Abstract
Maternal immune activation (MIA) model is increasingly well appreciated as a rodent model for the environmental risk factor of various psychiatric disorders. Numerous studies have demonstrated that MIA model is able to show face, construct, and predictive validity that are relevant to autism and schizophrenia. To model MIA, investigators often use viral mimic polyinosinic:polycytidylic acid (poly(I:C)) to activate the immune system in pregnant rodents. Generally, the offspring from immune activated dam exhibit behavioral abnormalities and physiological alterations that are associated with autism and schizophrenia. However, poly(I:C) injection with different dosages and at different time points could lead to different outcomes by perturbing brain development at different stages. Here we provide a detailed method of inducing MIA by intraperitoneal (i.p.) injection of 20 mg/kg poly(I:C) at mid-gestational embryonic 12.5 days (E12.5). This method has been shown to induce acute inflammatory response in the maternal-placental-fetal axis, which ultimately results in the brain perturbations and behavioral phenotypes that are associated with autism and schizophrenia.
Keywords: Immunology, Issue 109, Maternal infection, Maternal immune activation (MIA), Polyinosinic:polycytidylic acid (Poly(I:C)), Embryonic 12.5 days (E12.5), Interleukin-6 (IL-6), Maternal-placental-fetal axis, Cerebellar Purkinje cells, Hindbrain, Autism, Schizophrenia, Behavioral abnormalities
Introduction
The concept of maternal immune activation (MIA) originates from the epidemiology studies on the association of maternal infection with autism and schizophrenia1. Due to the absence of detectable replicating viral pathogens in the placenta or the fetal brain after maternal viral infection2,3, the effect of the infection on the offspring is hypothesized to be caused by the activation of the maternal immune system rather than the pathogens themselves.
To elucidate the cause-and-effect relationship between MIA and psychiatric disorders, injection of chemically synthesized, viral mimic double stranded RNA polyinosinic:polycytidylic acid (poly(I:C)) into pregnant rodents has been widely used as the animal model for MIA4,5. Poly(I:C) is recognized by toll-like receptor 3 (TLR3), and systemic administration of poly(I:C) induces viral-like acute inflammatory response. One of the mechanisms by which poly(I:C) produces behavioral abnormalities and neuropathologies in the offspring is by causing an imbalance of pro- and anti- inflammatory cytokines in the maternal-placental-fetal axis6. Several groups have adopted the MIA model to understand the etiology of psychiatric disorders7, and due to the diverse interests among research groups, various time points of immune activation have been used to achieve different perturbations on brain development and behaviors7.
The Paul H. Patterson laboratory at the California Institute of Technology adopts the strategy of injecting poly(I:C) into pregnant mice at embryonic 12.5 days (E12.5), which has successfully demonstrated that MIA is capable of inducing behavioral, neurological, and immunological changes in the offspring that are associated with autism and schizophrenia8-11. Our prior works show that MIA offspring display behavioral abnormalities (e.g., social impairment, communication deficit, repetitive behavior, anxiety-like behavior, and latent inhibition deficit8,10,12), immune dysregulation and cytokines imbalance8,13,14, alteration of fetal brain gene expression15, loss of Purkinje cell in lobule VII of cerebellum11, alteration of synaptic properties in hippocampus9, gene x environment interaction13, alteration of gut permeability, and gut microbiota composition16. Furthermore, therapeutic and prophylactic strategies are also developed from this model system13,16,17. By inducing MIA at E12.5, others have shown that MIA produces fetal microglial activation and cholinergic developmental alteration in basal forebrain18, strain specific interaction19, brain cerebral synaptosomal ultrastructural abnormalities, cerebral mitochondrial respiratory chain hyperfunction abnormalties, downregulation of cerebral synaptosomal molecules17, depressive-like behaviors, impairment in cognition and hippocampal long-term potentiation (LTP), and deficit of adult hippocampal neurogenesis20.
Here, we provide a detailed method of how to induce MIA at E12.5 by poly(I:C), as well as paradigms of how to apply this model to study the etiology of autism and schizophrenia. It is important to note that MIA is a risk factor for a variety of disorders4, and its outcomes are extremely sensitive to the time and method of induction as well as the husbandry of the pregnant dams. As such, even minor inconsistencies between laboratories often result in low reproducibility and/or different phenotypes in the offspring. Our method is specifically designed for those interested in studying MIA as an environmental risk factor for autism and schizophrenia, and the detailed description provided will help researchers improve the reproducibility of their data.
Protocol
All protocols were performed under the approval of the California Institute of Technology Institutional Animal Care and Use Committee (IACUC).
1. Preparation for Timed-mating Pairs
Use at least 6 dams for behavioral experiments and 3 for gene expression analysis. NOTE: Use double the number of estimated female mice, as only about 50% of the mice will be plugged from the timed-mating.
Use female mice with no prior pregnancy and at 8-16 weeks of age. NOTE: Female mice that have prior sexual exposure to male mice but have not been pregnant are acceptable.
House the female mice together and place the cages next to each other in order to synchronize their estrous cycle. Use a standard mice cage with a height of over 5.5 inches and a 75 square inch interior floor to allow housing of 5 adult mice. Label each female mouse using an ear punch.
2. Setting up Timed-mating Pair
Set up the timed-mating 0-2 hr before the dark cycle begins. Place two female mice in each cage. Take out the mice and weigh them individually. Record the body weight of each female mouse.
Place one male mouse into each cage with the two female mice. Use trio mating to minimize the paternal confounding effect.
