Abstract
Background
Age is a strong predictor of survival in patients with coronary artery disease. In elder patients with increasing co-morbidities percutaneous coronary intervention (PCI) is associated with more complications and worse outcome. The calculation of relative survival rates adjusts for the “background” mortality in the general population by correcting for age and gender. We analyzed if elder patients after elective PCI have a worse relative survival compared to younger patient groups.
Methods
A total of 8,342 patients who underwent elective PCI at two high volume centers between 1998 and 2009 were analyzed.
Results
The survival of our patients after PCI (observed survival) was slightly lower compared to the general population (expected survival) resulting in a slightly decreasing relative survival curve. In a multivariate Cox regression model age amongst others was a strong predictor of survival. Stratifying patients according to their age the relative survival curves of younger patients (Quartile 1: <58 years; 2,046 patients), elder patients (Quartile 3: 66–73 years; 2,090 patients) and very old patients (Quartile 4: >73 years; 2,307 patients) were similar. The relative survival of mid-aged patients (Quartile 2: 58–65 years; 1,899 patients) was better than that of all other patient groups. The profile of cardiovascular risk factors differs between the various groups resulting in different composition and burden of coronary plaques in an optical coherence tomography sub-study.
Conclusion
Patients after elective PCI have a slightly worse long-term survival compared to the age- and sex-matched general population. This is also true for different groups of age except for mid-aged patients between 58 and 63 years. Elder patients between 66 and 73 years and above 73 years have a similar relative survival compared to younger patients below 58 years, and might therefore have similar benefit from elective PCI.
Introduction
Elder patients with stable coronary artery disease (CAD) often suffer from multiple co-morbidities and are consequently more fragile. Accordingly, these patients experience increased rates of in-hospital complications like death, Q wave myocardial infarction (MI), stroke, renal failure and vascular complications after Percutaneous Coronary Intervention (PCI) [1,2]. In elder patients interventional complications were demonstrated to be the strongest predictor of hospital mortality after PCI [3]. Therefor, elder patients with CAD face significantly higher initial [4,5] but also long-term mortality [4,6] after catheterization. Recent guidelines point out this higher risk of complications during and after coronary revascularization in elder patients [7].
The feasibility to perform PCI safely has improved due to new techniques and treatment strategies. Accordingly, recent studies have demonstrated substantially higher technical success rates and considerably lower acute complication rates for coronary angioplasty compared with earlier years [8–13]. This is true especially in elder patients: improvements have been achieved despite the increase of the average age of elder patients admitted for PCI and despite the growing extent of CAD in these patients [8]. As a result, an increasing number of elder patients with more severe CAD nowadays undergo treatment by PCI [3,4,14–17]. However, recent guidelines still indicate that this group of patients is undertreated and under-represented in clinical trials [7].
In general, age per se is a strong predictor of mortality as elder people are at higher risk to die than younger people. Similarly in patients with CAD, higher age besides other factors is a strong predictor of outcome [2]. Accordingly, specific statistical assumptions are prerequisite for correct comparison of outcome between younger and elder patients after elective PCI. The calculation of relative survival is a statistical method, which adjusts the mortality rate of a patient population with a certain disease for the “background” mortality in the general population, to evaluate the mortality rate caused by this certain disease. The survival rate adjusted for this background mortality is called relative survival. Thereby, this method allows comparing patients in different groups of age in regard to differences in relative survival. The aim of this study is to compare the relative survival of patients in different groups of age with stable CAD, who underwent coronary intervention.
Methods
Study population
This multicenter observational cohort study retrospectively included consecutive patients who underwent elective PCI at two high volume centers (Medical University of Vienna [MUW], University Hospital of St. Pölten) between January 1st, 1998 and December 31st, 2009. Patients with an acute coronary syndrome were excluded from our analysis. Most of these patients suffered from a STEMI or NSTEMI and underwent acute coronary angiography. However, we also excluded patients with unstable angina class I to III [18]. Patients with significant and symptomatic valve disease and significant coronary artery disease did not undergo elective PCI but were assigned to valve repair or replacement and coronary artery bypass grafting. Between 2007 and 2009 a few patients with significant aortic stenosis underwent elective PCI before Transcatheter Aortic Valve Implantation. These patients were excluded from analysis.
Experienced interventionists approved all coronary angiograms. The coronary angioplasty approach and medical treatment were consistent with contemporary practices. The survival status of all patients included in the database was prospectively retrieved from the Austrian Death Registry database (Statistik Austria) at the due day (October 31st, 2010). After clarification of the survival status, patients were anonymized in the electronic data files by receiving a unique patient identification (ID). This patient identification was used for all analyses. The study is in line with the Declaration of Helsinki and was approved by the ethics committee of the MUW and the federal state of Lower Austria in St. Pölten. According to the ethics committee an informed consent was not required because of the retrospective inclusion of patients and analysis of their data in an anonymous format.
Data collection
Eligible patients were identified in the corresponding database of each catheter laboratory (MUW—Cardio-Report; University Hospital St. Pölten—Cathlab). Study data including baseline characteristics, co-morbidities, angiographic characteristics, and interventional results were extracted from the hospital information system (Krankenhausinformationssystem—KIS). In addition, re-angiograms were documented for each patient. All data were gathered in a comprehensive database, which had been established in co-operation with the Center for Medical Statistics, Informatics, and Intelligent Systems (CeMSIIS) of the MUW using the Research Documentation and Analysis (RDA) IT-system. At the end of the follow-up period, the data from the clinical database were synchronized with the Austrian Death Registry database (Statistik Austria) to check the life status and to evaluate the date and cause of death of each patient who died. This database comprises information on every case of death in Austria.
OCT subgroup analysis—data acquisition and analysis
For plaque characterization we analyzed optical coherence tomography (OCT) images from 40 patients including 10 consecutive patients in each group of age. OCT images were performed after final stent-optimization. The OCT images were obtained using a frequency domain—OCT system (St. Jude Medical Inc., St. Paul, MN, USA). This system is equipped with a turnable laser light source with a sweep range of 1.250 to 1.370nm. The optical fibre is encapsulated within a rotating torque wire built in a rapid-exchange 2.6-F catheter. The OCT imaging catheters were delivered over a 0.014 inch (0.0356cm) guide wire through a 6-F guiding catheter. Before image acquisition OCT images were calibrated adjusting the Z-offset to obtain accurate measurements. Images were acquired using a motorized pullback system at a speed of 20mm/s during flush of 4 to 6mL/s of iso-osmolar contrast (Iodixanol 320, VisipaqueTM, GE Health Care, Cork, Ireland) through the guiding catheter to replace blood flow and permit visualization of the vessel.
Plaque characteristics were measured manually by two independent observers. If there was disagreement between the observers, a consensus was made regarding the measurements of the plaque. A lipid plaque was defined as a low signal region with diffuse border and a maximum arc of lipid ≥ 90°. This maximum arc is an excellent discriminator of fibroatheroma [19] and together a fibrous cap thickness ≤85 μm identifies thin-cap fibroatheroma when combined with a fibrous cap thickness ≤85 μm [20]. Meassurements of fibrous cap thickness were not possible, as OCT recordings were performed after stenting. A calcified plaque was defined as a signal-poor or heterogeneous region with a sharply delineated border, an arc of calcification ≥ 40°, and the absence of a lipid plaque. Plaques, presenting as a homogenous, high backscattering region, were defined as fibrous plaques. In addition, also mixed types of plaque without fulfilling the criteria for a lipid or calcified plaque were classified as fibrous plaque. The arc angle (°) was measured with a protractor centered on the lumen. For calculation of lipid, calcification, and total plaque index we measured the arc of the according plaque in every cross-sectional frame (every 200 μm) and multiplied the averaged arc by the length of the plaque. Maximal plaque thickness (mm) was measured as the maximum distance occupied by the plaque in a line extrapolated from the centre of the vessel lumen. Maximal cross-sectional area (mm2) was measured at the same place by tracing the outline of the plaque. In lipid plaques with considerable amount of fibrous content maximal cross-sectional area and maximal plaque thickness were evaluated in the fibrous section of the plaque. Examples for lipid, fibrous and calcified plaques are shown in Fig 1.
Fig 1. OCT—examples of plaque.
