Abstract
Genome sequencing plays a key role in understanding the genetic diversity of Mycobacterium tuberculosis (M.tb). The genotype-specific character of M. tb contributes to tuberculosis severity and emergence of drug resistance. Strains of M. tb complex can be classified into seven lineages. The Nonthaburi (NB) genotype, belonging to the Indo-Oceanic lineage (lineage 1), has a unique spoligotype and IS6110-RFLP pattern but has not previously undergone a detailed whole genome analysis. In addition, there is not much information available on the whole genome analysis of M. tb isolates from tuberculous meningitis (TBM) patients in public databases. Isolates CSF3053, 46-5069 and 43-13838 of NB genotype were obtained from the cerebrospinal fluids of TBM Thai patients in Siriraj Hospital, Bangkok. The whole genomes were subjected to high throughput sequencing. The sequence data of each isolate were assembled into draft genome. The sequences were also aligned to reference genome, to determine genomic variations. Single nucleotide polymorphisms (SNPs) were obtained and grouped according to the functions of the genes containing them. They were compared with SNPs from 1,601 genomes, representing the seven lineages of M. tb complex, to determine the uniqueness of NB genotype. Susceptibility to first-line, second-line and other antituberculosis drugs were determined and related to the SNPs previously reported in drug-resistant related genes. The assembled genomes have an average size of 4,364,461 bp, 4,154 genes, 48 RNAs and 64 pseudogenes. A 500 base pairs deletion, which includes ppe50, was found in all isolates. RD239, specific for members of Indo Oceanic lineage, and RD147c were identified. A total of 2,202 SNPs were common to the isolates and used to classify the NB strains as members of sublineage 1.2.1. Compared with 1,601 genomes from the seven lineages of M. tb complex, mutation G2342203C was found novel to the isolates in this study. Three mutations (T28910C, C1180580T and C152178T) were found only in Thai NB isolates, including isolates from previous study. Although drug susceptibility tests indicated pan-susceptibility, non-synonymous SNPs previously reported to be associated with resistance to anti-tuberculous drugs; isoniazid, ethambutol, and ethionamide were identified in all the isolates. Non-synonymous SNPs were found in virulence genes such as the genes playing roles in apoptosis inhibition and phagosome arrest. We also report polymorphisms in essential genes, efflux pumps associated genes and genes with known epitopes. The analysis of the TBM isolates and the availability of the variations obtained will provide additional resources for global comparison of isolates from pulmonary tuberculosis and TBM. It will also contribute to the richness of genomic databases towards the prediction of antibiotic resistance, level of virulence and of origin of infection.
Keywords: Draft genome, M. tuberculosis Nonthaburi genotype, Meningitis, Genetic signatures, Thailand, Whole genome sequence
Introduction
Tuberculosis (TB) remains a global threat despite efforts targeted towards its control. With recent advances in next generation sequencing, the analysis of bacterial whole genome sequences has contributed significantly to the understanding of virulence factors and antibiotic resistance of pathogenic bacteria (Koser et al., 2013; Leopold et al., 2014). Currently, there are software tools and databases that are used for predicting bacterial genotype, lineages and drug resistance profile from mycobacterial whole genome sequence data (Benavente et al., 2015; Coll et al., 2015). Availability of more whole genome data (processed and unprocessed), especially from genotypes not currently available, will contribute immensely to the profiling of pathogens.
Although tuberculosis is a curable disease, 9.0 million new cases and 1.5 million TB deaths were recorded in 2013 (Zumla et al., 2015). This is due in part to incomplete understanding of the variations that contribute to the pathogenesis and antibiotic resistance of Mycobacterium tuberculosis. There are two broad types of clinical TB disease; pulmonary (PTB) in which the site of infection is the lung and extra-pulmonary, including the more severe tuberculous meningitis (TBM), in which the bacteria cross the blood brain barrier to get into the cerebrospinal fluid (CSF) of the patient. The morbidity and mortality rate of TBM is higher than PTB (Thwaites, van Toorn & Schoeman, 2013). The genotype of the infecting mycobacterium has been shown to be one of the factors that contribute to the severity of the disease and can play a role in emergence of drug resistance, susceptibility to TBM, host response and in transmissibility (Ford et al., 2013; Lopez et al., 2003; Nahid et al., 2010; Thwaites et al., 2008). However the genetic factors that determine the association of different lineages of mycobacteria with different level of disease severity remain largely unknown.
There have been controversies in associating specific genotypes with morbidity or mortality from TB. A study in Thailand associated the modern Beijing genotype with a more severe disease progression when compared with other lineages (Faksri et al., 2011). However, in a study conducted in HIV patients in Vietnam, modern Beijing genotype had lower mortality rates than those infected with other lineages (Tho et al., 2012). Comparing strains isolated from TBM across genotypes on a whole genome scale may provide better understanding of factors that contribute to the severity of the disease.
IS6110 based restriction fragment length polymorphism (RFLP) is an internationally recognized method for genotyping mycobacteria (Thierry et al., 1990; van Embden et al., 1993). Nonthaburi strains of M. tuberculosis were first identified in Thailand by its IS6110-RFLP patterns, usually containing 9-14 bands. Subsequent spoligotyping revealed that the Nonthaburi type has a spoligotype octal code 674000003413771 specifying the East-Asian India 2 Nonthaburi (EAI2-Nonthaburi) genotype (Palittapongarnpim et al., 1997). It has been reported in lower percentages from many countries such as the Netherlands, Australia, USA, Sweden, Saudi Arabia, Tunisia, and Taiwan. However, the origin of the isolates is likely to be South East Asia, as more isolates are from countries such as Indonesia, Laos PDR, Vietnam, Cambodia, Philippines and Thailand (Demay et al., 2012).
As of this date, only relatively little information is available on the genetic characteristics of the Nonthaburi strains. Three Nonthaburi strains were isolated from the CSF samples of TBM patients at Siriraj Hospital, Mahidol University, Thailand. For a deeper understanding of the characteristics of these isolates, genome-wide scale analysis and drug susceptibility pattern to anti-tuberculosis drugs were performed and compared to the reference strain M. tuberculosis H37Rv (NC_000962.3). The single nucleotide polymorphism (SNPs) common to the isolates were compared with SNPs from 1,601 genomes from the 7 different lineages and various sublineages of M. tuberculosis complex (MTBC). The whole genome sequence of the isolates were assembled into draft genomes, annotated and have been deposited into NCBI database for public access. Prior to our study, there was no complete or draft genome belonging to the Nonthaburi genotype of M. tuberculosis in the database.
Methods
Selection of strains
Three isolates, CSF3053, 46-5069 and 43-13838, identified to belong to Nonthaburi genotype by IS6110-RFLP, were selected from the stock of samples collected from the CSF of TBM patients at the Drug Resistant Tuberculosis Research Fund Laboratory, Department of Microbiology, Faculty of Medicine Siriraj Hospital, Mahidol University, Thailand.
Genomic DNA extraction
Stock culture of selected strains, stored at −70 °C in MH79 broth containing 15% glycerol, were subcultured on Loewenstein-Jensen medium and incubated for 4 weeks at 37 °C. DNA extraction was carried out using cetyltrimethylammonium bromide (CTAB)-lysozyme enzymatic method as earlier described (Larsen et al., 2007).
Spoligotyping
Spacer oligonucleotide typing, a polymerase chain reaction (PCR) based method used in typing M. tuberculosis was performed following the methods earlier described (Gori et al., 2005).
Whole genome sequencing and analysis
Genomic DNA samples isolated from the three isolates were sequenced at Macrogen Inc., Seoul, South Korea on the HiSeq 2000 platform with insert size of 300 bp (Illumina, San Diego, CA, USA) yielding 100 bp paired end reads. The qualities of the sequences were assessed with FastQC software (www.bioinformatics.babraham.ac.uk/projects/fastqc) to determine the parameters used for trimming. Bases with quality of less than 5, reads with average of quality less than 20 for every four bases, and reads with lengths that are less than 45 bases were discarded using Trimmomatic software (Bolger, Lohse & Usadel, 2014) (version 0.33). The trimmed sequences were aligned to the reference strain M. tuberculosis H37Rv (NC_000962.3) using the short reads aligner, Bowtie2 (version 2.2.0) (Langmead & Salzberg, 2012). The genomic coverage was estimated using Bedtools (version 2.18) (Quinlan & Hall, 2010). The fold coverage is estimated as the number of reads supporting a particular nucleotide position on the genome. Variant calling was performed on the aligned sequences using the Genome Analysis Tool Kit (GATK) (version 3.3) haplotype caller (McKenna et al., 2010) with minimum calling confidence threshold set at phred score 30. Point allelic variation at any position within the genome when compared with the reference H37Rv genome (NC_000962.3) is considered a single nucleotide polymorphism (SNP).