3. Vaginal Plug Check (E0.5)
Check the female mice 0-4 hr after the dark cycle ends. Gently lift the female mouse from the base of the tail and allow the mouse to grab the wire grid, cage top, or cage edge.
Search for signs of vaginal plug: vaginal plug is a visible whitish mass in the vaginal opening. If the plug is not obvious, gently insert a 200 µl pipette tip into the vaginal opening. Resistance from coagulation confirms vaginal plug formation. Check the vaginal plug once per day. NOTE: Vaginal plug is only an indication that copulation has occurred, and therefore does not guarantee pregnancy. Vaginal plug may be difficult to identify in certain strains of mice. Seek help from an experienced mice caregiver to increase the accuracy of plug identification.
Move the plugged mice to a new cage and group house them. Define the day of vaginal plug appearance as embryonic 0.5 day (E0.5).
4. On E10.5-11.5, Check for Pregnancy
Gently lift the base of the tail and allow the mouse to grab the wire grid, cage top, or cage edge. Observe the abdomen area to check for signs of pregnancy, e.g., a bulge in the abdomen (Figure 1). NOTE: The sign of pregnancy is always more obvious at E11.5 than at E10.5.
Weigh the mice to verify pregnancy. The pregnant mouse generally weighs about 20% heavier at E10.5 and 30% heavier at E12.5 compared to a non-pregnant mouse (Figure 2). Single house the pregnant mice in clean cages.
5. 20 mg/kg Poly(I:C) Preparation
Use at least 6 dams per group for behavioral experiments and 3 per group for gene expression analysis. Prepare the corresponding amount of poly(I:C) solution.
Weigh the poly(I:C) powder in a 50 ml conical tube to make a final concentration of 40 mg/ml. In order to minimize and standardize the stress caused by injection volume, inject 5 µl of poly(I:C) solution for each gram of mouse body weight (5 µl/g, or 5 ml/kg). To induce MIA, we inject 20 mg of poly(I:C) per kg. NOTE: The concentration of poly(I:C) solution needed is (20 mg/kg)/(5 ml/kg) = 4 mg/ml. Since the poly(I:C) powder contains 10% poly(I:C), we need to make a final concentration of (4 mg/ml)/0.1= 40 mg/ml of poly(I:C) solution. CAUTION: The poly(I:C) powder is very light. Be careful not to lose any powder while transferring from the spatula to the conical tube.
Add the corresponding volume of 0.9% sodium chloride (saline) into the conical tube and close the cap. Dissolve the powder by gently rolling the saline droplet over the poly(I:C) powder until the solution is clear. Centrifuge the conical tube at 800 x g for 1 min (Figure 3C). Filter the poly(I:C) solution by 0.22 µm filter. Transfer the poly(I:C) solution to an 1.5 or 2 ml microcentrifuge tube. Optional: Use a spectrophotometer to measure the 260/280 ratio of the poly(I:C) solution. Ensure the ratio is between 1.54-1.82 (Figure 3) as described by the manufacturer. NOTE: The saline used to dissolve poly(I:C) powder and serve as control injection should be sterile and pharmaceutical grade.
6. Saline or Poly(I:C) Injection on E12.5
Weigh the mouse on the scale and put it back into the cage. Use the following formula to calculate the volume of injection for both saline and poly(I:C) mice: body weight (g) multiplied by 5 = desired injection volume (µl). Use a 0.3 ml insulin syringe to draw up the calculated volume of saline or 20 mg/kg poly(I:C) solution, e.g., 150 µl for a 30 g mouse.
Gently pick up the mouse by the base of the tail and place it on top of the cage. Handle the mouse gently to avoid confounding effects induced by stress. Insert the needle, bevel up, at approximately 20° relative to the mouse into the center of its two upper nipples (Figure 4), and inject the saline or poly(I:C) solution. NOTE: The needle should be replaced for each mouse after injection.
Place the mouse back to its home cage. Place the cages in a quiet, low traffic room to avoid the other confounding effects.
7. The Day After Poly(I:C) Injection (E13.5)
Check the injected mice on the following morning. Use a scale to measure body weight. NOTE: Poly(I:C) injected pregnant mice usually gain less weight than saline injected mice on the day after injection.
Do not disturb the mice until the offspring are born.
8. Gauge of MIA Offspring
- In order to minimize the confounding effects of MIA induction, follow the guidelines listed below to gauge the usage of MIA offspring.
- Exclude the litters from preterm or delayed birth, or if the litter size is smaller than 5.
- Euthanize the excessive pups by CO2 if the litter size is larger than 10.
9. Optional: Examine the Acute Inflammatory Response After MIA
Sacrifice the pregnant mice 3 hr after saline or poly(I:C) injection using the method described in the institution's animal care committee-approved protocol. NOTE: Cervical dislocation is often preferred as it minimizes the effect of CO2/euthanasia drugs on the dam and embryo.
- Collect the spleen from the adult pregnant mice and isolate the placenta and fetal brain under the stereomicroscope.
- For spleen collection, lay the mouse on the dissection plate and spray 70% ethanol on the abdominal area of the pregnant mice. Use a pair of sterile scissors to make a vertical cut in the center of abdominal area below the ribcage through the skin.
- The spleen sits in the left superior abdominal quadrant. Use forceps to isolate the spleen. Roll the spleen on delicate task wipers to remove the fatty tissue around the organ. Collect roughly 50 mg of splenic tissue and place them individually in Eppendorf tubes with 500 µl RNA stabilize buffer or TRIzol to prevent nucleic acid degradation. Store the samples at -80 °C freezer until use.
