoma is the second most common type of NET following insulinomas (22). The tumors exhibit the abnormal secretion of gastrin and most commonly occur in the pancreas, followed by the duodenum. In the present case, the tumor was located in an annular pancreas encircling the descending portion of the duodenum. Gastrinoma may be divided into two subtypes: Sporadic gastrinoma (SG), which accounts for 75% of all gastrinomas, and multiple endocrine neoplasia type-1 (MEN-1) gastrinoma (23). MEN-1 patients usually develop hyperparathyroidism, pancreatic NETs and pituitary adenoma. Although the majority of gastrinomas grow slowly, 50–60% are malignant and have metastasized at diagnosis (24). In patients with metastatic malignant gastrinoma, the 5-year survival rate is 20–38% (24). The most common metastatic sites are the peripheral lymph nodes and liver. Pre-operative diagnosis is difficult due to the lack of specific symptoms, and the management of gastrinoma remains controversial due to the rarity of the tumor.
An annular pancreas is a congenital abnormality in which a ring of pancreatic tissue encircles the descending portion of the duodenum; this was first reported by Tiedemann in 1818 and was termed an annular pancreas by Ecker in 1862 (25). The incidence of annular pancreases is low, with a previous autopsy series indicating a prevalence of 0.005–0.015% (26). An annular pancreas is usually associated with peptic ulcers, duodenal obstruction, pancreatitis and obstructive jaundice (27).
The common clinical manifestations of gastrinoma include abdominal pain, secretory diarrhea, esophagitis and hypercalcemia (28). Abdominal pain is the most common presentation and is usually caused by peptic ulcers (4). In the present literature review, the most common initial symptom was also abdominal pain. Thus, a gastrinoma may be misdiagnosed as a peptic ulcer. Peptic ulcers caused by gastrinoma should be distinguished from cases of annular pancreas, as compression from the annulus to the duodenum may lead to the development of peptic ulcers. In cases where a patient presents with a refractory ulcer and diarrhea, a diagnosis of gastrinoma must be considered. For patients with suspected gastrinoma, the serum gastrin level should be measured. The normal level of serum gastrin is <100 pg/ml, however, in gastrinoma patients, levels may significantly exceed the upper limit. In the present literature review, the gastrin levels ranged from 137 to 1,550 pg/ml (normal range, 5–100 pg/ml). However, hypergastrinemia may also be observed in patients with atrophic gastritis or gastric outlet obstruction. Thus, analysis of gastric acid secretion should be applied for the differential diagnosis of gastrinoma (28). The patient in the present case exhibited normal PTH and blood calcium levels, and was subsequently diagnosed with SG. In addition, CgA has been used as a tumor marker for NETs, with a sensitivity of 59% and a specificity of 100% in MEN-1 (29).
The majority of gastrinomas are identified within the gastrinoma triangle, which consists of the duodenum, pancreatic head and hepatoduodenal ligament (30). The tumor should be located prior to surgery, and ultrasonography is the standard choice of diagnostic technique due to its convenience and cost-effectiveness; however, the sensitivity of the procedure is low. Thus, CT, MRI, selective angiography and somatostatin receptor scintigraphy, which exhibit sensitivities of 51, 57, 57 and 78%, respectively (31), are recommended for further evaluation. Recently, endoscopic ultrasound has been applied widely due to its high sensitivity. Furthermore, it has been suggested that intraoperative ultrasound should be routinely used to identify pancreatic lesions (32). However, the final diagnosis of gastrinoma depends on the pathological and immunohistochemical analysis. The most important feature for the diagnosis of NETs is positive immunostaining for CgA and Syn. Furthermore, gastrin immunostaining may be used to differentiate gastrinoma from other pancreatic NETs. In the present case, the tumor specimen was positive for Syn (+++), CgA (+++) and gastrin (++), with Ki-67 levels of 10%, which strongly indicated a gastrinoma.