SnpEFF (Cingolani et al., 2012) (version 4.0) software was used to annotate the SNPs. The SNPs were filtered using standard hard filtering parameters according to GATK Best Practices Recommendations (DePristo et al., 2011; Van der Auwera et al., 2013). Variants with QualByDepth <2.0, FisherStrand >60, RMSMapping quality <40, MappingQualityRankSumTest <−12.5 and ReadPosRankSumTest <−8 were filtered. All SNPs were confirmed using Integrated Genomic Viewer (IGV) (James et al., 2011) (version 2.0). The SNPs were further grouped according to the functions of the genes in which they were found in the genome when compared to the reference genome H37Rv (NC_000962.3). We evaluated SNPs in groups of genes considered to be essential, drug resistance related, virulence related, contain known epitopes and associated with efflux pumps.
The Whole Genome Shotgun project has been deposited at DDBJ/EMBL/GenBank under the accession numbers LGCH00000000, LGCG00000000and LGCF00000000. The versions described in this paper are LGCH01000000, LGCG01000000and LGCF01000000for CSF3053, 46-5069 and 43-13838 respectively. The raw sequences have been deposited to the short read archive (SRA) of NCBI under accession numbers SRX1094547, SRX1094546and SRX1094545for isolates CSF3053, 46-5069 and 43-13838 respectively.
Determination of principle genetic group, lineage and sequence type
Nucleotide alleles at positions 7585 and 2154724 were investigated to determine the principal genetic group of the isolates as earlier defined (Sreevatsan et al., 1997). To determine the lineage of the isolates, SNPs specific to different lineages as earlier reported (Coll et al., 2014b) were investigated.
Draft genome assembly
The paired-end raw reads of the isolates were assembled into draft genomes by using the de novo assembly algorithm of CLC Genomics Workbench (version 7.5) which works by using a de Bruijn graph (http://www.clcbio.com/). The minimum contig output was set at 200 bp long. Annotation of the draft genome was performed by Rapid Annotation using Subsystem Technology (RAST) (http://www.nmpdr.org/) and by NCBI Prokaryotic Genome Annotation Pipeline (PGAP) (http://www.ncbi.nlm.nih.gov/genome/annotation_prok/).
Comparison of Nonthaburi isolates with isolates from other lineages
The SNPs that are common to the three isolates were compared with 92,000 SNPs from 1,601 genomes of MTBC previously reported (Coll et al., 2014a) (http://pathogenseq.lshtm.ac.uk/phytblive/index.php). These include 121, 390, 189, 856, 17, 11, and 6 genomes from lineages 1, 2, 3, 4, 5, 6, and 7 respectively. Eleven samples from M. bovis were also included.
Large sequence polymorphism determination
Regions of differences when compared with reference strain H37Rv (NC_000926.3) were determined by using the indel and structural variants determination tool of CLC Genomics Workbench (version 7.5) (http://www.clcbio.com) and Bedtools (version 2.18) (Quinlan & Hall, 2010). The regions of deletions were confirmed with PCR using primers CF (CATCCGCACCGAACCTGTAA) and CR (AACCGTTCACGACAAGCAAC), AF (GCCCAACCTGATTGGTTTCG) and AR (CAAACGCTCGCCATGATCTC), BF (TCGACTGCCATACAACCTGC) and BR (ACTTCCGGTGGTAACAGTGC) respectively for RD239, RD147c and newly identified deletion of 500 bp between 3501224-3501724 (M. tuberculosis H37Rv (NC_000962.3 genome numbering). The reactions were performed with initial denaturation at 94 °C and 30 cycles of denaturation for 1 min, annealing of primers at 60 °C for 1 min and extension with platinum Taq DNA polymerase for 1 min at 68 °C. Final extensions were performed at 68 °C for 10 min. The reactions were performed as recommended by the manufacturer of the DNA polymerase.
Drug susceptibility testing
The susceptibility of the isolates to first line drugs and other second-line anti-tuberculosis drugs was investigated using the standard agar proportion method (Larsen et al., 2007). The drug concentrations used in the test comprise 0.2 mg/l isoniazid, 1.0 mg/l rifampicin, 2.0 mg/l streptomycin, 5.0 mg/l ethambutol, 1.0 mg/l linezolid, 6.0 mg/l amikacin, 5.0 mg/l ethionamide, 2.0 mg/l paraaminosalycic acid, 2.0 mg/ml ofloxacin, 2.0 mg/l moxifloxacin, 2.0 mg/l gatifloxacin, 1.0 mg/ml sitafloxacin, 6.0 mg/l kanamycin, 2.0 mg/l ciprofloxacin, 2.0 mg/l levofloxacin, and 3.0 mg/l clarithromycin. Growth equal to or more than 1% on drug containing media compared to drug free media was recorded as drug resistance. The phenotypic drug testing was performed on the initial isolates from the patients and repeated on the stock cultures.
Ethical approval
The study was approved by the Institutional review board (IRB) of Faculty of Medicine Siriraj Hospital, Mahidol University SiEC No. 152/2549.
Results and Discussion
For the three isolates CSF3053, 46-5069 and 43-13838, an average of 99.1% of raw reads mapped to the reference genome. On the average, 99.8% of the reference was covered to at least 1-fold coverage. The depth across all the positions covered by the reads was about 1,056-fold on the average (Table 1).
Table 1. Statistics of whole genome sequencing, genome assembly and annotation.
Gross statistics of the whole genome sequence data, mapping of reads, assembly of draft genome and annotation for isolates CSF-3053, 46-5069 and 43-13838. Length of reference genome (M. tuberculosis H37Rv, NC_000962.3) is 4,411,532 base pairs.
| Isolate | Total reads | % of reads mapped to reference | % of Reference covered | Number of contigs | N50 | Fold coverage of positions in the genome | GC content (%) | Number of predicted Genes | No. of predicted RNA genes | No. of predicted pseudo genes |
|---|---|---|---|---|---|---|---|---|---|---|
| CSF-3053 | 50,004,564 | 99.96 | 99.78 | 159 | 69,028 | 1329.0 | 65.5 | 4153 | 48 | 62 |
| 46-5069 | 44,478,206 | 98.67 | 99.82 | 173 | 63,852 | 920 | 65.5 | 4159 | 48 | 63 |
| 43-13838 | 40,767,970 | 98.69 | 99.80 | 177 | 63,019 | 920 | 65.5 | 4150 | 48 | 67 |
Notes.
- GC
- guanine/cytocine
Genome assembly
The sequences of the isolates were assembled and annotated as described in Methods. 159 contigs with N50 of 69,028, 173 contigs with N50 of 63,852, and 177 contigs with N50 of 63,019 contigs were obtained for CSF3053, 43-5069 and 46-13838 respectively. All isolates have 65.5 % guanine/cytosine (GC) content, typical of mycobacteria. The draft genomes have an average size of 4,364,461 bp, 4,154 genes, 48 rRNAs and 64 pseudogenes. Details of the assembly and annotation are shown in Table 1.
Single nucleotide polymorphisms
Point allelic variations at any position within the genome when compared with the reference H37Rv genome (NC_000962.3) were investigated.
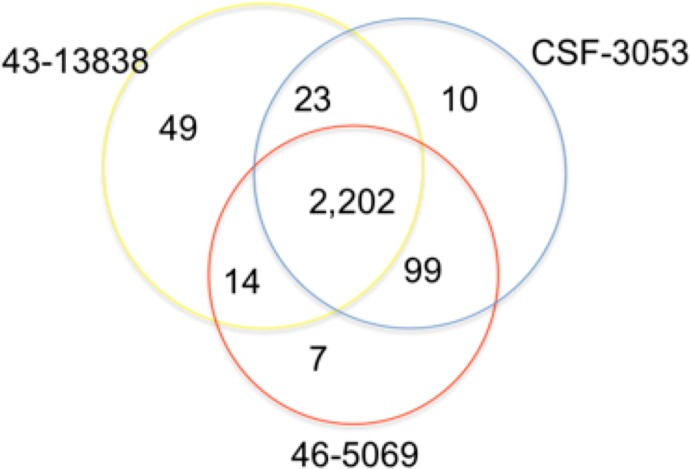
In total, 2,202 positions were found to have similar allelic changes (SNPs) in all isolates as shown in Fig. 1. 1,963 are in coding regions (754 synonymous, 1209 (61.6%) non synonymous) and 239 are intergenic. In this study, CSF3053, 46-5069 and 43-13838 have 10, 7 and 49 unique SNPs respectively. 43-13838 and CSF3053 have 23 SNPs in common, CSF3053 and 46-5069 have 99 SNPs in common, while 43-13838 and 46-5069 have 7 SNPs in common. Using the SNPs, the isolates were found to belong to lineage 1 with the presence of allele C/A and G/C at positions 2154724 and 7585 resulting in katG R463L and gyrA S95T respectively (Sreevatsan et al., 1997). Using a recently developed SNP barcode (Coll et al., 2014a), the isolates were found to be specific to Indo Oceanic lineage 1.2.1, with nucleotide changes G/A at position 615938, C/A at position 3479545, G/C at position 4244420 and G/C at position 9260.