- For placenta collection, gently pull out the uterine horns from the body. Place the uterine horns in a Petri dish filled with autoclaved phosphate buffered saline (PBS) and leave the dish on ice. Use two forceps to tear open the uterine wall and expose the amniotic sac.
- Carefully use the forceps to open the amniotic sac without damaging the placenta and the fetus. Separate the placenta from the fetus and cut the umbilical cord as close to the placenta as possible. Remove the amniotic sac debris from the placenta. Place the placenta individually in Eppendorf tubes with 500 µl RNA stabilize buffer or TRIzol. Store the sample at -80 °C freezer until use.
- For fetal brain collection, store the fetus in RNA stabilize buffer from step 9.2.2 until dissection. Tease off the pial membrane from the ventricle of the brain using delicate #5 forceps under the stereomicroscope. Carefully remove all the membrane from the fetal brain. Place the fetal brains individually in Eppendorf tubes with 500 µl RNA stabilize buffer or TRIzol. Store the sample at -80 °C freezer until use.
Extract the RNA from the tissue and convert the RNA to cDNA. Analyze the Il6 gene expression in the cDNA by real-time PCR .
Extract the RNA from the tissues by following the manufacture protocol of TRIzol. If the tissues are stored in RNA stabilization buffer. Thaw the sample and spin down the buffer. Transfer the tissue by tip to another eppendorf with 500 µl TRIzol and continue RNA extraction.
Use a spectrophotometer to measure the concentration, 260/280 and 260/230 ratio of the RNA. Ensure the ratios are above 2.0.
Eliminate the DNA contamination by treated the RNA with DNase I. Mix 1 µg RNA, 1 µl 10X reaction buffer, 1 µl RNase-Free DNase, and nuclease-free water to a final volume of 10 µl. Incubate the master mix at 37 °C for 30 min. Add 1 µl DNase stop solution to terminate the reaction. Incubate the mixture at 65 °C for 10 min.
Reverse transcribe the RNA to cDNA. Use 20 µl reactions, and up to 1 µg of total RNA per reaction. Mix 4 µl 5x reverse transcription (RT) reaction mix buffer, 1 µl reverse transcriptase, 11 µl RNA template after the treatment with DNase I and 4 µl nuclease-free H2O to make a final 20 µl solution. Program the thermal cycler conditions for RT. A generic condition include: Step 1: 25 °C for 5 min; Step 2: 42 °C for 30 min; Step 3: 85 °C for 5 min; Step 4: hold at 4 °C. Dilute the cDNA 5 times. For short-term storage, store the cDNA at 4°C. For long-term storage, freeze the cDNA at -20 °C.
- Analyze the Il6 gene expression in the cDNA by real-time PCR and normalize the Il6 gene expression to β-actin gene expression.
- Make a master mix for Il6 and β-actin detection. Use 25 µl reaction, the master mix for each gene includes 12.5 µl SYBR Green Mix, 0.5 µl of 10 µM forward primer, 0.5 µl of 10 µM reverse primer and 6.5 µl nuclease-free H2O.
- Place 5 µl 5x diluted cDNA at the bottom of the well of optical 96-well reaction plate. Pipette 20 µl of master mix to each well to make a final 25 µl PCR mix. Triplicate each gene for each sample. Seal the plate by using optical adhesive film. Spin down the plate at 800 x g for 3 min at 4 °C Use the standard program of real-time PCR: Step 1: 50 °C for 2 min; Step 2: 95 °C for 10 min; Step 3: 40 cycles of 95 °C for 15 sec and temperature 60 °C for 1 min. Add an additional step for dissociation curve.
10. Examine the Behavioral Abnormalities in MIA Adult Offspring (Optional):
Perform the behavior tests at the ages of 8-12 weeks. Change the cage bedding at least 3 days before the test. Acclimate the mice to the behavioral testing room at least 30 min before the test.
For prepulse inhibition (PPI), restrain the mice in plexiglass cylinder with a piezo-electric sensor underneath it. Acclimate the mice to the PPI chamber for 5 min. Expose the mice with 6 trials of 120 dB white noise (startle).
Then expose the mice with randomized mixtures of 14 trials of background noise, 14 trials of Startle, 14 trials of prepulse 5 dB (5 dB higher than background noise) + Startle (PPI5), and 14 trials of prepulse 15 dB (15 dB higher than background noise + Startle (PPI15).
Average the startle response for the first 6 trials of 120 dB trials. Average the startle response for the other individual stimulations. Covert the value to prepulse inhibition by (Startle - PPI5 or PPI15)/Startle.
For open-field test, place the mouse in the corner of an open square arena (50 x 50 x 50 cm3). Record the trajectory of the mouse for 10 min through a ceiling mounted camera. Define the center of the arena as the center zone (17 x 17 cm2). Quantify the distance traveled, center zone entries, and time spent in center zone manually or by image-based automatic analysis software.
For marble burying test, acclimate the mouse to a test cage with compressed 5cm deep, clean, Aspen pine bedding for 10 min. Return the mouse to its homecage. Gently place 20 navy blue 1.5 cm diameter glass marbles in the fashion of 4 x 5 arrangement. Place the mouse back to the test cage with marbles for 10 min. Count the marbles that has been buried in the 10 min testing duration.
11. Examine the Cerebellar Neuropathology in MIA Adult Offspring (Optional):
Euthanize the saline and MIA adult offspring by anesthetic. Perfuse the mouse with 50 ml PBS and subsequently 50 ml 4% paraformaldehyde through the cardiovascular system.