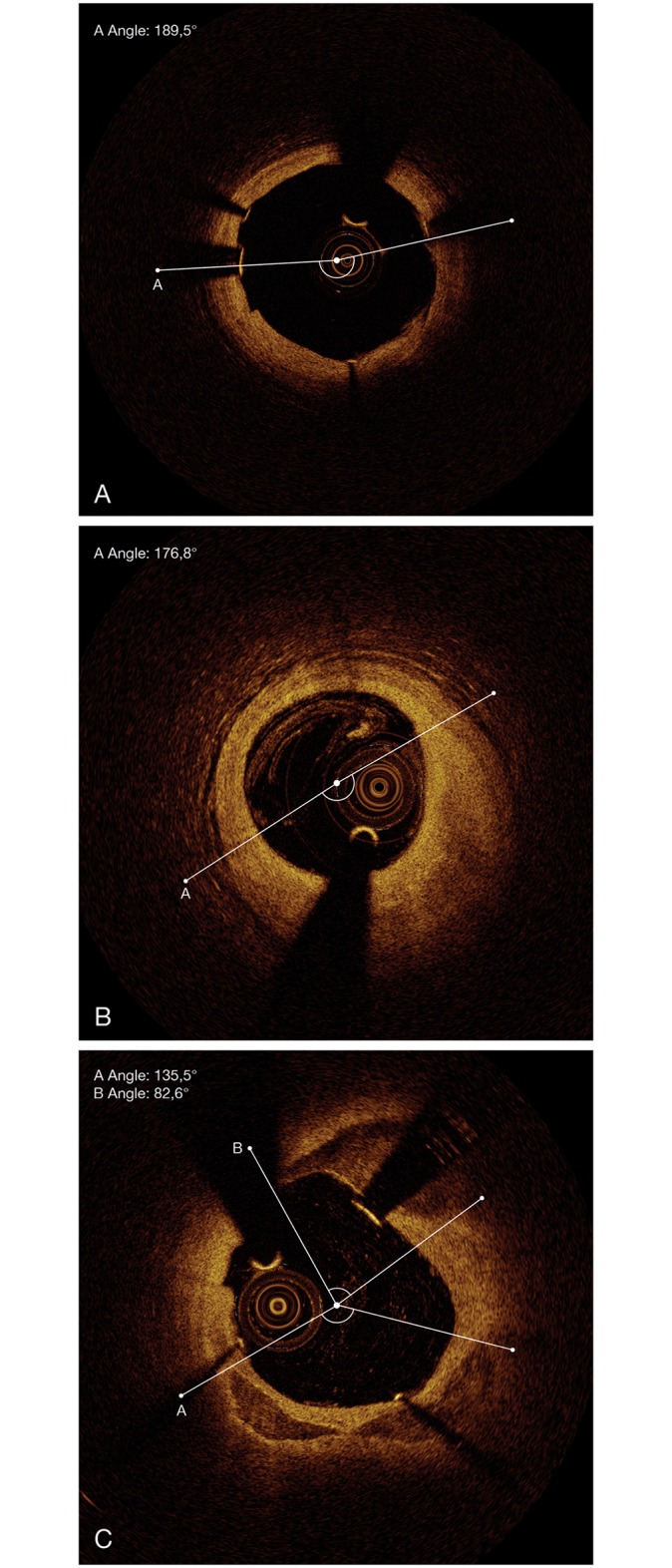
A. fibrous plaque. B. lipid plaque. C. calcified plaque.
Primary outcome
In our analysis the primary outcome was defined as survival time after elective PCI—both observed and relative (as compared with a matched normal population). Whereas observed survival gives the rate of surviving patients in an observed group, relative survival compares observed survival with the survival expected according to the age-, sex- and follow-up year-matched population estimated from the life table provided by Statistik Austria.
Statistical methods
Continuous variables are presented as mean ± SD or median with interquartile range (IQR) as appropriate. Categorical variables are presented as counts or percentages as appropriate. Continuous variables were compared using a 1-factor analysis of variance followed by a Bonferroni procedure. Ordinal data were compared using a Kruskal-Wallis test followed by Shaffer-corrected U tests. Categorical data were compared using a chi-square test. To evaluate the impact of possible risk factors on survival, univariate Cox regression models were calculated. Those risk factors, which were available in a large proportion of patients (at least 4,900 patients) and showed statistically significant influence in the univariate model, were further analyzed in a multivariate model. To compare the cohort survival with the according age and gender matched background population, multiplicative relative survival models were used. This method compares patients in different groups of age (quartiles) in regard to differences in relative survival. For this comparison, online life tables provided by Statistik Austria were used [21]. Relative survival curves according to Hakulinen's method were plotted. Analysis was performed using R 3.0.1. All p-values <0.05 were considered as statistically significant. No correction for multiplicity was done.
Results
Baseline and angiographic characteristics
A total of 8,342 patients with stable CAD, 2,944 male and 5,398 female patients with a median age of 66 years, underwent PCI in two high volume centers (MUW n = 3,906, University Hospital St. Pölten n = 4,436) during the observational period. The patient characteristics are given in Table 1. Interestingly, 24% of patients suffered from a prior MCI, 17% had a prior PCI, and 9% had a prior coronary artery bypass graft (CABG). Arterial hypertension (70% of all patients) and treated hyperlipidemia (70%) were the most common risk factors. 25% of patients had a history of diabetes, with 18% of patients suffering from non-insulin-dependent diabetes mellitus (NIDDM) and 7% suffering from insulin-dependent diabetes mellitus (IDDM). The cardiovascular medication and angiographic characteristics are listed in Tables 2 and 3. Sixty percent of the patients had a one-vessel disease, while 18% of the patients had a two- and 22% had a three-vessel disease. About one-fifth (17%) of the patients had a chronic total occlusion. Most of the patients (6,583 patients– 79%) underwent one procedure within the observation period, whereas 1,759 (21%) patients had two or more procedures (Table 3).
Table 1. Baseline.
| All, n = 8,342 | Quartile 1, n = 2,046 | Quartile 2, n = 1,899 | Quartile 3, n = 2,090 | Quartile 4, n = 2,307 | p | |
|---|---|---|---|---|---|---|
| Age, years (n), median (IQR) | (8342), 66 (58/74) | (2046), 52 (46/55) | (1899), 62 (60/64) | (2090), 70 (68/71) | (2307), 78 (76/81) | <0,0001 *†‡§¶# |
| Gender | <0.0001 *द# | |||||
| - Female, n (%) | 2944 (35) | 545 (27) | 534 (28) | 737 (35) | 1,128 (49) | |
| - Male, n (%) | 5398 (65) | 1501 (73/73/28) | 1365 (72/72/25) | 1353 (65/65/25) | 1179 (51/51/22) | |
| Height, cm (n), Median (IQR) | (6415), 171 (165/178) | (1575), 173 (166/180) | (1803), 173 (166/178) | (1616), 170 (165/176) | (1421), 168 (160/173) | <0.0001 †‡§¶# |
| Weight, kg (n), Median (IQR) | (6437), 80 (70/90) | (1577), 82 (71/94) | (1807), 83 (74/92) | (1621), 80 (71/90) | (1432), 74 (66/82) | <0.0001 †‡§¶# |
| Body-Mass-Index, kg/m2 (n), Mean±SD | (5712), 27.7±5 | (1565), 27.7±5 | (1540), 28.3±5 | (1372), 27.8±4 | (1235), 26.6±4 | <0.0001 *‡§¶# |
| Heart rate, bpm (n), Median (IQR) | (5366), 68 (60/78) | (1472), 69 (60/79) | (1440), 67 (60/78) | (1310), 68 (60/77) | (1144), 68 (60/78) | <0.246 |
| Arterial hypertension, n (%) | 4981 (70) | 1027 (56) | 1397 (71) | 1368 (78) | 1189 (75) | <0.0001 *†‡§¶# |
| Systolic blood pressure, mmHg (n), Median (IQR) | (5213), 135 (120/150) | (1415), 130 (120/140) | (1409), 134 (121/149) | (1264), 139 (125/150) | (1125), 140 (125/150) | <0.0001 *†‡§¶ |
| Diastolic blood pressure, mmHg (n), Median (IQR) | (5213), 80 (70/85) | (1415), 80 (70/88) | (1409), 80 (70/88) | (1264), 80 (70/85) | (1125), 74 (66/80) | <0.0001 †‡§¶# |
| Diabetes mellitus | ||||||
| - NIDDM, n (%) | 1255 (18) | 198 (11) | 363 (19) | 352 (20) | 342 (22) | <0.0001 *†‡¶ |
| - IDDM, n (%) | 522 (7) | 103 (6) | 138 (7) | 155 (9) | 126 (8) | 0,002 †‡§ |
| Hyperlipidemia, n (%) | 5026 (70) | 1171 (64) | 1492 (76) | 1311 (74) | 1052 (66) | <0.0001 *†¶# |
| Total cholesterol, mg/dl (n), Median (IQR) | (2489), 185 (155/217) | (595), 192 (158/222) | (666), 191 (160/221) | (627), 178 (154/212) | (601), 178 (150/213) | <0.0001 †‡§¶ |
| HDL, mg/dl (n), Median (IQR) | (1551), 47 (39/57) | (357), 45 (38/54) | (424), 47 (39/59) | (391), 48 (39/57) | (379), 48 (40/59) | 0.003 *‡ |
| LDL, mg/dl (n), Median (IQR) | (1562), 113 (86/141) | (363), 121 (92/151) | (425), 116 (88/142) | (392), 110 (83/138) | (382), 106 (83/133) | 0.002 †‡ |
| Serum-creatine,mg/dl (n), Median (IQR) | (2948), 1.03 (0.89/1.21) | (872), 0.95 (0.85/1.09) | (738), 1.02 (0.89/1.18) | (708), 1.05 (0.92/1.22) | (630), 1.14 (0.95/1.37) | <0.0001 *†‡¶# |
| Creatine-clearance **, ml/min (n), Median (IQR) | (2611), 79 (59/100) | (872), 98 (74/120) | (738), 82 (65/99) | (708), 70 (55/84) | (630), 49 (35/61) | <0.0001 *†‡§¶# |
| Smoker* | ||||||
| - current, n (%) | 1208 (17) | 556 (30) | 382 (20) | 183 (10) | 87 (6) | <0,0001 *†‡§¶# |
| - previous, n (%) | 1539 (22) | 349 (19) | 494 (25) | 402 (23) | 294 (19) | <0.0001 *†¶# |
| Family history, n (%) | 1220 (17) | 418 (23) | 324 (17) | 261 (15) | 217 (14) | <0.0001 *†‡¶ |
| Central vascular disease, n (%) | 893 (13) | 106 (6) | 217 (11) | 261 (15) | 309 (19) | <0.0001 *†‡§¶# |
| Peripheral vascular disease, n (%) | 710 (10) | 86 (5) | 175 (9) | 212 (12) | 237 (15) | <0.0001 *†‡§¶# |
| Prior myocardial infarction, n (%) | 1715 (24) | 329 (18) | 465 (24) | 454 (26) | 467 (29) | <0.0001 *†‡¶# |
| Prior PCI, n (%) | 1194 (17) | 225 (12) | 362 (19) | 357 (20) | 250 (16) | <0.0001 *†‡¶# |
| Prior coronary artery bypass graft, n (%) | 638 (9) | 60 (3) | 150 (8) | 206 (12) | 222 (14) | <0.0001 *†‡§¶ |
| Pacemaker, n (%) | 272 (4) | 21 (1) | 35 (2) | 71 (4) | 145 (9) | <0.0001 *†‡§¶# |
| CRT, n (%) | 16 (0.2) | 0 | 6 (0.3) | 5 (0.3) | 5 (0.3) | 0.134 |
| LVEF (levogram), % (n), Median (IQR) | (428), 56 (41/68) | (100), 60 (47/69) | (134), 58 (42/69) | (102), 55 (41/66) | (92), 51 (34/65) | 0.035 ‡ |
| NT-proBNP, pg/ml (n), Median (IQR) | (810), 595 (154/2259) | (208), 195 (72/1601) | (202), 373 (109/1417) | (183), 652 (251/2259) | (217), 1313 (406/4810) | <0.0001 ঠ|
| NYHA class | <0.0001 *†‡§¶# | |||||
| - Class I, n (%) | 4132 (58) | 1228 (67) | 1160 (59) | 978 (56) | 766 (48) | |
| - Class II-III, n (%) | 3017 (42) | 608 (33) | 798 (41) | 784 (45) | 827 (52) | |