Surgical management remains the only curative treatment for gastrinoma (5). Fraker et al (33) reported that gastrinoma patients administered conservative treatment exhibited a higher risk of liver metastasis (23%) compared with those who received surgery (3%). In the present study, of the 17 cases, 11 patients underwent surgery and 6 patients received conservative therapy due to metastasis or patient choice. Surgical resection should be performed at laparotomy rather than laparoscopically, as the primary tumor is not usually observed on pre-operative imaging studies, since the tumors are submucosal in the duodenum and frequently metastasize to the lymph nodes (34). Prior to surgery, proton pump inhibitors (PPIs) should be applied to maintain gastric acid levels at <5 mmol/h. For gastrinoma of the pancreas, tumor enucleation is recommended, while for gastrinoma of the duodenum, due to its multiple distribution, the duodenum should be lanced and the lesions resected. Notably, lymph node dissection should be performed even if no primary tumor is identified due to the possibility that the primary tumor may have occurred in the (peripancreatic) lymph nodes. Lymph nodes along the celiac trunk and hepatic ligament should also be dissected (34,35).
Conservative treatment is only recommended for patients who are unsuitable for surgery or for patients with widespread metastasis. Gastric hypersecretion may be controlled effectively using PPIs and H2-blockers (36). However, PPIs are the optimal choice due to their long duration of efficacy, which allows a dose to be administered once or twice a day. Somatostatins, such as octreotide, have also been used to inhibit the hypersecretion of gastric acid (37). However, a routine dose has not been established and randomized trials are required. Chemotherapy may be administered for patients with widespread metastasis. The first-line treatment is combined therapy with streptozotocin and 5-fluorouracil or doxorubicin (22). Controversy remains with regard to whether chemotherapy prolongs survival time (38), and radiotherapy is not generally recommended.
In conclusion, the present study reported that case of a 68-year-old female patient diagnosed with gastrinoma, who was successfully treated by surgical resection. Gastrinoma is a rare disease. Complete removal of the lesion is the main curative treatment. Conservative treatment is only recommended for patients unable to undergo surgery or those with widespread metastasis. Therefore, further study is required to evaluate alternative conservative treatments.
References
- 1.Hung PD, Schubert ML, Mihas AA. Zollinger-Ellison Syndrome. Curr Treat Options Gastroenterol. 2003;6:163–170. doi: 10.1007/s11938-003-0017-6. [DOI] [PubMed] [Google Scholar]
- 2.Andersen DK. Current diagnosis and management of Zollinger-Ellison syndrome. Ann Surg. 1989;210:685–703. doi: 10.1097/00000658-198912000-00002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zollinger RM, Ellison EH. Primary peptic ulcerations of the jejunum associated with islet cell tumors of the pancreas. 1955. CA Cancer J Clin. 1989;39:231–247. doi: 10.3322/canjclin.39.4.231. [DOI] [PubMed] [Google Scholar]
- 4.Roy PK, Venzon DJ, Shojamanesh H, Abou-Saif A, Peghini P, Doppman JL, Gibril F, Jensen RT. Zollinger-Ellison syndrome. Clinical presentation in 261 patients. Medicine (Baltimore) 2000;79:379–411. doi: 10.1097/00005792-200011000-00004. [DOI] [PubMed] [Google Scholar]
- 5.Norton JA, Fraker DL, Alexander HR, Venzon DJ, Doppman JL, Serrano J, Goebel SU, Peghini PL, Roy PK, Gibril F, Jensen RT. Surgery to cure the Zollinger-Ellison syndrome. N Engl J Med. 1999;341:635–644. doi: 10.1056/NEJM199908263410902. [DOI] [PubMed] [Google Scholar]