Remove the brain carefully and postfix the brain in 4% paraformaldehyde overnight at 4°C. Rinse the brain with PBS three times and store the brain in PBS with 0.02% sodium azide in a 4 °C fridge until use. NOTE: The postfixed brain can be stored in PBS with 0.02% sodium azide in the fridge for up to a month.
Section the brain into 50 µm thin slices sagittally using vibratome. Collect the brain sections using a soft brush pen. Terminate the endogenous peroxidase activity by incubating the sections in 0.6% hydrogen peroxide for 30 min at room temperature.
Incubate the sections in anti-calbindin antibody diluted in the blocking buffer (10% goat serum with 0.1% Triton X-100 and 0.02% sodium azide in PBS) overnight at room temperature. Then incubate the sections in corresponding biotinylated secondary antibody diluted in blocking buffer for 2 hr at room temperature.
Incubate the sections in avidin DH - Biotinylated peroxidase H complex buffer to detect the biotin for 1 hr at room temperature. Develop the sections by using peroxidase substrate to visualize the antigen. Wash the sections with PBS three times in between the steps. Mount the sections onto microscope slides. Image the sections under the microscope.
Representative Results
Injection of 20 mg/kg poly(I:C) at E12.5 could evoke an acute inflammatory response in the maternal-placental-fetal axis and precipitate a chronic effect to brain development and behavioral phenotypes12,13. Elevated levels of a proinflammatory cytokine, Interleukin (IL)-6, is a reliable indicator of acute inflammatory response after MIA. The peak time for Il6 gene expression in the placenta and fetal brain is at 3 hr post-poly(I:C) injection12,13. We have shown that poly(I:C) induces higher Il6 gene expression across the maternal spleen-placenta-fetal brain (Figure 5) (Data adapted from Wu et al., 2015)13. Researchers could combine this paradigm with other treatment to study the factors that might alter acute inflammatory response after MIA, e.g., maternal diets, genetic mutant mice, anti-inflammatory drugs, etc.
On the other hand, behavioral changes are also hallmarks of the MIA model. Several behavioral tests can be conducted on the MIA adult offspring to demonstrate different autistic and schizophrenic-like behaviors. Generally, when compared to saline offspring, MIA offspring display fewer center zone entries and shorter center zone duration spent in open-field test. They also display more frequent burying behaviors in marble burying test and lower PPI (Figure 6) (Data adapted from Wu et al., 2015)13. Loss of Purkinje cells in cerebellum lobule VII is another hallmark of MIA offspring (Shi et al., 2009). When stained with calbindin antibody, there are fewer calbindin-positive Purkinje cells in the cerebellum lobule VII of MIA offspring compared to the saline control offspring (Figure 7).
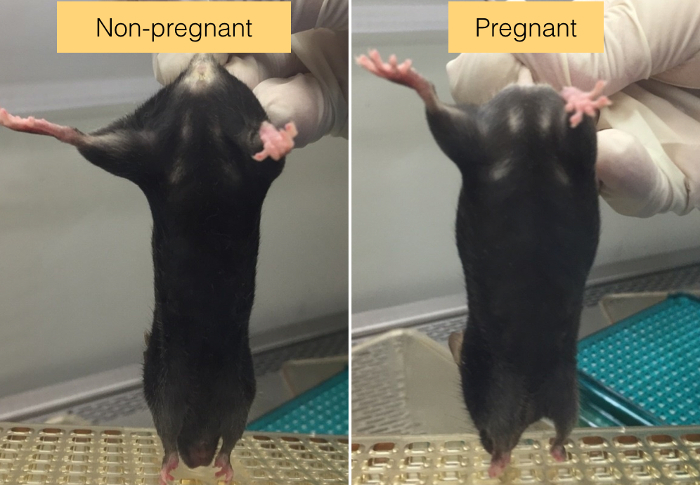 Figure 1. The sign of pregnant mice. Mid-gestation mouse can be identified by the bulge in its abdomen area (Arrow). Inbred C57BL/6 mouse strain is used here as an example. Please click here to view a larger version of this figure.
Figure 1. The sign of pregnant mice. Mid-gestation mouse can be identified by the bulge in its abdomen area (Arrow). Inbred C57BL/6 mouse strain is used here as an example. Please click here to view a larger version of this figure.
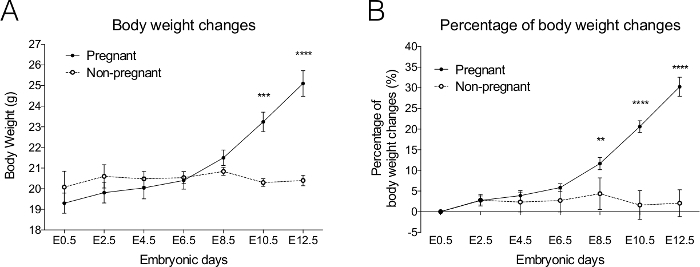 Figure 2. Body weight changes in pregnant C57BL/6N mice from embryonic (E) 0.5-12.5 days. When comparing the pregnant and non-pregnant mice with vaginal plugs present at E0.5, the(A) body weight and (B) percentage of body weight undergoes a dramatic increase at E10.5 in pregnant mice. Pregnant n = 10, Non-pregnant n = 5. Data are presented as Mean ± SE. *** p <0.001, **** p <0.0001. Please click here to view a larger version of this figure.
Figure 2. Body weight changes in pregnant C57BL/6N mice from embryonic (E) 0.5-12.5 days. When comparing the pregnant and non-pregnant mice with vaginal plugs present at E0.5, the(A) body weight and (B) percentage of body weight undergoes a dramatic increase at E10.5 in pregnant mice. Pregnant n = 10, Non-pregnant n = 5. Data are presented as Mean ± SE. *** p <0.001, **** p <0.0001. Please click here to view a larger version of this figure.