| CCS class | <0.0001 *†‡§ | |||||
| - Class I, n (%) | 3508 (49) | 1001 (55) | 974 (50) | 804 (46) | 729 (46) | |
| - Class II, n (%) | 2160 (30) | 512 (28) | 577 (30) | 575 (33) | 496 (31) | |
| - Class III, n (%) | 1481 (21) | 323 (18) | 407 (21) | 383 (22) | 368 (23) |
** calculated with the Cockcroft-Gault formula.
Significant differences according to quartiles:
* = Q1 vs. Q2,
† = Q1 vs. Q3,
‡ = Q1 vs. Q4,
§ = Q2 vs. Q3,
¶ = Q2 vs. Q4,
# = Q3 vs. Q4.
Quartile 1 = <58 years;
Quartile 2 = 58–65 years;
Quartile 3 = 66–73 years;
Quartile 4 = >73 years.
Abbreviations:
CCS = canadian cardiovascular society;
CRT = cardiac resynchronization therapy;
HDL = high density lipoprotein;
IDDM = insulin dependent diabetes mellitus;
LDL = low density lipoprotein;
LVEF = left ventricular ejection fraction;
NIDDM = non insulin dependent diabetes mellitus;
NT-proBNP = n-terminal pro-brain natriuretic peptide;
NYHA = new york heart association;
p = p-value;
PCI = percutaneous coronary intervention.
Table 2. Medical therapy.
| All, n = 8,342 | Quartile 1, n = 2,046 | Quartile 2, n = 1,899 | Quartile 3, n = 2,090 | Quartile 4, n = 2,307 | p | |
|---|---|---|---|---|---|---|
| ACE inhibitor, n (%) | 3223 (52) | 733 (53) | 889 (50) | 828 (49) | 773 (52) | <0.0001 *†‡ |
| ARB, n (%) | 1504 (24) | 298 (21) | 426 (24) | 407 (25) | 373 (26) | 0.016 †‡ |
| Aldosteron-antagonist, n (%) | 577 (9) | 101 (7) | 122 (7) | 139 (9) | 215 (15) | <0.0001 †‡§¶# |
| Beta-blocker, n (%) | 4166 (67) | 954 (76) | 1194 (64) | 1086 (65) | 932 (63) | <0.0001 *†‡ |
| Statin, n (%) | 4706 (71) | 1080 (66) | 1420 (76) | 1207 (72) | 999 (68) | <0.0001 *†§¶# |
| Aspirin, n (%) | 7001 (98) | 1813 (99) | 1323 (98) | 1712 (97) | 1553 (98) | 0.004 †‡§ |
| P2Y12 inhibitor, n (%) | 7010 (98) | 1800 (98) | 1922 (98) | 1729 (98) | 1559 (98) | 0.926 |
| Dual antiplatelet therapy, n (%) | 6866 (96) | 1800 (98) | 1868 (95) | 1679 (95) | 1519 (95) | <0.0001 *†‡ |
| Calcium-channal blocker, n (%) | 1637 (26) | 291 (19) | 489 (27) | 438 (27) | 419 (29) | <0.0001 *†‡ |
| Nitrates, n (%) | 1326 (22) | 191 (13) | 351 (20) | 381 (25) | 403 (29) | <0.0001 *†‡§¶# |
Significant differences according to quartiles:
* = Q1 vs. Q2,
† = Q1 vs. Q3,
‡ = Q1 vs. Q4,
§ = Q2 vs. Q3,
¶ = Q2 vs. Q4,
# = Q3 vs. Q4.
Quartile 1 = <58 years;
Quartile 2 = 58–65 years;
Quartile 3 = 66–73 years;
Quartile 4 = >73 years.
Abbreviations:
ACE = angiotensin-converting-enzyme;
ARB = angiotensin receptor blocker;
p = p-value.
Table 3. Angiographic characteristics.
| All, n = 8,342 | Quartile 1, n = 2,046 | Quartile 2, n = 1,899 | Quartile 3, n = 2,090 | Quartile 4, n = 2,307 | p | |
|---|---|---|---|---|---|---|
| Vessel disease | 0.482 | |||||
| - 1VD, n (%) | 4292 (60) | 1130 (62) | 1149 (59) | 1066 (61) | 947 (59) | |
| - 2VD, n (%) | 1270 (18) | 313 (17) | 375 (19) | 302 (17) | 280 (18) | |
| - 3VD, n (%) | 1587 (22) | 393 (21) | 434 (22) | 394 (22) | 366 (23) | |
| Chronic total occlusion, n (%) | 1203 (17) | 287 (16) | 338 (17) | 308 (18) | 270 (17) | 0.441 |
| Number of procedures | 0.032 ‡¶# | |||||
| - one, n (%) | 6,583 (79) | 1,599 (78) | 1,486 (78) | 1,620 (78) | 1,878 (81) | |
| - two, n (%) | 1,490 (18) | 372 (18) | 349 (18) | 399 (19) | 370 (16) | |
| - > two, n (%) | 269 (3) | 75 (4) | 64 (4) | 71 (3) | 59 (3) | |
| Patients treated with | ||||||
| - drug-eluting stent, n (%) | 1699 (80) | 347 (84) | 470 (79) | 465 (80) | 417 (78) | 0,142 |
| - bare-metal stent, n (%) | 427 (20) | 67 (16) | 126 (21) | 117 (20) | 117 (22) | 0,142 |
| Troponin T, ng/ml | ||||||
| - before PCI (n), Median (IQR) | (144), 0.03 (0.01/0.05) | (32), 0.02 (0/0.06) | (31), 0.02 (0/0.05) | (32), 0.03 (0.02/0.04) | (49), 0.04 (0.01/0.05) | 0.771 |
| - day 1 after PCI (n), Median (IQR) | (144), 0.03 (0.01/ 0.06) | (32), 0.04 (0.01/0.07) | (31), 0.03 (0.02/0.05) | (32), 0.03 (0.02/0.05) | (49), 0.03 (0.01/0.06) | 0.277 |
| Creatine kinase, U/l | ||||||
| - Before PCI (n), Median (IQR) | (1169), 79 (52/115) | (276), 84 (56/119) | (280), 87 (58/117) | (296), 83 (52/117) | (317), 68 (46/105) | 0.560 |
| - Day 1 after PCI (n), Median (IQR) | (1161), 66 (46/98) | (276), 69 (48/101) | (280), 68 (49/107) | (296), 69 (45/97) | (317), 62 (43/91) | 0.878 |
| Re-catheterizations | 0.291 | |||||
| - < 30 days, n (%) | 610 (7) | 130 (6) | 125 (7) | 166 (8) | 189 (8) | |
| - 30days– 6 months, n (%) | 387 (5) | 102 (5) | 84 (4) | 111 (5) | 90 (4) | |
| - 6 months– 1 year, n (%) | 258 (3) | 79 (4) | 62 (3) | 59 (3) | 58 (3) | |
| - > 1 year, n (%) | 504 (6) | 136 (7) | 142 (8) | 134 (6) | 92 (4) |
Significant differences according to quartiles:
‡ = Q1 vs. Q4,
¶ = Q2 vs. Q4,
# = Q3 vs. Q4.
Quartile 1 = <58 years;
Quartile 2 = 58–65 years;
Quartile 3 = 66–73 years;
Quartile 4 = >73 years.
Abbreviations:
p = p-value;
VD = vessel disease.
OCT substudy
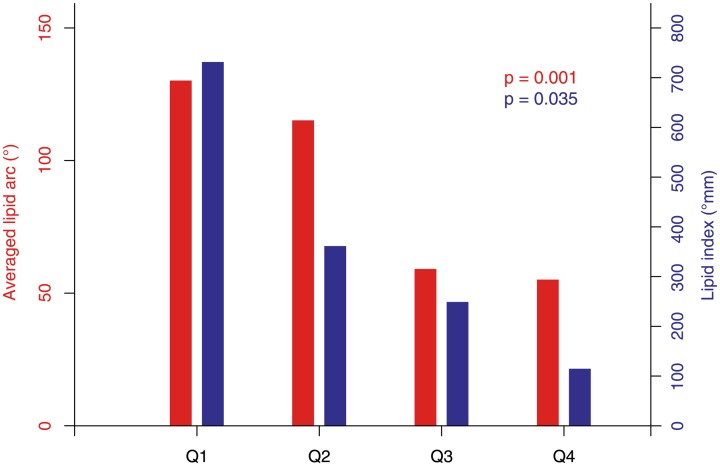
Lipid plaques are most frequent in the youngest patient group. Incidence and size of lipid plaques are decreasing with age (Fig 2). In contrast, the presence of calcification as well as the total amount of plaque burden is increasing with age. Accordingly, the highest amount of calcified plaques, as well as of total plaque burden, was detected in the oldest patient group (Table 4).