Figure 1. Distribution of single nucleotide polymorphisms in isolates CSF3053, 46-5069 and 43-13838.
Venn diagram showing the distribution of the single nucleotide polymorphisms (SNPs) observed in isolates CSF-3053 (blue), 46-5069 (red) and 43-13838 (yellow). CSF3053, 46-5069 and 43-13838 have 10, 7 and 49 unique SNPs respectively. 43-13838 and CSF3053 have 23 SNPs in common, CSF3053 and 46-5069 have 99 SNPs in common, while 43-13838 and 46-5069 have 14 SNPs in common. 2,202 SNPs are common to all isolates.
The 2,202 SNPs that were found to be common to the isolates in this study were compared with 92,000 SNPs from 1,601 genomes of MTBC that were analyzed previously. These include 121, 390, 189, 856, 17, 11, and 6 genomes from lineages 1, 2, 3, 4, 5, 6, and 7, respectively. Eleven samples from M. bovis were also included (Coll et al., 2014a). The common SNPs were used to position the strains on a phylogenetic tree compared to other strains and lineages of MTBC as shown in Fig. S1. Nucleotide change G/C at position 2342203 was found only in the isolates in this study when compared with the 1,601 MTBC genomes. There is evidence from macrophage systems that strain-to-strain variability affects phenotypic outcomes (McEvoy et al., 2012). Phylogeographic strain variation may therefore have considerable effect on the development of new diagnostic tools, vaccines and drugs.
SNP C/T at position 3378828 was reported to be unique to members of lineage 1 (Coll et al., 2014a). Although this SNP was found in many genomes belonging to lineage 1, we found out that it was absent in the three isolates in this study and in 6 other Nonthaburi isolates from Thailand and the Netherlands used in previous studies which are grouped under lineage 1. This indicates that the allele change at this position may be specific only to a sub-branch of lineage 1. Synonymous SNP T/C at position 28910, non-synonymous SNP C/T at position 152178 resulting in Thr344Ile in pepA gene and intergenic SNP C/T at position 1180580 were found only in Nonthaburi isolates from Thailand. They were not found in any genome belonging to lineages 2, 3, 4, 5, 6 and 7. Within lineage 1, these SNPs were found only in Thai Nonthaburi isolates, from previous study (Coll et al., 2014a), and the isolates in this study. They were however absent in the Nonthaburi genotype isolates from the Netherlands. pepA gene is a probable serine protease with the exact function unknown. It is in the intermediary metabolism and respiration functional category. Its mRNA was found to be upregulated after 96 h of starvation (Betts et al., 2002), suggesting its role in the adaptation of mycobacteria to extreme conditions. The association of the SNPs at these positions with Thailand warrants further investigation.
Large sequence polymorphism
Region of difference RD239 that is specific to lineage 1 of MTBC and previously reported RD147c, not specific to lineage 1, were found in all the three isolates. In addition, a region of deletion of 500 bp between 3501224-3501724 (M. tuberculosis H37Rv (NC_000962.3 genome numbering) comprising Rv3135 (ppe50), was observed in all isolates. The details of the deletions as well as the affected open reading frames are shown in Table 2. The deletions were confirmed with PCR (see Figs. S2, S3 and S4). The PE-PPE protein class, while not well characterized, represents the third most abundant category of mycobacterial proteins and showed the most consistent expression during infection (Kruh et al., 2010). Although PPE50 has a yet unknown function, it was listed among promising therapeutic target in tuberculosis treatment based on its expression, and homology to human and other microbial proteins (Raman, Yeturu & Chandra, 2008). The deletion of this gene may be a means of evading recognition by the host immune system.
Table 2. Regions of deletion and affected open reading frames found in isolates CSF-3053, 46-5069 and 43-13838.
All regions were confirmed by PCR reaction as described in methods.
| Region in reference genome (H37Rv, NC_000962.3) | Length | Region of difference | Open reading frame (ORF) affected |
|---|---|---|---|
| 1718912–1721213 | 2302 | RD147c | Rv1526c |
| Rv1525 (wbbL2) | |||
| Rv1526c | |||
| 3501225–3501723 | 499 | This study | Rv3135 |
| 4092082–4092921 | 840 | RD239 | Rv3651 |
Deletions have been shown to have a wide range of effects on M. tuberculosis including association with an increased probability of transmission (Tsolaki et al., 2004).
Polymorphisms in drug resistance associated genes
Despite being isolated from patients with severe form of tuberculosis, drug susceptibility tests results show that the three isolates are susceptible to first line drugs; isoniazid, rifampicin, ethambutol and streptomycin, and to quinolones: ciprofloxacin, ofloxacin, gatifloxacin, moxifloxacin, levofloxacin, and sitafloxacin. They were also found to be susceptible to linezolid, amikacin, ethionamide, paraaminosalicylic acid, kanamycin and clarithromycin.
However, 37 SNPs were found in drug-resistant related genes reported in TBdream database and other earlier published reports (Sandgren et al., 2009). Nineteen are synonymous while 18 are non synonymous. Non synonymous mutations Gly312Ser of kasA gene and Ile73Thr in efpA were previously reported to be associated with isoniazid resistance (Mdluli et al., 1998; Ramaswamy et al., 2003), but were found in our isolates. Association between these mutations and resistance to isoniazid needs to be confirmed. iniA gene and Rv1592c were reported to be associated with tolerance to isoniazid (Colangeli et al., 2005; Ramaswamy et al., 2003). In our analysis, mutations His481Gln in iniA gene and Ile322Val in Rv1592c were found. These positions may not be associated with the supposed roles of these genes in isoniazid resistance.
Polymorphism exists at position 237 of nudC in M. tuberculosis isolates (Wang et al., 2011). In particular, the amino acid change Gln237Pro in nudC is found in the Indo Oceanic and West African lineages. It was demonstrated to prevent dimer formation and results in the loss of activity of the enzyme. It was also shown to degrade the active forms of isoniazid and ethionamide (Wang et al., 2011). We however found this codon change in all isolates in this study. This suggests the non-involvement of the amino acid change at this position in resistance to both drugs.
Mutations Cys110Tyr in embR, Thr270Ile and Asn394Asp in embC, Pro913Ser in embA and Glu378Ala in embB, were previously reported to be involved in ethambutol resistance (Ramaswamy et al., 2000; Srivastava et al., 2009). However, these mutations were found in this study. Mutation Ser257Pro in rmlD was suspected to be involved in isoniazid and ethambutol resistance (Ramaswamy et al., 2000). This was however found in all isolates considered in this study. Mutations Glu21Gln in gyrA, Ile322Val in Rv1592c, Arg463Leu in katG, and Arg93Leu in cycA were found to be common to the isolates in this study. They have also been reported to be common to pan-susceptible and drug-resistant M. tuberculosis sequence type 10 Beijing isolates (Regmi et al., 2015). Our results confirm that these mutations are polymorphic rather than being involved in drug resistance. The details of the synonymous and non-synonymous SNPs found in drug-resistant related genes and the predicted protein variation effects are shown in Table 3.
Table 3. Common SNPs found in drug resistance related genes in isolates CSF-3053, 46-5069 and 43-13838.