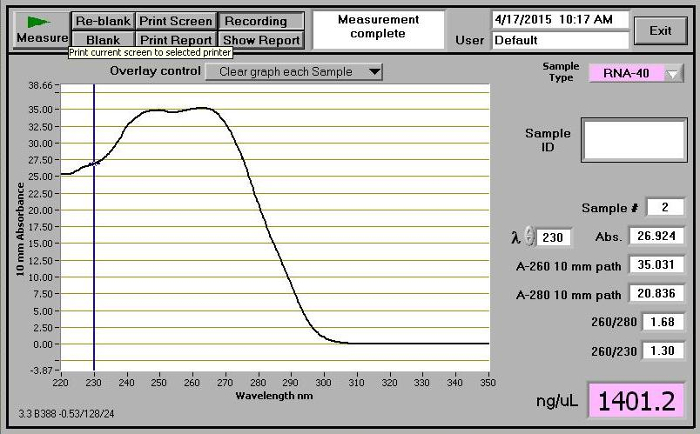 Figure 3. Absorbance measurement of poly(I:C) solution. Make sure the 260/280 ratio is between 1.54-1.82. Please click here to view a larger version of this figure.
Figure 3. Absorbance measurement of poly(I:C) solution. Make sure the 260/280 ratio is between 1.54-1.82. Please click here to view a larger version of this figure.
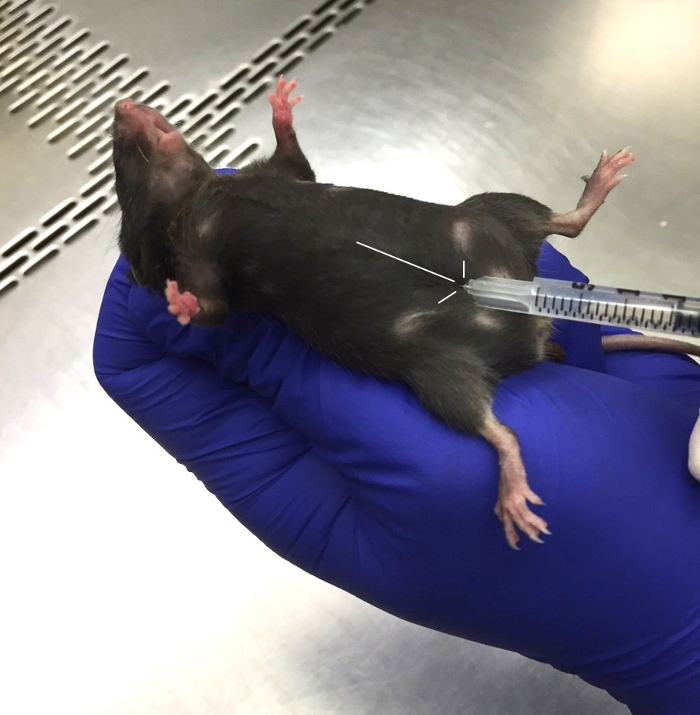 Figure 4. Poly(I:C) injection at embryonic (E) 12.5 days. Scruff the pregnant mouse gently. The injection site is located at the mid-point of the upper nipples. Inject the needle, bevel up, at approximately 20° angle relative to the mouse. Inbred C57BL/6 mouse strain is used here as an example. Please click here to view a larger version of this figure.
Figure 4. Poly(I:C) injection at embryonic (E) 12.5 days. Scruff the pregnant mouse gently. The injection site is located at the mid-point of the upper nipples. Inject the needle, bevel up, at approximately 20° angle relative to the mouse. Inbred C57BL/6 mouse strain is used here as an example. Please click here to view a larger version of this figure.
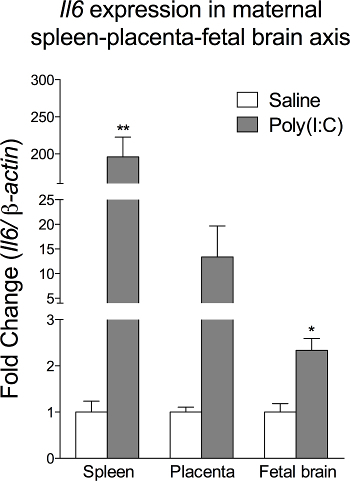 Figure 5. Poly(I:C) injection increases Il6 gene expression in the spleen-placenta-fetal brain axis of the dam. Data are presented as Mean ± SEM. * p <0.05, ** p <0.01 (The data is originally from Wu et al., 2015 and modified for the article13). Please click here to view a larger version of this figure.
Figure 5. Poly(I:C) injection increases Il6 gene expression in the spleen-placenta-fetal brain axis of the dam. Data are presented as Mean ± SEM. * p <0.05, ** p <0.01 (The data is originally from Wu et al., 2015 and modified for the article13). Please click here to view a larger version of this figure.