Fig 2. Averaged lipid arc and lipid index.
Averaged lipid arc and lipid index stratified according to age into four quartiles.
Table 4. OCT analysis.
| All, n = 40 | Quartile 1, n = 10 | Quartile 2, n = 10 | Quartile 3, n = 10 | Quartile 4, n = 10 | p | |
|---|---|---|---|---|---|---|
| Type of plaque | ||||||
| - LP/ FP/CP (n) | 17/12/11 | 9/1/0 | 6/3/1 | 0/6/4 | 2/2/6 | <0.0001 †‡§ |
| Lipid content | ||||||
| - Present (n) | 27 | 10 | 6 | 5 | 6 | 0,081 |
| - Averaged lipid arc (°, Mean±SD) | 97±45 | 130±36 | 115±38 | 59±18 | 55±14 | <0.0001 †‡§¶ |
| - Lipid index (°mm, Mean±SD) | 514±522 | 729±753 | 360±392 | 248±279 | 114±167 | 0.017 |
| Calcification | ||||||
| - Present (n) | 16 | 2 | 2 | 4 | 8 | 0,019 ঠ|
| - Averaged calcification arc (°, Mean±SD) | 53±15 | 53±13 | 44±14 | 62±21 | 51±12 | 0.566 |
| - Calcification index (°mm, Mean±SD) | 55±15 | 54±13 | 47±14 | 64±22 | 53±13 | 0.599 |
| Total plaque extent | ||||||
| - Averaged arc of total plaque (°, Mean±SD) | 174±52 | 156±53 | 146±43 | 193±32 | 197±62 | 0.054 |
| - Total plaque index, (°mm, Mean±SD) | 2314±1894 | 1989±1458 | 1599±1142 | 2222±1238 | 3387±2797 | 0.155 |
| - Maximal area (mm2, Mean±SD) | 3.79±1.53 | 3.45±2.01 | 2.68±0.88 | 4.11±0.96 | 4.87±1.35 | 0.006 ¶ |
| - Maximal thickness (mm2, Mean±SD) | 0.79±0.21 | 0.68±0,17 | 0.64±0.13 | 0.87±0.18 | 0.94±0.19 | 0.001 †‡§¶ |
Significant differences according to quartiles:
† = Q1 vs. Q3,
‡ = Q1 vs. Q4,
§ = Q2 vs. Q3,
¶ = Q2 vs. Q4.
Quartile 1 = <58 years;
Quartile 2 = 58–65 years;
Quartile 3 = 66–73 years;
Quartile 4 = >73 years.
Abbreviations:
CP = calcified plaque;
FP = fibrous plaque;
LP = lipid plaque;
p = p-value.
Mortality
During the observation period 2,009 (24%) patients died. The most common causes of death were cardiovascular (56%) and cancer (18%). Of course, mortality rate rose with increasing age at a death rate from 13% (in patients aged less than 58 years) up to 44% (in elder patients aged over 73 years). Interestingly, the incidence of cardiovascular death and death from cancer was similar in all groups of age (Table 5).
Table 5. Cause of death **.
| All, n = 8,342 | Quartile 1, n = 2,046 | Quartile 2, n = 1,899 | Quartile 3, n = 2,090 | Quartile 4, n = 2,307 | p | |
|---|---|---|---|---|---|---|
| Total death, n (%) | 2,009 (24) | 257 (13) | 351 (19) | 516 (25) | 885 (38) | <0,0001 *†‡§¶# |
| Cardiovascular, n (%) | 1,132 (56) | 132 (52) | 200 (57) | 276 (53) | 524 (59) | 0,063 |
| Cancer, n (%) | 365 (18) | 42 (16) | 68 (19) | 106 (21) | 149 (17) | 0,269 |
| Gastrointestinal, n (%) | 45 (2) | 8 (13) | 7 (2) | 12 (2) | 18 (2) | 0,758 |
| Respiratory, n (%) | 86 (4) | 11 (4) | 12 (3) | 25 (5) | 38 (4) | 0,792 |
| Non-natural, n (%) | 73 (4) | 14 (6) | 9 (3) | 16 (3) | 34 (4) | 0,252 |
| Other, n (%) | 308 (16) | 50 (19) | 55 (16) | 81 (16) | 122 (14) | 0,166 |
** Percentages of total death refer to the number of all patients in the corresponding study group. Percentages of different causes of death refer to the number of total deaths in the corresponding study group.
Significant differences according to quartiles:
* = Q1 vs. Q2,
† = Q1 vs. Q3,
‡ = Q1 vs. Q4,
§ = Q2 vs. Q3,
¶ = Q2 vs. Q4,
# = Q3 vs. Q4.
Quartile 1 = <58 years;
Quartile 2 = 58–65 years;
Quartile 3 = 66–73 years;
Quartile 4 = >73 years.
Abbreviations:
p = p-value.
Univariate and multivariate results
Table 6 lists the results of the univariate analysis. In a multivariate model (Table 7) age was the most important predictor of survival. The corresponding Hazard ratio (HR) of 1.043 means that an additional year of age increases the Hazard by 4.3%. Furthermore, the presence of peripheral vascular disease, IDDM, and NIDDM, prior MI, central vascular disease, and prior CABG increase the Hazard as well. In contrast, treated hyperlipidemia, family history, and prior PCI reduce the Hazard. Interestingly, a positive family history is associated with a better survival. This phenomenon may result from the fact that especially younger patients are characterized by a positive family history, and younger patients per se have a better survival than elder patients.
Table 6. Univariate results.
| HR | low95 | up95 | p | |
|---|---|---|---|---|
| Age | 1.039 | 1.034 | 1.045 | <0.0001 |
| Gender | 1.068 | 0.956 | 1.193 | 0.242 |
| Height | 0.553 | 0.303 | 1.012 | 0.055 |
| Weight | 0.995 | 0.992 | 0.999 | 0.015 |
| Body mass index | 0.983 | 0.969 | 0.997 | 0.017 |
| Heart rate, bpm | 1.015 | 1.011 | 1.018 | <0.0001 |
| Arterial hypertension | 1.041 | 0.930 | 1.165 | 0.482 |
| Systolic blood pressure, mmHg | 0.998 | 0.995 | 1.001 | 0.285 |
| Diastolic blood pressure, mmHg | 0.988 | 0.983 | 0.993 | <0.0001 |
| Diabetes mellitus—NIDDM | 1.277 | 1.124 | 1.452 | <0.0001 |
| Diabetes mellitus—IDDM | 1.564 | 1.321 | 1.853 | <0.0001 |
| Treated hyperlipidemia | 0.799 | 0.716 | 0.891 | <0.0001 |
| Smoker—current | 0.868 | 0.749 | 1.006 | 0.059 |
| Smoker—previous | 1.116 | 0.988 | 1.262 | 0.078 |
| Family history | 0.702 | 0.596 | 0.826 | <0.0001 |
| Central vascular disease | 1.494 | 1.303 | 1.713 | <0.0001 |
| Peripheral vascular disease, n (%)* | 2.009 | 1.751 | 2.305 | <0.0001 |
| Prior myocardial infarction, n (%)* | 1.219 | 1.088 | 1.365 | 0.001 |
| Prior PCI, n (%)* | 1.009 | 0.883 | 1.153 | 0.892 |
| Prior coronary artery bypass graft, n (%)* | 1.277 | 1.089 | 1.498 | 0.003 |
| PM, n (%)* | 1.620 | 1.295 | 2.027 | <0.0001 |
| CRT, n (%) | 2.452 | 1.167 | 5.154 | 0.018 |
| NYHA class | 1.236 | 1.114 | 1.371 | <0.0001 |
| CCS class | 0.894 | 0.835 | 0.956 | 0.001 |
| ACE inhibitor | 1.078 | 0.966 | 1.202 | 0.179 |
| ARB | 1.048 | 0.914 | 1.202 | 0.504 |
| ACE inhibitor or ARB | 1.138 | 1.014 | 1.276 | 0.028 |
| Aldosteron-antagonist | 2.129 | 1.815 | 2.497 | <0.0001 |
| Beta-blocker | 0.938 | 0.838 | 1.050 | 0.265 |
| Statin | 0.826 | 0.736 | 0.927 | 0.001 |
| Aspirin | 0.968 | 0.722 | 1.298 | 0.828 |
| P2Y12 inhibitor | 3.019 | 1.436 | 6.349 | 0.004 |
| Dual antiplatelet therapy | 1.138 | 0.880 | 1.473 | 0.324 |
| Calcium-channel blocker | 1.071 | 0.947 | 1.211 | 0.272 |
| Nitrates | 1.188 | 1.050 | 1.344 | 0.006 |
| Vessel disease | 1.026 | 0.965 | 1.092 | 0.409 |
| Chronic total occlusion | 1.177 | 1.033 | 1.342 | 0.014 |
| Number of procedures | 0.938 | 0.873 | 1.007 | 0.079 |
| Re-catheterizations | 0.945 | 0.912 | 0.980 | 0.002 |
Abbreviations:
ACE = angiotensin-converting-enzyme;
ARB = angiotensin receptor blocker;
CCS = canadian cardiovascular society;
CRT = cardiac resynchronization therapy;
HR = hazard ratio;
IDDM = insulin dependent diabetes mellitus;
Low95 = lower bound for 95% confidence interval;
LVEF = left ventricular ejection fraction;
NIDDM = non insulin dependent diabetes mellitus;
NT-proBNP = n-terminal pro-brain natriuretic peptide;
NYHA = new york heart association;
p = p-value;
PCI = percutaneous coronary interventio;
PM = pacemaker;
Up95 = upper bound for 95% confidence interval.