The reference genome positions, nucleotide change, amino acid change and effect of single nucleotide polymorphisms in drug resistance related genes that are common to isolates CSF3053, 46-5069 and 43-13838. The protein variation was determined by Protein Variation Effect Analyzer (PROVEAN), a web based protein variation analysis tool (Choi et al., 2012).
| Position in reference genome (H37Rv, NC_000962.3) | Nucleotide change | Amino acid change | Protein variation effect | Gene | Associated drug | References |
|---|---|---|---|---|---|---|
| 6112 | G>C | Met291Ile | Deleterious | gyrB | Quinolones | Guillemin, Jarlier & Cambau (1998) |
| 7362 | G>C | Glu21Gln | Neutral | gyrA | Quinolones | Guillemin, Jarlier & Cambau (1998) |
| 7585 | G>C | Ser95Thr | Neutral | gyrA | Quinolones | Guillemin, Jarlier & Cambau (1998) and Kapur et al. (1995) |
| 8452 | C>T | Ala384Val | Deleterious | gyrA | Quinolones | Guillemin, Jarlier & Cambau (1998) |
| 9143 | T>C | Ile614Ile | gyrA | Quinolones | Guillemin, Jarlier & Cambau (1998) | |
| 9260 | G>C | Leu653Leu | gyrA | Quinolones | Guillemin, Jarlier & Cambau (1998) | |
| 9304 | G>A | Gly668Asp (N) | Neutral | gyrA | Quinolones | Guillemin, Jarlier & Cambau (1998) |
| 412280 | T>G | His481Gln | Neutral | iniA | Ethambutol | Ramaswamy et al. (2003) |
| 575368 | T>C | Asp7Asp | Rv0486 | Isoniazid/Ethionamide | Projahn et al. (2011) | |
| 763031 | T>C | Ala1081Ala | rpoB | Rifampicin | Taniguchi et al. (1996) | |
| 763531 | G>C | Pro54Pro | rpoC | Rifampicin | Comas et al. (2012) | |
| 763884 | C>T | Ala172Val | Neutral | rpoC | Rifampicin | Comas et al. (2012) |
| 763886 | C>A | Arg173Arg | rpoC | Rifampicin | Comas et al. (2012) | |
| 1406312 | A>G | His343His | Rv1258c | Streptomycin | Siddiqi et al. (2004) | |
| 1417019 | C>T | Cys110Tyr | Deleterious | embR | Ethambutol | Ramaswamy et al. (2000) |
| 1674162 | C>T | Gly241Gly | fabG1 | Isoniazid | Lavender et al. (2005) | |
| 1792777 | T>C | Ile322Val | Neutral | Rv1592c | Isoniazid | Ramaswamy et al. (2003) |
| 1792778 | T>C | Glu321Glu | Rv1592c | Isoniazid | Ramaswamy et al. (2003) | |
| 2154724 | C>A | Arg463Leu | Neutral | katG | Isoniazid | Heym et al. (1995) |
| 2518132 | C>T | Thr6Thr | kasA | Isoniazid | Lee et al. (1999) | |
| 2519048 | G>A | Gly312Ser | Neutral | kasA | Isoniazid | Lee et al. (1999) |
| 2521342 | T>C | Asp200Asp | accD6 | Isoniazid | Ramaswamy et al. (2003) | |
| 3154414 | A>G | Ile73Thr | Neutral | efpA | Isoniazid | Ramaswamy et al. (2003) |
| 3571834 | T>G | Gln237Pro | Neutral | nudC | Isoniazid/Ethionamide | Wang et al. (2011) |
| 3647041 | A>G | Ser257Pro | Neutral | rmlD | Ethambutol | Ramaswamy et al. (2000) |
| 3647591 | A>G | Asn73Asn | rmlD | Ethambutol | Ramaswamy et al. (2000) | |
| 4049254 | G>A | Leu243Leu | folP1 | Para-aminosalicylic acid | Mathys et al. (2009) | |
| 4240671 | C>T | Thr270Ile | Neutral | embC | Ethambutol | Ramaswamy et al. (2000) |
| 4241042 | A>G | Asn394Asp | Deleterious | embC | Ethambutol | Ramaswamy et al. (2000) |
| 4242643 | C>T | Arg927Arg | embC | Ethambutol | Ramaswamy et al. (2000) | |
| 4243580 | G>A | Val116Val | embA | Ethambutol | Telenti et al. (1997) | |
| 4244420 | G>C | Val396Val | embA | Ethambutol | Telenti et al. (1997) | |
| 4245969 | C>T | Pro913Ser | Deleterious | embA | Ethambutol | Ramaswamy et al. (2000) and Telenti et al. (1997) |
| 4247578 | G>A | Leu355Leu | embB | Ethambutol | Telenti et al. (1997) | |
| 4247646 | A>C | Glu378Ala | Neutral | embB | Ethambutol | Telenti et al. (1997) |
| 4407588 | T>C | Ala205Ala | rsmG | Streptomycin | Okamoto et al. (2007) | |
| 4407873 | C>A | Val110Val | rsmG | Streptomycin | Okamoto et al. (2007) |
Polymorphisms in virulence genes, efflux pump related genes, and essential genes
Oftentimes, mutations provide selective advantage to an organism in a particular environment. Some non-synonymous mutations in rpoC gene have been shown to result in higher competitiveness in vitro and have higher fitness in vivo evidenced by their prevalence across patient populations (Comas et al., 2012). In this study, we found Ala172Val mutation in rpoC gene in all isolates.
We also sought to determine polymorphisms in genes that play important roles in the survival and pathogenesis of M. tuberculosis. Of particular interest are the genes that are involved in the evasion of the host immune system. SNPs in 37 mycobacteria virulence related genes were found to be common to the isolates. Twenty nine of the SNPs are non-synonymous. Polyketide synthases (PKs) are group of genes involved in the synthesis of polyketides which are structurally complex compounds produced by organisms for survival advantage. Some mycobacteria PKs genes such as pks15, pks1, pks10, pks12, pks5, and pks7 are known to be involved in virulence (Reed et al., 2004; Rousseau et al., 2003; Sirakova et al., 2003; Tsenova et al., 2005). Insertion of 7 base pairs was found in pks15/1 junction in all isolates. The presence of the 7 base pair insertion leads to a frame shift that results in the loss of stop codon of pks15. This results in a continuous transcription of pks15 and pks1. This was previously associated with the more virulent phenotype of the modern Beijing family, but such claim has since been refuted as it can be found across the seven lineages. The implication of the insertion needs further experiments to understand. Two mutations Ile474Met and Thr604Ala were found in nuoG gene. nuoG is a probable NADH dehydrogenase, reported to be involved in apoptosis inhibition (Velmurugan et al., 2007). Mutation Arg463Leu was found in katG, a gene previously implicated in inhibiting antimicrobial effectors of the macrophage (Ng et al., 2004). Protein kinases such as pknD and pknG are important virulent factors of M. tuberculosis. pknD has been reported to play a role in the infection of the host’s central nervous system by M. tuberculosis (Be, Bishai & Jain, 2012; Cowley et al., 2004). Gln472Pro mutation in pknD was found in all isolates. virS is a transcription regulator that belongs to AraC family. Its attenuation in a mouse model resulted in an increased animal survival (Gupta, Jain & Tyagi, 1999; Singh et al., 2003). We found mutation Leu316Arg in this gene in all isolates.
Stop codon was gained after Arg305 in PStA1, an inorganic phosphate ABC transporter. Stop codon was however lost in Rv1504. The stop codon was replaced with glutamine as codon 200. Rv1504 and PstA1 were reported to be involved in the adaptation and survival of mycobacteria in macrophages (Brodin et al., 2010; Rengarajan, Bloom & Rubin, 2005).
Non-synonymous and synonymous SNPs were found in other genes involved in various other functions related to virulence such as synthesis of complex and simple lipids, cell wall proteins, lipoproteins, cholesterol metabolism, secretion systems, protein kinases, metal transporter proteins, two component systems and other proteins of unknown functions (Table S1).
Efflux pumps play roles in drug resistance, cell physiology, detoxification and virulence of M. tuberculosis (Nikaido, 2009). Ten synonymous SNPs and 15 non-synonymous SNPs were found in efflux pump related genes. One stop codon was gained by Rv2994, a predicted transmembrane protein involved in efflux system (Table S2).
Twenty eight SNPs were observed in genes with known epitopes, 11 are synonymous while 17 are non-synonymous (Table S3).
In addition, 316 SNPs were found in essential genes, 135 are synonymous, 181 are non-synonymous. A start codon was lost in pabB gene. pabB is a cell membrane associated gene that encodes para-aminobenzoate synthetase component-I involved in the biosynthesis of p-aminobenzoate, a precursor of folate biosynthesis (Sassetti, Boyd & Rubin, 2003; Zheng et al., 2008). The details of the position, nucleotide change, amino acid change and the genes involved are presented in Table S4.
The association of the SNPs or deletions reported in this study to TBM needs further investigations. This can be done by comparing them with variations from PTB cases, to determine exclusive associations with TBM. Furthermore, The involvement of the reported allelic changes in the functions of the various genes from which they were found can be verified by site directed mutagenesis in laboratory strains of M. tuberculosis, and subsequent animal experiments.
Conclusion
Genetic factors that contribute to the ability of infecting mycobacteria in causing TBM remain largely unknown. We have presented a detailed analysis of the polymorphism existing in the genome of Nonthaburi isolates from TBM patients, when compared to reference strain M. tuberculosis H37Rv (NC_000962.3). The polymorphisms were compared to 1,601 genomes representing the members of the 7 MTBC lineages. Uniqueness of certain SNPs to certain genotypes, countries or region such as found in this study may be useful epidemiologically to determine the origin of an infection and potential level of disease severity. We have also presented the first draft genomes of M. tuberculosis Nonthaburi genotype.