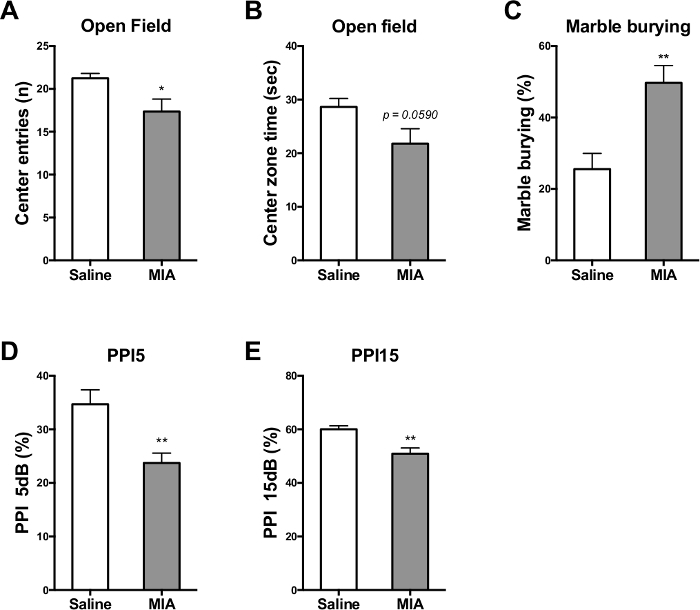 Figure 6. MIA adult offspring exhibit behavioral abnormalities associated with the endophenotypes of autism and schizophrenia. In the open-field test, MIA offspring (A) enter the center zone less frequently and (B) spend less time in the center zone. (C) In the marble burying test, MIA offspring bury more marbles than saline control offspring. MIA offspring exhibit prepulse inhibition (PPI) deficit in (D) prepulse 5 dB and (E) prepulse 15 dB. Mean ± SEM. * p <0.05, ** p <0.01 (The data is originally from Wu et al., 2015 and modified for the article13). Please click here to view a larger version of this figure.
Figure 6. MIA adult offspring exhibit behavioral abnormalities associated with the endophenotypes of autism and schizophrenia. In the open-field test, MIA offspring (A) enter the center zone less frequently and (B) spend less time in the center zone. (C) In the marble burying test, MIA offspring bury more marbles than saline control offspring. MIA offspring exhibit prepulse inhibition (PPI) deficit in (D) prepulse 5 dB and (E) prepulse 15 dB. Mean ± SEM. * p <0.05, ** p <0.01 (The data is originally from Wu et al., 2015 and modified for the article13). Please click here to view a larger version of this figure.
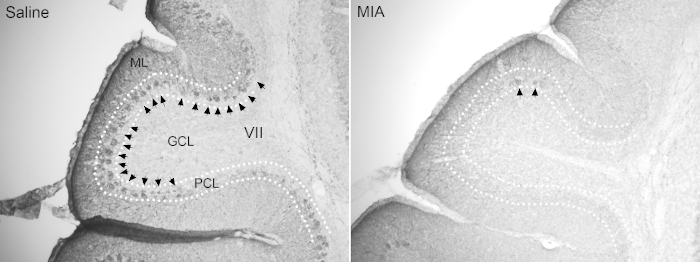 Figure 7. MIA adult offspring exhibit neuropathological abnormalities associated with the endophenotypes of autism. Loss of Purkinje cells in lobule VII has been shown in MIA adult offspring. Arrow: Calbindin+ Purkinje cells. ML: molecular layer, GCL: granule cell layer, PCL: Purkinje cell layer. Please click here to view a larger version of this figure.
Figure 7. MIA adult offspring exhibit neuropathological abnormalities associated with the endophenotypes of autism. Loss of Purkinje cells in lobule VII has been shown in MIA adult offspring. Arrow: Calbindin+ Purkinje cells. ML: molecular layer, GCL: granule cell layer, PCL: Purkinje cell layer. Please click here to view a larger version of this figure.
Discussion
MIA induction at different time windows perturbs different brain development events in rodents, and consequently leads to different behavioral abnormalities and neuropathologies in the offspring. Here, we described a protocol to induce MIA in mice with poly(I:C) injection at E12.5. This method of MIA induction leads to behavioral, neurological, immunological, and gastrointestinal abnormalities associated with autism and schizophrenia in the offspring8,10,11,16. It is important to note that, because MIA is an environmental risk factor, the phenotype of the offspring is expected to have higher variation than that of mice from genetic models of neurodevelopmental disorders. Thus, to accurately and consistently generate autism and schizophrenia-related phenotypes through MIA, it is especially important to minimize additional variables that may confound the results.
The timeline described in the protocol should be followed closely. Correct injection time window would ensure that MIA perturbs the offspring brain development at the desired stage. Induction of MIA at E12.5 in mouse corresponds to perturbations near the end of the first trimester in human gestation21- a period during which significant brain development occurs, including development of Purkinje cells and caudate putamen, and neurogenesis in the cortical layer. Further investigation will be needed to prove the association between the loss of Purkinje cells in MIA cerebellum and the early perturbation of brain development.
While consistent treatment, examination, and even husbandry of the dams would minimize the effect of maternal stress on the offspring phenotype, our protocol still contains caveats that researchers should be aware of. For example, we single house the dams the day before the injection of poly(I:C) and throughout the rest of pregnancy. Our strategy is to leave the pregnant mice in an undisturbed environment. However, this action could induce stress response in the maternal environment. Although we do not know whether this isolation-induced stress response interferes with the development of the offspring, the MIA effect can be observed by following our procedure.
In addition, the strain and genetic background of the mice needs to be chosen and tested carefully for MIA induction. So far, the effect of MIA is mostly replicated in wild-type C57BL/6N mice8,10,13,16. Others have also observed the MIA effect in BTBR mouse strain19. It has been shown that poly(I:C) could elicit different immune responses in genetically engineered mice13,22-24, which may introduce additional confounding effects to the offspring.