Table 7. Multivariate results.
| HR | low95 | up95 | p | |
|---|---|---|---|---|
| Age | 1.033 | 1.024 | 1.041 | <0.0001 |
| Peripheral vascular disease | 1.886 | 1.547 | 2.300 | <0.0001 |
| Heart rate | 1.011 | 1.007 | 1.016 | <0.0001 |
| Aldosterone antagonists | 1.405 | 1.121 | 1.761 | 0.003 |
| Treated hyperlipidemia | 0.805 | 0.679 | 0.953 | 0.012 |
| NYHA class | 1.282 | 1.096 | 1.500 | 0.002 |
| Diastolic blood pressure | 0.992 | 0.985 | 0.998 | 0.013 |
| CCS class | 0.881 | 0.795 | 0.978 | 0.170 |
| Pacemaker | 1.382 | 1.030 | 1.855 | 0.031 |
| Re-catheterization | 0.944 | 0.895 | 0.996 | 0.034 |
| Family history | 0.795 | 0.634 | 0.996 | 0.046 |
Abbreviations:
CCS = canadian cardiovascular society;
HR = hazard ratio;
Low95 = lower bound for 95% confidence interval;
NYHA = new york heart association;
p = p-value;
Up95 = upper bound for 95% confidence interval.
Relative survival
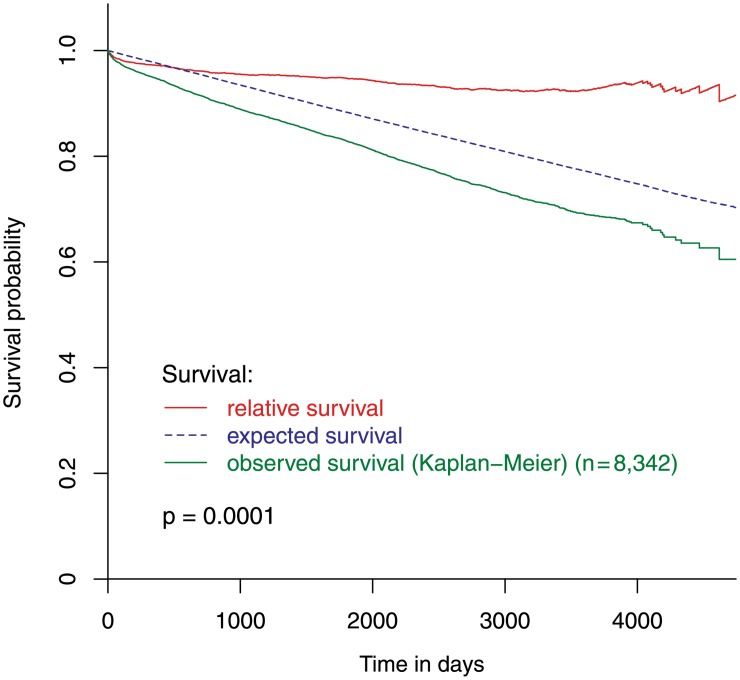
The observed patient survival was worse than the expected survival based on age- and gender-matched background population (p<0.001). The difference between these two groups continuously increased until the end of the observed time period. Hence, the relative survival curve starts almost horizontally and only slightly decreases afterwards (Fig 3).
Fig 3. Observed, expected and relative survival of all study-patients.
The observed survival of all study-patients, the expected survival of the sex- and age-matched general population, and the resulting relative survival of all study-patients.
Baseline and angiographic characteristics according to age
Patients were stratified according to their age by using quartiles. Quartile 1 (Q1: <58 years) comprised 2,046 patients, Quartile 2 (Q2: 58–65 years) 1,899 patients, Quartile 3 (Q3: 66–73 years) 2,090 patients, and Quartile 4 (Q4: >73 years) 2,307 patients. Baseline characteristics are given in Table 1. Interestingly, Quartile 1 includes the highest proportion of patients with positive family history (23%). Younger patients are highly characterized by having more current smokers and having a higher level of total and LDL cholesterol, whereas elder patients have a higher incidence of diabetes, elevated blood pressure, and chronic renal impairment (Table 1). Table 3 shows the angiographic characteristics according to age groups. Of note, the severity of CAD was distributed relatively homogenous across age groups with about 50% of patients suffering from a one-vessel disease and about 25% of patients suffering from a two- and three-vessel disease.
Mortality according to age
In all quartiles cardiovascular death was the most common cause of death. As expected, most people died in Quartile 4, however the proportion between cardiovascular death and other causes of death was similar in each group. The exact figures are shown in Table 5.
Observed and relative survival according to age
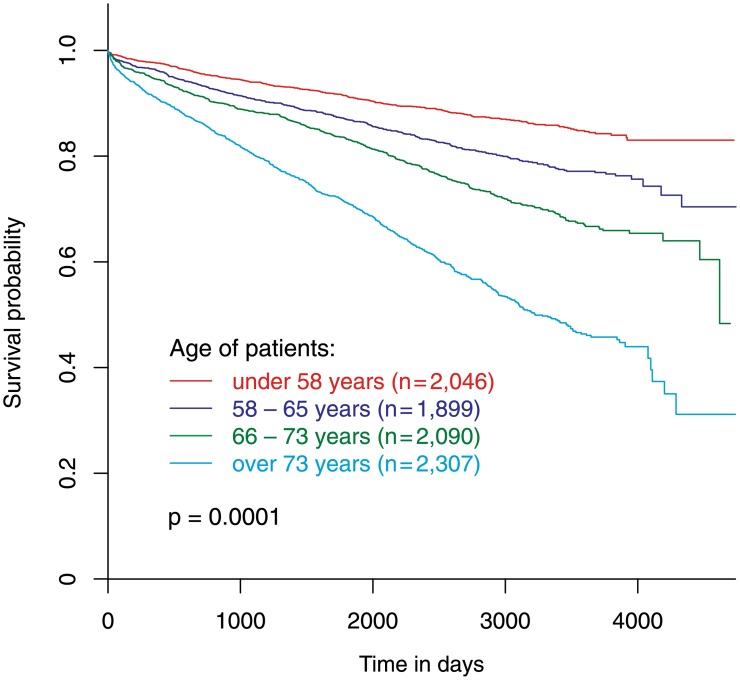
In a multivariate Cox regression model age was the most important predictor of survival (Table 7). Large differences in observed survival can be found between different age groups (p = 0.001) (Fig 4). Even the second age quartile has a statistically significant worse survival than the first age quartile (the youngest patient group) with a hazard ratio of 1.59 (p<0.001). Third age quartile and forth age quartile show hazard ratios of 2.29 (p<0.001) and 4.29 (p<0.001), respectively.
Fig 4. Observed survival according to age quartiles.
Observed survival of patients stratified according to age into four quartiles.
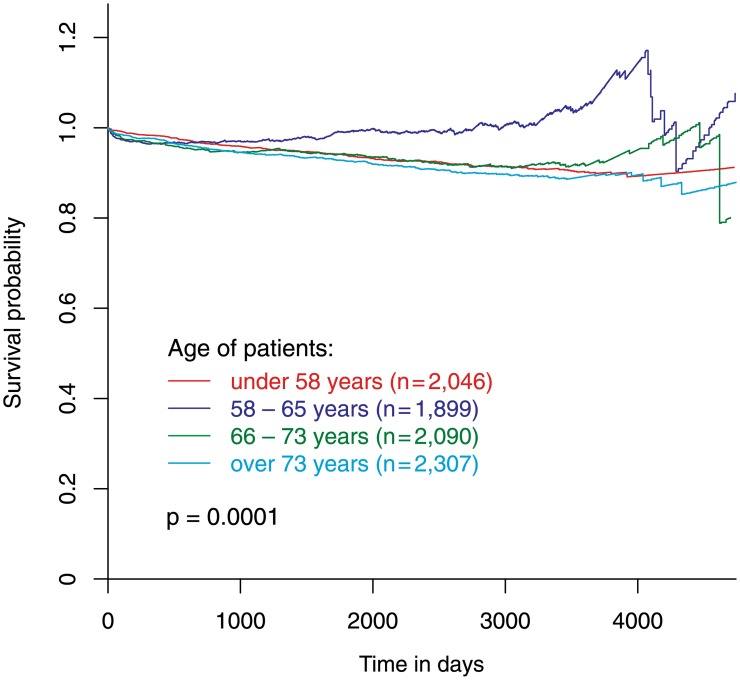
The relative survival of the younger population (Q1: <58 years, 2,046 patients), the elder population (Q3: 66–73 years, 2,090 patients) and the very old population (Q4: >73 years, 2,307 patients) was similar. The relative survival of mid-aged patients (Q2: 58–65 years, 1,899 patients) was significantly better (p = 0.001) than the relative survival of all other patient groups (Fig 5).
Fig 5. Relative survival according to age quartiles.
Relative survival of patients stratified according to age into four quartiles.
Discussion
Our analysis indicates that patients with stable CAD, who undergo elective PCI, have a slightly worse survival (observed) than the general population (expected survival). In our patients mortality-risk continuously increased with age similar to the general population. When stratifying patients according to their age, the relative survival of mid-aged patients between 58 and 65 years was better than the relative survival of younger and elder patient groups (Fig 5). The relative survival of elder patients—between 66 and 73 years and above 73 years—was similar to the relative survival of younger patients below 58 years suggesting a similar benefit from PCI in these patient groups (Fig 5). These differences are paralleled by different profiles of cardiovascular risk factors and consecutive different patterns of plaque morphology.