Many studies have reported the SNPs playing roles in drug resistance in many drug-resistant related genes. These have majorly formed the basis for the development of some databases. It is equally important to report polymorphisms found in these genes from drug susceptible strains so that SNPs that are not involved in resistance to drugs but present in the drug resistance related genes could be filtered out in the process of predicting drug resistance. Our results will also form a basis for comparison with other genotypes of mycobacteria isolated from the CSF of TBM or sputum of PTB patients in order to identify potential factors contributing to TBM.
Supplemental Information
SNPs common to isolates CSF-3053, 46-5069 and 43-13838 compared to 92,000 SNPs from 1,601 genomes of M. tuberculosis complex members (Coll et al., 2014a; Coll et al., 2014b) were used to position the isolates as belonging to subineage 1.2.1.
Figure 2: PCR products using primers CF: GCCCAACCTGATTGGTTTCG and CR: CAAACGCTCGCCATGATCTC for RD239 Primers were designed to cover region 4092041-4092947. Expected size is 907 bp Lane 1: 1 kb DNA plus ladder Lane 2: M. tuberculosis (H37Rv, NC_000962.3) Lane 3: CSF3053 Lane 4: 46-5069 Lane 5: 43-13838. Figure 3: PCR products using primers AF: GCCCAACCTGATTGGTTTCG and AR: CAAACGCTCGCCATGATCTC for RD147c Primers were designed to cover region 1718833-1721268. Expected sixe is 2436 Lane 1: 1 kb DNA plus ladder Lane 2: M. tuberculosis (H37Rv NC_000962.3) Lane 3: CSF3053 Lane 4: 46-5069 Lane 5: 43-13838. Figure 4: PCR products using primers BF: GCCCAACCTGATTGGTTTCG and BR: CAAACGCTCGCCATGATCTC for 500 bp deletion. Primers were designed to cover region 3501124-3501822. Expected size is 699 bp Lane 1: 1 kb DNA plus ladder Lane 2: M. tuberculosis (H37Rv NC_000962.3) Lane 3: CSF3053 Lane 4: 46-5069 Lane 5: 43-13838.
Table S1: The positions, nucleotide change, amino acid change and effect of single nucleotide polymorphisms in virulence genes that are common to isolates CSF3053, 46-5069 and 43-13838. The protein variation was determined by Protein Variation Effect Analyzer (PROVEAN), a web based protein variation analysis tool (Choi et al., 2012). Table S2: The positions, nucleotide change, amino acid change and effect of single nucleotide polymorphisms in efflux pump related genes that are common to isolates CSF3053, 46-5069 and 43-13838. The protein variation was determined by Protein Variation Effect Analyzer (PROVEAN), a web based protein variation analysis tool (Choi et al., 2012).
The positions, nucleotide change , amino acid change and effect of single nucleotide polymorphisms in genes with known epitopes that are common to isolates CSF3053, 46-5069 and 43-13838. The protein variation was determined by Protein Variation Effect Analyzer (PROVEAN), a web based protein variation analysis tool (Choi et al., 2012).
The reference genome positions, nucleotide change, amino acid change and effect of single nucleotide polymorphisms in essential genes that are common to isolates CSF3053, 46-5069 and 43-13838. The protein variation was determined by Protein Variation Effect Analyzer (PROVEAN). A web based protein variation analysis tool (Choi et al., 2012).
Funding Statement
The financial support for the study are from Mahidol University as postdoctoral scholarship to BOC via AC and some material support from JST/NSTDA grant No. P-12-01777. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Additional Information and Declarations
Competing Interests
The authors declare there are no competing interests.
Author Contributions
Olabisi Oluwabukola Coker conceived and designed the experiments, performed the experiments, analyzed the data, wrote the paper, prepared figures and/or tables, reviewed drafts of the paper.
Angkana Chaiprasert conceived and designed the experiments, analyzed the data, contributed reagents/materials/analysis tools, wrote the paper, reviewed drafts of the paper.
Chumpol Ngamphiw performed the experiments, analyzed the data, reviewed drafts of the paper.
Sissades Tongsima analyzed the data, reviewed drafts of the paper.
Sanjib Mani Regmi performed the experiments, reviewed drafts of the paper.
Taane G. Clark reviewed drafts of the paper, suggested the position of NB in phylogenetic tree.
Rick Twee Hee Ong, Yik-Ying Teo and Therdsak Prammananan contributed reagents/materials/analysis tools, reviewed drafts of the paper.
Prasit Palittapongarnpim reviewed drafts of the paper.
Ethics
The following information was supplied relating to ethical approvals (i.e., approving body and any reference numbers):
InstitutionL Review Board (IRB) of Faculty of Medicine Siriraj Hospital, Mahidol University SiEC No. 152/2549.
DNA Deposition
The following information was supplied regarding the deposition of DNA sequences:
DDBJ/EMBL/GenBank with accession numbers LGCH01000000, LGCG01000000and LGCF01000000for CSF3053, 46-5069 and 43-13838 respectively. The raw sequences have been deposited to the short read archive (SRA) of NCBI under accession numbers SRX1094547, SRX1094546and SRX1094545for isolates CSF3053, 46-5069 and 43-13838 respectively.
Data Availability
The following information was supplied regarding data availability:
The raw sequences have been deposited to the short read archive (SRA) of NCBI under accession numbers SRX1094547, SRX1094546and SRX1094545for isolates CSF3053, 46-5069 and 43-13838 respectively.
References
- Be, Bishai & Jain (2012).Be NA, Bishai WR, Jain SK. Role of Mycobacterium tuberculosis pknD in the pathogenesis of central nervous system tuberculosis. BMC Microbiology. 2012;12:7. doi: 10.1186/1471-2180-12-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benavente et al. (2015).Benavente ED, Coll F, Furnham N, McNerney R, Glynn JR, Campino S, Pain A, Mohareb FR, Clark TG. PhyTB: phylogenetic tree visualisation and sample positioning for M. tuberculosis. BMC Bioinformatics. 2015;16:155. doi: 10.1186/s12859-015-0603-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Betts et al. (2002).Betts JC, Lukey PT, Robb LC, McAdam RA, Duncan K. Evaluation of a nutrient starvation model of Mycobacterium tuberculosis persistence by gene and protein expression profiling. Molecular Microbiology. 2002;43:717–731. doi: 10.1046/j.1365-2958.2002.02779.x. [DOI] [PubMed] [Google Scholar]
- Bolger, Lohse & Usadel (2014).Bolger AM, Lohse M, Usadel B. Trimmomatic: a flexible trimmer for illumina sequence data. Bioinformatics. 2014;30(15):2114–2120. doi: 10.1093/bioinformatics/btu170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brodin et al. (2010).Brodin P, Poquet Y, Levillain F, Peguillet I, Larrouy-Maumus G, Gilleron M, Ewann F, Christophe T, Fenistein D, Jang J, Jang MS, Park SJ, Rauzier J, Carralot JP, Shrimpton R, Genovesio A, Gonzalo-Asensio JA, Puzo G, Martin C, Brosch R, Stewart GR, Gicquel B, Neyrolles O. High content phenotypic cell-based visual screen identifies Mycobacterium tuberculosis acyltrehalose-containing glycolipids involved in phagosome remodeling. PLoS Pathogens. 2010;6:e1905. doi: 10.1371/journal.ppat.1001100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi et al. (2012).Choi Y, Sims GE, Murphy S, Miller JR, Chan AP. Predicting the functional effect of amino acid substitutions and indels. PLoS ONE. 2012;7(10):e1905. doi: 10.1371/journal.pone.0046688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cingolani et al. (2012).Cingolani P, Platts A, Wang le L, Coon M, Nguyen T, Wang L, Land SJ, Lu X, Ruden DM. A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly. 2012;6(2):80–92. doi: 10.4161/fly.19695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colangeli et al. (2005).Colangeli R, Helb D, Sridharan S, Sun J, Varma-Basil M, Hazbon MH, Harbacheuski R, Megjugorac NJ, Jacobs WR, Jr, Holzenburg A, Sacchettini JC, Alland D. The Mycobacterium tuberculosis iniA gene is essential for activity of an efflux pump that confers drug tolerance to both isoniazid and ethambutol. Molecular Microbiology. 2005;55:1829–1840. doi: 10.1111/j.1365-2958.2005.04510.x. [DOI] [PubMed] [Google Scholar]