In terms of the number of biological replicates, we use at least 6 litters per group for behavioral or physiological assays and 3 litters per group for gene expression, protein, or immunohistochemistry approaches. We test all offspring within a litter instead of using one or two offspring within a litter to avoid experimental bias. We also average the data for all offspring within a litter, and use that average as one data point (such that n = the number of litters) to avoid inappropriate statistics13. The data from each gender are also pooled because no obvious gender difference has been observed in the MIA model13.
The rationale behind gauging MIA offspring by litter size is to minimize the variation caused by maternal nursing and caring. Litter size has been shown to correlate with stereotypic behaviors in C57BL/6 mice25. We speculate that this could be due to the assignment of resources during maternal nursing and caring. To avoid this confounding effect, our standard desired litter size is 6-10 for C57BL/6 mice.
Even when following the protocol closely, the experimenter may encounter an overly weak or excessively potent immune response in pregnant mice after poly(I:C) injection. To avoid this, it is essential to validate the pyrogenic and cytokinogenic activity of different batches of poly(I:C). Different batches and lots of poly(I:C) from the same company could lead to different levels of inflammatory response in mice; strikingly, injecting identical doses of poly(I:C) from different batches can result in up to 10 fold differences in plasma IL-6 levels26. Determining the dosage and batch of poly(I:C) for further MIA experiment by the readout of non-pregnant female IL-6 measurement could help reduce the variation of effect from MIA induction.
If it is necessary to modify the dosage for specific batches of poly(I:C), it is also important to modify the concentration of the poly(I:C) solution for injection such that the volume injected remains roughly 5 µl per gram of mouse weight. For example, according to the manufacturer's procedure, poly(I:C) is supposedly reconstituted at ~10 mg/ml of water to yield a polynucleotide in physiological phosphate buffered solution. Compared to our 40 mg/ml solution, the lower concentration would increase the volume of injection by 4 folds. For pregnant mice, a large volume of buffer injection could potentially increase the pressure on the embryos and introduce undesired, confounding effects to the offspring. Therefore, we choose to dissolve poly(I:C) powder in 0.09% sodium chloride with a higher final concentration in order to reduce the volume of injection.
IL-6 is chosen to be the marker of acute inflammatory response because prior research has identified IL-6 signaling as a critical factor in mediating the effects of MIA on brain development and behavior. Following poly(I:C) treatment, IL-6 is the most upregulated cytokine in maternal blood and placenta10,12. Most importantly, Smith et al. demonstrated that of several highly upregulated cytokines, only IL-6 exhibited both necessity and sufficiency for induction of MIA-associated brain and behavioral abnormalities. That is, maternal injection of anti-IL-6 could effectively prevent abnormal behavioral and transcriptomic abnormalities in MIA offspring, while neutralizing other cytokines did not10. Moreover, maternal injection of recombinant IL-6 alone (without poly(I:C)) reproduces MIA-associated transcriptional and behavioral abnormalities seen in the MIA fetal brain and offspring12,15.
Compared to other methods to induce MIA (e.g., lipopolysaccharide (LPS) or influenza), poly(I:C) is relatively safe and provides an easy way to control the intensity of the immune response. Although the inflammatory response elicited by maternal poly(I:C) administration is acute and transient, its effect on the offspring behaviors and brain development is profound. Overall, once the technique of inducing MIA through poly(I:C) injection at E12.5 is mastered, one can then proceed to perform different behavioral and biological assays to examine the effect of this environmental risk factor on the offspring. At the same time, one could also combine the method with genetic modifications or other treatments before, during, or after pregnancy to investigate the causes and possible therapeutic approaches to autism and schizophrenia.
Disclosures
The authors have nothing to disclose.
Acknowledgments
We would like to honor the late Dr. Paul H. Patterson for his contributions to the progress of MIA model, autism, and schizophrenia research. We acknowledge Sarkis K. Mazmanian for his great support on this protocol; Ruben M. Bayon, Yvette Garcia-Flores, Karen C. Lencioni, and Leslie A. Neumann for administrative assistance; Ali Khoshnan and Jan C. Ko for the assistance on filming; Elaine Y. Hsiao and Natalia Malkova for their advice on MIA induction; Jeffrey S. Cochrane, Joaquin Gutierrez, Kwan F. Lee, Jaime Rodriguez, Lorena C. Sandoval, and Natalie A. Verduzco for their expert animal husbandry. This work was supported by the NIH Conte Center Award (NIH 5P50MH086383-04, to Paul H. Patterson); Autism Speaks (#7670, to Paul H. Patterson); Simons Foundation (#322839, to Sarkis K. Mazmanian); NIH Training Grant (NIH/NRSA T32GM07616 to K.-H.C.); Caltech Summer Undergraduate Research Fellowship (SURF) (to Z.Y.); Amgen Scholars Program at Caltech (to Z.Y.); and Postdoctoral Fellowship from National Science Council, Taiwan (NSC 101-2917-I-564-039, to W.-L.W.).