- 6.Rindi G, Arnold R, Bosman FT, Capella C, Klimstra DS, Klöppel G, Komminoth P, Solcia E. Nomenclature and classification of neuroendocrine neoplasms of the digestive system. In: Bosman FT, Carneiro F, Hruban RH, Theise ND, editors. WHO Classification of Tumours of the Digestiva System. 4th. Vol. 3. IARC Press; Lyon: 2010. pp. 13–14. [Google Scholar]
- 7.Wang F, Wen MR, Li Y, Wang M. A case report of gastrinoma in duodenum diagnosed by ultrasound. Lin Chuang Chao Sheng Yi Xue Za Zhi. 2008;10(475):484. (In Chinese) [Google Scholar]
- 8.Wang D, Peng L, Wang XL. Report of 2 cases of gastrinoma and literature review. Zhong Guo Pu Wai Ji Chu Yu Lin Chuang Za Zhi. 2010;17:81–82. (In Chinese) [Google Scholar]
- 9.Li FQ, Li XZ, Zeng QD. Two case reports of gastrinoma. Zhong Guo Pu Tong Wai Ke Za Zhi. 2006;15:160. (In Chinese) [Google Scholar]
- 10.Hou MH, Yan HJ, Lu SC, Ma K. Analysis of misdiagnosis of 2 cases of gastrinoma. Shan Dong Yi Yao. 2009;49:103. (In Chinese) [Google Scholar]
- 11.Zhan YG, Ji YT, Liu H. Gastrinoma with hepatic metastasis: A case report. Zhong Yi Yao Lin Chuang Za Zhi. 2012;24:766–767. (In Chinese) [Google Scholar]
- 12.Yang J, Wang Z. Gastrinoma with hepatic metastasis: A case report and research status. Lin Chuang Yi Yao Shi Jian. 2008;17:148–149. (In Chinese) [Google Scholar]
- 13.Hou XL, Qin YT, Qi S. Repeated perforation of digestive tract caused by gastrinoma: A case report. Lin Chuang Wai Ke Za Zhi. 2005;13:100. (In Chinese) [Google Scholar]
- 14.Dai JJ, Qian JQ, Ke GX, Wang WJ. Gastrinoma without peptic ulcer: A case report. Zhong Guo Shi Yong Nei Ke Za Zhi. 2010;4:369–370. 393. (In Chinese) [Google Scholar]
- 15.Zhang YY, Liu JM. Hepatic encephalopathy and death caused by gastrinoma and hepatic metastasis: One case report. Zhong Hua Nei Fen Mi Dai Xie Za Zhi. 2011;27:690–691. (In Chinese) [Google Scholar]
- 16.Rajiv PK, Wang W, Zhou ZW. Gastrinoma at pancreatic tail with hepatic metastasis: A case report. Guang Dong Yi Yao. 2009;30:890. (In Chinese) [Google Scholar]
- 17.Wang YF. A case report of gastrinoma. Zhong Guo Yi Shi Za Zhi. 2008;10:1409. 1411. (In Chinese) [Google Scholar]
- 18.Chen X, Wang ZW. Gastrinoma with hepatic metastasis: A case report. Zhong Guo Zhong Liu Lin Chuang. 2004;31:896. (In Chinese) [Google Scholar]
- 19.Wu X, Shi J, Liu H, Xu S. A case report of gastrinoma. Shi Jie Zhong Liu Za Zhi. 2003;4:332. (In Chinese) [Google Scholar]
- 20.Wang CY. A case report of gastrinoma. Han Shao Ji Bing Za Zhi. 2003;10:27–28. (In Chinese) [Google Scholar]
- 21.Shen J, Wang HY, Zhou CP, Liang BL. Pancreatic gastrinoma with hepatic metastasis. Zhong Hua Fang She Xue Za Zhi. 2007;41:436–437. (In Chinese) [Google Scholar]
- 22.Saclarides TJ. Gastrinoma. In: Millikan KW, Saclarides TJ, editors. Common Surgical Diseases: An Algorithmic Approach to Problem Solving. 1st. Springer Science and Business Media; New York, NY: 1998. pp. 135–138. [DOI] [Google Scholar]
- 23.Brandi ML, Gagel RF, Angeli A, Bilezikian JP, Beck-Peccoz P, Bordi C, Conte-Devolx B, Falchetti A, Gheri RG, Libroia A, et al. Guidelines for diagnosis and therapy of MEN type 1 and type 2. J Clin Endocrinol Metab. 2001;86:5658–5671. doi: 10.1210/jcem.86.12.8070. [DOI] [PubMed] [Google Scholar]
- 24.Fendrich V, Langer P, Waldmann J, Bartsch DK, Rothmund M. Management of sporadic and multiple endocrine neoplasia type 1 gastrinomas. Br J Surg. 2007;94:1331–1341. doi: 10.1002/bjs.5987. [DOI] [PubMed] [Google Scholar]