- Coll et al. (2014a).Coll F, McNerney R, Guerra-Assuncao JA, Glynn JR, Perdigao J, Viveiros M, Portugal I, Pain A, Martin N, Clark TG. A robust SNP barcode for typing Mycobacterium tuberculosis complex strains. Nature Communications. 2014a;5 doi: 10.1038/ncomms5812. Article 4812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coll et al. (2015).Coll F, McNerney R, Preston MD, Guerra-Assuncao JA, Warry A, Hill-Cawthorne G, Mallard K, Nair M, Miranda A, Alves A, Perdigao J, Viveiros M, Portugal I, Hasan Z, Hasan R, Glynn JR, Martin N, Pain A, Clark TG. Rapid determination of anti-tuberculosis drug resistance from whole-genome sequences. Genome Medicine. 2015;7:51. doi: 10.1186/s13073-015-0164-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coll et al. (2014b).Coll F, Preston M, Guerra-Assuncao JA, Hill-Cawthorn G, Harris D, Perdigao J, Viveiros M, Portugal I, Drobniewski F, Gagneux S, Glynn JR, Pain A, Parkhill J, McNerney R, Martin N, Clark TG. PolyTB: a genomic variation map for Mycobacterium tuberculosis. Tuberculosis. 2014b;94:346–354. doi: 10.1016/j.tube.2014.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Comas et al. (2012).Comas I, Borrell S, Roetzer A, Rose G, Malla B, Kato-Maeda M, Galagan J, Niemann S, Gagneux S. Whole-genome sequencing of rifampicin-resistant Mycobacterium tuberculosis strains identifies compensatory mutations in RNA polymerase genes. Nature Genetics. 2012;44:106–110. doi: 10.1038/ng.1038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cowley et al. (2004).Cowley S, Ko M, Pick N, Chow R, Downing KJ, Gordhan BG, Betts JC, Mizrahi V, Smith DA, Stokes RW, Av-Gay Y. The Mycobacterium tuberculosis protein serine/threonine kinase PknG is linked to cellular glutamate/glutamine levels and is important for growth in vivo. Molecular Microbiology. 2004;52:1691–1702. doi: 10.1111/j.1365-2958.2004.04085.x. [DOI] [PubMed] [Google Scholar]
- DePristo et al. (2011).DePristo M, Banks E, Poplin R, Garimella K, Maguire J, Hartl C, Philippakis A, del Angel G, Rivas MA, Hanna M, McKenna A, Fennell T, Kernytsky A, Sivachenko A, Cibulskis K, Gabriel S, Altshuler D, Daly M. A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nature Genetics. 2011;43:491–498. doi: 10.1038/ng.806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Demay et al. (2012).Demay C, Liens B, Burguiere T, Hill V, Couvin D, Millet J, Mokrousov I, Sola C, Zozio T, Rastogi N. SITVITWEB–a publicly available international multimarker database for studying Mycobacterium tuberculosis genetic diversity and molecular epidemiology. Infection, Genetics and Evolution. 2012;12:755–766. doi: 10.1016/j.meegid.2012.02.004. [DOI] [PubMed] [Google Scholar]
- Faksri et al. (2011).Faksri K, Drobniewski F, Nikolayevskyy V, Brown T, Prammananan T, Palittapongarnpim P, Prayoonwiwat N, Chaiprasert A. Epidemiological trends and clinical comparisons of Mycobacterium tuberculosis lineages in Thai TB meningitis. Tuberculosis. 2011;91:594–600. doi: 10.1016/j.tube.2011.08.005. [DOI] [PubMed] [Google Scholar]
- Ford et al. (2013).Ford CB, Shah RR, Maeda MK, Gagneux S, Murray MB, Cohen T, Johnston JC, Gardy J, Lipsitch M, Fortune SM. Mycobacterium tuberculosis mutation rate estimates from different lineages predict substantial differences in the emergence of drug-resistant tuberculosis. Nature Genetics. 2013;45:784–790. doi: 10.1038/ng.2656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gori et al. (2005).Gori A, Bandera A, Marchetti G, Degli Esposti A, Catozzi L, Nardi GP, Gazzola L, Ferrario G, van Embden JD, van Soolingen D, Moroni M, Franzetti F. Spoligotyping and Mycobacterium tuberculosis. Emerging Infectious Diseases. 2005;11:1242–1248. doi: 10.3201/eid1108.040982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guillemin, Jarlier & Cambau (1998).Guillemin I, Jarlier V, Cambau E. Correlation between quinolone susceptibility patterns and sequences in the A and B subunits of DNA gyrase in Mycobacteria. Antimicrobial Agents and Chemotherapy. 1998;42:2084–2088. doi: 10.1128/aac.42.8.2084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gupta, Jain & Tyagi (1999).Gupta S, Jain S, Tyagi AK. Analysis, expression and prevalence of the Mycobacterium tuberculosis homolog of bacterial virulence regulating proteins. FEMS Microbiology Letters. 1999;172:137–143. doi: 10.1111/j.1574-6968.1999.tb13461.x. [DOI] [PubMed] [Google Scholar]
- Heym et al. (1995).Heym B, Alzari PM, Honore N, Cole ST. Missense mutations in the catalase-peroxidase gene, katG, are associated with isoniazid resistance in Mycobacterium tuberculosis. Molecular Microbiology. 1995;15:235–245. doi: 10.1111/j.1365-2958.1995.tb02238.x. [DOI] [PubMed] [Google Scholar]
- James et al. (2011).James TR, Helga T, Wendy W, Mitchell G, Eric SL, Gad G, Jill PM. Integrative genomics viewer. Nature Biotechnology. 2011;29:24–26. doi: 10.1038/nbt.1754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kapur et al. (1995).Kapur V, Li LL, Hamrick MR, Plikaytis BB, Shinnick TM, Telenti A, Jacobs WR, Jr, Banerjee A, Cole S, Yuen KY. Rapid Mycobacterium species assignment and unambiguous identification of mutations associated with antimicrobial resistance in Mycobacterium tuberculosis by automated DNA sequencing. Archives of Pathology & Laboratory Medicine. 1995;119:131–138. [PubMed] [Google Scholar]
- Koser et al. (2013).Koser CU, Bryant JM, Becq J, Torok ME, Ellington MJ, Marti-Renom MA, Carmichael AJ, Parkhill J, Smith GP, Peacock SJ. Whole-genome sequencing for rapid susceptibility testing of M. tuberculosis. The New England Journal of Medicine. 2013;369:290–292. doi: 10.1056/NEJMc1215305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kruh et al. (2010).Kruh NA, Troudt J, Izzo A, Prenni J, Dobos KM. Portrait of a pathogen: the Mycobacterium tuberculosis proteome in vivo. PLoS ONE. 2010;5:e1905. doi: 10.1371/journal.pone.0013938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Langmead & Salzberg (2012).Langmead B, Salzberg S. Fast gapped-read alignment with Bowtie 2. Nature Methods. 2012;9:357–359. doi: 10.1038/nmeth.1923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larsen et al. (2007).Larsen MH, Biermann K, Tandberg S, Hsu T, Jacobs WR., Jr Genetic manipulation of Mycobacterium tuberculosis. Current Protocols in Microbiology. 2007 doi: 10.1002/9780471729259.mc10a02s6. Chapter 10:Unit 10A 12. [DOI] [PubMed] [Google Scholar]
- Lavender et al. (2005).Lavender C, Globan M, Sievers A, Billman-Jacobe H, Fyfe J. Molecular characterization of isoniazid-resistant Mycobacterium tuberculosis isolates collected in Australia. Antimicrobial Agents and Chemotherapy. 2005;49:4068–4074. doi: 10.1128/AAC.49.10.4068-4074.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee et al. (1999).Lee AS, Lim IH, Tang LL, Telenti A, Wong SY. Contribution of kasA analysis to detection of isoniazid-resistant Mycobacterium tuberculosis in Singapore. Antimicrobial Agents and Chemotherapy. 1999;43:2087–2089. doi: 10.1128/aac.43.8.2087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leopold et al. (2014).Leopold SR, Goering RV, Witten A, Harmsen D, Mellmann A. Bacterial whole-genome sequencing revisited: portable, scalable, and standardized analysis for typing and detection of virulence and antibiotic resistance genes. Journal of Clinical Microbiology. 2014;52:2365–2370. doi: 10.1128/JCM.00262-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lopez et al. (2003).Lopez B, Aguilar D, Orozco H, Burger M, Espitia C, Ritacco V, Barrera L, Kremer K, Hernandez-Pando R, Huygen K, van Soolingen D. A marked difference in pathogenesis and immune response induced by different Mycobacterium tuberculosis genotypes. Clinical and Experimental Immunology. 2003;133:30–37. doi: 10.1046/j.1365-2249.2003.02171.