References
- Brown AS. Epidemiologic studies of exposure to prenatal infection and risk of schizophrenia and autism. Dev Neurobiol. 2012;72(10):1272–1276. doi: 10.1002/dneu.22024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fatemi SH, et al. The viral theory of schizophrenia revisited: abnormal placental gene expression and structural changes with lack of evidence for H1N1 viral presence in placentae of infected mice or brains of exposed offspring. Neuropharmacology. 2012;62(3):1290–1298. doi: 10.1016/j.neuropharm.2011.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi L, Tu N, Patterson PH. Maternal influenza infection is likely to alter fetal brain development indirectly: the virus is not detected in the fetus. Int J Dev Neurosci. 2005;23(2-3):299–305. doi: 10.1016/j.ijdevneu.2004.05.005. [DOI] [PubMed] [Google Scholar]
- Knuesel I, et al. Maternal immune activation and abnormal brain development across CNS disorders. Nat Rev Neurol. 2014;10(11):643–660. doi: 10.1038/nrneurol.2014.187. [DOI] [PubMed] [Google Scholar]
- Meyer U. Prenatal poly(i:C) exposure and other developmental immune activation models in rodent systems. Biol Psychiatry. 2014;75(4):307–315. doi: 10.1016/j.biopsych.2013.07.011. [DOI] [PubMed] [Google Scholar]
- Meyer U, Feldon J, Yee BK. A review of the fetal brain cytokine imbalance hypothesis of schizophrenia. Schizophr Bull. 2009;35(5):959–972. doi: 10.1093/schbul/sbn022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boksa P. Effects of prenatal infection on brain development and behavior: a review of findings from animal models. Brain Behav Immun. 2010;24(6):881–897. doi: 10.1016/j.bbi.2010.03.005. [DOI] [PubMed] [Google Scholar]
- Hsiao EY, McBride SW, Chow J, Mazmanian SK, Patterson PH. Modeling an autism risk factor in mice leads to permanent immune dysregulation. Proc Natl Acad Sci U S A. 2012;109(31):12776–12781. doi: 10.1073/pnas.1202556109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito HT, Smith SE, Hsiao E, Patterson PH. Maternal immune activation alters nonspatial information processing in the hippocampus of the adult offspring. Brain Behav Immun. 2010;24(6):930–941. doi: 10.1016/j.bbi.2010.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith SE, Li J, Garbett K, Mirnics K, Patterson PH. Maternal immune activation alters fetal brain development through interleukin-6. J Neurosci. 2007;27(40):10695–10702. doi: 10.1523/JNEUROSCI.2178-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi L, et al. Activation of the maternal immune system alters cerebellar development in the offspring. Brain Behav Immun. 2009;23(1):116–123. doi: 10.1016/j.bbi.2008.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsiao EY, Patterson PH. Activation of the maternal immune system induces endocrine changes in the placenta via IL-6. Brain Behav Immun. 2011;25(4):604–615. doi: 10.1016/j.bbi.2010.12.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu WL, et al. The interaction between maternal immune activation and alpha 7 nicotinic acetylcholine receptor in regulating behaviors in the offspring. Brain Behav Immun. 2015. [DOI] [PMC free article] [PubMed]
- Garay PA, Hsiao EY, Patterson PH, McAllister AK. Maternal immune activation causes age- and region-specific changes in brain cytokines in offspring throughout development. Brain Behav Immun. 2013;31:54–68. doi: 10.1016/j.bbi.2012.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garbett KA, Hsiao EY, Kalman S, Patterson PH, Mirnics K. Effects of maternal immune activation on gene expression patterns in the fetal brain. Transl Psychiatry. 2012;2:e98. doi: 10.1038/tp.2012.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsiao EY, et al. Microbiota modulate behavioral and physiological abnormalities associated with neurodevelopmental disorders. Cell. 2013;155(7):1451–1463. doi: 10.1016/j.cell.2013.11.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naviaux RK, et al. Antipurinergic therapy corrects the autism-like features in the poly(IC) mouse model. PLoS One. 2013;8(3):e57380. doi: 10.1371/journal.pone.0057380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pratt L, Ni L, Ponzio NM, Jonakait GM. Maternal inflammation promotes fetal microglial activation and increased cholinergic expression in the fetal basal forebrain: role of interleukin-6. Pediatr Res. 2013;74(4):393–401. doi: 10.1038/pr.2013.126. [DOI] [PubMed] [Google Scholar]
- Schwartzer JJ, et al. Maternal immune activation and strain specific interactions in the development of autism-like behaviors in mice. Transl Psychiatry. 2013;3:e240. doi: 10.1038/tp.2013.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan D, et al. Long-term effects of maternal immune activation on depression-like behavior in the mouse. Transl Psychiatry. 2014;4:e363. doi: 10.1038/tp.2013.132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Workman AD, Charvet CJ, Clancy B, Darlington RB, Finlay BL. Modeling transformations of neurodevelopmental sequences across mammalian species. J Neurosci. 2013;33(17):7368–7383. doi: 10.1523/JNEUROSCI.5746-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abazyan B, et al. Prenatal interaction of mutant DISC1 and immune activation produces adult psychopathology. Biol Psychiatry. 2010;68(12):1172–1181. doi: 10.1016/j.biopsych.2010.09.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer U, et al. Adult behavioral and pharmacological dysfunctions following disruption of the fetal brain balance between pro-inflammatory and IL-10-mediated anti-inflammatory signaling. Mol Psychiatry. 2008;13(2):208–221. doi: 10.1038/sj.mp.4002042. [DOI] [PubMed] [Google Scholar]
- Vuillermot S, et al. Prenatal immune activation interacts with genetic Nurr1 deficiency in the development of attentional impairments. J Neurosci. 2012;32(2):436–451. doi: 10.1523/JNEUROSCI.4831-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bechard A, Nicholson A, Mason G. Litter size predicts adult stereotypic behavior in female laboratory mice. J Am Assoc Lab Anim Sci. 2012;51(4):407–411. [PMC free article] [PubMed] [Google Scholar]
- Harvey L, Boksa P. A stereological comparison of GAD67 and reelin expression in the hippocampal stratum oriens of offspring from two mouse models of maternal inflammation during pregnancy. Neuropharmacology. 2012;62(4):1767–1776. doi: 10.1016/j.neuropharm.2011.11.022. [DOI] [PubMed] [Google Scholar]