Cardiovascular risk factors and plaque morphology
Cardiovascular risk factors differ considerably between various groups of age. Younger patients are highly characterized by having more current smokers and having a higher level of total and LDL cholesterol. This risk profile in younger patients is also documented by other authors [22,23]. Their higher genetic predisposition for coronary artery disease is reflected by a higher incidence of positive family history. In contrast, elder patients are characterized by a distinct pattern of cardiovascular risk factors with a higher incidence of diabetes, elevated blood pressure, and chronic renal impairment. However, they have lower total and LDL-cholesterol levels and are less frequently current smokers. A similar distribution of risk factors was recently documented in patients with ACS [24].
Recent analyses showed, that specific risk factors cause certain plaque characteristics. Current smokers are more likely to have lipid plaques and OCT-defined vulnerable plaques [25]. Similarly, LDL cholesterol levels are positively correlated to the extent of lipid plaques [26]. On the other hand, chronic renal impairment and diabetes are accompanied by an increased plaque burden with higher calcium content [27,28]. These findings confirm the results of our OCT sub-analysis: younger patients, who were more frequently smokers and had elevated total and LDL-cholesterol levels, had higher rates of large lipid plaques compared to elder patients. These plaques, also called fibroatheroma, include thin-cap atheroma and may cause acute coronary syndrome when rupturing. Elder patients with higher rates of diabetes and chronic renal impairment were characterized by a considerably lower rate of large lipid plaques but a higher rate of calcified plaques and an increased plaque burden.
Plaque morphology and age
Age-dependent differences in plaque composition have already been described by some authors: several analyses indicate that plaque burden and calcium content increase with age [24,29,30]. These findings are confirmed in our analysis. Moreover, the incidence of thin-cap fibroatheroma (TCFA), defined as a necrotic core (lipid plaque) situated next to the vessel lumen, may change with age. In the PROSPECT study, which used virtual-histology intravascular ultrasound (VH-IVUS) for identification of a necrotic core (lipid pool), thickcap-FA increased in patients above 65 years, whereas TCFA were, in trend, more frequent in patients below 65 years [24]. However, VH-IVUS has been reported to overestimate the extent of a lipid plaque [31,32]. A fact, that may especially apply for plaques with multiple components, typically found in the elderly. In contrast, OCT has a higher sensitivity (90% to 92%) to identify lipid-rich plaques [33]. Accordingly, OCT analyses found out that thick-cap FA consists of considerably smaller lipid pools but a higher amount of fibrosis and calcification area compared to TCFA [34]. OCT criteria for a TCFA include the presence of a lipid plaque with an arc > 90° and a thin fibrous cap [35]. Recent data [36] documented that an arc of lipid >80° can excellently identify TCFA (area under the curve 0.86). In accordance with these findings, we found significant lipid plaques—suspicious to be a TCFA—mainly in younger (<58 years) and mid-aged patients (58–65 years). Elder patients predominantly had smaller lipid plaques, not fulfilling the criteria for a TCFA, embedded in large mixed plaques with fibrosis and frequent calcific components. Two reports, that compared plaque morphology in younger and elder patient groups, found similar results [36,37].
Possible explanations for age-dependent differences in relative survival
In younger patients a more aggressive course of disease can be assumed, as their coronary artery disease occurs earlier during life-time. Due to a high rate of positive family history a genetic disposition is likely in many of these patients [38,39]. Compared to elder patients, higher cholesterol levels and a higher incidence of smoking result in larger lipid plaques suspicious to be TCFA. Few data reported that this patient group has a worse long-term survival [23,40], as also confirmed by our analysis.
In elder patients the greater plaque burden indicates a more advanced stage of disease and is accompanied with a higher incidence of comorbidities like peripheral vascular disease, central vascular disease, prior MI, and prior CABG. This finding is well documented in other large observational studies [41,42], presumably resulting from a long history of various cardiovascular risk factors, and may enlarge the survival gap between CAD patients and the general population. Despite these unfavorable factors on long-term outcome, elder patients have a similar relative survival compared to our younger patient group. Accordingly, elective PCI can be suggested to be a useful tool to treat these patients. Another factor may contribute to the better relative survival in these patients. The acute coronary syndrome (ACS) represents a major complication during the career of a patient with CAD. Current guidelines support the use of an early invasive strategy (early in-hospital coronary angiography followed by revascularization, if appropriate) for patients with moderate-risk and high-risk NSTEMI, rather than a selective approach based on recurrent symptoms or evidence of ongoing ischemia [43,44]. Interestingly, the risk for adverse events with an early instead of a selective invasive approach decreases with age [45,46]. Accordingly, patients aged above 75 years gain the greatest survival benefit from the use of an early invasive approach as demonstrated recently in an analysis of a large database including more than 11,000 patients [47]. However, the same analysis showed that an age above 75 years was the strongest negative predictor for the use of cardiac catheterization. The underestimation of risk by physicians was the most common reason for choosing a conservative strategy. During the long-term follow-up of our patients this factor may worsen the outcome in elder patient groups compared to the younger.
Mid-aged patients between 58 and 65 years have a less aggressive severity of disease compared to younger patients, a less advanced stage of disease and is accompanied with a higher incidence of comorbidities. These patients may gain the most benefit from treatment of their lipid plaques. Compared with medical treatment alone, PCI reduces the subsequent risk of spontaneous MI in patients with CAD [48,49]. As the incidence of STEMI has an age-peek between 55 and 70 years and rapidly decreases thereafter [50], patients in this group of age, with the highest risk to suffer from a STEMI, may benefit most from elective PCI. Our data support this suggestion.
Cause of death
The cardiovascular mortality of 56% in our study is in accordance with other studies, which report death-rates due to cardiac causes (not cardiovascular) of below or equal to 50% in patients with stable coronary artery disease [51,52]. When comparing the main causes of death including cardiovascular causes, cancer, gastro-intestinal diseases, diseases of the respiratory system, non-natural and other causes between our study population and the general population [21], the distribution of these causes is rather similar except the following two: Interestingly, less study patients died from cancer compared to the general population (18% versus 26%). One explanation for this finding may be the fact that, not surprisingly, our study population more frequently died from cardiovascular reasons, and therefore had a lower chance to experience cancer compared to the general population. In addition, the statin therapy in patients with CAD may contribute to the lower rate of cancer death [53]. Similar results, a lower death rate from cancer and a higher rate of cardiovascular deaths were reported by the authors of the GENetic DEterminants of Restenosis (GENDER) study, who also compared CAD patients after elective PCI and the general population [54].
Limitations
As a limitation of our analysis our control group, the general population of Austria, also includes patients with stable CAD. Some of these patients do not seek help from a doctor and others are treated medically and are not sent to the catheter laboratory. These patients may have a worse prognosis to patients treated in the catheter laboratory and compared to the general population. Moreover, some subjects in the general population also underwent PCI in other catheter laboratories. This part of the control group can also be suggested to have a worse prognosis than the general population and a similar prognosis to our study population. Despite of these patients, the mortality rate of our patient population was slightly higher compared to the general population. However, the main message from our analysis results from the comparison of the relative survival between different groups of age. This comparison is not influenced by the background noise of CAD patients in the general population as this background noise can be suggested to be similar in all groups. In addition, the long follow-up includes patients with constant changes in the therapeutic and interventional strategy. On average, medical therapy of our patients is comparable to treatment regimen in studies dedicated to provide optimized medical treatment to these patients [51]. Eighty percent of our patients had been treated with drug-eluting stents and only 20% received bare-metal stents. It can not be excluded that a higher rate of drug-eluting-stents may slightly reduce the survival disadvantage of patients who undergo elective PCI compared to the total population. Accordingly, a potential improvement in survival may also slightly reduce the relative survival benefit of mid-aged patients compared to younger and elder patients. Upcoming new treatment options for additional lipid-lowering may have a more important impact. Our analysis emphasizes the necessity for therapeutic improvement.
Clinical implications
Younger patients below 58 years (median 52 years) with stable coronary artery disease suffer from a more aggressive type of disease with higher total and LDL cholesterol levels and larger lipid plaques than elder patients. More aggressive lipid lowering strategies should be implemented especially in these patients to improve survival. Further studies will evaluate if screening for lipid plaques (non-invasive or invasive) will be reasonable in these patients. Elder patients suffer from a distinct type of coronary artery disease with higher blood pressure levels, higher rates of diabetes and chronic renal impairment. These patients are characterized by fewer and smaller lipid plaques but more calcification and greater plaque burden associated with late complications like peripheral vascular disease, central vascular disease, or prior MI. Although a large network meta-analysis including more than 90,000 mid-aged patients (median age 62, interquartile range 51/69) provided evidence for improved survival with new generation drug eluting stents compared with medical treatment alone [55], the benefit of PCI in elder patients is unclear. Our analysis clearly demonstrate that elder patients with stable CAD who undergo PCI have a similar relative survival like younger patients. Mid-aged CAD patients have a similar survival compared to the age-matched general population and a slightly better outcome than younger and elder patient groups. These patients can be suggested to benefit most from elective PCI, as they are at high risk to experience a STEMI due to a higher probability to have a vulnerable thin-cap atheroma and elective PCI may be an effective treatment to prevent this event [48,49].
Data Availability
All relevant data are available in the paper.
Funding Statement
All authors have reported that they have no relationships relevant to the contents of this paper to disclose. Furthermore the authors received no specific funding for this work.