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mathys et al. (2009).Mathys V, Wintjens R, Lefevre P, Bertout J, Singhal A, Kiass M, Kurepina N, Wang XM, Mathema B, Baulard A, Kreiswirth BN, Bifani P. Molecular genetics of para-aminosalicylic acid resistance in clinical isolates and spontaneous mutants of Mycobacterium tuberculosis. Antimicrobial Agents and Chemotherapy. 2009;53:2100–2109. doi: 10.1128/AAC.01197-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McEvoy et al. (2012).McEvoy CR, Cloete R, Muller B, Schurch AC, van Helden PD, Gagneux S, Warren RM, Gey van Pittius NC. Comparative analysis of Mycobacterium tuberculosis pe and ppe genes reveals high sequence variation and an apparent absence of selective constraints. PLoS ONE. 2012;7:e1905. doi: 10.1371/journal.pone.0030593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKenna et al. (2010).McKenna A, Hanna M, Banks E, Sivachenko A, Cibulskis K, Kernytsky A, Garimella K, Altshuler D, Gabriel S, Daly M, DePristo MA. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Research. 2010;20:1297–1303. doi: 10.1101/gr.107524.110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mdluli et al. (1998).Mdluli K, Slayden RA, Zhu Y, Ramaswamy S, Pan X, Mead D, Crane DD, Musser JM, Barry CE., 3rd Inhibition of a Mycobacterium tuberculosis beta-ketoacyl ACP synthase by isoniazid. Science. 1998;280:1607–1610. doi: 10.1126/science.280.5369.1607. [DOI] [PubMed] [Google Scholar]
- Nahid et al. (2010).Nahid P, Bliven EE, Kim EY, Mac Kenzie WR, Stout JE, Diem L, Johnson JL, Gagneux S, Hopewell PC, Kato-Maeda M, Tuberculosis Trials C Influence of M. tuberculosis lineage variability within a clinical trial for pulmonary tuberculosis. PloS One. 2010;5:e1905. doi: 10.1371/journal.pone.0010753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ng et al. (2004).Ng VH, Cox JS, Sousa AO, MacMicking JD, McKinney JD. Role of KatG catalase-peroxidase in mycobacterial pathogenesis: countering the phagocyte oxidative burst. Molecular Microbiology. 2004;52:1291–1302. doi: 10.1111/j.1365-2958.2004.04078.x. [DOI] [PubMed] [Google Scholar]
- Nikaido (2009).Nikaido H. Multidrug resistance in bacteria. Annual Review of Biochemistry. 2009;78:119–146. doi: 10.1146/annurev.biochem.78.082907.145923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okamoto et al. (2007).Okamoto S, Tamaru A, Nakajima C, Nishimura K, Tanaka Y, Tokuyama S, Suzuki Y, Ochi K. Loss of a conserved 7-methylguanosine modification in 16S rRNA confers low-level streptomycin resistance in bacteria. Molecular Microbiology. 2007;63:1096–1106. doi: 10.1111/j.1365-2958.2006.05585.x. [DOI] [PubMed] [Google Scholar]
- Palittapongarnpim et al. (1997).Palittapongarnpim P, Luangsook P, Tansuphaswadikul S, Chuchottaworn C, Prachaktam R, Sathapatayavongs B. Restriction fragment length polymorphism study of Mycobacterium tuberculosis in Thailand using IS6110 as probe. The International Journal of Tuberculosis and Lung Disease. 1997;1:370–376. [PubMed] [Google Scholar]
- Projahn et al. (2011).Projahn M, Koser CU, Homolka S, Summers DK, Archer JA, Niemann S. Polymorphisms in isoniazid and prothionamide resistance genes of the Mycobacterium tuberculosis complex. Antimicrobial Agents and Chemotherapy. 2011;55:4408–4411. doi: 10.1128/AAC.00555-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quinlan & Hall (2010).Quinlan AR, Hall IM. BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics. 2010;26(6):841–842. doi: 10.1093/bioinformatics/btq033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raman, Yeturu & Chandra (2008).Raman K, Yeturu K, Chandra N. targetTB: a target identification pipeline for Mycobacterium tuberculosis through an interactome, reactome and genome-scale structural analysis. BMC Systems Biology. 2008;2:109. doi: 10.1186/1752-0509-2-109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramaswamy et al. (2000).Ramaswamy SV, Amin AG, Goksel S, Stager CE, Dou SJ, El Sahly H, Moghazeh SL, Kreiswirth BN, Musser JM. Molecular genetic analysis of nucleotide polymorphisms associated with ethambutol resistance in human isolates of Mycobacterium tuberculosis. Antimicrobial Agents and Chemotherapy. 2000;44:326–336. doi: 10.1128/AAC.44.2.326-336.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramaswamy et al. (2003).Ramaswamy SV, Reich R, Dou SJ, Jasperse L, Pan X, Wanger A, Quitugua T, Graviss EA. Single nucleotide polymorphisms in genes associated with isoniazid resistance in Mycobacterium tuberculosis. Antimicrobial Agents and Chemotherapy. 2003;47:1241–1250. doi: 10.1128/AAC.47.4.1241-1250.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reed et al. (2004).Reed MB, Domenech P, Manca C, Su H, Barczak AK, Kreiswirth BN, Kaplan G, Barry CE., 3rd A glycolipid of hypervirulent tuberculosis strains that inhibits the innate immune response. Nature. 2004;431:84–87. doi: 10.1038/nature02837. [DOI] [PubMed] [Google Scholar]
- Regmi et al. (2015).Regmi SM, Coker OO, Kulawonganunchai S, Tongsima S, Prammananan T, Viratyosin W, Thaipisuttikul I, Chaiprasert A. Polymorphisms in drug-resistant-related genes shared among drug-resistant and pan-susceptible strains of sequence type 10, Beijing family of Mycobacterium tuberculosis. International Journal of Mycobacteriology. 2015;4:67–72. doi: 10.1016/j.ijmyco.2014.11.050. [DOI] [PubMed] [Google Scholar]
- Rengarajan, Bloom & Rubin (2005).Rengarajan J, Bloom BR, Rubin EJ. Genome-wide requirements for Mycobacterium tuberculosis adaptation and survival in macrophages. Proceedings of the National Academy of Sciences of the United States of America. 2005;102:8327–8332. doi: 10.1073/pnas.0503272102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rousseau et al. (2003).Rousseau C, Sirakova TD, Dubey VS, Bordat Y, Kolattukudy PE, Gicquel B, Jackson M. Virulence attenuation of two Mas-like polyketide synthase mutants of Mycobacterium tuberculosis. Microbiology. 2003;149:1837–1847. doi: 10.1099/mic.0.26278-0. [DOI] [PubMed] [Google Scholar]
- Sandgren et al. (2009).Sandgren A, Strong M, Muthukrishnan P, Weiner BK, Church GM, Murray MB. Tuberculosis drug resistance mutation database. PLoS Medicine. 2009;6(2):e1905. doi: 10.1371/journal.pmed.1000002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sassetti, Boyd & Rubin (2003).Sassetti CM, Boyd DH, Rubin EJ. Genes required for mycobacterial growth defined by high density mutagenesis. Molecular Microbiology. 2003;48:77–84. doi: 10.1046/j.1365-2958.2003.03425.x. [DOI] [PubMed] [Google Scholar]
- Siddiqi et al. (2004).Siddiqi N, Das R, Pathak N, Banerjee S, Ahmed N, Katoch VM, Hasnain SE. Mycobacterium tuberculosis isolate with a distinct genomic identity overexpresses a tap-like efflux pump. Infection. 2004;32:109–111. doi: 10.1007/s15010-004-3097-x. [DOI] [PubMed] [Google Scholar]
- Singh et al. (2003).Singh A, Jain S, Gupta S, Das T, Tyagi AK. mymA operon of Mycobacterium tuberculosis: its regulation and importance in the cell envelope. FEMS Microbiology Letters. 2003;227:53–63. doi: 10.1016/S0378-1097(03)00648-7. [DOI] [PubMed] [Google Scholar]
- Sirakova et al. (2003).Sirakova TD, Dubey VS, Kim HJ, Cynamon MH, Kolattukudy PE. The largest open reading frame (pks12) in the Mycobacterium tuberculosis genome is involved in pathogenesis and dimycocerosyl phthiocerol synthesis. Infection and Immunity. 2003;71:3794–3801. doi: 10.1128/IAI.71.7.3794-3801.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sreevatsan et al. (1997).Sreevatsan S, Pan X, Stockbauer KE, Connell ND, Kreiswirth BN, Whittam TS, Musser JM. Restricted structural gene polymorphism in the Mycobacterium tuberculosis complex indicates evolutionarily recent global dissemination. Proceedings of the National Academy of Sciences of the United States of America. 1997;94:9869–9874. doi: 10.1073/pnas.94.18.9869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Srivastava et al. (2009).Srivastava S, Ayyagari A, Dhole TN, Nyati KK, Dwivedi SK. emb nucleotide polymorphisms and the role of embB306 mutations in Mycobacterium tuberculosis resistance to ethambutol. International Journal of Medical Microbiology: IJMM. 2009;299:269–280. doi: 10.1016/j.ijmm.2008.07.001. [DOI] [PubMed] [Google Scholar]