- 25.Kiernan PD, ReMine SG, Kiernan PC, ReMine WH. Annular pancreas: Mayo clinic experience from 1957 to 1976 with review of the literature. Arch Surg. 1980;115:46–50. doi: 10.1001/archsurg.1980.01380010038007. [DOI] [PubMed] [Google Scholar]
- 26.Alahmadi R, Almuhammadi S. Annular pancreas: A cause of gastric outlet obstruction in a 20-year-old patient. Am J Case Rep. 2014;15:437–440. doi: 10.12659/AJCR.891041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Urayama S, Kozarek R, Ball T, Brandabur J, Traverso L, Ryan J, Wechter D. Presentation and treatment of annular pancreas in an adult population. Am J Gastroenterol. 1995;90:995–999. [PubMed] [Google Scholar]
- 28.Imamura M, Nakamoto Y, Uose S, Komoto I, Awane M, Taki Y. Diagnosis of functioning pancreaticoduodenal neuroendocrine tumors. J Hepatobiliary Pancreat Sci. 2015;22:602–609. doi: 10.1002/jhbp.209. [DOI] [PubMed] [Google Scholar]
- 29.Nehar D, Lombard-Bohas C, Olivieri S, Claustrat B, Chayvialle JA, Penes MC, Sassolas G, Borson-Chazot F. Interest of Chromogranin A for diagnosis and follow-up of endocrine tumours. Clin Endocrinol (Oxf) 2004;60:644–652. doi: 10.1111/j.1365-2265.2004.02030.x. [DOI] [PubMed] [Google Scholar]
- 30.Stabile BE, Morrow DJ, Passaro E., Jr The gastrinoma triangle: Operative implications. Am J Surg. 1984;147:25–31. doi: 10.1016/0002-9610(84)90029-1. [DOI] [PubMed] [Google Scholar]
- 31.Alexander HR, Fraker DL, Norton JA, Bartlett DL, Tio L, Benjamin SB, Doppman JL, Goebel SU, Serrano J, Gibril F, Jensen RT. Prospective study of somatostatin receptor scintigraphy and its effect on operative outcome in patients with Zollinger-Ellison syndrome. Ann Surg. 1998;228:228–238. doi: 10.1097/00000658-199808000-00013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Norton JA, Alexander HR, Fraker DL, Venzon DJ, Gibril F, Jensen RT. Does the use of routine duodenotomy (DUODX) affect rate of cure, development of liver metastases, or survival in patients with Zollinger-Ellison syndrome? Ann Surg. 2004;239:617–626. doi: 10.1097/01.sla.0000124290.05524.5e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Fraker DL, Norton JA, Alexander HR, Venzon DJ, Jensen RT. Surgery in Zollinger-Ellison syndrome alters the natural history of gastrinoma. Ann Surg. 1994;220:320–330. doi: 10.1097/00000658-199409000-00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Norton JA, Jensen RT. Resolved and unresolved controversies in the surgical management of patients with Zollinger-Ellison syndrome. Ann Surg. 2004;240:757–773. doi: 10.1097/01.sla.0000143252.02142.3e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Norton JA, Alexander HR, Fraker DL, Venzon DJ, Gibril F, Jensen RT. Possible primary lymph node gastrinoma: Occurrence, natural history, and predictive factors: A prospective study. Ann Surg. 2003;237:650–659. doi: 10.1097/01.SLA.0000064375.51939.48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Metz DC, Pisegna JR, Fishbeyn VA, Benya RV, Jensen RT. Control of gastric acid hypersecretion in the management of patients with Zollinger-Ellison syndrome. World J Surg. 1993;17:468–480. doi: 10.1007/BF01655106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.de Herder WW, Lamberts SW. Clinical endocrinology and metabolism. Gut endocrine tumours. Best Pract Res Clin Endocrinol Metab. 2004;18:477–495. doi: 10.1016/j.beem.2004.08.003. [DOI] [PubMed] [Google Scholar]
- 38.Arnold R, Rinke A, Schmidt Ch, Hofbauer L. Endocrine tumours of the gastrointestinal tract: Chemotherapy. Best Pract Res Clin Gastroenterol. 2005;19:649–656. doi: 10.1016/j.bpg.2005.04.004. [DOI] [PubMed] [Google Scholar]