References
- 1.Batchelor WB, Anstrom KJ, Muhlbaier LH, Grosswald R, Weintraub WS, O'Neill WW, et al. Contemporary outcome trends in the elderly undergoing percutaneous coronary interventions: results in 7,472 octogenarians. J Am Coll Cardiol. 2000. September;36(3):723–30. . [DOI] [PubMed] [Google Scholar]
- 2.Klein LW, Block P, Brindis RG, McKay CR, McCallister BD, Wolk M, et al. Percutaneous coronary interventions in octogenarians in the American College of Cardiology-National Cardiovascular Data Registry: development of a nomogram predictive of in-hospital mortality. J Am Coll Cardiol. 2002. August 7;40(3):394–402. . [DOI] [PubMed] [Google Scholar]
- 3.Bauer T, Mollmann H, Weidinger F, Zeymer U, Seabra-Gomes R, Eberli F, et al. Predictors of hospital mortality in the elderly undergoing percutaneous coronary intervention for acute coronary syndromes and stable angina. Int J Cardiol. 2011. September 1;151(2):164–9. 10.1016/j.ijcard.2010.05.006 [DOI] [PubMed] [Google Scholar]
- 4.Munoz JC, Alonso JJ, Duran JM, Gimeno F, Ramos B, Garcimartin I, et al. Coronary stent implantation in patients older than 75 years of age: clinical profile and initial and long-term (3 years) outcome. Am Heart J. 2002. April;143(4):620–6. . [DOI] [PubMed] [Google Scholar]
- 5.Kelsey SF, Miller DP, Holubkov R, Lu AS, Cowley MJ, Faxon DP, et al. Results of percutaneous transluminal coronary angioplasty in patients greater than or equal to 65 years of age (from the 1985 to 1986 National Heart, Lung, and Blood Institute's Coronary Angioplasty Registry). Am J Cardiol. 1990. November 1;66(15):1033–8. . [DOI] [PubMed] [Google Scholar]
- 6.Roe MT, Li S, Thomas L, Wang TY, Alexander KP, Ohman EM, et al. Long-term outcomes after invasive management for older patients with non-ST-segment elevation myocardial infarction. Circ Cardiovasc Qual Outcomes. 2013. May 1;6(3):323–32. 10.1161/CIRCOUTCOMES.113.000120 [DOI] [PubMed] [Google Scholar]
- 7.Montalescot G, Sechtem U, Achenbach S, Andreotti F, Arden C, Budaj A, et al. 2013 ESC guidelines on the management of stable coronary artery disease: the Task Force on the management of stable coronary artery disease of the European Society of Cardiology. Eur Heart J. 2013. October;34(38):2949–3003. 10.1093/eurheartj/eht296 [DOI] [PubMed] [Google Scholar]
- 8.Thompson RC, Holmes DR Jr., Grill DE, Mock MB, Bailey KR. Changing outcome of angioplasty in the elderly. J Am Coll Cardiol. 1996. January;27(1):8–14. . [DOI] [PubMed] [Google Scholar]
- 9.Johnman C, Oldroyd KG, Mackay DF, Slack R, Pell AC, Flapan AD, et al. Percutaneous coronary intervention in the elderly: changes in case-mix and periprocedural outcomes in 31,758 patients treated between 2000 and 2007. Circ Cardiovasc Interv. 2010. August;3(4):341–5. 10.1161/CIRCINTERVENTIONS.109.928705 [DOI] [PubMed] [Google Scholar]
- 10.Park SJ, Park DW. New era of left main coronary artery treatment strategy. Cardiovasc Interv Ther. 2012. January;27(1):1–7. 10.1007/s12928-011-0085-9 [DOI] [PubMed] [Google Scholar]
- 11.Cheng JM, Onuma Y, Piazza N, Nuis RJ, Van Domburg RT, Serruys PW. Comparison of five-year outcome of octogenarians undergoing percutaneous coronary intervention with drug-eluting versus bare-metal stents (from the RESEARCH and T-SEARCH Registries). Am J Cardiol. 2010. November 15;106(10):1376–81. 10.1016/j.amjcard.2010.07.007 [DOI] [PubMed] [Google Scholar]
- 12.Ranchord AM, Prasad S, Seneviratne SK, Simmonds MB, Matsis P, Aitken A, et al. Same-day discharge is feasible and safe in the majority of elderly patients undergoing elective percutaneous coronary intervention. J Invasive Cardiol. 2010. July;22(7):301–5. . [PubMed] [Google Scholar]
- 13.Lee MS, Zimmer R, Pessegueiro A, Jurewitz D, Tobis J. Outcomes of nonagenarians who undergo percutaneous coronary intervention with drug-eluting stents. Catheter Cardiovasc Interv. 2008. March 1;71(4):526–30. 10.1002/ccd.21382 [DOI] [PubMed] [Google Scholar]
- 14.Kobayashi Y, Mehran R, Mintz GS, Dangas G, Moussa I, Lansky AJ, et al. Comparison of in-hospital and one-year outcomes after multiple coronary arterial stenting in patients > or = 80 years old versus those <80 years old. Am J Cardiol. 2003. August 15;92(4):443–6. . [DOI] [PubMed] [Google Scholar]
- 15.Chauhan MS, Kuntz RE, Ho KL, Cohen DJ, Popma JJ, Carrozza JP Jr., et al. Coronary artery stenting in the aged. J Am Coll Cardiol. 2001. March 1;37(3):856–62. . [DOI] [PubMed] [Google Scholar]
- 16.Bauer T, Koeth O, Junger C, Heer T, Wienbergen H, Gitt A, et al. Effect of an invasive strategy on in-hospital outcome in elderly patients with non-ST-elevation myocardial infarction. Eur Heart J. 2007. December;28(23):2873–8. . [DOI] [PubMed] [Google Scholar]
- 17.Marcolino MS, Simsek C, de Boer SP, van Domburg RT, van Geuns RJ, de Jaegere P, et al. Short- and long-term outcomes in octogenarians undergoing percutaneous coronary intervention with stenting. EuroIntervention. 2012. December 20;8(8):920–8. 10.4244/EIJV8I8A141 [DOI] [PubMed] [Google Scholar]
- 18.Deckers JW. Classification of myocardial infarction and unstable angina: a re-assessment. Int J Cardiol. 2013. September 10;167(6):2387–90. 10.1016/j.ijcard.2013.01.008 [DOI] [PubMed] [Google Scholar]
- 19.Brown AJ, Obaid DR, Costopoulos C, Parker RA, Calvert PA, Teng Z, et al. Direct Comparison of Virtual-Histology Intravascular Ultrasound and Optical Coherence Tomography Imaging for Identification of Thin-Cap Fibroatheroma. Circ Cardiovasc Imaging. 2015. October;8(10):e003487 . Central PMCID: PMC4596008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mintz GS. Optical Coherence Tomography and Virtual-Histology Intravascular Ultrasound: Strange Bedfellows?…or Not? Circ Cardiovasc Imaging. 2015. October;8(10):e004045 10.1161/CIRCIMAGING.115.004045 [DOI] [PubMed] [Google Scholar]
- 21.Healthreport 2011, STATISTIK AUSTRIA, Bundesanstalt Statistik, Österreich, Verlag Österreich GmbH, ISBN 978-3-902791-61-0. Available:http://www.statistik.at/web_de/services/publikationen/4/index.html?includePage=detailedView&pubId=543§ionName=Gesundheit.