- Taniguchi et al. (1996).Taniguchi H, Aramaki H, Nikaido Y, Mizuguchi Y, Nakamura M, Koga T, Yoshida S. Rifampicin resistance and mutation of the rpoB gene in Mycobacterium tuberculosis. FEMS Microbiology Letters. 1996;144:103–108. doi: 10.1111/j.1574-6968.1996.tb08515.x. [DOI] [PubMed] [Google Scholar]
- Telenti et al. (1997).Telenti A, Philipp WJ, Sreevatsan S, Bernasconi C, Stockbauer KE, Wieles B, Musser JM, Jacobs WR., Jr The emb operon, a gene cluster of Mycobacterium tuberculosis involved in resistance to ethambutol. Nature Medicine. 1997;3:567–570. doi: 10.1038/nm0597-567. [DOI] [PubMed] [Google Scholar]
- Thierry et al. (1990).Thierry D, Cave MD, Eisenach KD, Crawford JT, Bates JH, Gicquel B, Guesdon JL. IS6110, an IS-like element of Mycobacterium tuberculosis complex. Nucleic acids research. 1990;18:188. doi: 10.1093/nar/18.1.188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tho et al. (2012).Tho DQ, Torok ME, Yen NT, Bang ND, Lan NT, Kiet VS, van Vinh Chau N, Dung NH, Day J, Farrar J, Wolbers M, Caws M. Influence of antituberculosis drug resistance and Mycobacterium tuberculosis lineage on outcome in HIV-associated tuberculous meningitis. Antimicrobial Agents and Chemotherapy. 2012;56:3074–3079. doi: 10.1128/AAC.00319-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thwaites et al. (2008).Thwaites G, Caws M, Chau TT, D’Sa A, Lan NT, Huyen MN, Gagneux S, Anh PT, Tho DQ, Torok E, Nhu NT, Duyen NT, Duy PM, Richenberg J, Simmons C, Hien TT, Farrar J. Relationship between Mycobacterium tuberculosis genotype and the clinical phenotype of pulmonary and meningeal tuberculosis. Journal of Clinical Microbiology. 2008;46:1363–1368. doi: 10.1128/JCM.02180-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thwaites, van Toorn & Schoeman (2013).Thwaites GE, van Toorn R, Schoeman J. Tuberculous meningitis: more questions, still too few answers. Lancet Neurol. 2013;12:999–1010. doi: 10.1016/S1474-4422(13)70168-6. [DOI] [PubMed] [Google Scholar]
- Tsenova et al. (2005).Tsenova L, Ellison E, Harbacheuski R, Moreira AL, Kurepina N, Reed MB, Mathema B, Barry CE, 3rd, Kaplan G. Virulence of selected Mycobacterium tuberculosis clinical isolates in the rabbit model of meningitis is dependent on phenolic glycolipid produced by the bacilli. The Journal of Infectious Diseases. 2005;192:98–106. doi: 10.1086/430614. [DOI] [PubMed] [Google Scholar]
- Tsolaki et al. (2004).Tsolaki AG, Hirsh AE, DeRiemer K, Enciso JA, Wong MZ, Hannan M, Goguet de la Salmoniere YO, Aman K, Kato-Maeda M, Small PM. Functional and evolutionary genomics of Mycobacterium tuberculosis: insights from genomic deletions in 100 strains. Proceedings of the National Academy of Sciences of the United States of America. 2004;101:4865–4870. doi: 10.1073/pnas.0305634101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van der Auwera et al. (2013).Van der Auwera GA, Carneiro M, Hartl C, Poplin R, del Angel G, Levy-Moonshine A, Jordan T, Shakir K, Roazen D, Thibault J, Banks E, Garimella K, Altshuler D, Gabriel S, DePristo M. From FastQ data to high-confidence variant calls: the genome analysis toolkit best practices pipeline. Current Protocols in Bioinformatics. 2013;43:11.10.1–11.10.33. doi: 10.1002/0471250953.bi1110s43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Embden et al. (1993).van Embden JD, Cave MD, Crawford JT, Dale JW, Eisenach KD, Gicquel B, Hermans P, Martin C, McAdam R, Shinnick TM, Small PM. Strain identification of Mycobacterium tuberculosis by DNA fingerprinting: recommendations for a standardized methodology. Journal of Clinical Microbiology. 1993;31:406–409. doi: 10.1128/jcm.31.2.406-409.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Velmurugan et al. (2007).Velmurugan K, Chen B, Miller JL, Azogue S, Gurses S, Hsu T, Glickman M, Jacobs WR, Jr, Porcelli SA, Briken V. Mycobacterium tuberculosis nuoG is a virulence gene that inhibits apoptosis of infected host cells. PLoS Pathogens. 2007;3:e1905. doi: 10.1371/journal.ppat.0030110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang et al. (2011).Wang XD, Gu J, Wang T, Bi LJ, Zhang ZP, Cui ZQ, Wei HP, Deng JY, Zhang XE. Comparative analysis of mycobacterial NADH pyrophosphatase isoforms reveals a novel mechanism for isoniazid and ethionamide inactivation. Molecular Microbiology. 2011;82:1375–1391. doi: 10.1111/j.1365-2958.2011.07892.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng et al. (2008).Zheng H, Lu L, Wang B, Pu S, Zhang X, Zhu G, Shi W, Zhang L, Wang H, Wang S, Zhao G, Zhang Y. Genetic basis of virulence attenuation revealed by comparative genomic analysis of Mycobacterium tuberculosis strain H37Ra versus H37Rv. PLoS ONE. 2008;3:e1905. doi: 10.1371/journal.pone.0002375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zumla et al. (2015).Zumla A, George A, Sharma V, Herbert RH, Baroness Masham of I. Oxley A, Oliver M. The WHO 2014 global tuberculosis report–further to go. The Lancet Global Health. 2015;3:e10–12. doi: 10.1016/S2214-109X(14)70361-4. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
SNPs common to isolates CSF-3053, 46-5069 and 43-13838 compared to 92,000 SNPs from 1,601 genomes of M. tuberculosis complex members (Coll et al., 2014a; Coll et al., 2014b) were used to position the isolates as belonging to subineage 1.2.1.
Figure 2: PCR products using primers CF: GCCCAACCTGATTGGTTTCG and CR: CAAACGCTCGCCATGATCTC for RD239 Primers were designed to cover region 4092041-4092947. Expected size is 907 bp Lane 1: 1 kb DNA plus ladder Lane 2: M. tuberculosis (H37Rv, NC_000962.3) Lane 3: CSF3053 Lane 4: 46-5069 Lane 5: 43-13838. Figure 3: PCR products using primers AF: GCCCAACCTGATTGGTTTCG and AR: CAAACGCTCGCCATGATCTC for RD147c Primers were designed to cover region 1718833-1721268. Expected sixe is 2436 Lane 1: 1 kb DNA plus ladder Lane 2: M. tuberculosis (H37Rv NC_000962.3) Lane 3: CSF3053 Lane 4: 46-5069 Lane 5: 43-13838. Figure 4: PCR products using primers BF: GCCCAACCTGATTGGTTTCG and BR: CAAACGCTCGCCATGATCTC for 500 bp deletion. Primers were designed to cover region 3501124-3501822. Expected size is 699 bp Lane 1: 1 kb DNA plus ladder Lane 2: M. tuberculosis (H37Rv NC_000962.3) Lane 3: CSF3053 Lane 4: 46-5069 Lane 5: 43-13838.
Table S1: The positions, nucleotide change, amino acid change and effect of single nucleotide polymorphisms in virulence genes that are common to isolates CSF3053, 46-5069 and 43-13838. The protein variation was determined by Protein Variation Effect Analyzer (PROVEAN), a web based protein variation analysis tool (Choi et al., 2012). Table S2: The positions, nucleotide change, amino acid change and effect of single nucleotide polymorphisms in efflux pump related genes that are common to isolates CSF3053, 46-5069 and 43-13838. The protein variation was determined by Protein Variation Effect Analyzer (PROVEAN), a web based protein variation analysis tool (Choi et al., 2012).
The positions, nucleotide change , amino acid change and effect of single nucleotide polymorphisms in genes with known epitopes that are common to isolates CSF3053, 46-5069 and 43-13838. The protein variation was determined by Protein Variation Effect Analyzer (PROVEAN), a web based protein variation analysis tool (Choi et al., 2012).
The reference genome positions, nucleotide change, amino acid change and effect of single nucleotide polymorphisms in essential genes that are common to isolates CSF3053, 46-5069 and 43-13838. The protein variation was determined by Protein Variation Effect Analyzer (PROVEAN). A web based protein variation analysis tool (Choi et al., 2012).
Data Availability Statement
The following information was supplied regarding data availability:
The raw sequences have been deposited to the short read archive (SRA) of NCBI under accession numbers SRX1094547, SRX1094546and SRX1094545for isolates CSF3053, 46-5069 and 43-13838 respectively.