- 22.Davidson L, Wilcox J, Kim D, Benton S, Fredi J, Vaughan D. Clinical features of precocious acute coronary syndrome. Am J Med. 2014. February;127(2):140–4. 10.1016/j.amjmed.2013.09.025 [DOI] [PubMed] [Google Scholar]
- 23.Ertelt K, Genereux P, Mintz GS, Brener SJ, Kirtane AJ, McAndrew TC, et al. Clinical profile and impact of family history of premature coronary artery disease on clinical outcomes of patients undergoing primary percutaneous coronary intervention for ST-elevation myocardial infarction: analysis from the HORIZONS-AMI Trial. Cardiovasc Revasc Med. 2014. Nov-Dec;15(8):375–80. 10.1016/j.carrev.2014.09.002 [DOI] [PubMed] [Google Scholar]
- 24.Ruiz-Garcia J, Lerman A, Weisz G, Maehara A, Mintz GS, Fahy M, et al. Age- and gender-related changes in plaque composition in patients with acute coronary syndrome: the PROSPECT study. EuroIntervention. 2012. December 20;8(8):929–38. 10.4244/EIJV8I8A142 [DOI] [PubMed] [Google Scholar]
- 25.Buljubasic N, Akkerhuis KM, de Boer SP, Cheng JM, Garcia-Garcia HM, Lenzen MJ, et al. Smoking in Relation to Coronary Atherosclerotic Plaque Burden, Volume and Composition on Intravascular Ultrasound. PloS One. 2015;10(10):e0141093 Central PMCID: PMC4619630. 10.1371/journal.pone.0141093 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kataoka Y, Hammadah M, Puri R, Duggal B, Uno K, Kapadia SR, et al. Plaque microstructures in patients with coronary artery disease who achieved very low low-density lipoprotein cholesterol levels. Atherosclerosis. 2015. October;242(2):490–5. 10.1016/j.atherosclerosis.2015.08.005 [DOI] [PubMed] [Google Scholar]
- 27.Marso SP, Mercado N, Maehara A, Weisz G, Mintz GS, McPherson J, et al. Plaque composition and clinical outcomes in acute coronary syndrome patients with metabolic syndrome or diabetes. JACC Cardiovascular imaging. 2012. March;5(3 Suppl):S42–52. 10.1016/j.jcmg.2012.01.008 [DOI] [PubMed] [Google Scholar]
- 28.Baber U, Stone GW, Weisz G, Moreno P, Dangas G, Maehara A, et al. Coronary plaque composition, morphology, and outcomes in patients with and without chronic kidney disease presenting with acute coronary syndromes. JACC Cardiovascular imaging. 2012. March;5(3 Suppl):S53–61. 10.1016/j.jcmg.2011.12.008 [DOI] [PubMed] [Google Scholar]
- 29.Garcia-Garcia HM, Serruys PW, Mintz GS, Saito S, Klaus V, Margolis P, et al. Synergistic effect of cardiovascular risk factors on necrotic core in coronary arteries: a report from the global intravascular radiofrequency data analysis registry. JACC Cardiovascular imaging. 2009. May;2(5):629–36. 10.1016/j.jcmg.2009.01.008 [DOI] [PubMed] [Google Scholar]
- 30.Virmani R, Kolodgie FD, Burke AP, Farb A, Schwartz SM. Lessons from sudden coronary death: a comprehensive morphological classification scheme for atherosclerotic lesions. Arterioscler Thromb Vasc Biol. 2000. May;20(5):1262–75. . [DOI] [PubMed] [Google Scholar]
- 31.Thim T, Hagensen MK, Wallace-Bradley D, Granada JF, Kaluza GL, Drouet L, et al. Unreliable assessment of necrotic core by virtual histology intravascular ultrasound in porcine coronary artery disease. Circ Cardiovasc Imaging. 2010. July;3(4):384–91. 10.1161/CIRCIMAGING.109.919357 [DOI] [PubMed] [Google Scholar]
- 32.Obaid DR, Calvert PA, Gopalan D, Parker RA, Hoole SP, West NE, et al. Atherosclerotic plaque composition and classification identified by coronary computed tomography: assessment of computed tomography-generated plaque maps compared with virtual histology intravascular ultrasound and histology. Circ Cardiovasc Imaging. 2013. September;6(5):655–64. 10.1161/CIRCIMAGING.112.000250 [DOI] [PubMed] [Google Scholar]
- 33.Yabushita H, Bouma BE, Houser SL, Aretz HT, Jang IK, Schlendorf KH, et al. Characterization of human atherosclerosis by optical coherence tomography. Circulation. 2002. September 24;106(13):1640–5. . [DOI] [PubMed] [Google Scholar]
- 34.Miyamoto Y, Okura H, Kume T, Kawamoto T, Neishi Y, Hayashida A, et al. Plaque characteristics of thin-cap fibroatheroma evaluated by OCT and IVUS. JACC Cardiovascular imaging. 2011. June;4(6):638–46. 10.1016/j.jcmg.2011.03.014 [DOI] [PubMed] [Google Scholar]
- 35.Tearney GJ, Regar E, Akasaka T, Adriaenssens T, Barlis P, Bezerra HG, et al. Consensus standards for acquisition, measurement, and reporting of intravascular optical coherence tomography studies: a report from the International Working Group for Intravascular Optical Coherence Tomography Standardization and Validation. J Am Coll Cardiol. 2012. March 20;59(12):1058–72. 10.1016/j.jacc.2011.09.079 [DOI] [PubMed] [Google Scholar]
- 36.Kataoka Y, Hammadah M, Puri R, Duggal B, Uno K, Kapadia SR, et al. Plaque microstructures in patients with coronary artery disease who achieved very low low-density lipoprotein cholesterol levels. Atherosclerosis. 2015. October;242(2):490–5. 10.1016/j.atherosclerosis.2015.08.005 [DOI] [PubMed] [Google Scholar]
- 37.Kataoka Y, Hammadah M, Puri R, Duggal B, Uno K, Kapadia SR, et al. Plaque vulnerability at non-culprit lesions in obese patients with coronary artery disease: Frequency-domain optical coherence tomography analysis. Eur J Prev Cardiol. 2015. October;22(10):1331–9. 10.1177/2047487315598711 [DOI] [PubMed] [Google Scholar]
- 38.Cole JH, Sperling LS. Premature coronary artery disease: clinical risk factors and prognosis. Curr Atheroscler Rep. 2004. March;6(2):121–5. . [DOI] [PubMed] [Google Scholar]
- 39.Hou X, Chen X, Shi J. Genetic polymorphism of MTHFR C677T and premature coronary artery disease susceptibility: A meta-analysis. Gene. 2015. July 1;565(1):39–44. 10.1016/j.gene.2015.03.062 [DOI] [PubMed] [Google Scholar]
- 40.Cole JH, Sperling LS. Premature coronary artery disease: clinical risk factors and prognosis. Curr Atheroscler Rep. 2004. March;6(2):121–5. . [DOI] [PubMed] [Google Scholar]
- 41.Zamorano JL, Garcia-Moll X, Ferrari R, Greenlaw N. Demographic and clinical characteristics of patients with stable coronary artery disease: results from the CLARIFY registry in Spain. Revista espanola de cardiologia (English ed). 2014. July;67(7):538–44. . [DOI] [PubMed] [Google Scholar]
- 42.Bortnick AE, Epps KC, Selzer F, Anwaruddin S, Marroquin OC, Srinivas V, et al. Five-year follow-up of patients treated for coronary artery disease in the face of an increasing burden of co-morbidity and disease complexity (from the NHLBI Dynamic Registry). Am J Cardiol. 2014. February 15;113(4):573–9. 10.1016/j.amjcard.2013.10.039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Anderson JL, Adams CD, Antman EM, Bridges CR, Califf RM, Casey DE Jr., et al. ACC/AHA 2007 guidelines for the management of patients with unstable angina/non-ST-Elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines for the Management of Patients With Unstable Angina/Non-ST-Elevation Myocardial Infarction) developed in collaboration with the American College of Emergency Physicians, the Society for Cardiovascular Angiography and Interventions, and the Society of Thoracic Surgeons endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation and the Society for Academic Emergency Medicine. J Am Coll Cardiol. 2007. August 14;50(7):e1–e157. . [DOI] [PubMed] [Google Scholar]
- 44.Bassand JP, Hamm CW, Ardissino D, Boersma E, Budaj A, Fernandez-Aviles F, et al. Guidelines for the diagnosis and treatment of non-ST-segment elevation acute coronary syndromes. Portuguese journal of cardiology. 2008. September;27(9):1063–143. . [PubMed] [Google Scholar]
- 45.Bach RG, Cannon CP, Weintraub WS, DiBattiste PM, Demopoulos LA, Anderson HV, et al. The effect of routine, early invasive management on outcome for elderly patients with non-ST-segment elevation acute coronary syndromes. Ann Intern Med. 2004. August 3;141(3):186–95. . [DOI] [PubMed] [Google Scholar]
- 46.Anderson HV, Cannon CP, Stone PH, Williams DO, McCabe CH, Knatterud GL, et al. One-year results of the Thrombolysis in Myocardial Infarction (TIMI) IIIB clinical trial. A randomized comparison of tissue-type plasminogen activator versus placebo and early invasive versus early conservative strategies in unstable angina and non-Q wave myocardial infarction. J Am Coll Cardiol. 1995. December;26(7):1643–50. . [DOI] [PubMed] [Google Scholar]
- 47.Bagnall AJ, Goodman SG, Fox KA, Yan RT, Gore JM, Cheema AN, et al. Influence of age on use of cardiac catheterization and associated outcomes in patients with non-ST-elevation acute coronary syndromes. Am J Cardiol. 2009. June 1;103(11):1530–6. 10.1016/j.amjcard.2009.01.369 [DOI] [PubMed] [Google Scholar]
- 48.Bangalore S, Pursnani S, Kumar S, Bagos PG. Percutaneous coronary intervention versus optimal medical therapy for prevention of spontaneous myocardial infarction in subjects with stable ischemic heart disease. Circulation. 2013. February 19;127(7):769–81. 10.1161/CIRCULATIONAHA.112.131961 [DOI] [PubMed] [Google Scholar]
- 49.Wald DS, Morris JK, Wald NJ, Chase AJ, Edwards RJ, Hughes LO, et al. Randomized trial of preventive angioplasty in myocardial infarction. N Engl J Med. 2013. September 19;369(12):1115–23. 10.1056/NEJMoa1305520 [DOI] [PubMed] [Google Scholar]
- 50.Kyto V, Sipila J, Rautava P. Gender, age and risk of ST segment elevation myocardial infarction. Eur J Clin Invest. 2014. October;44(10):902–9. 10.1111/eci.12321 [DOI] [PubMed] [Google Scholar]
- 51.Boden WE, O'Rourke RA, Teo KK, Hartigan PM, Maron DJ, Kostuk WJ, et al. Optimal medical therapy with or without PCI for stable coronary disease. N Engl J Med. 2007. April 12;356(15):1503–16. . [DOI] [PubMed] [Google Scholar]
- 52.Stone GW, Gao R, Kimura T, Kereiakes DJ, Ellis SG, Onuma Y, et al. 1-year outcomes with the Absorb bioresorbable scaffold in patients with coronary artery disease: a patient-level, pooled meta-analysis. Lancet. 2016. January 25 . [DOI] [PubMed] [Google Scholar]
- 53.Ridker PM, Danielson E, Fonseca FA, Genest J, Gotto AM Jr., Kastelein JJ, et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N Engl J Med. 2008. November 20;359(21):2195–207. 10.1056/NEJMoa0807646 [DOI] [PubMed] [Google Scholar]
- 54.Verschuren JJ, Trompet S, Tio RA, de Winter RJ, Doevendans PA, Jukema JW. Ten-year mortality risk of patients undergoing elective PCI: long-term follow-up of the GENetic Determinants of Restenosis (GENDER) study: No increased mortality risk by restenosis, only by coronary artery disease itself. Netherlands heart journal. 2013. February;21(2):101–5. 10.1007/s12471-012-0370-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Windecker S, Stortecky S, Stefanini GG, da Costa BR, Rutjes AW, Di Nisio M, et al. Revascularisation versus medical treatment in patients with stable coronary artery disease: network meta-analysis. BMJ. 2014;348:g3859 10.1136/bmj.g3859 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All relevant data are available in the paper.